Abstract
Objectives
The study aimed to determine whether exposure to a volcanic eruption was associated with increased prevalence of physical and/or mental symptoms.
Design
Cohort, with non-exposed control group.
Setting
Natural disasters like volcanic eruptions constitute a major public-health threat. The Icelandic volcano Eyjafjallajökull exposed residents in southern Iceland to continuous ash fall for more than 5 weeks in spring 2010. This study was conducted during November 2010–March 2011, 6–9 months after the Eyjafjallajökull eruption.
Participants
Adult (18–80 years of age) eruption-exposed South Icelanders (N=1148) and a control population of residents of Skagafjörður, North Iceland (N=510). The participation rate was 72%.
Main outcome measures
Physical symptoms in the previous year (chronic), in the previous month (recent), General Health Questionnaire (GHQ-12) measured psychological morbidity.
Results
The likelihood of having symptoms during the last month was higher in the exposed population, such as; tightness in the chest (OR 2.5; 95% CI 1.1 to 5.8), cough (OR 2.6; 95% CI 1.7 to 3.9), phlegm (OR 2.1; 95% CI 1.3 to 3.2), eye irritation (OR 2.9; 95% CI 2.0 to 4.1) and psychological morbidity symptoms (OR 1.3; 95% CI 1.0 to 1.7). Respiratory symptoms during the last 12 months were also more common in the exposed population; cough (OR 2.2; 95% CI 1.6 to 2.9), dyspnoea (OR 1.6; 95% CI 1.1 to 2.3), although the prevalence of underlying asthma and heart disease was similar. Twice as many in the exposed population had two or more symptoms from nose, eyes or upper-respiratory tract (24% vs 13%, p<0.001); these individuals were also more likely to experience psychological morbidity (OR 4.7; 95% CI 3.4 to 6.5) compared with individuals with no symptoms. Most symptoms exhibited a dose–response pattern within the exposed population, corresponding to low, medium and high exposure to the eruption.
Conclusions
6–9 months after the Eyjafjallajökull eruption, residents living in the exposed area, particularly those closest to the volcano, had markedly increased prevalence of various physical symptoms. A portion of the exposed population reported multiple symptoms and may be at risk for long-term physical and psychological morbidity. Studies of long-term consequences are therefore warranted.
Keywords: Epidemiology, Public Health
Article summary.
Article focus
There is scarcity of data on potential health effects of volcanic eruptions.
The Icelandic population registers enabled us to perform a large population-based cohort study of self-reported symptoms of all residents in affected areas of the Eyjafjallajökull eruption during spring 2010. Residents in northern Iceland who had minimal exposure to ash from the eruption were recruited as non-exposed population.
We hypothesised that physical and mental symptoms would reflect the individual's exposure to the volcanic eruption, measured by the proximity of residency area to the eruption.
Key messages
Residents exposed to the eruption had a higher prevalence of respiratory and mental symptoms after adjusting for age, gender, education and smoking status.
Within the exposed region there was a dose–response pattern, the highest symptom prevalence was found in those living closest to the volcano, indicating that the symptoms found in the study may be attributed to exposure to the eruption.
There was a strong association between reporting more than one physical symptom and poor mental health. This finding indicates a clustering of symptoms in sensitised or fragile individuals.
Strengths and limitations of this study
An important strength of the study is that it includes the total population of the exposed region and a matched sample from a non-exposed region. The response rate was 72%.
A limiting factor is that the study relies on self-reported symptoms and that the degree of exposure is based solely on proximity of residency to the eruption.
Permission has been granted to follow this cohort in health registries through personal identification numbers, enabling us to study potential long-term health effects of the eruption.
Introduction
Throughout history, human societies have been exposed to natural disasters like volcanic eruptions and earthquakes. In 2010, 300 000 individuals were killed worldwide in natural disasters.1Accurate information on mortality and long-term health consequences of natural disasters is instrumental to strengthen risk management and decrease their negative health impact.2
The eruption of the Eyjafjallajökull volcano in Iceland, which lasted from 14 April to 20 May 2010, made headlines worldwide, not least because of extensive effects on international flight traffic. Direct ash fall from the eruption was estimated at around 250 million tons, the rural regions in Iceland south and south-east of the volcano were most severely affected.3 4 Ash fall was continuous for about 6 weeks, and following the eruption the ash was frequently resuspended in the area.4 5 The surface of the fresh ash particles contained reactive salts and as much as 20% of the particles (by mass) were less than 10 μm in aerodynamic diameter and could enter the lower respiratory tract.6 A study of local residents (N=207) was conducted immediately after the eruption ended. Participants were examined by a physician and to ascertain respiratory health, standardised spirometry was performed before and after bronchodilator usage. Adult participants also answered questionnaires about mental and physical health. Ash exposure was associated with high prevalence of eye and upper-airway irritation (25% and 50%, respectively), and exacerbation of pre-existing asthma but did not contribute to serious health problems or impair respiratory function compared with controls. In total, 39% showed symptoms of psychological morbidity as measured by the General Health Questionnaire (GHQ).7 In another study residents of the region expressed a need for more detailed information on the potential health effects of the ash fall.8 Meanwhile, the impact on long-term health of the residents remains to be explored.
Previous studies on volcanic ash exposure and health have shown increased respiratory morbidity and asthma attacks,9 10 and increased hospital visits for respiratory illness in association with some eruptions11 but not in others.12 Also, increased irritation of the respiratory tract from short-term exposure to volcanic gases and ash.13 Long-term exposure to sulphuric gases (often emitted in volcanic eruptions) were found to be associated with increased prevalence of chronic bronchitis and cardiorespiratory symptoms in some studies.14 15 In addition to direct physical health hazards, experiencing floods, lahars, as well as being exposed to prolonged ash suspension can be a threat to mental health. Stress levels may increase dramatically and have been shown to contribute to psychological morbidity such as post-traumatic stress syndrome or depression.16 17
Health effects of long-term exposure to a volcanic eruption are important both from a scientific and healthcare standpoint.18 Iceland's population-based registries and strong infrastructure present an important opportunity to study such health impacts, particularly in terms of long-term follow-up.
Utilising the Icelandic population-based registers to identify all residents living in the vicinity of Eyjafjallajökull, the aim of this study was to investigate their self-reported physical and mental health 6–9 months after the volcanic eruption. We hypothesised that residents of the Eyjafjallajökull area, particularly those most exposed, would be at increased risk of physical and psychological symptoms compared to a non-exposed population in North Iceland.
Methods
Study area
The exposed area in South Iceland is mostly farmland with a few villages. It has several active volcanoes19 which along with sand plains and river beds are a source of dust storms.20 21 Apart from traffic on the national highway and agricultural activities, there are no major sources of anthropogenic particles in the area.
Almost from the onset of the eruption, the Environment Agency of Iceland monitored concentrations of inhalable particulate matter (PM10) in up to three locations in the study area. The official health limit for PM10, 50 µg/m3 daily averages, was surpassed more than half of the days between 7 May and 6 June 2010, when air quality was continuously monitored in the most severely affected areas.4 Monitoring continued after the eruption ended and until the end of our study period (end of March 2011), ash was repeatedly resuspended and the mean 24 h concentration of PM10 particles was 41 µg/m3. The official health limit of 50 µg/m3 daily average was exceeded 25 times, mostly during summer and fall of 2010. From November 2010 onwards the number of exceedences declined rapidly.5
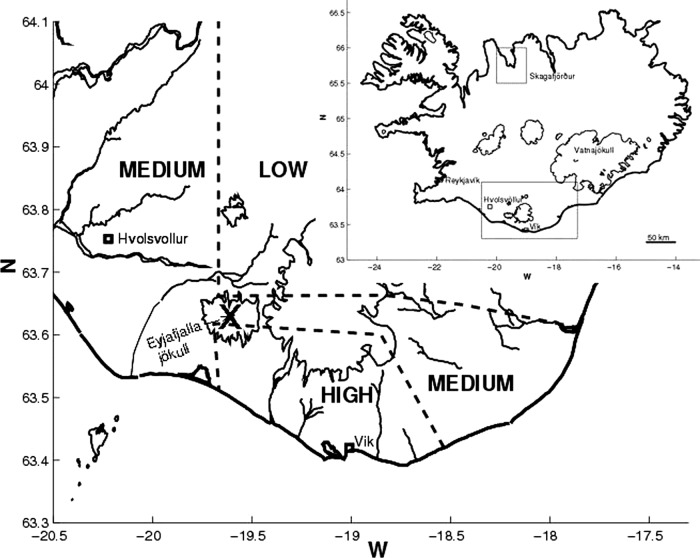
In addition to a non-exposed control area in North Iceland, the study area was divided into low, medium and high-exposure regions in South Iceland (figure 1) based on satellite images of the eruption plume (coarse time resolution), information about the emission intensity3 and observations of ash deposits on the ground.4 22 Models calculated with FLEXPART show similar ash deposits, ranging from approximately 1000 g/m2 in the region just south of the volcano, down to about 200 g/m2 near Vík some 50 km further east.4
Figure 1.
Map of Iceland and the study areas. Inserted map of Iceland shows the location of Skagafjörður (non-exposed, control area) in the north and of the exposed area in South Iceland. The larger map of the exposed area shows Eyjafjallajökull (marked with X) and the low, medium and high ash exposure areas.
During the eruption, the prevailing wind was from the north-west, causing the heaviest ash fall south and east of the volcano. While ash deposition was relatively low in the western part of the medium-exposure region, the volcano was in full view there and thus these residents were more visually exposed to the volcano than in other regions. The lowland regions south and west of the glacier are prone to flooding and many residents were evacuated because of glacial outburst floods in the first days of the eruption.
Study population
The study population consisted of all residents in the municipalities closest to Eyjafjallajökull volcano (predefined by postal codes), identified in the population-based registry (Statistics Iceland). Most live in farmlands (N=1207) and the rest in small townships (N=859).23 By these means we identified 1615 inhabitants who were 18–80 years of age, resided in the exposed area during the eruption, could be reached and spoke Icelandic fluently. In addition, a sample of 697 demographically matched (age, gender and urban/rural habitation) residents from a non-exposed area in Northern Iceland was included as control group. Sheep and dairy farming are predominant in both areas.
Data collection
Initially, all participants in the exposed group received a letter including information about the study and an invitation to participate. Some days after the letters were sent the recipients were contacted by telephone and asked whether they were willing to take part, and if so, whether they preferred to reply on paper or online. Subsequently, questionnaires or email invitations were sent and a week later a combined thank-you/reminder card was sent by post or email. If needed, the participants were reminded again by phone. A similar protocol was used for the control group, with the exception that the introductory letter stated that a questionnaire would be sent a few days later, unless participation was declined.
Questionnaires were sent to the exposed population between 19 November and 28 December 2010 (6–7 months after the eruption ended) and the last replies were received in March 2011. The control group received questionnaires between 26 January and 4 February 2011, the last replies were received in April 2011.
The questionnaires contained no information that revealed the identity of the respondent, instead, they had a running number which could be linked to the person's ID number through a list which was kept separately and securely to enable later follow-up. A few of the returned questionnaires lacked most of the required information and were excluded from the analysis (n=13).
Questionnaires
The questionnaires contained questions concerning demographic background and current well-being, including various physical and psychological symptoms. We used standard questions from the screening part of the European Community Respiratory Health questionnaire24 and assessed underlying disease by asking “Has a medical doctor ever told you that you had the following diseases: asthma, heart disease, emphysema, chronic bronchitis or chronic obstructive pulmonary disease (COPD)” with the response alternatives ‘Yes’ or ‘No’ by each option. To assess recent symptoms, we asked “Have the following symptoms disrupted your daily activities during the previous month” followed by a list of various symptoms from, for example, the respiratory system, skin or eyes, or relating to pain. We also asked about smoking ‘Have you ever smoked’, ‘Yes’ or ‘No’, and ‘Have you smoked during the last month’, ‘Yes’ or ‘No’. Questions on regular use of medication were ‘Do you take medication regularly, that is, once per week or more often’ followed by listing asthma medication, analgesics, blood-pressure-lowering medication and sleep medication/antidepressants/tranquillizers/medication for other mental health problems. Current psychological morbidity was evaluated from the General Health Questionnaire-12-item version (GHQ-12),25 26 a non-specific screening tool for psychological morbidity which measures anxiety, loss of self-confidence and social dysfunction.27 We used a binary cut-off score of >2.
Database and coding
The online survey was built with LimeSurvey.28 Participants replying online accessed the survey using a unique identifier sent to them by email. Questionnaire replies on paper were entered into LimeSurvey according to uniform guidelines set by the researchers.
Statistical analysis
We first calculated descriptive statistics, contrasting background characteristics in the exposed and non-exposed population using χ2 tests (p applied to all categories within demographic characteristics). Logistic regression was used to determine ORs associated with residence in (1) the exposed and non-exposed regions and (2) the low, medium, or high-exposure areas within the exposed region. All models were adjusted for a priori selected variables: age, gender, smoking status (never, former and current) and education level, ORs and 95% CI were calculated from the outputs. A CI not including 1.0 or a p value of 0.05 or less was considered statistically significant. A Venn diagram was drawn to show the interrelationship between key nasal, eye or upper respiratory symptoms (cough and/or phlegm without having a cold, eye irritation or itch, and sneeze, stuffed, or runny nose). Demographic characteristics, risk factors and comorbidities of those reporting multiple symptoms were explored using χ2 tests and logistic regression.
IBM SPSS 1929 was used for data analysis. Individuals who had not replied to all relevant questions were excluded from the regression models.
The study was approved by The Icelandic Data Protection Authority (no. S4878/2010) and The Science Bioethics Committee (no. VSNb2010080002/03.7), all participants gave informed consent.
Results
Valid questionnaires were obtained from 1148 of 1615 from the exposed population (71%) and 510 of 697 (73%) from the non-exposed population. A higher proportion of the exposed population could not be reached or found (10.8% vs 7.2%; p=0.005), and more refused to participate (17.8% vs 14.6%; p=0.069). The exposed and non-exposed participants were similar with respect to demographic characteristics; age, education levels and occupational, marital and financial status (table 1).
Table 1.
Participation and demographic characteristics of the exposed (South Iceland) and non-exposed (North Iceland) populations
| Exposed | Non-exposed | ||||
|---|---|---|---|---|---|
| % (n/N) |
% (n/N) | p Value | |||
| Target population (identified in total population registers) | 1811 | 751 | |||
| Could not be found or reached | 10.8 (196/1811) | 7.2 (54/751) | 0.01 | ||
| Study population | 1615 | 697 | |||
| Refused to participate | 17.8 (286/1615) | 14.6 (102/697) | 0.07 | ||
| Originally agreed to participate | 1329 | 595 | |||
| Explained non-participation* | 7.0 (93/1329) | 6.1 (36/595) | 0.44 | ||
| Unexplained non-participation† | 6.6 (88/1329) | 8.2 (49/595) | 0.20 | ||
| Response rate (participants/study population) | 71 (1148/1615) | 73 (510/697) | 0.31 | ||
| Demographic characteristics | |||||
| Male | 49.0 (562) | 51.4 (262) | 0.36 | ||
| Female | 51.0 (586) | 48.6 (248) | 0.36 | ||
| Age categories | |||||
| 18–23 | 11.1 (128/1148) | 8.2 (42/510) | 0.07 | ||
| 24–30 | 8.6 (99/1148) | 9.0 (46/510) | 0.79 | ||
| 31–40 | 15.2 (175/1148) | 14.3 (73/510) | 0.55 | ||
| 41–50 | 20.3 (233/1148) | 21.4 (109/510) | 0.62 | ||
| 51–60 | 19.3 (222/1148) | 22.2 (113/510) | 0.19 | ||
| 61–70 | 15.9 (183/1148) | 16.5 (84/510) | 0.79 | ||
| 71–80 | 9.4 (108/1148) | 8.4 (43/510) | 0.52 | ||
| Education | |||||
| No formal education | 5.4 (61/1134) | 4.8 (24/501) | 0.62 | ||
| Primary education | 35.9 (407/1134) | 30.9 (155/501) | 0.05 | ||
| Secondary education | 33.4 (379/1134) | 37.7 (189/501) | 0.09 | ||
| Professional or university education | 20.6 (234/1134) | 23.8 (119/501) | 0.16 | ||
| Other education* | 4.7 (53/1134) | 2.8 (14/501) | 0.08 | ||
| Marital status | |||||
| Married or cohabitating | 72.4 (831/1148) | 76.6 (391/510) | 0.07 | ||
| Single or divorced | 18.3 (210/1148) | 15.5 (79/510) | 0.17 | ||
| Relationship—no cohabitation | 6.8 (78/1148) | 4.7 (24/510) | 0.10 | ||
| Widow or widower | 2.5 (29/1148) | 3.1 (16/510) | 0.48 | ||
| Household size | |||||
| 1 adult | 13.8 (151/1096) | 15.4 (76/494) | 0.40 | ||
| 2 adults | 51.4 (563/1096) | 56.1 (277/494) | 0.40 | ||
| 3 adults | 21.3 (233/1096) | 18.0 (89/494) | 0.14 | ||
| ≥4 adults | 13.6 (149/1096) | 10.5 (52/494) | 0.09 | ||
| Occupational status | |||||
| Full-time job | 60.4 (683/1130) | 61.0 (310/507) | 0.79 | ||
| Part-time job | 9.1 (103/1130) | 11.6 (59/507) | 0.11 | ||
| Unemployed | 3.5 (40/1130) | 1.2 (6/507) | 0.01 | ||
| Student | 6.9 (78/1130) | 5.7 (28/507) | 0.29 | ||
| Homemaker or maternity leave | 9.4 (99/1130) | 7.8 (40/507) | 0.56 | ||
| Retired | 6.1 (69/1130) | 6.3 (32/507) | 0.87 | ||
| On disability or sick leave | 5.1 (58/1130) | 6.3 (32/507) | 0.33 | ||
| Financial situation | |||||
| Very good | 4.6 (52/1136) | 4.3 (22/510) | 0.81 | ||
| Good | 23.9 (271/1136) | 26.3 (134/510) | 0.92 | ||
| Acceptable (‘making ends meet’) | 55.6 (632/1136) | 56.1 (286/510) | 0.87 | ||
| Bad | 13.5 (153/1136) | 12.0 (61/510) | 0.40 | ||
| Very bad (‘indebted or bankruptcy’) | 2.5 (28/1136) | 1.4 (7/510) | 0.16 | ||
|
Exposure areas‡ |
|||||
| Smoking status | Low | Medium | High | Non-exposed area | |
| Never smoker | 57.2 (87/152) | 58.5 (377/644) | 54.0 (190/352) | 54.3 (277/510) | 0.31 |
| Former smoker | 28.9 (44/152) | 24.5 (158/644) | 26.1 (92/352) | 26.3 (134/510) | 0.69 |
| Current smoker | 13.8 (21/152) | 16.9 (109/644) | 19.9 (70/352) | 19.4 (99/510) | 0.33 |
*Dropped out because of the nature of the questions, because they did not think the study applied to them, or because of illness or old age.
†Did not reply, could not be reached for reminders, did not respond to reminders or returned empty questionnaires.
‡The exposed area was divided into three areas by levels of exposure with regard to magnitude of ash fall, see figure 1. The p value is based on comparison between the non-exposed and the sum of the exposed area.
Analysis 1: exposed versus non-exposed
Respiratory symptoms such as waking up with a feeling of tightness in the chest, breathlessness, cough and phlegm in the last 12 months were more prevalent in the exposed population. After adjusting for gender, age, education and smoking status, the exposed population was more likely to report symptoms like tightness in chest (OR 2.0; 95% CI 1.3 to 3.0), coughing without a cold (OR 2.2; 95% CI 1.6 to 2.9) and having chronic bronchitis (OR 1.9; 95% CI 1.1 to 3.1; table 2). In addition, bothersome physical symptoms during the last month were more common in the exposed population; these were shortness of breath (OR 2.1; 95% CI 1.2 to 3.6), cough (OR 2.6; 95% 1.7 to 3.9), phlegm (OR 2.1; 95% CI 1.3 to 3.2) and eye irritation (OR 2.9; 95% CI 2.0 to 4.1). Back pain, myalgia and insomnia were less prevalent in the exposed population. Psychological morbidity was marginally more common in the exposed population (OR 1.3; 95% CI 1.0 to 1.7), as was the use of blood-pressure-lowering medication (OR 1.3; 95% CI 1.0 to 1.7), while the use of analgesics was less common (OR 0.7; 95% CI 0.5 to 1.0; table 3).
Table 2.
Risk of respiratory symptoms (ECHRS) in a population exposed to the Eyjafjallajökull volcanic eruption compared to a non-exposed population
| Non-exposed |
Exposed |
|||
|---|---|---|---|---|
| % (n/N) | % (n/N) | OR (95% CI)* | p Value | |
| Wheezing (last 12 months) | 10.2 (51/498) | 15.9 (177/1110) | 1.8 (1.3 to 2.5) | 0.001 |
| If yes, breathlessness at the same time | 56.3 (27/48) | 57.0 (94/165) | 1.2 (0.6 to 2.4) | 0.56 |
| If yes, do you wheeze without a cold | 66.0 (31/47) | 70.7 (118/167) | 1.2 (0.6 to 2.5) | 0.59 |
| Nocturnal chest tightness (last 12 months) | 6.6 (33/500) | 12.1 (135/1115) | 2.0 (1.3 to 3.0) | 0.003 |
| Breathlessness at rest | 5.4 (27/500) | 7.7 (85/1103) | 1.4 (0.9 to 2.3) | 0.13 |
| Coughing without a cold | 15.9 (80/502) | 28.2 (314/1114) | 2.2 (1.6 to 2.9) | <0.001 |
| Nocturnal cough (last 12 months) | 18.8 (95/504) | 23.2 (258/1110) | 1.3 (1.0 to 1.7) | 0.06 |
| Morning winter cough | 11.6 (60/504) | 12.0 (133/1111) | 1.0 (0.7 to 1.4) | 0.99 |
| Nocturnal or daytime winter cough | 9.2 (46/498) | 11.0 (121/1105) | 1.3 (0.9 to 1.8) | 0.23 |
| If yes, is it chronic† | 75.0 (30/40) | 67.2 (78/116) | 0.5 (0.2 to 1.4) | 0.19 |
| Morning winter phlegm | 10.2 (51/500) | 14.4 (159/1104) | 1.5 (1.1 to 2.1) | 0.02 |
| Nocturnal or daytime winter phlegm | 5.8 (29/497) | 8.1 (89/1097) | 1.5 (1.0 to 2.4) | 0.08 |
| If yes, is it chronic† | 96.4 (27/28) | 86.9 (73/84) | 0.3 (0.0 to 2.4) | 0.25 |
| Dyspnoea | 7.8 (39/498) | 11.8 (131/1106) | 1.6 (1.1 to 2.3) | 0.02 |
| Nasal allergy and hay fever | 19.1 (96/502) | 19.1 (213/1116) | 1.1 (0.8 to 1.4) | 0.73 |
| Allergic rhinitis | 23.0 (115/501) | 29.5 (327/1109) | 1.4 (1.1 to 1.8) | 0.007 |
| Physician diagnosed conditions‡ | ||||
| Asthma | 14.3 (71/498) | 11.9 (132/1111) | 0.8 (0.6 to 1.1) | 0.17 |
| Asthma diagnosis was confirmed by an MD | 85.5 (59/69) | 95.9 (117/122) | 3.9 (1.2 to 12.5) | 0.03 |
| Heart disease | 6.2 (31/503) | 8.0 (89/1115) | 1.4 (0.9 to 2.2) | 0.15 |
| Chronic bronchitis | 4.2 (21/503) | 7.0 (78/1107) | 1.9 (1.1 to 3.1) | 0.02 |
| Emphysema | 2.0 (10/502) | 1.9 (21/1109) | 1.0 (0.5 to 2.3) | 0.96 |
| Chronic obstructive pulmonary disease (COPD) | 0.8 (4/500) | 1.3 (14/1105) | 1.7 (0.5 to 5.2) | 0.36 |
*OR and 95% CI from multivariate logistic regression adjusted for age Category, gender, education and smoking status.
†Chronic: more than 3 months/year.
‡Answering ‘Yes’ to ‘Has a physician ever told that you had (the disease)?’.
Table 3.
Risk of recent symptoms (physical and psychological) and drug use in a population exposed to the Eyjafjallajökull volcanic eruption compared to a non-exposed population
| Non-exposed |
Exposed |
|||
|---|---|---|---|---|
| % (n/N) | % (n/N) | OR (95% CI)* | p Value | |
| Respiratory symptoms† | ||||
| Shortness of breath | 3.5 (17/488) | 6.7 (72/1074) | 2.1 (1.2 to 3.6) | 0.011 |
| Feeling of tightness in chest | 1.8 (9/491) | 3.6 (38 /1070) | 2.5 (1.1 to 5.8) | 0.03 |
| Cough and phlegm† | ||||
| Cough | 6.4 (31/488) | 15.3 (166/1085) | 2.6 (1.7 to 3.9) | <0.001 |
| Phlegm | 5.5 (27/488) | 11.3 (122/1079) | 2.1 (1.3 to 3.2) | <0.001 |
| Irritation symptoms† | ||||
| Dry throat | 3.4 (17/494) | 10.1 (110/1089) | 3.1 (1.8 to 5.3) | <0.001 |
| Eye irritation and itch | 8.6 (42/487) | 20.6 (224/1085) | 2.9 (2.0 to 4.1) | <0.001 |
| Skin rash/eczema | 5.1 (25/487) | 6.2 (67/1075) | 1.2 (0.8 to 1.9) | 0.39 |
| Musculoskeletal symptoms† | ||||
| Back pain | 23.0 (116/494) | 18.2 (196/1075) | 0.7 (0.5 to 0.9) | 0.012 |
| Myalgia | 24.2 (120/496) | 20.1 (216/1073) | 0.7 (0.6 to 1.0) | 0.024 |
| Sleep† and mental health | ||||
| Insomnia | 16.9 (84/497) | 13.7 (148/1078) | 0.8 (0.6 to 1.0) | 0.08 |
| Psychological morbidity‡ | 19.0 (95/500) | 24.6 (278/1129) | 1.3 (1.0 to 1.7) | 0.05 |
| Regular drugs use (at least once per week) | ||||
| Asthma medication | 4.7 (24/510) | 3.4 (39/1147) | 0.7 (0.4 to 1.1) | 0.12 |
| Analgesics | 11.4 (58/510) | 8.7 (100/1147) | 0.7 (0.5 to 1.0) | 0.04 |
| Any drug for depression, anxiety, sleeping and other mental symptoms | 14.9 (76/510) | 12.5 (144/1148) | 0.8 (0.6 to 1.1) | 0.12 |
| Blood pressure-lowering medication | 19.6 (100/510) | 22.6 (259/1148) | 1.3 (1.0 to 1.7) | 0.10 |
*OR and 95% CI from multivariate logistic regression adjusted for age category, gender, education and smoking status.
†Answers ‘Yes, to a moderate extent’ or ‘Yes, to much extent’ to the question ‘Have any of the following symptoms disturbed your daily activities during the last month?’.
‡Psychological morbidity was derived from GHQ-12 referring to ‘the previous weeks’, using a binary cut-off score of >2.
Analysis 2: low, medium and high exposure
The prevalence of most respiratory symptoms during the last year increased with ash exposure. Adjusting for gender, age, education and smoking, the likelihood of waking up with a feeling of tightness in the chest was higher in the medium-exposure and high-exposure regions (OR 2.1; 95% CI 1.0 to 4.2) and (OR 3.1; 95% CI 1.5 to 6.6), respectively. Chronic morning phlegm was only increased in the high-exposure region (OR, 2.3; 95% CI 1.2 to 4.4; table 4).
Table 4.
Risk of respiratory symptoms (ECHRS) in a population exposed to Eyjafjallajökull volcanic eruption by exposure level at the residence
| Low exposure* |
Medium exposure* |
High exposure* |
||||
|---|---|---|---|---|---|---|
| OR (95% CI)† | % (n/N) | OR (95% CI)† | % (n/N) | OR (95% CI)† | % (n/N) | |
| Wheezing (last 12 months) | 1 (ref) | 14.3 (21/147) | 1.1 (0.6 to 1.8) | 14.6 (91/623) | 1.4 (0.8 to 2.3) | 19.1 (65/340) |
| If yes, breathlessness at the same time | 1 (ref) | 47.6 (10/21) | 1.2 (0.4 to 3.2) | 54.5 (48/88) | 1.8 (0.6 to 5.4) | 64.3 (36/56) |
| If yes, do you wheeze without having a cold | 1 (ref) | 71.4 (15/21) | 0.8 (0.2 to 2.5) | 67.4 (60/89) | 1.1 (0.3 to 3.9) | 75.4 (43/57) |
| Nocturnal chest tightness in (last 12 months) | 1 (ref) | 6.0 (9/149) | 2.1 (1.0 to 4.2) | 11.4 (71/624) | 3.1 (1.5 to 6.6) | 16.1 (55/342) |
| Breathlessness at rest | 1 (ref) | 2.7 (4/146) | 3.3 (1.2 to 9.3) | 8.2 (51/619) | 3.3 (1.1 to 9.7) | 8.9 (30/338) |
| Coughing without having a cold | 1 (ref) | 19.5 (29/149) | 2.0 (1.3 to 3.1) | 31.1 (194/623) | 1.6 (1.0 to 2.6) | 26.6 (91/342) |
| Nocturnal cough (last 12 months) | 1 (ref) | 13.6 (20/147) | 2.1 (1.3 to 3.5) | 25.0 (155/619) | 2.0 (1.2 to 3.4) | 24.1 (83/344) |
| Cough in the morning in winter | 1 (ref) | 6.7 (10/149) | 2.2 (1.1 to 4.3) | 13.7 (85/620) | 1.6 (0.8 to 3.3) | 11.1 (38/342) |
| Cough during the day or night in winter | 1 (ref) | 7.5 (11/147) | 1.7 (0.9 to 3.4) | 12.1 (75/619) | 1.3 (0.6 to 2.7) | 10.2 (35/342) |
| If yes, it is chronic‡ | 1 (ref) | 70.0 (7/10) | 0.6 (0.1 to 3.6) | 63.9 (46/72) | 1.1 (0.2 to 6.9) | 70.6 (24/34) |
| Morning winter phlegm | 1 (ref) | 8.3 (12/145) | 1.7 (0.9 to 3.2) | 13.7 (85/620) | 2.3 (1.2 to 4.4) | 18.3 (62/339) |
| Nocturnal or daytime winter phlegm | 1 (ref) | 4.9 (7/144) | 1.5 (0.6 to 3.3) | 6.9 (42/613) | 2.4 (1.0 to 5.5) | 11.8 (40/340) |
| If yes, is it chronic‡ | 1 (ref) | 85.7 (6/7) | 0.7 (0.0 to 20.8) | 92.5 (37/40) | 0.5 (0.2 to 15.7) | 81.1 (30/37) |
| Dyspnoea | 1 (ref) | 6.7 (10/144) | 1.9 (1.0 to 3.7) | 6.9 (42/613) | 2.4 (1.2 to 4.9) | 11.8 (40/340) |
| Nasal allergy and hay fever | 1 (ref) | 17.2 (25/145) | 1.2 (0.7 to 1.9) | 19.4 (122/628) | 1.1 (0.7 to 1.9) | 19.2 (66/345) |
| Allergic rhinitis | 1 (ref) | 22.8 (33/145) | 1.5 (0.9 to 2.2) | 29.5 (184/624) | 1.7 (1.1 to 2.7) | 32.4 (110/340) |
| Physician diagnosed conditions§ | ||||||
| Asthma | 1 (ref) | 17.2 (25/145) | 0.6 (0.4 to 1.0) | 10.5 (65/622) | 0.7 (0.4 to 1.2) | 12.3 (42/342) |
| Asthma diagnosis was confirmed by an MD | 1 (ref) | 95.5 (21/22) | 0.3 (0.0 to 3.2) | 93.7 (59/63) | Na¶ | 100 (37/37) |
| Heart disease | 1 (ref) | 10.1 (15/149) | 0.8 (0.4 to 1.4) | 7.2 (45/628) | 0.8 (0.4 to 1.7) | 8.6 (29/338) |
| Chronic bronchitis | 1 (ref) | 6.1 (9/147) | 1.3 (0.6 to 2.7) | 6.8 (42/620) | 1.3 (0.6 to 3.0) | 7.9 (27/340) |
| Emphysema | 1 (ref) | 1.4 (2/146) | 1.6 (0.3 to 7.5) | 1.8 (11/623) | 1.4 (0.3 to 7.1) | 2.4 (8/340) |
| Chronic obstructive pulmonary disease | 1 (ref) | 0.7 (1/146) | 2.5 (0.3 to 20.2) | 1.5 (9/619) | 1.7 (0.2 to 14.2) | 1.2 (4/340) |
*Regions are seen in figure 1.
†OR and 95% CI from multivariate logistic regression adjusted for age category, gender, education and smoking status.
‡Chronic: more than 3 months/year.
§Answering ‘Yes’ to ‘Has a physician ever told that you had (the disease)?’
¶Cannot divide with 0.
The experience of recent symptoms increased with exposure; cough (medium exposure OR 3.6; 95% CI 1.6 to 8.1; high exposure OR 4.5; 95% CI 2.0 to 10.2), phlegm (medium exposure OR 4.2; 95% CI 1.5 to 11.8, high exposure OR 6.0; 95% CI 2.1 to 17.1) and insomnia (medium exposure OR 2.4; 95% CI 1.2 to 5.0, high exposure OR 2.8; 95% CI 1.3 to 5.9; table 5). No significant associations were observed between level of exposure and feeling of tightness in the chest, psychological morbidity, use of analgesic-lowering and blood-pressure-lowering drugs, or physician-diagnosed disease, though a non-significant trend was observed with some outcomes. ORs not adjusted for age, gender, education or smoking were similar to the adjusted ones.
Table 5.
Risk of recent symptoms (physical and psychological) and drug use in a population exposed to Eyjafjallajökull volcanic eruption by exposure level at the residence
| Low exposure* |
Medium exposure* |
High exposure* |
||||
|---|---|---|---|---|---|---|
| OR (95% CI)† | % (n/N) | OR (95% CI)† | % (n/N) | OR (95% CI)† | % (n/N) | |
| Respiratory symptoms‡ | ||||||
| Shortness of breath | 1 (ref) | 2.8 (4/144) | 2.9 (1.0 to 8.5) | 6.8 (41/600) | 3.3 (1.1 to 9.9) | 8.2 (27/330) |
| Feeling of tightness in chest | 1 (ref) | 1.4 (2/145) | 3.4 (0.8 to 15.1) | 3.9 (23/597) | 3.1 (0.7 to 14.5) | 4.0 (13/328) |
| Cough and phlegm‡ | ||||||
| Cough | 1 (ref) | 4.9 (7/143) | 3.6 (1.6 to 8.1) | 15.7 (95/607) | 4.5 (2.0 to 10.2) | 19.1 (64/335) |
| Phlegm | 1 (ref) | 2.8 (4/142) | 4.2 (1.5 to 11.8) | 10.8 (65/603) | 6.0 (2.1 to 17.1) | 15.9 (53/334) |
| Irritation symptoms‡ | ||||||
| Dry throat | 1 (ref) | 2.1 (3/145) | 6.7 (2.0 to 21.6) | 11.2 (68/608) | 6.7 (2.0 to 22.2) | 11.6 (39/336) |
| Eye irritation and itch | 1 (ref) | 8.3 (12/144) | 3.4 (1.8 to 6.5) | 21.5 (130/606) | 3.6 (1.9 to 7.0) | 24.5 (82/335) |
| Skin rash or eczema | 1 (ref) | 2.1 (3/146) | 3.0 (0.9 to 10.1) | 6.0 (36/600) | 4.3 (1.3 to 14.3) | 8.5 (28/329) |
| Musculoskeletal symptoms‡ | ||||||
| Back pain | 1 (ref) | 15.4 (22/143) | 1.3 (0.8 to 2.1) | 18.0 (108/599) | 1.2 (0.7 to 2.1) | 19.8 (66/333) |
| Myalgia | 1 (ref) | 16.6 (24/145) | 1.3 (0.8 to 2.1) | 20.0 (120/600) | 1.3 (0.8 to 2.3) | 22.0 (72/328) |
| Sleep and mental health | ||||||
| Insomnia‡ | 1 (ref) | 6.3 (9/143) | 2.4 (1.2 to 5.0) | 13.8 (83/601) | 2.8 (1.3 to 5.9) | 16.8 (56/334) |
| Psychological morbidity§ | 1 (ref) | 20.0 (30/150) | 1.2 (0.8 to 1.9) | 24.8 (157/634) | 1.3 (0.8 to 2.1) | 26.4 (91/345) |
| Regular drugs use (at least once per week) | ||||||
| Asthma medication | 1 (ref) | 3.9 (6/152) | 0.7 (0.3 to 1.9) | 2.5 (16/644) | 1.2 (0.4 to 3.2) | 4.8 (17/352) |
| Analgesics | 1 (ref) | 7.2 (11/152) | 1.5 (0.7 to 2.9) | 9.0 (58/644) | 1.2 (0.6 to 2.6) | 8.8 (31/352) |
| Any drug for depression, anxiety, sleeping and other mental symptoms | 1 (ref) | 5.3 (8/152) | 3.6 (1.7 to 7.8) | 13.7 (88/644) | 2.8 (1.3 to 6.3) | 13.6 (48/352) |
| Blood pressure-lowering medication | 1 (ref) | 19.7 (30/152) | 1.7 (1.1 to 2.8) | 22.8 (147/644) | 1.4 (0.8 to 2.4) | 23.3 (82/352) |
*Regions are seen in figure 1.
†OR and 95% CI from multivariate logistic regression adjusted for age category, gender, education and smoking status.
‡Answering ‘Yes, to a moderate extent’ or ‘Yes, to much extent’ to the question ‘Have any of the following symptoms disturbed your daily activities during the last month?’.
§Psychological morbidity was derived from GHQ-12 referring to ‘the previous weeks’, using a binary cut-off score of >2.
Analysis 3: multiple symptoms
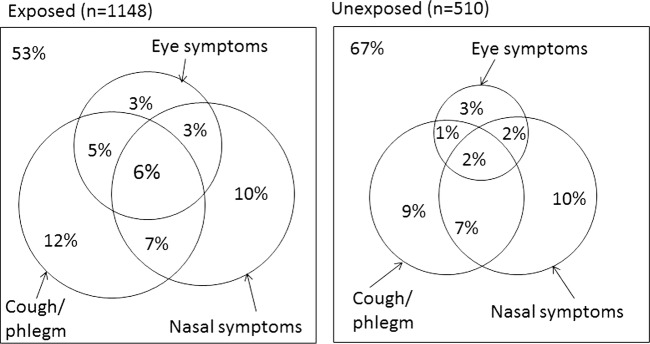
A subgroup within both populations reported multiple symptoms from nose, eyes or upper respiratory organs. The proportion reporting two or more symptoms was larger in the exposed population than in the non-exposed (23.8% vs 12.9%, data not shown), and there was a significant overlap in reporting one or more symptoms, see Venn diagram (figure 2). Within the exposed population the proportion was 13.3% in the low-exposure area, 24.7% in the medium-exposure area and 26.7% in the high-exposure area. In the exposed area, those who reported multiple key symptoms were more likely to be women (58.1% women vs 41.9% men, p<0.015), and have asthma, compared with those with no symptoms (26.9% vs 3.4% p<0.001).
Figure 2.
Venn diagram of exposed and non-exposed participants reporting one or more key symptom 6–9 months after the Eyjafjallajökull eruption. Eye symptoms: irritation, itch or other discomfort; nasal symptoms: sneeze or runny nose without having a cough; cough and/or phlegm: often cough without having a cold and/or phlegm during winter. The numbers do not add up due to rounding.
Analysing the association between exposure and psychological morbidity and adjusting for multiple symptoms, we found that having multiple symptoms was associated with psychological morbidity (OR 4.7; 95% CI 3.4 to 6.5), irrespective of exposure level.
Discussion
Our study found that 6–9 months after the Eyjafjallajökull eruption ended the participants from exposed areas reported increased wheezing, cough and phlegm, as well as recent eye and skin irritation. Participants from medium-exposure and high-exposure regions experienced significantly higher rates of upper respiratory, skin and eye irritation symptoms than those from the low-exposure region. This suggests a dose-dependent relationship of the Eyjafjallajökull ash exposure on physical symptoms.
Many of the recent physical and mental symptoms were only marginally more prevalent in the high than the medium exposure area, indicating that there is a threshold beyond which additional exposure does not result in increased morbidity. Reporting two or more key respiratory symptoms was more common in the exposed population. Compared with the non-exposed, the exposed population reported only marginally higher prevalence of psychological morbidity. However, psychological morbidity was reported to be much higher in the subgroup reporting two or more symptoms, indicating that those with many symptoms represent a more sensitive subgroup within the population which should be especially targeted in preventive actions.
The main strengths of this study, our ability to identify the whole population experiencing a volcanic eruption as well as the high participation rate, both minimise the risk of selection bias. In addition, the internal response rate (answers to specific items) was high. The exposed and non-exposed populations were demographically similar and adjustment for age, gender and education further reduces the risk of confounding. Chronic illness prevalence in this study is comparable between the two areas, suggesting that the environment and occupational exposures are not dissimilar in the two areas, both characterised by sheep and dairy farming.
Regarding the limitations of the study, we have no information on the health status of the two populations before the eruption or the health status of non-respondents, and cannot exclude the possibility that the groups may have differed before the eruption. Although the study benefits overall from the high response rate, we have limited information on non-responders and therefore it remains uncertain to what extent, if at all, attrition affects our comparison across exposure areas. Another limitation of the study is that all symptoms are self-reported that may confer misclassification of symptoms. However, it is unlikely that this bias differs across exposure categories.
Classification of exposure areas (low, medium and high), which was based on estimated ash fall, may be imprecise and actual ash exposure may also vary within the exposure areas due to local weather conditions, terrain or housing quality. Yet, if our classification is unclear or erroneous, this would reasonably result in compromised dose-dependent effects seen in our study and rather decrease the measured effects. It is also possible that exposures other than ash fall, for example, noise, visibility or living in lowlands exposed to glacial outburst floods, are significant contributors to the psychological morbidity which we observed in this study.
The exposed region is varied with respect to population density and occupation; the high-exposure area has a higher proportion of farmers, who spend more time outside, which may exaggerate the observed difference between the medium-exposure and low-exposure areas. On the other hand, residents of the high-exposure area may have been more vigilant in avoiding exposure, which would reduce the difference between the exposure areas. Data collection for the exposed group went on in November–December, and January–February for the non-exposed group, which may induce bias with respect to respiratory symptoms, as the seasonal influenza peaked during February and March in 2011.30 However, this would attenuate the observed difference seen in respiratory symptoms.
Before the eruption of Eyjafjallajökull, dust storms frequently compromised air quality in the exposed area;20 21 however, a study from 2004 on Icelandic farmers found no difference in respiratory symptoms between controls sampled from the national population and farmers, or among farmers in different regions of Iceland.31 Chronic disease prevalence is similar in the exposed and non-exposed areas, further suggesting that the dust storms occurring before the eruption have no lasting effect on people's health. Also, the dose–response character of symptoms with respect to exposure to the volcano suggests that the symptoms are associated with the eruption.
Our findings of high rates of cough and eye irritation after the eruption are consistent with other studies, for example of the Mount St Helens eruption, where the number of emergency room visits, especially for respiratory conditions in those with underlying illness increased threefold to fivefold in the weeks following the eruption. Eye irritation was also more common in loggers exposed to Mount St Helens ash, and the amount of eye mucus seemed to be dose-dependent on the ash density.32
Dose-response and threshold effects of urban-type airborne particles on health have been explored in epidemiological studies,33 but rarely in humans exposed to volcanic ash. A Japanese study of asthma treatment and volcanic ash exposure found worsening of symptoms in asthmatics in areas with more than 100 g/m2 ash, but not in areas with less ash fall.34 The psychological morbidity found in the current study (20–26%) was lower than that found in the survey of the most exposed area right after the Eyjafjallajökull eruption ended (39%).7 This may indicate that residents have somewhat adapted to the strain following the eruption. Although a disaster with more dramatic consequences, a similar trend was found in a Japanese study of evacuees from a volcanic area where 66.1% showed signs of psychological morbidity (GHQ-30) 6 months after evacuation, while 4 years later the rate had fallen to 45.6%.17 In our study, psychological morbidity and insomnia were most common in the high-exposure group, as was the regular intake of drugs for depression, anxiety, sleep problems or other mental symptoms. Dose–response trends were found between psychological morbidity and exposure to the Mount St Helens eruption,16 indicating possible long-term risk of further psychological morbidity in the high-exposure group.
At this point, we cannot speculate about the effects of financial loss because of damages to property, this will be addressed in future studies.
The results from this study have implications for planners and authorities, as it indicates risk groups particularly susceptible to adverse reactions after exposure to volcanic ash. Also, the study design and registration of the participants enables follow-up of the exposed group, both directly in a new study, as well as in hospital-, medicine- and mortality registers.
Conclusions
In this population-based study we documented a high prevalence of respiratory symptoms 6–9 months following the volcanic eruption in Eyjafjallajökull, especially among those most exposed. Also, subgroups who reported more than one physical symptom were more prone to experience psychological difficulties. The study reveals that the adverse health effects of a volcanic eruption may last for many months beyond the eruption and the immediate disaster relief services provided. This is important for health authorities to bear in mind.
These findings are an incentive for further studies, for example, on predictive factors for morbidity, the health of children and long-term follow-up. Important knowledge may be gained from such studies to help develop mitigation measures at future eruptions.
Supplementary Material
Acknowledgments
The study was funded through a grant from the Icelandic Government, the funding source had no influence on the design, analysis or reporting of this study. We thank Hildur Friðriksdóttir, MA (University of Iceland) for administrative support. Last but not least, we thank the participants for their cooperation.
Footnotes
Contributors: All the authors conceived and designed the study and had full access to the data. AH, UAV, ÞBK and GP gathered the data and HKC, AH, UAV and GP did the statistical analyses. ÞÞ provided estimates of ash fall density. HKC and AH drafted the article, which was critically revised by all the authors, who also approved the final report. GP (guarantor) supervised the study, had full access to all the data in the study and carries the final responsibility for deciding to submit it for publication, and takes responsibility for the integrity of the data and the accuracy of the data analysis.
Funding: The study and article was funded through a grant from the Icelandic Government, the funding source had no influence on the design, analysis or reporting of this study. We do not wish to share the data used in this study. We thank Hildur Friðriksdóttir, M.A. (University of Iceland) for administrative support. Last but not least, we thank the participants for their cooperation.
Competing interests: None.
Ethics approval: The study was approved by The Icelandic Data Protection Authority (no. S4878/2010) and The Science Bioethics Committee (no. VSNb2010080002/03.7).
Provenance and peer review: Not commissioned; externally peer reviewed.
Data sharing statement: GP holds all the data used in this study. The data set holds additional information about health of children and mental health, which the authors seek to publish later. The data are anonymised, but could hold identifiable details, thus we do not wish to share the data.
References
- 1.Guha-Sapir D, Vos F, Below R.Annual disaster statistical review: Numbers and trends 2010. Louvain-la-Neuve (Belgium): Centre for Research on the Epidemiology of Disasters (CRED): 2011 p. 50. Report No. 280. [accessed 2012 March 27]. http://www.cred.be/sites/default/files/ADSR_2010.pdf.
- 2.Lancet Natural disasters—taking a longer term view. Lancet 2011;377:439. [DOI] [PubMed] [Google Scholar]
- 3.Stohl A, Prata AJ, Eckhardt S, et al. Determination of time- and height-resolved volcanic ash emissions for quantitative ash dispersion modeling: the 2010 Eyjafjallajökull eruption. Atmos Chem Phys Discuss 2011;11:5541–88 [Google Scholar]
- 4.Thorsteinsson T, Jóhannsson T, Stohl A, et al. High levels of particulate matter in Iceland due to direct ash emissions by the Eyjafjallajökull eruption and resuspension of deposited ash. J Geophys Res 2012;117:1–9. [Google Scholar]
- 5.Jóhannsson T. Internal Report 2011. Reykjavík, Iceland: Environmental Agency of Iceland, 2012 [Google Scholar]
- 6.Gislason SR, Hassenkam T, Nedel S, et al. Characterization of Eyjafjallajökull volcanic ash particles and a protocol for rapid risk assessment. P Natl Acad Sci USA 2011;108:7307–12 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 7.Carlsen H, Gíslason T, Benediktsdottir B, et al. A survey of early health effects of the Eyjafjallajökull 2010 eruption in Iceland—a population-based study. BMJ Open 2012;2:e000343 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 8.Bird D, Gísladóttir G. Residents’ attitudes and behaviour before and after the 2010 Eyjafjallajökull eruptions—a case study from southern Iceland. Bull Volcanol 2012;74:1263–79 [Google Scholar]
- 9.Horwell C, Baxter P. The respiratory health hazards of volcanic ash: a review for volcanic risk mitigation. Bull Volcanol 2006;69:1–24 [Google Scholar]
- 10.Yano E, Yokoyama Y, Nishii S. Chronic pulmonary effects of volcanic ash: an epidemiologic study. Arch Environ Health: Int J 1986;41:94–9 [DOI] [PubMed] [Google Scholar]
- 11.Buist AS, Bernstein RS, Johnson LR, et al. Evaluation of physical health effects due to volcanic hazards: human studies. Am J Public Health 1986;76(Suppl):66–75 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 12.Hickling J, Clements M, Weinstein P, et al. Acute health effects of the Mount Ruapehu (New Zealand) volcanic eruption of June 1996. Int J Environ Health Res 1999;9:97–107 [Google Scholar]
- 13.Hansell AL, Horwell CJ, Oppenheimer C. The health hazards of volcanoes and geothermal areas. Occup Environ Med 2006;63:149–56 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 14.Longo BM, Rossignol A, Green JB. Cardiorespiratory health effects associated with sulphurous volcanic air pollution. Public Health 2008;122:809–20 [DOI] [PubMed] [Google Scholar]
- 15.Durand M, Wilson JG. Spatial analysis of respiratory disease on an urbanized geothermal field. Environ Res 2006;101:238–45 [DOI] [PubMed] [Google Scholar]
- 16.Shore JH, Tatum EL, Vollmer WM. Evaluation of mental effects of disaster, Mount St. Helens eruption. Am J Public Health 1986;76(Suppl):76–83 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 17.Ohta Y, Araki K, Kawasaki N, et al. Psychological distress among evacuees of a volcanic eruption in Japan: a follow-up study. Psychiatry ClinNeurosci 2003;57:105–11 [DOI] [PubMed] [Google Scholar]
- 18.Small C, Naumann T. The global distribution of human population and recent volcanism. Glob Environ Chang—Part B: Environ Hazards 2003;3:93–109 [Google Scholar]
- 19.Thordarson T, Larsen G. Volcanism in Iceland in historical time: volcano types, eruption styles and eruptive history. J Geodyn 2007;43:118–52 [Google Scholar]
- 20.Arnalds Ó. Dust sources and deposition of aeolian materials in Iceland. Iceland Agric Sci 2010;23:3–29 [Google Scholar]
- 21.Prospero JH, Bullard JE, Hodgkins R. High-latitude dust over the North Atlantic: inputs from Icelandic Proglacial dust storms. Science 2012;334:1078–82 [DOI] [PubMed] [Google Scholar]
- 22.Larsen G, Höskuldsson Á. Ash layer from Eyjafjallajökull April 17 2010. Reykjavík, Iceland: Institute of Earth Sciences. 2010. (accessed 1 Nov 2011) http://jardvis.hi.is/Apps/WebObjects/HI.woa/swdocument/1015817/2010-04-17_GLARE+%C3%B6skulag_Eyjafj%C3%B6ll.pdf
- 23.Statistics Iceland HagstofaÍslands—Talnaefni » Mannfjöldi » PX. (accessed 1 Sept 2011) http://hagstofa.is/Hagtolur/Mannfjoldi/Byggdakjarnar,-postnumer,-hverfi
- 24.Burney PG, Luczynska C, Chinn S, et al. The European Community Respiratory Health Survey. Eur Respir J 1994;7:954–60 [DOI] [PubMed] [Google Scholar]
- 25.Goldberg D, Williams P. A user's Guide to the General Health Questionnaire. Oxford: NFER-Nelson Publishing Company Ltd, 1988 [Google Scholar]
- 26.Goldberg DP, Gater R, Sartorius N, et al. The validity of two versions of the GHQ in the WHO study of mental illness in general health care. Psychol Med 1997;27:191–7 [DOI] [PubMed] [Google Scholar]
- 27.Hankins M. The reliability of the twelve-item general health questionnaire (GHQ-12) under realistic assumptions. BMC Public Health 2008;14:355. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 28. LimeSurvey Project Team, Schmitz C. LimeSurvey: An Open Source survey tool. Hamburg, Germany: LimeSurvey Project, 2012. [Google Scholar]
- 29.IBM IBM SPSS Statistics Version 19. New York: IBM, 2010 [Google Scholar]
- 30.Gudnason G, Gudnason T. Influenza-like illness this winter. EPI—ICE January–March 2011. 7th edn. Reykjavík, Iceland: Landlæknisembættið Directorate of Health, 2011, 2–3 [Google Scholar]
- 31.Sigurdarson ST, Gudmundsson G, Sigurvinsdottir L, et al. Respiratory disorders are not more common in farmers. Results from a study on Icelandic animal farmers. Respir Med 2008;102:1839–43 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 32.Bernstein R, Baxter P, Falk H, et al. Immediate public health concerns and actions in volcanic eruptions: lessons from the Mount St. Helens eruptions, May 18–October 18, 1980 . Am J Public Health 1986;76(Suppl):25–38 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 33.Daniels MJ, Dominici F, Samet JM, et al. Estimating particulate matter-mortality dose-response curves and threshold levels: an analysis of daily time-series for the 20 largest US cities. Am J Epidemiol 2000;152:397–406 [DOI] [PubMed] [Google Scholar]
- 34.Shimizu Y, Dobashi K, Hisada T, et al. Acute impact of volcanic ash on asthma symptoms and treatment. Int J Immunopathol Pharmacol 2007;20(2 Suppl):9–14 [DOI] [PubMed] [Google Scholar]
Associated Data
This section collects any data citations, data availability statements, or supplementary materials included in this article.