Abstract
Background
Changes in right ventricular (RV) morphology are associated with morbidity and mortality in heart and lung disease. We examined the association of abnormal RV structure and function with the risk of heart failure (HF) or cardiovascular death in a population-based multiethnic sample free of clinical cardiovascular disease at baseline.
Methods and Results
The Multi-Ethnic Study of Atherosclerosis (MESA) performed cardiac magnetic resonance imaging (MRI) on 5098 participants between 2000–2002 with follow-up for incident heart failure and cardiovascular death (“death”) until January 2008. RV volumes and mass were available for 4204 participants. The study sample (N = 4,144) was 61.4 ± 10.1 years old and 47.6 % male. The presence of RV hypertrophy (increased RV mass) was associated with a more than twice the risk of heart failure or death after adjustment for demographics, body mass index, education, C-reactive protein level, hypertension, and smoking status (HR = 2.52, 95%CI 1.55–4.10, p < 0.001) and a doubling of risk (or more) with left ventricular mass at the mean value or lower (p for interaction = 0.05).
Conclusions
RV hypertrophy was associated with the risk of heart failure or death in a multi-ethnic population free of clinical cardiovascular disease at baseline.
Keywords: right ventricle, pulmonary heart disease, magnetic resonance imaging, pulmonary hypertension, survival
The role of the right ventricle (RV) in determining outcomes in a variety of heart and lung diseases has become well-recognized.1, 2 RV volumes and RV ejection fraction (RVEF) are associated with the risk of death in patients with pulmonary arterial hypertension (PAH),3, 4 and recent studies have demonstrated that RVEF is also associated with survival in more common cardiovascular diseases, such as chronic heart failure.5, 6 Cor pulmonale has long been described in the setting of chronic obstructive pulmonary disease and has historically been associated with an increased risk of death.7
RV structure and function are challenging to measure non-invasively due to the shape and mode of contraction of the RV, which defy many of the geometric assumptions necessary for measurement using standard transthoracic echocardiography. The thin free wall of the RV in individuals without significant cardiopulmonary disease is particularly difficult to accurately measure. Magnetic resonance imaging (MRI) is considered the gold standard for non-invasive assessment of the RV, since no geometric assumptions are necessary. However, expense, availability, and patient tolerance limit the use of MRI in large epidemiologic cohorts.
RV changes may not only serve as a sensitive indicator of subclinical disease of the left ventricle (LV) or the lungs but also directly contribute to the onset of clinical heart failure (HF) (by contributing to manifestations such as dyspnea or lower extremity edema) or cardiovascular death. Age, sex, race, and other factors affect RV morphology;8–10 however, the clinical impact of extreme changes in RV structure and function on long-term outcomes is currently unknown.
We hypothesized that RV hypertrophy, RV enlargement, and RV systolic dysfunction are associated with the risk of HF or cardiovascular death in a large, prospective cohort study of adults without clinical cardiovascular disease at baseline.
METHODS
Subjects
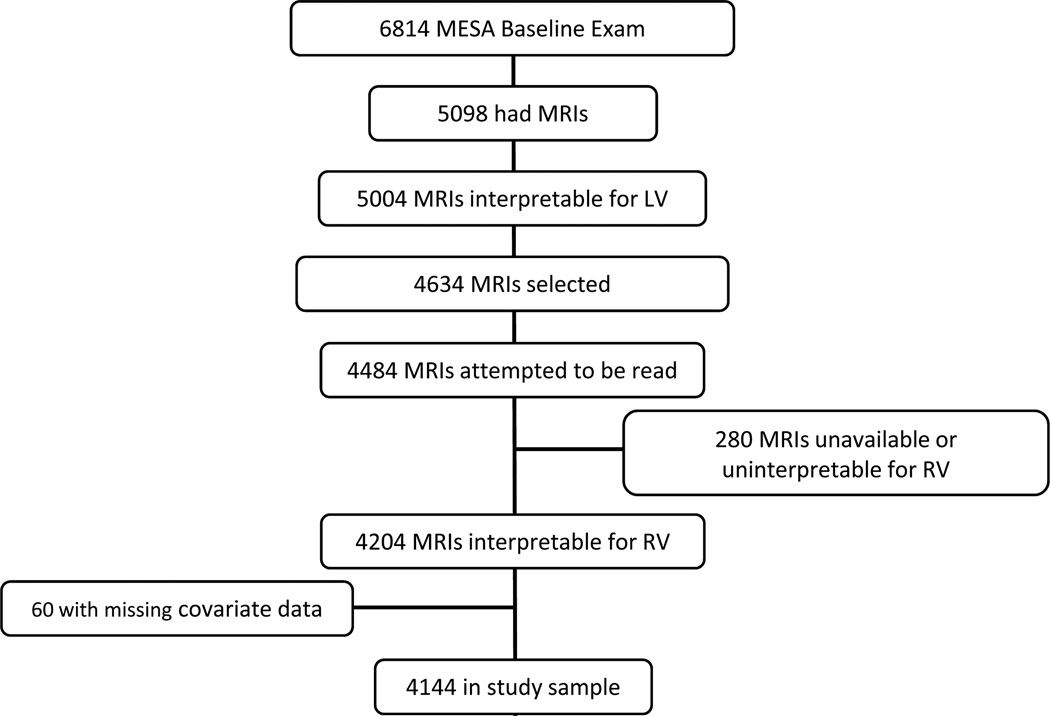
The Multi-Ethnic Study of Atherosclerosis (MESA) is a multicenter prospective cohort study designed to investigate the prevalence, correlates, and progression of subclinical cardiovascular disease in Caucasians, African-Americans, Hispanics, and Chinese-Americans without clinical cardiovascular disease at baseline.11 In 2000–2002, MESA recruited 6814 men and women aged 45–84 years old from six U.S. communities: Forsyth County, NC; Northern Manhattan and the Bronx, NY; Baltimore City and Baltimore County, MD; St. Paul, MN; Chicago, IL; and Los Angeles, CA. Exclusion criteria included clinical cardiovascular disease (physician diagnosis of heart attack, stroke, transient ischemic attack, heart failure, angina, current atrial fibrillation, any cardiovascular procedure), weight > 136 kgs (300 lbs), pregnancy, or impediment to long-term participation. Hypertension, diabetes mellitus, and hyperlipidemia were not considered as clinical cardiovascular diseases by design of the parent study, and subjects with these conditions were therefore eligible for inclusion.11 The MESA-Right Ventricle Study is an ancillary study supported by an NIH grant that planned for the selection of 4634 participants with interpretable cardiac MRIs at the baseline examination for measurement of RV morphology, which was completed in 4204 (Figure 1). Participants were sampled without regard to clinical variables.
Figure 1.
Study sample
The protocols of MESA were approved by the Institutional Review Boards of all collaborating institutions and the National Heart Lung and Blood Institute (NHLBI). NHLBI staff participated in the design of the MESA study.
Cardiac Magnetic Resonance Imaging Measures
The cardiac MRI protocol and interpretation of RV parameters have been previously described.8, 12, 13 All imaging was performed on 1.5 T magnets with a 4-element phased-array surface coil positioned anteriorly and posteriorly and electrocardiographic gating. Imaging consisted of fast gradient echo cine images with temporal resolution ≤ 50 ms.
Methods for interpretation of LV and RV parameters have been previously reported.8, 12–14 Cardiac MRI examinations were transmitted to the reading center at Johns Hopkins University in Baltimore, MD using the DICOM transfer protocol. Image analysis was done on Windows workstations using the QMASS software (v4.2, Medis, the Netherlands). Images were magnified to 250%. Image contrast was set to 55, image brightness was set to 55, and window width and level were set using the auto function in QMASS to minimum and maximum pixel values of 0 and 238 respectively.
The endocardial and epicardial borders of the RV were traced manually on the short axis cine images at the end-systolic and end-diastolic phase. Full visualization of the correct placement of RV contours relied on evaluation of cine images to determine the demarcation between the right atrium and RV. Contours were modified at basal slices of the heart by carefully identifying the tricuspid valve so as to exclude the right atrium and avoid overestimation of the volumes. The outflow tract was included in the RV volume. Papillary muscles and trabeculae were included in the RV volumes and excluded from RV mass, as is commonly done for LV mass.15, 16
RV end-diastolic volume (RVEDV) and RV end-systolic volume (RVESV) were calculated using Simpson's rule by summation of areas on each slice multiplied by the sum of slice thickness and image gap. RV mass was determined at the end-diastole phase as the difference between end-diastolic epicardial and endocardial volumes multiplied by the specific gravity of myocardium (1.05 g/ml).12 The septum was not included in the measurement of RV mass. RVEF was calculated by dividing the difference between RVEDV and RVESV by RVEDV. RV hypertrophy, RV enlargement, and RV dysfunction were respectively defined as RV mass, RVEDV (or RVESV) and RVEF above/below the 95th/5th %ile using sex-specific normative equations based on age, height, and weight derived from a healthy, normal-weight, non-smoking subpopulation of the MESA cohort.9 These parameters were indexed to body size and therefore do not require indexing (e.g., for body surface area) in the analyses.
The intra-reader intraclass correlation coefficient from random, blinded re-reads of 229 scans for RV mass was 0.94 and was 0.99 and 0.89 for RVEDV and RVEF from 230 scans. The inter-reader intraclass correlation coefficients from random, blinded re-reads of 240 scans for RV mass and RVEDV were 0.89 and 0.96 respectively and 0.80 for RVEF.
Covariates
Race/ethnicity was self-reported during the baseline MESA exam according to 2000 US Census criteria as race (Caucasian, African-American, Chinese) and ethnicity (Hispanic or non-Hispanic). Participants self-identifying as Hispanic were categorized as Hispanic. Standard methods were used to ascertain body mass index, waist and hip circumference, self-reported intentional exercise, history of blood clots, use of aspirin, hormone therapy, statins, or antihypertensive medication, smoking status (classified as never, former, or current) and pack-years, blood pressure, hypertension, diabetes mellitus, highly-sensitive plasma C-reactive protein levels, hemoglobin A1c, creatinine, insulin and plasma interleukin-6 levels.17
End Points
We followed the cohort for incident cardiovascular events from 2000–2002 through January 2008. At intervals of 9–12 months, a telephone interviewer contacted each participant to inquire about all interim hospital admissions, cardiovascular outpatient diagnoses and procedures, and deaths. In addition, MESA occasionally identified additional medical encounters through cohort clinic visits, participant call-ins, medical record abstractions or obituaries. In order to verify self-reported diagnoses, we requested copies of all death certificates and medical records for all hospitalizations and selected outpatient cardiovascular diagnoses and procedures. We also obtained next of kin interviews for out of hospital cardiovascular deaths.
Trained personnel abstracted any hospital records suggesting possible cardiovascular events. They recorded symptoms, history and biomarkers; scanned ECGs, echocardiograms, catheterization reports, outpatient records, and other relevant diagnostic and procedure reports; and transmitted these to the coordinating center. The coordinating center collated the abstracted or original endpoint records and sent them to two paired physicians for independent endpoint classification and assignment of incidence dates. Cardiologists or cardiovascular physician epidemiologists reviewed non-neurovascular endpoints; neurologists reviewed all neurovascular endpoints. If the reviewing pair disagreed on the classification, they adjudicated differences. If disagreements persisted, the full review committee made the final classification. Causes of death for cardiovascular deaths were assigned through committee review.
The definitions of HF and cardiovascular death were similar to those used in multiple other large cardiovascular cohort studies. Definite or probable HF required HF symptoms, such as shortness of breath or edema. In addition to symptoms, probable HF required HF diagnosed by a physician and the patient receiving medical treatment for HF. Definite HF required one or more other criteria, such as pulmonary edema/congestion by chest radiograph; dilated ventricle or poor LV function by echocardiocardiography or ventriculography; or evidence of LV diastolic dysfunction. We considered participants not meeting any criteria, including just a physician diagnosis of HF without any other evidence, as not having HF. Cardiovascular deaths (referred to as “deaths” from here forward) included fatal coronary heart disease, fatal stroke, and other fatal cardiovascular diseases. All personnel performing data collection and determination of HF and death end points were unaware of the RV measurements.
Statistical Analysis
Continuous variables were expressed as means and standard deviations (SDs) or ranges. Categorical variables were expressed as percentages. Cox proportional hazards models were used to estimate hazard ratios for the association of RV parameters with time to HF or death. Participants were censored at the time of non-cardiovascular death. We estimated unadjusted models for RV hypertrophy, RV enlargement, and RV dysfunction.9 We then adjusted for age, sex, body mass index (BMI), race/ethnicity, education, and C-reactive protein (CRP) levels (which were forced into the models) and other covariates which changed the RV parameter coefficient by > 15% in any of the models using purposeful selection of covariates.18
By protocol, the MESA parent study suggests the inclusion of some measure of socioeconomic status (e.g., education) in the analysis. Anthropometrics, diabetes status, hypertension, smoking status, blood pressure, cholesterol and lipoprotein levels, creatinine, CRP levels, total minutes of intentional exercise, use of antihypertension medication or statins, and site were measured at the baseline examination (2000–2002) and at three subsequent examinations (between 2002–2004, 2004–2005, and 2005–2007) and were assessed as time-varying covariates. Interactions of the RV parameters with age, sex, race/ethnicity, and LV parameters were assessed. The proportional hazards assumption was tested for all models using Schoenfeld residuals and model fit was assessed with log-log plots. The population attributable risk of RV hypertrophy adjusted for confounders with 95% CI was calculated using published methods.19
Additional analyses were performed after adjusting for spirometry measures (forced expiratory volume in 1 sec (FEV1), forced vital capacity (FVC)), urine cotinine, and percent emphysema measured on the lung windows of cardiac computed tomography (CT) in a subset of participants (n = 2704) selected between 2004 – 2006 for another ancillary study (MESA-Lung).20, 21 Adjusting for FEV1 and FVC individually accounts for the presence of significant lung disease, since these parameters accounted for > 90% of the variance in the FEV1/FVC ratio and FVC reflects restriction. SAS (Version 9.3) or R (Version 2.14) was used for all analyses. P values < 0.05 were considered statistically significant.
RESULTS
MESA included 6814 participants (Figure 1). Of these, 5098 had cardiac MRIs of which 5004 were interpretable for LV measures. The MESA-Right Ventricle Study selected 4634 scans from the 5004 (without regard to age, sex, or race) and attempted to read 4484, of which 4204 were available and interpretable for RV morphology. Participants with interpretable scans (n = 4204) were similar to those without interpretable scans (n = 280) (Supplemental Table 1). Table 1 shows the characteristics of the study sample with complete covariate data (N = 4144), compared to those excluded (N = 2670). The mean age of the study sample was 61.4 ± 10.1 years, and 47.6 % were men. Thirty-nine percent self-identified as Caucasian, 26.1 % as African-American, 22.0 % as Hispanic, and 12.5 % Chinese. The mean BMI was 27.9 ± 5.0 kg/m2. The study sample was slightly younger and had somewhat lower body mass index compared to those excluded. Supplemental Table 2 shows the mean ± SD for all RV measures, stratified by sex and race/ethnicity.
Table 1.
Characteristics of the study population at baseline
| Variable | Value | Study Sample (N=4,144) |
Excluded (N=2,670) |
|---|---|---|---|
| Age (yrs) | 61.4 +/− 10.1 | 63.3 +/− 10.4 | |
| Sex | Male | 1,972 (47.6) | 1,241 (46.5) |
| BMI (kg/m2) | 27.9 +/− 5.0 | 29.1 +/− 6.1 | |
| Race/ethnicity | Caucasian | 1,628 (39.3) | 994 (37.2) |
| African American | 1,083 (26.1) | 810 (30.3) | |
| Hispanic | 913 (22.0) | 583 (21.8) | |
| Chinese | 520 (12.5) | 283 (10.6) | |
| Education Level | < High School | 673 (16.2) | 552 (20.9) |
| High School/GED | 772 (18.6) | 464 (17.5) | |
| < College (> High School) |
1,176 (28.4) | 761 (28.7) | |
| Bachelor's Degree | 747 (18.0) | 424 (16.0) | |
| Graduate Degree | 776 (18.7) | 446 (16.8) | |
| Systolic Blood Pressure (mmHg) |
125.4 +/− 21.0 | 128.4 +/− 22.1 | |
| Diastolic Blood Pressure (mm Hg) |
71.9 +/− 10.2 | 72.0 +/− 10.4 | |
| Hypertension | 1,774 (42.8) | 1,284 (48.1) | |
| Diabetes mellitus | Normal (Blood glucose <100 mg/dl) |
3,114 (75.3) | 1,878 (70.8) |
| Impaired Fasting Glucose (Blood glucose = 100–125 mg/dl) |
545 (13.2) | 394 (14.9) | |
| Untreated Diabetes (Blood glucose ≥ 126 mg/dl) |
101 (2.4) | 78 (2.9) | |
| Treated Diabetes | 378 (9.1) | 302 (11.4) | |
| Statin use | 607 (14.7) (N = 4142) |
402 (15.1) (N =2669) |
|
| C-reactive protein (CRP) (mg/L) |
3.5 +/− 5.6 | 4.2 +/− 6.3 | |
| Smoking Status | Never | 2,142 (51.7) | 1,276 (48.2) |
| Former | 1,481 (35.7) | 1,006 (38.0) | |
| Current | 521 (12.6) | 366 (13.8) | |
| Pack Years | 10.8 +/− 22.7 (N =4097) |
12.3 +/− 21.5 (N = 2623) |
Data shown as mean ± standard deviation or n (%).
The median follow-up time was 5.8 yrs and the total follow-up time was 23,264 person-years. There were 78 incident HF events (2.0%) (17 probable and 61 definite) and 34 deaths (0.8%) during follow-up. Of the deaths, 21 (61.8%) were due to coronary heart disease, 4 (11.8%) were from stroke, and 9 (26.5%) were from other cardiovascular diseases. Eleven participants who died had a preceding HF event, which was considered as the end point in those individuals. The five-year risk of HF and death was 1.6% and 0.7% respectively.
Participants with RV hypertrophy (n = 302) had more than triple the risk of HF or death compared to those with normal RV mass (Table 2). This association persisted after adjustment for age, sex, race/ethnicity, BMI, education, CRP, hypertension and smoking status (Table 2, Figure 2A). RV hypertrophy was still associated with the risk of HF or death with effect modification by LV mass (p for interaction = 0.05) (Table 2, Figures 2B–C). RV hypertrophy was more strongly associated with outcome at lower levels of LV mass. The population attributable risk of RV hypertrophy in the adjusted model was 12.4% (95%CI 1.9% – 22.7%).
Table 2.
Proportional hazards models for abnormal RV morphology and HF or death
| HR | 95%CI | P value | |
|---|---|---|---|
| RV hypertrophy | |||
| Unadjusted | 3.20 | 1.98 – 5.17 | < 0.001 |
| Adjusted* | 2.52 | 1.55 – 4.10 | < 0.001 |
| Adjusted + LV Mass (at mean, 145 g) | 1.93 | 1.06 – 3.54 | 0.03 |
| RV enlargement (RVEDV) | |||
| Unadjusted | 1.65 | 0.92– 2.96 | 0.09 |
| Adjusted | 1.43 | 0.79 – 2.60 | 0.23 |
| Adjusted + LVEDV | 0.84 | 0.45 – 1.56 | 0.58 |
| RV enlargement (RVESV) | |||
| Unadjusted | 2.07 | 1.08 – 3.97 | 0.03 |
| Adjusted* | 1.92 | 0.99 – 3.75 | 0.05 |
| Adjusted + LVESV | 1.09 | 0.54 – 2.18 | 0.81 |
| RV systolic dysfunction | |||
| Unadjusted | 0.82 | 0.33 – 2.02 | 0.67 |
| Adjusted | 0.76 | 0.31 – 1.88 | 0.55 |
| Adjusted + LVEF | 0.46 | 0.18 –1.15 | 0.10 |
Adjusted for age, sex, race/ethnicity, BMI, education, CRP, hypertension, and smoking status.
Figure 2.
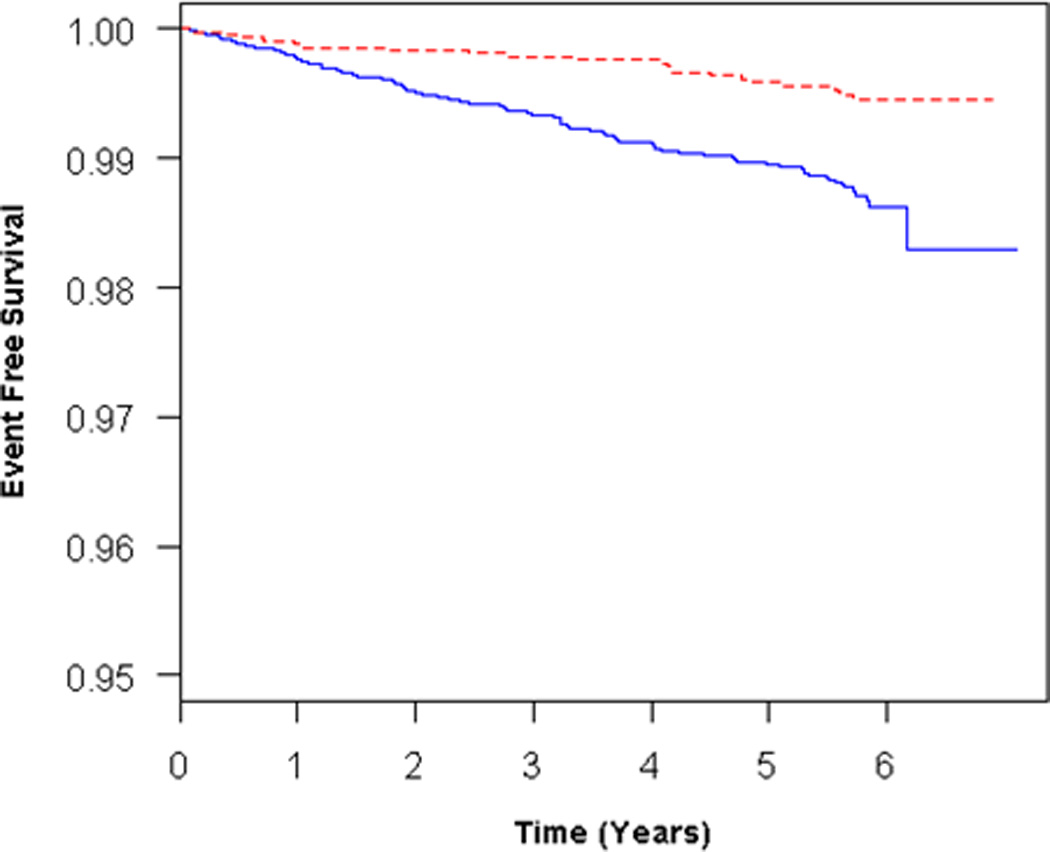
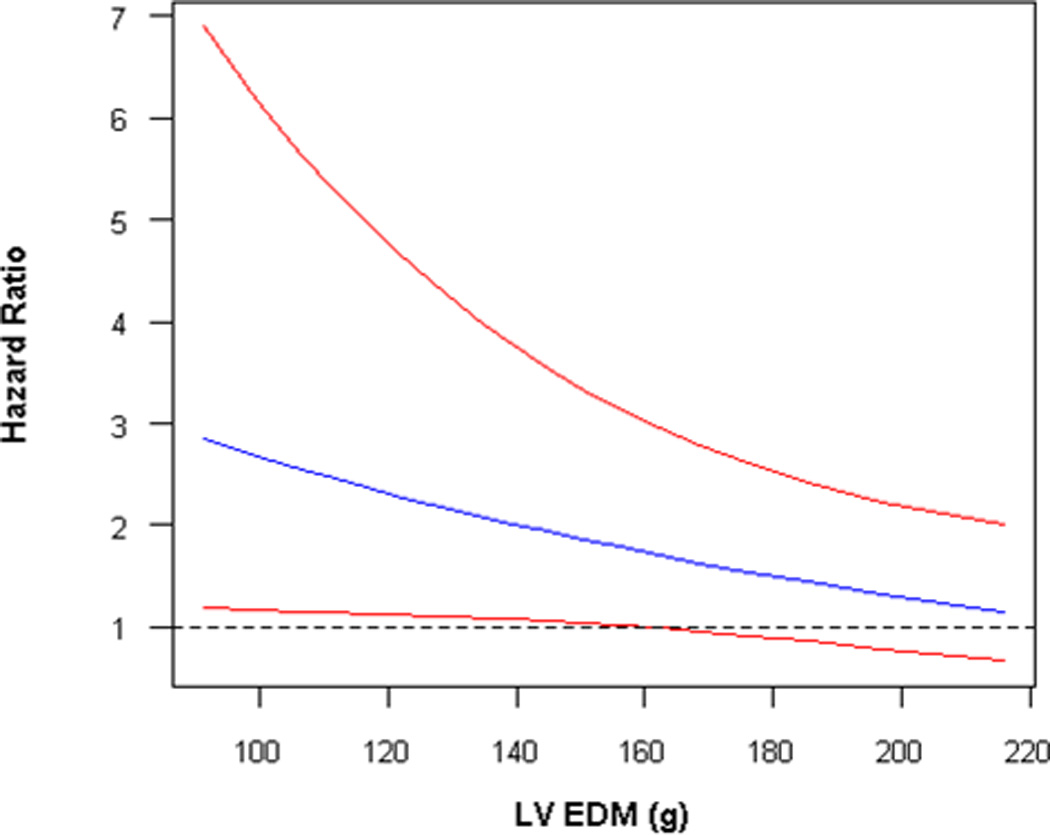
A) Adjusted survival curves for RV hypertrophy vs normal, adjusted for age, sex, race/ethnicity, BMI, education, CRP, hypertension, and smoking status, and B) Adjusted survival curves for RV hypertrophy vs normal, adjusted for all covariates + LV mass at mean LV mass (145 g). Solid blue = RV hypertrophy, dashed red = normal RV mass. C) Interaction of RV hypertrophy with LV mass in terms of the risk of HF or death. Solid blue = estimate, solid red = 95% confidence intervals.
Participants with RV enlargement (RVEDV, n = 308) tended to have an increased risk of HF or death in unadjusted and adjusted models (Table 2, Figure 3A), but these findings were not statistically significant. Further adjustment for LVEDV reduced the effect estimate (Table 2, Figure 3B). RV enlargement (at end-systole) (RVESV, n = 204) was associated with an increased risk of HF or death in the unadjusted and possibly in the adjusted models (Table 2, Figure 4A). Adjustment for LVESV weakened this association (Figure 4B). RV systolic dysfunction (RVEF, n = 248) was not associated with outcomes in unadjusted analysis or after adjustment for covariates (Table 2). There were no significant interactions of the RV parameters with age, sex, race/ethnicity, so that stratified analyses were not required. Results from analyses using only HF as the end point were similar to those from analyses using the combined end point of HF or cardiovascular death (Supplemental Tables 3–4).
Figure 3.
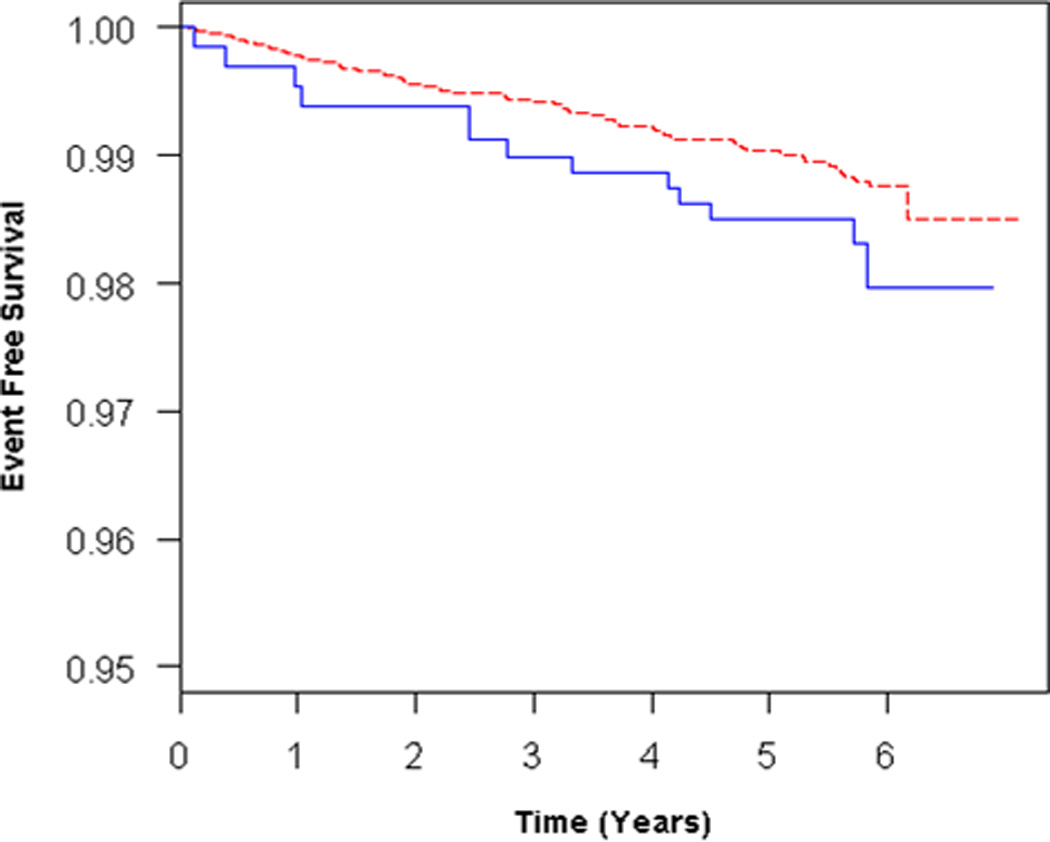
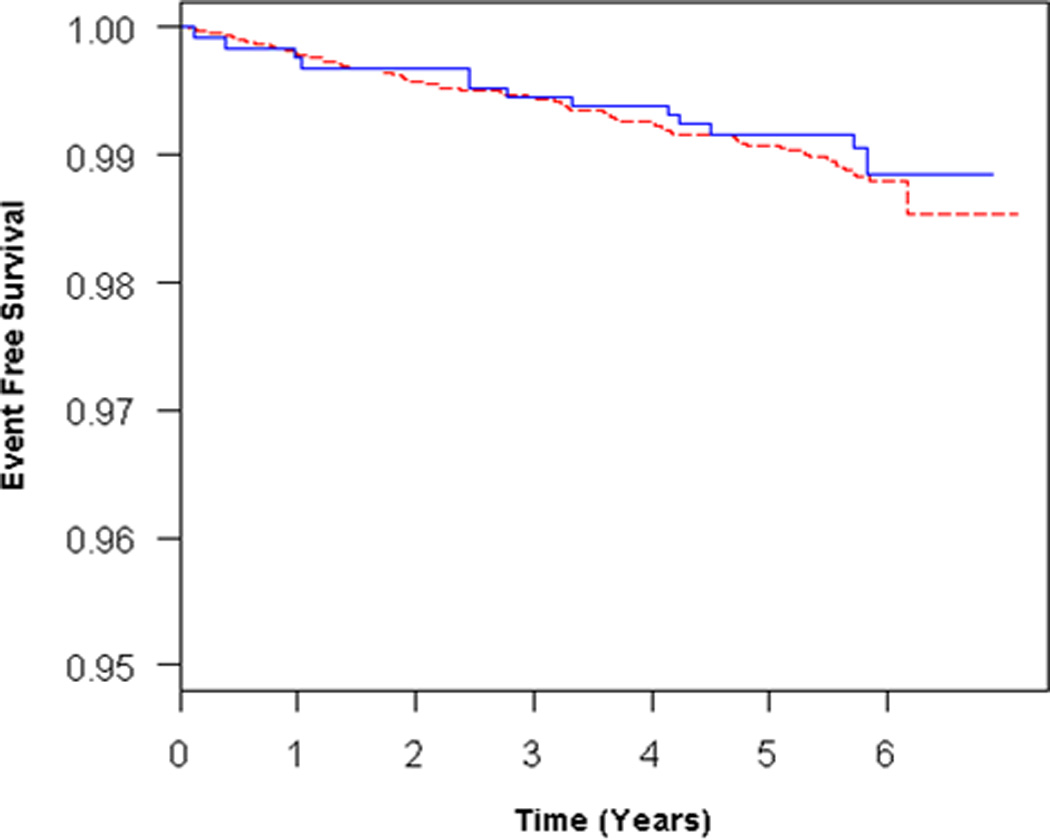
Adjusted survival curves for RV enlargement (RVEDV) vs normal, adjusted for A) age, sex, race/ethnicity, BMI, education, CRP, hypertension, and smoking status and B) all covariates + LVEDV. Solid blue = RV enlargement, dashed red = normal RVEDV.
Figure 4.
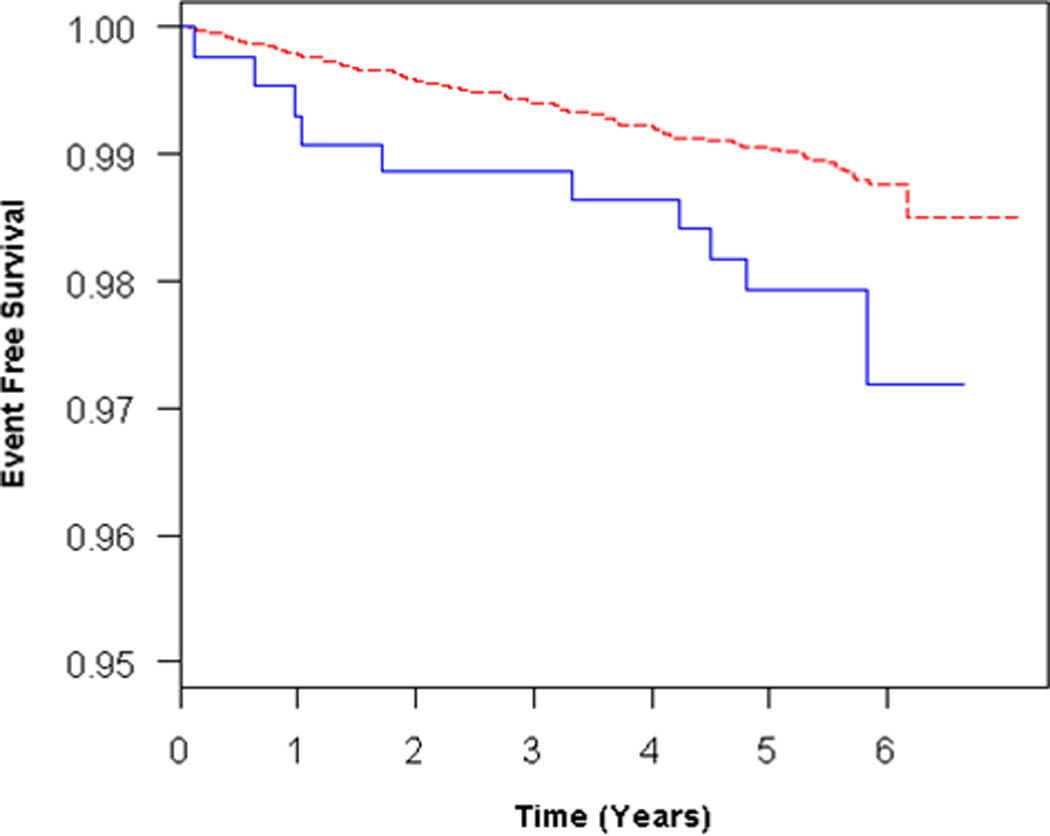
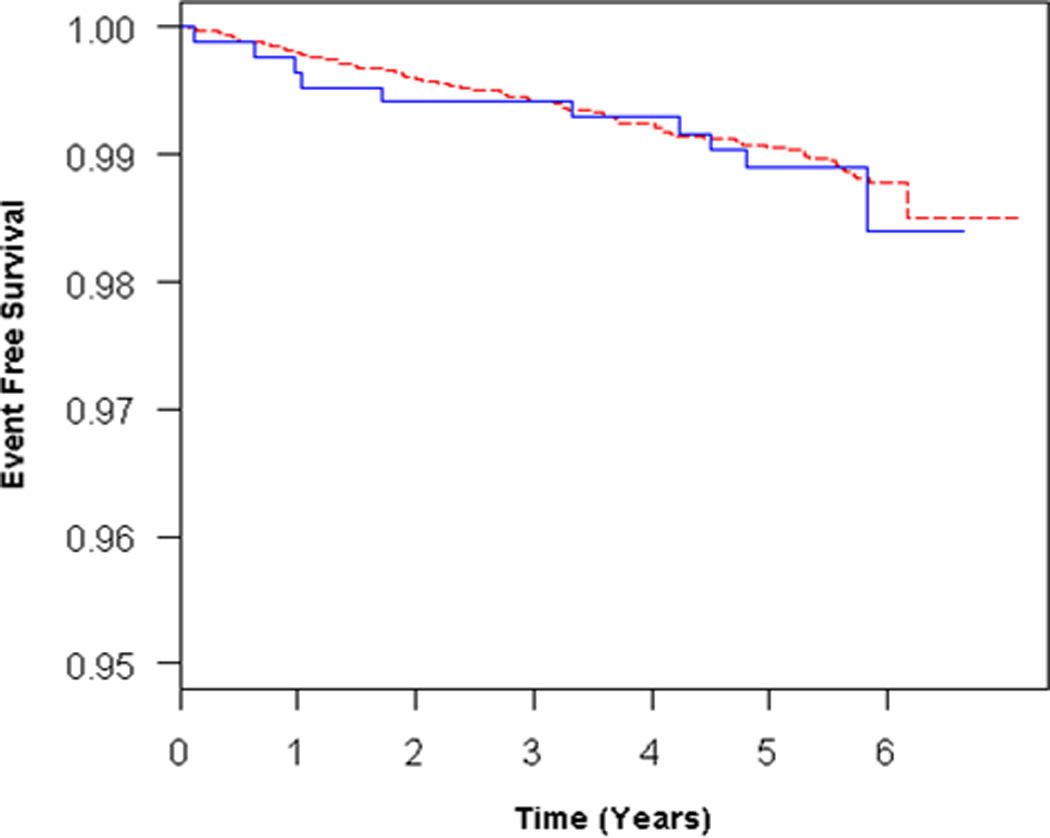
Adjusted survival curves for RV enlargement (RVESV) vs normal, adjusted for A) age, sex, race/ethnicity, BMI, education, CRP, hypertension, and smoking status and B) all covariates + LVESV. Solid blue = RV enlargement, dashed red = normal RVEDV.
The effect estimates after adjustment for pulmonary function are shown in Supplemental Table 5 (65% of the study sample, n = 2704). We reduced the number of covariates in the adjusted models to avoid overfitting as there were only 34 endpoints in this subset. RV hypertrophy was significantly associated with HF and death after adjustment for age, FEV1, FVC, cotinine levels, and percent emphysema (HR = 2.60, 95% CI 1.07–6.32, p = 0.04). This association appeared to persist after accounting for LV mass (HR at mean LV mass = 2.67, 95%CI 0.95 – 7.51, p = 0.06; p for interaction= 0.01). Other RV parameters were not associated with outcomes, although some of these analyses had very few end points in certain subgroups.
DISCUSSION
We have shown that RV morphology in a multi-ethnic population of US adults without clinical cardiovascular disease at baseline has significant associations with the risk of HF and cardiovascular death. Drawn from the only large, population-based study of RV structure and function to our knowledge, these results are novel and run counter to the traditional teaching that the RV does not have physiologic import in healthy adults. Participants with RV hypertrophy had a significant increase in the risk of HF or death, independent of other confounders and LV mass. RV hypertrophy had a stronger association with outcome in the setting of lower LV mass. RV hypertrophy and its causes may account for one out of ten cases of incident HF or death in individuals without clinical cardiovascular disease at baseline. RV enlargement (abnormal RVESV and RVEDV) was possibly associated with a higher risk of events independent of confounders, however these associations were not present after consideration of the respective LV parameters.
RV morphology plays an integral role in certain diseases of the lungs and heart. Several studies of pulmonary arterial hypertension (PAH) have shown that lower cardiac output or a history of RV failure are associated with an increased risk of death.3, 22–24 Studies using cardiac imaging have similarly shown significant associations between RV structure and function and survival in PAH and CHF.4–6, 25, 26 While pulmonary hypertension and increased RV afterload are the major cause of the changes in RV structure and function in PAH and CHF, it is the adaptive ability of the RV which determines survival independent of the severity of the pulmonary vascular disease. Therefore, while increased RV afterload is also the most likely cause of the RV morphologic changes in our study, the RV findings may provide important insights into the mechanisms and risk of adverse outcomes in the long-term.
There are several explanations for why RV hypertrophy may predict an increased risk of clinically-diagnosed HF, which is usually attributable to LV dysfunction. Increased LV end-diastolic pressure from LV systolic or diastolic dysfunction causes elevated left atrial pressure and pulmonary hypertension, increasing RV pressure and wall stress. RV hypertrophy could therefore better reflect increased LV end-diastolic pressure over time (e.g., during activity, stress, and sleep) than does a single quantitative MRI measure of the LV (LV mass or LVEF). In this way, RV hypertrophy could indicate a higher risk of developing clinical HF by serving as a sensitive “barometer” of LV function. Of course, increased RV afterload may also be seen in conditions such as sleep-disordered breathing and/or subclinical pulmonary vascular disease, which could cause both RV hypertrophy and an increased risk of HF and death.
Alternatively, RV hypertrophy could directly contribute to the onset of clinical HF. Dyspnea or lower extremity edema were required for a diagnosis of HF in this study. Impaired RV filling could lead to dyspnea and increased RV (and subsequently right atrial) pressure causing edema. Therefore, RV hypertrophy (with or without concomitant LV disease) could increase the incidence of clinically-diagnosed HF.
Finally, underlying pathophysiologic processes (such as hypoxemia or inflammation) could impact on both the RV and LV. The independent association of RV hypertrophy with events however suggests an RV-specific (or disproportionate) relationship with the risk of long-term outcome. This association was stronger at lower levels of LV mass; patients with LV hypertrophy may have an increased risk of events directly attributable to the LV pathology so that the effect of RV hypertrophy may be more dramatic in its absence. Regardless of the mechanism of this finding, the strength and persistence of the association in multiple analyses implies a potentially significant public health impact.
Enlargement of RVESV appeared to be associated with a higher risk of HF or death, a finding which was non-significant after adjustment for LVESV. Others have shown that greater RVESV is associated with an increased risk of mortality in CHF.27 RV enlargement at end-systole could impact on LV filling or function via ventricular interaction or less likely serve as a nidus for arrhythmias. Reduced RV systolic function was not associated with HF or death in unadjusted or adjusted models. Valvular regurgitation or “pseudo-normalization” of RVEF by RV diastolic dysfunction could explain these results.
There are several limitations to our study. Not all MESA participants were able to tolerate MRI, and not all scans were interpretable. The slightly lower reading success rate compared to the LV was likely attributable to the technical demands of interpreting the very thin RV free wall. However, this is the largest study of RV morphology ever performed with a heterogeneous study sample with > 20,000 person-years of follow-up, making the conclusions particularly generalizable to a multi-ethnic adult population. Pulmonary hemodynamic measurements are an important determinant of RV morphology, but were not feasible in 4000 community-based participants. Our definitions of abnormal RV parameters were based on fast gradient echo MRI sequences, which are less precise than currently used acquisition methods which could affect generalizability. However, any measurement error (as long as non-differential in terms of outcome) would bias to the null, so that RV hypertrophy may be even more strongly related to HF or death than we have shown.
In conclusion, RV hypertrophy is associated with the risk of clinical HF or cardiovascular death in a multi-ethnic adult US population without clinical cardiovascular disease at baseline. The strong association of increased RV mass in unselected community-based participants warrants further studies of the role of increased RV afterload in contributing to the risk of HF or death.
Supplementary Material
The roles of right ventricular (RV) structure and function in health and disease have not been well-studied. While historical animal studies suggested that the RV did not contribute importantly to cardiac function, more recent studies of patients with diseases such as pulmonary arterial hypertension and congestive heart failure have shown that RV size and ejection fraction are independent predictors of survival. Whether the RV has important an important role in community-based adults is unknown. This study showed that the presence of RV hypertrophy (measured by magnetic resonance imaging) in adults without clinical cardiovascular disease at baseline was associated with an increased risk of heart failure or cardiovascular death. This association held after accounting for left ventricular (LV) mass, however was stronger in participants with lower LV mass. There are several explanations for this finding. Patients with LV hypertrophy may have an increased risk of events directly attributable to the LV pathology, explaining why the effect of RV hypertrophy is stronger in its absence. RV hypertrophy could indicate a higher risk of heart failure by serving as a sensitive “barometer” of LV function. Increased RV afterload may also be seen in conditions such as sleep-disordered breathing and/or subclinical pulmonary vascular disease, which could cause both RV hypertrophy and an increased risk of events. Alternatively, RV hypertrophy could directly contribute to the onset of heart failure. Clinical cardiovascular-disease free adults with RV hypertrophy are at higher overall risk of heart failure and death, warranting further studies of mechanisms and potential treatment interventions.
ACKNOWLEDGEMENTS
All authors participated in the design of the study and data acquisition and analysis. All authors provided critical revision of the manuscript and take responsibility for the entire content. SMK had full access to all of the data in the study and takes responsibility for the integrity of the data and the accuracy of the data analysis. There are no conflicts of interest. This manuscript has been reviewed by the MESA Investigators for scientific content and consistency of data interpretation with previous MESA publications and significant comments have been incorporated prior to submission for publication. The authors thank the other investigators, staff, and participants of the MESA and MESA-Lung Studies for their valuable contributions. A full list of participating MESA Investigators and institutions can be found at http://www.mesa-nhlbi.org.
Funding Sources: This study was funded by National Institutes of Health R01-HL086719, R01-HL077612, K24- HL103844, N01-HC95159 through N01-HC95169
Footnotes
Publisher's Disclaimer: This is a PDF file of an unedited manuscript that has been accepted for publication. As a service to our customers we are providing this early version of the manuscript. The manuscript will undergo copyediting, typesetting, and review of the resulting proof before it is published in its final citable form. Please note that during the production process errors may be discovered which could affect the content, and all legal disclaimers that apply to the journal pertain.
Disclosures: None
REFERENCES
- 1.Haddad F, Doyle R, Murphy DJ, Hunt SA. Right ventricular function in cardiovascular disease, part II: pathophysiology, clinical importance, and management of right ventricular failure. Circulation. 2008;117:1717–1731. doi: 10.1161/CIRCULATIONAHA.107.653584. [DOI] [PubMed] [Google Scholar]
- 2.Haddad F, Hunt SA, Rosenthal DN, Murphy DJ. Right ventricular function in cardiovascular disease, part I: Anatomy, physiology, aging, and functional assessment of the right ventricle. Circulation. 2008;117:1436–1448. doi: 10.1161/CIRCULATIONAHA.107.653576. [DOI] [PubMed] [Google Scholar]
- 3.Kawut SM, Horn EM, Berekashvili KK, Garofano RP, Goldsmith RL, Widlitz AC, Rosenzweig EB, Kerstein D, Barst RJ. New predictors of outcome in idiopathic pulmonary arterial hypertension. Am J Cardiol. 2005;95:199–203. doi: 10.1016/j.amjcard.2004.09.006. [DOI] [PubMed] [Google Scholar]
- 4.van Wolferen SA, Marcus JT, Boonstra A, Marques KM, Bronzwaer JG, Spreeuwenberg MD, Postmus PE, Vonk-Noordegraaf A. Prognostic value of right ventricular mass, volume, and function in idiopathic pulmonary arterial hypertension. Eur Heart J. 2007;28:1250–1257. doi: 10.1093/eurheartj/ehl477. [DOI] [PubMed] [Google Scholar]
- 5.Meyer P, Filippatos GS, Ahmed MI, Iskandrian AE, Bittner V, Perry GJ, White M, Aban IB, Mujib M, Dell'Italia LJ, Ahmed A. Effects of right ventricular ejection fraction on outcomes in chronic systolic heart failure. Circulation. 2010;121:252–258. doi: 10.1161/CIRCULATIONAHA.109.887570. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 6.Ghio S, Gavazzi A, Campana C, Inserra C, Klersy C, Sebastiani R, Arbustini E, Recusani F, Tavazzi L. Independent and additive prognostic value of right ventricular systolic function and pulmonary artery pressure in patients with chronic heart failure. J Am Coll Cardiol. 2001;37:183–188. doi: 10.1016/s0735-1097(00)01102-5. [DOI] [PubMed] [Google Scholar]
- 7.France AJ, Prescott RJ, Biernacki W, Muir AL, MacNee W. Does right ventricular function predict survival in patients with chronic obstructive lung disease? Thorax. 1988;43:621–626. doi: 10.1136/thx.43.8.621. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 8.Chahal H, Johnson C, Tandri H, Jain A, Hundley WG, Barr RG, Kawut SM, Lima JA, Bluemke DA. Relation of cardiovascular risk factors to right ventricular structure and function as determined by magnetic resonance imaging (results from the multi-ethnic study of atherosclerosis) Am J Cardiol. 2010;106:110–116. doi: 10.1016/j.amjcard.2010.02.022. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 9.Kawut SM, Lima JA, Barr RG, Chahal H, Jain A, Tandri H, Praestgaard A, Bagiella E, Kizer JR, Johnson WC, Kronmal RA, Bluemke DA. Sex and race differences in right ventricular structure and function: the multi-ethnic study of atherosclerosis-right ventricle study. Circulation. 2011;123:2542–2551. doi: 10.1161/CIRCULATIONAHA.110.985515. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 10.Ventetuolo CE, Ouyang P, Bluemke DA, Tandri H, Barr RG, Bagiella E, Cappola AR, Bristow MR, Johnson C, Kronmal RA, Kizer JR, Lima JA, Kawut SM. Sex hormones are associated with right ventricular structure and function: The MESA-right ventricle study. Am J Respir Crit Care Med. 2011;183:659–667. doi: 10.1164/rccm.201007-1027OC. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 11.Bild DE, Bluemke DA, Burke GL, Detrano R, Diez Roux AV, Folsom AR, Greenland P, Jacob DR, Jr, Kronmal R, Liu K, Nelson JC, O'Leary D, Saad MF, Shea S, Szklo M, Tracy RP. Multi-ethnic study of atherosclerosis: objectives and design. Am J Epidemiol. 2002;156:871–881. doi: 10.1093/aje/kwf113. [DOI] [PubMed] [Google Scholar]
- 12.Natori S, Lai S, Finn JP, Gomes AS, Hundley WG, Jerosch-Herold M, Pearson G, Sinha S, Arai A, Lima JA, Bluemke DA. Cardiovascular function in multi-ethnic study of atherosclerosis: normal values by age sex, and ethnicity. AJR Am J Roentgenol. 2006;186:S357–S365. doi: 10.2214/AJR.04.1868. [DOI] [PubMed] [Google Scholar]
- 13.Tandri H, Daya SK, Nasir K, Bomma C, Lima JA, Calkins H, Bluemke DA. Normal reference values for the adult right ventricle by magnetic resonance imaging. Am J Cardiol. 2006;98:1660–1664. doi: 10.1016/j.amjcard.2006.07.049. [DOI] [PubMed] [Google Scholar]
- 14.Bluemke DA, Kronmal RA, Lima JA, Liu K, Olson J, Burke GL, Folsom AR. The relationship of left ventricular mass and geometry to incident cardiovascular events: the MESA (Multi-Ethnic Study of Atherosclerosis) study. J Am Coll Cardiol. 2008;52:2148–2155. doi: 10.1016/j.jacc.2008.09.014. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 15.Vogel-Claussen J, Finn JP, Gomes AS, Hundley GW, Jerosch-Herold M, Pearson G, Sinha S, Lima JA, Bluemke DA. Left ventricular papillary muscle mass: relationship to left ventricular mass and volumes by magnetic resonance imaging. J Comput Assist Tomogr. 2006;30:426–432. doi: 10.1097/00004728-200605000-00013. [DOI] [PubMed] [Google Scholar]
- 16.Winter MM, Bernink FJ, Groenink M, Bouma BJ, van Dijk AP, Helbing WA, Tijssen JG, Mulder BJ. Evaluating the systemic right ventricle by CMR: the importance of consistent and reproducible delineation of the cavity. J Cardiovasc Magn Reson. 2008;10:40. doi: 10.1186/1532-429X-10-40. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 17.MESA Manual of Operations: Field center and laboratory procedures. [Accessed December 23, 2009];University of Washington. Available at: http://www.mesa-nhlbi.org/manuals.aspx.
- 18.Hosmer DW, Lemeshow S. Applied survival analysis : regression modeling of time to event data. New York: Wiley; 1999. [Google Scholar]
- 19.Spiegelman D, Hertzmark E, Wand HC. Point and interval estimates of partial population attributable risks in cohort studies: examples and software. Cancer Causes Control. 2007;18:571–579. doi: 10.1007/s10552-006-0090-y. [DOI] [PubMed] [Google Scholar]
- 20.Barr RG, Bluemke DA, Ahmed FS, Carr JJ, Enright PL, Hoffman EA, Jiang R, Kawut SM, Kronmal RA, Lima JA, Shahar E, Smith LJ, Watson KE. Percent emphysema, airflow obstruction, and impaired left ventricular filling. N Engl J Med. 2010;362:217–227. doi: 10.1056/NEJMoa0808836. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 21.Hankinson JL, Kawut SM, Shahar E, Smith LJ, Stukovsky KH, Barr RG. Performance of American Thoracic Society-recommended spirometry reference values in a multiethnic sample of adults: the multi-ethnic study of atherosclerosis (MESA) lung study. Chest. 2010;137:138–145. doi: 10.1378/chest.09-0919. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 22.Thenappan T, Shah SJ, Rich S, Tian L, Archer SL, Gomberg-Maitland M. Survival in pulmonary arterial hypertension: a reappraisal of the NIH risk stratification equation. Eur Respir J. 2010;35:1079–1087. doi: 10.1183/09031936.00072709. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 23.McLaughlin VV, Shillington A, Rich S. Survival in primary pulmonary hypertension: the impact of epoprostenol therapy. Circulation. 2002;106:1477–1482. doi: 10.1161/01.cir.0000029100.82385.58. [DOI] [PubMed] [Google Scholar]
- 24.Sitbon O, Humbert M, Nunes H, Parent F, Garcia G, Herve P, Rainisio M, Simonneau G. Long-term intravenous epoprostenol infusion in primary pulmonary hypertension: prognostic factors and survival. J Am Coll Cardiol. 2002;40:780–788. doi: 10.1016/s0735-1097(02)02012-0. [DOI] [PubMed] [Google Scholar]
- 25.Forfia PR, Fisher MR, Mathai SC, Housten-Harris T, Hemnes AR, Borlaug BA, Chamera E, Corretti MC, Champion HC, Abraham TP, Girgis RE, Hassoun PM. Tricuspid annular displacement predicts survival in pulmonary hypertension. Am J Respir Crit Care Med. 2006;174:1034–1041. doi: 10.1164/rccm.200604-547OC. [DOI] [PubMed] [Google Scholar]
- 26.Ghio S, Klersy C, Magrini G, D'Armini AM, Scelsi L, Raineri C, Pasotti M, Serio A, Campana C, Vigano M. Prognostic relevance of the echocardiographic assessment of right ventricular function in patients with idiopathic pulmonary arterial hypertension. Int J Cardiol. 2010;140:272–278. doi: 10.1016/j.ijcard.2008.11.051. [DOI] [PubMed] [Google Scholar]
- 27.Bourantas CV, Loh HP, Bragadeesh T, Rigby AS, Lukaschuk EI, Garg S, Tweddel AC, Alamgir FM, Nikitin NP, Clark AL, Cleland JG. Relationship between right ventricular volumes measured by cardiac magnetic resonance imaging and prognosis in patients with chronic heart failure. Eur J Heart Fail. 2011;13:52–60. doi: 10.1093/eurjhf/hfq161. [DOI] [PubMed] [Google Scholar]
Associated Data
This section collects any data citations, data availability statements, or supplementary materials included in this article.