Abstract
Vaccination strategies for protection against a number of respiratory pathogens must induce T-cell populations in both the pulmonary airways and peripheral lymphoid organs. In this study, we show that pulmonary immunization using plasmid DNA formulated with the polymer polyethyleneimine (PEI-DNA) induced antigen-specific CD8+ T cells in the airways that persisted long after antigen local clearance. The persistence of the cells was not mediated by local lymphocyte proliferation or persistent antigen presentation within the lung or airways. These vaccine-induced CD8+ T cells effectively mediated protective immunity against respiratory challenges with vaccinia virus and influenza virus. Moreover, this protection was not dependent upon the recruitment of T cells from peripheral sites. These findings demonstrate that pulmonary immunization with PEI-DNA is an efficient approach for inducing robust pulmonary CD8+ T-cell populations that are effective at protecting against respiratory pathogens.
Introduction
Virus-specific CD8+ T cells contribute to the resolution of infection by a number of respiratory viruses. Following clearance of these viruses, the virus-specific T cells contract in numbers and a stable pool of memory cells is established that persists in the peripheral blood and lymphoid tissues. These memory T cells also accumulate in the pulmonary airways and parenchyma.1 Studies in mice and humans using sendai virus, influenza virus, and respiratory syncytial virus suggest that memory CD8+ T cells in the pulmonary airways may provide a first line of defense against secondary virus infections.2, 3, 4
Vaccines that induce strong antiviral CD8+ T-cell responses should confer protective immunity against such respiratory pathogens. Pulmonary immunization strategies have been developed that generate potent immune responses and protect against aerosolized challenge viruses.5, 6, 7 For optimal protection against these pathogens, immunization should induce memory CD8+ T cells that reside in the pulmonary compartment, systemic effector-memory CD8+ T cells that can rapidly migrate to the airways, and a reservoir of central memory CD8+ T cells that can expand in numbers following exposure to virus.8 Therefore, an effective vaccine against respiratory viruses should induce both pulmonary and systemic memory T-cell populations.
We have previously shown that pulmonary immunization with a plasmid DNA vaccine formulated with the cationic polymer polyethyleneimine (PEI; PEI-DNA) resulted in antigen expression in the lungs and the consequent generation of antigen-specific CD8+ T cells in the systemic circulation, the lung mucosa, and other mucosal compartments.9 We have also demonstrated that systemic antigen-specific CD8+ T cells differentiated into effector-memory and central-memory populations following pulmonary PEI-DNA immunization. The present studies were initiated to further characterize the immune responses generated in the airways following pulmonary PEI-DNA immunization and determine the contribution of PEI-DNA vaccine-induced CD8+ T cells to protective immunity against a respiratory viral challenge. We demonstrate that pulmonary PEI-DNA immunization induces antigen-specific CD8+ T cells that persist in the airways and these resident cells confer protection against infection by respiratory viruses.
Results
Pulmonary administration of plasmid DNA formulated with PEI induces persistent antigen-specific CD8+ T cells in the lungs
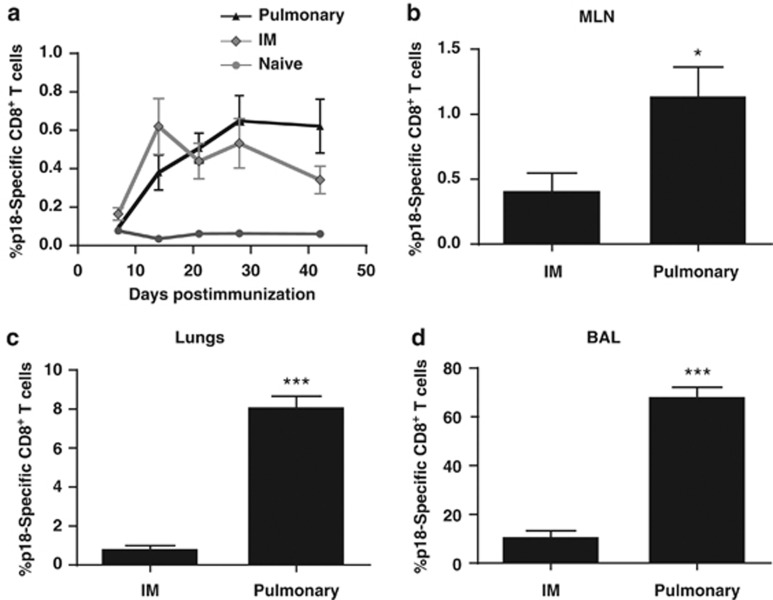
Induction of antigen-specific CD8+ T-cell responses in the lungs by pulmonary mucosal immunization may contribute to protection against inhaled pathogens. We therefore studied the effect of pulmonary DNA administration on the kinetics of CD8+ T-cell responses in the systemic circulation and in the pulmonary compartment. For this pulmonary immunization, we formulated a plasmid DNA immunogen with the polymer PEI, as we previously showed that formulation with PEI improves plasmid DNA expression in the lungs and enhances pulmonary vaccination-induced systemic CD8+ T-cell responses.9 This was not the case for immunization by the intramuscular (IM) route where formulation with PEI diminished plasmid DNA expression and did not enhance vaccine-induced CD8+ T-cell responses. Based on previous experiments, we determined that a pulmonary dose of 40 μg plasmid DNA per mouse, given on two consecutive days, elicited the best responses (data not shown). We immunized Balb/c mice by the pulmonary route with 40 μg HXBc2 gp120 DNA complexed with PEI and by the IM route with 40 μg HXBc2 gp120 DNA solution. Vaccine elicited CD8+ T-cell responses were measured using the p18/H-2Dd tetramer to detect cell populations specific for the 10-amino-acid H-2Dd-restricted dominant epitope of HXBc2 gp120.10 Peripheral blood mononuclear cell p18-specific CD8+ T-cell responses were comparable in magnitude following pulmonary or IM DNA-gp120 immunizations (Figure 1a). The kinetics of the p18-specific responses elicited by the two formulations and routes of administration were different, with peripheral blood p18-specific responses induced by pulmonary PEI-DNA-gp120 immunization reaching peak levels more slowly than those induced by IM immunization.
Figure 1.
Effect of the route of immunization on systemic and mucosal p18-specific CD8+ T-cell responses. Mice were immunized by the intramuscular (IM) route with 40 μg of plasmid DNA expressing HIV-1 HXB2 gp120 protein or by the pulmonary route with 40 μg of this DNA formulated with polyethyleneimine (PEI). (a) Kinetics of the p18-specific CD8+ T-cell response in the peripheral blood of immunized mice. p18-specific CD8+ T-cell responses in (b) mediastinal lymph nodes (MLN), (c) pulmonary lymphocytes and (d) broncho-alveolar lavage (BAL) lymphocytes of mice on day 42 following IM immunization with DNA-gp120 or by the pulmonary route with PEI-DNA-gp120. Data represent means of 4–8 mice per group±s.e.
We then assessed whether pulmonary DNA immunization could induce persistent immunity in the lungs. We isolated vaccine-elicited CD8+ T cells from three anatomic compartments: the mediastinal lymph nodes (MLN), the pulmonary airways, and the pulmonary parenchyma. To assess the persistence of pulmonary cellular immunity, CD8+ T-cell responses were measured 6 weeks following immunization. p18-Specific CD8+ T-cell responses were significantly higher in the MLN, lungs, and airways of the mice immunized by the pulmonary than by the IM route (Figure 1b–d). Pulmonary immunization with PEI-DNA-empty plasmid in conjunction with IM DNA-gp120 did not increase the magnitude of p18-specific responses in the peripheral blood or in the pulmonary compartments compared with IM DNA-gp120 immunization (see Supplementary Figure S1 online). Therefore, accumulation of antigen-specific CD8+ T cells in the pulmonary compartment following pulmonary administration of PEI-DNA is not an artifact resulting from the effects of PEI on the innate immune system or the recruitment of antigen-specific cells from the periphery. These findings establish that pulmonary PEI-DNA immunization can induce CD8+ T-cell immunity in the pulmonary compartment that persists for a prolonged period of time.
Persistence of CD8+ T cells in the lungs following pulmonary DNA immunization is not a consequence of ongoing antigen presentation or antigen-specific CD8+ T-cell proliferation in the airways
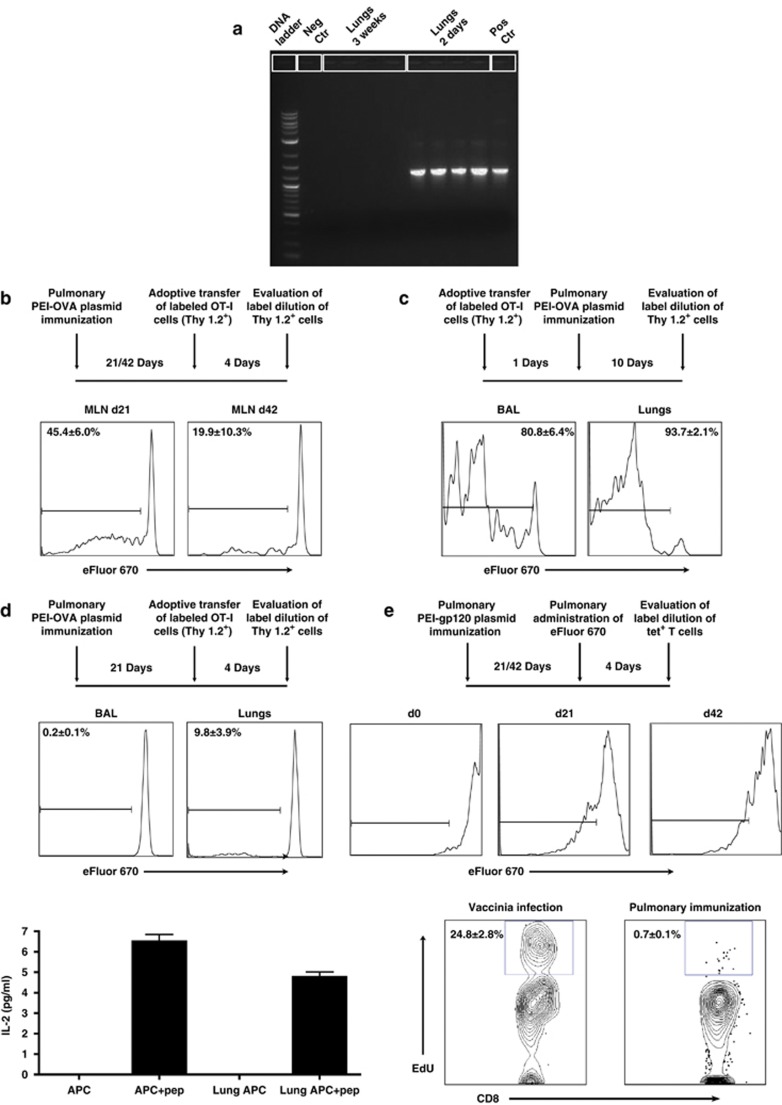
We then explored the mechanism underlying the persistence of antigen-specific CD8+ T cells in the lungs and airways following pulmonary PEI-DNA immunization. We first assessed how long plasmid DNA was present in the lungs following immunization as this would have a direct impact on the duration of antigen presentation. PCR analysis of lungs from immunized mice did not detect plasmid DNA 3 weeks following immunization and showed that plasmid DNA was not retained in the lungs to account for the prolonged presence of CD8+ T cells (Figure 2a). Next, we examined whether the persistence of antigen-specific CD8+ T cells was a consequence of ongoing local antigen presentation. To address this question, we evaluated the duration of antigen presentation in the draining lymph nodes of the lungs. We immunized B6-PL (Thy1.1+) mice by the pulmonary route with PEI-DNA-OVA. Three and 6 weeks postimmunization, CD8+ T cells purified from naive OT-I (Thy1.2+) mice were labeled with eFluor 670 and adoptively transferred by the intravenous route into the immunized mice. We reasoned that if presentation of the vaccine antigen was ongoing, the antigen-specific transferred cells would react by proliferating, and we would detect this proliferation by measuring eFluor 670 fluorescence decay. As shown in Figure 2b, antigen presentation to the donor OT-I cells in the MLN decreased over time, but could be detected for up to 6 weeks. This suggested that there was persistent presentation of antigen in the draining lymph nodes following pulmonary DNA immunization, long after the antigen was cleared.9 As antigen presentation could also be mediated in the lung by resident DCs, we evaluated whether there was persistent presentation in the airways and lung parenchyma. Labeled OT-I cells (Thy1.2+) were transferred intratracheally to B6-PL (Thy1.1+) mice and cell proliferation was measured as an indicator of antigen presentation. We detected antigen presentation in the airways and lung parenchyma 10 days following immunization (Figure 2c); however, by day 21 antigen presentation was no longer detected in this anatomic compartment (Figure 2d). To directly evaluate antigen presentation by cells in the lung compartment, we used a major histocompatibility complex class I presentation assay. Single-cell suspensions from lungs of pulmonary immunized mice were depleted of CD4/CD5/CD8 and co-cultured with a T-cell hybridoma that secretes IL-2 upon recognition of the OVA CD8+ T-cell–specific SIINFEKL peptide presented on antigen-presenting cells (APCs). This would directly measure whether SIINFEKL is still presented in the lung compartment by APCs. There was no detectable antigen presentation in the lungs of pulmonary immunized mice 6 weeks following immunization although APCs from those animals were capable of presenting when peptide was present (Figure 2d). This demonstrates that antigen presentation is not sustained in the lungs or airways long term. Phenotypic analysis of OVA-specific CD8+ T cells isolated from broncho-alveolar lavage (BAL) 6 weeks following immunization presented low expression of CD11a and CD103 (see Supplementary Table S1 online). As antigen-specific CD8+ T cells lose CD11a when they reach the airways and CD103 is expressed during sustained antigen presentation, these findings support our observations of persistence of antigen-specific CD8+ T cells in the airways in the absence of local ongoing antigen presentation.
Figure 2.
Persistence of pulmonary CD8+ T cells is independent of the duration of antigen presentation in the airways and lungs or local T-cell proliferation. (a) Lungs were harvested from B6 mice 2 days and 3 weeks post pulmonary PEI-DNA-OVA immunization and plasmid DNA isolated from the tissue. The OVA sequence was amplified from plasmid DNA by PCR. The plasmid DNA-OVA used for immunization served as positive control (pos ctr) while plasmid DNA without the OVA insert served as the negative control (neg ctr). (b) B6.PL (Thy 1.1+) mice were immunized by the pulmonary route with PEI-DNA-OVA and 21 and 42 days later, received adoptively transferred labeled OT-I cells (Thy 1.2+) by intravenous injection. The dilution of eFluor 670 in donor Thy1.2+ tetramer+ CD8+ T cells was evaluated in mediastinal lymph nodes (MLN) of the immunized mice 4 days post transfer. Representative histograms show the average percentages±s.e. of proliferating Thy1.2+ tetramer+ CD8+ T cells (3–4 mice per group). (c) Labeled OT-I cells (Thy 1.2+) were adoptively transferred to the airway lumen of B6.PL mice (Thy 1.1+). One day later, mice were immunized by the pulmonary route with PEI-DNA-OVA, and 10 days later, donor tetramer-binding CD8+ T cells were isolated from the BAL and lungs, and evaluated for the dilution of eFluor 670 staining. (d) B6-PL (Thy 1.1+) mice were immunized by the pulmonary route with PEI-DNA-OVA and 21 days later received adoptively transferred labeled OT-I cells (Thy 1.2+) in their airway lumens. The dilution of eFluor 670 in donor Thy1.2+ tetramer+ CD8+ T cells was evaluated 4 days later in BAL and lungs. Representative histograms show the average percentages±s.e. of proliferating Thy1.2+ tetramer+ CD8+ T cells (6–8 mice per group). Local antigen presentation was also evaluated in lungs of B6 mice immunized by the pulmonary route with PEI-DNA-OVA. Lungs were harvested 6 weeks following immunization and co-cultured with the RF.33.70 hybridoma overnight. The levels of IL-2 secretion from the RF33.70 hybridoma in response to SIINFEKL presentation were determined using a mouse IL-2 enzyme-linked immunosorbent assay. Bars represent the average±s.e. of secreted IL-2 (3 mice per group). (e) Mice were inoculated by the pulmonary route with PEI-DNA-gp120, and 3 and 6 weeks later eFluor 670 was applied to the airways to stain local resident T cells. Four days later, the dilution of eFluor 670 staining of tetramer+ CD8+ T cells in the BAL was assessed. Histograms representative of 3–8 mice per time point are shown. Proliferation of antigen-specific CD8+ T cells was also evaluated in the BAL of immunized Balb/c mice 6 weeks following pulmonary PEI-DNA-gp120 immunization. Mice were injected with 250 μg 5-ethynyl-2′-deoxyuridine (EdU) intraperitoneally 12 h before sacrifice. Cells isolated from the BAL were stained with monoclonal antibodies to CD4 and CD8, fixed, permeabilized, and EdU detected with the Alexa Fluor 647 Click-iT EdU flow cytometry assay kit. Representative plot show the average percentages±s.e. of EdU incorporation by BAL CD8+ T cells (3 mice per group). BAL CD8+ T cells from mice infected intranasally with rVac-gp160 were used as a positive control for local proliferation. APC, antigen-presenting cells; BAL, broncho-alveolar lavage; ctr, control; IL-2, interleukin 2; pep, peptide; PEI-DNA-OVA, polyethyleneimine-DNA-Ovalbumin complexes.
The persistence of antigen-specific CD8+ T cells in the airways following pulmonary immunization may also be a consequence of local cell proliferation. To assess local airway CD8+ T-cell proliferation, Balb/c mice were immunized by the pulmonary route with PEI-DNA-gp120, and 3 or 6 weeks later eFluor 670 was applied directly to the airways. Four days following this regional staining, proliferation of antigen-specific CD8+ T cells was measured. We found that airway CD8+ T cells did not expand 3 and 6 weeks following immunization. Rather, they underwent only 1–2 rounds of proliferation. This could be a consequence of local T-cell turnover rather than clonal expansion (Figure 2e). The cell division of airway antigen-specific CD8+ T cells was further evaluated using the 5-ethynyl-2′-deoxyuridine (EdU) assay that directly measures DNA synthesis. Airway CD8+ T cells from mice infected intranasally with vaccinia virus proliferated 6 days following infection and were used as a positive control for the assay. Airway CD8+ T cells from pulmonary PEI-DNA-gp120 immunized mice did not show significant levels of proliferation at 6 weeks following immunization. These findings are consistent with the conclusion that antigen presentation is not sustained in the lungs or airways long term. Collectively, these data suggest that the persistence of antigen-specific CD8+ T cells in the airways is not due to ongoing antigen presentation in the airways or local expansion of resident cells; rather, this population is either comprised of long-lived tissue-resident CD8+ cells or is maintained by persistent antigen presentation in the draining MLN.
Pulmonary PEI-DNA immunization confers immune protection against a respiratory virus challenge
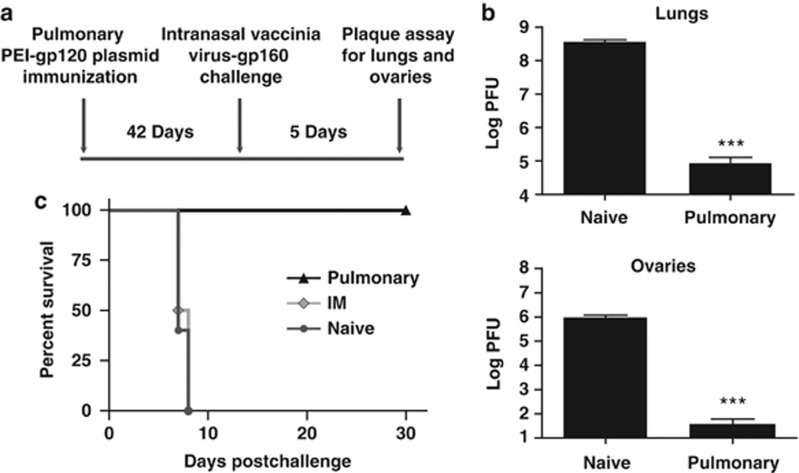
We then determined whether a single pulmonary administration of PEI-DNA generated protective immunity against an upper airway viral infection. Mice were immunized by the pulmonary route with PEI-DNA-gp120 and challenged by the intranasal route 6 weeks after immunization with 2 × 106 PFU rVac-gp160, 10 times the LD50 of this virus in Balb/c mice (Figure 3a). Lungs and ovaries from challenged mice were harvested and analyzed for viral load using a vaccinia virus plaque assay. As shown in Figure 3b, viral titers in the lungs and ovaries of these immunized mice were significantly lower than the titers in lungs and ovaries of the challenged naive mice (P<0.001). To determine whether viral load was associated with the consequences of infection, we monitored the survival following challenge of naive mice and the mice immunized by IM and pulmonary routes. Unvaccinated mice and mice immunized by the IM route mice succumbed to infection 8 days following challenge, whereas 100% of the mice immunized by the pulmonary route were protected against intranasal recombinant vaccinia virus challenge (Figure 3c). These data demonstrate that while immunization by the IM route induces no protective immunity against this challenge, immunization with a single pulmonary inoculation of PEI-DNA provides protection against a lethal intranasal challenge with vaccinia virus.
Figure 3.
Pulmonary PEI-DNA immunization protects against intranasal vaccinia virus challenge. (a) Mice were primed by intramuscular (IM) inoculation with 40 μg DNA-gp120 or pulmonary inoculation of PEI-DNA-gp120 and challenged 6 weeks later by intranasal instillation of a lethal dose (2 × 106 PFU) of vaccinia virus-gp160. Protective immunity was monitored by evaluating (b) viral titers in the lungs and ovaries, and (c) mouse survival. Statistically significant differences between groups were determined using the Student's t-test (***P<0.001). Data represent means of 4–9 mice per group±s.e. PEI, polyethyleneimine; PFU, plaque-forming units.
Protective immunity following PEI-DNA immunization by the pulmonary route is mediated by CD8+ T cells
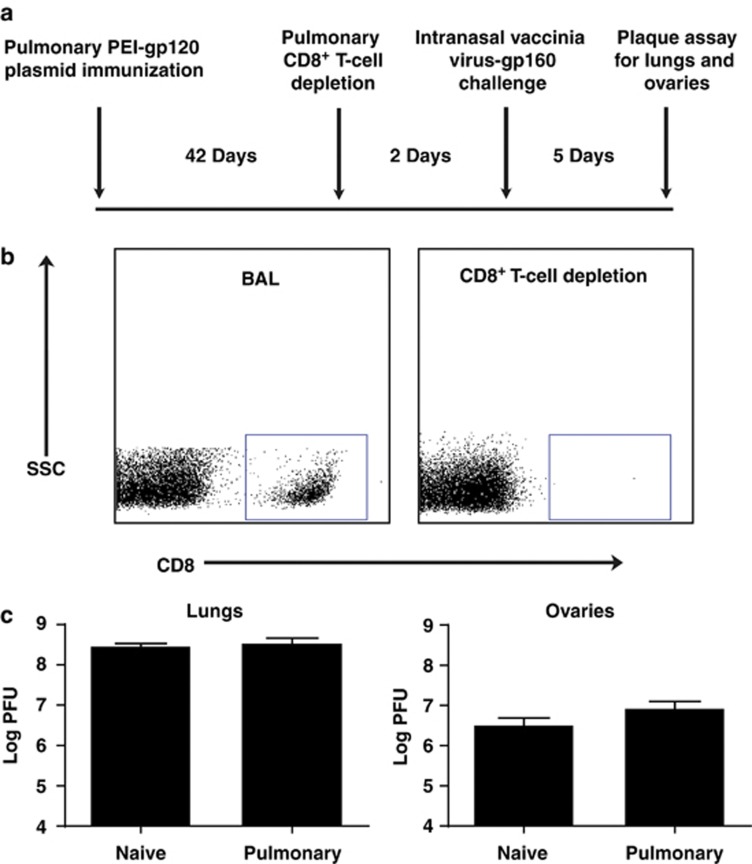
Pulmonary T cells can have a key role in protective immunity against pathogens by eliminating the pathogens at the site of entry, before they disseminate throughout the body. In a previous study, we were able to recover antigen-specific CD8+ T cells from the pulmonary airways long after antigen expression from the DNA vaccine was no longer detectable.9 We therefore hypothesized that these cells contributed to the protection we observed following pulmonary immunization. To test this hypothesis, mice were immunized by the pulmonary route with PEI-DNA-gp120 and 6 weeks later their airway CD8+ T cells were depleted using a monoclonal anti-CD8 antibody administered by the pulmonary route. Two days following anti-CD8 antibody treatment, mice were intranasally challenged with 2 × 106 PFU rVac-gp160, and 5 days later viral load was analyzed in their lungs and ovaries (Figure 4a). To confirm the adequacy of the CD8+ T-cell depletion, BAL from the immunized mice that were treated with the anti-CD8 monoclonal antibody were analyzed for the presence of cells stained with an anti-CD8 antibody (Figure 4b). No CD8+ cells were found, indicating that administration of the depleting monoclonal antibody to the airways results in efficient depletion of CD8+ T cells from the pulmonary compartment (Figure 4b and data not shown). Viral loads in the lungs and ovaries of CD8+ T-cell–depleted, immunized mice were comparable with those of unvaccinated mice (Figure 4c). These findings provide evidence that protection against intranasal recombinant vaccinia virus challenge following pulmonary PEI-DNA immunization is mediated by CD8+ T cells.
Figure 4.
Protective immunity following pulmonary PEI-DNA-gp120 immunization is mediated by CD8+ T cells. (a) Mice were inoculated by the pulmonary route with PEI-DNA-gp120 and 6 weeks later CD8+ T cells were depleted by pulmonary instillation of depleting antibodies. Mice were challenged 2 days later by intranasal instillation of a lethal dose (2 × 106 PFU) of vaccinia virus-gp160. Tissue viral titers were evaluated 5 days post-challenge. (b) Depletion of CD8+ T cells in BAL of immunized mice. (c) Vaccinia virus-gp160 titers in lungs of CD8+ T-cell–depleted mice. Data represent means of 3–5 mice per group±s.e. BAL, broncho-alveolar lavage; PEI-DNA-gp120, polyethyleneimine-DNA-gp120 complexes; PFU, plaque-forming units; SSC, side scatter.
Resident pulmonary CD8+ T cells can protect mice against an intranasal viral challenge independent of peripheral T-cell recruitment
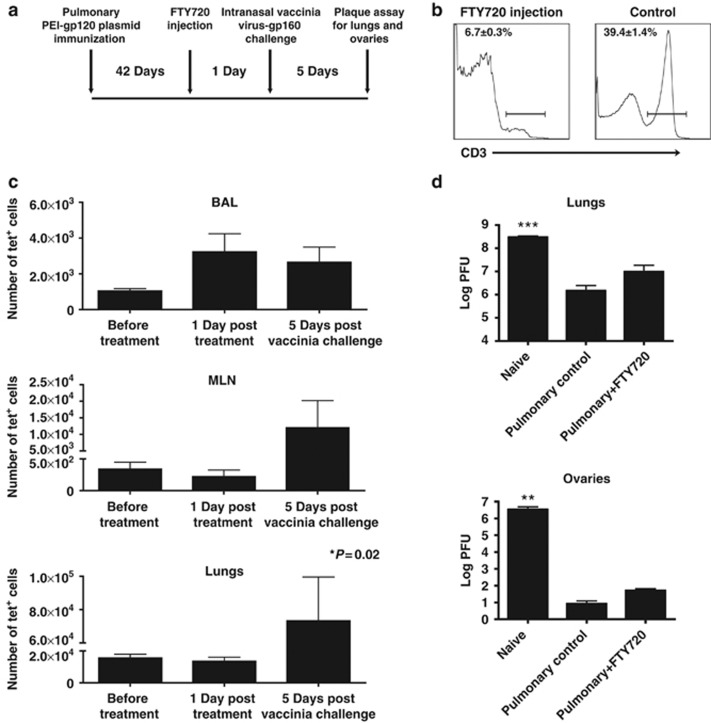
The present findings indicated that pulmonary PEI-DNA immunization generated protective immunity against an intranasal viral challenge, and this protection was associated with the persistence of CD8+ T cells in the pulmonary compartment long after immunization. We sought to determine whether protection against an intranasal recombinant vaccinia virus infection was solely mediated by airways resident CD8+ T cells, or whether recruitment of peripheral T cells into the lungs is necessary for clearing the infection. To explore this issue, we used FTY720 (Fingolimod) treatment, which has been shown to block T-cell trafficking from both lymphoid organs and non-lymphoid tissues.11 This treatment allowed us to isolate the CD8+ T cells in the airways and evaluate whether they were sufficient to protect against infection in the absence of an influx of T cells from the periphery and the lung parenchyma. Balb/c mice were immunized by the pulmonary route with PEI-DNA-gp120 and 6 weeks later were injected with FTY720 to block lymphocyte trafficking (Figure 5a). One day following FTY720 administration, mice were challenged by the intranasal route with 2 × 106 PFU rVac-gp160, and lungs and ovaries were harvested 5 days later for viral load analysis. To assess the drug's effect, we measured the levels of circulating T cells and antigen-specific CD8+ T cells in the mucosal compartment before challenge. FTY720 administration resulted in a peripheral blood lymphopenia, consistent with an efficacious treatment (Figure 5b). There were no significant differences in the numbers of tetramer-positive CD8+ T cells in the lungs, MLN, and BAL following FTY720 treatment (Figure 5c). Despite the treatment-induced blockade of the peripheral T-cell supply, immunized animals were protected against intranasal recombinant vaccinia virus challenge (Figure 5d). The viral loads in the lungs and ovaries of mice immunized by the pulmonary route with PEI-DNA that were treated or not treated with FTY720 were significantly lower than the viral loads in these organs in unvaccinated mice. Importantly, there were no significant differences in the viral titers of the immunized mice that received the FTY720 treatment and the mice that did not receive the treatment. Importantly, the numbers of tetramer-specific CD8+ T cells in the BAL following vaccinia virus infection remained similar to what they were before infection, indicating that there was no local recruitment of cells from the lung parenchyma (Figure 5c). These data suggest that antigen-specific CD8+ T cells in the airways induced by pulmonary PEI-DNA immunization can mediate immune protection against a viral challenge without requiring recruitment of additional lymphocytes from the periphery.
Figure 5.
Protection following pulmonary DNA immunization is independent of the peripheral T-cell supply. (a) Mice were immunized by the pulmonary route with PEI-DNA-gp120, and 6 weeks later the mice were treated with FTY720. The following day the mice were challenged by intranasal instillation of a lethal dose (2 × 106 PFU) of vaccinia virus-gp160. Five days later, lungs and ovaries were harvested and assessed for viral titers. (b) The average percentages ± s.e. of circulating T cells in the blood of 5–7 mice per group with or without FTY720 treatment. (c) The total numbers of tetramer-positive CD8+ T cells (mean±s.e.) before FTY720 treatment, 1 day post treatment and 5 days following intranasal vaccinia virus-gp160 challenge. (d) Viral titers in the lungs and ovaries of unimmunized, pulmonary immunized, and pulmonary immunized+FTY720-treated mice that were challenged intranasally with vaccinia virus-gp160. Data represent means of 3–5 mice per group±s.e. BAL, broncho-alveolar lavage; MLN, mediastinal lymph nodes; PEI-DNA-gp120, polyethyleneimine-DNA-gp120 complexes; PFU, plaque-foming units.
Antigen-specific CD8+ T cells induced by pulmonary PEI-DNA immunization protect against an influenza virus challenge
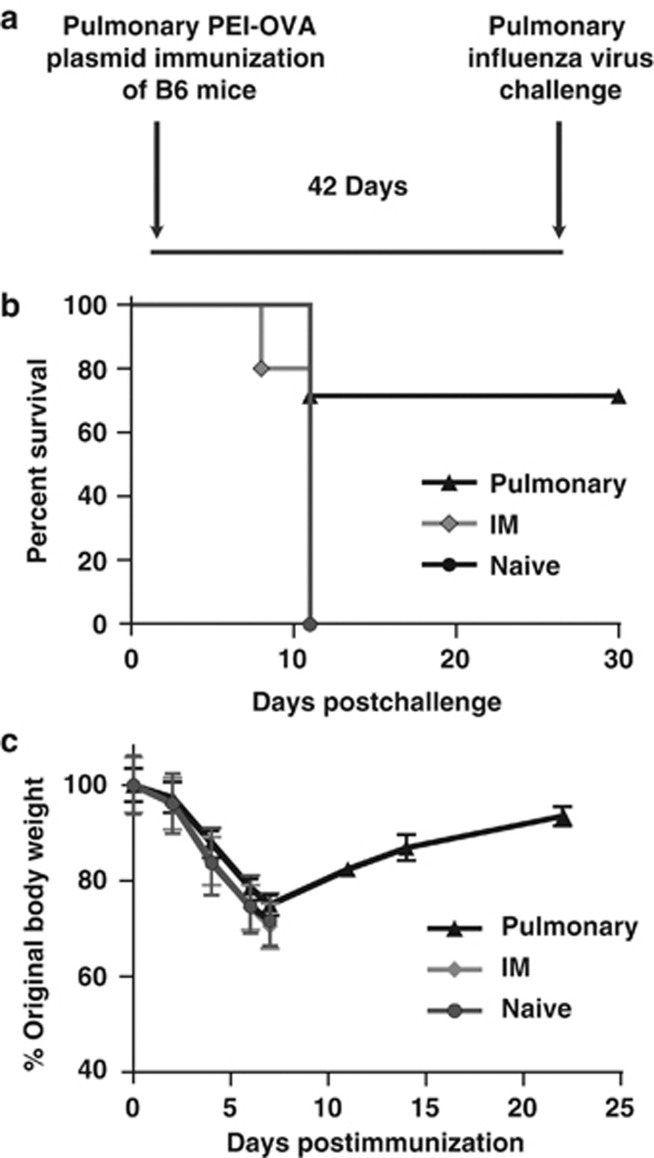
Finally, we assessed the protection conferred by pulmonary immunization with PEI-DNA against influenza virus. We immunized mice by the pulmonary route with PEI-DNA-OVA, and 6 weeks later challenged the vaccinated mice by the intranasal route with 105 EID50 of PR8 influenza virus encoding the OVA257–264 (SIINFEKL) epitope (Figure 6a). This challenge system allowed us to assess protective immunity against influenza and also determine whether the protection was CD8+ T-cell mediated, as only SIINFEKL-specific CD8+ T cells could contribute to protection against infection with this recombinant virus. As shown in Figure 6b, the influenza virus challenge was lethal in unvaccinated mice. Likewise, the challenge was lethal in all mice immunized by the IM route. However, we observed significant protection in the mice immunized by the pulmonary route after challenge with influenza virus with a 71% survival rate. Mice immunized by the pulmonary route lost 25% of their body weight by day 7 post challenge, but regained weight back to 94% of their starting weight by day 22 (Figure 6c). These findings provide further evidence to support the potential utility of pulmonary PEI-DNA immunization for inducing CD8+ T-cell immune responses for protection against respiratory pathogens.
Figure 6.
CD8+ T-cell immunity induced by pulmonary DNA immunization protects against influenza virus challenge. (a) C57BL/6 mice were immunized by the pulmonary route with PEI-DNA-OVA, and 6 weeks later they were infected intranasally with 105 EID50 PR8-SIINFEKL influenza virus. (b) Protective immunity was assessed by the survival rates of mice post infection and (c) mouse weight loss following challenge. Data represent means of 5–7 mice per group±s.e. IM, intramuscular; PEI-DNA-gp120, polyethyleneimine-DNA-gp120 complexes.
Discussion
In these studies, we show that pulmonary PEI-DNA immunization induced protective immunity against viral pathogens that initiate infections via the pulmonary route. This protection was mediated by long-lived memory CD8+ T-cell populations residing within the airways. These persistent effector-memory CD8+ T cells are similar to those observed in the airways after influenza and respiratory syncytial virus infections are resolved.2, 12 Although their mechanism of action has not been clearly defined, memory CD8+ T cells in the airways have been shown to be poorly cytolytic and mediate protective cellular immunity by controlling viral load during the early stages of infection, possibly through secretion of cytokines such as interferon-γ.13, 14
Although the persistence of pathogen-specific CD8+ T cells in the airways has been shown following a number of pulmonary infections and vaccination,2, 4, 15 the mechanisms responsible for their maintenance in the airways has been a subject of much debate and investigation. Persistence of memory CD8+ T cells in the airways could be a consequence of prolonged local antigen presentation, local homeostatic proliferation, or recruitment of memory cells from the circulation.16 We sought to define which mechanisms are responsible for the persistence of antigen-specific CD8+ T cells in the airways following pulmonary PEI-DNA immunization. We showed that despite a brief duration of vaccine antigen presentation in the airways, we detected prolonged antigen presentation in draining lymph nodes. This finding is consistent with the presence of residual antigen in the draining lymph nodes, even though we could not detect antigen expression for more than 5 days following pulmonary PEI-DNA delivery using the in vivo imaging system (IVIS) technology9 or after 3 weeks using PCR. In fact, persistent antigen presentation has been shown following acute viral infections and is thought to contribute to sustained immune responses.17, 18 The priming location of the antigen-specific CD8+ T cells could also affect the maintenance of these cells in the airways and pulmonary-primed memory CD8+ T cells can be reactivated by residual antigen in the MLN and are preferentially recruited to the airways.19 Our data suggests that processed T-cell antigens in the MLN are retained following pulmonary PEI-DNA immunization without plasmid DNA expression, although it is possible that small amounts of plasmid DNA (below the limits of detection in our assays) have been retained in some tissues or cells such as macrophages and dendritic cells. We did not observe the proliferation of antigen-specific (OT-I) CD8+ T cells when adoptively transferred at 6 weeks following pulmonary PEI-DNA immunization. Rather, we observed 1–2 cycles of CD8+ T-cell proliferation, consistent with homeostatic turnover of these cells. This type of proliferation has been described in respiratory virus-induced CD8+ T cells and is a well-characterized mechanism for the maintenance of peripheral T-cell memory.20 This finding highlights the difference between immunization using plasmid DNA and recombinant adenovirus constructs, as the latter was recently shown to generate renewable airway luminal memory CD8+ T cells.21
The presence of memory CD8+ T cells in tissues at sites of pathogen entry can provide for an effective first line of defense against infections. Consistent with this idea, we observed significantly lower viral loads in the lungs and ovaries of mice immunized with PEI-DNA via the pulmonary route than IM immunized and unimmunized mice. More importantly, the decrease in viral loads we observed translated into host protection; 100% of the animals immunized with PEI-DNA-gp120 via the pulmonary route survived an intranasal challenge with a lethal dose of rVac-gp160, whereas unimmunized and IM immunized mice did not survive past day 8 post challenge. Our data indicates that memory CD8+ T cells in pulmonary PEI-DNA-immunized mice mediated this protection, presumably by limiting respiratory infection at the site of virus entry and blocking significant spread of the virus to systemic compartments, as suggested by the low viral titers in the ovaries. The protective efficacy of pulmonary immunization with PEI-DNA was also confirmed in an influenza virus challenge model in which pulmonary PEI-DNA immunized mice had higher survival rates following infection than unimmunized and IM immunized mice. It was reported that memory T cells in the lungs are exclusively of an effector-memory phenotype both during and following infection, and can be effective at protecting the lung mucosa from infection.22, 23 Our findings suggest that PEI-DNA complexes are an efficient immunization platform for generating sustainable memory CD8+ T cells that are protective against respiratory virus infections.
Having demonstrated that pulmonary PEI-DNA immunization generated CD8+ T cells in the pulmonary compartment that can mediate protection against respiratory viral challenge, we sought to establish whether those cells were sufficient for the observed protection, or if recruitment of effector memory CD8+ T cells from the circulation was required. When systemic T-cell trafficking was blocked by FTY720 treatment, and inhibited recruitment of lung-resident antigen-specific CD8+ T cells into the airways (Figure 5), we observed no diminution in immune protection against intranasal rVac-gp160 challenge in mice that received PEI-DNA via the pulmonary route. These data suggest that vaccine-induced airways memory CD8+ T cells have an immediate effector function and are present in the airways in sufficient numbers to protect against a pulmonary viral infection without cell recruitment from the circulation. These data also demonstrate that a non-viral vector can generate effective effector CD8+ T cells in the pulmonary compartment that can mediate immune protection against pulmonary viral infection long after antigen is cleared.
Data suggest that DNA vaccines can be formulated in novel ways to maximize the magnitude and duration of antigen expression in vivo. PEI has been shown to be an efficient and well-tolerated transfection agent for local and systemic administration.24, 25, 26 We previously showed that although PEI enhanced DNA expression in the lungs following pulmonary administration, gene expression was short-lived.9 Despite this brief duration of antigen expression in the lungs, this study shows that it was sufficient to induce protective immunity against respiratory viral challenge.
In the present study, we used a non-invasive pulmonary aerosol administration of the vaccine immunogen and formulated the DNA vaccine with the cationic polymer PEI. There is an ongoing debate in the literature as to whether direct lung immunization is superior to intranasal immunization. The latter vaccine delivery approach has been extensively studied, is an easy method of vaccine delivery, and is clinically approved for influenza immunization in humans. Many studies have shown that intranasal immunization protects against infections.21, 27, 28 However, a recent study by Song et al.29 has demonstrated that intranasal delivery of recombinant adenovirus is less effective in generating humoral and cellular immune responses in the pulmonary airways of nonhuman primates than aerosol delivery. The results of the present study complements those experiments in arguing that depositing a vaccine immunogen directly in the lungs may be the most efficient way to induce protective immunity in the pulmonary mucosa.
Methods
DNA vectors. The codon-optimized HIV-1 HXB2 env and the OVA genes were cloned separately into the VRC vector (DNA-gp120 and DNA-OVA, respectively). The empty VRC vector was provided by Dr G. Nabel (Vaccine Research Center, National Institute of Allergy and Infectious Diseases, National Institutes of Health). Plasmids were prepared using the Endo-free plasmid Giga kit (QIAGEN, Valencia, CA) and had endotoxin levels <0.1 EU μg−1 DNA.
Preparation of PEI-DNA complexes. Plasmid DNA was complexed with in vivo-jetPEI (Polyplus transfection, Illkirch, France) according to the manufacturer's guidelines. Specifically, 0.7 mg ml−1 plasmid DNA solution in 5% glucose was mixed with jetPEI solution to achieve a final DNA concentration of 0.4 mg ml−1 and an N/P ratio of 7.5. The mixture was incubated for at least 30 min at room temperature in order for the complexes to form before being injected into the mice.
Mice and immunizations. Balb/c female mice, 8–12-weeks old, were purchased from Charles River Laboratories (Wilmington, MA). C57BL/6 (B6) mice, B6.PL mice carrying the Thy1.1 allele, and transgenic OT-I mice were purchased from the Jackson Laboratory (Bar Harbor, ME) and maintained under specific pathogen-free conditions. Research on mice was approved by the Beth Israel Deaconess Institutional Animal Care and Use Committee. Mice were immunized IM with 40 μg of DNA in solution in a 100-μl total injection volume (50 μl delivered into each quadriceps muscle). Non-invasive aerosol applications were performed using the technique described by Bivas-Benita et al.30 In short, 50 μl formulation consisting of 40 μg DNA complexed to jetPEI was sprayed directly into the airways of mice with the Penn-Century MicroSprayer (Penn-Century, Wyndmoor, PA). Identical procedures were used for the gp120 and OVA-expressing plasmids.
Lymphocyte isolation for tetramer and phenotypic analysis of T cells. Blood samples from individual mice were collected in RPMI 1640 supplemented with 40 U ml−1 heparin and peripheral blood mononuclear cells were isolated by density gradient centrifugation using Lympholyte-M (Cedarlane, Burlington, NC). Cells collected from the gradient interface were washed with phosphate-buffered saline (PBS) containing 2% fetal bovine serum and stained at room temperature for 20 min with phycoerythrin-conjugated H-2Dd/p18 or SIINFEKL tetramer. Cells were then stained with anti-CD3e-APC-Cy7 (145-2C11) and anti-CD8a-PerCP-Cy5.5 (53-6.7) from BD Biosciences (San Jose, CA) at room temperature for an additional 15 min. Following staining, cells were washed once and fixed with PBS containing 2% paraformaldehyde.
Lungs and MLN were removed aseptically and the airway luminal cells were removed from the lungs by four consecutive BALs through a cannula in a volume of 2 ml PBS to ensure efficient recovery. Following the lavage, lungs and MLN were cut into small pieces using straight scissors and incubated with 1.33 mg ml−1 collagenase-D and 0.2 mg ml−1 DNase (Roche, Indianapolis, IN) in HBSS for 1 hour and 30 min, respectively, at 37 °C with vigorous shaking. Tissue fragments were then crushed through a 70-μm pore size filter and cells were washed once. Lung cells were resuspended in 40% Percoll (Sigma-Aldrich, St Louis, MO), layered over 67% Percoll, and centrifuged at 1,900 r.p.m. for 25 min. The lymphocytes were present in the interface between the two Percoll layers. For tissue tetramer staining, lymphocytes were collected from the gradient interface, washed, and stained as described above for blood samples. MLN samples were washed twice after the filtration step and similarly stained. Samples were analyzed using an LSR II flow cytometer (BD Biosciences, San Jose, CA) and FlowJo software (Tree Star, Ashland, OR).
PCR analysis of plasmid DNA. B6 mice were pulmonary immunized with PEI-DNA-OVA and DNA was isolated from their lungs using the Qiagen DNeasy kit (Qiagen, Valencia, CA). For the PCR reaction, the forward primer (5′-CTCAAAAGACAGCGGCCGCGCCACCATGGGCTCCATCGGCGCAGCAAGCA) and the reverse primer (5′-AGCTTTCGGATCCTTAAGGGGAAACACATCTGCCAAAGAAGAGAACGGCG) were used with the Platinum PCR SuperMix High Fidelity for PCR amplification (Invitrogen, Grand Island, NY). The plasmid DNA-OVA used for immunization served as positive control for the reaction. Each reaction consisted of 45 μl Platinum PCR SuperMix High Fidelity, 200 nm final concentration of each primer solution, and 200 ng genomic DNA template. PCR products were then subjected to agarose gel electrophoresis and 2-log DNA ladder was used to determine the amplicon's mass (New England BioLabs, Ipswich, MA).
In vivo antigen presentation assays. Lymphocytes from spleens and lymph nodes of OT-I mice were washed with PBS and incubated with an equal volume of 10 μm eFluor 670 (eBioscience, San Diego, CA) in PBS for 10 min at 37 °C, at a final concentration of 5 μm. To quench the label, ice-cold RPMI 1640 complete medium was added, incubated on ice for 5 min, and then cells were washed twice with PBS. To evaluate antigen presentation in MLN, 5 × 106 labeled cells in 200 μl were adoptively transferred into B6.PL mice by tail vein injection. For pulmonary evaluation of antigen presentation, cells were resuspended in 50 μl PBS and instilled intratracheally in B6.PL mice. BAL, lungs, and MLN were harvested at different time points and the level of eFluor 670 dilution was determined by flow cytometry using Live/Dead fixable aqua dead-cell stain (Invitrogen), specific phycoerythrin-labeled tetramers, anti-CD3e-APC-Cy7 (145-2C11; BD Biosciences), anti-CD8a-PerCP-Cy5.5 (53-6.7; BD Biosciences), and anti-Thy1.2-eFluor 450 (53-2.1; eBioscience). To assess direct antigen presentation in the lung, lungs were removed and digested as described above. Single-cell suspensions were depleted of T cells by MACS separation using CD4, CD5 and CD8 MicroBeads according to the manufacturer's guidelines (Miltenyi Biotec, Auburn, CA). RF33.70 hybridoma cells (a gift from Dr K. Rock, University of Massachusetts, Worcester, MA), which produce IL-2 upon specific recognition of SIINFEKL presented by APCs, were co-cultured for 24 h with lung APCs in a 96-well plate (5 × 104 cells/well). Supernatants were collected and IL-2 levels were determined by the OptEIA mouse IL-2 ELISA kit (BD Biosciences, San Jose, CA) according to the manufacturer's instructions.
In vivo airway lymphocyte proliferation. For luminal staining of antigen-specific CD8+ T cells, a 25 mm solution of eFluor 670 was prepared in dimethyl sulfoxide. This stock solution was further diluted with 5% glucose solution to a final concentration of 2 mm. Mice immunized with PEI-DNA-gp120 by the pulmonary route were administered 50 μl of the 2 mm eFluor 670 solution using the same non-invasive application method we used for immunization, and eFluor 670 dilution was determined 4 days later. Proliferation was further evaluated using the thymidine analog EdU. Pulmonary immunized animals were administered 250 μg of EdU in PBS intraperitoneally. Mice infected intranasally with replication-competent NYCBH strain vaccinia virus expressing HIV-1 B10 (rVac-gp160) were used as positive control. One day after EdU administration, mice were killed and cells isolated from BAL. Cells were stained with anti-CD8a-PerCP-Cy5.5 (53-6.7; BD Biosciences) and evaluated for incorporated EdU using the Alexa Fluor 647 Click-iT EdU flow cytometry assay kit according to the manufacturer's guidelines (Invitrogen).
In vivo CD8+ T-cell depletion. CD8+ T-cell depletion in mice was accomplished with sterile-purified rat IgG2b anti-mouse CD8 monoclonal antibody (YTS 169.4, Bio-X-cell; West Lebanon, NH). In all, 50 μl of a 2 mg ml−1 antibody solution were sprayed in the lungs of mice by non-invasive pulmonary administration 6 weeks following their immunization. Two days following this treatment, mice were challenged intranasally with 2 × 106 pfu rVac-gp160 and viral titers were evaluated 5 days post challenge in lungs and ovaries using plaque forming assays on CV-1 cells.
Blockage of peripheral T-cell recruitment. Immunized mice were injected i.p. with 200 μl of 0.4 mg ml−1 FTY720 solution (Cayman Chemical, Ann Arbor, MI) to block T-cell recruitment to the lungs. The efficacy of the treatment was confirmed by measuring CD3+ lymphocytes in the peripheral blood of the mice.
Recombinant vaccinia virus challenge. Groups of mice were challenged 6 weeks after a plasmid DNA immunization with replication-competent NYCBH strain vaccinia virus expressing HIV-1 B10 (rVac-gp160). Mice were anesthetized by intraperitoneal injection with a ketamine (100 mg kg−1)/xylazine (10 mg kg−1) mixture and 2 × 106 pfu rVac-gp160 (10 × LD50) was instilled intranasally. Infected mice were monitored for survival and viral titers in the lungs and ovaries. For the evaluation of viral titers, mice were sacrificed 5 days following vaccinia virus challenge and their lungs and ovaries were harvested and frozen at −80 °C until analysis. A day before the assay, tissues were thawed, homogenized in 100 mm Tris buffer, pH=8, and frozen again overnight. On the day of the assay the cells were sonicated for 2 min and viral titers were evaluated by plaque forming assay on CV-1 cells. Each mouse tissue was evaluated in duplo plates to assure reproducibility.
Influenza virus challenge. Antigen-specific CD8+ T-cell responses induced by ovalbumin plasmid immunization were evaluated using the genetically modified influenza strain A/PR/8/34 (PR8, H1N1) encoding the OVA257–264 (SIINFEKL) Kb-restricted epitope in the neuraminidase protein.31 C57BL6 mice were immunized by the pulmonary route with PEI-DNA-OVA and 6 weeks later challenged intranasally with 105 EID50 of the influenza strain.
Statistical analysis. Immune response data are presented as mean±s.e. for each experimental group. Statistical analyses were performed using the two-sided Student's t-test or one-way analysis of variance, and P<0.05 was considered significant.
Acknowledgments
We thank Michelle Lifton, Kathryn Furr, Evita Grant and Ralf Geiben-Lynn for technical assistance. We also extent our gratitude to Dr Michael Santosuosso and Mazal Elnekave for helpful discussions. This work was supported by the NIAID Center for HIV/AIDS Vaccine Immunology grant AI-067854.
The authors declared no conflict of interest.
Footnotes
SUPPLEMENTARY MATERIAL is linked to the online version of the paper at http://www.nature.com/mi
Supplementary Material
References
- Kohlmeier J.E., Woodland D.L. Immunity to respiratory viruses. Annu. Rev. Immunol. 2009;27,:61–82. doi: 10.1146/annurev.immunol.021908.132625. [DOI] [PubMed] [Google Scholar]
- Hogan R.J., et al. Activated antigen-specific CD8+ T cells persist in the lungs following recovery from respiratory virus infections. J. Immunol. 2001;166,:1813–1822. doi: 10.4049/jimmunol.166.3.1813. [DOI] [PubMed] [Google Scholar]
- de Bree G.J., van Leeuwen E.M., Out T.A., Jansen H.M., Jonkers R.E., van Lier R.A. Selective accumulation of differentiated CD8+ T cells specific for respiratory viruses in the human lung. J. Exp. Med. 2005;202,:1433–1442. doi: 10.1084/jem.20051365. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Woodland D.L., Scott I. T cell memory in the lung airways. Proc. Am. Thorac. Soc. 2005;2,:126–131. doi: 10.1513/pats.200501-003AW. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Bivas-Benita M., et al. Pulmonary delivery of chitosan-DNA nanoparticles enhances the immunogenicity of a DNA vaccine encoding HLA-A*0201-restricted T-cell epitopes of Mycobacterium tuberculosis. Vaccine. 2004;22,:1609–1615. doi: 10.1016/j.vaccine.2003.09.044. [DOI] [PubMed] [Google Scholar]
- Lagranderie M., et al. BCG-induced protection in guinea pigs vaccinated and challenged via the respiratory route. Tuber. Lung Dis. 1993;74,:38–46. doi: 10.1016/0962-8479(93)90067-8. [DOI] [PubMed] [Google Scholar]
- Wayne Conlan J., Shen H., Kuolee R., Zhao X., Chen W. Aerosol-, but not intradermal-immunization with the live vaccine strain of Francisella tularensis protects mice against subsequent aerosol challenge with a highly virulent type A strain of the pathogen by an alphabeta T cell- and interferon gamma-dependent mechanism. Vaccine. 2005;23,:2477–2485. doi: 10.1016/j.vaccine.2004.10.034. [DOI] [PubMed] [Google Scholar]
- Hikono H., et al. T-cell memory and recall responses to respiratory virus infections. Immunol. Rev. 2006;211,:119–132. doi: 10.1111/j.0105-2896.2006.00385.x. [DOI] [PubMed] [Google Scholar]
- Bivas-Benita M., et al. Efficient generation of mucosal and systemic antigen-specific CD8+ T-cell responses following pulmonary DNA immunization. J. Virol. 2010;84,:5764–5774. doi: 10.1128/JVI.02202-09. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Takahashi H., et al. Induction of broadly cross-reactive cytotoxic T cells recognizing an HIV-1 envelope determinant. Science. 1992;255,:333–336. doi: 10.1126/science.1372448. [DOI] [PubMed] [Google Scholar]
- Yopp A.C., Ledgerwood L.G., Ochando J.C., Bromberg J.S. Sphingosine 1-phosphate receptor modulators: a new class of immunosuppressants. Clin. Transplant. 2006;20,:788–795. doi: 10.1111/j.1399-0012.2006.00570.x. [DOI] [PubMed] [Google Scholar]
- Ostler T., Hussell T., Surh C.D., Openshaw P., Ehl S. Long-term persistence and reactivation of T cell memory in the lung of mice infected with respiratory syncytial virus. Eur. J. Immunol. 2001;31,:2574–2582. doi: 10.1002/1521-4141(200109)31:9<2574::aid-immu2574>3.0.co;2-v. [DOI] [PubMed] [Google Scholar]
- Kohlmeier J.E., Cookenham T., Roberts A.D., Miller S.C., Woodland D.L. Type I interferons regulate cytolytic activity of memory CD8(+) T cells in the lung airways during respiratory virus challenge. Immunity. 2010;33,:96–105. doi: 10.1016/j.immuni.2010.06.016. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Hogan R.J., Zhong W., Usherwood E.J., Cookenham T., Roberts A.D., Woodland D.L. Protection from respiratory virus infections can be mediated by antigen-specific CD4(+) T cells that persist in the lungs. J. Exp. Med. 2001;193,:981–986. doi: 10.1084/jem.193.8.981. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Gallichan W.S., Rosenthal K.L. Long-lived cytotoxic T lymphocyte memory in mucosal tissues after mucosal but not systemic immunization. J. Exp. Med. 1996;184,:1879–1890. doi: 10.1084/jem.184.5.1879. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Woodland D.L., Kohlmeier J.E. Migration, maintenance and recall of memory T cells in peripheral tissues. Nat. Rev. Immunol. 2009;9,:153–161. doi: 10.1038/nri2496. [DOI] [PubMed] [Google Scholar]
- Turner D.L., Cauley L.S., Khanna K.M., Lefrancois L. Persistent antigen presentation after acute vesicular stomatitis virus infection. J. Virol. 2007;81,:2039–2046. doi: 10.1128/JVI.02167-06. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Zammit D.J., Turner D.L., Klonowski K.D., Lefrancois L., Cauley L.S. Residual antigen presentation after influenza virus infection affects CD8 T cell activation and migration. Immunity. 2006;24,:439–449. doi: 10.1016/j.immuni.2006.01.015. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Takamura S., Roberts A.D., Jelley-Gibbs D.M., Wittmer S.T., Kohlmeier J.E., Woodland D.L. The route of priming influences the ability of respiratory virus-specific memory CD8+ T cells to be activated by residual antigen. J. Exp. Med. 2010;207,:1153–1160. doi: 10.1084/jem.20090283. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Surh C.D., Boyman O., Purton J.F., Sprent J. Homeostasis of memory T cells. Immunol. Rev. 2006;211,:154–163. doi: 10.1111/j.0105-2896.2006.00401.x. [DOI] [PubMed] [Google Scholar]
- Jeyanathan M., et al. Murine airway luminal antituberculosis memory CD8 T cells by mucosal immunization are maintained via antigen-driven in situ proliferation, independent of peripheral T cell recruitment. Am. J. Respir. Crit. Care Med. 2010;181,:862–872. doi: 10.1164/rccm.200910-1583OC. [DOI] [PubMed] [Google Scholar]
- Kohlmeier J.E., Woodland D.L. Memory T cell recruitment to the lung airways. Curr. Opin. Immunol. 2006;18,:357–362. doi: 10.1016/j.coi.2006.03.012. [DOI] [PubMed] [Google Scholar]
- Sallusto F., Geginat J., Lanzavecchia A. Central memory and effector memory T cell subsets: function, generation, and maintenance. Annu. Rev. Immunol. 2004;22,:745–763. doi: 10.1146/annurev.immunol.22.012703.104702. [DOI] [PubMed] [Google Scholar]
- Gautam A., Densmore C.L., Xu B., Waldrep J.C. Enhanced gene expression in mouse lung after PEI-DNA aerosol delivery. Mol. Ther. 2000;2,:63–70. doi: 10.1006/mthe.2000.0087. [DOI] [PubMed] [Google Scholar]
- Demeneix B., Behr J.P. Polyethylenimine (PEI) Adv. Genet. 2005;53,:217–230. [PubMed] [Google Scholar]
- Bonnet M.E., Erbacher P., Bolcato-Bellemin A.L. Systemic delivery of DNA or siRNA mediated by linear polyethylenimine (L-PEI) does not induce an inflammatory response. Pharm. Res. 2008;25,:2972–2982. doi: 10.1007/s11095-008-9693-1. [DOI] [PubMed] [Google Scholar]
- Perrone L.A., et al. Intranasal vaccination with 1918 influenza virus-like particles protects mice and ferrets from lethal 1918 and H5N1 influenza virus challenge. J. Virol. 2009;83,:5726–5734. doi: 10.1128/JVI.00207-09. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Wu T.H., Hutt J.A., Garrison K.A., Berliba L.S., Zhou Y., Lyons C.R. Intranasal vaccination induces protective immunity against intranasal infection with virulent Francisella tularensis biovar A. Infect. Immun. 2005;73,:2644–2654. doi: 10.1128/IAI.73.5.2644-2654.2005. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Song K., et al. Genetic immunization in the lung induces potent local and systemic immune responses. Proc. Natl. Acad. Sci. USA. 2010;107,:22213–22218. doi: 10.1073/pnas.1015536108. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Bivas-Benita M., Zwier R., Junginger H.E., Borchard G. Non-invasive pulmonary aerosol delivery in mice by the endotracheal route. Eur. J. Pharm. Biopharm. 2005;61,:214–218. doi: 10.1016/j.ejpb.2005.04.009. [DOI] [PubMed] [Google Scholar]
- Jenkins M.R., Webby R., Doherty P.C., Turner S.J. Addition of a prominent epitope affects influenza A virus-specific CD8+ T cell immunodominance hierarchies when antigen is limiting. J. Immunol. 2006;177,:2917–2925. doi: 10.4049/jimmunol.177.5.2917. [DOI] [PubMed] [Google Scholar]
Associated Data
This section collects any data citations, data availability statements, or supplementary materials included in this article.