Abstract
Purpose
Insulin is a commonly used additive in chondrogenic media for differentiating mesenchymal stem cells (MSCs). The indispensability of other bioactive factors like TGF-β or dexamethasone in these medium formulations has been shown, but the role of insulin is unclear. The purpose of this study was to investigate whether insulin is essential for MSC chondrogenesis and if there is a dose-dependent effect of insulin on MSC chondrogenesis.
Methods
We cultivated human MSCs in pellet culture in serum-free chondrogenic medium with insulin concentrations between 0 and 50 μg/ml and assessed the grade of chondrogenic differentiation by histological evaluation and determination of glycosaminoglycan (GAG), total collagen and DNA content. We further tested whether insulin can be delivered in an amount sufficient for MSC chondrogenesis via a drug delivery system in insulin-free medium.
Results
Chondrogenesis was not induced by standard chondrogenic medium without insulin and the expression of cartilage differentiation markers was dose-dependent at insulin concentrations between 0 and 10 μg/ml. An insulin concentration of 50 μg/ml had no additional effect compared with 10 μg/ml. Insulin was delivered by a release system into the cell culture under insulin-free conditions in an amount sufficient to induce chondrogenesis.
Conclusions
Insulin is essential for MSC chondrogenesis in this system and chondrogenic differentiation is influenced by insulin in a dose-dependent manner. Insulin can be provided in a sufficient amount by a drug delivery system. Therefore, insulin is a suitable and inexpensive indicator substance for testing drug release systems in vitro.
Introduction
Tissue engineering of articular cartilage is a promising approach for the repair of articular cartilage injuries. Besides chondrocytes, mesenchymal progenitor cells are a candidate cell source for cartilage tissue engineering [1]. The major advantages of mesenchymal progenitor cells compared to chondrocytes are the abundant availability and the possibility of minimally invasive extraction by bone marrow aspiration.
IGF-1 has been shown to enhance extracellular matrix (ECM) biosynthesis in chondrocytes [2–5] and chondrocyte-based tissue engineering products [6, 7]. Insulin is structurally similar to IGF-1 and can activate the IGF-1 receptor, and insulin has been shown to be a potent stimulator of ECM production in chondrocyte-based tissue engineering products [8, 9].
In vitro chondrogenesis of mesenchymal progenitor cells can be induced by a strictly defined chondrogenic medium as described by Johnstone et al. [1]. TGF-β1 and dexamethasone, which are part of this or similar chondrogenic media, have been shown to be essential for in vitro chondrogenesis of mesenchymal progenitor cells [10, 11]. Furthermore, chondrogenesis is enhanced by IGF-1 [12–14]. Besides TGF-β1 and dexamethasone, in several chondrogenic media insulin (often as one of the components of ITS premix) represents a third bioactive substance with growth factor-like character [1]. However, the indispensability and a dose-dependent effect of insulin on MSC chondrogenesis have not been demonstrated yet.
In this study, we show that insulin is an essential additive for chondrogenic differentiation of mesenchymal progenitor cells and that it influences the grade of chondrogenic differentiation dose-dependently. Furthermore, insulin can be delivered in an amount sufficient for chondrogenesis by a drug delivery system and, therefore, is an inexpensive and suitable indicator substance for drug delivery systems tested for mesenchymal progenitor cell-based cartilage tissue engineering products.
Materials and methods
Cell isolation
Human bone marrow was obtained from the iliac crest of four patients undergoing surgery with approval of the local ethics committee. Mesenchymal stem cells (MSCs) were isolated by density gradient fractionation and plastic adhesion and grown in monolayer culture in Dulbecco’s modified Eagle’s medium (DMEM) low glucose (Invitrogen, Karlsruhe, Germany) with 10 % fetal calf serum (Invitrogen) at 37 °C with 5 % CO2 until 80 % confluence.
Cell differentiation
Cells were trypsinized and aliquots of the cell suspensions containing 200,000 cells were transferred to 15-ml conical tubes and aggregates were formed by five minute centrifugation at 250 g. Cultures were maintained in Dulbecco’s modified Eagle’s medium (Invitrogen) with high glucose content, pyruvate (1 mM), ascorbate 2-phosphate (37.5 μg/ml), dexamethasone (10−7 M) (all from Sigma Aldrich, Steinheim, Germany) and TGF-β1 (10 ng/ml) (R&D Systems, Wiesbaden, Germany). ITS + 3 from Sigma contains human transferrin at 0.55 mg/ml, selenite at 0.5 μg/ml, linoleic acid at 0.47 mg/ml, oleic acid at 0.47 mg/ml, bovine serum albumin at 50 mg/ml and insulin at 1 mg/ml. A stock solution containing the same ingredients except for insulin was prepared and 1 % of this admixture was added to the differentiation medium. Insulin was added to the medium, leading to final insulin concentrations of 0, 1, 5, 10 (like using 1 % ITS + 3) and 50 μg/ml. The medium was changed three times per week and the cell aggregates were harvested on day 21 for histological evaluation and determination of the biochemical composition. Four independent experiments with cells obtained from four donors were carried out.
Drug delivery system
In an additional experiment, an insulin-loaded biodegradable matrix was added to the pellet culture under insulin-free and otherwise identical conditions in order to investigate if a sufficient amount of insulin can be delivered during cell culture. The insulin-loaded matrices were manufactured as previously described [15]. In brief, Dynasan 16 (glycerol tripalmitate, Sasol, Witten, Germany) was sterilized for two hours at 160 °C and subsequently tempered for three days at 55 °C. The lipid was powdered in a mortar and sieved through a sieve with a pore size of 106 μm under aseptic conditions. For the preparation of the desired insulin/lipid powder mixture, human insulin (Sanofi-Aventis, Frankfurt/Main, Germany) was dissolved in 0.01 N HCl and double-distilled water and filtered through a 0.22-μm filter (Corning, Germany). The insulin solution was added to mortars filled with sterile Dynasan 116 to achieve an insulin concentration of 2 %. The mixtures were freeze-dried and manually homogenized in a mortar. Cylindrical matrix discs (2 mm height, 2 mm diameter) were obtained by manual compression of accurately weighed amounts of the insulin-loaded lipid powder in a custom-made die, applying a force of approximately 250 N for ten seconds. Cylinders with an average weight of 7.0 mg were used for the experiment.
Insulin ELISA
In the experiment employing the drug delivery matrices, cell culture media were collected at the time of media change and frozen at −80 °C. The insulin content was determined by ELISA in appropriate dilutions according to the manufacturer’s instructions (Mercodia, Uppsala, Sweden). The absorption was measured at 450 nm on a plate reader (Shimadzu, Duisburg, Germany).
Histology and biochemical composition
For histological evaluation, aggregates were formalin-fixed, paraffin-embedded and stained with Toluidine blue (Sigma) for glycosaminoglycans. The average wet weight of the cell aggregates was determined by weighing pooled aggregates (six to eight aggregates per experimental group) and calculating the mean value. Pooled aggregates (six to eight per experimental group) were used for biochemical analysis of DNA content, collagen content, and glycosaminoglycan (GAG) content. They were freeze-dried and digested with 1 ml of papainase (Cell Systems, St. Katharinen, Germany) solution (3.2 U/ml in buffer) for 18 hours at 60 °C. The DNA content in the aggregates was assessed using Hoechst 33258 dye (Polysciences, Warrington, PA, USA) [16]. Hydroxyproline content was determined spectrophotometrically after acid hydrolysis and reaction with p-dimethylaminobenzaldehyde (Merck, Darmstadt, Germany) and chloramine-T [17]. The total amount of collagen was calculated using a hydroxyproline to collagen ratio of 1:10 [18]. Sulfated GAG content was determined spectrophotometrically at 540 nm after the reaction with dimethylmethylene blue (Sigma), using bovine chondroitin sulfate (ICN, Aurora, OH, USA) as standard [19].
Statistical analysis
The data shown are pooled data of four independent experiments with cells of four different donors. Values are normalized to the group containing 10 μg/ml insulin (as cultured with ITS) in the particular experiment. Statistical evaluation was carried out using a non-parametric test (Kruskal-Wallis one way analysis of variance on ranks) in SigmaStat for Windows version 2.03 (Systat Software GmbH, Erkrath, Germany).
Results
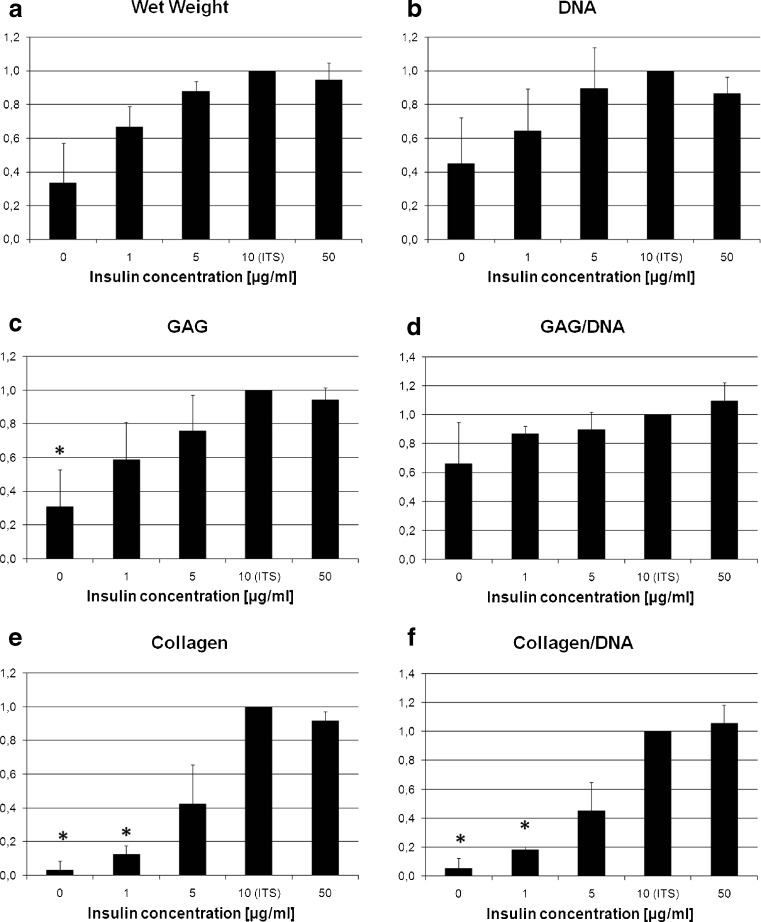
The histological evaluation revealed an insulin-dependent increase in the metachromatic Toluidine blue staining of the cell aggregates as a measure for glycosaminoglycan content between 0 and 10 μg/ml in the medium. Macroscopically, the size of the pellets also increased in this range with increasing insulin concentrations. No difference in pellet size and histological appearance could be detected between 10 and 50 μg/ml insulin (Fig. 1).
Fig. 1.
Toluidine blue stain of aggregates on day 21 as a measure of glycosaminoglycan content. a 0 μg/ml insulin. b 1 μg/ml insulin. c 5 μg/ml insulin. d 10 μg/ml insulin. e 50 μg/ml insulin
Wet weight (Fig. 2a), DNA content (Fig. 2b), total GAG content (Fig. 2c) and total collagen content (Fig. 2e) increased with increasing insulin concentrations between 0 and 10 μg/ml. GAG normalized to DNA (Fig. 2d) and collagen normalized to DNA (Fig. 2f) as a measure of the extracellular matrix production per cell, and therefore for the differentiation state, which also increased dose-dependently between 0 and 10 μg/ml insulin. GAG accumulation per aggregate was significantly lower with 0 μg/ml insulin compared to regular chondrogenic medium containing 10 μg/ml insulin (Fig. 2c). Collagen content per aggregate and collagen content normalized to DNA were significantly lower with 0 and 1 μg/ml insulin compared to regular chondrogenic medium (10 μg/ml insulin) (Fig. 2e, f). All values seemed to plateau at an insulin concentration of 10 μg/ml; increase of the insulin concentration in the medium to 50 μg/ml had no effect compared to 10 μg/ml. The absolute values for the biochemical composition and wet weight in the 10 μg/ml insulin group (control) were: wet weight 1.53 mg per aggregate, DNA content 1.65 μg per aggregate, GAG content 36.3 μg per aggregate, collagen content 35.0 μg per aggregate.
Fig. 2.
Wet weight (a), DNA content (b), GAG content (c), GAG content normalized to DNA (d), total collagen content (e) and collagen content normalized to DNA (f) on day 21, here shown relative to the values for the concentration of 10 μg/ml insulin (as in regular chondrogenic medium) (*p < 0.05)
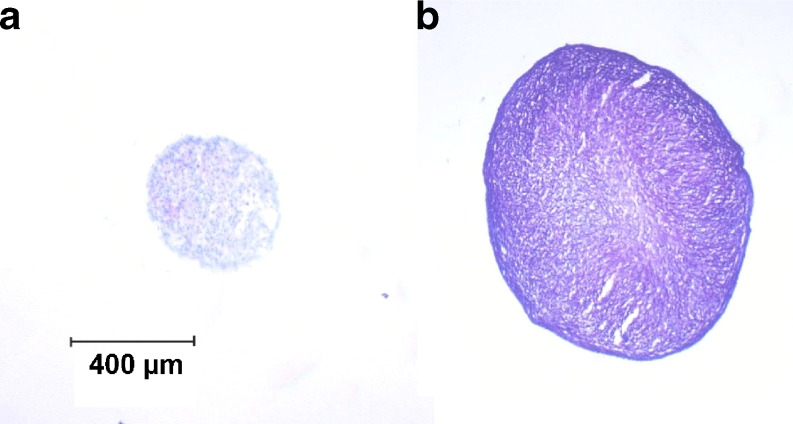
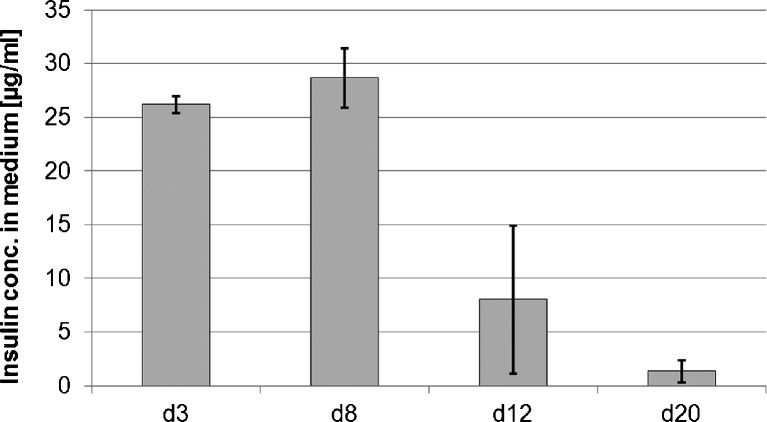
Employing the insulin-loaded drug delivery system in insulin-free medium, the induction of chondrogenic differentiation by insulin released from the drug delivery system could be clearly observed (Fig. 3). The matrix provided high insulin levels until day eight (Fig. 4). The insulin concentration in the medium decreased thereafter.
Fig. 3.
Toluidine blue stain of aggregates on day 21 as a measure of glycosaminoglycan content. a 0 μg/ml insulin without drug delivery system. b 0 μg/ml insulin with drug delivery system
Fig. 4.
Insulin concentration in the medium obtained with the drug delivery system over time (without additional exogenous insulin). n = 2; error bars: minimum and maximum value
Discussion
In this study, employing the pellet culture system, insulin was demonstrated to elicit dose-dependent effects on the extent of chondrogenesis in differentiating mesenchymal progenitor cells, as shown with regard to histological appearance and biochemical composition. Well in agreement with the obtained results, similar effects of insulin on chondrocyte-based tissue engineering products have previously been observed [9]. Insulin is homologous to parts of the IGF-1 molecule [8]; IGF-1 is a potent stimulator of extracellular matrix production in chondrocytes [2–5] and has also anabolic effects on mesenchymal progenitor cells undergoing chondrogenesis at a concentration of 100 ng/ml in the medium [12, 14, 20]. Insulin activates the IGF-1 receptor with a 100-fold lower binding constant [8, 9] and insulin binding to the insulin receptor in heterodimeric insulin/IGF-1 hybrid receptors can subsequently activate the IGF-1 receptor [21]. The maximum stimulation in our experiment was observed at an insulin concentration of 10 μg/ml, 100-fold the concentration of IGF-1 used in the experiments mentioned above. In this context, it is likely that the effect of insulin is caused by either direct or indirect activation of the IGF-1 receptor.
The low DNA content on day 21 in the group without insulin compared to the other treatment groups and the trend with increased DNA content up to an insulin concentration of 10 μg/ml showed that the cell number per pellet was dose-dependent. A dose-dependent effect of IGF-1 on cell proliferation in chondrocytes [22] and osteoblast culture [23] has been described previously and a similar effect of insulin and IGF-1 on cell proliferation in chick embryo chondrogenic cells has been reported [24]. In murine bone marrow-derived MSCs, IGF-1 has a proliferative effect [12]. In dental pulp cells, IGF-1, IGF-2 and insulin increase cell proliferation and differentiation dose-dependently [25]. These data support our findings.
Histological and biochemical evaluation showed a dose-dependent increase in chondrogenic differentiation of human MSCs at insulin concentrations between 0 and 10 μg/ml. In other studies investigating the effect of insulin/IGF-1 on extracellular matrix production also increased GAG and collagen production were observed, yet a more distinct effect on GAG deposition than collagen production was reported [6, 9]. In this study investigating MSC in the pellet culture system, we have seen a more impressive effect on collagen than on GAG deposition, but this may be explained by the different cell type used and the different cell culture system (chondrocytes in a scaffold-based system in the previous studies) [6, 9].
In general, the results of this study show that there is a positive dose-dependent effect of insulin on the chondrogenic differentiation of human MSCs. This and the low costs make insulin, with the MSC pellet culture system, a suitable and affordable factor for testing drug delivery systems for chondrogenic differentiation in vitro. In the experiment employing the drug delivery matrices, we could show that insulin can be delivered by a release system in vitro in a dose that is sufficient for chondrogenic differentiation. The release system used in this experiment provided high insulin concentrations until day eight. This was sufficient to induce chondrogenesis in aggregate culture of MSCs, shown by the histological results.
The ultimate meaning of drug delivery systems is the in vivo application for tissue repair. It has to be noted that, for this purpose, the drug release system and the cell culture system presented here are not directly suitable. For clinical applications, ideally the drug delivery system should be integrated into the matrix of a tissue engineering product, so that no additional material needs to be implanted and the bioactive factors will be released close to their target cells. Furthermore, we used a well established culture model that is frequently used for in vitro experiments, but clinically not applicable. Nevertheless, based on the results of the model system presented here, insulin may be used in a clinically more relevant setting for chondrogenic differentiation of mesenchymal stem cells using a scaffold- or hydrogel-based three-dimensional tissue engineering system.
References
- 1.Johnstone B, Hering TM, Caplan AI, Goldberg VM, Yoo JU. In vitro chondrogenesis of bone marrow-derived mesenchymal progenitor cells. Exp Cell Res. 1998;238(1):265–272. doi: 10.1006/excr.1997.3858. [DOI] [PubMed] [Google Scholar]
- 2.Barone-Varelas J, Schnitzer TJ, Meng Q, Otten L, Thonar EJ. Age-related differences in the metabolism of proteoglycans in bovine articular cartilage explants maintained in the presence of insulin-like growth factor I. Connect Tissue Res. 1991;26(1–2):101–120. doi: 10.3109/03008209109152167. [DOI] [PubMed] [Google Scholar]
- 3.Davies LC, Blain EJ, Gilbert SJ, Caterson B, Duance VC. The potential of IGF-1 and TGFbeta1 for promoting “adult” articular cartilage repair: an in vitro study. Tissue Eng Part A. 2008;14(7):1251–1261. doi: 10.1089/ten.tea.2007.0211. [DOI] [PubMed] [Google Scholar]
- 4.Loeser RF, Chubinskaya S, Pacione C, Im HJ. Basic fibroblast growth factor inhibits the anabolic activity of insulin-like growth factor 1 and osteogenic protein 1 in adult human articular chondrocytes. Arthritis Rheum. 2005;52(12):3910–3917. doi: 10.1002/art.21472. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 5.Tyler JA. Insulin-like growth factor 1 can decrease degradation and promote synthesis of proteoglycan in cartilage exposed to cytokines. Biochem J. 1989;260(2):543–548. doi: 10.1042/bj2600543. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 6.Blunk T, Sieminski AL, Gooch KJ, Courter DL, Hollander AP, Nahir AM, Langer R, Vunjak-Novakovic G, Freed LE. Differential effects of growth factors on tissue-engineered cartilage. Tissue Eng. 2002;8(1):73–84. doi: 10.1089/107632702753503072. [DOI] [PubMed] [Google Scholar]
- 7.Elder BD, Athanasiou KA. Systematic assessment of growth factor treatment on biochemical and biomechanical properties of engineered articular cartilage constructs. Osteoarthritis Cartilage. 2008;17(1):114–123. doi: 10.1016/j.joca.2008.05.006. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 8.Schmid C. Insulin-like growth factors. Cell Biol Int. 1995;19(5):445–457. doi: 10.1006/cbir.1995.1088. [DOI] [PubMed] [Google Scholar]
- 9.Kellner K, Schulz MB, Gopferich A, Blunk T. Insulin in tissue engineering of cartilage: a potential model system for growth factor application. J Drug Target. 2001;9(6):439–448. doi: 10.3109/10611860108998778. [DOI] [PubMed] [Google Scholar]
- 10.Derfoul A, Perkins GL, Hall DJ, Tuan RS. Glucocorticoids promote chondrogenic differentiation of adult human mesenchymal stem cells by enhancing expression of cartilage extracellular matrix genes. Stem Cells. 2006;24(6):1487–1495. doi: 10.1634/stemcells.2005-0415. [DOI] [PubMed] [Google Scholar]
- 11.Buxton AN, Bahney CS, Yoo JU, Johnstone B. Temporal exposure to chondrogenic factors modulates human mesenchymal stem cell chondrogenesis in hydrogels. Tissue Eng Part A. 2011;17(3–4):371–380. doi: 10.1089/ten.tea.2009.0839. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 12.Longobardi L, O’Rear L, Aakula S, Johnstone B, Shimer K, Chytil A, Horton WA, Moses HL, Spagnoli A. Effect of IGF-I in the chondrogenesis of bone marrow mesenchymal stem cells in the presence or absence of TGF-beta signaling. J Bone Miner Res. 2006;21(4):626–636. doi: 10.1359/jbmr.051213. [DOI] [PubMed] [Google Scholar]
- 13.Uebersax L, Merkle HP, Meinel L. Insulin-like growth factor I releasing silk fibroin scaffolds induce chondrogenic differentiation of human mesenchymal stem cells. J Control Release. 2008;127(1):12–21. doi: 10.1016/j.jconrel.2007.11.006. [DOI] [PubMed] [Google Scholar]
- 14.Worster AA, Brower-Toland BD, Fortier LA, Bent SJ, Williams J, Nixon AJ. Chondrocytic differentiation of mesenchymal stem cells sequentially exposed to transforming growth factor-beta1 in monolayer and insulin-like growth factor-I in a three-dimensional matrix. J Orthop Res. 2001;19(4):738–749. doi: 10.1016/S0736-0266(00)00054-1. [DOI] [PubMed] [Google Scholar]
- 15.Appel B, Maschke A, Weiser B, Sarhan H, Englert C, Angele P, Blunk T, Gopferich A. Lipidic implants for controlled release of bioactive insulin: effects on cartilage engineered in vitro. Int J Pharm. 2006;314(2):170–178. doi: 10.1016/j.ijpharm.2005.11.049. [DOI] [PubMed] [Google Scholar]
- 16.Kim YJ, Sah RL, Doong JY, Grodzinsky AJ. Fluorometric assay of DNA in cartilage explants using Hoechst 33258. Anal Biochem. 1988;174(1):168–176. doi: 10.1016/0003-2697(88)90532-5. [DOI] [PubMed] [Google Scholar]
- 17.Woessner JF., Jr The determination of hydroxyproline in tissue and protein samples containing small proportions of this amino acid. Arch Biochem Biophys. 1961;93:440–447. doi: 10.1016/0003-9861(61)90291-0. [DOI] [PubMed] [Google Scholar]
- 18.Hollander AP, Heathfield TF, Webber C, Iwata Y, Bourne R, Rorabeck C, Poole AR. Increased damage to type II collagen in osteoarthritic articular cartilage detected by a new immunoassay. J Clin Invest. 1994;93(4):1722–1732. doi: 10.1172/JCI117156. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 19.Farndale RW, Buttle DJ, Barrett AJ. Improved quantitation and discrimination of sulphated glycosaminoglycans by use of dimethylmethylene blue. Biochim Biophys Acta. 1986;883(2):173–177. doi: 10.1016/0304-4165(86)90306-5. [DOI] [PubMed] [Google Scholar]
- 20.Fukumoto T, Sperling JW, Sanyal A, Fitzsimmons JS, Reinholz GG, Conover CA, O’Driscoll SW. Combined effects of insulin-like growth factor-1 and transforming growth factor-beta1 on periosteal mesenchymal cells during chondrogenesis in vitro. Osteoarthr Cartil. 2003;11(1):55–64. doi: 10.1053/joca.2002.0869. [DOI] [PubMed] [Google Scholar]
- 21.Pandini G, Frasca F, Mineo R, Sciacca L, Vigneri R, Belfiore A. Insulin/insulin-like growth factor I hybrid receptors have different biological characteristics depending on the insulin receptor isoform involved. J Biol Chem. 2002;277(42):39684–39695. doi: 10.1074/jbc.M202766200. [DOI] [PubMed] [Google Scholar]
- 22.Nixon AJ, Lillich JT, Burton-Wurster N, Lust G, Mohammed HO. Differentiated cellular function in fetal chondrocytes cultured with insulin-like growth factor-I and transforming growth factor-beta. J Orthop Res. 1998;16(5):531–541. doi: 10.1002/jor.1100160503. [DOI] [PubMed] [Google Scholar]
- 23.Yeh LC, Adamo ML, Olson MS, Lee JC. Osteogenic protein-1 and insulin-like growth factor I synergistically stimulate rat osteoblastic cell differentiation and proliferation. Endocrinology. 1997;138(10):4181–4190. doi: 10.1210/en.138.10.4181. [DOI] [PubMed] [Google Scholar]
- 24.Quarto R, Campanile G, Cancedda R, Dozin B. Modulation of commitment, proliferation, and differentiation of chondrogenic cells in defined culture medium. Endocrinology. 1997;138(11):4966–4976. doi: 10.1210/en.138.11.4966. [DOI] [PubMed] [Google Scholar]
- 25.Onishi T, Kinoshita S, Shintani S, Sobue S, Ooshima T. Stimulation of proliferation and differentiation of dog dental pulp cells in serum-free culture medium by insulin-like growth factor. Arch Oral Biol. 1999;44(4):361–371. doi: 10.1016/S0003-9969(99)00007-2. [DOI] [PubMed] [Google Scholar]