Abstract
Objectives
To characterise and confirm the presence of Mansonella ozzardi microfilariae in the cornea by biomicroscopy and corneal confocal microscopy.
Design
Cross-sectional study.
Settings
Clinical practice study in patients from rural communities in Coari city on the Solimões river, Amazonas state, Brazil.
Participants
The eyes of 212 consecutive volunteer patients were examined using a flash light and their blood checked for the presence of microfilariae by an expert microscopist. Patients with suspicious corneal lesions (characterised as nummular keratitis) were submitted to biomicroscopy, fundoscopy and corneal confocal microscopy evaluation (CCME). In two patients, a biopsy of the limbal conjunctiva adjacent to the nummular keratitis was carried out and blood collected from the surgical wound for microfilariae investigation by thick blood film examination.
Primary and secondary outcome measures
Positive correlation between corneal biomicroscopic and confocal lesions and M ozzardi microfilaremia.
Results
Of the 212 patients, 56 (26.4%) were positive for microfilaremia. 22 patients with nummular keratitis identified under flash light examination underwent biomicroscopy and CCME. Corneal lesions were positively correlated to microfilaremia (p=0.0001). At biomicroscopy, lesions were classified as quiescent or active. At CCME, lesions were categorised as circular or filiform. The associations between corneal lesions, CCME findings and microfilaremia are shown.
Conclusions
We describe M ozzardi microfilariae in the cornea and the associated eye pathology. Further studies using ocular tissue PCR and other imaging techniques would be helpful.
Keywords: Ophthalmology, Parasitology
Article summary.
Article focus
To describe corneal lesions associated with Mansonella ozzardi microfilaremia.
To describe corneal confocal microscopy images in M ozzardi microfilaremia.
To correlate ophthalmological clinical signs with the presence of microfilaremia in some patients.
Key messages
Biomicroscopic investigation and corneal confocal microscopy can identify M ozzardi microfilariae in the cornea.
M ozzardi microfilaria can be pathogenic to the eye.
Further studies using ocular tissue PCR and other imaging techniques would be helpful.
Strengths and limitations of this study
The study describes a very common but under-studied disease in a remote area of Brazil.
This is the first description of M ozzardi corneal lesions using confocal microscopy.
A major limitation is the absence of a control group and follow-up of patients due to field access difficulties.
Introduction
Hundreds of types of filariae have been described. Mansonella ozzardi is one of the few causing infection in humans and was first reported as being associated with corneal disease in 1998.1 M ozzardi is a filaria exclusively found in the America continent and one of the causes of mansonelliasis.2 In Brazil it is found in the states of Roraima, Mato Grosso and Amazonas.3–5 The microfilariae of M ozzardi are found in peripheral blood and can be identified by their morphological characteristics or by molecular biology.6–11
The clinical features of mansonelliasis are not well described. Infected individuals are generally asymptomatic or have signs and symptoms common to other infections such as fever, cold legs, joint pain and headache.12 Signs and symptoms may also include skin and ocular lesions1 13 14 and be associated with Wolbachiae, which are bacterial endosymbionts of insects and many filarial nematodes and whose products trigger inflammatory responses.14
Coari city on the Solimões river (04°08′S, 63°07′W) has a high prevalence of M ozzardi infection. Studies showed an average infection rate of between 13.3% and 18.9%, with a higher prevalence in rural areas and older patients.2 14 Other studies conducted in non-endemic areas found no M ozzardi infection.15 Onchocerciasis is not found in this region of the Amazon.16
The objective of the present study is to characterise and confirm the presence of M ozzardi microfilariae in the cornea and to demonstrate the positive correlation between corneal biomicroscopic and confocal lesions and M ozzardi microfilaremia.
Methods
This was a cross-sectional, clinical practice study of 212 consecutive volunteer patients from rural communities of Coari city on the Solimões river, Amazonas state, Brazil, examined in August 2010. All patients had their eyes examined using a flash light and their blood checked for the presence of microfilariae by an expert microscopist. Peripheral blood infection by M ozzardi was determined by light microscopy examination (200× and 400×) of Giemsa-stained thick smears of peripheral blood obtained by digital puncture with a sterile disposable lancet,6 Knott and polycarbonate membrane filtration and by a new identification protocol using PCR (M Martins et al, unpublished observations).
Patients with nummular keratitis, similar to those previously described in the literature,1 13 14 at unilateral or bilateral eye examination were subjected to bilateral biomicroscopy, fundoscopy and corneal confocal microscopy evaluation (CCME) using the Rostock Cornea Module of the Heidelberg Retina Tomograph. Slit lamp photographs were taken. The CCME was performed on the central cornea of both eyes and on the corneal regions affected by nummular keratitis.
Two patients underwent biopsy of the limbal conjunctiva adjacent to the area of nummular keratitis and blood was collected from the surgical wound for microfilariae investigation by thick blood film examination.
Inclusion criteria were volunteering for a blood check for M ozzardi microfilaremia and signing the informed consent form or have it signed by a legal representative. Exclusion criteria were not volunteering or unable to sign the informed consent form.
This was a prospective study and was approved by the UNIFESP Ethics Committee (number 0767/10). All participants signed the protocol's informed consent form.
Data were presented in contingency tables and Fisher's exact test was used to compare proportions. p Values less than 0.05 were considered statistically significant. Analyses were carried out in Stata V.11 (College Station, Texas, USA).
Results
Of the 212 patients, 56 (26.4%) were positive for microfilaremia. Twenty two patients with nummular keratitis identified by flash light examination underwent biomicroscopy and CCME. Four of the patients had bilateral lesions. The statistically significant association between microfilaremia and corneal lesions is shown in table 1 (p=0.0001).
Table 1.
Association between corneal lesions and microfilaremia detected by Giemsa staining, Knott or polycarbonate membrane filtration and/or PCR*
| Corneal lesions |
|||
|---|---|---|---|
| Positive (+) | Negative (−) | Total | |
| Microfilaremia | |||
| Positive (+) | 14 (63.6%) | 42 (22%) | 56 |
| Negative (−) | 8 (33.4%) | 148 (88%) | 156 |
| Total | 22 | 190 | 212 (100%) |
*p=0.0001.
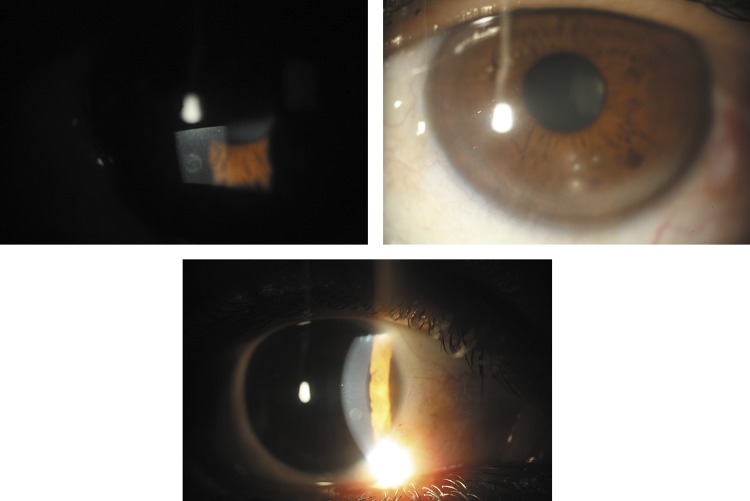
At biomicroscopy, some lesions were characterised as nummular, mid periphery and anterior stromal corneal opacities, 0.5–1.0 mm in diameter with a central mottled appearance surrounded by an opaque halo, and with a marked boundary between the white opacity and the clear cornea. Other lesions were characterised by nummular keratitis, also located at the mid periphery and anterior stroma. In all cases there was a normal translucent area between the corneal lesions and the limbus and an absence of corneal neovascularisation or other changes (figure 1). The clinical characteristics of the lesions as well as their high prevalence exclude the possibility of trauma or other known causes of the differential diagnosis, specifically onchocerciasis. The association between nummular keratitis and microfilaremia is shown in table 2 (p=0.1347).
Figure 1.
Clinical features. (Left and bottom) Central irregular ‘mottled’ appearance surrounded by an opaque halo; (right) four typical keratitis lesions with a normal translucent area between the lesions and the limbus, but no corneal neovascularisation.
Table 2.
Association between corneal lesions and microfilaremia detected by Giemsa staining, Knott or polycarbonate membrane filtration and/or PCR*
| Corneal lesions |
|||
|---|---|---|---|
| Keratitis | Opacities | Total | |
| Microfilaremia | |||
| Positive (+) | 10 (76.9%) | 4 (44%) | 14 |
| Negative (−) | 3 (23.1%) | 5 (56%) | 8 |
| Total | 13 | 9 | 22 (100%) |
*p=0.1347.
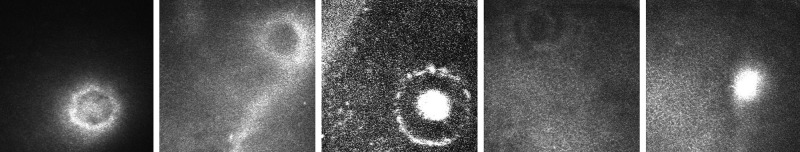
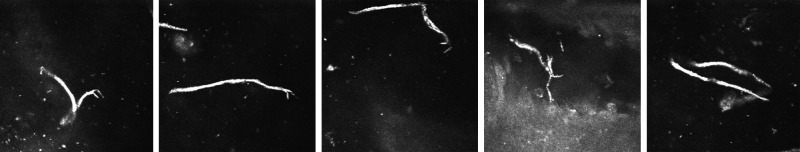
Among the 22 patients with corneal lesions at biomicroscopy, 12 also had suspected related lesions at CCME. These confocal suspected lesions displayed two patterns: circular (increased reflectivity, measuring about 80 µm in diameter, located at the sub-epithelial level, as shown in figure 2) and linear (filiform lesions 250–300 µm in length and 10–15 µm in width, with one ‘C’-shaped end and the other tapered, as shown in figure 3). Five patients had only circular lesions, one patient had only linear lesions and six patients had both. The association between confocal lesions and microfilaremia is shown in table 3 (p=0.4266).
Figure 2.
Five different sub-epithelial circular lesions (about 80 µm in diameter) in different patients. Images are 400 µm×400 µm.
Figure 3.
Filiform lesions located above the epithelium basal layer about 300 µm long and 10 µm wide, with one ‘C’-shaped end and the other tapered, all in the same patient, who had a limbal conjunctiva thick blood smear positive for Mansonella ozzardi. Images 400 µm×400 µm.
Table 3.
Association between confocal lesions and microfilaremia (detected by Giemsa staining, Knott or polycarbonate membrane filtration and/or PCR)*
| Confocal lesions |
|||
|---|---|---|---|
| Positive (+), circular/linear/both | Negative (−) | Total | |
| Microfilaremia | |||
| Positive (+) | 2 (25%)/1 (12.5%)/5 (62.5%) | 6 | 14 |
| Negative (−) | 3 (75%)/0/1 (25%) | 4 | 8 |
| Total | 12 | 10 | 22 |
*p=0.4266.
Examination of thick blood smears from the limbal conjunctiva in two patients with linear lesions confirmed microfilaremia. Figure 4 compares the M ozzardi microfilaria seen in the thick blood smear with the confocal microscopy image.
Figure 4.
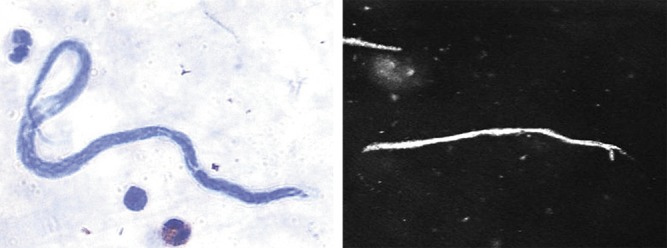
Microfilaria as detected by thick blood smear and corneal confocal image from a patient who underwent both tests.
Discussion
There is a high prevalence of M ozzardi in some riverside communities in the Amazon with the increasing occupation of the rainforest.2 16 Previous studies in Coari city showed average infection rates of between 13.3% and 18.9%, with a higher prevalence in rural areas and older patients.2 14 Other studies conducted in non-endemic areas show no M ozzardi infection.15 Prevalence was 26.4% in the present study.
Presumed keratitis caused by M ozzardi has also been described in Brazil by previous authors, with no other associated aetiology being identified.1 13 14 Comparison with other filarial diseases shows some similarities with onchocerciasis, but onchocerciasis has a different clinical picture and is rare in Brazil: it has different microfilaria tail characteristics and is only found in the Yanomami Indian Reservation close to the Venezuelan border.17 18 None of the patients had lived in or visited Onchocerca areas.
A study conducted in Pauiní on the Purus river identified 20 non-Indian habitants with peripheral corneal opacities among 524 patients examined. No microfilariae were seen in the skin snips and Mansonella sp. was only found in the blood of the two patients from whom it was possible to collect blood.1 Another study conducted in São Gabriel da Cachoeira (on the Negro river) showed a positive association between M ozzardi and corneal lesions as patients with ocular changes also had M ozzardi microfilariae in blood samples.13 However, all skin biopsies were negative for microfilaria including Onchocerca. Cohen et al also reported a positive relationship between keratitis and microfilaremia in the Amazon.14 This association was also found to be statistically significant in our study, as shown in table 1 (p=0.0001).
Corneal lesions were classified as either corneal opacities or keratitis, as shown in figure 1. Keratitis was more common among patients positive for microfilaremia, while corneal opacity was more common among patients negative for microfilaria, but neither finding was statistically significant (table 2, p=0.1347). One explanation for negative microfilaremia in patients with keratitis could be false negative blood tests, while chronic infection may explain the finding of microfilaremia in patients with corneal opacity .
As in previous studies,1 13 14 we were unable to detect filariae by biomicroscopy in any ocular tissue, but corneal lesions similar to those previously described were found. Using CCME, we detected lesions which had not been previously reported. Patients positive for microfilaremia had a higher proportion of confocal lesions (table 3, p=0.4266). Confocal lesions were categorised as linear or circular. More patients with lesions identified by CCME were positive for microfilaremia and more patients negative for microfilaremia had circular lesions, although this was not statistically significant (table 3, p=0.4266). This suggests that linear lesions are associated with active disease and circular lesions with inflammatory scars, associated or not with active disease. Linear lesions were larger (250–300 µm) on CCME than in microbiological studies (149–240 µm).6 The microfilariae seen in ocular tissue are probably dead and could have been enlarged due to an inflammatory reaction. In addition, circular lesions could be cross-sections of the remnants of adult worms (26–49 mm in length by 00.7–0.15 mm in diameter).6
Unaffected patients were not tested because of fieldwork difficulties. We carried out confocal microscopy in both eyes of all 22 patients with keratitis. In the 18 with unilateral keratitis, the non-affected eye showed no alterations on confocal microscopy.
New molecular biology techniques for M ozzardi identification6–11 could be helpful in future PCR studies of ocular tissue to confirm the association between microfilaremia and ocular lesions.
Conclusion
We describe M ozzardi microfilariae in the cornea and the associated eye pathology. Further studies using ocular tissue PCR and other imaging techniques would be helpful.
Supplementary Material
Acknowledgments
We wish to thank the following members of the Mansonella-Brazil Group: M Borborema, P Cunha, G Fontes, JF Medeiros, FAC Pessoa and T Zanelato. We also thank Nakano EM for helping with confocal microscopy analysis.
Footnotes
Contributors: LMMV and RBJ participated in the conception and design of the study, and drafted and wrote the manuscript. LMMV, RBJ, JMC, MJC and MM participated in the collection, analysis and interpretation of data, and in the revision and final approval of the manuscript.
Funding: FAPESP (Fundação de Amparo à Pesquisa do Estado de São Paulo or Foundation for Research Support of São Paulo State) funded this study.
Competing interests: None.
Ethics approval: UNIFESP Ethics Committee (number 0767/10) approved this study.
Data sharing statement: Further information such as population data and additional images of ophthalmological examination (biomicroscopy and confocal microscopy) can be obtained from the corresponding author.
Provenance and peer review: Not commissioned; externally peer reviewed.
References
- 1.Branco BC, Chamon W, Belfort R, et al. Ocular findings in Pauiní (Southwest of the Brazilian Amazon) and possible corneal lesions caused by Mansonella. Arq Bras Oftalmol 1998;61:647–82 [Google Scholar]
- 2.Martins M, Pessoa FA, de Medeiros MB, et al. Mansonella ozzardi in Amazonas, Brazil: prevalence and distribution in the municipality of Coari, in the middle Solimões River. Mem Inst Oswaldo Cruz 2010;105:246–53 [DOI] [PubMed] [Google Scholar]
- 3.Deane MP. Sobre a incidência de filárias humanas em Manaus, estado do Amazonas. Rev Serv Esp Saude Publ 1949;2:849–58 [Google Scholar]
- 4.Oliveira wr. (Filarial infestation in inhabitants of Vila Pereira, territory of Roraima (Brazil)). Rev Inst Med Trop Sao Paulo 1963;5:287–8 [PubMed] [Google Scholar]
- 5.D'Andretta, Pio da Silva CM, Kameyana F. Ocorrência da mansonelose entre índios do alto Xingu. Rev Soc Bras Med Trop 1969;3:11 [Google Scholar]
- 6.Orihel TC, Eberhard ML. Mansonella ozzardi: a redescription with comments on its taxonomic relationships. Am J Trop Med Hyg 1982;31:1142–7 [DOI] [PubMed] [Google Scholar]
- 7.Morales-Hojas R, Post RJ, Shelley AJ, et al. Characterisation of nuclear ribosomal DNA sequences from Onchocerca volvulus and Mansonella ozzardi (Nematoda: Filarioidea) and development of a PCR-based method for their detection in skin biopsies. Int J Parasitol 2001;31:169–77 Erratum in: Int J Parasitol 2001;31(8):850–1 [DOI] [PubMed] [Google Scholar]
- 8.Vera LJ, Basano S de A, Camargo J de S, et al. Improvement of a PCR test to diagnose infection by Mansonella ozzardi. Rev Soc Bras Med Trop 2011;44:380–2 [DOI] [PubMed] [Google Scholar]
- 9.Tang TH, López-Vélez R, Lanza M, et al. Nested PCR to detect and distinguish the sympatric filarial species Onchocerca volvulus, Mansonella ozzardi and Mansonella perstans in the Amazon Region. Mem Inst Oswaldo Cruz 2010;105:823–8 [DOI] [PubMed] [Google Scholar]
- 10.Degese MF, Cabrera MG, Krivokapich SJ, et al. Contribution of the PCR assay to the diagnosis of Mansonella ozzardi in endemic areas of Argentina. Rev Argent Microbiol 2012;44:97–100 Spanish.Genetic [PubMed] [Google Scholar]
- 11.Marcos LA, Arrospide N, Recuenco S, et al. Genetic characterization of atypical mansonella (Mansonella) ozzardi microfilariae in human blood samples from Northeastern Peru. Am J Trop Med Hyg 2012;87:491–4 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 12.Batista D, Cerqueira NL, Moraes MA. (Epidemiology of Manson's disease in a locality in the interior of Amazonas). Rev Assoc Med Bras 1960;6:176–84 [PubMed] [Google Scholar]
- 13.Garrido C, Campos M. First report of presumed parasitic keratitis in Indians from the Brazilian Amazon. Cornea 2000;19:817–19 [DOI] [PubMed] [Google Scholar]
- 14.Cohen JM, Ribeiro JA, Martins M. Ocular manifestations in mansonelliasis. Arq Bras Oftalmol 2008;71:167–71 Portuguese [DOI] [PubMed] [Google Scholar]
- 15.Basano S de A, Camargo J de S, Vera LJ, et al. Investigation of the occurrence of Mansonella ozzardi in the State of Rondônia, Western Amazonia, Brazil. Rev Soc Bras Med Trop 2011;44:600–3 [DOI] [PubMed] [Google Scholar]
- 16.Medeiros JF, Py-Daniel V, Barbosa UC, et al. (Occurrence of Mansonella ozzardi (Nematoda, Onchocercidae) in riverine communities of the Purus river, Boca do Acre municipality, Amazonas State, Brazil). Cad Saude Publica 2009;25:1421–6 [DOI] [PubMed] [Google Scholar]
- 17.Belfort Jr R, Moraes. M—Oncocercose ocular no Brasil. AMB Rev Assoc Med Bras 1979;25:123–7 [PubMed] [Google Scholar]
- 18.Sauerbrey M. The Onchocerciasis Elimination Program for the Americas (OEPA). Ann Trop Med Parasitol 2008;102 Suppl 1:25–9 [DOI] [PubMed] [Google Scholar]
Associated Data
This section collects any data citations, data availability statements, or supplementary materials included in this article.