Abstract
In the last decade, huge steps forward have been made in the field of cartilage regeneration. The most recent trend for treating chondral/osteochondral lesions is based on the application of smart biomaterials that could lead to “in situ” regeneration of not only cartilage, but also subchondral bone, preferably through a single step procedure to reduce the costs and the morbidity for the patient. This innovative approach is currently under investigation as several “scaffolds” have been proposed in clinical practice, with or without the aid of cells, with the opportunity, in the second case, of bypassing the strict limits imposed by cell manipulation regulations. Furthermore, the fascinating potential of mesenchymal stem cells has recently opened new paths of research to discover how and whether these powerful entities can really contribute to tissue regeneration. The first clinical trials have been published but further high quality research is needed to understand their mechanisms of action, their limits, and their clinical efficacy.
Keywords: Cartilage, Osteochondral, Regeneration, Tissue engineering, Scaffold, Mesenchymal stem cells, One-step surgery
Introduction
Cartilage regeneration is not a new concept. Autologous chondrocyte implantation (ACI) was the first clinical application of cartilage regeneration and was first performed 25 years ago, in 1987, for the treatment of isolated femoral condyle lesions, with the pioneers of this ambitious treatment approach reporting their results in 1994 [1]. In the following years, several studies demonstrated both the production of a hyaline-like articular surface and a successful clinical outcome at mid- to long-term follow-up. More recently, Peterson et al reported good results, with a 92 % satisfaction rate in a series of 224 cases at 13 years of follow-up. Besides the clinical improvement, ACI has also been shown to result in viable regenerative tissue at long term followup, as documented by Vasiliadis et al despite evidence of some osteophytes, cysts, and edema on magnetic resonance imaging (MRI) [2•, 3]. However, these positive results have to be weighed against several problems, both from the biological and surgical point of views. Classic first-generation ACI requires a more extensive approach depending on the location of the defect, which increases the risk of joint stiffness or arthrofibrosis, as frequently observed with this procedure. Moreover, there is a frequent occurrence of periosteal hypertrophy that takes place between 3 and 7 months after surgery in 10 %–25 % of cases, often requiring revision surgery [4, 5]. Some authors have shown a reoperation rate of up to 42 %, due to joint stiffness, and have indicated that the use of a periosteal flap increases the risk of complications during the recovery period and results in a more difficult rehabilitation [6, 7].
The latest bioengineering research offers new technologies and new surgical treatment options for cartilage lesions. The use of three-dimensional (3-D) structures for cell growth has been shown to allow the maintenance of a chondrocyte differentiated phenotype [8] and to overcome most of the biological and surgical concerns raised by first-generation methods [7, 8]. Following these principles, Matrix-assisted autologous chondrocyte transplantation (MACT) procedures have been developed using several scaffolds [9•].
MACT was introduced into clinical practice in Europe between 1998 and 1999, and since then a considerable number of clinical studies have been published with promising good mid-term results [10–22]. The ease in handling of the bioengineered tissues allows for the use of minimally invasive surgical approaches, and even arthroscopic procedures have been developed and routinely applied in clinical practice [23, 24].
Despite the different solutions developed and the promising results, the properties of healthy cartilage tissue are still unmatched by any available treatment [9•]. Moreover, in the US the Food and Drug Administration (FDA) has not yet approved MACT, and even in countries where this treatment is available some fundamental aspects are limiting its clinical use, namely its cost and requirement of a 2-step surgery.
In an attempt to overcome these limitations, different treatment options are being developed (Fig. 1) aimed at avoiding cell manipulation and its inherent regulatory obstacles, while also simplifying surgical procedures. We will describe in detail these new trends for cartilage regenerative treatment.
Fig. 1.
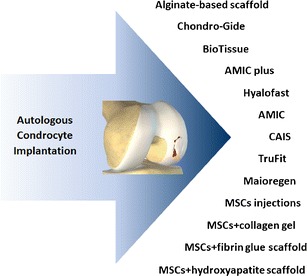
New trends for cartilage repair
Scaffold based procedures
The solutions proposed for providing both scaffold and cells in a one-step surgery are varied [25–29]. Cole et al [26] harvested healthy cartilage tissue from an unaffected area of the injured joint, mechanically fragmented and then embedded it into a 3-D polymeric reabsorbable scaffold (copolymer foam of 35 % polycaprolactone (PCL) and 65 % polyglycolic acid (PGA), reinforced with polydioxanone (PDO) mesh; Advanced Technologies and Regenerative Medicine, Raynham, Massachusetts, USA), that was implanted into the articular cartilage defect. The results of this cartilage autograft implantation system (CAIS) were reported in a randomized study demonstrating better subjective results at 2 years compared with microfracture. MRI evaluation demonstrated no differences in defect filling, tissue integration, or subchondral cysts, although more intralesional osteophyte formation was documented in the microfracture group.
Bone marrow concentrate (BMC) can be used instead of chondrocytes to provide mesenchymal stem cells (MSCs) to seed a scaffold. Buda et al [27] reported the use of a hyaluronic acid membrane (Hyalofast; Fidia Advanced Biopolymers, Abano Terme, Italy) filled with bone marrow concentrate and covered with a layer of platelet-rich fibrin in a one-step procedure. The authors reported overall good results in their series of 20 patients with clinical improvement demonstrated at 2 year follow-up, and 80 % graft integration and 70 % defect fill on MRI. Gobbi et al [28] used a similar approach, applying both scaffold and bone marrow concentrate, but in their case, they used an activated and clotted bone marrow concentrate to fill the defect, which was subsequently covered by a collagen I/III matrix (Chondro-Gide; Geistlich, Wolhusen, Switzerland) sutured and sealed with fibrin glue. Fifteen middle-aged patients were treated and evaluated at 2 years, showing promising results for lesions of all sizes, although the best results were obtained in smaller, isolated lesions. MRI showed coverage of the lesion with hyaline-like tissue, as confirmed by the histologic biopsy analysis.
Almqvist et al [29] proposed another one-step cell-based strategy, implanting mature human allogenic chondrocytes in a biodegradable alginate-based scaffold (Sigma, St Louis, Missouri, USA) in 21 patients, and observed no adverse reactions and significant clinical improvement at 2 year follow-up, although hyaline-like tissue was only found in a small percentage of patients.
As an alternative to cell seeded scaffolds, another treatment approach that is gaining interest involves the implantation of acellular biomaterials for “in situ” cartilage regeneration by stimulating bone marrow stem-cell recruitment and differentiation induced by the scaffold. In fact, an ideal graft would be an off-the-shelf product from both a surgical and commercial standpoint. The potential of creating a cell-free implant that is “smart” enough to provide the joint with the appropriate stimuli to induce orderly and durable tissue regeneration is attractive, and new biomaterials have been recently proposed to induce “in situ” cartilage regeneration after direct transplantation onto the defect site.
One of these cell-free procedures is autologous matrix-induced chondrogenesis (AMIC), a technique that combines microfracture with the implantation of a porcine collagen type-I/III bilayer scaffold to stabilise the blood clot in a one step procedure. Gille et al [30] reported highly satisfactory results in 87 % of the 27 patients evaluated at a mean follow up of 37 months, with MRI showing moderate-to-complete filling and a normal-to-hyperintense signal in most cases. Patrascu et al [31] used another scaffold, an absorbable non-woven polyglycolic acid textile treated with hyaluronic acid (BioTissue AG, Zurich, Switzerland), as a sponge to hold the blood clot and progenitor cells. The scaffold is fixed to the lesion site with resorbable treads after a standard microfracture procedure is performed. The authors reported the successful treatment of a 6 cm2 post-traumatic medial femoral condyle defect after 2 years. Pascarella et al applied a similar approach [32] combining a collagen patch (Chondro-Gide; Geistlich, Wolhusen, Switzerland) with 15-mm deep perforations made with a 2-mm Kirschner wire, to exploit the advantages of the Pridie technique, which may allow a greater number of MSCs to enrich the membrane. They reported good results in 19 patients evaluated at a median 2-year follow-up. Dhollander et al [33] proposed an AMIC “plus” technique for the treatment of patellar lesions. In a pilot study of 5 patients, the classic AMIC procedure was combined with the application of platelet-rich plasma gel, which aimed at further enhancing the healing response through the platelet-derived growth factors. While good clinical results were demonstrated at 2 years, there is not enough conclusive data to determine the effectiveness of this combined approach. Recently, Schiavone Panni et al [34] reported the use of a modified AMIC technique (drilling+fibrin glue) involving 17 patients evaluated at a mean of 36 months, with 76.5 % of patients satisfied or extremely satisfied and a 58.8 % reduction of defect area and subchondral bone edema at MRI. Finally, Kusano et al [35], in a retrospective evaluation of 38 patients undergoing AMIC for the treatment of chondral and osteochondral defects evaluated at a mean follow-up of 28 months, reported an overall improvement in both groups, with the largest improvements in the osteochondral subgroup. Although MRI showed that tissue filling was present, it was generally incomplete and heterogeneous.
The increasing awareness of the role of subchondral bone in the pathogenesis of articular surface pathology has led to the development of new biphasic products. The bilayer structure allows the entire osteochondral unit to be addressed by reproducing the different biological and functional requirements of both bone and cartilage tissues, which is particularly important in the case of chondral defects with bony involvement [36–38].
Only 2 scaffolds for osteochondral regeneration are currently commercially available for clinical application. One is a bilayer porous polylactic-co-glycolic acid (PLGA)-calcium-solfate biopolymer (TruFit; Smith & Nephew, Andover, Massachusetts, USA). Results after implantation of this osteochondral graft substitute are controversial with no available information on long-term durability. Barber et al demonstrated in a CT study that the plugs do not show any evidence of bone ingrowth, osteoconductivity, or integration, but rather lead to subchondral cyst formation in all cases [39]. Conversely, Bedi et al [40] reported that even an unfavorable mid-term MRI can significantly improve with time, while Carmont et al [41] suggested that although an intermediate postoperative interval can be associated with unfavourable MRI images, the plug appearance may significantly improve at further follow-up. Thus they recommended perseverance, and despite delayed incorporation and maturation of the graft with the surrounding tissue, the authors reported clinical improvement in an 18-year-old footballer at 2 years.
The second osteochondral scaffold is a nanostructured 3-layer biomimetic scaffold (Maioregen; Fin-Ceramica S.p.A., Faenza, Italy) with a porous composite structure, mimicking the osteochondral anatomy with a cartilaginous Type I collagen layer containing a smooth surface, an intermediate tide-mark-like layer consisting of a combination of Type I collagen (60 %) and hydroxyapatite (40 %), and a lower layer composed of a mineralized blend of Type I collagen (30 %) and hydroxyapatite (70 %) to mimic the sub-chondral bone. This scaffold was introduced into clinical practice as a cell-free approach following animal studies showing good results in terms of both cartilage and bone tissue formation, and is hypothesized to induce in situ regeneration through stem cells coming from the surrounding bone marrow [37, 42]. After a promising preliminary evaluation of early implant stability [43], longer term results have been recently reported. A case report on a 46-year-old man affected by multi-focal degenerative chondral lesions documented good results at 1 year, with return to previous level of athletic activity and restoration of the articular surface [44]. Subsequently, a pilot study of 28 patients affected by chondral and osteochondral lesions confirmed these positive findings showing a slower recovery in older, less active patients and poorer results in patellar lesions. However at 2-year follow-up, good results were reported in all patients with both clinical and MRI evaluations showing the potential of this osteochondral one-step procedure for the treatment of both chondral and osteochondral lesions [45].
Cell based procedures
Newer cell-based strategies for the treatment of cartilage lesions are based on the use of MSCs, rather than on differentiated chondrocytes.
MSCs are a population of non-hematopoietic stem cells first identified by Friedenstein et al [46] in 1970 within bone marrow. They are able to differentiate into a variety of connective tissues such as bone, cartilage, fat, tendon and ligament, marrow stroma, and others [47–49], and for this reason MSCs are currently emerging as a powerful alternative source of cells in orthopaedic tissue engineering.
The feasibility, efficacy and safety of autologous MSC implantation for the treatment of cartilage defects were first reported by Wakitani et al [50] in a rabbit osteochondral defect model. These results were confirmed by Caplan et al [51] using the same animal model, demonstrating that MSCs transplanted into full-thickness cartilage defects can recreate the layered arrangement of articular cartilage.
Subsequently, numerous researchers have demonstrated that the regenerative effects of MSCs are due to their ability to stimulate tissue repair while also providing an immunomodulatory and anti-inflammatory effect, through direct cell–cell interaction or secretion of bioactive molecules [52]. Moreover, it has been shown that these properties are likely the reason that patients receiving treatment with allogeneic human MSCs did not show anti-allogeneic MSCs antibody production [53]. However, different factors, such as isolation methods, culture surface, culture medium, seeding density as well as donor age and disease stage can influence the expansion and differentiation capacity of MSCs [52].
Currently, MSCs can be isolated from sources other than bone marrow, such as adipose tissue, umbilical cord blood, synovial membrane, synovial fluid, periosteum, dermis, trabecular bone, infrapatellar fat pad, muscle, and articular cartilage with similar phenotypic characteristics but different proliferation and differentiation potentials [49]. For example, synovium-derived MSCs show the highest chondrogenic capacity, followed by bone marrow-derived and periosteum derived MSCs [52, 54]. An in vitro study performed by Sakaguchi et al [54] confirmed that synovium derived MSCs are superior in terms of chondrogenesis, while bone marrow, synovium, and periosteum derived MSCs are superior in osteogenesis.
In human clinical settings, there are few papers regarding MSC application. While current reports are promising, they are still preliminary, due to small sample sizes, short term follow-up and variations among treatment protocols [58–63, 64•, 65–67].
BMSCs were the first MSCs type identified and the ease of collection along with the relatively high concentration of MSCs still make it a commonly used source of MSCs [55], which can be employed as a cell suspension expanded by culture (BMSCs) or just as BMC [56]. Various biomaterials have been applied as vehicles for intra-articular MSCs delivery [57], in particular collagen hydrogel. The first evidence of BMSCs embedded in collagen hydrogel and covered with periosteum was described by Wakitani, et al [58]. Twelve osteoarthritic knees, undergoing high tibial osteotomy, were concurrently treated with this novel construct, whereas 12 patients underwent high tibial osteotomy alone. At 16 months, while similar clinical improvement was found in both groups, better arthroscopic and histologic scores were seen in the cell-treated group. These promising results led to a new study performed by the same research group [59] 2 years later, using BMSCs and a collagen construct in 2 patients suffering from full-thickness cartilage defects. Patients demonstrated clinical improvement at 6 months, which was maintained at 4- and 5-year follow-up. Good results were also described in a case report by Kuroda et al [60], who implanted a collagen hydrogel and BMSCs on a full thickness cartilage defect of a 31-year-old male judo player. They reported hyaline-like tissue at histologic evaluation and a return to sport at the previous level just 1 year after surgery. More recently, Kasemkijwattana et al [61] reported good defect filling and repair tissue stiffness with cultured BMSCs on a collagen scaffold in 2 patients with knee osteoarthritis.
A case report performed by Adachi et al [62] investigated the use of a novel construct based on BMSCs and hydroxyapatite ceramic for knee osteochondral defects showing both cartilage and subchondral bone tissue regeneration.
Haleem et al [63] used BMSCs on a platelet fibrin glue scaffold for the treatment of knee articular cartilage defects in 5 patients. Patients had improvement in symptoms at 12 months and MRI evaluation revealed complete defect filling in 3 patients, whereas in the remaining 2 cases incomplete congruity was observed.
Recently Nejadnik et al [64•] compared the ACI technique with BMSCs implantation, obtaining a similar pattern of clinical and subjective improvement in 72 full-thickness knee cartilage defects, with BMSCs representing an advantage in terms of costs and donor site morbidity.
Intra-articular injections of MSCs have gained a lot of interest due to the simplicity of the technique and ease of administration; however it is likely only suitable for the early stages of the disease when the defect is restricted to the cartilage layer, whereas a scaffold or matrix would be required in cases of large subchondral bone exposure [57]. A case report by Centeno et al [65] reported encouraging results after treating a knee cartilage lesion with a simple approach consisting of a single injection of BMSCs. At 6 months, MRI showed an increase of cartilage and meniscus volume and improvement in range of motion and pain scores. Intra-articular injections of BMSCs were used also by Davatchi et al [66] in 4 osteoarthritic knees, reporting marked improvement in subjective parameters but less favourable outcome concerning physical performance. An interesting example of improving an already existing technique was described by Gigante et al [67], who used AMIC augmented with BMSCs to treat 5 patients with medial femoral condyle lesions. Upon a second look arthroscopy, they found a nearly normal arthroscopic appearance, although hyaline-like matrix was seen only in 1 case on histology.
Currently, adipose derived MSCs (ADMSCs) obtained from lipo-aspirates represents a great opportunity for cartilage tissue engineering due to their abundance and easy availability [68, 69]. It has been reported that 1 hurdle for their use is the lower chondrogenic potential, but this problem could be overcome using a combination of TGF-b and BMP, potent inducers of MSCs chondrogenesis [52, 70]. Pak et al [71] reported a significant increase in cartilage thickness on MRI and functional status improvement at 3 months using injections of adipose-derived stem-cell concentrate in the treatment of 2 osteoarthritic knees. MSCs were applied together with dexamethasone, platelet rich plasma, and hyaluronic acid injections.
Discussion
Cartilage regeneration is a challenging problem faced by both surgeons in the operating room and basic scientists in the laboratory aimed at restoring a hyaline-like tissue with normal biomechanical characteristics.
The use of 3-D structures for cell growth has been shown to promote the maintenance of a chondrocyte differentiated phenotype, while also simplifying the implant procedure. There are several scaffolds currently in clinical use offering a comparable clinical outcome to first-generation ACI, while overcoming most of the biological and surgical limitations [9•]. As polymers can be designed to have a wide range of properties and can be easily modified depending on the biological/surgical strategy, many more are being developed. Several other natural and synthetic scaffolds for cartilage regeneration are under investigation and will soon be available for clinical applications [7, 9•]. In particular, hydrogels are an attractive evolution of cartilage tissue engineering, while another innovation comes from photopolymerization, whereby liquid or gel scaffolds can be injected into the injury site and then polymerized by exposure to ultraviolet light allowing for a less invasive procedure. It is also possible to encapsulate cells within the gels obtaining a scaffold with uniformly distributed cells, thus offering potential surgical and biological advantages [72].
To further improve the available procedures, different strategies are being studied, mainly focusing on 2 aspects: simplifying the surgical technique and adopting more powerful agents to stimulate tissue regeneration.
One-step cell-free procedures have been developed to avoid the problems related to chondrocyte culture and expansion in scaffolds and also to reduce costs and surgical time. In fact, there is an increasing awareness that the role of scaffolds is not only to deliver cells, but to enhance tissue regeneration. For this reason, the use of cell-free scaffolds has been proposed and is gaining popularity. Among these new treatment options, osteochondral scaffolds have been proposed to treat lesions where the subchondral layer is also involved in the pathologic process.
Regenerative techniques promote the restoration of articular cartilage with a hyaline-like tissue, and the use of scaffolds has simplified and further improved the potential of this treatment approach, but the properties of the healthy cartilage tissue are still unmatched by any available substitute. Therefore, MSCs are being studied, given their regenerative potential with the possibility of producing a more physiologic repair. Results of these new procedures seem to be promising, but their real potential has still to be demonstrated and many aspects can be further improved. Moreover, for the time being, there is no agreement about the effective superiority of 1 regenerative approach over the others, and both results and indications remain controversial. One explanation for the contradictory and inconclusive findings in the literature may be that regenerative procedures may lead to a hyaline-like tissue through a remodeling process, thus leading to superior clinical results detectable only after 2–3 years of follow-up. Unfortunately, due to the recent development of these techniques, there are no high level studies with adequate long-term follow-up.
The regulation of tissue healing and regeneration are complex processes and further biological studies, as well as systematic long-term evaluation of the emerging treatment options, are necessary to clarify the role of the many variables that could influence the results, such as cells, growth factors, and different biomaterial properties. Future evaluations of these new techniques must also involve comparative studies with current, proven treatment options.
Conclusions
New regenerative procedures are emerging as promising treatments for chondral and osteochondral lesions. One-step scaffold-based strategies have been recently developed to simplify the procedure and further improve the results. While several scaffolds are currently available for clinical application, well-designed studies comparing the effectiveness of the various options are lacking. Another emerging strategy involves the use of MSCs in order to provide a source of cells with higher regenerative potential, but this approach is still in its infancy.
Randomized controlled trials are necessary to evaluate these new regenerative approaches, to clearly demonstrate their potential, their limits, their indications, and also to highlight advantages and disadvantages with respect to the currently available procedures for the treatment of chondral and osteochondral lesions.
Acknowledgments
Disclosure
No potential conflicts of interest relevant to this article were reported.
Contributor Information
Elizaveta Kon, Email: e.kon@biomec.ior.it.
Giuseppe Filardo, Email: g.filardo@biomec.ior.it.
Alice Roffi, Email: a.roffi@biomec.ior.it.
Luca Andriolo, Email: lucas.andriolo@gmail.com.
Maurilio Marcacci, Email: m.marcacci@biomec.ior.it.
References
Papers of particular interest, published recently, have been highlighted as: • Of importance
- 1.Brittberg M, Lindahl A, Nilsson A, Ohlsson C, Isaksson O, Peterson L. Treatment of deep cartilage defects in the knee with autologous chondrocyte transplantation. N Engl J Med. 1994;331:889–95. doi: 10.1056/NEJM199410063311401. [DOI] [PubMed] [Google Scholar]
- 2.Peterson L, Vasiliadis HS, Brittberg M, Lindahl A. Autologous chondrocyte implantation: a long-term follow-up. Am J Sports Med. 2010;38:1117–1124. doi: 10.1177/0363546509357915. [DOI] [PubMed] [Google Scholar]
- 3.Vasiliadis HS, Danielson B, Ljungberg M, McKeon B, Lindahl A, Peterson L. Autologous chondrocyte implantation in cartilage lesions of the knee: long-term evaluation with magnetic resonance imaging and delayed gadolinium-enhanced magnetic resonance imaging technique. Am J Sports Med. 2010;38:943–949. doi: 10.1177/0363546509358266. [DOI] [PubMed] [Google Scholar]
- 4.Peterson L, Brittberg M, Kiviranta I, Akerlund EL, Lindahl A. Autologous chondrocyte transplantation. Biomechanics and long-term durability. Am J Sports Med. 2002;30:2–12. doi: 10.1177/03635465020300011601. [DOI] [PubMed] [Google Scholar]
- 5.Browne JE, Anderson AF, Arciero R, Mandelbaum B, Moseley JB, Jr, Micheli LJ, et al. Clinical outcome of autologous chondrocyte implantation at 5 years in US subjects. Clin Orthop Relat Res. 2005;436:237–245. doi: 10.1097/00003086-200507000-00036. [DOI] [PubMed] [Google Scholar]
- 6.Micheli LJ, Browne JE, Erggelet C, Fu F, Mandelbaum B, Moseley JB, Zurakowski D. Autologous chondrocyte implantation of the knee: multicenter experience and minimum 3-year follow-up. Clin J Sport Med. 2001;11:223–228. doi: 10.1097/00042752-200110000-00003. [DOI] [PubMed] [Google Scholar]
- 7.Kon E, Delcogliano M, Filardo G, Montaperto C, Marcacci M. Second generation issues in cartilage repair. Sports Med Arthrosc. 2008;16:221–229. doi: 10.1097/JSA.0b013e31818cdbc5. [DOI] [PubMed] [Google Scholar]
- 8.Grigolo B, Lisignoli G, Piacentini A, Fiorini M, Gobbi P, Mazzotti G, et al. Evidence for redifferentiation of human chondrocytes grown on a hyaluronan-based biomaterial (HYAff 11): molecular, immunohistochemical and ultrastructural analysis. Biomaterials. 2002;23:1187–1195. doi: 10.1016/S0142-9612(01)00236-8. [DOI] [PubMed] [Google Scholar]
- 9.Kon E, Verdonk P, Condello V, Delcogliano M, Dhollander A, Filardo G, et al. Matrix-assisted autologous chondrocyte transplantation for the repair of cartilage defects of the knee: systematic clinical data review and study quality analysis. Am J Sports Med. 2009;37(Suppl 1):156S–166S. doi: 10.1177/0363546509351649. [DOI] [PubMed] [Google Scholar]
- 10.Behrens P, Bitter T, Kurz B, Russlies M. Matrix-associated autologous chondrocyte transplantation/implantation (MATC/MACI)–5-year follow –up. Knee. 2006;13:194–202. doi: 10.1016/j.knee.2006.02.012. [DOI] [PubMed] [Google Scholar]
- 11.Ferruzzi A, Buda R, Faldini C, Vannini F, Di Caprio F, Luciani D, Giannini S. Autologous chondrocyte implantation in the knee joint: open compared with arthroscopic technique. Comparison at a minimum follow-up of five years. J Bone Joint Surg Am. 2008;90(Suppl 4):90–101. doi: 10.2106/JBJS.H.00633. [DOI] [PubMed] [Google Scholar]
- 12.Kon E, Martino A, Filardo G, Tetta C, Busacca M, Iacono F, et al. Second-generation autologous chondrocyte transplantation: MRI findings and clinical correlations at a minimum 5-year follow-up. Eur J Radiol. 2011;79:382–388. doi: 10.1016/j.ejrad.2010.04.002. [DOI] [PubMed] [Google Scholar]
- 13.Kon E, Filardo G, Berruto M, Benazzo F, Zanon G, Della Villa S, Marcacci M. Articular cartilage treatment in high-level male soccer players: a prospective comparative study of arthroscopic second-generation autologous chondrocyte implantation vs microfracture. Am J Sports Med. 2011;39:2549–2557. doi: 10.1177/0363546511420688. [DOI] [PubMed] [Google Scholar]
- 14.Kon E, Filardo G, Condello V, Collarile M, Martino A, Zorzi C, Marcacci M. Second-Generation Autologous Chondrocyte Implantation: results in patients older than 40 years. Am J Sports Med. 2011;39:1668–1675. doi: 10.1177/0363546511404675. [DOI] [PubMed] [Google Scholar]
- 15.Kon E, Gobbi A, Filardo G, et al. Arthroscopic second-generation autologous chondrocyte implantation compared with microfracture for chondral lesions of the knee: prospective nonrandomized study at 5 years. Am J Sports Med. 2009;37:33–41. doi: 10.1177/0363546508323256. [DOI] [PubMed] [Google Scholar]
- 16.Gobbi A, Kon E, Berruto M, et al. Patellofemoral full-thickness chondral defects treated with second-generation autologous chondrocyte implantation: results at 5 years' follow-up. Am J Sports Med. 2009;37:1083–1092. doi: 10.1177/0363546509331419. [DOI] [PubMed] [Google Scholar]
- 17.Filardo G, Kon E, Berruto M, Di Martino A, Patella S, Marcheggiani Muccioli GM, et al. Arthroscopic second generation autologous chondrocytes implantation associated with bone grafting for the treatment of knee osteochondritis dissecans: results at 6 years. Knee. 2011;Nov 22. [DOI] [PubMed]
- 18.Filardo G, Kon E, Di Martino A, Patella S, Altadonna G, Balboni F, et al. Second-generation arthroscopic autologous chondrocyte implantation for the treatment of degenerative cartilage lesions. Knee Surg Sports Traumatol Arthrosc. 2011;Nov 1. [DOI] [PubMed]
- 19.Filardo G, Kon E, Martino A, Iacono F, Marcacci M. Arthroscopic second-generation autologous chondrocyte implantation: a prospective 7-year follow-up study. Am J Sports Med. 2011;39:2153–2160. doi: 10.1177/0363546511415658. [DOI] [PubMed] [Google Scholar]
- 20.Nehrer S, Dorotka R, Domayer S, Stelzeneder D, Kotz R. Treatment of full-thickness chondral defects with hyalograft C in the knee: a prospective clinical case series with 2 to 7 years' follow-up. Am J Sports Med. 2009;37 Suppl 1:81S–7S. [DOI] [PubMed]
- 21.Ebert JR, Robertson WB, Woodhouse J, Fallon M, Zheng MH, Ackland T, Wood DJ. Clinical and magnetic resonance imaging-based outcomes to 5 years after matrix-induced autologous chondrocyte implantation to address articular cartilage defects in the knee. Am J Sports Med. 2011;39:753–763. doi: 10.1177/0363546510390476. [DOI] [PubMed] [Google Scholar]
- 22.Macmull S, Jaiswal PK, Bentley G, Skinner JA, Carrington RW, Briggs TW. The role of autologous chondrocyte implantation in the treatment of symptomatic chondromalacia patellae. Int Orthop. 2012;36:1371–7. [DOI] [PMC free article] [PubMed]
- 23.Marcacci M, Zaffagnini S, Kon E, Visani A, Iacono F, Loreti I. Arthroscopic autologous chondrocyte transplantation: technical note. Knee Surg Sports Traumatol Arthrosc. 2002;10:154–159. doi: 10.1007/s00167-001-0275-6. [DOI] [PubMed] [Google Scholar]
- 24.Erggelet C, Sittinger M, Lahm A. The arthroscopic implantation of autologous chondrocytes for the treatment of full-thickness cartilage defects of the knee joint. Arthroscopy. 2003;19:108–110. doi: 10.1053/jars.2003.50025. [DOI] [PubMed] [Google Scholar]
- 25.Gobbi A, Bathan L. Biological approaches for cartilage repair. J Knee Surg. 2009;22:36–44. doi: 10.1055/s-0030-1247726. [DOI] [PubMed] [Google Scholar]
- 26.Cole BJ, Farr J, Winalski CS, Hosea T, Richmond J, Mandelbaum B, et al. Outcomes after a single-stage procedure for cell-based cartilage repair: a prospective clinical safety trial with 2-year follow-up. Am J Sports Med. 2011;39:1170–9. [DOI] [PubMed]
- 27.Buda R, Vannini F, Cavallo M, Grigolo B, Cenacchi A, Giannini S. Osteochondral lesions of the knee: a new one-step repair technique with bone-marrow-derived cells. J Bone Joint Surg Am. 2010;92 Suppl 2:2–11. [DOI] [PubMed]
- 28.Gobbi A, Karnatzikos G, Scotti C, Mahajan V, Mazzucco L, Grigolo B. One-step cartilage repair with bone marrow aspirate concentrated cells and collagen matrix in full-thickness knee cartilage lesions: results at 2-year follow-up. Cartilage. 2011;2:286–299. doi: 10.1177/1947603510392023. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 29.Almqvist KF, Dhollander AA, Verdonk PC, Forsyth R, Verdonk R, Verbruggen G. Treatment of cartilage defects in the knee using alginate beads containing human mature allogenic chondrocytes. Am J Sports Med. 2009;37:1920–1929. doi: 10.1177/0363546509335463. [DOI] [PubMed] [Google Scholar]
- 30.Gille J, Schuseil E, Wimmer J, Gellissen J, Schulz AP, Behrens P. Mid-term results of Autologous Matrix-Induced Chondrogenesis for treatment of focal cartilage defects in the knee. Knee Surg Sports Traumatol Arthrosc. 2010;18:1456–64. [DOI] [PubMed]
- 31.Patrascu JM, Freymann U, Kaps C, Poenaru DV. Repair of a post-traumatic cartilage defect with a cell-free polymer-based cartilage implant: a follow-up at two years by MRI and histological review. J Bone Joint Surg Br. 2010;92:1160–1163. doi: 10.1302/0301-620X.92B8.24341. [DOI] [PubMed] [Google Scholar]
- 32.Pascarella A, Ciatti R, Pascarella F, Latte C, Salvatore MG, Liguori L, Iannella G. Treatment of articular cartilage lesions of the knee joint using a modified AMIC technique. Knee Surg Sports Traumatol Arthrosc. 2010;18:509–513. doi: 10.1007/s00167-009-1007-6. [DOI] [PubMed] [Google Scholar]
- 33.Dhollander AA, De Neve F, Almqvist KF, Verdonk R, Lambrecht S, Elewaut D, et al. Autologous matrix-induced chondrogenesis combined with platelet-rich plasma gel: technical description and a five pilot patients report. Knee Surg Sports Traumatol Arthrosc. 2011;19:536–42. [DOI] [PubMed]
- 34.Schiavone Panni A, Cerciello S, Vasso M. The manangement of knee cartilage defects with modified amic technique: preliminary results. Int J Immunopathol Pharmacol. 2011;24(1 Suppl 2):149–152. doi: 10.1177/03946320110241s228. [DOI] [PubMed] [Google Scholar]
- 35.Kusano T, Jakob RP, Gautier E, Magnussen RA, Hoogewoud H, Jacobi M. Treatment of isolated chondral and osteochondral defects in the knee by autologous matrix-induced chondrogenesis (AMIC). Knee Surg Sports Traumatol Arthrosc. 2011;Dec 25. [DOI] [PubMed]
- 36.Pape D, Filardo G, Kon E, van Dijk CN, Madry H. Disease-specific clinical problems associated with the subchondral bone. Knee Surg Sports Traumatol Arthrosc. 2010;18:448–62. [DOI] [PubMed]
- 37.Kon E, Mutini A, Arcangeli E, Delcogliano M, Filardo G, Nicoli Aldini N, et al. Novel nanostructured scaffold for osteochondral regeneration: pilot study in horses. J Tissue Eng Regen Med. 2010;4:300–8. [DOI] [PubMed]
- 38.Kon E, Filardo G, Delcogliano M, Fini M, Salamanna F, Giavaresi G. Platelet autologous growth factors decrease the osteochondral regeneration capability of a collagen-hydroxyapatite scaffold in a sheep model. BMC Musculoskelet Disord. 2010;11:220. [DOI] [PMC free article] [PubMed]
- 39.Barber FA, Dockery WD. A computed tomography scan assessment of synthetic multiphase polymer scaffolds used for osteochondral defect repair. Arthroscopy. 2011;27:60–64. doi: 10.1016/j.arthro.2010.06.023. [DOI] [PubMed] [Google Scholar]
- 40.Bedi A, Foo LF, Williams RJ, Potter HG, the Cartilage Study Group. The maturation of synthetic scaffolds for osteochondral donor sites of the knee: an MRI and T2-Mapping Analysis. Cartilage. 2010;1:20–8. [DOI] [PMC free article] [PubMed]
- 41.Carmont MR, Carey-Smith R, Saithna A, Dhillon M, Thompson P, Spalding T. Delayed incorporation of a TruFit plug: perseverance is recommended. Arthroscopy. 2009;25:810–4. [DOI] [PubMed]
- 42.Kon E, Delcogliano M, Filardo G, Fini M, Giavaresi G, Francioli S, et al. Orderly osteochondral regeneration in a sheep model using a novel nano-composite multilayered biomaterial. J Orthop Res. 2010;28:116–24. [DOI] [PubMed]
- 43.Kon E, Delcogliano M, Filardo G, Pressato D, Busacca M, Grigolo B, et al. A novel nano-composite multi-layered biomaterial for treatment of osteochondral lesions: technique note and an early stability pilot clinical trial. Injury. 2010;41:693–701. [DOI] [PubMed]
- 44.Kon E, Delcogliano M, Filardo G, et al. Novel nano-composite multi-layered biomaterial for the treatment of multifocal degenerative cartilage lesions. Knee Surg Sports Traumatol Arthrosc. 2009;17:1312–1325. doi: 10.1007/s00167-009-0819-8. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 45.Kon E, Delcogliano M, Filardo G, Busacca M, Di Martino A, Marcacci M. Novel nano-composite multilayered biomaterial for osteochondral regeneration: a pilot clinical trial. Am J Sports Med. 2011;39:1180–90. [DOI] [PubMed]
- 46.Friedenstein AJ, Gorskaja JF, Kulagina NN. Fibroblast precursors in normal and irradiated mouse hematopoietic organs. Exp Hematol. 1976;4:267–274. [PubMed] [Google Scholar]
- 47.Smith GD, Knutsen G, Richardson JB. A clinical review of cartilage repair techniques. J Bone Joint Surg Br. 2005;87:445–449. doi: 10.1302/0301-620X.87B4.15971. [DOI] [PubMed] [Google Scholar]
- 48.Caplan AI, Dennis JE. Mesenchymal stem cells as trophic mediators. J Cell Biochem. 2006;98:1076–1084. doi: 10.1002/jcb.20886. [DOI] [PubMed] [Google Scholar]
- 49.Lodi D, Iannitti T, Palmieri B. Stem cells in clinical practice: applications and warnings. J Exp Clin Cancer Res. 2011;30:9. doi: 10.1186/1756-9966-30-9. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 50.Wakitani S, Goto T, Pineda SJ, Young RG, Mansour JM, Caplan AI, Goldberg VM. Mesenchymal cell-based repair of large, full-thickness defects of articular cartilage. J Bone Joint Surg Am. 1994;76:579–92. [DOI] [PubMed]
- 51.Caplan AI, Elyaderani M, Mochizuki Y, Wakitani S, Goldberg VM. Principles of cartilage repair and regeneration. Clin Orthop Relat Res. 1997;342:254–69. [PubMed]
- 52.Chen FH, Tuan RS. Mesenchymal stem cells in arthritic diseases. Arthritis Res Ther. 2008;10:223–234. doi: 10.1186/ar2514. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 53.Sundin M, Ringdén O, Sundberg B, Nava S, Götherström C, Le Blanc K. No alloantibodies against mesenchymal stromal cells, but presence of anti-fetal calf serum antibodies, after transplantation in allogeneic hematopoietic stem cell recipients. Haematologica. 2007;92:1208–15. [DOI] [PubMed]
- 54.Sakaguchi Y, Sekiya I, Yagishita K, Muneta T. Comparison of human stem cells derived from various mesenchymal tissues. Superiority of synovium as a cell source. Arthritis Rheum. 2005;52:2521–2529. doi: 10.1002/art.21212. [DOI] [PubMed] [Google Scholar]
- 55.Mosna F, Sensebé L, Krampera M. Human bone marrow and adipose tissue mesenchymal stem cells: a user's guide. Stem Cells Dev. 2010;19:1449–1470. doi: 10.1089/scd.2010.0140. [DOI] [PubMed] [Google Scholar]
- 56.Krampera M, Pizzolo G, Aprili G, Franchini M. Mesenchymal stem cells for bone, cartilage, tendon and skeletal muscle repair. Bone. 2006;39:678–683. doi: 10.1016/j.bone.2006.04.020. [DOI] [PubMed] [Google Scholar]
- 57.Noth U, Steinert AF, Tuan RS. Technology insight: adult mesenchymal stem cells for osteoarthritis therapy. Nat Clin Pract Rheumatol. 2008;4:371–380. doi: 10.1038/ncprheum0816. [DOI] [PubMed] [Google Scholar]
- 58.Wakitani S, Imoto K, Yamamoto T, Saito M, Murata N, Yoneda M. Human autologous culture expanded bone marrow mesenchymal cell transplantation for repair of cartilage defects in osteoarthritic knees. Osteoarthritis Cartilage. 2002;10:199–206. [DOI] [PubMed]
- 59.Wakitani S, Mitsuoka T, Nakamura N, Toritsuka Y, Nakamura Y, Horibe S. Autologous bone marrow stromal cell transplantation for repair of full-thickness articular cartilage defects in human patellae: two case reports. Cell Transplant. 2004;13:595–600. [DOI] [PubMed]
- 60.Kuroda R, Ishida K, Matsumoto T, Akisue T, Fujioka H, Mizuno K, et al. Treatment of a full-thickness articular cartilage defect in the femoral condyle of an athlete with autologous bone-marrow stromal cells. Osteoarthritis Cartilage. 2007;15:226–31. [DOI] [PubMed]
- 61.Kasemkijwattana C, Hongeng S, Kesprayura S, et al. Autologous bone marrow mesenchymal stem cells implantation for cartilage defects: two cases report. J Med Assoc Thai. 2011;94:395–400. [PubMed] [Google Scholar]
- 62.Adachi N, Ochi M, Deie M, Ito Y. Transplant of mesenchymal stem cells and hydroxyapatite ceramics to treat severe osteochondral damage after septic arthritis of the knee. J Rheumatol. 2005;32:1615–1618. [PubMed] [Google Scholar]
- 63.Haleem AM, Singergy AA, Sabry D, Atta HM, Rashed LA, Chu CR, et al. The clinical use of human culture-expanded autologous bone marrow mesenchymal stem cells transplanted on Platelet-Rich Fibrin Glue in the treatment of Articular Cartilage Defects: a pilot study and preliminary results. Cartilage. 2010;1:253–61. [DOI] [PMC free article] [PubMed]
- 64.• Nejadnik H, Hui JH, Feng Choong EP, Tai BC, Lee EH. Autologous bone marrow-derived mesenchymal stem cells vs autologous chondrocyte implantation: an observational cohort study. Am J Sports Med. 2010;38:1110–6. The Comparison Between Autologous Chondrocyte Implantation And Autologous Bone Marrow-Derived Mesenchymal Stem Cells Implantation Showed The Potential Advantage Of The New Cell-Based Approach, With Similar Results But Lower Cost And Morbidity With Respect To The Classic ACI Technique. [DOI] [PubMed]
- 65.Centeno CJ, Busse D, Kisiday J, Keohan C, Freeman M, Karli D. Increased knee cartilage volume in degenerative joint disease using percutaneously implanted, autologous mesenchymal stem cells. Pain Physician. 2008;11:343–53. [PubMed]
- 66.Davatchi F, Abdollahi BS, Mohyeddin M, Shahram F, Nikbin B. Mesenchymal stem cell therapy for knee osteoarthritis. Preliminary report of four patients. Int J Rheum Dis. 2011;14:211–5. [DOI] [PubMed]
- 67.Gigante A, Calcagno S, Cecconi S, Ramazzotti D, Manzotti S, Enea D. Use of collagen scaffold and autologous bone marrow concentrate as a one-step cartilage repair in the knee: histological results of second-look biopsies at 1 year follow-up. Int J Immunopathol Pharmacol. 2011;24(1 Suppl 2):69–72. [DOI] [PubMed]
- 68.Wagner W, Wein F, Seckinger A, Frankhauser M, Wirkner U, Krause U, et al. Comparative characteristics of mesenchymal stem cells from human bone marrow, adipose tissue, and umbilical cord blood. Exp Hematol. 2005;33:1402–16. [DOI] [PubMed]
- 69.Schäffler A, Büchler C. Concise review: adipose tissue-derived stromal cells–basic and clinical implications for novel cell-based therapies. Stem Cells. 2007;25:818–827. doi: 10.1634/stemcells.2006-0589. [DOI] [PubMed] [Google Scholar]
- 70.Im GI, Lee JH. Repair of osteochondral defects with adipose stem cells and a dual growth factor-releasing scaffold in rabbits. J Biomed Mater Res B Appl Biomater. 2010;92:552–560. doi: 10.1002/jbm.b.31552. [DOI] [PubMed] [Google Scholar]
- 71.Pak J. Regeneration of human bones in hip osteonecrosis and human cartilage in knee osteoarthritis with autologous adipose-tissue-derived stem cells: a case series. J Med Case Rep. 2011;5:296. doi: 10.1186/1752-1947-5-296. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 72.Selmi TA, Verdonk P, Chambat P, Dubrana F, Potel JF, Barnouin L, et al. Autologous chondrocyte implantation in a novel alginate-agarose hydrogel: outcome at two years. J Bone Joint Surg Br. 2008;90:597–604. [DOI] [PubMed]


