Abstract
Intracellular recycling pathways play critical roles in internalizing membrane and fluid phase cargo and in balancing the inflow and outflow of membrane and cell surface molecules. To identify proteins involved in the regulation of endocytic recycling, we used an shRNA trafficking library and screened for changes in the surface expression of CD1a antigen-presenting molecules that follow an endocytic recycling route. We found that silencing of the ADP-ribosylation factor (Arf)-like small GTPase Arl13b led to a decrease in CD1a surface expression, diminished CD1a function, and delayed CD1a recycling, suggesting that Arl13b is involved in the regulation of endocytic recycling traffic. Arl13b appears to be required for the major route of endocytic trafficking, causing clustering of early endosomes and leading to the accumulation of endocytic cargo. Moreover, Arl13b colocalized with markers of the endocytic recycling pathway followed by CD1a, namely Arf6 and Rab22a. We also detected an interaction between Arl13b and the actin cytoskeleton. Arl13b was previously implicated in cilia formation and function. Our present results indicate a previously unidentified role for Arl13b in endocytic recycling traffic and suggest a link between Arl13b function and the actin cytoskeleton.
Keywords: immune response, lipid antigens, ciliary traffic, vesicle traffic
Eukaryotic cells sample the extracellular milieu to take up fluid and receptor-bound ligands. Many receptors release their ligands in the mildly acidic environment of early endosomes. From this compartment, also known as the sorting endosome, membrane-bound receptors and other plasma membrane proteins can recycle back to the plasma membrane, whereas fluid phase cargo and membrane proteins destined for utilization or degradation follow the endocytic pathway to late endosomes and lysosomes. Recycling back to the plasma membrane can occur via a fast recycling pathway or, alternatively, a slow recycling pathway through the perinuclear endocytic recycling compartment (ERC). Cells take up considerable amounts of membrane from the cell surface; thus, recycling pathways are essential to return membrane, membrane proteins, and lipids to the cell surface, balancing the internalized flow. Along with this function, regulated recycling has been shown to be important for cytokinesis, cell adhesion, morphogenesis, cell fusion, and even learning and memory (1).
However, the machinery involved in the sorting and trafficking of recycling cargo through the ERC remains incompletely understood. We previously reported that CD1a, an MHC class I-like antigen-presenting molecule that presents lipids rather than peptides to T cells, follows an endocytic recycling pathway similar to that used by MHC class I and other cargo internalized independent of clathrin (2). This pathway has been shown to depend on the small GTPases Rab22a and Arf6 (3, 4).
To uncover previously unknown proteins involved in regulation of the endocytic recycling pathway followed by CD1a and other clathrin-independent cargo, we developed an shRNA library targeting the main families of proteins involved in membrane trafficking, including Rab, Arf, Arf-like (Arl), soluble N-ethylmaleimide-sensitive factor attachment protein receptor (SNARE), endosomal sorting complex required for transport (ESCRT) proteins, and other trafficking proteins (5). To identify proteins involved in endocytic recycling, we screened for the shRNAs that block CD1a recycling and function. We found that silencing of the small GTPase Arl13b impaired CD1a expression at the cell surface and its function in lipid antigen presentation. Moreover, Arl13b silencing resulted in characteristic clustering of early endosome antigen (EEA) 1-positive early endosomes and coclustering of recycling cargo, as well as cargo destined for lysosomes. These findings indicate a role for Arl13b in the regulation of endocytic trafficking, complementing previous studies reporting a role in the biology of the primary cilium and Sonic Hedgehog (Shh) signaling (6, 7). Furthermore, we found an association between tubular structures labeled by Arl13b and actin filaments and an interaction between Arl13b and actin that provide mechanistic insight into how Arl13b could regulate endocytic recycling traffic.
Results
Arl13b Silencing Leads to Reduced CD1a Surface Expression and Function.
We assembled an shRNA library targeting members of protein families known to be involved in membrane trafficking, including Rabs, Arfs/Arls, SNAREs, ESCRT, tetraspanins, and adaptor protein complexes, among others (Dataset S1). The library included five hairpins per gene and covered 211 genes. It has been shown that in the case of recycling membrane proteins, such as transferrin receptor (TfR), perturbations in trafficking often lead to changes in surface expression of the protein (8). Given that CD1a molecules follow an endocytic recycling pathway (2), we used flow cytometry to screen HeLa:CD1a stable transfectants for changes in CD1a surface expression after RNA silencing.
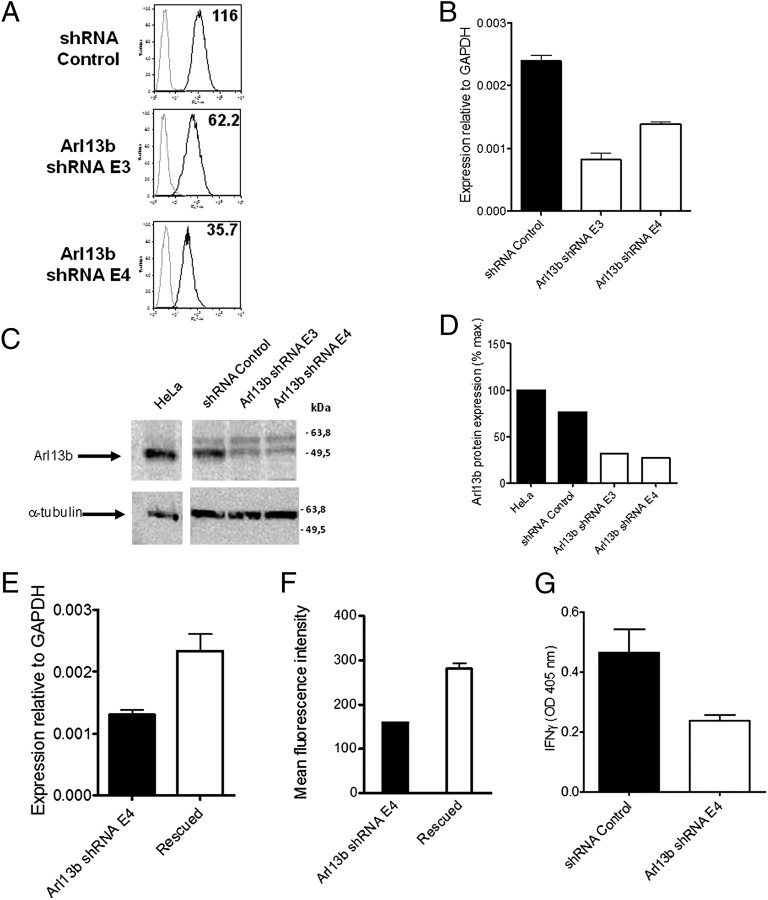
We chose candidate shRNAs exhibiting statistically significant differences in cell surface expression from controls, in which more than one hairpin against a certain gene exhibited the same trend, and which could be repeated in at least three separate assays. After screening ∼1,050 shRNAs in 96-well plates, we selected a candidate that provided a particularly robust reduction in CD1a surface expression. Two hairpins targeting the Arl13b protein resulted in a ∼50–70% reduction in the mean fluorescence intensity measurement of CD1a surface expression (Fig. 1A). We next verified that the hairpins that provided reduced CD1a surface expression and function correspondingly decreased the expression of ARL13B. By quantitative PCR, we found that Arl13b mRNA levels were reduced by 62% with the E3 hairpin and by 43% with the E4 hairpin (Fig. 1B). Western blot analysis showed a 68.5% reduction in Arl13b protein levels with the E3 hairpin and a 73.4% reduction with the E4 hairpin (Fig. 1 C and D).
Fig. 1.
Arl13b silencing leads to a decrease in CD1a surface expression and function. (A) HeLa:CD1a stable transfectant cells were transduced with lentiviruses encoding Arl13b-targeting shRNA (E3 and E4) or control (Mission) and selected in puromycin for 1 wk. CD1a surface levels were analyzed by flow cytometry (black lines). Gray lines correspond to isotype controls. The number in each panel represents the geometric mean fluorescence intensity. Results are representative of at least two independent experiments. (B) Arl13b mRNA levels were analyzed by quantitative RT-PCR. Error bars represent SD of triplicate measurements, and the results are representative of at least three independent experiments. (C) Arl13b protein expression levels from total cell lysates were analyzed by Western blot using anti-Arl13b affinity-purified Ab. α-tubulin was used as an internal loading control. (D) Quantification of band intensities from C using ImageJ software. (E and F) HeLa:CD1a:shRNA cells were transfected with Arl13b (“rescued”) or an empty vector (“Arl13b shRNA E4”) and selected in blasticydin. Arl13b mRNA levels were analyzed by quantitative RT-PCR (E), and CD1a surface levels were analyzed by flow cytometry (F). Error bars represent the SE of four cell lines for rescued and two lines for Arl13b shRNA E4. Analysis of Arl13b mRNA levels (E) was done in triplicate. (G) Cells transduced with Arl13b-targeting shRNA or control (Mission) were incubated with 0.11 μg/mL sonicated antigen and CD8-2 T cells, which recognize DDM presented by CD1a. After 22 h, the release of IFN-γ was measured by ELISA. Error bars represent SE of two Arl13b-targeting shRNAs and two control samples, each analyzed in triplicate. The results are representative of three independent experiments.
Despite this reduction in Arl13b expression, RNAi can induce off-target effects, that is, silencing of an additional gene that could be responsible for the observed phenotype. To confirm that the observed phenotype resulted specifically from Arl13b silencing, we carried out rescue experiments by stably overexpressing Arl13b. Among the stable cell lines with Arl13b gene expression rescued (i.e., increased, as determined by mRNA levels) with either Arl13b WT or a rescue construct not subject to shRNA silencing owing to silent point mutations, we also found markedly higher CD1a surface expression in four of the five lines (Fig. 1 E and F).
We next examined whether Arl13b silencing affected CD1a function. For this, we incubated HeLa:CD1a cells transduced with shRNAs targeting Arl13b or negative control shRNA with dideoxymycobactin (DDM) lipid antigen and corresponding antigen-specific T cells (9). T-cell recognition of the CD1a-presented antigen resulted in IFN-γ secretion, as determined by ELISA of the supernatants. An approximate twofold reduction in IFN-γ production was noted when Arl13b was silenced (Fig. 1G). Taken together, these results indicate that silencing of Arl3b leads to decreased CD1a surface expression and reduced function in CD1a antigen presentation to T cells.
Arl13b Silencing Causes Early Endosome Clustering and Delayed CD1a Recycling.
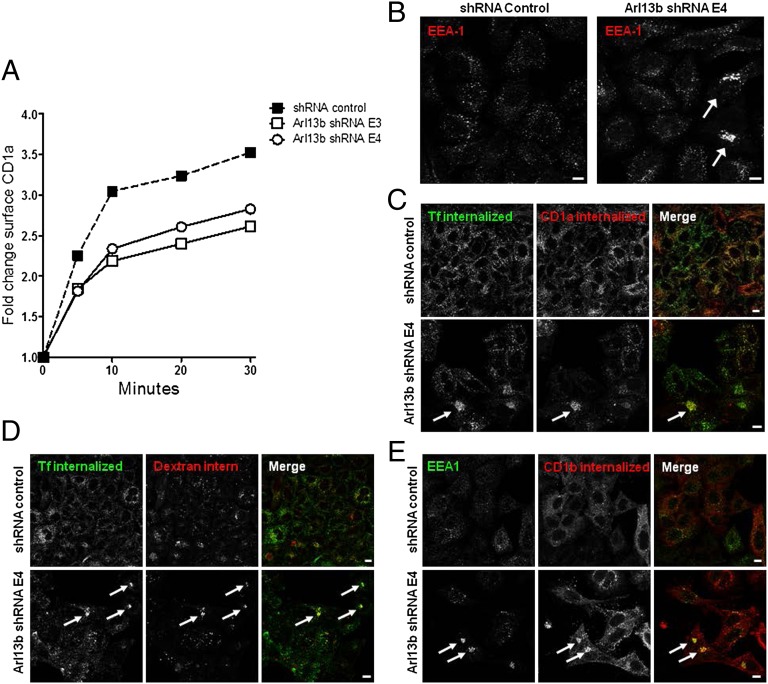
To determine whether the decrease in CD1a surface expression resulted from a decrease in protein recycling, we followed the reappearance on the cell surface of CD1a/anti-CD1a complexes that had been previously internalized. We found that the recycling of CD1a was impaired through the silencing of Arl13b by the E3 and E4 shRNAs (Fig. 2A), suggesting that Arl13b is involved in the regulation of endocytic recycling trafficking of CD1a.
Fig. 2.
Arl13b silencing impairs CD1a recycling and results in early endosome clustering. (A) HeLa cells were transiently cotransfected with CD1a and Arl13b-targeting shRNA (E3 and E4) or control (Mission), selected in puromycin, and incubated with anti-CD1a Ab on ice and then at 37 °C to allow for internalization. After the surface-bound Ab was stripped in a brief acidic wash, cells were incubated at 37 °C for different periods to allow for recycling. Cells were then stained with anti-mouse secondary Ab and analyzed by flow cytometry. The results (in arbitrary units) are normalized for recycling at time 0. The results are representative of three independent experiments. (B–E) HeLa:CD1a (B and C) or HeLa:CD1b (E) stable transfectant cells or HeLa untransfected cells (D) were transfected with Arl13b-targeting shRNA (Lower) or control (Upper) and selected in puromycin. (B) Cells were fixed, permeabilized, and stained with anti-EEA1 primary Ab, followed by Alexa Fluor 546-conjugated anti-mouse secondary Ab. (C) Cells were serum-starved for 30 min and then incubated with anti-CD1a mAb for 30 min on ice. Cells were then washed and incubated for 15 min at 37 °C with Alexa Fluor 488-conjugated transferrin (green). After the surface-bound Ab was stripped in a brief acidic wash, cells were fixed, permeabilized, and labeled with Alexa Fluor 546-conjugated anti-mouse to detect anti-CD1a (red). (D) Cells were serum-starved for 30 min and incubated with Alexa Fluor 488-conjugated transferrin (green) and Alexa Fluor 546-conjugated dextran (red) for 15 min. After washing, cells were chased for 20 min and then fixed. (E) Cells were incubated with anti-CD1b mAb for 30 min on ice, and then washed and incubated for 30 min at 37 °C. After the surface-bound Ab was stripped as in B, cells were fixed, permeabilized, stained for EEA1 with a rabbit polyclonal Ab (green), and labeled with Alexa Fluor 546-conjugated anti-mouse to detect anti-CD1b (red). Arrows indicate cargo clustering. (Scale bars: 10 μm.)
To provide further insight into the reason for the blockade of CD1a recycling from Arl13b silencing, we analyzed the morphology of the main compartments of the secretory and endocytic pathways by confocal microscopy. We found clustering of early endosomes labeled with anti-EEA1 mAb, albeit in a small subset of cells and only after prolonged silencing by shRNA (Fig. 2B). We confirmed this result using two other markers that localize to early/recycling endosomes, namely Sorting nexin 1 and TfR, and observed that both molecules coclustered with EEA1 in cells in which Arl13b was silenced (Fig. S1).
Based on our finding that CD1a surface expression and recycling were affected by Arl13b silencing, we also tested whether CD1a-positive endosomes became clustered when Arl13b was silenced. We allowed HeLa cells to internalize transferrin (Tf) and CD1a bound to an Ab, and found coclustering of internalized CD1a and Tf (Fig. 2C). This finding suggests that Arl13b silencing results in a blockade of endocytic recycling trafficking that goes through the early endosomes. To investigate this futher, we performed a double-pulse/chase of Tf, which recycles through early and recycling endosomes, along with a pulse/chase of dextran, which follows the endocytic pathway through early endosomes to late endosomes and finally to lysosomes. In control cells, we found no colocalization between Tf and dextran as they trafficked to different compartments after the pulse/chase period. However, we detected coclustering of both molecules in Arl13b-silenced cells, suggesting a blockade of trafficking out of early/sorting endosomes (Fig. 2D). This blockade affects both endocytic recycling cargo, such as Tf and CD1a, and cargo that follows the endocytic pathway to lysosomes, such as dextran. To confirm this finding, we examined the distribution of the CD1b isoform, which, unlike CD1a, traffics exclusively to lysosomes (10). Strikingly, when CD1b was internalized in complex with an Ab, it clustered in EEA1-positive early endosomes (Fig. 2E) rather than trafficking to lysosomes, further suggesting that the trafficking of cargo through early endosomes, including cargo destined for lysosomes, is blocked when Arl13b is silenced.
Arl13b Localizes to Tubular-Vesicular Structures of the Arf6- and Rab22a- Dependent Recycling Pathway.
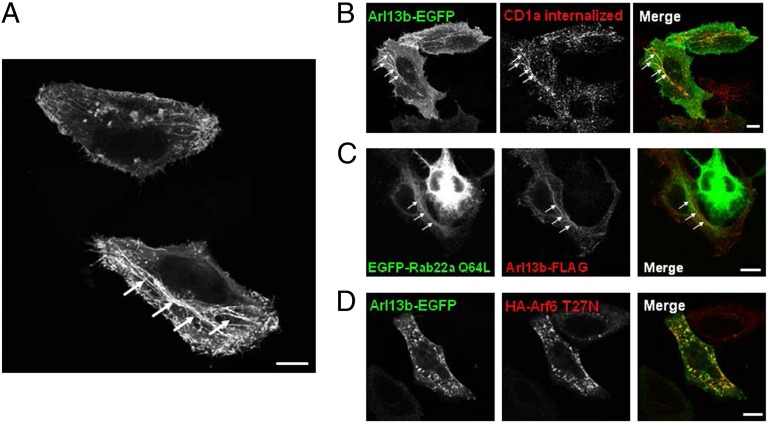
Arl13b has been found to associate with primary cilia in several cell types, including fibroblasts and retinal pigment epithelium cells (6, 7, 11). However, HeLa cells do not form cilia (12). When we investigated the localization of Arl13b in HeLa cells by expressing Arl13b fused with EGFP at the C terminus (7), we observed tubular-vesicular intracellular staining, as well as plasma membrane staining (Fig. 3A). Importantly, we noted a similar intracellular distribution when we expressed FLAG-tagged Arl13b, indicating that the EGFP tag did not affect localization of Arl13b (Fig. S2A). Given that Arl13b has been identified as a ciliary protein (7), we also confirmed that Arl13b-EGFP in ciliated NIH 3T3 mouse fibroblasts localizes to the primary cilium, suggesting that transfected Arl13b-EGFP can be used to follow the trafficking of this protein (Fig. S2B).
Fig. 3.
Arl13b-EGFP colocalizes with CD1a, Rab22a, and Arf6 in HeLa cells. (A) HeLa cells were transiently transfected with Arl13b-EGFP and analyzed by confocal microscopy. Arrows indicate Arl13b-decorated tubular structures. (B) HeLa cells were cotransfected with CD1a and Arl13b-EGFP and incubated with anti-CD1a mAb for 30 min on ice. Cells were then washed and incubated for 30 min at 37 °C. After the surface-bound Ab was stripped in a brief acidic wash, cells were fixed, permeabilized, and labeled with Alexa Fluor 546-conjugated anti-mouse secondary Ab to detect anti-CD1a (red). (C) HeLa cells were transiently cotransfected with EGFP-Rab22a Q64L and Arl13b-FLAG, fixed, permeabilized, and stained with Cy3-conjugated mouse monoclonal anti-FLAG Ab (red). (D) HeLa cells were transiently cotransfected with Arl13b-EGFP and HA-Arf6 T27N, fixed, permeabilized, and stained with anti-HA tag polyclonal Ab followed by Alexa Fluor 546-conjugated anti-rabbit secondary Ab (red). (Scale bars: 10 μm.)
CD1a and other cargo internalized independent of clathrin follow an endocytic recycling pathway regulated by Arf6 and Rab22a (2, 3). Interestingly, Rab22a localizes to tubular recycling structures in which CD1a localizes as well (2, 3). Therefore, to determine whether the tubules labeled with Arl13b-EGFP also contained CD1a and Rab22a, we transfected Arl13b-EGFP in HeLa cells stably transfected with CD1a, bound anti-CD1a Ab to CD1a, and allowed the complexes to internalize. We observed a striking colocalization between Arl13b-EGFP and internalized CD1a in both tubular and vesicular structures (Fig. 3B). To analyze the colocalization with Rab22a, we transfected Arl13b-FLAG and the constitutively active form of Rab22a (Rab22a Q64L) tagged with EGFP, and found that they both colocalized along tubules (Fig. 3C). This suggests that Arl13b in HeLa cells localizes to the endocytic recycling pathway followed by CD1a and other cargo internalized independent of clathrin (3).
We next investigated the colocalization of Arl13b-EGFP with Arf6. Overexpression of Arf6 T27N dominant-negative mutant leads to the accumulation of the protein in recycling vesicles in which clathrin-independent cargo, such as CD1a, accumulates as well (10). Strikingly, we observed Arl13b-EGFP colocalization in Arf6 T27N-positive recycling vesicles in HeLa cells (Fig. 3D), as well as in NIH 3T3 cells (Fig. S3A). Moreover, in NIH 3T3 cells overexpressing the Arf6 Q67L constitutively active mutant, which sequesters cargo that normally follows the Arf6-dependent pathway (2, 4), we observed an accumulation of vesicles containing both Arl13b and Arf6 (Fig. S3B). These results suggest that Arl13b might be involved in regulating the Arf6- and Rab22a-dependent endocytic recycling pathway followed by CD1a.
Arl13b Associates with the Actin Cytoskeleton.
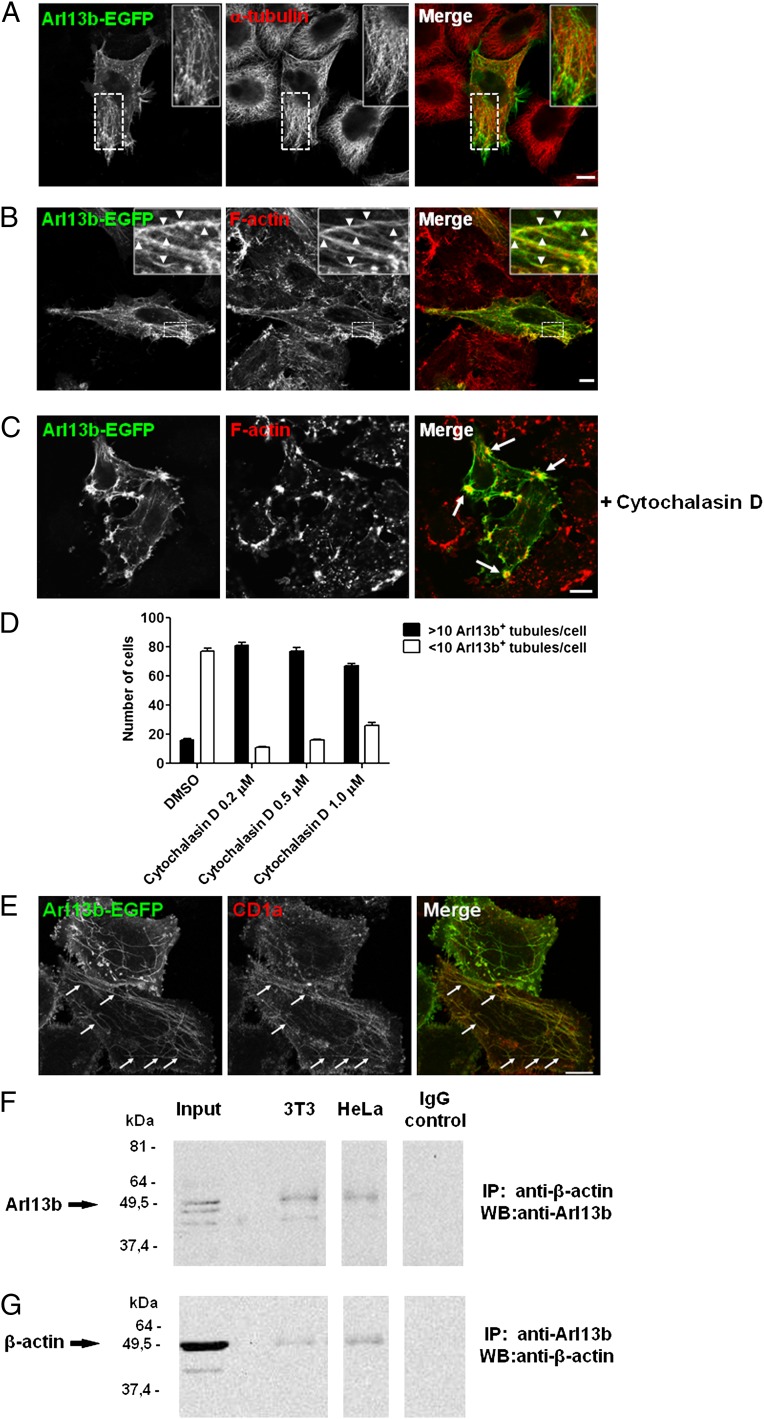
Links between Arl proteins and microtubules are well documented. Arl2 and Arl3 bind proteins that are tubulin-folding cofactors and regulate microtubule-dependent processes (13, 14). Moreover, Arl8b has been shown to bind to microtubules (15). On the other hand, Arl4d has been shown to be involved in actin remodeling (16). Because Arl13b is associated with primary cilia, which are microtubule-based structures, we tested for a link between Arl13b and microtubules. We compared the distribution of tubules labeled with Arl13b-EGFP and microtubules labeled with anti–α-tubulin mAb and found a low degree of colocalization between Arl13b and microtubules (Fig. 4A). However, some functional association with microtubules may exist as treatment with nocodazole, which depolimerizes microtubules, rescued Arl13b silencing-induced endosomal clustering (Fig. S4 A and B).
Fig. 4.
Arl13b interacts with actin. (A–C) HeLa cells were transfected with Arl13b-EGFP for 24 h, treated with 5 µM of cytochalasin D for 45 min (C) or untreated, fixed, permeabilized, and stained with anti–α-tubulin Ab (A) or Alexa Fluor 568-conjugated phalloidin, which labels filamentous actin (B and C) (red). Arrowheads indicate actin filaments that colocalize with Arl13b tubules. Arrows indicate plasma membrane accumulations of actin and Arl13b. (D and E) HeLa:CD1a stable transfectant cells were transiently transfected with Arl13b-EGFP for 24 h, treated with cytochalasin D for 45 min at the indicated concentrations (0.2 µM in E), fixed, permeabilized, and stained with anti-CD1a mAb (red). The number of Arl13b tubules per cell was counted (n = 95) and grouped into cells with >10 and <10 tubules per cell (D). Arrows indicate Arl13b and CD1a colocalization in tubules (E). Results are representative of two independent experiments. (F and G) HeLa and NIH 3T3 cell lysates were immunoprecipitated with mouse monoclonal anti–β-actin (F) or rabbit polyclonal anti-Arl13b (G) Ab, separated by SDS/PAGE, and immunoblotted with anti-Arl13b (F) or anti–β-actin (G) Ab, followed by HRP-conjugated anti-rabbit or anti-mouse secondary Ab, respectively. Rabbit and mouse IgG were used as negative controls. (Scale bars: 10 μm.)
We then investigated a possible link between Arl13b and other cytoskeletal elements, such as actin filaments. Strikingly, we found Arl13b-EGFP localization in actin-rich regions at the plasma membrane and significant colocalization between tubules labeled by Arl13b-EGFP and filamentous actin stress fibers (Fig. 4B). This suggests that the recycling tubules identified by the presence of Arl13b-EGFP (Fig. 3A) are linked to the actin cytoskeleton.
To further test the association between Arl13b and the actin cytoskeleton, we used cytochalasin D, which depolymerizes the actin cytoskeleton. After treating the cells with 5 µM cytochalasin D, we observed a retraction of the actin stress fibers, followed by Arl13b-EGFP (Fig. 4C). Arf6-labeled tubules have been shown to increase and trap recycling cargo at low doses of cytochalasin D (17); thus, we used cytochalasin D in lower doses (0.2–1 µM), and observed a striking increase in the number of Arl13b-EGFP tubules per cell (Fig. 4D). Moreover, these tubules contained CD1a, as expected (Fig. 4E). Finally, we were able to coimmunoprecipitate Arl13b and actin when we used either anti-Arl13b or anti–β-actin Ab for immunoprecipitation in both HeLa and NIH 3T3 cells (Fig. 4 F and G). Importantly, when we silenced Arl13b, immunoprecipitation of actin demonstrated no band corresponding to Arl13b, confirming the specificity of the signal obtained when both proteins are coimmunoprecipitated (Fig. S5A). In contrast, we detected no interaction between Arl13b and tubulin (Fig. S5B).
Given that Arl13b-EGFP–labeled tubules colocalize with actin filaments and Arl13b interacts with actin, we tested whether tubules containing CD1a, which also colocalize with Arl13b-EGFP (Fig. 3B), colocalize with actin in untransfected cells. We observed colocalization between CD1a-labeled tubules and actin filaments, demonstrating that this is not a result of Arl13b-EGFP overexpression (Fig. S6).
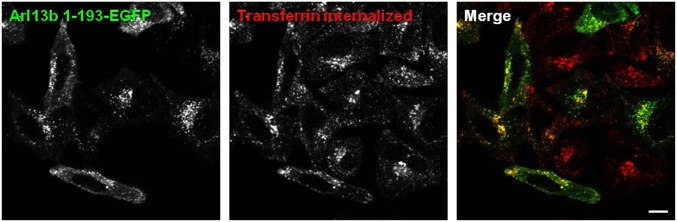
C-Terminal Domain of Arl13b Is Required for Tubular and Plasma Membrane Localization of the Protein.
Arl13b differs significantly from all other Arl proteins in having a long C-terminal tail that, apart from a coiled coil and proline-rich regions, has no motifs that suggest its function. To investigate the role of Arl13b C-terminal tail, we deleted the last 235 residues of the protein to create Arl13b 1–193–EGFP truncated protein, and found that when overexpressed, this protein displayed a very different intracellular distribution that became punctate without the striking plasma membrane or tubular localization of Arl13b WT (Fig. 5). Moreover, the labeled vesicles colocalized partially with endocytic recycling cargo, such as Tf (Fig. 5), and also with early endosome and recycling endosome markers, such as EEA1 and Rab11a, respectively (Fig. S7 A and B), suggesting that the C-terminal tail of Arl13b not present on other Arls is necessary for localization to tubular recycling structures and the plasma membrane. Importantly, the colocalization of Arl13b 1–193–EGFP with endocytic recycling cargo further supports the link between this protein and the endocytic recycling pathway, and the absence of strong tubular localization of Arl13b 1–193 indicates that the overlap between Arl13b WT-labeled tubules and actin filaments is specific. Nevertheless, in cells expressing Arl13b 1–193–EGFP, CD1a was found in tubules in which Arl13b 1–193 was detected only faintly, suggesting that the recycling tubules containing CD1a are not ablated, and confirming that Arl13b 1–193 has a markedly decreased capacity to decorate the tubular structures containing cargo such as CD1a (Fig. S7C).
Fig. 5.
Arl13b 1–193 truncation mutant displays a punctate distribution and colocalizes with transferrin. HeLa cells were transfected with Arl13b 1–193-EGFP, starved, pulsed with Alexa Fluor 546-conjugated transferrin (red) for 30 min and fixed. (Scale bar: 10 μm.)
Discussion
Our results indicate that Arl13b is involved in the regulation of endocytic recycling traffic, as indicated by the altered recycling of CD1a when Arl13b is silenced. The defect is a more general phenomenon that also affects endocytic trafficking to late endosomes and lysosomes, as demonstrated by the coclustering of two proteins that follow this route, dextran and CD1b, with markers for early endosomes in Arl13b-silenced cells. Moreover, in a dynamic pulse-chase assay, the progression of dextran along the endocytic pathway was delayed because it colocalized with Tf, which follows the endocytic recycling pathway. Thus, our data indicate that defects in early/sorting endosomes caused by Arl13b silencing could lead to perturbations in both endocytic recycling trafficking and endocytic trafficking to late endosomes and lysosomes.
Previous studies have shown that mutation of Arl13b causes deficient Shh signaling, presumably owing to abnormalities in primary cilia, leading to developmental defects (6). That and other studies have reported localization of Arl13b to cilia, but the function of this protein remains undefined (7, 11). Interestingly, recent studies have suggested a link between the ERC and cilia. Rab11, a marker for the ERC, localizes at the base of primary cilia and is essential for ciliogenesis (18). Moreover, ERC markers colocalize with the pericentrosomal preciliary compartment, which stores ciliary proteins during the early phase of ciliogenesis (19). Another study found that a dominant-negative mutant of Rab5 involved in early endosome homotypic fusion affects ciliary trafficking of the protein Kim1 (20). Taken together, the foregoing findings and our data on Arl13b point to the existence of membrane traffic pathways linking early endosomes, the ERC, and primary cilia, and suggest that these pathways are essential for both the proper localization of primary cilium proteins and endocytic recycling. Thus, it is tempting to speculate that Arl13b regulates such trafficking pathways.
We found a distribution of Arl13b in HeLa cells along the plasma membrane, as well as intracellular tubular-vesicular staining. In some cells, the tubules extended throughout the length of the cell and appeared similar to the recycling tubules that contain Rab22a and CD1a described by us and by others (2, 3). These tubules were extensively colocalized with CD1a-positive and Rab22a-positive tubules, indicating that they could serve as recycling structures. Moreover, Arl13b colocalizes with Arf6, similar to CD1a.
Our findings also suggest a link between Arl13b and the actin cytoskeleton. We detected a high degree of colocalization between Arl13b-labeled tubules and actin filaments. Interestingly, another Arl protein, Arl4d, has been linked to actin (16); however, Arl4d seems to regulate actin indirectly through Arf guanine nucleotide exchange factors (GEFs), whereas we found that Arl13b interacts with actin, although we did not evaluate whether the interaction is direct or via effectors. A recent study showed that actin filaments can mediate long-range transport of recycling endosomes to the cell periphery (21). In addition, the recycling of clathrin-independent cargo, such as MHC class I, is blocked by latrunculin A, which inhibits actin polymerization (3). Moreover, actin is involved in the formation of tubular endosomes that recycle β2-adrenergic receptor, another molecule internalized independent of clathrin (22). Also of interest, a role for the actin cytoskeleton in ciliogenesis has been established (19). That study also found that silencing of a regulator of cargo sorting at early endocytic compartments causes the accumulation of Smoothened (Smo), an essential component of Shh that normally localizes to primary cilia, in EEA1-positive early endosomes. Of note, the trafficking pathway of Smo is controversial (23, 24). Given the recent report that Arl13b regulates entry of Smo into cilia (25), our results strongly support a model in which Smo uses the endocytic recycling pathway to traffic to primary cilia, and could explain the defects in Shh signaling observed in Arl13b-deficient mice.
Notably, we observed only limited staining overlap between Arl13b-labeled tubules and α-tubulin, with no coimmunoprecipitation. Nevertheless, we found evidence of a functional role of microtubules in the early endosome clustering caused by Arl13b silencing, given that this phenotype was alleviated when microtubules were depolymerized. In addition, the depolymerization of microtubules with nocodazole affected the stability of the tubules labeled by Arl13b-EGFP. Thus, it is possible that the actin and microtubule cytoskeletons cooperate to localize early/sorting endosomes at the cell periphery, as recently proposed by Ohashi et al. (26).
In conclusion, we present evidence supporting a previously unappreciated role for Arl13b in endocytic recycling traffic, which now promises important functions in parallel to those shown previously in ciliogenesis.
Materials and Methods
shRNA Library Screening.
All lentiviruses were produced in accordance with protocols publicly available at the Broad Institute’s RNAi Consortium Web site (www.broadinstitute.org/rnai/public/resources/protocols). pLKO.1 plasmid was obtained from the RNAi Consortium. For lentiviral transduction, HeLa CD1a cells were plated at 15,000 cells per well in 96-well flat-bottomed plates in 6 μg/mL polybrene (hexadimethrine bromide; Sigma-Aldrich) with 20 μL of viral supernatant. At 48 h after transduction, infected cells were selected by the addition of puromycin (Sigma-Aldrich) to a final concentration of 2.5 μg/mL. At 3 d after selection, cells were detached using 0.05% trypsin-EDTA in PBS (Invitrogen) and stained with PE-conjugated anti-CDa mAb (BD Pharmingen) in flow cytometry buffer [PBS and 2% (vol/vol) FBS]. Cells were analyzed for CD1a cell surface levels on a BD Biosciences LSR-II cytometer equipped with a high-throughput sampler. Data were acquired using FACSDiva (BD Biosciences) and analyzed using FlowJo (Tree Star). For more details on the materials and methods used, please see SI Materials and Methods.
Supplementary Material
Acknowledgments
We thank Dr. D. B. Moody (Brigham and Women’s Hospital, Harvard Medical School), Dr. J. G. Donaldson [National Heart, Lung, and Blood Institute, National Institutes of Health (NIH)], Dr. T. Caspary (Emory University), and Dr. K. Kontani (University of Tokyo) for the kind gifts of reagents; and Rosa Grenha for technical assistance. This work was supported by NIH Grant RO1 AI028973 (to M.B.B.) and by a postdoctoral fellowship from the Arthritis Foundation, a Marie Curie International Reintegration Grant, and a grant from Câmara Municipal de Oeiras (to D.C.B.).
Footnotes
The authors declare no conflict of interest.
This article contains supporting information online at www.pnas.org/lookup/suppl/doi:10.1073/pnas.1218272110/-/DCSupplemental.
References
- 1.Grant BD, Donaldson JG. Pathways and mechanisms of endocytic recycling. Nat Rev Mol Cell Biol. 2009;10(9):597–608. doi: 10.1038/nrm2755. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 2.Barral DC, et al. CD1a and MHC class I follow a similar endocytic recycling pathway. Traffic. 2008;9(9):1446–1457. doi: 10.1111/j.1600-0854.2008.00781.x. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 3.Weigert R, Yeung AC, Li J, Donaldson JG. Rab22a regulates the recycling of membrane proteins internalized independently of clathrin. Mol Biol Cell. 2004;15(8):3758–3770. doi: 10.1091/mbc.E04-04-0342. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 4.Brown FD, Rozelle AL, Yin HL, Balla T, Donaldson JG. Phosphatidylinositol 4,5-bisphosphate and Arf6-regulated membrane traffic. J Cell Biol. 2001;154(5):1007–1017. doi: 10.1083/jcb.200103107. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 5.Garg S, et al. Lysosomal trafficking, antigen presentation, and microbial killing are controlled by the Arf-like GTPase Arl8b. Immunity. 2011;35(2):182–193. doi: 10.1016/j.immuni.2011.06.009. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 6.Caspary T, Larkins CE, Anderson KV. The graded response to Sonic Hedgehog depends on cilia architecture. Dev Cell. 2007;12(5):767–778. doi: 10.1016/j.devcel.2007.03.004. [DOI] [PubMed] [Google Scholar]
- 7.Hori Y, Kobayashi T, Kikko Y, Kontani K, Katada T. Domain architecture of the atypical Arf-family GTPase Arl13b involved in cilia formation. Biochem Biophys Res Commun. 2008;373(1):119–124. doi: 10.1016/j.bbrc.2008.06.001. [DOI] [PubMed] [Google Scholar]
- 8.Benmerah A, Poupon V, Cerf-Bensussan N, Dautry-Varsat A. Mapping of Eps15 domains involved in its targeting to clathrin-coated pits. J Biol Chem. 2000;275(5):3288–3295. doi: 10.1074/jbc.275.5.3288. [DOI] [PubMed] [Google Scholar]
- 9.Moody DB, et al. T cell activation by lipopeptide antigens. Science. 2004;303(5657):527–531. doi: 10.1126/science.1089353. [DOI] [PubMed] [Google Scholar]
- 10.Sugita M, et al. Separate pathways for antigen presentation by CD1 molecules. Immunity. 1999;11(6):743–752. doi: 10.1016/s1074-7613(00)80148-x. [DOI] [PubMed] [Google Scholar]
- 11.Cevik S, et al. Joubert syndrome Arl13b functions at ciliary membranes and stabilizes protein transport in Caenorhabditis elegans. J Cell Biol. 2010;188(6):953–969. doi: 10.1083/jcb.200908133. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 12.Alieva IB, Gorgidze LA, Komarova YA, Chernobelskaya OA, Vorobjev IA. Experimental model for studying the primary cilia in tissue culture cells. Membr Cell Biol. 1999;12(6):895–905. [PubMed] [Google Scholar]
- 13.Zhou C, Cunningham L, Marcus AI, Li Y, Kahn RA. Arl2 and Arl3 regulate different microtubule-dependent processes. Mol Biol Cell. 2006;17(5):2476–2487. doi: 10.1091/mbc.E05-10-0929. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 14.Gillingham AK, Munro S. The small G proteins of the Arf family and their regulators. Annu Rev Cell Dev Biol. 2007;23:579–611. doi: 10.1146/annurev.cellbio.23.090506.123209. [DOI] [PubMed] [Google Scholar]
- 15.Okai T, et al. Novel small GTPase subfamily capable of associating with tubulin is required for chromosome segregation. J Cell Sci. 2004;117(Pt 20):4705–4715. doi: 10.1242/jcs.01347. [DOI] [PubMed] [Google Scholar]
- 16.Li CC, et al. ARL4D recruits cytohesin-2/ARNO to modulate actin remodeling. Mol Biol Cell. 2007;18(11):4420–4437. doi: 10.1091/mbc.E07-02-0149. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 17.Radhakrishna H, Donaldson JG. ADP-ribosylation factor 6 regulates a novel plasma membrane recycling pathway. J Cell Biol. 1997;139(1):49–61. doi: 10.1083/jcb.139.1.49. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 18.Knödler A, et al. Coordination of Rab8 and Rab11 in primary ciliogenesis. Proc Natl Acad Sci USA. 2010;107(14):6346–6351. doi: 10.1073/pnas.1002401107. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 19.Kim J, et al. Functional genomic screen for modulators of ciliogenesis and cilium length. Nature. 2010;464(7291):1048–1051. doi: 10.1038/nature08895. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 20.Boehlke C, et al. Differential role of Rab proteins in ciliary trafficking: Rab23 regulates smoothened levels. J Cell Sci. 2010;123(Pt 9):1460–1467. doi: 10.1242/jcs.058883. [DOI] [PubMed] [Google Scholar]
- 21.Schuh M. An actin-dependent mechanism for long-range vesicle transport. Nat Cell Biol. 2011;13(12):1431–1436. doi: 10.1038/ncb2353. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 22.Puthenveedu MA, et al. Sequence-dependent sorting of recycling proteins by actin-stabilized endosomal microdomains. Cell. 2010;143(5):761–773. doi: 10.1016/j.cell.2010.10.003. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 23.Milenkovic L, Scott MP, Rohatgi R. Lateral transport of Smoothened from the plasma membrane to the membrane of the cilium. J Cell Biol. 2009;187(3):365–374. doi: 10.1083/jcb.200907126. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 24.Wang Y, Zhou Z, Walsh CT, McMahon AP. Selective translocation of intracellular Smoothened to the primary cilium in response to Hedgehog pathway modulation. Proc Natl Acad Sci USA. 2009;106(8):2623–2628. doi: 10.1073/pnas.0812110106. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 25.Larkins CE, Aviles GD, East MP, Kahn RA, Caspary T. Arl13b regulates ciliogenesis and the dynamic localization of Shh signaling proteins. Mol Biol Cell. 2011;22(23):4694–4703. doi: 10.1091/mbc.E10-12-0994. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 26.Ohashi E, et al. Receptor sorting within endosomal trafficking pathway is facilitated by dynamic actin filaments. PLoS ONE. 2011;6(5):e19942. doi: 10.1371/journal.pone.0019942. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 27.Peters PJ, et al. Overexpression of wild-type and mutant ARF1 and ARF6: Distinct perturbations of nonoverlapping membrane compartments. J Cell Biol. 1995;128(6):1003–1017. doi: 10.1083/jcb.128.6.1003. [DOI] [PMC free article] [PubMed] [Google Scholar]
Associated Data
This section collects any data citations, data availability statements, or supplementary materials included in this article.