Abstract
Asthma is a disease of airway inflammation that in most cases fails to resolve. The resolution of inflammation is an active process governed by specific chemical mediators, including D-series resolvins. Here, we determined the impact of resolvin D1 (RvD1) and aspirin-triggered resolvin D1 (AT-RvD1) on the development of allergic airway responses and their resolution. Mice were allergen sensitized and RvD1, AT-RvD1 (1, 10 or 100 ng) or vehicle were administered at select intervals before or after aerosol allergen challenge. RvD1 markedly decreased airway eosinophilia and mucus metaplasia, in part by decreasing IL-5 and IκBα degradation. For the resolution of established allergic airway responses, AT-RvD1 was even more efficacious than RvD1, leading to a marked decrease in the resolution interval for lung eosinophilia, decrements in select inflammatory peptide and lipid mediators and more rapid resolution of airway hyper-reactivity to methacholine. Relative to RvD1, AT-RvD1 resisted metabolic inactivation by macrophages and AT-RvD1 significantly enhanced macrophage phagocytosis of IgG-OVA-coated beads in vitro and in vivo, a new pro-resolving mechanism for the clearance of allergen from the airways. In conclusion, RvD1 and AT-RvD1 can serve as important modulators of allergic airway responses by decreasing eosinophils and pro-inflammatory mediators and promoting macrophage clearance of allergen. Together, these findings identify D-series resolvins as potential pro-resolving therapeutic agents for allergic responses.
Keywords: Resolution, asthma, mediators, lung, D-series resolvins, omega-3 fatty acids
Introduction
Asthma is a chronic inflammatory disease of the airways without curative therapy (1). Allergen-induced airway responses are common in asthma and notable for infiltration and accumulation of eosinophils and T lymphocytes in the airways. These leukocytes, as well as lung resident cells, can produce soluble mediators that promote airway inflammation, mucus metaplasia and hyper-responsiveness, which are cardinal features of asthma (1). Most patients with asthma have symptoms that are readily controllable by standard asthma therapies, including β2-adrenergic agonists, low doses of inhaled corticosteroids or leukotriene modifiers (1). However, 5–10% of asthmatic individuals have poorly controlled disease with frequent exacerbations or symptoms that are refractory to current therapy(2).
During inflammation, the essential omega-3 fatty acid docosahexaenoic acid (DHA, C22:6) is available for enzymatic transformation to several anti-inflammatory and pro-resolving mediators, including D-series resolvins (3). Pro-resolving mediators are generated from DHA in asthmatic airways (4) and during experimental allergic airway responses (4, 5), and these DHA-derived mediators can promote clearance of eosinophils and T-cells (4, 6). Epidemiological evidence has uncovered a significant relationship between dietary fish ingestion and a lower prevalence of allergy and asthma (7) and DHA is enriched in cold water fish. In addition, DHA is present in airway mucosa and, in comparison to healthy individuals, mucosal levels of DHA decrease in asthmatic individuals, particularly in relation to the omega-6 fatty acid arachidonic acid (C20:4) (8). Resolvin D1 (RvD1) is enzymatically derived from DHA and has recently been identified to inhibit neutrophil activation (9–11), regulate cytokines (12, 13) and protect after ischemia-reperfusion second organ injury (14, 15) and LPS-induced acute lung injury (16). Here, we determined the actions of RvD1 and its aspirin-triggered epimer AT-RvD1 on the course and resolution of allergic airway responses in a murine experimental model of asthma.
Materials and Methods
Animals
After approval by the institutional review board of Harvard Medical Area (protocol 03618), five to seven week old FVB male mice (body weights 20–25 g, Charles River Laboratories) were housed in isolation cages in pathogen–free conditions on a light-dark cycle with light from 7:00 to 20:00 at 25°C. Mice were fed a standard diet (Laboratory Rodent Diet 5001; PMI Nutrition International), containing 4.5% total fat with 0.26% omega-3 fatty acids and less than 0.01% C20:4, and were provided water ad libitum.
Murine asthma model
Mice were sensitized by intraperitoneal injection on protocol days 0 and 7 with 10 µg ovalbumin (OVA, Grade III; Sigma) plus 1 mg aluminum hydroxide as adjuvant (Sigma) in 0.2 ml saline. On protocol days 14, 15, 16 and 17, mice were aerosol challenged with 6% (wt/vol) OVA for 25 min per day as in (17). Some mice received RvD1 (10 ng) or vehicle (0.01% (vol/vol) ethanol) in saline by intravenous injection 1 h before mice were euthanized on protocol day 18 after first sensitization. In a second cohort, mice received RvD1 (1–100 ng) or vehicle (0.1% (vol/vol) ethanol) in saline by intravenous injection 30 min before OVA aerosol challenge on days 14, 15, 16 and 17 after the first sensitization. Analyses were performed 24 h after the last OVA nebulization. In a third cohort, mice received RvD1 (10–100 ng), AT-RvD1 (100 ng) or vehicle (0.1% (vol/vol) ethanol) alone by intravenous injection on protocol days 18, 19 and 20 and tissue samples were obtained for analyses during the resolution phase of the model on protocol days 21 and/or 25. In a fourth cohort, mice received RvD1 (100 ng), AT-RvD1 (100 ng) or vehicle (0.5% (vol/vol) ethanol intranasally in 20 µL of saline on protocol days 18, 19 and 20 and tissue samples were analyzed on protocol day 21. Doses were chosen based on prior reports with the E-series resolvin E1 in this model (17) and with RvD1 and AT-RvD1 in murine models of acute inflammation (11). Resolution was quantitated by calculation of the resolution index (time interval from maximum to half-maximum) for BALF eosinophils (as in (17, 18)).
Bronchoalveolar lavage
Bronchoalveolar lavage (BAL) was performed with two 1-ml aliquots of PBS with 0.6 mM EDTA. Cells in BAL fluids (BALFs) were resuspended in PBS and counted using a hemocytometer. Cytospin preparations were done by cytocentrifugation (265 g; StatSpin) and cells were stained with Wright-Giemsa (Sigma) for quantification of leukocyte subsets; at least 200 cells per slide were counted.
Histology and immunohistochemistry
In select animals, lungs were collected, fixed with 10% (vol/vol) buffered formalin at a perfusion pressure of 20 cm H2O and embedded in paraffin for staining with hematoxylin and eosin or periodic acid–Schiff (PAS) reagent (Sigma) as in (17). Additional sections were immunostained for rabbit anti-mouse ALX/FPR2 (1:50; Santa Cruz Biotechnology) and its isotype control, labeled with biotinylated goat anti-rabbit IgG (1:100; Santa Cruz Biotechnology), and developed using the Vectastain Elite ABC kit (Vector Laboratories) and diaminobenzidine (Sigma-Aldrich) as a substrate. NIH ImageJ 1.36b imaging software (National Institute of Health, Bethesda, MD, USA) was used to quantitate ALX/FPR2 receptor abundance. Scores for peribronchiolar inflammatory cell infiltrates in sections from each lung were graded as in (19), namely a score of 0 indicated the absence of inflammatory cell infiltrates; a score of 1, <5 layers of inflammatory cells in <50% of the bronchiolar submucosa; 2, >5 layers of inflammatory cells in < 50% of the bronchiolar submucosa; 3, <5 layers of inflammatory cells in >50% of the bronchiolar submucosa; and 4, >5 layers of inflammatory cells in >50% of the bronchiolar submucosa (19). To quantitate the level of mucus expression in the airway, the number of PAS staining (PAS+) cells in individual bronchioles was counted as described in (20).
Measurement of lung resistance
For measurement of airway hyper-responsiveness, mice were anesthetized with sodium pentobarbital (80 mg/kg, i.p.) and mechanically ventilated with a FlexiVent mouse ventilator (SciReq) and aerosolized methacholine (0, 1, 3, 10, 30 and 100 mg/ml) was delivered in-line through the inhalation port for 10 seconds. Lung resistance was determined as the mean of ten measurements obtained for each concentration of methacholine and was reported as the percent increase from baseline (PBS nebulization). No BAL or histological analysis was performed on mice undergoing measurement of airway hyper-responsiveness.
Cytokines and lipid mediators
Samples of cell-free BALF (centrifuged for 10 min at 300 g) were assigned an individual code and peptide mediators were measured by bead array (Aushon BioSystems) and lipid mediators by enzyme-linked immunoassay (ELISA; LTB4 (Cayman Chemical), and LXA4 (Neogen)).
Western blots
Lungs were homogenized with a hand held glass dounce. Samples were denatured and equal amounts of protein (50 µg) were loaded per lane prior to electrophoresis with 12% (w/v) SDS-PAGE gels. After transfer onto a polyvinylidene difluoride membrane (0.2 µm pore size) using the Mini-PROTEAN 3 apparatus (Bio-Rad Ltd), immunoblots were probed with primary antibodies to IκBα (1:1000 dilution, Santa Cruz Biotechnology) followed by a donkey anti-rabbit IgG conjugated to horseradish peroxidase (1:5000 dilution, GE Healthcare) and were visualized using ECL (GE Healthcare). An antibody against β-actin (1:500 dilution, Santa Cruz Biotechnology) was used as a loading control. The immunoblotted membranes were detected using a Chemiluminescence Substrate kit (Thermo Scientific) and visualized on the FluorChem® HD2 Imaging System (Alpha Innotech). Densitometric analysis was performed using integrated density analysis provided by the AlphaEaseFC software.
Gene expression
Lungs were obtained and snap frozen. RNA was extracted with Trizol and reverse transcribed. The cDNA was used as a template for the amplification of murine ALX/FPR2 [GeneID: 14289] by real-time PCR using a Stratagene real-time PCR machine (model #Mx 3005). Fold change was calculated as 2−ΔΔCT for the difference between the CT value for the gene of interest and the respective CT value for ppia (ΔCT) compared to day 0.
Lung macrophage isolation and allergen clearance
Macrophages from control or OVA-sensitized and -challenged animals were obtained at protocol day 21 from BAL as described in (21). Phagocytosis was determined in vitro using AMJ2-C8 (murine alveolar macrophages cell line, ATCC) or freshly obtained murine alveolar macrophages (mAlvMacs). As in (22), cells were placed on coverslips in 96-well plates (2 × 105 cells/well) in media (RPMI 1640 + 10% FCS containing L-glutamine and antibiotics) and incubated overnight at 37°C. Non-adherent cells were removed and mAlvMacs were supplemented with fresh medium. Macrophages (cell line or from BAL) were treated with RvD1, AT-RvD1 (0.1, 10 or 100 nM) or vehicle (ethanol <0.01%) and incubated in the dark (20 min, 37°C). Rabbit anti-OVA IgG-antibody-coated polybead® microsphere beads were prepared (according to the manufacturer’s instructions (Polysciences, Inc)) and added to the cells at a ratio of 13 beads/cell. Immediately (time 0) or after 15 min, cells were washed with PBS and paraformaldehyde (4%) was added. After 30 min, cells were washed again with PBS and FITC-conjugated goat anti-rabbit antibody (1:150) was added (35 min, room temperature, in the dark). Supernatants were removed, and after washing in PBS, the cells on the coverslips were mounted for fluorescent microscopy. Beads were counted in both light and fluorescence images that were acquired for ≥ 50 cells in each incubation. Since antibodies are not membrane permeable, only adherent non-internalized beads are fluorescent. This allows for distinction between internalized and cell adherent beads. To quantify particle internalization, the number of surface-bound beads was counted from the fluorescence images, and the total number of beads from the non-fluorescent images. The phagocytosis index was determined by subtracting the number of fluorescent beads from the total number of beads (non-fluorescent images) to derive the number of internalized beads. For each cell counted, the number of internalized beads was divided by the total number of beads to derive its phagocytosis index.
Macrophage phagocytosis of allergen in vivo
Mice were sensitized to OVA as described above. On protocol day 14, RvD1 (100 ng), AT-RvD1 (100 ng) or vehicle (0.1% (vol/vol) ethanol) alone were administered i.p. in 100 µl of sterile saline as in (18). After 5 min, rabbit anti-OVA IgG-antibody-coated beads were injected i.p. The number of beads approximated 13 latex beads per macrophage based on the anticipated total number of macrophages (~1 × 106) expected from control mice (non-stimulated) (23). After 15 min, peritoneal contents were collected by lavage (2 × 3 ml aliquots of PBS with 0.6 mM EDTA). Paraformaldehyde was immediately added (4% final concentration) to peritoneal lavage fluids. After 30 min, cells were washed with PBS and FITC-conjugated goat anti-rabbit antibody (1:150) was added (35 min, room temperature, in the dark). After centrifugation (265 g, 5 min), the supernatants were removed and cells were washed in PBS. Cells were mounted for fluorescent microscopy by cytospin (265g; StatSpin) and the phagocytosis index was calculated as described above.
RvD1 and AT-RvD1 further metabolism and inactivation
AMJ2-C8 cells were suspended (2 × 106/ml) in PBS with CaCl2, MgCl2 and dextrose (10mM) and warmed (37°C, 5min) prior to incubation for 30 min (37°C) in the presence of RvD1, AT-RvD1 (100 nM) or vehicle (ethanol 0.1%). Indomethacin (100 µM, Sigma) was added to select cells just prior to RvD1. Reactions were stopped with 2.5 volumes of iced methanol and stored at −20°C (≥ 12h) until extraction. d4-PGE2 was added as an internal standard to calculate recovery during C18 solid phase column extraction. The methyl formate eluate was taken for LC-MS/MS based quantitation of RvD1, AT-RvD1 and the RvD1 metabolite 17-oxo-RvD1 (as in (11, 24)). Briefly, metabolism to 17-oxo-RvD1 was determined by monitoring at m/z = 373 and the presence of at least three additional prominent daughter ions, including m/z 355 [-H2O]; 337 [-2H2O]; 329 [373-CO2]; 275 [373-CHO-CH2-(CH)2-CH2-CH3]; 231 [373-CHOH-CH2-(CH)2-CO2] and 141 [CHO-CH2-(CH)2-(CH2)2-COO-]. The quantities of 17-oxo-RvD1 relative to its precursors (RvD1, AT-RvD1) were expressed as percent conversion to the metabolite.
Statistical analysis
Statistical significance was assessed by the Student’s t-test and one-way analysis of variance (ANOVA). When significant differences were identified, individual comparisons were subsequently performed with the Tukey’s test. P values less than 0.05 were considered significant.
Results
RvD1 counters the development of allergic airway responses
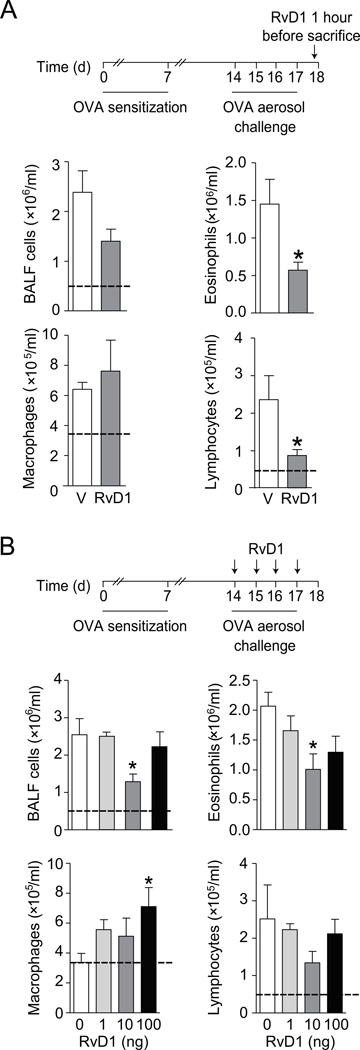
To determine if RvD1 could regulate allergic airway inflammation, a single dose of RvD1 (10 ng) or vehicle (0.01% ethanol) was given intravenously to animals on protocol day 18 after allergen sensitization and challenge with OVA. Leukocytes in the airways were measured 1h later in bronchoalveolar lavage fluids (BALFs). Total BALF cells were decreased after intravenous RvD1, with significant decrements in BALF eosinophils and lymphocytes (Fig. 1A). BALF eosinophils were decreased by approximately 60% from 1.45 ± 0.33 (vehicle) to 0.57 ± 0.10 (RvD1) (mean × 106/ml ± S.E.M. for n = 7, p < 0.05) and BALF lymphocytes were also decreased by approximately 60% from 2.4 ± 0.6 (vehicle) to 0.87 ± 0.2 (RvD1) (mean × 105/ml ± S.E.M. for n = 7, p < 0.05). In OVA naïve animals, BALF eosinophils were undetectable and BALF lymphocytes were 0.49 ± 0.22 (mean × 105/ml ± S.E.M. for n = 3) (Fig. 1).
FIGURE 1.
RvD1 decreases allergic lung inflammation. (A). Mice were sensitized and challenged with OVA prior to one dose of RvD1 (10 ng, i.v.) or vehicle 1 hour before BAL on protocol day 18 (inset). Total leukocytes and leukocyte subsets were enumerated (see Methods). (B) A second cohort of sensitized and challenged mice received RvD1 (1–100 ng, i.v.) or vehicle 30 minutes prior to OVA aerosol challenge on 4 successive days (protocol days 14–17) (inset) and BALF leukocytes were enumerated on protocol day 18. The dashed black line indicates the cells from OVA naïve, non-allergic control mice. Results represent the mean ± s.e.m of two or more independent experiments with three or more mice per group per experiment. * P < 0.05 vs. vehicle.
To determine if RvD1 could prevent the development of allergic airway responses, 1, 10 or 100 ng or vehicle (0.1% ethanol) was given to sensitized animals intravenously 30 min prior to each daily allergen aerosol challenge for a period of 4 days (see Methods). RvD1 again led to significant decrements in BALF total cells, in particular eosinophils (Fig. 1B). 10 ng of RvD1 was the most potent dose. Of interest, the magnitude of RvD1’s inhibition at this dosing schedule was similar to a single administration given 1h prior to BAL (Fig. 1A, 1B). The RvD1 dose response relationship for inhibition of BALF eosinophils and lymphocyte numbers appeared to be bell-shaped as neither 1 nor 100 ng of RvD1 was more effective than 10 ng. In contrast, RvD1 gave a dose dependent increase in BALF macrophages (Fig. 1B).
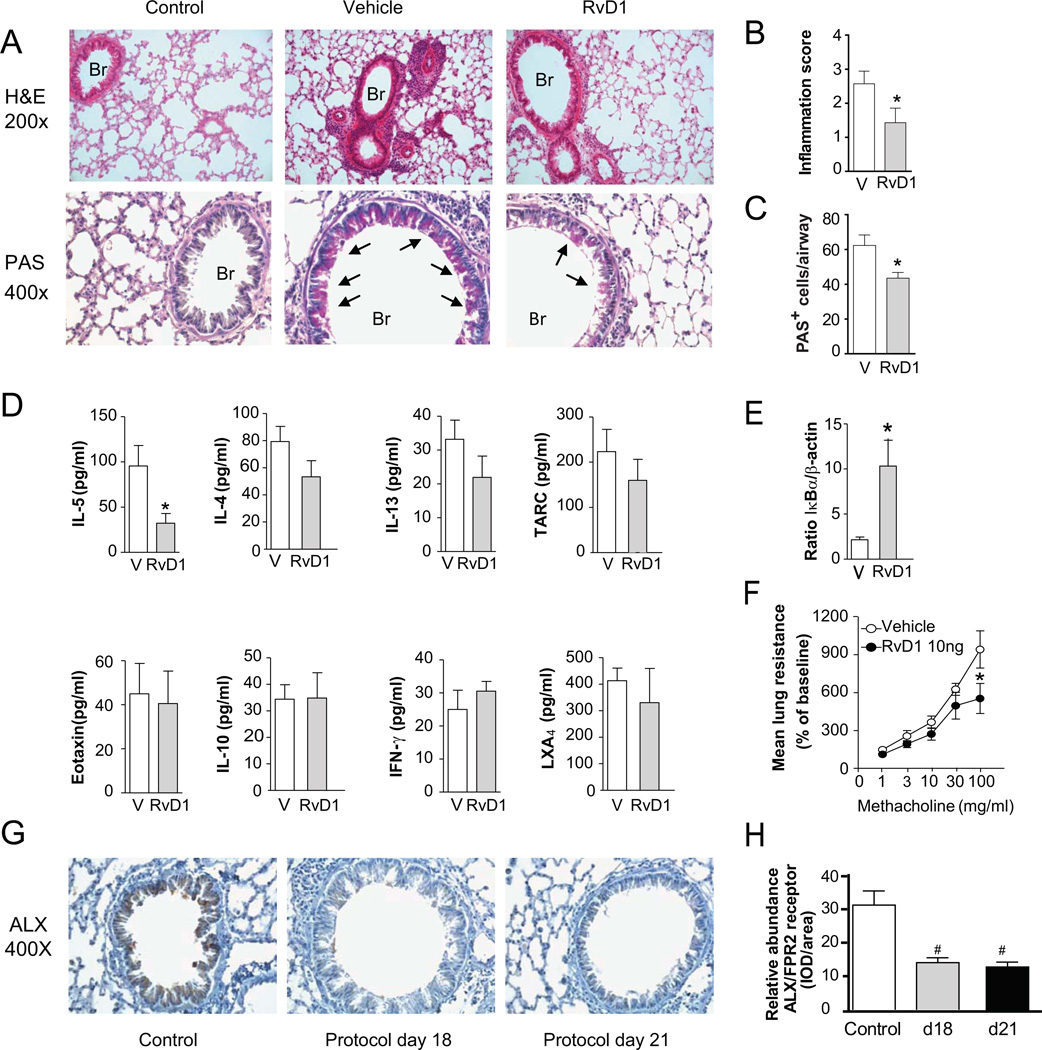
Leukocyte infiltration into the lung parenchyma was markedly decreased by RvD1. Histopathological analyses revealed the RvD1 (10 ng, i.v.) decreased leukocyte accumulation in the peribronchial regions and decreased mucus metaplasia in bronchial epithelial cells (Fig. 2A–C). Cell-free BALFs were analyzed by cytokine bead array and ELISA to determine the impact of intravenous RvD1 on levels of select inflammatory mediators. RvD1 administration led to significantly lower levels of interleukin-5 (IL-5), approximately 60% lower than vehicle (Fig. 2D). Levels of IL-4, IL-13 and TARC were also decreased but did not reach statistical significance at this sample size. There was no significant effect of RvD1 on eotaxin, IL-10, IFN-γ or lipoxin A4 (LXA4). RvD1 (10 ng) significantly reduced lung IκBα degradation (Fig. 2E). In addition, airway hyper-responsiveness to inhaled methacholine was dampened by RvD1 (10 ng, i.v.), reaching statistical significance only at 100 mg/ml methacholine (Fig. 2F).
FIGURE 2.
RvD1 prevents the development of allergic airway responses. Allergen sensitized mice received RvD1 (10 ng, i.v.) or vehicle 30 minutes prior to OVA aerosol challenge on 4 successive days (protocol day 14–17). On protocol day 18, (A) lung tissue inflammation was assessed by hematoxylin and eosin staining (H&E, original magnification, 200×) and the presence of mucus metaplasia was determined by periodic acid–Schiff reagent (original magnification, 400×) staining of lung sections. Arrows indicate mucus (magenta) containing goblet cells; Br, bronchus. Quantitative measures of (B) inflammation and (C) mucus metaplasia were determined in lung sections (see Methods). (D) BALF levels of peptide and lipid mediators were measured by immunoassay and (E) lung IκBα was determined by Western blotting (see Methods). (F) Mean lung resistance (% of baseline) after aerosolized methacholine was determined by Flexivent (see Methods). (G) Representative images for identification of ALX/FPR2 receptors by imunohistochemistry in lung sections from control mice and protocol days 18 and 21 (magnification: 400×). (H) Relative abundance of lung ALX/FPR2 staining in the lung. Results represent the mean ± s.e.m of two or more independent experiments with three or more mice per group per experiment. * P< 0.05 vs. vehicle. # P< 0.05 vs. control.
RvD1 can interact with ALX/FPR2 receptors to transduce, in part, its counter-regulatory actions (9, 10, 25), so the expression of murine ALX/FPR2 receptors in the lung was determined by quantitative PCR and immunohistochemistry. Compared to baseline mice prior to allergen sensitization and challenge (ΔCT 6.37 ± 0.33), lung ALX/FPR2 expression was decreased at both peak inflammation (protocol day 18) (ΔCT 7.05 ± 0.20) and early resolution (protocol day 21) (ΔCT 7.43 ± 0.24; P<0.05 for n = 3, mean ± S.E.M.). These findings were consistent with a fold change of 0.64 (day 18) and 0.49 (day 21), respectively, in gene expression. Lung immunohistochemistry for ALX/FPR2 abundance gave similar findings as qPCR. Prominent ALX/FPR2 staining was present at baseline in airway epithelial cells and alveolar macrophages, and decreased during the allergic airway responses at protocol day 18 and 21 (Fig. 2 G, H).
RvD1 and AT-RvD1 enhance the resolution of airway inflammation
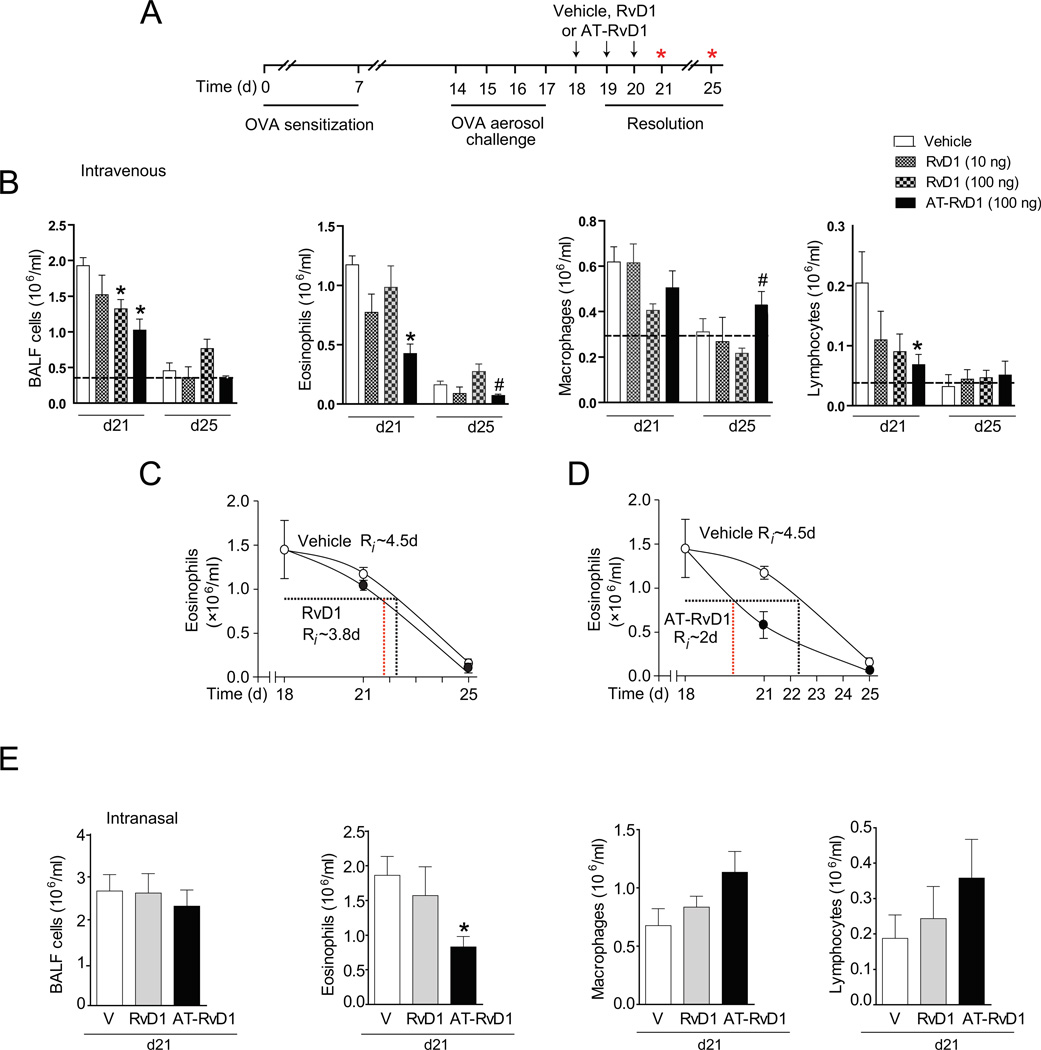
In view of RvD1’s protective actions in the airway, we next determined the influence of RvD1 and its aspirin-triggered 17(R)-epimer (AT-RvD1) on the resolution of established airway inflammation (Fig. 3). After animals were OVA sensitized and aerosol challenged, RvD1 (10–100 ng), AT-RvD1 (100 ng) or vehicle (0.1% ethanol) were administered intravenously for 3 consecutive days (protocol days 18–20), and no further allergen challenges were performed (Fig. 3A). BALF leukocytes were enumerated on days 21 and 25. During resolution (day 21), RvD1-treated mice displayed a dose dependent decrease in total BALF cells with associated changes in eosinophils, macrophages and lymphocytes (Fig. 3B). Compared to RvD1, an equivalent dose (100 ng) of AT-RvD1 led to even more significant decrements in BALF total cells, eosinophils and lymphocytes (Fig. 3B). With AT-RvD1, BALF eosinophils decreased from 1.17 ± 0.07 (vehicle) to 0.42 ± 0.08 (AT-RvD1) (mean × 106/ml ± S.E.M. for n > 6, p < 0.05). In addition, AT-RvD1 decreased BALF lymphocytes from 2.05 ± 0.52 (vehicle) to 0.67 ± 0.2 (AT-RvD1) (mean × 105/ml ± S.E.M. for n > 6, p < 0.05). At day 25, BALF eosinophil and lymphocyte numbers were near baseline. Of interest, AT-RvD1 led to small, but significant decreases in eosinophils and increases in macrophages at this time point (Fig. 3B).
FIGURE 3.
RvD1 and AT-RvD1 promote the resolution of airway inflammation. (A) RvD1 (10 or 100 ng), AT-RvD1 (100 ng) or vehicle was administered (i.v.) to OVA sensitized and challenged mice on protocol days 18–20 (inset) after cessation of OVA exposure, and (B) BALF total cells, eosinophils, macrophages and lymphocytes were evaluated on protocol day 21. The dashed black line indicates the cells from OVA naïve, non-allergic control mice. (C) The resolution interval (Ri) for RvD1 (100ng) and (D) AT-RvD1 (100 ng) for BALF eosinophils was determined. (E) RvD1 (100 ng), AT-RvD1 (100 ng) or vehicle was administered directly to the airway (intranasal) in OVA sensitized and challenged mice on protocol days 18–20 after cessation of OVA exposure. BALF total cells, eosinophils, macrophages and lymphocytes were evaluated on protocol day 21. Results represent the mean ± S.E.M. for two or more independent experiments with three or more mice per group per experiment. * P < 0.05 vs. vehicle, #P < 0.05 vs. RvD1.
The resolution interval is defined as the time required for cell numbers to decrease to 50% of the maximum at peak inflammation (18), and in vehicle-exposed mice, the endogenous resolution interval for BALF eosinophils was approximately 4.5d (Fig. 3C, 3D). RvD1-treated mice displayed a modestly decreased resolution interval for BALF eosinophils (less than 4d for 100 ng) (Fig. 3C). Of note, the resolution interval for BALF eosinophils was markedly decreased with AT-RvD1 to ~2d – approximately 45% of the resolution interval with vehicle (Fig. 3D), indicative of more rapid resolution of acute allergic inflammation.
To determine the impact of airway delivery, mice were given daily intranasal RvD1 (100 ng), AT-RvD1 (100 ng) or vehicle (0.5% ethanol) on protocol days 18–20. Exposure to AT-RvD1, but not RvD1, at this dose led to significant decreases in BALF eosinophils at day 21 (Fig. 3E). BALF eosinophils decreased from 1.86 ± 0.27 (vehicle) to 0.83 ± 0.15 (AT-RvD1) (mean × 106/ml ± S.E.M. for n > 5, p < 0.05). Intranasal AT-RvD1 also increased BALF macrophages and lymphocytes. Overall, significant differences were not obtained for changes in total BALF cell numbers (Fig. 3E).
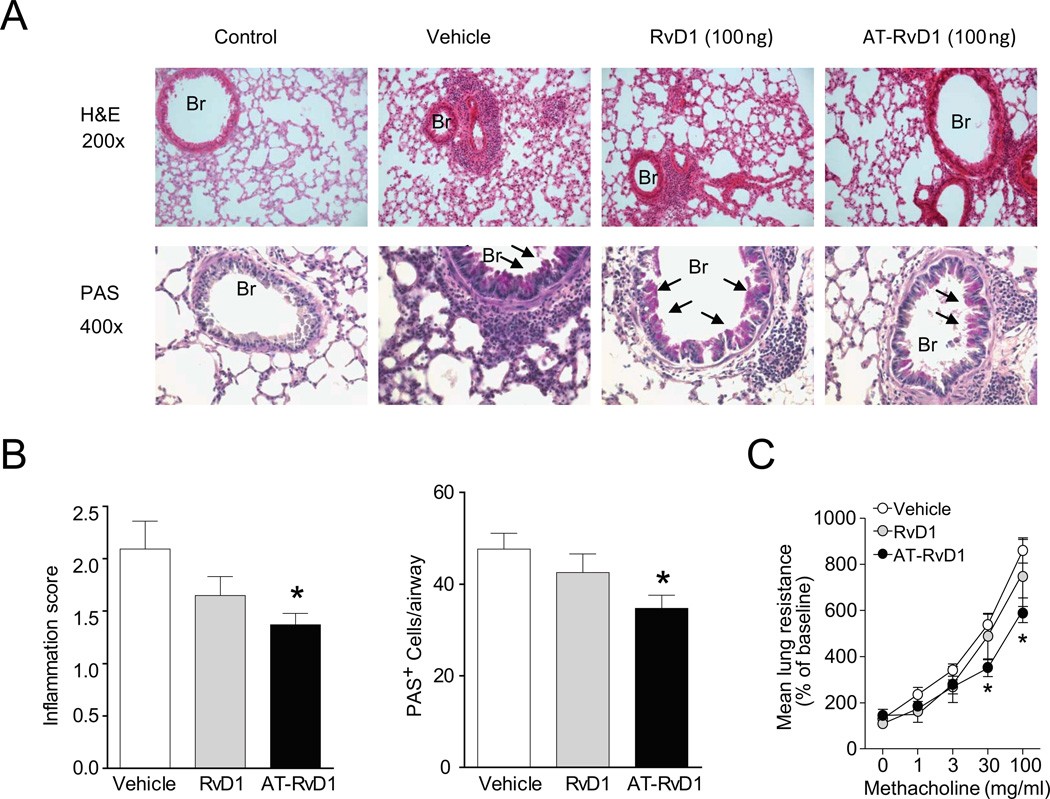
AT-RvD1 accelerates the resolution of lung inflammation and airway mucus metaplasia and hyper-responsiveness
The pro-resolving actions of AT-RvD1 for airway leukocytes were also evident for lung inflammation and airway mucus metaplasia and hyper-responsiveness. AT-RvD1 was more potent than RvD1 for each of these metrics (Fig. 4). Lung histopathology demonstrated that AT-RvD1 facilitated the resolution of leukocyte infiltration with less reactive bronchial epithelial cells and mucus metaplasia, as detected by H&E and PAS staining, respectively (Fig. 4A, B). These changes were quantitated by determining an inflammation score (see Methods) and enumerating the numbers of PAS+ cells in the airways, both of which were significantly reduced with AT-RvD1 (Fig. 4B). In addition, mice treated with AT-RvD1 displayed significant protection from methacholine-induced increases in mean lung resistance (Fig. 4C). Thus, AT-RvD1 gave potent pro-resolving actions for allergic airway responses.
FIGURE 4.
Impact of RvD1 and AT-RvD1 on the resolution of airway inflammation, mucus and hyper-reactivity. (A) Lung tissue sections were obtained at protocol day 21 from mice given RvD1 (100 ng), AT-RvD1 (100 ng) or vehicle and stained with hematoxylin and eosin (H&E, original magnification, 200×) or periodic acid–Schiff reagent (PAS, original magnification, × 400) (see Methods). Arrows indicate examples of mucus (magenta) containing goblet cells; Br, bronchus. (B) Quantitative analyses of lung inflammation and bronchial PAS-positive cells in lung sections were performed (see Methods). (C) Airway hyper-responsiveness to aerosolized methacholine was assessed on day 21 by measuring the mean lung resistance (% of baseline) (see Methods). Results represent the mean ± s.e.m for two or more independent experiments with three or more mice per group per experiment. *P < 0.05 vs. vehicle.
AT-RvD1 treatment regulates airway mediators
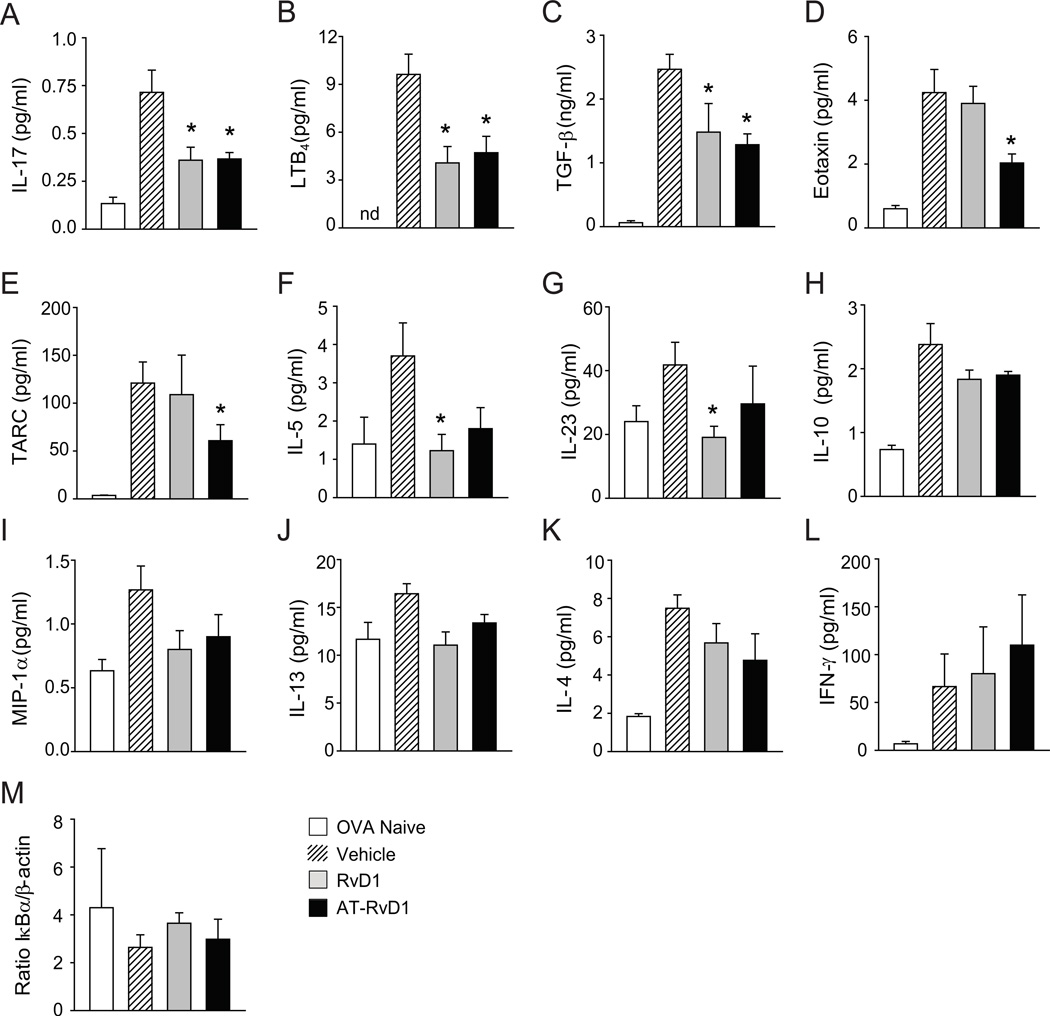
To uncover pro-resolving mechanisms, BALF levels of peptide and lipid mediators were measured during early resolution (protocol day 21). Exposure to both RvD1 and AT-RvD1 led to significantly decreased levels of IL-17, LTB4 and TGF-β (Fig. 5A–C). AT-RvD1 alone led to significant decrements in eotaxin and TARC (Fig. 5D–E) and RvD1 gave significant regulation of IL-5 and IL-23 (Fig. 5F–G). AT-RvD1 also decreased both of these mediators, but the results did not reach statistical significance at this sample size. No significant changes were observed in IL-10, MIP-1α, IL-13, IL-4 or IFN-γ (Fig. 5H–L). Because RvD1 regulates specific miRNAs to decrease NF-κB signaling in acute inflammation (12) and RvD1 down-regulated IκBα degradation to prevent the development of allergic responses (Fig. 2E), the impact of AT-RvD1 and RvD1 on this measure of NF-κB pathway activation was next determined during resolution. In contrast to their administration during induction of the airway inflammation, neither AT-RvD1 nor RvD1 led to significant changes in IκBα degradation at this resolution time point (Fig. 5M).
FIGURE 5.
RvD1 and AT-RvD1 selectively regulate inflammatory mediators during resolution of allergic inflammation. (A–I) BALF levels of peptide and lipid mediators were measured in OVA naïve, non-allergic control mice (white) and on protocol day 21 in materials obtained from mice given RvD1 (100 ng, grey), AT-RvD1 (100 ng, black) or vehicle (hatched) (see Methods). (M) Lung tissue was obtained on protocol day 21 during resolution and was subjected to Western blotting analyses for IκBα (see Methods). Results represent the mean ± s.e.m for two or more independent experiments with three or more mice per group per experiment. *P< 0.05 vs. vehicle. nd, not detected.
RvD1 is metabolized by alveolar macrophages
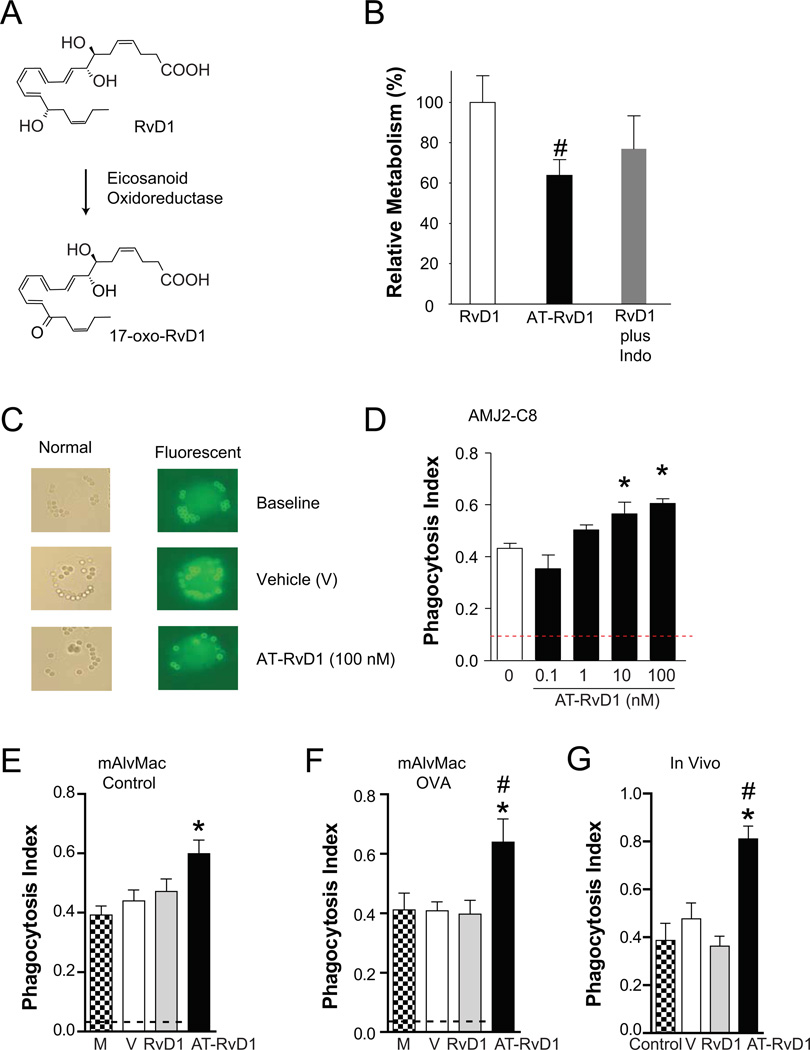
Recombinant eicosanoid oxidoreductase and murine lung rapidly converts RvD1 to an inactive 17-oxo-RvD1 metabolite and AT-RvD1 resists this rapid inactivation (11). Because the in vitro and in vivo results with AT-RvD1 were more potent than RvD1, the metabolism of these compounds was next determined with a murine alveolar macrophage cell line (AMJ2-C8). Cells were exposed (30 min, 37°C) in vitro to either RvD1 or AT-RvD1. Lipids were extracted and taken to LC-MS/MS for analyses of RvD1 and AT-RvD1 metabolism (see Methods). In parallel, some cells incubated with RvD1 were also exposed to indomethacin, which inhibits the eicosanoid oxidoreductase (25) – an enzyme that is highly expressed in the lung (26) and capable of converting the alcohol at carbon 17 position to a ketone (9) (Fig. 6A). Metabolic inactivation of AT-RvD1 by the murine alveolar macrophages was significantly decreased relative to RvD1 (63.8 ± 7.5% with AT-RvD1 versus RvD1, mean ± S.E.M. for n=3, P < 0.05). Indomethacin also decreased RvD1 inactivation by approximately 25% (Fig. 6B).
FIGURE 6.
AT-RvD1 resists metabolic inactivation by macrophages and stimulates macrophage clearance of allergen. Murine alveolar macrophages cells (AMJ2-C8) were incubated (30 minutes, 37°C) in the presence of RvD1 (in some cases with indomethacin) or AT-RvD1. Lipids were extracted and (A) RvD1 metabolites were identified and quantitated by LC-MS/MS analyses (see Methods). (B) Values are expressed as the relative percent 17-oxo-RvD1 metabolite relative to the total starting compound. (C) Macrophage phagocytosis of allergen was determined using rabbit anti-OVA IgG-coated beads (2 µm) that are detectable by light microscopy. In non-permeabilized cells, a fluorophore tagged antibody (FITC-conjugated goat anti-rabbit antibody) was used to distinguish adherent (fluorescent) from internalized (non-fluorescent) beads. A phagocytosis index was calculated after 15 min in the presence of RvD1, AT-RvD1 (0.1, 10 and/or 100 nM), vehicle (V) or media alone (M) using (D) AMJ2-C8 cells in vitro or (E,F) alveolar macrophages (mAlvMacs) ex vivo that were isolated from BALF of (E) control or (F) OVA-sensitized and –challenged mice (Protocol day 21) (see Methods). The dashed black line indicates the baseline phagocytosis index. (G) The phagocystosis index was determined in vivo in OVA-sensitized or control mice 15 min after i.p. injection of OVA IgG-coated beads. In some animals, RvD1 (100 ng), AT-RvD1 (100 ng) or vehicle (0.1% ethanol) were given i.p. 5 min before introduction of the beads. Results represent the mean ± S.E.M. for two or more independent experiments. *P< 0.05 vs. vehicle, #P < 0.05 vs. RvD1.
AT-RvD1 enhances macrophage clearance of allergen by phagocytosis
Because AT-RvD1 increased BALF macrophages in vivo during resolution (Fig. 3) and macrophages play important pro-resolving roles in clearing inflamed tissues (24), the impact of AT-RvD1 and RvD1 on macrophage phagocytosis of allergen was determined. AMJ2-C8 cells (in vitro) and mAlvMacs that were isolated from BALF on protocol day 21 from control animals or mice that were OVA-sensitized and aerosol–challenged (ex vivo) were exposed to IgG-OVA coated beads (see Methods, Fig. 6C). In the nanomolar range, AT-RvD1 increased the macrophage phagocytosis index within 15 min (37°C) for allergen coated beads (Fig. 6D–G). The increased phagocytosis index was concentration-dependent for AT-RvD1 with AMJ2-C8 cells (Fig. 6D). AT-RvD1 also increased the allergen phagocytosis index for mAlvMacs from control and OVA allergic mice (Fig. 6E–F). It was not feasible to deliver the OVA coated beads to alveoli in live animals, so the in vivo allergen phagocytosis index was determined for peritoneal macrophages in OVA-sensitized mice. AT-RvD1 also significantly increased this pro-resolving functional response in vivo.
Discussion
Here, we have provided evidence for both anti-inflammatory and pro-resolving actions for RvD1 and its epimer AT-RvD1 in allergic airway responses. When given to OVA-sensitized and aerosol challenged mice, RvD1 reduced the development of allergic airway responses. RvD1 most potently regulated eosinophilia with decreases in levels of IL-5, an important Th2 cytokine that serves as a chemoattractant and activator of eosinophils. BALF levels of the related Th2 cytokines IL-4 and IL-13 were also decreased, and IκBα in the lung was significantly increased by RvD1, suggesting decreased activation of NF-κB. RvD1 also reduced bronchial mucus metaplasia with a more modest effect on airway hyper-responsiveness to methacholine. Levels of the counter-regulatory mediators IL-10 and LXA4 were not increased, indicating non-redundant anti-inflammatory signaling circuits for RvD1.
To determine if these anti-inflammatory properties for RvD1 would also enhance the resolution of allergic airway responses, RvD1 was given after allergen sensitization and challenge was complete. RvD1 led to significant decrements in allergic lung inflammation 1 hour after administration and during early resolution, leading to an approximate 10% decrease in the eosinophil resolution interval. An equivalent dose of AT-RvD1 (100 ng, ~0.005 mg/kg) provided significantly greater pro-resolving actions, decreasing the eosinophil resolution interval by more than 50% – an approximate doubling of the pace of resolution relative to RvD1. Both AT-RvD1 and RvD1 can serve as an agonist at ALX/FPR2 receptors to transduce, in part, their pro-resolving actions (9, 10, 25, 27). ALX/FPR2 was broadly expressed in airway epithelial cells and alveolar macrophages and dynamically regulated during allergic airway responses that led to decreased receptor abundance. These changes are similar to those observed in human asthma (28). In addition to regulation of ALX/FPR2 receptors, pro-resolving signaling can also be decreased by metabolic inactivation of RvD1 and AT-RvD1 (11). Although enzymatically generated from DHA by distinct biosynthetic pathways, RvD1 and AT-RvD1 are diastereomers, differing only in stereochemistry at carbon 17 (reviewed in (29)). This change in stereochemistry for AT-RvD1 provides a significant increase in the compound’s half-life in vivo, secondary to resistance to metabolic inactivation by eicosanoid oxidoreductases (11, 25). Here, AT-RvD1 had a decreased rate of metabolic inactivation relative to RvD1, which likely contributed to AT-RvD1’s enhanced in vitro and in vivo pharmacological actions. In addition, the reduced efficacy of airway administration relative to the intravenous route was notable given the capacity for lung macrophages to inactivate both RvD1 and AT-RvD1.
During resolution, AT-RvD1 led to significant decrements in select inflammatory mediators, namely BALF IL-17, eotaxin, TARC, TGF-β and LTB4. Unlike when given prior to aerosol challenge, neither RvD1 nor AT-RvD1 significantly changed lung IκBα during resolution. In the setting of allergen driven inflammation, inhaled allergen needs to be cleared in order to facilitate resolution. AT-RvD1 increased alveolar macrophage phagocytosis of allergen coated beads in vitro in a concentration-dependent manner and AT-RvD1 increased the peritoneal macrophage allergen phagocytosis index in vivo. Alveolar and peritoneal macrophages differ in several respects, in particular their uptake of particulate and soluble antigen that is spatiotemporally regulated in the airway (30–32). Because of safety concerns delivering the beads directly into the airway and the animals had been systemically sensitized by intraperitoneal injection, peritoneal macrophages were chosen for in vivo analysis. A potential caveat to our findings is that the increased phagocytic capacity of peritoneal relative to alveolar macrophages may over estimate the role of lung macrophages in vivo in the clearance of allergen delivered directly into the respiratory tract; however, AT-RvD1 promoted increased allergen engulfment by both peritoneal macrophages in vivo and alveolar macrophages in vitro. Together, these results point to more rapid allergen clearance by AT-RvD1 exposed macrophages as a pro-resolving mechanism for allergic airway responses, including airway inflammation, mucus metaplasia, and hyper-responsiveness to methacholine.
The development of airway inflammation in asthma is characterized by leukocyte accumulation and in many individuals is notable for a Th2 cytokine gene expression signature (33). Among the inflammatory mediators implicated in eosinophil recruitment are the Th2 cytokine IL-5 and eotaxins (2). In addition, NF-κB activation plays an essential role in the development of airway eosinophilia in this model of asthma (34). LTB4 can also serve as an eosinophil chemoattractant, as LTB4 receptor (BLT1)-deficient mice do not develop eosinophilic inflammation or airway hyperresponsiveness in an experimental asthma model (35). Here, we have identified RvD1 and AT-RvD1 as potent regulators of murine IL-5, eotaxin and LTB4 and eosinophil accumulation after allergen challenge. Eosinophils are a significant source of the pro-fibrotic growth factor TGF-β1 (36) that contributes to airway remodeling in asthma pathogenesis (37, 38). The decrease in eosinophils and TGF-β levels by RvD1 and AT-RvD1 suggest a beneficial action in preventing airway remodeling, but further study using RvD1 stable analogs in longer term models is needed to address this outcome.
In the murine experimental model used here, the timely resolution of allergic airway responses is governed by regulation of IL-17, not Th2 cytokines (17). This is in sharp contrast to the pivotal roles for Th2 cytokines during the development of allergic lung inflammation. IL-17 can be generated by several cell types in asthmatic lung, most notably Th-17 cells that are a subset of CD4+ T helper cells characterized by the production of IL-17 and whose population expansion and survival depends upon IL-23. IL-17 has been linked to the pathogenesis of many inflammatory diseases, is present in the airways of asthmatic patients and can induce lung inflammation, airway hyper-reactivity and mucus production (17, 39–42). Similar to resolvin E1 and lipoxin A4 stable analog (17), RvD1 and AT-RvD1 also regulated IL-17 to promote resolution. Also like RvE1, RvD1 decreased BALF IL-23 during resolution. In addition to decreasing the generation of pro-phlogistic mediators, pro-resolving mediators are defined in part by enhancing macrophage mediated clearance of inflamed tissue (29), and here, AT-RvD1 enhanced the phagocytic uptake of IgG-OVA-coated beads by macrophages in vitro and in vivo. Thus, mechanisms for RvD1 and AT-RvD1 actions are distinct for preventing the development of allergic airway responses (i.e., decreased Th2 cytokines and NF-κB) and in promoting their resolution (i.e., regulation of IL-23 and IL-17 and increased allergen clearance).
In conclusion, these results demonstrate protective anti-inflammatory and pro-resolving actions for D-series resolvins in airway inflammation. RvD1 and AT-RvD1 decreased eosinophil recruitment and activation, Th2 cytokines early and Th17 cytokines late, airway mucus metaplasia and airway hyper-reactivity to methacholine and AT-RvD1 promoted the resolution of allergic airway responses by enhancing allergen clearance. Together, these results point to D-series resolvins as a candidate therapeutic modality in allergic disorders and provide a window into potential mechanisms for the association between changes in airway mucosal omega-3 fatty acids and asthma.
Acknowledgments
This research was supported in part by the US National Institutes of Health grants AI068084 (BDL), P01-GM095467 (BDL, CNS) and GM38765 (CNS) and a Brazilian postdoctoral fellowship from Coordenação de Aperfeiçoamento de Pessoal de Nível Superior (CAPES) (process number 0616-08/7) (APR).
Abbreviations
- AT-RvD1
aspirin-triggered resolvin D1 (7S,8R,17R-trihydroxy-4Z,9E,11E,13Z,15E,19Z-docosahexaenoic acid)
- BALF
bronchoalveolar lavage fluid
- DHA
docosahexaenoic acid
- LC-MS/MS
liquid chromatography-tandem mass spectrometry
- LTs
leukotrienes
- LXA4
lipoxin A4
- RvD1
resolvin D1 (7S,8R,17S-trihydroxy-4Z,9E,11E,13Z,15E,19Z-docosahexaenoic acid)
References
- 1.Fanta CH. Asthma. N Engl J Med. 2009;360:1002–1014. doi: 10.1056/NEJMra0804579. [DOI] [PubMed] [Google Scholar]
- 2.Busse WW, Lemanske RF., Jr Asthma. N Engl J Med. 2001;344:350–362. doi: 10.1056/NEJM200102013440507. [DOI] [PubMed] [Google Scholar]
- 3.Serhan CN, Hong S, Gronert K, Colgan SP, Devchand PR, Mirick G, Moussignac RL. Resolvins: a family of bioactive products of omega-3 fatty acid transformation circuits initiated by aspirin treatment that counter proinflammation signals. J Exp Med. 2002;196:1025–1037. doi: 10.1084/jem.20020760. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 4.Levy BD, Kohli P, Gotlinger K, Haworth O, Hong S, Kazani S, Israel E, Haley KJ, Serhan CN. Protectin D1 is generated in asthma and dampens airway inflammation and hyperresponsiveness. J Immunol. 2007;178:496–502. doi: 10.4049/jimmunol.178.1.496. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 5.Bilal S, Haworth O, Wu L, Weylandt KH, Levy BD, Kang JX. Fat-1 transgenic mice with elevated omega-3 fatty acids are protected from allergic airway responses. Biochimie Biophysica Acta. 2011;1812:1164–1169. doi: 10.1016/j.bbadis.2011.05.002. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 6.Ariel A, Fredman G, Sun YP, Kantarci A, Van Dyke TE, Luster AD, Serhan CN. Apoptotic neutrophils and T cells sequester chemokines during immune response resolution through modulation of CCR5 expression. Nat Immunol. 2006;7:1209–1216. doi: 10.1038/ni1392. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 7.Schwartz J, Weiss ST. The relationship of dietary fish intake to level of pulmonary function in the first National Health and Nutrition Survey (NHANES I) Eur Respir J. 1994;7:1821–1824. doi: 10.1183/09031936.94.07101821. [DOI] [PubMed] [Google Scholar]
- 8.Freedman SD, Blanco PG, Zaman MM, Shea JC, Ollero M, Hopper IK, Weed DA, Gelrud A, Regan MM, Laposata M, Alvarez JG, O'Sullivan BP. Association of cystic fibrosis with abnormalities in fatty acid metabolism. New England Journal of Medicine. 2004;350:560–569. doi: 10.1056/NEJMoa021218. [DOI] [PubMed] [Google Scholar]
- 9.Krishnamoorthy S, Recchiuti A, Chiang N, Yacoubian S, Lee CH, Yang R, Petasis NA, Serhan CN. Resolvin D1 binds human phagocytes with evidence for proresolving receptors. Proc Natl Acad Sci U S A. 2010;107:1660–1665. doi: 10.1073/pnas.0907342107. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 10.Norling LV, Dalli J, Flower RJ, Serhan CN, Perretti M. Resolvin D1 limits PMN recruitment to inflammatory loci: receptor dependent actions. Arteriosclerosis, Thrombosis, and Vascular Biology. 2012 doi: 10.1161/ATVBAHA.112.249508. in press. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 11.Sun YP, Oh SF, Uddin J, Yang R, Gotlinger K, Campbell E, Colgan SP, Petasis NA, Serhan CN. Resolvin D1 and its aspirin-triggered 17R epimer. Stereochemical assignments, anti-inflammatory properties, and enzymatic inactivation. J Biol Chem. 2007;282:9323–9334. doi: 10.1074/jbc.M609212200. [DOI] [PubMed] [Google Scholar]
- 12.Recchiuti A, Krishnamoorthy S, Fredman G, Chiang N, Serhan CN. MicroRNAs in resolution of acute inflammation: identification of novel resolvin D1-miRNA circuits. FASEB J. 2011;25:544–560. doi: 10.1096/fj.10-169599. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 13.Tian H, Lu Y, Sherwood AM, Hongqian D, Hong S. Resolvins E1 and D1 in Choroid-retinal Endothelial Cells and Leukocytes: Biosynthesis and Mechanisms of Anti-inflammatory Actions. Invest Ophthalmol Vis Sci. 2009 doi: 10.1167/iovs.08-3146. [DOI] [PubMed] [Google Scholar]
- 14.Duffield JS, Hong S, Vaidya VS, Lu Y, Fredman G, Serhan CN, Bonventre JV. Resolvin D series and protectin D1 mitigate acute kidney injury. J Immunol. 2006;177:5902–5911. doi: 10.4049/jimmunol.177.9.5902. [DOI] [PubMed] [Google Scholar]
- 15.Kasuga K, Yang R, Porter TF, Agrawal N, Petasis NA, Irimia D, Toner M, Serhan CN. Rapid appearance of resolvin precursors in inflammatory exudates: novel mechanisms in resolution. J Immunol. 2008;181:8677–8687. doi: 10.4049/jimmunol.181.12.8677. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 16.Wang B, Gong X, Wan JY, Zhang L, Zhang Z, Li HZ, Min S. Resolvin D1 protects mice from LPS-induced acute lung injury. Pulm Pharmacol Ther. 2011;24:434–441. doi: 10.1016/j.pupt.2011.04.001. [DOI] [PubMed] [Google Scholar]
- 17.Haworth O, Cernadas M, Yang R, Serhan CN, Levy BD. Resolvin E1 regulates interleukin 23, interferon-gamma and lipoxin A4 to promote the resolution of allergic airway inflammation. Nat Immunol. 2008;9:873–879. doi: 10.1038/ni.1627. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 18.Bannenberg GL, Chiang N, Ariel A, Arita M, Tjonahen E, Gotlinger KH, Hong S, Serhan CN. Molecular circuits of resolution: formation and actions of resolvins and protectins. J Immunol. 2005;174:4345–4355. doi: 10.4049/jimmunol.174.7.4345. [DOI] [PubMed] [Google Scholar]
- 19.Fulkerson PC, Fischetti CA, Hassman LM, Nikolaidis NM, Rothenberg ME. Persistent effects induced by IL-13 in the lung. American Journal of Respiratory, Cell and Molecular Biology. 2006;169:2117–2126. doi: 10.1165/rcmb.2005-0474OC. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 20.Bilsborough J, Chadwick E, Mudri S, Ye X, Henderson WRJ, Waggie K, Hebb L, Shin J, Rixon M, Gross JA, Dillon SR. TACI-Ig prevents the development of airway hyperresponsiveness in a murine model of asthma. Clinical and Experimental Allergy. 2008;38:1959–1968. doi: 10.1111/j.1365-2222.2008.03099.x. [DOI] [PubMed] [Google Scholar]
- 21.Levy BD, Romano M, Chapman HA, Reilly JJ, Drazen J, Serhan CN. Human alveolar macrophages have 15-lipoxygenase and generate 15(S)-hydroxy-5,8,11-cis-13-trans-eicosatetraenoic acid and lipoxins. Journal of Clinical Investigation. 1993;92:1572–1579. doi: 10.1172/JCI116738. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 22.Gilberti RM, Joshi GN, Knecht DA. The phagocytosis of crystalline silica particles by macrophages. Am J Respir Cell Mol Biol. 2008;39:619–627. doi: 10.1165/rcmb.2008-0046OC. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 23.Zhang X, Goncalves R, Mosser DM. The isolation and characterization of murine macrophages. Current Protocols in Immunology. 2008;Chapter 14(Unit 14.11) doi: 10.1002/0471142735.im1401s83. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 24.Serhan CN, Yang R, Martinod K, Kasuga K, Pillai PS, Porter TF, Oh SF, Spite M. Maresins: novel macrophage mediators with potent antiinflammatory and proresolving actions. J Exp Med. 2009;206:15–23. doi: 10.1084/jem.20081880. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 25.Krishnamoorthy S, Recchiuti A, Chiang N, Fredman G, Serhan CN. Resolvin D1 Receptor Stereoselectivity and Regulation of Inflammation and Proresolving MicroRNAs. Am J Pathol. 2012 doi: 10.1016/j.ajpath.2012.01.028. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 26.Hong S, Porter TF, Lu Y, Oh SF, Pillai PS, Serhan CN. Resolvin E1 metabolome in local inactivation during inflammation-resolution. J Immunol. 2008;180:3512–3519. doi: 10.4049/jimmunol.180.5.3512. [DOI] [PubMed] [Google Scholar]
- 27.Perretti M, Chiang N, La M, Fierro IM, Marullo S, Getting SJ, Solito E, Serhan CN. Endogenous lipid- and peptide-derived anti-inflammatory pathways generated with glucocorticoid and aspirin treatment activate the lipoxin A4 receptor. Nature Medicine. 2002;8:1296–1302. doi: 10.1038/nm786. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 28.Planaguma A, Kazani S, Marigowda G, Haworth O, Mariani TJ, Israel E, Bleecker ER, Curran-Everett D, Erzurum SC, Calhoun WJ, Castro M, Chung KF, Gaston B, Jarjour NN, Busse WW, Wenzel SE, Levy BD. Airway lipoxin A4 generation and lipoxin A4 receptor expression are decreased in severe asthma. American Journal of Respiratory & Critical Care Medicine. 2008;178:574–582. doi: 10.1164/rccm.200801-061OC. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 29.Serhan CN, Chiang N, Van Dyke TE. Resolving inflammation: dual anti-inflammatory and pro-resolution lipid mediators. Nat Rev Immunol. 2008;8:349–361. doi: 10.1038/nri2294. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 30.Byersdorfer CA, Chaplin DD. Visualization of early APC/T cell interactions in the mouse lung following intranasal challenge. J Immunol. 2001;167:6756–6764. doi: 10.4049/jimmunol.167.12.6756. [DOI] [PubMed] [Google Scholar]
- 31.Jakubzick C, Helft J, Kaplan TJ, Randolph GJ. Optimization of methods to study pulmonary dendritic cell migration reveals distinct capacities of DC subsets to acquire soluble versus particulate antigen. J Immunol Methods. 2008;337:121–131. doi: 10.1016/j.jim.2008.07.005. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 32.Thornton EE, Looney MR, Bose O, Sen D, Sheppard D, Locksley R, Huang X, Krummel MF. Spatiotemporally separated antigen uptake by alveolar dendritic cells and airway presentation to T cells in the lung. J Exp Med. 2012 doi: 10.1084/jem.20112667. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 33.Woodruff PG, Boushey HA, Dolganov GM, Barker CS, Yang YH, Donnelly S, Ellwanger A, Sidhu SS, Dao-Pick TP, Pantoja C, Erle DJ, Yamamoto KR, Fahy JV. Genome-wide profiling identifies epithelial cell genes associated with asthma and with treatment response to corticosteroids. Proc Natl Acad Sci U S A. 2007;104:15858–15863. doi: 10.1073/pnas.0707413104. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 34.Yang L, Cohn L, Zhang DH, Homer R, Ray A, Ray P. Essential role of nuclear factor kappaB in the induction of eosinophilia in allergic airway inflammation. J Exp Med. 1998;188:1739–1750. doi: 10.1084/jem.188.9.1739. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 35.Terawaki K, Yokomizo T, Nagase T, Toda A, Taniguchi M, Hashizume K, Yagi T, Shimizu T. Absence of leukotriene B4 receptor 1 confers resistance to airway hyperresponsiveness and Th2-type immune responses. J Immunol. 2005;175:4217–4225. doi: 10.4049/jimmunol.175.7.4217. [DOI] [PubMed] [Google Scholar]
- 36.Wong DT, Elovic A, Matossian K, Nagura N, McBride J, Chou MY, Gordon JR, Rand TH, Galli SJ, Weller PF. Eosinophils from patients with blood eosinophilia express transforming growth factor beta 1. Blood. 1991;78:2702–2707. [PubMed] [Google Scholar]
- 37.Alcorn JF, Rinaldi LM, Jaffe EF, van Loon M, Bates JH, Janssen-Heininger YM, Irvin CG. Transforming growth factor-beta1 suppresses airway hyperresponsiveness in allergic airway disease. Am J Respir Crit Care Med. 2007;176:974–982. doi: 10.1164/rccm.200702-334OC. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 38.McMillan SJ, Xanthou G, Lloyd CM. Manipulation of allergen-induced airway remodeling by treatment with anti-TGF-beta antibody: effect on the Smad signaling pathway. J Immunol. 2005;174:5774–5780. doi: 10.4049/jimmunol.174.9.5774. [DOI] [PubMed] [Google Scholar]
- 39.Alcorn JF, Crowe CR, Kolls JK. TH17 cells in asthma and COPD. Annu Rev Physiol. 2010;72:495–516. doi: 10.1146/annurev-physiol-021909-135926. [DOI] [PubMed] [Google Scholar]
- 40.Al-Ramli W, Prefontaine D, Chouiali F, Martin JG, Olivenstein R, Lemiere C, Hamid Q. T(H)17-associated cytokines (IL-17A and IL-17F) in severe asthma. J Allergy Clin Immunol. 2009;123:1185–1187. doi: 10.1016/j.jaci.2009.02.024. [DOI] [PubMed] [Google Scholar]
- 41.Chen Y, Thai P, Zhao YH, Ho YS, DeSouza MM, Wu R. Stimulation of airway mucin gene expression by interleukin (IL)-17 through IL-6 paracrine/autocrine loop. J Biol Chem. 2003;278:17036–17043. doi: 10.1074/jbc.M210429200. [DOI] [PubMed] [Google Scholar]
- 42.Lajoie S, Lewkowich IP, Suzuki Y, Clark JR, Sproles AA, Dienger K, Budelsky AL, Wills-Karp M. Complement-mediated regulation of the IL-17A axis is a central genetic determinant of the severity of experimental allergic asthma. Nat Immunol. 2010;11:928–935. doi: 10.1038/ni.1926. [DOI] [PMC free article] [PubMed] [Google Scholar]