Abstract
Organochlorine pesticides (OCPs) are persistent endocrine disruptors. OCPs cross the placenta; this prenatal exposure has been associated with adverse pregnancy outcomes. We investigated associations between prenatal exposure to OCPs and gestational age and birth weight in 600 infants born between 1960 and 1963. The primary OCP was 1,1,1-trichloro-2,2-bis(p-chlorophenyl)ethane (p,p′-DDT), its primary metabolite, 1,1′-dichloro-2,2'-bis(p-chlorophenyl)ethylene(p,p′-DDE) and the contaminant, 1,1,1-trichloro-2-(p-chlorophenyl)-2-(o-chlorophenyl)-ethane (o,p′-DDT). Regression analysis indicated that for each natural log unit increase in p,p′-DDT, birth weight increased by 274 grams (95% CI 122, 425) when controlling for p,p′-DDE and o,p′-DDT. At a given level of p,p′-DDT exposure, o,p′-DDT and p,p′-DDE were associated with decreased birth weight. p,p′-DDE was negatively associated with length of gestation, controlling for p,p′-DDT and o,p′-DDT. These findings suggest opposing associations between exposure to p,p′-DDT and p,p′-DDE and birth weight. We did not find evidence to support mediation by maternal thyroid hormone status nor that the association differed by sex.
Keywords: endocrine disruptors, organochlorine pesticides, DDT, prenatal exposures, birth weight, length of gestation
1. Introduction
Although banned in the 1970s, organochlorine pesticide (OCP) exposure is ubiquitous as these compounds and their metabolites persist in the environment, are lipophilic and bioaccumulate. Perhaps the best studied OCP is dichlorodiphenyltrichloroethane (DDT). DDT is a composite term frequently used to include the active ingredient 1,1,1-trichloro-2,2-bis(p-chlorophenyl)ethane (p,p′-DDT), its contaminant 1,1,1-trichloro-2-(p-chlorophenyl)-2-(o-chlorophenyl)-ethane (o,p′-DDT) and primary metabolite 1,1′-dichloro-2,2'-bis(p-chlorophenyl)ethylene (p,p′-DDE). Reproductive outcomes following DDT exposure have been studied in wildlife since the early 1960s [1]. As a result of adverse findings in those studies, numerous studies in humans were initiated. Indeed, because OCPs cross the placenta [2] hypotheses regarding decreased fetal growth, as well as developmental outcomes have been proposed.
Human exposure to p,p′-DDT and o,p′-DDT occurs primarily through direct contact with pesticide application and diet, while the source of exposure to p,p′-DDE depends on whether exposure to p,p′-DDT is active. During active exposure, humans convert p,p′-DDT to p,p′-DDE but otherwise p,p′-DDE exposure occurs largely via diet. The greater detectability and higher levels of p,p′-DDE seen in humans is a result of the longer half-life of this compound in the environment, and consequent bio-accumulation in the food chain, relative to its counterparts. Following the ban of DDT in the early 1970s, blood concentrations of p,p′-DDT, p,p′-DDE, and o,p′-DDT have decreased by approximately five fold [3, 4].
OCPs are endocrine disrupting compounds (EDCs) [5, 6]. Due to the structural similarity of p,p′-DDT and p,p′-DDE to thyroid hormones, it is plausible that p,p′-DDT or p,p′-DDE may interfere with the synthesis, transport, receptor binding or action of these hormones [7]. Though the epidemiologic evidence is conflicting, studies examining prenatal exposure to p,p′-DDT and p,p′-DDE and infant thyroid hormone levels have suggested that exposure to these OCPs is associated with increases in thyroid stimulating hormone (TSH) [8], decreases in free thyroxine (fT4) [8, 9], decreases in total thyroxine (TT4) [10], and/or decreases in (total triiodothyronine (TT3) [11, 12]. Still, other studies report no such associations [13, 14]. Both exposure to OCPs and perturbed thyroid function during pregnancy are posited to result in similar adverse pregnancy outcomes [3, 15]. Thus, it is plausible to hypothesize a mediation mechanism by which impaired maternal thyroid function, resulting from exposure to OCPs, negatively impacts the developing fetus in utero.
At present, some evidence suggests there are independent associations between adverse pregnancy outcomes and both OCP exposure [16–18] and impaired maternal thyroid function during pregnancy [15, 19, 20]. In animal studies, prenatal p,p′-DDT exposure has been associated with premature delivery [21, 22] and increased fetal resorption or abortion [21, 23]. In epidemiologic studies, associations between exposure to p,p′-DDT and p,p′- p,p′-DDE and fetal growth are less consistent. Longnecker et al. (2001) found increased odds ratios relating maternal p,p′-DDE levels above ≥ 60μg/L with preterm delivery and being for small for gestational age [16]. Similarly, Wolff et al. (2007) found an inverse association between maternal p,p′-DDE concentrations and birth weight [18]. Other studies also have found p,p′-DDT to be associated with preterm delivery [24], although many studies have found no evidence of significant associations with birth outcomes [17, 25–27]. These discrepant results may be due, in part, to issues of exposure characterization. Exposure to DDT is complex due to both contamination (o,p′-DDT) and the presence of a major metabolite (p,p′-DDE); each compound reflects timing, level and sources of exposure and host metabolism.
Studies have likewise provided evidence favoring an association between maternal thyroid dysfunction and adverse pregnancy outcomes. Several have suggested that in comparison to euthyroid mothers, mothers with hypothyroidism (elevated TSH and decreased fT4) or subclinical hypothyroidism (elevated TSH and fT4 within the normal range) are more likely to deliver preterm infants [28–30] and, to a lesser extent, infants with low birth weights [31]; the risk of these complications is greater in overtly hypothyroid mothers compared to those with sub-clinical hypothyroidism [15]. Previous studies of OCP exposure and pregnancy outcomes have not considered possible mediation pathways. Because of the vital role thyroid hormone plays in growth and development of the fetus, a mediation pathway warrants further exploration.
Here, we add to the literature by considering associations between p,p′-DDT, p,p′-DDE and o,p′-DDT and birth weight in a well characterized cohort from the 1960s. We a priori hypothesize that DDT and its components will be associated with decreased birth weight and decreased gestational age. We also explore the possible role of maternal thyroid status as a mediator.
2. Methods
2.1. Study design
The CHDS is a prospective birth cohort designed to observe women, their pregnancies and their offspring [32]. The cohort recruited members of the Kaiser Health Plan who were receiving obstetric care at the Oakland, California Kaiser Foundation Hospital and nearby East Bay clinics from 1959 through 1966. Women were enrolled at the confirmation of their pregnancies and followed through delivery. The CHDS includes data on 20,754 pregnancies and 19,044 live births. All live births were followed through at least age 5 years, with some subsets of children whose families remained in the San Francisco Bay area being observed through adolescence. At 12 months only 48 (0.2%) subjects were lost to follow up and at age 5 years 89.4% were still under observation.
Detailed information on maternal and paternal characteristics was obtained through maternal interview. Maternal and pediatric medical records were abstracted to obtain information about maternal prenatal care measures, labor and delivery, and child's serial growth (weight and height) through age 5. Serum samples were collected from the women during each trimester of pregnancy and postpartum. After fractionation, serum samples were frozen at −20°C, stored and archived. Subjects provided informed consent in accordance with practices in the 1960s.
The data in the present analysis are derived from a study of organochlorine compounds (OCs), thyroid hormone levels and neurocognitive outcomes at ages 5, 9–11 and 15–17. The study base for the present analysis is drawn from a subset of CHDS children who participated in neurocognitive examinations through successive follow-up at ages 5, 9–11 and 15–17 (n=1752). Of these participants, we analyzed a sample of 600 infants (300 male, 300 female). The 600 were selected at random from the 500 males and 453 females who remained eligible after excluding subjects with inadequate second trimester sera (for the measurement of thyroid hormones), inadequate postpartum maternal serum (for the measurement of OCs) and subjects with missing gestational age. Pregnancy serum samples used for this analysis were drawn between 1960 and 1963, when DDT was still in active use.
2.2. Laboratory analyses
All serum samples were stored at −20 °C, with minimal thaw-refreeze cycles, until analyzed in 2007–2008. OCs were measured in sera collected immediately post partum (within three days of delivery) from the mother. Serum levels of DDT (p,p′-DDT, p,p′-DDE, o,p′-DDT) were measured. Serum levels of 11 polychlorinated biphenyl (PCB) congeners (66, 74, 99, 118, 138, 153, 170, 180, 187, 194 and 203) were also obtained. PCB and DDT assays were performed at both the Environmental Chemistry Laboratory of the California Department of Toxic Substances Control and at the Mount Sinai School of Medicine. The assay methods are detailed in a previous publication [33]. Serum triglyceride and cholesterol levels were measured in the laboratory of Dr. Nadir Rafai at The Children's Hospital, Harvard Medical School.
Previous reports have established that a wide array of organochlorines are detectable in CHDS serum and that measures between laboratories are comparable [34, 35]. The limits of detection (LOD) for both laboratories are also well below those generally considered satisfactory, ranging from 0.10 to 0.80 ng/mL for p,p′-DDE and p,p′-DDT, and from 0.03 to 0.40 for o,p′-DDT. Minimum percent of samples with detectable levels were 99.9% for p,p′-DDE, 99.7% for p,p′-DDT and 93.3% for o,p′-DDT. The maximum coefficients of variations calculated from quality control samples were 13.1, 28.3 and 25.5 for p,p′-DDE, p,p′-DDT and o,p′-DDT respectively.
Maternal thyroid function during pregnancy was assessed using free thyroxine (FT4) and thyroid stimulating hormone (TSH) measured in archived samples taken in the second trimester. All thyroid assays were performed at The Children's Hospital, Harvard Medical School. FT4 and TSH were measured using immunoassays on the Roche E Modular system (Roche Diagnostics, Indianapolis, IN). For FT4, the detection limit was 0.023 ng/dL and the day-to-day imprecision values at concentrations of 0.68, 1.64 and 3.94 ng/dL were 3.5, 3.3 and 6.6%, respectively. For TSH, the detection limit was 0.005 uIU/mL and the day-to-day imprecision values at concentrations of 0.084, 0.91 and 3.96 uIU/mL were 5.4, 2.1 and 1.8%, respectively. In a preliminary study (Factor-Litvak, unpublished data), we found that second trimester serum samples from pregnant women stored at −20 C for approximately 20 years had values of total T4 and TSH comparable to that of fresh sera.
2.3. Description of potential covariates
Potential covariates included maternal race, smoking status, occupation, education, age, body mass index (BMI) and parity and infant sex. Where possible, covariates were analyzed as continuous variables in regression models (i.e. maternal age and BMI), although for descriptive tables all variables were expressed categorically. Categories of variables included maternal race (white, black and mixed-race/other), smoking (never, former and current), occupation (employed and house-wife/part-time worker), education (less than a high-school diploma, a high school diploma only, a high school diploma and some college or trade school and college graduates), age (15–24, 25–29, 30–34, and ≥ 35 years), BMI (underweight (<18.5 kg/m2), normal weight (18.5–24.99 kg/m2), overweight (25–29.99 kg/m2) and obese (≥30 kg/m2)), parity (0, 1, 2 and ≥3) and infant sex (male and female).
Pre-pregnancy maternal weight was not ascertained for all participants; thus we used weight and height measured at first interview to calculate maternal BMI. Weight was adjusted to compensate for variation in gestational age by regressing weight on gestational age using the locally weighted scatterplot smoothing (LOWESS) technique [36]. Adjusted weight was then imputed as the fitted mean weight at 104 days of gestation, the median gestational age at first interview, plus each mother's residual from the locally weighted regression and scatterplot smoothing technique fit. This procedure removes differences in weight due to differences in gestational age at first interview. Maternal weight gain during pregnancy was thus calculated by subtracting the last measured pre-delivery weight from the adjusted first weight.
Finally, as in previous studies, the sum of PCB congeners (PCBtotal) was examined as a covariate in regression models [17]. In addition, for further comparability with other studies, we also examined as a covariate PCB congener 153 (PCB153) because it is one of the most prevalent congeners and is present at high concentrations [37, 38].
2.4. Description of prenatal outcomes
Infant birth weight and length of gestation were extracted from medical records. Length of gestation was determined by mothers' self-reported last menstrual period and recorded delivery date. There were 14 women in the sample with gestational length greater than 44 weeks; excluding these women did not change the findings.
Birth weight in grams and length of gestation in weeks were examined as continuous measures and were the primary outcomes in the study. We also calculated percentiles of birth weight-for-gestational-age, using race stratified tables as previously reported [39]. A small-for-gestational age (SGA) variable was created from these percentiles and was defined as birth weight less than the 10th percentile for gestational age. There were very few low-birth weight (N=11) and preterm (N=21) infants in our sample; thus the sample was not sufficiently powered to analyze these. We considered SGA as a secondary outcome.
2.5. Statistical analysis
The distributions of all variables were examined. Variables with skewed distributions were transformed to reduce impact of extreme values e.g. OCP concentrations were transformed using the natural logarithmic function and TSH concentrations were transformed using the square root function. Correlations between DDT components (p,p′-DDT, p,p′-DDE and o,p′-DDT), and between DDT components and PCBs (PCBtotal and PCB153) were also examined using the Spearman's rank correlation coefficients. Preliminary analyses compared the 600 infants selected for the study to those not selected using Chi-squares for categorical variables and t-tests for continuous variables with similar within sample variances.
We used multiple linear regression analysis to assess the associations between exposures and either birth weight or gestational age, with and without adjustment for potential confounders. For SGA, logistic regression models were used with the same set of predictors and control variables. Potential confounders were selected based on those previously reported to be associated with birth weight or gestational age in the literature or those known to confound the relationship between OCPs and pregnancy outcomes, which included infant sex, maternal age, race, education, BMI, occupation, marital status, parity, smoking status and exposure to PCBs (PCBtotal and PCB153). All models adjusted for the lab (ECL vs. MSSM) in which OCPs were measured and serum triglycerides and cholesterol transformed using the natural logarithmic function [40]. Potential covariates were included in a `core' regression model if they were associated with birth weight or gestational age. The core regression model did not include the exposure of interest. After exposure was included, covariates were deleted from final regression models if their exclusion resulted in an unsubstantial change (<0.5 standard error) in the estimated regression coefficient relating exposure to outcome.
Final linear models for the birth weight outcome were adjusted for the following covariates: maternal race, age at delivery, smoking status during pregnancy, BMI at first visit, infant sex, and linear and quadratic terms for length of gestation centered at its mean. Linear models for birth weight percentiles were adjusted for maternal age, BMI at first visit, employment status and smoking status during pregnancy, as well as infant sex. Linear models for length of gestation were adjusted for maternal race, age at delivery, employment status during pregnancy and infant sex.
We excluded subjects missing covariates (N = 21, 21, and 8 for the birth weight, birth weight percentile and length of gestation outcomes, respectively). For an additional 22 women, OCP, total cholesterol and triglyceride concentrations were unable to be measured due to a laboratory problem; these subjects did not differ from the remaining on important variables, including the outcomes. Further taking into consideration the exclusion of the 14 participants with length of gestation greater than 44 weeks, final sample sizes for birth weight, birth weight percentile and length of gestation models were 543, 543, and 556 respectively.
DDT was the main exposure in the study. We examined each component of DDT exposure separately, that is p,p′-DDT (the primary component), o,p′-DDT (a contaminant) and p,p′-DDE (the primary metabolite). We also examined the joint effects by including all three components in the same models, to account for total exposure to DDT. We explored the dose-response pattern by categorizing each component into tertiles.
We examined whether the associations between DDT (each component and considered jointly) and birth outcomes were mediated by maternal thyroid function using linear regression analysis [12]. In these analyses, FT4 and TSH were included separately in the regression models; we considered the following as evidence for mediation: reductions in the magnitude of the estimated regression coefficient for the exposure of interest, i.e. OCPs, as well as associations between FT4 and/or TSH and exposure and outcome.
Final analyses examined sex specific associations between exposures and each birth outcome as reported previously [16]. We stratified models by sex to examine sex specific associations between all DDT components and outcome.
All analyses were performed using SAS version 9.2.
3. Results
Our sample had a mean birth weight of 3387 grams and mean gestational age of 40.2 weeks (Table 1). There were 87 (14.8%) SGA infants in our sample and the mean percentile of birth weight-for-gestational age was 43.2%. Comparisons between the 600 randomly selected subjects from the larger pool of all Adolescent Study participants show no significant differences in relevant variables (birth weight, number of previous live births, percent male infants, mothers' education at birth, mothers' smoking status during pregnancy). Significant differences in gestational age were reported, with days of gestation being three days longer in those selected (p<0.001). Table 2 presents the distributions of maternal concentrations of p,p′-DDT, p,p′-DDE and o,p′-DDT. Spearman correlation coefficients were 0.73 between p,p′-DDT and p,p′-DDE, 0.74 between p,p′-DDT and o,p′-DDT, and 0.57 between p,p′-DDE and o,p′-DDT. Spearman correlation coefficients were 0.51, 0.58, and 0.36 between PCBtotal and p,'-DDT, PCBtotal and p,p′-DDE, and PCBtotal and o,p′-DDT, respectively. Concentrations of p,p′-DDT, p,p′-DDE and o,p′-DDT were higher in non-white mothers, older mothers and those who had not achieved a high-school diploma (Table 3). In addition, mothers who never smoked had significantly higher p,p′-DDT and o,p′-DDT concentrations, and obese mothers had higher levels of p,p′-DDT and o,p′-DDT compared with underweight mothers.
Table 1.
Sample features between those included in the study and those excluded
| Variable | Selected (N=600) | Not Selected (N=1420) | |||||
|---|---|---|---|---|---|---|---|
|
| |||||||
| N | Mean | SD | N | Mean | SD | p-value | |
|
|
|||||||
| Birth Weight (g) | 600 | 3387.8 | 493.3 | 1420 | 3353.7 | 533.0 | 0.92 |
| Gestation (days; LMP to term) | 600 | 282.1 | 12.7 | 1404 | 279.1 | 16.3 | <.001 |
| Number of Previous Live Births | 600 | 1.7 | 1.7 | 1420 | 1.9 | 1.8 | 0.09 |
| % | % | ||||||
| Male | 300 | 50.0 | 703 | 49.5 | 0.84 | ||
| Mother Education at Child's Birth | |||||||
| < High School | 74 | 12.3 | 169 | 11.9 | 0.58 | ||
| High School Graduate | 202 | 33.7 | 477 | 33.6 | |||
| Some College /Trade School | 200 | 33.3 | 444 | 31.3 | |||
| College Graduate or Higher | 123 | 20.5 | 329 | 23.2 | |||
| Missing | 1 | 0.2 | 1 | 0.1 | |||
| Maternal Smoking During Pregnancy | |||||||
| Never Smoked | 315 | 52.5 | 662 | 46.6 | 0.16 | ||
| Smokes Now | 180 | 30.0 | 490 | 34.5 | |||
| Smoked Until Current Pregnancy | 40 | 6.7 | 119 | 8.4 | |||
| Former Smoker | 61 | 10.2 | 136 | 9.6 | |||
| Missing | 4 | 0.7 | 13 | 0.9 | |||
Data are presented as number (N), mean and standard deviation (SD) or percent (%).
Table 2.
Distribution of maternal pesticide (ng/mL) and thyroid concentrations.
| Percentiles |
|||||||
|---|---|---|---|---|---|---|---|
| Exposure categorya | No. | 25th | 50th | 75th | Mian ± SD | Min | Max |
| p,p′-DDT | 579 | 7.9 | 10.8 | 15.1 | 12.9 ± 7.7 | 1.5 | 71.1 |
| ECL | 459 | 7.9 | 10.5 | 14.5 | 12.2 ± 6.5 | 3.5 | 52.9 |
| MSSM | 120 | 8.7 | 12.7 | 16.6 | 15.3 ± 10.7 | 1.5 | 71.1 |
| p,p′-DDE | 578 | 30.0 | 38.7 | 52.2 | 44.2 ± 23.2 | 4.98 | 285.4 |
| ECL | 458 | 29.7 | 37.3 | 49.5 | 42.3 ± 22.2 | 11.8 | 285.4 |
| MSSM | 120 | 33.7 | 47.8 | 60.9 | 513 ± 25.8 | 5.0 | 158.1 |
| o,p′-DDT | 575 | 0.28 | 0.43 | 0.68 | 058 ± 0.55 | 0.01 | 4.46 |
| ECL | 459 | 0.27 | 0.41 | 0.64 | 0.56 ± 0.54 | 0.50 | 4.46 |
| MSSM | 116 | 0.31 | 0.46 | 0.86 | 0.66 ± 0.061 | 0.01 | 3.48 |
|
| |||||||
| FT4 (ng/dL) | 600 | 1.13 | 1.23 | 138 | 126 ± 0.21 | 0.76 | 2.46 |
| TSH (μIU/mL) | 600 | 0.86 | 1.29 | 1.92 | 153 ± 1.16 | 0.005 | 12.55 |
DDT variables are reported by laboratory: Environmental Chemistry Laboratory (ECL) and Mount Sinai School of Medicine (MSSM).
Table 3.
Concentrations of p,p′-DDT, p,p′-DDE and o,p′-DDT by sample characteristicsa
| Pesticides (mean ± SD) |
||||
|---|---|---|---|---|
| Sample Characteristics | N# | p,p′-DDT | p,p′-DDE | o,p′-DDT |
| Total sample | 579 | 12.9 ± 7.7 | 44.2 ± 23.2 | 0.58 ± 0.55 |
| Maternal race | ||||
| White§ | 412 | 11.5 ± 6.8 | 41.6 ± 19.4 | 0.47 ± 0.41 |
| Black | 115 | 15.5 ± 8.4* | 49.8 ± 32.8* | 0.85 ± 0.72* |
| Other | 52 | 18.0 ± 8.6* | 52.3 ± 22.0* | 0.87 ± 0.75* |
| Sex of infant | ||||
| Male§ | 285 | 12.5 ± 7.4 | 41.9 ± 20.1 | 0.54 ± 0.51 |
| Female | 294 | 13.2 ± 7.9 | 46.3 ± 25.7* | 0.61 ± 0.59 |
| Maternal smoking status | ||||
| Never§ | 307 | 13.5 ± 7.9 | 44.0 ± 25.7 | 0.63 ± 0.60 |
| Current | 174 | 11.8 ± 7.4* | 46.5 ± 21.1 | 0.50 ± 0.43* |
| Former | 94 | 12.7 ± 7.2 | 40.5 ± 18.0 | 0.56 ± 0.59 |
| Maternal occupation | ||||
| Employed | 234 | 12.7 ± 7.6 | 45.2 ± 25.2 | 0.57 ± 0.50 |
| House wife/Part-time§ | 338 | 12.8 ± 7.6 | 43.2 ± 21.6 | 0.58 ± 0.59 |
| Maternal education† | ||||
| No HS diploma | 70 | 15.8 ± 8.9* | 46.9 ± 22.3 | 0.89 ± 0.80* |
| HS diploma only§ | 194 | 12.1 ± 6.6 | 44.2 ± 24.3 | 0.53 ± 0.50 |
| HS + some college | 195 | 12.3 ± 6.6 | 43.8 ± 23.6 | 0.55 ± 0.52 |
| College graduate | 119 | 13.3 ± 9.5 | 43.0 ± 21.2 | 0.53 ± 0.46 |
| Maternal age categories | ||||
| 15–24 | 158 | 12.1 ± 6.6 | 45.3 ± 27.2 | 0.55 ± 0.46 |
| 25–29§ | 162 | 12.4 ± 7.5 | 43.5 ± 23.5 | 0.60 ± 0.65 |
| 30–34 | 141 | 13.0 ± 8.9 | 42.5 ± 20.7 | 0.57 ± 0.57 |
| ≥35 | 117 | 14.3 ± 7.4* | 45.7 ± 19.5 | 0.61 ± 0.50 |
| Maternal BMI categories | ||||
| Underweight | 19 | 11.5 ± 7.7 | 47.2 ± 24.1 | 0.44 ± 0.37 |
| Normal§ | 406 | 12.7 ± 7.9 | 45.5 ± 24.8 | 0.56 ± 0.54 |
| Over-weight | 105 | 12.8 ± 6.1 | 39.3 ± 16.5* | 0.59 ± 0.50 |
| Obese | 32 | 15.6 ± 9.1* | 43.2 ± 22.3 | 0.86 ± 0.81* |
| Parity | ||||
| 0§ | 163 | 12.5 ± 7.9 | 44.9 ± 26.7 | 0.57 ± 0.52 |
| 1 | 145 | 11.9 ± 7.8 | 41.3 ± 21.6 | 0.52 ± 0.48 |
| 2 | 111 | 13.2 ± 6.7 | 47.2 ± 21.9 | 0.54 ± 0.44 |
| ≥3 | 160 | 13.8 ± 7.9* | 44.0 ± 21.6 | 0.68 ± 0.69 |
Differences in OCP levels within categories of maternal covariates were evaluated using analysis of variance with Bonferroni corrections.
group size for variables in the sample of N=579 subjects with available p,p′-DDT measures [p,p′-DDT (N=579); p,p′-DDE (N=578); o,p′-DDT (N=575)]
HS = high-school;
reference category
p<0.05 for difference in the pesticide between two groups (a group vs. the reference).
No significant associations were found between OCPs and length of gestation in models adjusted only for serum lipids and laboratory when exposure was defined as each component of DDT separately (Table 4). After adjustment for the full set of covariates, for each natural log unit increase in p,p′-DDE we observed a non-significant 0.25 week decrease in length of gestation week decrease in length of gestation (95% CI: −0.58, 0.08) (Table 4). In the model with all components of DDT, each natural log unit increase in p,p′-DDE was associated with a 0.49 week reduction in length of gestation (95% CI: −0.97, −0.004) Results did not change when exposure was categorized by tertiles (data not shown). These associations diminished when PCBtotal was added as a covariate (estimated regression coefficient for p,p′-DDE reduced to −0.099 weeks (95% CI −0.49, 0.29)). Similar reductions were found when PCBtotal was replaced with PCB153.
Table 4.
Covariates-adjusted Regression Coefficient Estimates Relating Prenatal OCP Exposure to Birth Weight or Length of Gestation
| Birth Outcomes |
||||||||
|---|---|---|---|---|---|---|---|---|
| Birth weight (g) N=543 | Length of Gestation (days) n=556 | |||||||
|
| ||||||||
| badjusteda | 95%CI | badjustedb | 95%CI | badjusteda | 95%CI | badjustedc | 95%CI | |
|
|
|
|||||||
| Individual Organochlorines d | ||||||||
| p,p′-DDT | 44 | (−44, 133) | 92* | (7, 176) | −0.01 | (−0.32, 0.30) | −0.010 | (−0.32, 0.30) |
| p,p′-DDE | −117 | (−217, −17) | 0.5 | (−93, 94) | −0.25 | (−0.58, 0.08) | −0.25 | (−0.58, 0.08) |
| o,p′DDT | −44 | (−101, 14) | −3 | (−58, 52) | −0.15 | (−034, 0.031) | −0.064 | (−0.26, 0.13) |
| Joint Model | ||||||||
| p,p′-DDT | 364 | (206, 523) | 274** | (122, 425) | 0.35 | (−0.17, 0.87) | 0.39 | (−0.14, 0.92) |
| p,p′-DDE | −303 | (−500, −157) | −153* | (−296, −10) | −0.47 | (−0.96, 0.008) | −0.49* | −0.97, −0.004) |
| o,p′-DDT | −115 | (−202, −27) | −75+ | (−155, 5) | −0.16 | (−0.44, 0.12) | −0.08 | (−0.36, 0.21) |
Variables adjusted: laboratory, natural log-transformed serum triglycerides and cholesterol.
Variables adjusted: maternal race, age at delivery, smoking status during pregnancy, maternal first visit BMI, infant sex, linear and quadratic length of gestation (centered at mean), laboratory and natural log-transformed serum triglycerides and cholesterol.
Variables adjusted: maternal race, age at delivery, employment status, infant sex, laboratory and natural log-transformed serum triglycerides and cholesterol.
All DDT components are in units of ng/mL. In the models OCP measures are transformed by natural logarithm function.
0.05<p<0.10,
p<0.05,
p<0.01.
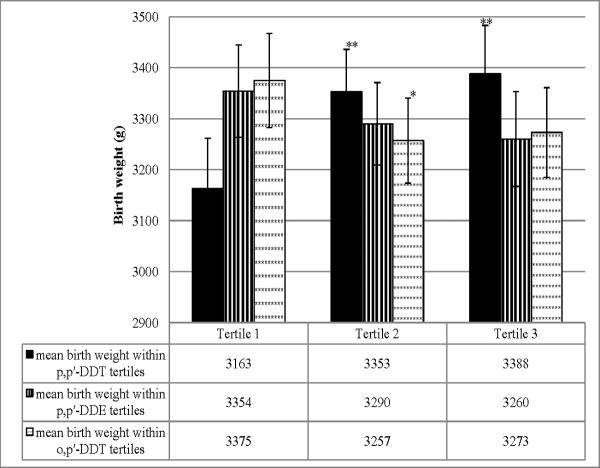
In utero exposure to p,p′-DDT was associated with increased birth weight (Table 4) and increased birth weight percentile for gestational age (Supplementary Table S1). For each natural log unit increase in p,p′-DDT concentration, adjusted birth weight increased by 92 grams (95% CI: 7, 176). When the three variables, p,p′-DDT, p,p′-DDE and o,p′-DDT, were included in the same model, adjusted birth weight increased by 274 grams (95% CI 122, 425), decreased by 173 grams (95% CI −296, −10) and decreased by 73 grams (95% CI −155, 5), for each natural log unit increase in the respective exposures (Table 4). Similarly, when included in the same model, adjusted percentiles of birth weight-for-gestational age increased by 14.4 percentiles (95% CI 5.1, 23.8), decreased by 8.7 percentiles (95% CI −17.7, 0.2), and decreased by 4.6 percentiles (95% CI −9.5, 0.4) for each log-unit increase in p,p′-DDT, p,p′-DDE, and o,p′-DDT exposure, respectively (Supplementary Table S1). When exposure was categorized as tertiles, a significant dose-response relationship was observed with covariate-adjusted mean birth weights increasing with tertiles of p,p′-DDT (p<0.05) and this effect was substantially magnified when o,p′-DDT and p,p′-DDE were considered concurrently in the model (p<0.01) (Figure 1). In the joint model, adjusted mean birth weight decreased with increasing tertiles of both p,p′-DDE and o,p′-DDT. Associations were unchanged when PCBtotal or PCB153 was controlled in the models. Addition of the adjusted maternal weight gain variable into the joint model did not change the interpretation of our results. p,p′-DDT remained positively associated with birth weight (b= 288; 95% CI: 143, 433) , while p,p′-DDE (b=−81; 95% CI: −219, 57) and o,p′-DDT (b=−85; 95% CI: −162, −9) remained negatively associated with birth weight; however, associations with p,p′-DDE lost statistical significance. Logistic regression models of SGA did not reach statistical significance, likely due to the small number of SGA infants in our sample (N=87), though the direction of associations was consistent with results of birth weight and percentile birth weight-for-gestational age analyses (Supplementary Table S2).
Figure 1. Adjusted mean birth weight in tertiles of p,p′-DDT, p,p′-DDE and o,p′-DDT exposure from the model with joint exposures.
Covariates-adjusted mean birth weight and 95% confidence intervals for tertiles of p,p′-DDT, p,p′-DDE and o,p′-DDT exposure derived from the model with joint exposures. All three compounds were included categorized as tertiles. Covariates adjusted included maternal race, age at delivery, smoking status during pregnancy, maternal BMI, infant sex, linear and quadratic terms for length of gestation centered at its mean, laboratory and log-transformed serum triglyceride and cholesterol. The lowest tertile of exposure was used as the reference in regression models. In this joint exposures model, a trend toward decreasing birth weight is observed with increasing p,p′-DDE and o,p′-DDT exposure at a fixed level of p,p′-DDT. Tertile cut-points for p,p′-DDT were 1.51–8.78, 8.89–13.42, and 13.43–71.05 for tertiles 1, 2 and 3, respectively. Tertile cut-points for p,p′-DDE were 4.98–33.21, 33.32–47.47, and 47.5–285.4 for tertiles 1, 2 and 3, respectively. Tertile cut-points for o,p′-DDT were 0.01–0.32, 0.33–0.54, 0.55–4.46 for tertiles 1, 2 and 3, respectively.
*p<0.05, **p<0.01.
We did not find evidence of mediation by maternal thyroid status on the association between pregnancy outcomes and exposure to the DDT metabolites because neither FT4 nor TSH was related to exposures or outcomes [Supplementary Tables S3 (FT4) and S4 (TSH)]. We also did not find evidence of sex differences in the OCP associations.
4. Discussion
Overall, we saw significant associations between exposure to the pesticide DDT and pregnancy outcomes. Our results suggest inverse associations between exposure to p,p′-DDE and length of gestation such that there was approximately a three day decrease for each natural log unit increase in p,p′-DDE; however, this association diminishes when PCBs are also controlled for in the model.
Our results also suggest a complex relationship between overall DDT exposure and birth weight, but findings support the hypothesis that DDT compounds influence fetal growth. When considered simultaneously, p,p′-DDT and p,p′-DDE had opposite associations with birth weight such that for each log unit increase in p,p′-DDT birth weight increased by 274 grams (95% CI 122, 425) and for each log unit increase in p,p′-DDE, birth weight decreased by 153 grams (95% CI −296, −10). Supplementary analysis examining the ratio of p,p′-DDT to p,p′-DDE support the concept that relative amounts of these compounds determine the direction of the associations observed (Supplementary Table S5). In adjusted regression analysis we found that with each unit increase in p,p′-DDT/p,p′-DDE ratio, birth weight significantly increased by 567 g (95% CI 171, 962). Results changed minimally when o,p′-DDT was also added to the model (b=571; 95% CI: 144, 998). This finding is consistent with what we see in linear regression analyses for joint exposure and provides added confidence in our findings. A higher proportion of p,p′-DDT predicted higher birth weight.
Figure 1 suggests that infants born to mothers in the lowest p,p′-DDT tertile also have the lowest birth weights (although still in the range considered clinically normal) and that the difference between the lowest tertile and highest tertile (i.e. approximately 200 grams) is similar to the well-known association between cigarette smoking during pregnancy and birth [41]. However, our findings suggest that the impact of exposure on birth weight depends on either the recency of exposure or differences in metabolism of p,p′-DDT. For example, although speculative, this result may be attributed to differences in genetic polymorphisms in the enzymes which are responsible for p,p′-DDT metabolism [42]. Alternatively, we speculate that the recency of exposure may be associated with either increased or decreased birth weight, as results using the ratio of p,p′-DDT to p,p′-DDE indicated that a increasing ratio predicts higher birth weight while a decreasing ratio predicts lower birth weight. In either case, our results demonstrate that DDT and its components disrupt fetal growth.
A 2005 study of the association of p,p′-DDT and p,p′-DDE with birth weight in a different sample from the CHDS found similar results [17]: a positive association for p,p′-DDT and a negative association for p,p′-DDE; however, neither was statistically significant. That study design was based on a nested case-control sample of hypospadias and cryptorchidism among 420 males in the CHDS and did not include an investigation of o,p′-DDT. These differences in study design (nested case-control vs. prospective), sample characteristics and size (420 males vs. 600 males and females) and method of exposure assessment (our study included exposure to o,p′-DDT and an examination of total exposure to DDT) may explain the differences in the two analyses.
To our knowledge, no other studies examining DDT and birth outcomes have reported assessing total DDT exposure by including all components in the same model. We posit that higher concentrations of p,p′-DDT and o,p′-DDT may represent more recent exposure to commercial DDT or lower individual metabolic conversion of p,p′-DDT to p,p′ DDE. Under this assumption, o,p′-DDT is a marker of recent exposure as it is the least persistent component of DDT, being quickly metabolized and eliminated from the body [43]. In contrast, p,p′-DDE is the most stable long-term marker since it is more slowly metabolized than p,p′-DDT and most persistent in the environment [43]. Thus, p,p′-DDE is often used as a proxy for p,p′-DDT. However during active DDT use, individual p,p′-DDE levels are considered to result from metabolic conversion following direct exposure to p,p′-DDT and also from ingestion of already metabolized p,p′-DDE in the environment. Therefore it is difficult to separate exposure from metabolism in interpreting an individual's p,p′-DDE levels. This underscores the importance of controlling for all components of the pesticide DDT when assessing total exposure.
Some researchers consider the ratio of p,p′-DDT to p,p′-DDE as the metric for recent exposure [16, 17]; this represents the timing of exposure as well as the rate of metabolic clearance. For example, women who are slower at eliminating p,p′-DDT or were more recently exposed to p,p′-DDT would have a higher ratio [44]. Such a metric was used by Cohn et al. (2003), to evaluate the associations between prenatal DDT exposure and time to pregnancy in daughters [45] and testicular cancer in son [44]. In those studies higher ratios of p,p′-DDT to p,p′-DDE were associated with longer time to pregnancy in daughters and testicular cancer in sons, suggesting that higher or more recent exposure to p,p′-DDT, or slower metabolism of p,p′-DDT was deleterious.
p,p′-DDT and p,p′-DDE may operate through different biological pathways; p,p′-DDT exhibits little estrogenic or androgenic activity, o,p′-DDT is thought to be weakly estrogenic, and p,p′-DDE has potent anti-androgenic activity [46]. As these components of DDT operate through different hormone receptors, they are likely to have different biological effects. We note that in utero exposure to high levels of estrogen is associated with increased birth weight [47, 48]. The effects of in utero exposure to environmental anti-androgens on birth weight are less established, although a few studies in males have postulated that androgen insufficiency may contribute to low-birth weight and, thus, later reproductive dysfunction, such as hypospadias [49]. Cohn et al. (2003) provides evidence in support of opposing effects of p,p′-DDT and p,p′-DDE, with maternal concentrations of p,p′-DDT associated with a reduced probability of pregnancy in daughters and maternal concentrations of p,p′-DDE associated with a raised probability [45]. Due to these differences, we conclude that modeling simultaneously for all major DDT components elucidates the compound specific associations.
Previous reports of associations between pesticide exposure and pregnancy outcomes are inconsistent. Discrepant findings result mainly from differences in classification and assessment of exposure and outcome variables, and population differences such as background levels of pesticides. In a prospective study of 2,380 subjects from the National Collaborative Perinatal Study (NCPP), Longnecker et al. (2001) assessed birth weight (continuous and dichotomized at 2500g), gestation (continuous and dichotomized at 37 weeks), and SGA in relation to p,p′-DDE exposure (<15 ng/mL, 15–29 ng/mL, 30–44 ng/mL, 45–59 ng/mL, and ≥60 ng/mL) [16]. Increasing categories of p,p′-DDE were associated with decreased birth weight and length of gestation, and increased odds of preterm birth, low-birth weight and SGA. For example, the adjusted mean birth weight was 150 grams lower for infants in the highest category of p,p′-DDE (≥ 60 ng/mL) compared to infants in the lowest (<15ng/mL). The authors also considered p,p′-DDT exposure and the ratio of p,p′-DDT/p,p′-DDE and did not find associations with either (no data shown). It is noteworthy that this population was similar to ours in terms of years of enrollment into the study (1959 – 1966 vs. 1960 – 1963 in our study) and median serum p,p′-DDE levels (25 ng/mL vs. 38.7 ng/mL in our study) which is higher than current population levels [4]. In addition, the range of p,p′-DDE levels in both studies were similar (3–178 ng/mL in NCPP vs 5–285 ng/mL in our study). o,p′-DDT levels were not reported. We also observe negative associations with p,p′-DDE and birth weight in our study, however these associations only reach a level of significance when p,p′-DDT and o,p′-DDT are jointly considered.
Studies of more recent cohorts often have much lower levels of OCPs, which could contribute to inconsistencies in findings. Wolff et al. (2007), in a cohort enrolled between 1998 and 2002, observed decreasing birth weight with increasing amounts of lipid-adjusted, log-transformed p,p′-DDE (b=−128, p=0.058) [18]. Neither p,p′-DDT nor o,p′-DDT were examined in this study and the median maternal serum p,p′-DDE concentration was 0.64 ng/mL, which is much lower than in our sample (38.7 ng/mL). Berkowitz et al. (1996) found no association between p,p′-DDE and preterm delivery in a sample of 40 U.S. mothers whose specimens were collected from 1990 to 1993 [25]. Median p,p′-DDE levels were low in this study (cases: 1.3 ng/mL, controls: 1.35 ng/mL) compared with ours. The smaller sample size, different study design and lower median levels of p,p′-DDE could all have contributed to the difference in findings between their study and ours. Other studies also conclude no association between p,p′-DDE and pregnancy outcomes [27, 50], however these studies are not consistent with ours with respect to biological specimen analyzed (maternal milk vs. serum) and year of specimen collection (1978–1982 [27] and 1993–1994 [49] vs. 1960s for the current study).
Most recently, Govarts et al. (2011) conducted a meta-analysis using 15 studies in 12 European cohorts which examined the association between p,p′-DDE and pregnancy outcomes including birth weight adjusted for gestational age [38]. All cohorts were established between 1990 and 2008 and thus had lower exposure levels (measured in maternal and cord blood samples and in breast milk) than the CHDS. p,p′-DDE in cord blood was used as the primary measure of pesticide exposure and estimates of cord blood levels were created for those studies with exposure metrics other than cord blood. Low-level exposure to p,p′-DDE was not associated with birth weight; each one unit increase in p,p′-DDE resulted in a non-significant seven gram decrease in birth weight (95% CI −18, 4) [38]. However, the median levels of p,p′-DDE in this study were quite low compared with our study (overall median: 0.528 ng/mL; range: 0.05 – 1.21 ng/mL). In addition, there was no examination of p,p′-DDT or o,p′-DDT.
We explored possible effect modification by sex; however, no consistent patterns or differences between male and female infants emerged. Some studies find suggestions of sex effects. For example, Longnecker et al (2001) found increased odds of preterm birth related to p,p′-DDE exposure in males compared to females; the adjusted odds ratios suggested a stronger effect with increasing p,p′-DDE concentrations, although the statistical test for interaction was not significant [16]. However others have not seen evidence of gender specific associations [38].
In addition to our consideration of the joint effect of p,p′-DDT, o,p′-DDT and p,p′-DDE, our study has many strengths. First, data were collected prospectively, avoiding recall bias and problems with temporality. High participation rates among those invited to join the full CHDS study reduced the potential for selection biases. Of those eligible, 75.5% of participants agreed to participate in 5-year examinations and 80% agreed to participate in the 15–17 year (adolescent) examination. From the CHDS adolescent cohort, we randomly selected our sample, and those selected did not appreciably differ in maternal characteristics from those who were not; thus, we have no reason to believe these factors affect the probability of selection into our study. Length of gestation in the selected participants was slightly longer compared to those not selected. Birth weight did not differ between those eligible and selected participants, thus it is unlikely that study selection bias could explain the p,p′-DDT association with birth weight.
Second, the expected relationships between known confounders and pregnancy outcomes (e.g. maternal smoking was associated with decreased birth weight, male infants exhibited higher birth weights, maternal BMI was associated with birth weight) are present in our sample, thus supporting the validity of our data (data not shown). Further, we controlled for all maternal characteristics shown to be significantly associated with DDT exposure and pregnancy outcomes in our sample. In length of gestation models, inclusion of other known risk factors (maternal BMI, smoking and parity) did not significantly alter results (data not shown). Thus, confounding by other maternal characteristics is unlikely to be responsible for our findings.
Third, the wide range of exposure allowed examination of the association across p,p′-DDT, p,p′-DDE and o,p′-DDT exposures from low to relatively high. Indeed, we found the significant difference in birth weight between the exposure groups of the highest tertile and the lowest tertile. Fourth, our outcomes were considered as continuous variables, rather than dichotomizing birth weight at 2500 g or length of gestation at 37 weeks, which we believe has the statistical advantage of avoiding the implied assumption of homogeneity of risk within dichotomies of the birth outcome [51]. As the percentage of infants born preterm or low birth weight was very low (1.8% and 3.5%, respectively) in our sample, we believe keeping the outcome as a continuous variable was preferable to an insufficiently-powered logistic regression analysis of either outcome as a dichotomous variable. Fifth, we found consistent results with another measure of fetal growth, birth weight percentile for gestational age.
A particular strength of this study is that we were able to test whether thyroid disruption mediates the effects of DDT and its components on pregnancy outcomes. The proposed mechanism is likely due to the competitive binding by certain OCs to transthyretin (TTR), the transport protein that carries T4 from maternal circulation to the placenta [7]. There is some evidence that OCPs can perturb thyroid function [12]. However, we did not find any evidence of mediation by maternal thyroid function in either birth weight or length of gestation analyses, suggesting that DDT and its components impact fetal growth via a different mechanism.
Our study also has several limitations. First, we were missing either laboratory data or data on covariates on approximately 10% of our sample. However, comparisons of subjects with missing data to those without missing data did not find appreciable differences. It should be noted that other authors [52] have found maternal weight gain during pregnancy to be an important confounder in analyses of environmental contaminants and birth weight. Because a question on pre-pregnancy weight was added later in the data collection, about 30% of mothers had missing data on pre-pregnancy weight. However we were able to adjust for maternal weight gain during pregnancy using a Lowess adjusted variable. The interpretation of our results does not change with the addition of the adjusted maternal weight gain variable into our final covariates-adjusted regression model for birth weight; however, associations with p,p′-DDE lose statistical significance.
Second, serum samples from these women were collected between 1960 and 1963 [45], a time when DDT use was at its peak, so exposure levels today are not comparable to exposure in this population. For example, based on the Fourth Report of the National Health and Nutrition Examination Survey (NHANES), levels of p,p′-DDT for females sampled in 2003–04 were below the limit of detection at both the 50th and 75th percentiles of serum concentrations and no geometric mean was calculated; however, the mean level of p,p′-DDE (whole weight) was 1.45 ng/g (95% CI 1.16, 1.82) [4]. Thus, while we may not be able to extrapolate these findings to the current exposure levels, our population allows us the unique opportunity to explore relationships that would be impossible to assess in contemporary serum. Additionally, post partum sera were used for the measurement of OCs. This was for a practical reason, as more sera were available for this time point. However, because OCs have a long half-life in adipose tissue we do not expect levels to vary during pregnancy. Indeed, Longnecker et al. found that OC levels measured in maternal blood collected serially in each trimester of pregnancy and early postpartum were highly correlated with minimal differences [53]. There may be concerns about multiple testing, however our analysis were a priori specified and analyses that were secondary or exploratory were stated as such. Lastly, we cannot rule out the possibility of confounding by a co-variable or other environmental contaminant that we did not measure.
5. Conclusions
Our findings have public health implications for infant and potentially later adult health; they are consistent with the notion that birth weight may be a marker of in utero exposures. Reductions in birth weight carries increased perinatal risk as well as implications for a variety of adverse health effects in childhood [54–56]. Our results also raise the possibility that OCP related alterations in birth weight may mediate the hypothesized relations between OCPs and metabolic disorders later in life (e.g. type II diabetes, obesity) [57–62] or certain cancers [63, 64]. The impact of prenatal p,p′-DDT exposure on birth weight reported here may shed light on mechanisms for altering these risk pathways which will be explored in future follow up of this cohort.
Supplementary Material
Research Highlights
Prenatal p,p′-DDT exposure is associated with increased birth weight.
The major metabolite, p,p′-DDE, is associated with decreased birth weight.
DDT is not associated with length of gestation.
The observed associations are not mediated by maternal thyroid hormone levels.
The observed associations did not differ by infant's sex.
Acknowledgements
Funded by R01ES012360 and P30ES009089 from the National Institute of Environmental Health Sciences. The funding source had no role in the research.
Glossary
- CHDS
Child Health and Development Studies
- OCP
organochlorine pesticide
- DDT
trade name of the organochlorine pesticide examined in this study which is comprised of three component parts
- p,p′-DDT
1,1,1-trichloro-2,2-bis(p-chlorophenyl)ethane
- p,p′-DDE
1,1′-dichloro-2,2'-bis(p-chlorophenyl)ethylene
- o,p′-DDT
1,1,1-trichloro-2-(p-chlorophenyl)-2-(o-chlorophenyl)-ethane
- tC
cholesterol
- tG
triglyceride
- FT4
free thyroxine
- TSH
thyroid stimulating hormone
- TTR
transthyretin
- PCB
polychlorinated biphenyl
- 95% CI
95% confidence interval
Footnotes
Publisher's Disclaimer: This is a PDF file of an unedited manuscript that has been accepted for publication. As a service to our customers we are providing this early version of the manuscript. The manuscript will undergo copyediting, typesetting, and review of the resulting proof before it is published in its final citable form. Please note that during the production process errors may be discovered which could affect the content, and all legal disclaimers that apply to the journal pertain.
Competing financial interests: The authors declare they have no competing financial interests.
References
- [1].Carson R. Silent spring. Houghton-Mufflin; Boston, MA: 1962. [Google Scholar]
- [2].Lopez-Espinosa MJ, Granada A, Carreno J, Savatoerra M, Olea-Serrano F, Olea N. Organochlorine pesticides in placentas from Southern Spain and some related factors. Placenta. 2007;28:631–8. doi: 10.1016/j.placenta.2006.09.009. [DOI] [PubMed] [Google Scholar]
- [3].Longnecker M, Rogan W, Lucier G. The human health effects of DDT (dichlorodiphenyltrichloroethane) and PCBS (polychlorinated biphenyls) and an overview of organochlorines in public health. Annu Rev Public Health. 1997;18:211–44. doi: 10.1146/annurev.publhealth.18.1.211. [DOI] [PubMed] [Google Scholar]
- [4].CDC (Centers for Disease Control and Prevention) Fourth National Report on Human Exposure to Environmental Chemicals. Centers for Disease Control and Prevention; Atlanta: 2009. [cited 2011 Aug 20]. Available from: http://www.cdc.gov/exposurereport/pdf/FourthReport.pdf. [Google Scholar]
- [5].Brouwer A, Morse DC, Lans MC, Schuur AG, Murk AJ, Klasson-Wehler E, et al. Interactions of persistent environmental organohalogens with the thyroid hormone system: mechanisms and possible consequences for animal and human health. Toxicol Ind Health. 1998;14:59–84. doi: 10.1177/074823379801400107. [DOI] [PubMed] [Google Scholar]
- [6].National Research Council . Hormonally active agents in the environment. National Academy Press; Washington, DC: 1999. [Google Scholar]
- [7].Cheek AO, Kow K, Chen J, McLachlan JA. Potential mechanisms of thyroid disruption in humans: interaction of organochlorine compounds with thyroid receptor, transthyretin, and thyroid-binding globulin. Environ Health Perspect. 1999;107:273–8. doi: 10.1289/ehp.99107273. [DOI] [PMC free article] [PubMed] [Google Scholar]
- [8].Lopez-Espinosa MJ, Vizcaino E, Murcia M, Llop S, Espada M, Seco V, et al. Association between thyroid hormone levels and 4,4′-DDE concentrations in pregnant women (Valencia, Spain) Environ Res. 2009;109:479–85. doi: 10.1016/j.envres.2009.02.003. [DOI] [PubMed] [Google Scholar]
- [9].Maervoet J, Vermeir G, Covaci A, Van Larebeke N, Koppen G, Shoeters G, et al. Association of thyroid hormone concentrations with levels of organochlorine compounds in cord blood of neonates. Environ Health Perspect. 2007;115:1780–6. doi: 10.1289/ehp.10486. [DOI] [PMC free article] [PubMed] [Google Scholar]
- [10].Asawasinsopon R, Prapamontol T, Prakobvitayakit O, Vaneesorn Y, Mangklabruks A, Hock B. The association between organochlorine and thyroid hormone levels in cord serum: a study from northern Thailand. Environ Int. 2006;32:554–9. doi: 10.1016/j.envint.2006.01.001. [DOI] [PubMed] [Google Scholar]
- [11].Álvarez-Pedrerol M, Ribas-Fitó N, Torrent M, Carrizo D, Grimalt JO, Sunyer J. Effects of PCBs, p,p′-DDT, p,p′-DDE, HCB and β-HCH on thyroid function in preschool children. Occup Environ Med. 2008;65:452–7. doi: 10.1136/oem.2007.032763. [DOI] [PubMed] [Google Scholar]
- [12].Takser L, Mergler D, Baldwin M, de Grosbois S, Smargiassi A, Lafond J. Thyroid hormones in pregnancy in relation to environmental exposure to organochlorine compounds and mercury. Environ Health Perspect. 2005;113:1039–45. doi: 10.1289/ehp.7685. [DOI] [PMC free article] [PubMed] [Google Scholar]
- [13].Lopez-Espinosa MJ, Vizcaino E, Murcia M, Fuentes V, Garcia AM, Rebagliato M, et al. Prenatal exposure to oragnochlorine compounds and neonatal thyroid stimulating hormone levels. J Expo Sci Environ Epidemiol. 2010;20:579–88. doi: 10.1038/jes.2009.47. [DOI] [PubMed] [Google Scholar]
- [14].Chevier J, Eskenazi B, Holland N, Bradman A, Barr DB. Effects of exposure to polychlorinated biphenyls and organochlorine pesticides on thyroid function during pregnancy. Am J Epidemiol. 2008;168:298–310. doi: 10.1093/aje/kwn136. [DOI] [PMC free article] [PubMed] [Google Scholar]
- [15].LaFranchi SH, Haddow JE, Hollowell JG. Is thyroid inadequacy during gestation a risk factor for adverse pregnancy and developmental outcomes? Thyroid. 2005;15:60–71. doi: 10.1089/thy.2005.15.60. [DOI] [PubMed] [Google Scholar]
- [16].Longnecker MP, Klebanoff MA, Zhou H, Brock JW. Association between maternal serum concentration of the DDT metabolite DDE and preterm and small-for-gestational-age babies at birth. Lancet. 2001;358:110–4. doi: 10.1016/S0140-6736(01)05329-6. [DOI] [PubMed] [Google Scholar]
- [17].Farhang L, Weintraub JM, Petreas M, Eskenazi B, Bhatia R. Association of DDT and DDE with Birth Weight and Length of Gestation in the Child Health and Development Studies, 1959–1967. American Journal of Epidemiology. 2005;162:717–25. doi: 10.1093/aje/kwi276. [DOI] [PubMed] [Google Scholar]
- [18].Wolff MS, Engel S, Berkowitz G, Teitelbaum , Siskind J, Barr DB, et al. Prenatal pesticide and PCB exposures and birth outcomes. Pediatr Res. 2007;61:243–50. doi: 10.1203/pdr.0b013e31802d77f0. [DOI] [PubMed] [Google Scholar]
- [19].Cleary-Goldman J, Malone FD, Lambert-Messerlian G, Sullivan L, Canick J, Porter TF, et al. Maternal thyroid hypofunction and pregnancy outcomes. Obstet Gynecol. 2008;112:85–92. doi: 10.1097/AOG.0b013e3181788dd7. [DOI] [PMC free article] [PubMed] [Google Scholar]
- [20].Abalovich M, Gutierrez S, Alcaraz G, Maccallini G, Garcia A, Levalle O. Overt and subclinical hypothryoidism complicating pregnancy. Thyroid. 2002;12:63–8. doi: 10.1089/105072502753451986. [DOI] [PubMed] [Google Scholar]
- [21].Hart MM, Whang-Peng J, Sieber SM, Fabro S, Adamson RH. Distribution and effects of DDT in the pregnant rabbit. Xenobiotica. 1972;2:567–74. doi: 10.3109/00498257209111084. [DOI] [PubMed] [Google Scholar]
- [22].DeLong RL, Gilmartin WG, Simpson JG. Premature births in California sea lions: association with high organochlorine pollutant residue levels. Science. 1973;181:1168–70. doi: 10.1126/science.181.4105.1168. [DOI] [PubMed] [Google Scholar]
- [23].Palmer K, Green S, Legator M. Dominant lethal study of p,p_-DDT in rats. Food Cosmet Toxicol. 1973;11:53–62. doi: 10.1016/0015-6264(73)90061-8. [DOI] [PubMed] [Google Scholar]
- [24].Ribas-Fito N, Sala M, Cardo E, Mazon C, De Muga ME, Verdu A, et al. Association of hexachlorobenzene and other organochlorine compounds with anthropometric measures at birth. Pediatr Res. 2002;52:163–7. doi: 10.1203/00006450-200208000-00006. [DOI] [PubMed] [Google Scholar]
- [25].Berkowitz GS, Lapinski RH, Wolff MS. The role of DDE and polychlorinated biphenyl levels in preterm birth. Arch Environ Contam Toxicol. 1996;30:139–41. doi: 10.1007/BF00211340. [DOI] [PubMed] [Google Scholar]
- [26].Torres-Arreola L, Berkowitz G, Torres-Sánchez L, López-Cervantes M, Cebrián ME, Uribe M, et al. Preterm birth in relation to maternal organochlorine serum levels. Ann Epidemiol. 2003;13:158–62. doi: 10.1016/s1047-2797(02)00424-6. [DOI] [PubMed] [Google Scholar]
- [27].Rogan WJ, Gladen BC, McKinney JD, Carreras N, Hardy P, Thullen J, et al. Neonatal effects of transplacental exposure to PCBs and DDE. J Pediatr. 1986;109:335–41. doi: 10.1016/s0022-3476(86)80397-3. [DOI] [PubMed] [Google Scholar]
- [28].Casey BM, Dashe JS, Wells CE, McIntire DD, Byrd W, Leveno KJ, et al. Subclinical hypothyroidism and pregnancy outcomes. Obstet Gynecol. 2005;105:239–45. doi: 10.1097/01.AOG.0000152345.99421.22. [DOI] [PubMed] [Google Scholar]
- [29].Stagnaro-Green A. Maternal Thyroid Disease and Preterm Delivery. J Clin Endocrinol Metab. 2009;94:21–5. doi: 10.1210/jc.2008-1288. [DOI] [PubMed] [Google Scholar]
- [30].Leung AS, Millar LK, Koonings PP, Montoro M, Mestman JH. Perinatal outcome in hypothyroid pregnancies. Obstet Gynecol. 1993;81:349–53. [PubMed] [Google Scholar]
- [31].Idris I, Srinivasan R, Simm A, Page RC. Maternal hypothyroidism in early and late gestation: effects on neonatal and obstetric outcome. Clin Endocrinol (Oxf) 2005;63:560–5. doi: 10.1111/j.1365-2265.2005.02382.x. [DOI] [PubMed] [Google Scholar]
- [32].van den Berg BJ, Christianson RE, Oechsli FW. The California Child Health and Development Studies of the School of Public Health, University of California at Berkeley. Paediatr Perinat Epidemiol. 1988;2:265–82. doi: 10.1111/j.1365-3016.1988.tb00218.x. [DOI] [PubMed] [Google Scholar]
- [33].Rogers E, Petreas M, Park J-S, Zhao G, Charles MJ. Evaluation of four capillary columns for the analysis of organochlorine pesticides, polychlorinated biphenyls, and polybrominated diphenyl ethers in human serum for epidemiologic studies. J Chromatogr B Analyt Technol Biomed Life Sci. 2004;813:269–85. doi: 10.1016/j.jchromb.2004.09.051. [DOI] [PubMed] [Google Scholar]
- [34].Park JS, Petreas M, Cohn BA, Cirillo PM, Factor-Litvak P. Hydroxylated PCB metabolites (OH-PCBs) in archived serum from 1950–60s California mothers: A pilot study. Environ Int. 2009;35:937–42. doi: 10.1016/j.envint.2009.04.002. [DOI] [PMC free article] [PubMed] [Google Scholar]
- [35].Sholtz RI, McLaughlin KR, Cirillo PM, Petreas M, Park J-S, Wolff MS, et al. Assaying organochlorines in archived serum for a large, long-term cohort: Implications of combining assay results from multiple laboratories over time. Environ Int. 2011;37:709–14. doi: 10.1016/j.envint.2011.01.013. [DOI] [PMC free article] [PubMed] [Google Scholar]
- [36].Cleveland WS. Robust locally weighted regression and smoothing scatterplots. J Am Stat Assoc. 1979;74:829–36. [Google Scholar]
- [37].Longnecker M, Wolff M, Gladen B, Brock J, Grandjean P, Jacobson J, et al. Comparison of polychlorinated biphenyl levels across studies of human neurodevelopment. Environ Health Perspect. 2003;111:65–70. doi: 10.1289/ehp.5463. [DOI] [PMC free article] [PubMed] [Google Scholar]
- [38].Govarts E, Nieuwenhuijsen M, Schoeters G, Ballester F, Bloemen K, de Boer M, et al. Birth weight and prenatal exposure to polychlorinated biphenyls (PCB) and dichlorodiphenyldichloroethylene (DDE): a meta-analysis within 12 European birth cohorts. Environ Health Perspect. 2011;120:162–70. doi: 10.1289/ehp.1103767. [DOI] [PMC free article] [PubMed] [Google Scholar]
- [39].Oken E, Kleinman K, Rich-Edwards J, Gillman M. A nearly continuous measure of birth weight for gestational age using a United States national reference. BMC Pediatr. 2003 Jul 8;3:6. doi: 10.1186/1471-2431-3-6. doi:10.1186/1471-2431-3-6. PMID: 12848901. [DOI] [PMC free article] [PubMed] [Google Scholar]
- [40].Schisterman EF, Whitcomb BW, Buck Louis GM, Louis TA. Lipid adjustment in the analysis of environmental contaminants and human health risks. Environ Health Perspect. 2005;113:853–7. doi: 10.1289/ehp.7640. [DOI] [PMC free article] [PubMed] [Google Scholar]
- [41].Kramer MS. Determinants of low birth weight: methodological assessment and meta-analysis. Bull World Health Organ. 1987;65:663–737. [PMC free article] [PubMed] [Google Scholar]
- [42].Messaros BM, Rossano MG, Liu G, Diamond MP, Friderici K, Nummy-Jernigan K, et al. Negative effects of serum p,p′-DDE on sperm parameters and modification by genetic polymorphisms. Environ Res. 2009;109:457–64. doi: 10.1016/j.envres.2009.02.009. [DOI] [PubMed] [Google Scholar]
- [43].Morgan D, Roan C. The metabolism of DDT in man. In: Hayes WJ, editor. Essays in Toxicology. Academic Press; New York: 1974. pp. 39–97. [Google Scholar]
- [44].Cohn BA, Cirillo PM, Christianson RE. Prenatal DDT exposure and testicular cancer: a nested case-control study. Arch Environ Occup Health. 2010;65:127–34. doi: 10.1080/19338241003730887. [DOI] [PMC free article] [PubMed] [Google Scholar]
- [45].Cohn BA, Cirillo PM, Wolff MS, Schwingl PJ, Cohen RD, Sholtz RI, et al. DDT and DDE exposure in mothers and time to pregnancy in daughters. Lancet. 2003;361:2205–6. doi: 10.1016/S0140-6736(03)13776-2. [DOI] [PubMed] [Google Scholar]
- [46].Kelce WR, Stone CR, Laws SC, Gray LE, Kemppainen JA, Wilson EM. Persistent DDT metabolite p,p′-DDE is a potent androgen receptor antagonist. Nature. 1995;375:581–5. doi: 10.1038/375581a0. [DOI] [PubMed] [Google Scholar]
- [47].Petridou E, Panagiotopoulou K, Katsouyanni K, Spanos E, Trichopoulos D. Tobacco smoking, pregnancy estrogens, and birth weight. Epidemiology. 1990;3:247–50. doi: 10.1097/00001648-199005000-00011. [DOI] [PubMed] [Google Scholar]
- [48].Kaijser M, Granath F, Jacobsen G, Cnattingius S, Ekbom A. Maternal pregnancy estriol levels in relation to anamnestic and fetal anthropometric data. Epidemiology. 2000;3:315–9. doi: 10.1097/00001648-200005000-00015. [DOI] [PubMed] [Google Scholar]
- [49].Hughes IA, Northstone K, Golding J, ALSPAC Study Team Reduced birth weight in boys with hypotspadias: an index of androgen dysfunction? Arch Dis Child Fetal Neonatal Ed. 2002 Jan 17;87:F150–1. doi: 10.1136/fn.87.2.F150. doi:10.1136/fn.87.2.F150. PMID: 12193526. [DOI] [PMC free article] [PubMed] [Google Scholar]
- [50].Gladen B, Shkiryak-Nyzhnyk Z, Chyslovska N, Zadorozhnaja T, Little R. Persistent organochlorine compounds and birth weight. Ann Epidemiol. 2003;13:151–7. doi: 10.1016/s1047-2797(02)00268-5. [DOI] [PubMed] [Google Scholar]
- [51].Savitz DA, Hertz-Picciotto I, Poole C, Olshan AF. Epidemiologic Measures of the Course and Outcome of Pregnancy. Epidemiologic Reviews. 2002;24:91–101. doi: 10.1093/epirev/mxf006. [DOI] [PubMed] [Google Scholar]
- [52].Eskenazi B, Harley K, Bradman A, Jewell NP, Barr DB, et al. Association of in utero organophosphate pesticide exposure and fetal growth and length of gestation in an agricultural population. Environ Health Perspec. 2004;112:1116–24. doi: 10.1289/ehp.6789. [DOI] [PMC free article] [PubMed] [Google Scholar]
- [53].Longnecker MP, Klebanoff MA, Gladen BC, Berendes HW. Serial levels of serum organochlorines during pregnancy and postpartum. Arch Environ Health. 1999;54:110–4. doi: 10.1080/00039899909602244. [DOI] [PubMed] [Google Scholar]
- [54].Jacobson JL, Jacobson SW. Intellectual impairment in children exposed to polychlorinated biphenyls in utero. New England Journal of Medicine. 1996;335:783–9. doi: 10.1056/NEJM199609123351104. [DOI] [PubMed] [Google Scholar]
- [55].Jurewicz J, Hanke W. Prenatal and childhood exposure to pesticides and neurobehavioral development: review of epidemiological studies. Int J Occup Med Environ Health. 2008;21:121–32. doi: 10.2478/v10001-008-0014-z. [DOI] [PubMed] [Google Scholar]
- [56].Eskenazi B, Marks AR, Bradman A, Harley K, Barr DB, Johnson C, et al. Organophosphate pesticide exposure and neurodevelopment in young Mexican-American children. Environ Health Perspect. 2007;115:792–8. doi: 10.1289/ehp.9828. [DOI] [PMC free article] [PubMed] [Google Scholar]
- [57].Beard J. DDT and human health. Sci Total Environ. 2006;355:78–89. doi: 10.1016/j.scitotenv.2005.02.022. [DOI] [PubMed] [Google Scholar]
- [58].Cox S, Niskar AS, Narayan KM, Marcus M. Prevalence of self-reported diabetes and exposure to organochlorine pesticides among Mexican Americans: Hispanic health and nutrition examination survey, 1982–1984. Environ Health Perspect. 2007;12:1747–52. doi: 10.1289/ehp.10258. [DOI] [PMC free article] [PubMed] [Google Scholar]
- [59].Karmaus W, Osuch JR, Eneli I, Mudd LM, Zhang J, Mikucki D, et al. Maternal levels of dichlorodiphenyl-dichloroethylene (DDE) may increase weight and body mass index in adult female offspring. Occup Environ Med. 2009;66:143–9. doi: 10.1136/oem.2008.041921. [DOI] [PubMed] [Google Scholar]
- [60].Gillman MW, Rifas-Shiman S, Berkey CS, Field AE, Colditz GA. Maternal gestational diabetes, birth weight, and adolescent obesity. Pediatrics. 2003 Mar 1;111:e221–6. doi: 10.1542/peds.111.3.e221. doi:0.1542/peds.111.3.e221. PMID: 12612275. [DOI] [PubMed] [Google Scholar]
- [61].Charney E, Goodman HC, McBride M, Lyon B, Pratt R, Breese B, et al. Childhood antecedents of adult obesity: do chubby infants become obese adults? NEJM. 1976;295:6–9. doi: 10.1056/NEJM197607012950102. [DOI] [PubMed] [Google Scholar]
- [62].Curhan GC, Chertow GM, Willett WC, Spiegelman D, Colditz GA, Manson JE, et al. Birth weight and adult hypertension and obesity in women. Circulation. 1996;94:1310–5. doi: 10.1161/01.cir.94.6.1310. [DOI] [PubMed] [Google Scholar]
- [63].Michels KB, Xue F. Role of birthweight in the etiology of breast cancer. Int J Cancer. 2006;119:2007–25. doi: 10.1002/ijc.22004. [DOI] [PubMed] [Google Scholar]
- [64].Ahlgren M, Wohlfahrt J, Olsen LW, Sørensen TIA, Melbye M. Birth weight and risk of cancer. Cancer. 2007;110:412–9. doi: 10.1002/cncr.22773. [DOI] [PubMed] [Google Scholar]
Associated Data
This section collects any data citations, data availability statements, or supplementary materials included in this article.