Abstract
Assemblies of saturated 1,2-diacyl-phosphatidylcholine lipid and hydrophobic dodecanethiol-capped 1.8 nm diameter gold nanocrystals were studied as a function of lipid chain length and the addition of the naturally-occurring oil, squalene. The gold nanocrystals formed various lipid-stabilized agglomerates, sometimes fusing with lipid vesicle bilayers. The nanocrystal assembly structure depended on the hydrocarbon chain length of the lipid fatty acids. Lipid with the shortest fatty acid length studied, dilauroyl-phosphatidylcholine, created extended chains of gold nanocrystals. Lipid with slightly longer fatty acid chains created planar sheets of nanocrystals. Further increases of the fatty acid chain length led to spherical agglomerates. The inclusion of squalene led to lipid- and nanocrystal-coated oil droplets.
Keywords: Gold nanocrystal, vesicle, liposome, squalene, phosphatidylcholine, emulsion, lipid bilayer, cryo TEM, phospholipids, saturated lipids
Introduction
Phosphatidylcholine lipids with saturated fatty acid chains are naturally-occurring,1–3,4 bio-degradable amphiphiles that aggregate into lipid bilayers when dispersed in water5–7 and form vesicles.8,9 The vesicles can serve as drug carriers by encapsulating hydrophilic molecules in an aqueous compartment or hosting hydrophobic molecules in the lipid bilayer.10–13 The lipid bilayer can also host hydrophobic metal,14,15 oxide,16 or semiconductor nanocrystals,17,18 which can give the vesicles new functionalities for biomedical imaging and therapy.19,20 Recently, it was reported that only hydrophobic nanocrystals smaller than about 5 nm diameter can incorporate into lipid bilayers,17,21 while larger nanocrystals aggregate and do not associate with the bilayers.16 However, the role of the lipid chemistry has not yet been considered, i.e., changes in lipid chain length and saturation.
In our recent work, we found that dodecanethiol-coated 1.8 nm diameter Au nanocrystals formed closely-packed monolayers in hen egg-derived phosphatidylcholine (eggPC) vesicle bilayers.14 A significant number of empty vesicles and Au nanocrystal clusters encapsulated in lipid micelles were also observed. EggPC is a mixture of phosphatidylcholine lipid, with chain lengths ranging from 14 to 18 carbons in length and about half of the fatty acid chains being saturated1 and we have considered whether the precise lipid composition affects how 1.8 nm Au nanocrystals incorporate into vesicles. The fatty acid chain length of saturated phosphatidylcholines influences the thickness,7 elastic bending modulus,22 and melting temperature of bilayers,1 so it seems likely that it would influence the incorporation of nanocrystals into the lipid bilayer.
Here, we show that hydrophobic 1.8 nm Au nanocrystals do not incorporate into the lipid bilayer of vesicles of phosphatidylcholine lipids with saturated hydrocarbon chains, including dilauroyl-phosphatidylcholine (DLPC, C12), dimyristoyl-phosphatidylcholine (DMPC, C14), dipalmitoyl-phosphatidylcholine (DPPC, C16), and distearoyl-phosphatidylcholine (DSPC, C18).
We also tested whether the addition of the liquid hydrocarbon, squalene, influences the assembly of saturated lipids and dodecanethiol-coated Au nanocrystals. A recent study reported that 2 nm diameter hydrophobic Au nanocrystals can only incorporate into surfactant bilayers swollen with n-dodecane.23 We tested squalene since it is a natural precursor to steroids and is frequently used to prepare oil-in-water emulsions for delivering hydrophobic drugs and vaccines,24,25 We found that squalene and phosphatidylcholine formed mixtures of lipid-coated squalene oil droplets and lipid vesicles. Dodecanethiol-capped Au nanocrystals added to the squalene/lipid dispersions did not mix into the lipid bilayers of vesicles, but accumulated near the surface of the squalene droplets, somewhat similar to the therapeutic vegetable oil emulsions prepared with iron nanocrystals and phospholipid stabilizers that others have studied.28–30
Experimental Details
Materials
Hydrogen tetrachloroaurate trihydrate (HAuCl4-3H2O, 99.999%), tetraoctylammonium bromide (TOAB, 98%), sodium borohydride (NaBH4, 98%), squalene, n-dodecane, and 1-dodecanethiol (98%) were obtained from Sigma-Aldrich. Chloroform, toluene, and ethanol (EtOH, 200 proof) were from Fisher Scientific. 1,2-dilauroyl-sn-glycero-3-phosphatidylcholine (DLPC, >99%), 1,2-dimyristoyl-sn-glycero-3-phosphatidylcholine (DMPC, >99%), 1,2-dipalmitoyl-sn-glycero-3-phosphatidylcholine (DPPC, >99%), and 1,2-distearoyl-sn-glycero-3-phosphatidylcholine (DSPC, >99%) were obtained from Avanti Polar Lipids. Deionized (DI) water was obtained from a Barnstead Nanopure Filtration System operating at a 17 ΩM resistance.
Gold Nanocrystal Synthesis
Dodecanethiol-capped 1.8 nm diameter Au nanocrystals were synthesized following literature procedures.14 In a 125 mL flask, 6.0 g of TOAB was dissolved in 73 mL of toluene by magnetic stirring (600 rpm). An aqueous gold solution (0.300 g HAuCl4-3H2O in 18 mL DI water) was added to the toluene phase and stirring was continued for 1 hour, resulting in complete phase transfer of the gold ions to the toluene phase. The toluene phase was extracted, placed in a clean flask, and stirred at 600 rpm. 2.3 mmol (550 μL) of 1-dodecanethiol was added, turning the solution from red to colorless. After stirring for 15 minutes, aqueous sodium borohydride (0.346 g NaBH4 in 18 mL DI water) was added. After stirring for 12 hours, the toluene phase was extracted and poured into glass centrifuge tubes. Ethanol was added to each tube as an antisolvent (20 mL EtOH: 5 mL toluene). After centrifugation at 9000 rpm for 6 minutes, the colorless supernatant was discarded. The gold nanocrystal precipitate was dispersed in 2 mL of toluene, combined into one centrifuge tube, and centrifuged at 10000 rpm for 3 minutes. The supernatant of well-dispersed nanocrystals was transferred to a clean glass centrifuge tube. The nanocrystal size distribution was further narrowed to isolate nanocrystals with the smallest diameter by adding 500 μL of ethanol to the dispersion, centrifuging at 9000 rpm for 6 minutes, transferring the supernatant to a new tube, and repeating this step once more. The nanocrystals were then isolated by precipitation by adding 20 mL of ethanol and centrifuging at 9000 rpm for 8 minutes. The final precipitate of nanocrystals was dispersed in 2 mL of toluene.
Vesicle Formation
Lipid films were dried from 1.0 mL chloroform dispersions of 30 μmol phosphatidylcholine (DLPC, DMPC, DPPC, or DSPC) and 3 mg of gold nanocrystals, corresponding to about 500 lipid molecules per nanocrystal. The 1 mL dispersion was placed in a 50 mL glass round bottom flask (Chemglass) and connected to a rotary evaporator (Buchi). Chloroform was evaporated at different temperatures depending on the lipid: DLPC and DMPC at 5°C, DPPC at 50°C, DSPC at 60°C. Compared to drying at room temperature, these temperatures reduced the amount of phase separation between lipid and nanocrystals (See light microscopy data in Supporting Information Figure S3). Once dry, the pressure in the flask was reduced to 50 mbar for 15 minutes. The flask was removed from the rotary evaporator and placed in a vacuum oven for 12 hours at room temperature to completely remove the chloroform.
Lipid films were hydrated with 1.0 mL of 0.1 μm pore-filtered DI water above the liquid crystal lamellar phase (Lα) temperature of each lipid:1 DLPC at 25°C; DMPC at 30°C; DPPC at 50°C; and DSPC at 60°C.31 The flasks with the dry lipid-nanocrystal films were briefly warmed in a circulating water bath to the desired temperature, followed by addition of water at the same temperature. The film was completely wetted by gently rotating the flask while held in the water bath. Then, the flask was sealed with parafilm, transferred to an ultrasonicator bath (Misonix, 600 mL water bath) adjusted to the hydration temperature for each lipid, and sonicated until the lipid-nanocrystal film completely lifted off of the glass surface (3 minutes). Subsequently, the dispersion was transferred by glass pipette to a clean 20 mL scintillation vial warmed to the lipid hydration temperature.32 The scintillation vials have thinner walls and a flat bottom, which are better for transmitting ultrasonic waves than the round bottom flasks for reducing particle size.33 The vial was sealed with parafilm and sonicated for an additional 12 minutes. The power delivered by the ultrasonicator was about 30 W,34 calculated from the measured temperature rise of the bath versus time near 25°C according to Taurozzi et al.35,36
After sonication, samples were transferred to 2 mL plastic centrifuge tubes, warmed to the hydration temperature in a water bath, and centrifuged at 1000g for 10 minutes to precipitate poorly dispersed nanocrystals. The supernatant was transferred to new tubes and maintained at the hydration temperature in a water bath. Samples were then extruded 10 times through two stacked polycarbonate membranes (100 nm diameter pores) using a stainless steel Lipex extruder (Northern Lipids), operating at 500 psi and equilibrated to the lipid hydration temperature using a water jacket connected to a circulating water bath. After extrusion, the samples were allowed to cool to room temperature overnight prior to imaging the next day.
Squalene Emulsions
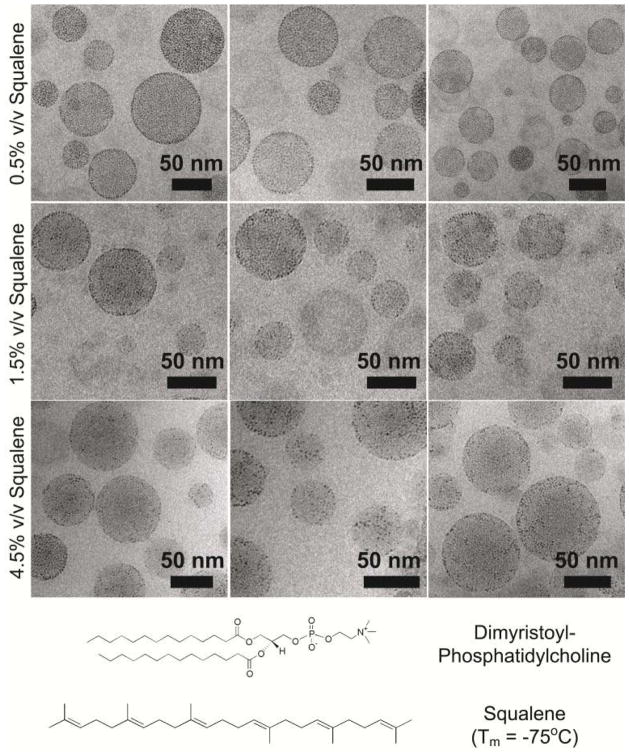
The vesicle formation process was repeated using 30 μmol of DLPC, DMPC, or DPPC, in addition to 3 mg of Au nanocrystals. Squalene was added to the chloroform dispersions of lipid and nanocrystals prior to rotary evaporation in varying amounts from 5, 10, 15, to 45 μL (0.5%, 1.0%, 1.5%, and 4.5% v/v respectively).
CryoTEM Imaging
CryoTEM was performed with an FEI Tecnai F20 TEM operated at 200 kV with a liquid nitrogen cooled stage. Specimens were prepared on C-flat holey carbon film TEM grids (Protochips, via Electron Microscopy Sciences) with 1.2 μm diameter holes and 1.3 μm spacing. TEM grids were exposed to glow discharge plasma for 30 seconds. TEM samples were vitrified using an automated Vitrobot (FEI). 2.5 μL of a dispersion was placed on a C-flat grid suspended in the Vitrobot chamber maintained at 95–100% relative humidity. The Vitrobot chamber temperature was 25°C unless specified otherwise.37 The Vitrobot blots the grid with filter paper (1 blot, 3.5 second blot time) and then plunges the grid into liquid ethane. The vitrified grid was transferred to a cryo grid storage box (Electron Microscopy Sciences) submerged in liquid nitrogen prior to imaging. The vitrified grid was transferred in liquid nitrogen to a Gatan CT3500 single tilt cryo TEM specimen holder on a cryo workstation (Gatan), followed by insertion into the microscope. Vesicles are imaged in the holey regions of the carbon film.
Results and Discussion
Saturated PC Lipid-Nanocrystal aggregates
Figure 1 shows cryoTEM images of saturated phosphatidylcholine lipids with various fatty acid chain length (C12-C18) combined with dodecanethiol-coated 1.8 nm diameter Au nanocrystals. The lipid forms vesicles, but unlike eggPC vesicles, the saturated PC lipid vesicles do not have any Au nanocrystals in their lipid bilayer.14 The nanocrystals form various aggregated structures coated with lipid.
Figure 1.
CryoTEM images of aggregated Au nanocrystals formed with various PC lipids with saturated hydrocarbon chains. All aqueous dispersions were vitrified at 25°C. DLPC and DMPC is in the liquid crystal phase, while DPPC and DSPC are in the gel phase. Red arrows identify nanocrystal aggregates coated with a thin lipid layer. Photographs of the dispersions are shown in the insets.
Disetearoyl- and Dipalmitoyl Phosphatidylcholine Aggregates
The morphology of the lipid-coated gold nanocrystal agglomerates varied with fatty acid length. For DSPC (C18) and DPPC (C16), the nanocrystals form lipid-coated agglomerates ranging from 10 to 50 nm in diameter. Both DPPC and DSPC lipid are in the lamellar gel phase (Lβ′) (at 25°C, the temperature of cryoTEM sample vitrification)1 and the vesicles exhibit planar, faceted membranes as has been observed previously for mixtures of cationic and anionic surfactants in the gel phase.38 The lipid-coated nanocrystal aggregates also had faceted surfaces. About half of the nanocrystal agglomerates in each lipid sample were found to be fused to vesicle bilayers most likely due to aggregation. This type of structure—of clustered nanocrystals associated with lipid bilayers—has also been proposed as a way to minimize distortions in lipid packing.14,19
Dimyristoyl- and Dilauroyl-Phosphatidylcholine Aggregates
The structures of the gold agglomerates with DMPC (C14) and DLPC (C12) differed significantly from those with DPPC and DSPC. DMPC and DLPC are both in the lamellar liquid crystal phase (Lα) at 25°C,1 making their bilayers much more fluid than those of DPPC and DSPC.1,22 Accordingly, DLPC and DMPC vesicles are round and spherical, not faceted.39,40 The DMPC-coated nanocrystal agglomerates were either solid spherical agglomerates (highlighted with red arrows in Figure 1) or flat monolayer sheets. When these structures fuse with the DMPC vesicles, the nanocrystals appear to be budding off the pure lipid bilayer, as if being forced out of the vesicle membrane. Nanocrystals with DLPC formed chains. These chains often terminated in pure lipid bilayers or larger spherical nanocrystal agglomerates. Furthermore, the chains seemed to form an interconnected network. The DLPC-stabilized Au nanocrystal chains resemble the thread-like micelles that form when eggPC vesicles are mixed with cetyltrimethylammonium chloride surfactant,41 yet pure DLPC lipid chains were not observed (Supporting Information). Perhaps the interfacial curvature of thread-like micelles of pure lipid is too high and the core of Au nanocrystals reduces this to stabilize the structure.22
Lipid-Squalene Oil Droplets with Au nanocrystals
Squalene was added to the dispersions to test whether it could swell the lipid bilayer and enable nanocrystals to accumulate in the lipid bilayer of the saturated lipid vesicles. The addition of squalene however led to the formation of lipid-coated squalene (oil) droplets, as shown in Figure 2. The Au nanocrystals were observed to accumulate on the droplet surface. Figure 3 shows DLPC and DPPC lipid and squalene droplets as well. At high Au nanocrystal concentrations, the Au nanocrystals form a tightly packed monolayer at the droplet surface. Squalene disrupts the Au nanocrystal chains formed with DLPC, although there were rare instances of Au nanocrystal chains still, as in the top right panel of Figure 3. The DPPC-squalene droplets loaded with Au nanocrystals were spherical and not faceted either above or below the Lβ′ to Lα phase transition temperature of pure DPPC. This is another indication that the observed droplets are not vesicles, as squalene does not alter the phase transition temperature of DPPC,26 and the nanocrystal-loaded droplet morphology remains spherical.
Figure 2.
CryoTEM images of squalene/DMPC lipid droplets with dodecanethiol-coated Au nanocrystals. Each row of images corresponds to different squalene concentrations.
Figure 3.
CryoTEM images of squalene emulsion droplets loaded with dodecanethiol-coated Au nanocrystals and stabilized with either DLPC or DPPC. The bottom right panel shows Au-loaded squalene droplets deforming when packed together in one region of the TEM grid. The squalene concentrations are all 1% v/v. The insets show photographs of the dispersions.
The partitioning of the hydrophobic nanocrystals to the squalene-water interface indicates that lipid is coating the droplets.42, 43,44 Dodecanethiol-coated gold nanocrystals disperse in pure squalene, but squalene-nanocrystal mixtures did not emulsify in water without lipid. The positioning of the hydrophobic Au nanocrystals at the squalene-lipid interface increases the separation between the water and squalene phases, and also reduces the contact between lipid molecules and squalene, which do not mix according to recent studies.26, 27 This is comparable to Pickering emulsions, which are stabilized by a combination of solid particles and surfactant, where the role of the surfactant is to change the interfacial tension so the particles reside at the oil/water interface to stabilize the emulsion.45, 46
Figure 4 summarizes the various saturated PC lipid-nanocrystal-squalene assemblies observed by cryoTEM. In each case, the lipid forms a coating around the assemblies and stabilizes their structure. The lipid chain length plays a determining role, varying the structure from chain-like aggregates to two-dimensional sheets of nanocrystals to three-dimensional solid aggregates of nanocrystals.
Figure 4.
Lipid/Au nanocrystal assemblies observed by cryoTEM. The PC lipids are drawn with blue polar headgroups and green fatty acid chains. The gold nanocrystals are brown circles. Squalene is shown in yellow.
Buoyancy of Au nanocrystal-loaded squalene/lipid droplets
Lipid-coated squalene droplets were observed to cream (negative settling velocity), particularly when Au nanocrystals were not added. For example, significant creaming was observed within 24 hours if insufficient sonication power was delivered or Au nanocrystals were not incorporated into the emulsions. The Au nanocrystals alter the buoyancy of the squalene droplets. Figure 5 shows the settling velocity v, calculated for the various types of particles observed by cryoTEM. Assuming the particles do not interact,47
Figure 5.
Settling velocities calculated for the different types of particles observed by cryoTEM. Positive velocity refers to motion in the direction of gravity and negative velocity refers to flotation or particle creaming. In the illustrations, lipids have a blue headgroup and green fatty acid chains, Au nanocrystals are brown circles, and squalene is yellow.
| (1) |
D is the diameter of the lipid-coated particle, g is 9.8 m/s2, and η is the viscosity of water at 25°C (0.89 mPa-s). mwater is the mass of water displaced by the particle, mlipid is the mass of lipid coating the particle, msqualene is the mass of squalene in the droplet, and mAu is the mass of the Au nanocrystals per droplet. As shown in Figure 5, settling is size dependent. (See Supporting Information for details regarding the calculation of v). The lipid-coated Au agglomerates settle the fastest and the Au nanocrystals act as a weighting agent in the squalene droplets, preventing creaming until the droplet diameter exceeds 130 nm.
Conclusions
The saturated phosphatidylcholine lipids, DLPC, DMPC, DPPC, and DSPC assemble into vesicles, but do not incorporate dodecanethiol-capped (1.8 nm diameter) Au nanocrystals into their lipid bilayers. Instead, lipid/nanocrystal agglomerates form with different morphologies depending on the fatty acid chain length. DLPC (C12) created chains of nanocrystals. DMPC (C14) stabilized both three-dimensional aggregates and thin monolayer sheets of nanocrystals. DPPC (C16) and DSPC (C18) stabilized only three-dimensional agglomerates of nanocrystals. In many cases, the lipid-coated nanocrystal agglomerates partially fused with vesicles.
The lipids forming membranes with more mechanical flexibility produced agglomerates with higher curvature surfaces. The Au nanocrystal chains formed with DLPC resemble thread-like micelles typically formed with detergents.41 There must be a synergy between the lipid and nanocrystals that stabilizes these structures since thread-like micelles do not form with pure DLPC. DMPC lipid forms slightly more rigid membranes than DLPC and was found to stabilize two-dimensional aggregates of Au nanocrystals. Others have reported that CdSe nanocrystals can incorporate into the bilayer of DMPC vesicles,17, 21 but the Au nanocrystals studied here did not. With DPPC and DSPC, the Au nanocrystals only formed spherical agglomerates that did not have highly curved edges like the DMPC and DLPC structures, which is consistent with higher bending elasticity of DPPC and DSPC lipid assemblies at 25°C.
Squalene did not encourage the nanocrystals to mix with the lipid bilayers, but created lipid-coated oil droplets in which the Au nanocrystals were found to accumulate at the oil-water interface. Nanocrystal-loaded emulsion particles have been established, such as the therapeutic vegetable oil emulsions prepared with iron nanocrystals and phospholipid stabilizers that others have studied,28–30 and so has the interfacial assembly of Au nanocrystals having mixed polar and non-polar surface functionalities to form Pickering emulsions.44 However the assembly of hydrophobic nanocrystals at a lipid-coated oil-water interface seems to be a new concept. The Au nanocrystals alter the density of the oil droplets and prevent creaming, which might be exploited in other surfactant-based separations. Furthermore the Au nanocrystals assembled at the squalene-water interface may be useful as a barrier to controlled release of molecules dissolved in the squalene phase,20, 27 though this remains to be demonstrated.
Supplementary Material
Acknowledgments
We thank the Texas A&M Microscopy and Imaging Center for use of their Tecnai F20 transmission electron microscope and sample preparation facilities, and Dr. Zhiping Luo for cryoTEM instruction. We acknowledge the Robert A. Welch Foundation (F-1464) and the National Institutes of Health (Grant no. R01 CA132032) for financial support of the research.
References
- 1.Marsh D. CRC Handbook of Lipid Bilayers. CRC Press, Inc; Boca Raton, FL: 1990. [Google Scholar]
- 2.Cole LK, Vance JE, Vance DE. Phosphatidylcholine biosynthesis and lipoprotein metabolism. Biochim Biophys Acta. 2012;1821:754–761. doi: 10.1016/j.bbalip.2011.09.009. [DOI] [PubMed] [Google Scholar]
- 3.Dashiti M, Kulik W, Hoek F, Veerman EC, Peppelenbosch MP, Rezaee F. A Phospholipidomic Analysis of All Defined Human Plasma Lipoproteins. Scientific Reports. 2011;1:1–11. doi: 10.1038/srep00139. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 4.We refer only to fatty acids with an even number of carbon atoms throughout this text.
- 5.Hermansson M, Hokynar K, Somerharju P. Mechanisms of glycerophospholipid homeostasis in mammalian cells. Prog Lipid Res. 2011;50:240–257. doi: 10.1016/j.plipres.2011.02.004. [DOI] [PubMed] [Google Scholar]
- 6.Bianco ID, Fidelio GD, Yu RK, Maggio B. Degradation of Dilauroylphosphatidylcholine by Phospholipase A2 in Monolayers Containing Glycosphingolipids. Biochem. 1991;30:1709–1714. doi: 10.1021/bi00220a037. [DOI] [PubMed] [Google Scholar]
- 7.Kucerka N, Nieh MP, Katsaras J. Fluid phase lipid areas and bilayer thicknesses of commonly used phosphatidylcholines as a function of temperature. Biochimica et Biophysica Acta. 2011;1808:2761–2771. doi: 10.1016/j.bbamem.2011.07.022. [DOI] [PubMed] [Google Scholar]
- 8.Lasic DD. The mechanism of vesicle formation. Biochem J. 1988;256:1–11. doi: 10.1042/bj2560001. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 9.Torchilin VP, Weissig V. Liposomes: A Practical Approach. 2. Oxford; New York: 2003. [Google Scholar]
- 10.Torchilin VP, Sawant RR. Liposomes as ‘smart’ pharmaceutical nanocarriers. Soft Matter. 2010;6:4026–4044. [Google Scholar]
- 11.Tsuchida E, Sou K, Nakagawa A, Sakai H, Komatsu T, Kobayashi K. Artificial Oxygen Carriers, Hemoglobin Vesicles and Albumin-Hemes, Based on Bioconjugate Chemistry. Bioconj Chem. 2009;20:1419–1440. doi: 10.1021/bc800431d. [DOI] [PubMed] [Google Scholar]
- 12.Kudlicki W, Katzen F, Peterson TC. Membrane protein expression: no cells required. Trends in Biotechnology. 2009;27:455–460. doi: 10.1016/j.tibtech.2009.05.005. [DOI] [PubMed] [Google Scholar]
- 13.Jesorka A, Orwar O. Liposomes: Technologies and Analytical Applications. Ann Rev Analytical Chem. 2008;1:801–832. doi: 10.1146/annurev.anchem.1.031207.112747. [DOI] [PubMed] [Google Scholar]
- 14.Rasch MR, Rossinyol E, Hueso JL, Goodfellow BW, Arbiol J, Korgel BA. Hydrophobic Gold Nanoparticle Self-Assembly with Phosphatidylcholine Lipid: Membrane-Loaded and Janus Vesicles. Nano Lett. 2010;10:3733–3739. doi: 10.1021/nl102387n. [DOI] [PubMed] [Google Scholar]
- 15.Park SH, Oh SG, Suh KD, Han SH, Chung DJ, Mun JY, Han SS, Kim JW. Control over micro-fluidity of liposomal membranes by hybridizing metal nanoparticles. Colloid Surf B-Biointerfaces. 2009;70:108–113. doi: 10.1016/j.colsurfb.2008.12.024. [DOI] [PubMed] [Google Scholar]
- 16.Amstad E, Kohlbrecher J, Muller E, Schweizer T, Textor M, Reimhult E. Triggered Release from Liposomes through Magnetic Actuation of Iron Oxide Nanoparticle Containing Membranes. Nano Lett. 2011;11:1664–1670. doi: 10.1021/nl2001499. [DOI] [PubMed] [Google Scholar]
- 17.Gopalakrishnan G, Danelon C, Izewska P, Prummer M, Bolinger PY, Geissbuhler I, Demurtas D, Dubochet J, Vogel H. Multifunctional lipid/quantum dot hybrid nanocontainers for controlled targeting of live cells. Angew Chem, Int Ed. 2006;45:5478–5483. doi: 10.1002/anie.200600545. [DOI] [PubMed] [Google Scholar]
- 18.Al-Jamal WT, Al-Jamal KT, Tian B, Lacerda L, Bomans PH, Frederik PM, Kostarelos K. Lipid-quantum dot bilayer vesicles enhance tumor cell uptake and retention in vitro and in vivo. ACS Nano. 2008;2:408–418. doi: 10.1021/nn700176a. [DOI] [PubMed] [Google Scholar]
- 19.Von White IG, Chen Y, Roder-Hanna J, Bothun GD, Kitchens CL. Structural and Thermal Analysis of Lipid Vesicles Encapsulating Hydrophobic Gold Nanoparticles. ACS Nano. 2012;6:4678–4685. doi: 10.1021/nn2042016. [DOI] [PubMed] [Google Scholar]
- 20.Chen Y, Bose A, Bothun GD. Controlled Release from Bilayer-Decorated Magnetoliposomes via Electromagnetic Heating. ACS Nano. 2010;4:3215–3221. doi: 10.1021/nn100274v. [DOI] [PubMed] [Google Scholar]
- 21.Wi HS, Lee K, Pak HK. Interfacial energy consideration in the organization of a quantum dot-lipid mixed system. J Phys-Condensed Matter. 2008;20:494211. [Google Scholar]
- 22.Marsh D. Lateral Pressure Profile, Spontaneous Curvature Frustration, and the Incorporation and Conformation of Proteins in Membranes. Biophys J. 2007;93:3884–3899. doi: 10.1529/biophysj.107.107938. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 23.Pansu B, Lecchi A, Constantin D, Imperor-Clerc M, Veber M, Dozov I. Insertion of Gold Nanoparticles in Fluid Mesophases: Size Filtering and Control of Interactions. J Phys Chem C. 2011;115:17682–17687. [Google Scholar]
- 24.Fox CB. Squalene Emulsion for Parenteral Vaccine and Drug Delivery. Molecules. 2009;14:3286–3312. doi: 10.3390/molecules14093286. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 25.Spanova M, Daum G. Squalene - biochemistry, molecular biology, process biotechnology, and applications. Euro J Lipid Sci and Tech. 2011;113:1299–1320. [Google Scholar]
- 26.Simon SA, Lis LJ, MacDonald RC, Kauffman JW. The Noneffect of a Large Linear Hydrocarbon, Squalene, on the Phosphatidylcholine Packing Structure. Biophys J. 1977;19:83–90. doi: 10.1016/S0006-3495(77)85570-7. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 27.Hauss T, Dante S, Dencher NA, Haines TH. Squalane is in the midplane of the lipid bilayer: implications for its function as a proton permeability barrier. Biochimica et Biophysica Acta (BBA)-Bioenergetics. 2002;1556:149–154. doi: 10.1016/s0005-2728(02)00346-8. [DOI] [PubMed] [Google Scholar]
- 28.Gianella A, Jarzyna PA, Mani V, Ramachandran S, Calcagno C, Tang J, Kann B, Dijk WJR, Thijssen VL, Griffioen AW, Storm G, Fayad ZA, Mulder WJM. Multifunctional Nanoemulsion Platform for Imaging Guided Therapy Evaluated in Experimental Cancer. ACS Nano. 2011;5:4422–4433. doi: 10.1021/nn103336a. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 29.Tilborg GAF, Cormode DP, Jarzyna PA, van der Toorn A, van der Pol SMA, van Bloois L, Fayad ZA, Storm G, Mulder WJM, de Vries HE, Dijkhuizen RM. Nanoclusters of Iron Oxide: Effect of Core Composition on Structure, Biocompatibility, and Cell Labeling Efficacy. Bioconjugate Chem. 2012;23:941–950. doi: 10.1021/bc200543k. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 30.Woodward RT, Olariu CI, Hasan EA, Yiu HHP, Rosseinsky MJ, Weaver JVM. Multi-responsive polymer-stabilized magnetic engineered emulsions as liquid-based switchable magneto-responsive actuators. Soft Matter. 2011;7:4335. [Google Scholar]
- 31.The lipids all disperse poorly in water at lower temperatures corresponding to the lamellar gel phase (Lβ′).
- 32.The scintillation vials, as well as the round bottom flasks, are thoroughly cleaned beforehand. They are sonicated in an aqueous solution of laboratory detergent, then rinsed 5 times with tap water and DI water, rinsed with EtOH, sonicated with chloroform, and then finally dried in an oven overnight.
- 33.Santos HM, Lodeiro C, Capelo-Martinez J-L. In: Ultrasound in Chemistry: Analytical Applications. Capelo-Martinez J-L, editor. Wiley-VCH Verlag GmbH & Co; Weinheim: 2009. [Google Scholar]
- 34.A sonication amplitude of 42kHz is stated on the instrument.
- 35.Taurozzi JS, Hackley VA, Wiesner MR. Ultrasonic dispersion of nanoparticles for environmental, health and safety assessment – issues and recommendations. Nanotoxicology. 2011;5:711–729. doi: 10.3109/17435390.2010.528846. [DOI] [PubMed] [Google Scholar]
- 36.Heat loss to the surroundings will be minimal near room temperature, so the measured temperature rise will correlate better with sonicator power.
- 37.Increasing the chamber temperature above 30°C was challenging because water begins to condense inside the chamber, which can easily ruin the specimen.
- 38.Dubois M, Deme B, Gulik-Krzywicid T, Dedleu JC, Vautrin C, Desert S, Perez E, Zemb T. Self-assembly of regular hollow icosahedra in salt-free catanionic solutions. Nature. 2001;411:672–675. doi: 10.1038/35079541. [DOI] [PubMed] [Google Scholar]
- 39.Blaurock AE, Gamble RC. Small phosphatidylcholine vesicles appear to be faceted below the thermal phase transition. J Membrane Biol. 1979;50:187–204. [Google Scholar]
- 40.Quemeneur F, Quilliet C, Faivre M, Viallat A, Pepin-Donat B. Gel Phase Vesicles Buckle into Specific Shapes. Phys Rev Lett. 2012;108:108303. doi: 10.1103/PhysRevLett.108.108303. [DOI] [PubMed] [Google Scholar]
- 41.Almgren M, Edwards K, Karlsson G. Cryo transmission electron microscopy of liposomes and related structures. Coll Surf A. 2000;174:3–21. [Google Scholar]
- 42.Binks BP. Particles as surfactantssimilarities differences. Curr Opinion Colloid Interface Sci. 2002;7:21–41. [Google Scholar]
- 43.Wang J, Yang F, Tan J, Liu G, Xu J, Sun D. Pickering Emulsions Stabilized by a Lipophilic Surfactant and Hydrophilic Platelike Particles. Langmuir. 2010;26:5397–5404. doi: 10.1021/la903817b. [DOI] [PubMed] [Google Scholar]
- 44.Glogowski E, He J, Russell TP, Emrick T. Mixed monolayer coverage on gold nanoparticles for interfacial stabilization of immiscible fluids. Chem Commun. 2005:4050–4052. doi: 10.1039/b503670d. [DOI] [PubMed] [Google Scholar]
- 45.Tambe DE, Sharma MM. Factors Controlling the Stability of Colloid-stabilized Emulsions. J Colloid Interface Sci. 1993;157:244–253. [Google Scholar]
- 46.Pichot R, Spyropoulos F, Norton IT. O/W emulsions stabilised by both low molecular weight surfactants and colloidal particles: The effect of surfactant type and concentration. J Colloid Interf Sci. 2010;352:128–135. doi: 10.1016/j.jcis.2010.08.021. [DOI] [PubMed] [Google Scholar]
- 47.Mason TG, Wilking JN, Meleson K, Chang CB, Graves SM. Nanoemulsions: formation, structure, and physical properties. J Phys: Condensed Matter. 2006;18:R635–R666. [Google Scholar]
Associated Data
This section collects any data citations, data availability statements, or supplementary materials included in this article.