Abstract
One-third of patients with medulloblastoma die due to recurrence after various treatments including radiotherapy. Although it has been postulated that cancer stem-like cells are radio-resistant and play an important role in tumor recurrence, the “stemness” of medulloblastoma cells surviving irradiation has not yet been elucidated. Using a medulloblastoma cell line ONS-76, cells that survived gamma irradiation were investigated on their “stemness” in vitro. From 10 500 cells, 20 radio-resistant clones were selected after gamma ray irradiation (5 Gy × two fractions) using the replica micro-well technique. These 20 resistant clones were screened for CD133 positivity by flow cytometry followed by side population assay, tumor sphere formation assay and clonogenic survival assay. Results revealed CD133 fractions were significantly elevated in three clones, which also exhibited significantly increased levels of tumor sphere formation ability and side population fraction. Clonogenic survival assay demonstrated that their radio-resistance was significantly higher than the parental ONS-76. This may support the hypothesis that a small number of cancer stem-like cells (CSCs) are the main culprits in local recurrence after radiotherapy, and disruption of the resistance mechanism of these CSCs is a critical future issue in improving the outcome of patients with medulloblastoma.
Keywords: medulloblastoma, radiation, CD133, cancer stem-like cell
INTRODUCTION
Medulloblastoma is the most common pediatric central nervous system (CNS) tumor with an incidence of 0.5 per 100 000 children in Japan, and 0.65 per 100 000 in Europe [1]. Approximately 75% of medulloblastomas develop in the cerebellar vermis and it is most commonly seen at ages 6–7 years of age [1]. Although the current standard care for medulloblastoma consists of maximum surgical removal followed by conventional radiotherapy and concomitant chemotherapy [2–3], approximately one-third of patients die due to tumor recurrence [4]. Recurrence occurs not only because of the invasive and metastatic nature of the tumor [2], but also due to the restrictions on radiation doses to the CNS in children [5–7]. Another biological factor leading to treatment failure in one-third of patients may be tumor heterogeneity; a small mingled cell population survives chemo-radiotherapy and regenerates the tumor mass although most of the tumor cells are relatively sensitive to chemo-radiotherapy.
Recently, it has been suggested that the cancer stem-like cells (CSCs) are radio-resistant and may be involved in malignant brain tumor recurrence after radiotherapy [8–11]. Among these, CSCs in medulloblastoma have been reported to be enriched in CD133-positive cells and to have tumor sphere formation ability [12–13]. In addition, it was reported that the side population fraction is closely related to the stemness of medulloblastoma cells through the Notch pathway [14]. Thus, to elucidate the mechanism of recurrence after chemo-radiotherapy in patients with medulloblastoma, it is essential to confirm that the medulloblastoma cells which survive irradiation contain radio-resistant CSCs.
Based on this background, we recreated the process of tumor recurrence after X-ray irradiation in vitro to prove the hypothesis that a small population of survivors is not only radio-resistant but also has the characteristics of CSCs.
MATERIALS AND METHODS
Cell line and culture condition
A human medulloblastoma cell line, ONS-76, was obtained from the RIKEN Cell Bank (Tsukuba, Ibaraki, Japan). ONS-76 was established from a primary culture of tumor tissue taken from a 2-year-old Japanese girl in 1987 [15]. It has been reported that ONS-76 possesses the neuronal cell markers neuron-specific enolase (NSE) and neurofilament protein (NFP) in the cytoplasm. Also, the ONS-76 line expresses class I and II major histocompatibility complex (MHC) following administration of interferon-gamma (IFN-γ). These phenomena suggest that ONS-76 can differentiate into both neural cells and glial cells; thus, ONS-76 may be kept in an undifferentiated or early differentiation state and may have neural stem cell-like properties [15]. Also, we have previously reported that ONS-76 cells form clear colonies with high plating efficiency [16], and a recent report defined that ONS-76 has wild-type p53 [17]. The cells were cultured in minimal essential medium (MEM) (Sigma-Aldrich Inc., Tokyo, Japan) containing 10% fetal bovine serum (FBS) (Nichirei Biosciences Inc., Tokyo, Japan), 100 mg/ml streptomycin and 100 U/ml penicillin (Sigma-Aldrich). The cells were incubated in a humidified atmosphere at 37°C with 5% CO2 in the air. For subcultures, cells were rinsed with Ca2+ and Mg2+-free phosphate buffered saline (PBS) (Sigma-Aldrich), and were dispersed with 0.25% trypsin containing 0.5 mM of ethylenediaminetetraacetate (EDTA) (Sigma-Aldrich).
Irradiation and dosimetry
ONS-76 cells in a logarithmic growing phase were irradiated with 137cesium γ-rays at a dose rate of approximately 0.9 Gy/min using Gammacell (Atomic Energy, Ottawa, ON, Canada). Irradiation was performed at room temperature. Dosimetry at the same position was performed using a small chip photoluminescence glass dosimeter (PLD) system (Dose Ace; Asahi Techno Glass Co., Japan).
Isolation of surviving cells after irradiation
To isolate surviving clones after irradiation, the modified replica micro-well method was performed (Supplemental Fig. 1) [18]. First, ONS-76 cells in flasks were irradiated with 5 Gy of γ-rays. Then, the cells were trypsinized and suspended in MEM, and 350 cells were counted and seeded into thirty 10-cm dishes (Falcon, Becton Dickinson, Franklin Lakes, NJ, USA). After two weeks of incubation, the 96 largest colonies were selected and sampled by custom-made cloning cylinders, and transferred to three identical 96-well plates (Falcon, Becton Dickinson). After incubation for 24 h, one of the three plates was irradiated with 5 Gy of γ-rays again and the three plates were incubated for another 5 days. Finally, the cells in the irradiated plate and one of two unirradiated plates were fixed and stained with 0.25% methylene blue (Wako Pure Chemical Industries Ltd, Osaka, Japan) in 90% ethanol solution. Comparing the two stained plates macroscopically, the deep-colored candidates were selected and harvested from the corresponding wells of the remaining plate.
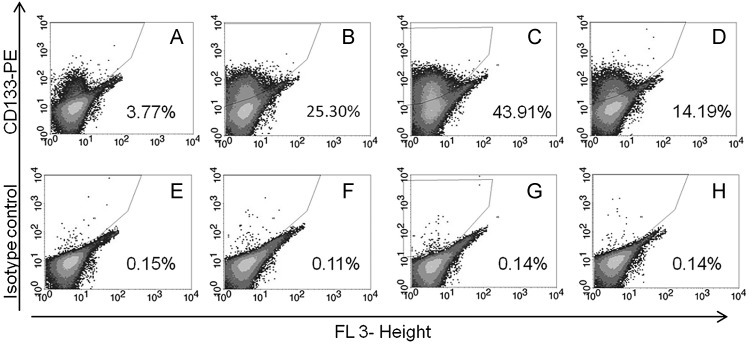
Fig. 1.
Results of CD133 positivity analyses on the selected clones and the parental cells by flow cytometry.
ONS-76 (A), ONS-F8 (B), ONS-B11 (C) and ONS-F11 (D) are results with CD133-PE antibody. ONS-76 (E), ONS-F8 (F), ONS-B11 (G) and ONS-F11 (H) are results with an isotype control antibody.
Immunocytochemistry by flow cytometry
The cells were detached by 0.25% trypsin-EDTA, and washed in PBS containing 2% FBS. Then, 2 × 106 cells were suspended in 80 µl of PBS with 2% FBS, and 20 µl of FcR blocking reagent (Miltenyi Biotec Inc., Tokyo, Japan) and 10 µl of anti-human CD133/1 mouse IgG1 antibody conjugated to phycoerythrin (PE) was added (Miltenyi Biotec). PE-labeled mouse IgG1 (BD Biosciences, Franklin Lakes, NJ, USA) was used as an isotype control. The cell suspension supplemented with these antibodies was incubated at 4°C in the dark. After 30 min of incubation, the cells were washed twice in PBS containing 2% FBS, and then propidium iodide (PI) (Sigma-Aldrich) was added at a final concentration of 1 µg/ml to eliminate dead cells. The treated cells were filtered through a 35-µm cell strainer before flow cytometry (BD FACSCalibur; BD Biosciences).
Tumor sphere limiting dilution assay
For limiting dilution assay, ONS-76 cells were dissociated with 0.25% trypsin-EDTA and suspended in PBS containing 2% FBS. Then, PI was added at a final concentration of 1 µg/ml, and PI-negative cells were sorted by MoFlo (Beckman Coulter Inc., Brea, CA, USA). The selected cells were counted and plated at numbers of 2, 5, 8, 10 and 20 cells per well of the ultra low attachment surface 96-well (Corning Inc., Lowell, MA, USA). The numbers of the cells were plated in 12 wells each of which contained 200 µl of serum-free medium (SFM) composed of Dulbecco's Modified Eagle Medium/Nutrient Mixture F-12 (DMEM/F12) medium (GIBCO, Life Technologies, Carlsbad, CA, USA), 20 ng/ml epidermal growth factor (EGF) (Sigma-Aldrich), 20 ng/ml basic fibroblast growth factor (bFGF) (Sigma-Aldrich) and 20 µl/ml B27 supplement (GIBCO, Life Technologies). During incubation, the cells in the wells were fed with 0.025 ml of SFM on Days 2, 4 and 6. Finally, wells that did not contain spheres were selected macroscopically and their numbers counted on Day 7. The percentage of wells without tumor spheres was calculated and plotted against the number of cells plated per well. From the regression curves, x-intercept values were extrapolated to represent the number of cells required to form at least one tumor sphere in each clone.
Side population analysis
The cells were trypsinized, washed and resuspended at 106 cells/ml in Hank's balanced salt solution (HBSS) (Lonza Walkersville Inc., Walkersville, MD, USA) containing 3% FBS and 10 mM HEPES (GIBCO, Life Technologies). Then, Hoechst 33342 (Dojindo, Kumamoto, Japan) was added at a final concentration of 8 µg/ml with or without 15 µM reserpine (Sigma-Aldrich). Reserpine was used here because it is an adenosine-triphosphate (ATP)-binding cassette (ABC) transporters antagonist. It has been used to evaluate or confirm the presence of side-populations as it is able to selectively extinguish side-populations by its specific function [19]. After incubation at 37°C/5% CO2 for 90 min with gentle agitation every 30 min, cells were washed twice with ice-cold HBSS containing 3% FBS and 10 mM HEPES, followed by and addition of PI to a final concentration of 1 µg/ml. Finally, the cells were filtered through a 35-µm cell strainer (Falcon, Becton Dickinson) to obtain single-cell suspensions, and placed on ice until analysis.
Aliquots of 5 × 105 or more cells were analyzed by MoFlo (Beckman Coulter). The Hoechst 33342 dye was excited by a 350-nm laser, and subjected to dual-wavelength analysis using a 457/50 (Hoechst blue) band-pass filter and 670/30 band-pass filter (Hoechst red). The PI was excited with a 488-nm laser and measured through the 670/30 band-pass filter for discrimination of dead cells.
Clonogenic survival assay
The cells in flasks were exposed to γ-rays with dose values of 1, 2, 4, 6, 8 and 10 Gy. After irradiation, the cells were trypsinized and counted, and the predetermined number of cells was plated onto five 60-mm dishes (Falcon, Becton Dickinson) at each dose point. After 14 days incubation, the colonies were fixed and stained with methylene blue solution 0.25% methylene blue (Wako Pure Chemical Industries) in 90% ethanol solution. The number of surviving colonies that include 50 cells or more was counted and averaged [20]. The survival curves were fitted to the linear-quadratic (LQ) model using DeltaGraph v.5.4 software (RedRock Software, Inc., Salt Lake City, UT, USA) as previously described [16].
Growth pattern and CD133-positive cell fraction analysis
The growth rates and CD133 positivity were analyzed simultaneously. Before the experiment, 1 × 106 cells were plated in T75 flasks (NUNC) and pre-incubated for 3 days. Then the cells were trypsinized and plated at a density of 1 × 106 cells/flask in T75 flasks. The numbers were counted using a Coulter Counter (Beckman Coulter) every 24 h. The ratios of CD133-positive cells were analyzed simultaneously by the above-mentioned method.
Statistical analysis
Experiments were performed three times each. The mean and standard deviations (SD) were calculated at each data point. Student's t test was used to analyze the significance of differences between groups. A probability (P) value of less than 0.05 was regarded as statistically significant. In addition, linear regression analysis and confidence interval estimation were performed on the results of colony survival assay in which the 10% survival values (D10) were obtained and surviving fraction at 2 Gy exposure (SF2) were calculated.
RESULTS
Isolation of surviving cells after irradiation
After γ-ray irradiation with 5 Gy, approximately 10 500 ONS-76 cells in total number were seeded in 30 dishes. Consequently, 1440 colonies (13.7%) were formed after 2 weeks of incubation. From these colonies, 96 large colonies were selected by eye. Then, after another 5 Gy of γ-ray irradiation, 20 clones with high proliferative potency were selected and used in the further analyses (Supplemental Fig. 2 and Supplemental Fig. 4).
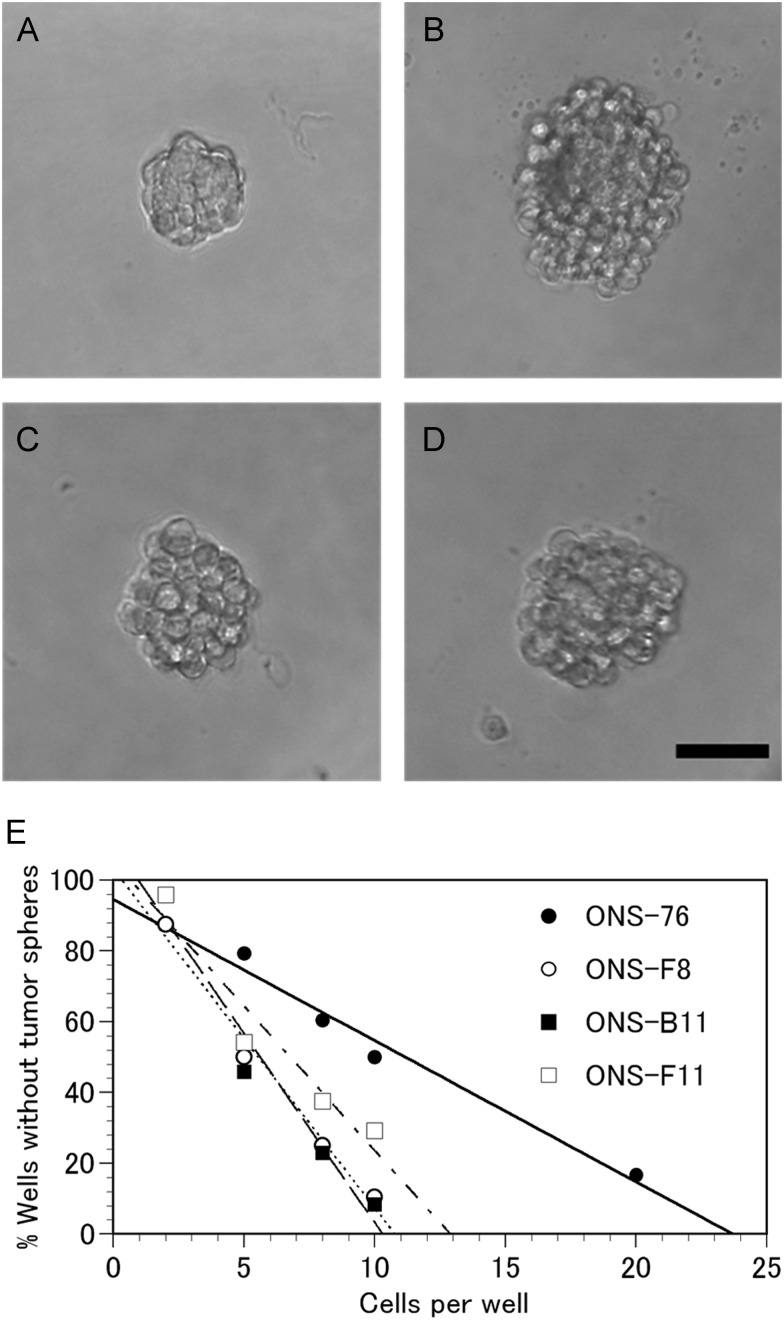
Fig. 2.
Results of the tumor sphere formation assay.
Photographs show tumor spheres from ONS-76 (A), ONS-F8 (B), ONS-B11 (C) and ONS-F11 (D). The black bar in the panel D indicates 300 µm in length. The tumor sphere formation abilities in ONS-76 (closed circles), ONS-F8 (open circles), ONS-B11 (closed squares) and ONS-F11 (open squares) were determined by the limiting dilution assay.
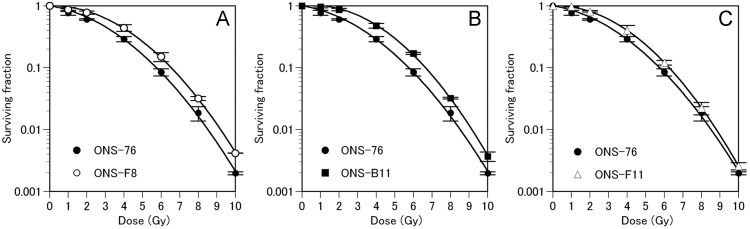
Fig. 4.
Results of the clonogenic survival assay.
(A) Survival curves of ONS-76 (closed circles) and ONS-F8 (open circles). (B) Survival curves of ONS-76 (closed circles) and ONS-B11 (closed circles). (C) Survival curves of ONS-76 (closed circles) and ONS-F11 (open triangles). The survival parameters were calculated by curve fitting with the LQ model. Error bars indicate standard deviation.
Screening of CD133 positivity
The results of CD133 positivity screening in the 20 clones are shown in Table 1. Parental ONS-76 contained 3.77% ± 0.09% CD133-positive cells (Fig. 1 and Table 1). The isolated clones showed a wide variation of CD133 positivities ranging from 0.06% to 43.91% (Table 1). Of these 20 clones, 12 clones showed a decreased CD133 positivity while 8 clones showed an increased CD133 positivity as compared with the parental ONS-76 cell line. Among 8 clones with increased CD133 level, 3 clones, namely ONS-F8, ONS-B11 and ONS-F11, showed significantly higher CD133-positive ratios of 25.30% ± 0.41%, 43.91% ± 2.08% and 14.19% ± 0.75%, respectively (Fig. 1 and Supplemental Fig. 3A). Thus, further analyses were focused on these three clones and the parental ONS-76 cell line.
Table 1.
CD133 positive ratios in 20 isolated clones
| Clones | CD133-positive ratio (%) | Clones | CD133-positive ratio (%) |
|---|---|---|---|
| ONS-76 | 3.77 | ||
| ONS-B11 | 43.91 | ONS-H1 | 0.98 |
| ONS-F8 | 25.30 | ONS-B5 | 0.98 |
| ONS-F11 | 14.19 | ONS-D2 | 0.43 |
| ONS-C7 | 7.21 | ONS-H7 | 0.35 |
| ONS-G8 | 6.34 | ONS-C8 | 0.3 |
| ONS-E10 | 6.29 | ONS-B10 | 0.3 |
| ONS-A6 | 5.85 | ONS-E4 | 0.24 |
| ONS-C1 | 5.35 | ONS-B3 | 0.23 |
| ONS-C10 | 3.46 | ONS-H8 | 0.2 |
| ONS-A10 | 1.06 | ONS-C5 | 0.06 |
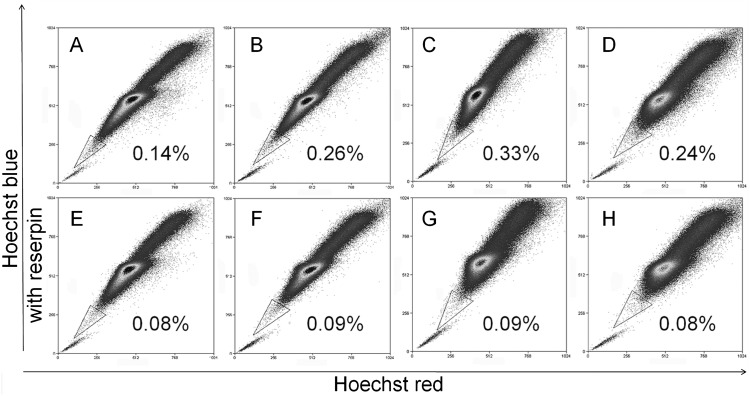
Fig. 3.
Results of the side-population analysis.
ONS-76 (A), ONS-F8 (B), ONS-B11 (C) and ONS-F11 (D) are results of flow cytometry with Hoechst 33342. ONS-76 (E), ONS-F8 (F), ONS-B11 (G) and ONS-F11 (H) are results after treatment with reserpine. Note that the side-population disappeared in E, F, G and H.
Tumor sphere formation ability
The results of tumor sphere formation assay of ONS-76, ONS-F8, ONS-B11 and ONS-F11 demonstrate that the calculated numbers of cells required to form at least one tumor sphere/well were 23.2 ± 0.9 in ONS-76, 10.8 ± 0.6 in ONS-F8, 10.3 ± 0.5 in ONS-B11 and 13.2 ± 2.3 in ONS-F11 (Fig. 2). Statistical analyses demonstrated that three isolated clones with high CD133 positivity have significantly higher tumor sphere formation ability as compared with the parental ONS-76 (Supplemental Fig. 3B).
Percentage of side population cells
As shown in Fig. 3A–D, the percentages of the side population in ONS-76, ONS-F8, ONS-B11 and ONS-F11 were 0.14% ± 0.02%, 0.26% ± 0.02%, 0.33% ± 0.05% and 0.24% ± 0.02%, respectively. In each case, the side population was decreased significantly after treatment with reserpine (15 mg/ml) (Fig. 3E–H). Statistical analyses demonstrated that these three isolated clones had significantly higher side population cell fractions as compared with the parental ONS-76 (Supplemental Fig. 3C).
Clonogenic survival assay
To validate the radio-resistance of the three clones with high CD133 positivity, standard clonogenic survival assay was performed, and the results are shown in Fig. 4. The mean data points for each radiation treatment were curve-fitted by the LQ model. The error bars on the survival curves indicate the SD calculated from three independent experiments. The 10% survival values (D10) were 5.68 ± 0.13 Gy in ONS-76, 6.53 ± 0.15 Gy in ONS-F8, 6.64 ± 0.08 Gy in ONS-B11 and 6.26 ± 0.07 Gy in ONS-F11, and the survival fractions at 2 Gy (SF2) were 0.64% ± 0.03% in ONS-76, 0.82% ± 0.06% in ONS-F8, 0.90% ± 0.05% in ONS-B11 and 0.81% ± 0.07% in ONS-F11 (Table 2). Considering a 95% confidence interval (CI), these three isolated clones had significantly higher SF2 values as compared with the parental ONS-76 (Table 2).
Table 2.
Calculated parameters of survival curves of ONS-76, ONS-F8, ONS-B11 and ONS-F11
| α(Gy−1) ± 95% CI | β(Gy−2) ± 95% CI | R2 | SF2 ± 95%CI | D10 (Gy) ± 95% CI | |
|---|---|---|---|---|---|
| ONS-76 | 0.13 ± 0.03 | 0.05 ± 0.003 | 0.99 | 0.64 ± 0.03 | 5.68 ± 0.13 |
| ONS-F8 | −0.01 ± 0.05 | 0.06 ± 0.005 | 0.99 | 0.82 ± 0.06 | 6.53 ± 0.14 |
| ONS-B11 | −0.07 ± 0.04 | 0.06 ± 0.005 | 0.99 | 0.90 ± 0.05 | 6.64 ± 0.08 |
| ONS-F11 | −0.016 ± 0.02 | 0.06 ± 0.004 | 0.99 | 0.81 ± 0.02 | 6.26 ± 0.07 |
Values of α and β with ±95% confidence interval (CI), and the coefficient of determination (R2) were calculated by curve fitting with linear-quadratic (LQ) model. Values of SF2 indicate surviving fractions at dose of 2 Gy. D10 indicates dose corresponding to 10% surviving fraction. Values of 95% CI were calculated from three independent experiments.
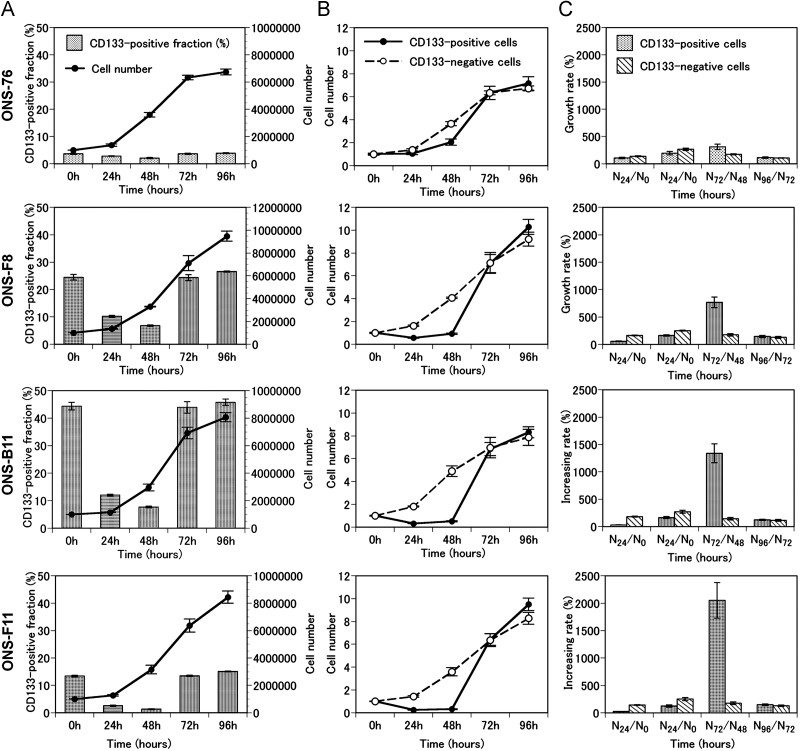
Growth pattern and CD133-positive cell fraction
As shown in Fig. 5A, the numbers of cells of the three clones became greater than that of ONS-76 after 96 h of incubation although their growth patterns were similar. Also, it was shown that compared with CD133-negative cells, the percentage of CD133-positive cells in the three clones decreased from 0 to 48 h, and they increased rapidly from 48 to 72 h (Fig. 5B). The calculated maximum growth rates of CD133-positive cells were 310% ± 50% in ONS-76, 768% ± 96% in ONS-F8, 1338% ± 174% in ONS-B11 and 2053% ± 323% in ONS-F11 (Fig. 5C), which were significantly greater than the growth rates of 300% or less in CD133-negative cells in every cell line.
Fig. 5.
Chronological changes of cell counts and CD133-positive fractions of the selected clones and the parental cells.
(A) Cell counts (line graphs) and CD133-positive fractions (bar graphs) of ONS-76, ONS-F8, ONS-B11 and ONS-F11 were plotted against time. (B) Cell counts of CD133-positive cells (solid lines) and CD133-negative cells (dotted lines) of ONS-76, ONS-F8, ONS-B11 and ONS-F11 were plotted against time. Cell counts were normalized to 1 at 0 h. (C) Increase rates (%) of CD133-positive and CD133-negative cells of ONS-76, ONS-F8, ONS-B11 and ONS-F11at the time-points indicated. Error bars indicate standard deviation.
DISCUSSION
It has been postulated that a very small number of CSCs in tumor tissue survive radiotherapy and lead to tumor regeneration in various malignant tumors [21–22]. However, it has not been clarified whether radio-resistant subpopulations possess stemness in medulloblastoma cells. In this study, we aimed to confirm that medulloblastoma cells that survived γ-ray irradiation contain cells with stem cell-like characteristics in vitro. Thus, we recreated the process of tumor recurrence in vitro using γ-ray irradiation followed by resistant cell selection and characterizing their stemness.
Several attempts have been made to establish radio-resistant cells after exposure to single or fractionated irradiations, and their radio-resistant nature have been analyzed [23–24]. Wei et al. selected radio-resistant cells from human fibrosarcoma HT1080 through exposure to 2 Gy per day, 5 days per week for 7 months. Their radio-resistant cells had different G-banded karyotypes compared with parental cells [23]. Also, Tang et al. established two radio-resistant sublines of human hepatocellular carcinoma HepG2 cells by exposure to 2 Gy of γ-rays for 10 days or 10 Gy for 2 days. They found radio-resistant cells had a greater extent of potentially lethal damage repair (PLDR) and over-expression of Raf-1 [24]. More recently, Kuwahara et al. established radio-resistant cells from a human hepatocellular carcinoma cell line, HepG2, in which the cells were exposed once to 2 Gy of α-particles with boron neutron capture, followed by 0.5 Gy of X-rays at every 12-h interval for more than 6 years associated with 2 Gy of X-rays per day for 30 consecutive days [25–26]. These reported methods were most likely intended to induce mutations by irradiation over a certain period of time to produce stably radio-resistant cells. In contrast, our purpose here was to recreate tumor recurrence after radiotherapy and to evaluate stemness in surviving cells. Thus, we used a modified replica micro-well method to select clones after 5 Gy of γ-irradiation twice, based on our previous data that 5 Gy corresponded to approximately 20% survival in ONS-76 (Supplemental Fig. 1) [16]. Although a comparison between non-irradiated and irradiated clones may demonstrate the effect of irradiation more clearly, the chances of harvesting radio-resistant clones from non-irradiated cells without selection would be very low. Thus we evaluated stemness only on clones isolated by the method described above.
CD133 was originally found as a marker on neural stem-like cells [27–28]. In addition, CD133-positive cells were reported to have strong tumor-initiating and multilineage capacities in glioblastoma and medulloblastoma cells [12–13, 29]. Therefore, CD133 is recognized as one of the reliable markers of CSCs in these tumors. In this study, 20 selected radio-resistant clones expressed CD133 with a range from 0.06 to 44%, and three clones showed predominantly higher levels of CD133 positivity as compared with parental ONS-76 (Table 1 and Fig. 1). In addition, we confirmed that these three clones were stable in maintaining CD133 positivity even after 10 passages. Since it has been reported that CD133-negative cells do not spontaneously become CD133-positive after cell division, and irradiation itself does not induce CD133 expression in CD133-negative tumor cells, these three clones were most likely derived from cells that were originally CD133-positive [8]. Thus, our observation may indicate that these CD133-positive clones have strong self-renewal capacity to produce CD133-positive cells in a symmetric cell division pattern. As for the other 17 clones with CD133-positive ratios ranging from 0.06–7.21%, some of them may possess radio-resistant features due to other mechanisms that are not directly related to stemness, however, further characterization of these clones is out of the scope of the present study.
Other characteristics of CSCs in glioblastoma and medulloblastoma cells are sphere formation ability in optimum media [12, 30], and higher Hoechst 33342 effluxes side-population [19, 31–34]. We demonstrated that the three clones with high CD133-positive ratios also had significantly higher D10 and SF2 values, sphere formation ability and side-population fractions as compared with the parental ONS-76 (Figs 2, 3, 4, Supplemental Fig. 3 and Table 2). These results agree well with the reported data, which have demonstrated that side-population cells were expressing high levels of self-renewal-related genes such as notch and PTEN in brain tumor cells [14, 35]. However, although we performed double staining of CD133 and side-population in FACS analysis, we could not find a close correlation between these in the three isolated radio-resistant clones, indicating that they may contain different subtypes of cells with regard to stemness.
We observed that CD133-positive cells did not proliferate during the first 48 h of the incubation period, and they grew very rapidly thereafter (Fig. 5B). This may demonstrate that CD133-positive cells proliferated by symmetric cell divisions after remaining in the resting (G0) state for a latent period. Furthermore, maximum growth rates of CD133-positive cells in the three isolated clones were significantly higher than those to the parental ONS-76. From these observations, these three isolated clones possess not only self-renewal potency but also potentially high proliferative ability in vitro (Fig. 5C).
As for the mechanism of radio-resistance of CSCs, Bao et al. reported that the expression levels of checkpoint-related proteins such as ATM, Rad17, Chk1 and Chk2 were highly up-regulated in CSCs after irradiation, resulting in accumulation of cells in the cell cycle arrest in order to repair damaged DNA [8]. In addition, Diehn et al. demonstrated that CSCs had lower reactive oxygen species (ROS) levels with less DNA damages, which may suggest that CSCs had high radiation-induced free radical scavenge ability and had lower incidence of radical-induced DNA damages [36]. Also, it has been reported that the self-renewal-related notch pathway was closely associated with increase of the CD133-positive cell fractions and increase of radio-resistance in brain tumor cells [37]. Another self-renewal-related gene, BMI-1, was also reported to enhance radio-resistance in brain tumor cells [38]. These molecular analyses of the mechanism of self-renewal and radio-resistance may add further information to the characterization of the three clones isolated in this study.
To improve tumor control we must initially clarify resistance mechanisms. Elucidating the role of medulloblastoma stem cells in tumor response to radiation will enhance our understanding of the current status of radiotherapy against this pediatric brain tumor, and may help direct our research to achieving a more favorable outcome. Thus, future studies should be aimed at defining a specific method to disrupt the resistance mechanism of CSCs and improve overall tumor control. As mentioned above, the checkpoint kinases or self-renewal related genes may be possible molecular targets. At this point, however, it is mandatory to proceed with studies, not only to clarify the clinical applicability of these molecules, but also to find other novel molecules that are responsible for both stemness and radio-resistance.
In conclusion, we recreated the phenomenon of medulloblastoma recurrence after radiotherapy in vitro, and demonstrated that at least three clones among 20 survivors with high proliferative proficiency after γ-irradiation possess cancer stemness. These results should contribute to elucidating the mechanism of recurrence after radiotherapy in patients with medulloblastoma.
Supplementary Material
ACKNOWLEDGEMENTS
This work was partly supported by a Grant-in-Aid from the Ministry of Education, Culture, Sports, Science & Technology of Japan, and a research fund from the University of Tsukuba, Japan.
REFERENCES
- 1.Peris-Bonet R, Martinez-Garcia C, Lacour B, et al. Childhood central nervous system tumours–incidence and survival in Europe (1978-1997): report from Automated Childhood Cancer Information System project. Eur J Cancer. 2006;42:2064–80. doi: 10.1016/j.ejca.2006.05.009. [DOI] [PubMed] [Google Scholar]
- 2.Oyharcabal-Bourden V, Kalifa C, Gentet JC, et al. Standard-risk medulloblastoma treated by adjuvant chemotherapy followed by reduced-dose craniospinal radiation therapy: a French Society of Pediatric Oncology Study. J Clin Oncol. 2005;23:4726–34. doi: 10.1200/JCO.2005.00.760. [DOI] [PubMed] [Google Scholar]
- 3.Gajjar A, Chintagumpala M, Ashley D, et al. Risk-adapted craniospinal radiotherapy followed by high-dose chemotherapy and stem-cell rescue in children with newly diagnosed medulloblastoma (St Jude Medulloblastoma-96): long-term results from a prospective, multicentre trial. Lancet Oncol. 2006;7:813–20. doi: 10.1016/S1470-2045(06)70867-1. [DOI] [PubMed] [Google Scholar]
- 4.Massimino M, Giangaspero F, Garre ML, et al. Childhood medulloblastoma. Crit Rev Oncol Hematol. 2011;79:65–83. doi: 10.1016/j.critrevonc.2010.07.010. [DOI] [PubMed] [Google Scholar]
- 5.Habrand JL, De Crevoisier R. Radiation therapy in the management of childhood brain tumors. Childs Nerv Syst. 2001;17:121–33. doi: 10.1007/s003810000365. [DOI] [PubMed] [Google Scholar]
- 6.Cervoni L, Cantore G. Medulloblastoma in pediatric age: a single-institution review of prognostic factors. Childs Nerv Syst. 1995;11:80–4. doi: 10.1007/BF00303809. discussion 85. [DOI] [PubMed] [Google Scholar]
- 7.Dennis M, Spiegler BJ, Hetherington CR, et al. Neuropsychological sequelae of the treatment of children with medulloblastoma. J Neurooncol. 1996;29:91–101. doi: 10.1007/BF00165522. [DOI] [PubMed] [Google Scholar]
- 8.Bao S, Wu Q, McLendon RE, et al. Glioma stem cells promote radioresistance by preferential activation of the DNA damage response. Nature. 2006;444:756–60. doi: 10.1038/nature05236. [DOI] [PubMed] [Google Scholar]
- 9.Chiou SH, Kao CL, Chen YW, et al. Identification of CD133-positive radioresistant cells in atypical teratoid/rhabdoid tumor. PLoS One. 2008;3:e2090. doi: 10.1371/journal.pone.0002090. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 10.Blazek ER, Foutch JL, Maki G. Daoy medulloblastoma cells that express CD133 are radioresistant relative to CD133- cells, and the CD133+ sector is enlarged by hypoxia. Int J Radiat Oncol Biol Phys. 2007;67:1–5. doi: 10.1016/j.ijrobp.2006.09.037. [DOI] [PubMed] [Google Scholar]
- 11.Vlashi E, Lagadec C, Vergnes L, et al. Metabolic state of glioma stem cells and nontumorigenic cells. Proc Natl Acad Sci USA. 2011;108:16062–67. doi: 10.1073/pnas.1106704108. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 12.Singh SK, Clarke ID, Terasaki M, et al. Identification of a cancer stem cell in human brain tumors. Cancer Res. 2003;63:5821–8. [PubMed] [Google Scholar]
- 13.Singh SK, Hawkins C, Clarke ID, et al. Identification of human brain tumour initiating cells. Nature. 2004;432:396–401. doi: 10.1038/nature03128. [DOI] [PubMed] [Google Scholar]
- 14.Fan X, Matsui W, Khaki L, et al. Notch pathway inhibition depletes stem-like cells and blocks engraftment in embryonal brain tumors. Cancer Res. 2006;66:7445–52. doi: 10.1158/0008-5472.CAN-06-0858. [DOI] [PubMed] [Google Scholar]
- 15.Tamura K, Shimizu K, Yamada M, et al. Expression of major histocompatibility complex on human medulloblastoma cells with neuronal differentiation. Cancer Res. 1989;49:5380–4. [PubMed] [Google Scholar]
- 16.Gerelchuluun A, Hong Z, Sun L, et al. Induction of in situ DNA double-strand breaks and apoptosis by 200 MeV protons and 10 MV X-rays in human tumour cell lines. Int J Radiat Biol. 2011;87:57–70. doi: 10.3109/09553002.2010.518201. [DOI] [PubMed] [Google Scholar]
- 17.Kunkele A, De Preter K, Heukamp L, et al. Pharmacological activation of the pp53 pathway by nutlin-3 exerts anti-tumoral effects in medulloblastomas. Neuro Oncol. 2012;14:859–69. doi: 10.1093/neuonc/nos115. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 18.Tsuboi K, Tsuchida Y, Endo K, et al. Isolation of radiosensitive and radioresistant mutants from a medulloblastoma cell line. Brain Tumor Pathol. 1997;14:19–25. doi: 10.1007/BF02478864. [DOI] [PubMed] [Google Scholar]
- 19.Ho MM, Ng AV, Lam S, et al. Side population in human lung cancer cell lines and tumors is enriched with stem-like cancer cells. Cancer Res. 2007;67:4827–33. doi: 10.1158/0008-5472.CAN-06-3557. [DOI] [PubMed] [Google Scholar]
- 20.Franken NA, Rodermond HM, Stap J, et al. Clonogenic assay of cells in vitro. Nat Protoc. 2006;1:2315–19. doi: 10.1038/nprot.2006.339. [DOI] [PubMed] [Google Scholar]
- 21.Rich JN. Cancer stem cells in radiation resistance. Cancer Res. 2007;67:8980–4. doi: 10.1158/0008-5472.CAN-07-0895. [DOI] [PubMed] [Google Scholar]
- 22.Visvader JE, Lindeman GJ. Cancer stem cells in solid tumours: accumulating evidence and unresolved questions. Nat Rev Cancer. 2008;8:755–68. doi: 10.1038/nrc2499. [DOI] [PubMed] [Google Scholar]
- 23.Wei K, Kodym R, Jin C. Radioresistant cell strain of human fibrosarcoma cells obtained after long-term exposure to x-rays. Radiat Environ Biophys. 1998;37:133–7. doi: 10.1007/s004110050106. [DOI] [PubMed] [Google Scholar]
- 24.Tang WY, Chau SP, Tsang WP, et al. The role of Raf-1 in radiation resistance of human hepatocellular carcinoma Hep G2 cells. Oncol Rep. 2004;12:1349–54. doi: 10.3892/or.12.6.1349. [DOI] [PubMed] [Google Scholar]
- 25.Kuwahara Y, Li L, Baba T, et al. Clinically relevant radioresistant cells efficiently repair DNA double-strand breaks induced by X-rays. Cancer Sci. 2009;100:747–52. doi: 10.1111/j.1349-7006.2009.01082.x. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 26.Kuwahara Y, Mori M, Oikawa T, et al. The modified high-density survival assay is the useful tool to predict the effectiveness of fractionated radiation exposure. J Radiat Res. 2010;51:297–302. doi: 10.1269/jrr.09094. [DOI] [PubMed] [Google Scholar]
- 27.Coskun V, Wu H, Blanchi B, et al. CD133+ neural stem cells in the ependyma of mammalian postnatal forebrain. Proc Natl Acad Sci USA. 2008;105:1026–31. doi: 10.1073/pnas.0710000105. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 28.Kania G, Corbeil D, Fuchs J, et al. Somatic stem cell marker prominin-1/CD133 is expressed in embryonic stem cell-derived progenitors. Stem Cells. 2005;23:791–804. doi: 10.1634/stemcells.2004-0232. [DOI] [PubMed] [Google Scholar]
- 29.Qiang L, Yang Y, Ma YJ, et al. Isolation and characterization of cancer stem like cells in human glioblastoma cell lines. Cancer Lett. 2009;279:13–21. doi: 10.1016/j.canlet.2009.01.016. [DOI] [PubMed] [Google Scholar]
- 30.Ghods AJ, Irvin D, Liu G, et al. Spheres isolated from 9L gliosarcoma rat cell line possess chemoresistant and aggressive cancer stem-like cells. Stem Cells. 2007;25:1645–53. doi: 10.1634/stemcells.2006-0624. [DOI] [PubMed] [Google Scholar]
- 31.Fukaya R, Ohta S, Yamaguchi M, et al. Isolation of cancer stem-like cells from a side population of a human glioblastoma cell line, SK-MG-1. Cancer Lett. 2010;291:150–7. doi: 10.1016/j.canlet.2009.10.010. [DOI] [PubMed] [Google Scholar]
- 32.Kondo T, Setoguchi T, Taga T. Persistence of a small subpopulation of cancer stem-like cells in the C6 glioma cell line. Proc Natl Acad Sci USA. 2004;101:781–6. doi: 10.1073/pnas.0307618100. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 33.Chiba T, Kita K, Zheng YW, et al. Side population purified from hepatocellular carcinoma cells harbors cancer stem cell-like properties. Hepatology. 2006;44:240–51. doi: 10.1002/hep.21227. [DOI] [PubMed] [Google Scholar]
- 34.Patrawala L, Calhoun T, Schneider-Broussard R, et al. Side population is enriched in tumorigenic, stem-like cancer cells, whereas ABCG2+ and ABCG2- cancer cells are similarly tumorigenic. Cancer Res. 2005;65:6207–19. doi: 10.1158/0008-5472.CAN-05-0592. [DOI] [PubMed] [Google Scholar]
- 35.Bleau AM, Hambardzumyan D, Ozawa T, et al. PTEN/PI3K/Akt pathway regulates the side population phenotype and ABCG2 activity in glioma tumor stem-like cells. Cell Stem Cell. 2009;4:226–35. doi: 10.1016/j.stem.2009.01.007. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 36.Diehn M, Cho RW, Lobo NA, et al. Association of reactive oxygen species levels and radioresistance in cancer stem cells. Nature. 2009;458:780–3. doi: 10.1038/nature07733. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 37.Wang J, Wakeman TP, Lathia JD, et al. Notch promotes radioresistance of glioma stem cells. Stem Cells. 2010;28:17–28. doi: 10.1002/stem.261. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 38.Facchino S, Abdouh M, Chatoo W, et al. BMI1 confers radioresistance to normal and cancerous neural stem cells through recruitment of the DNA damage response machinery. J Neurosci. 2010;30:10096–111. doi: 10.1523/JNEUROSCI.1634-10.2010. [DOI] [PMC free article] [PubMed] [Google Scholar]
Associated Data
This section collects any data citations, data availability statements, or supplementary materials included in this article.