Abstract
We compare the whole-genome sequences of two multidrug-resistant clinical Acinetobacter baumannii isolates recovered in the same patient before (ABIsac_ColiS susceptible to colistin and rifampin only) and after (ABIsac_ColiR resistant to colistin and rifampin) treatment with colistin and rifampin. We decipher all the molecular mechanisms of antibiotic resistance, and we found mutations in the rpoB gene and in the PmrAB two-component system explaining resistance to rifampin and colistin in ABIsac_ColiR, respectively.
TEXT
Acinetobacter baumannii is an emerging multidrug-resistant (MDR) pathogen that is responsible for community- and hospital-acquired infections that are difficult to control and to treat (1, 2). This bacterium is intrinsically highly resistant to several antimicrobial agents, but increasing resistance to other antibiotics has been reported during the last decade, especially resistance to carbapenems, and the antibiotic resistance of A. baumannii is now recognized as a significant health problem because of the limited options for antibiotic therapy (1, 2). In these MDR strains, colistin is often the last resort for treatment, but colistin-resistant clinical isolates have been reported recently leading to pan-drug-resistant bacteria (1, 3, 4). We have recently reported such clinical pan-drug-resistant bacteria in a French patient with a bloodstream infection occurring after colistin therapy (4). An imipenem-resistant but colistin- and rifampin-susceptible A. baumannii isolate was recovered initially from a bronchoalveolar lavage (BAL) specimen from this French patient who suffered from pneumonia (ABIsac_ColiS). After 4 weeks of treatment with colistin and rifampin, a colistin- and rifampin-resistant isolate was recovered from a tracheal aspirate (ABIsac_ColiR). Here we report the whole-genome sequence comparison of these two clinical isolates to decipher whether the two MDR isolates actually were derived from a single clone of a colistin- and rifampin-resistant isolate being selected by antibiotic treatment.
High-throughput sequencing technologies are now widespread and could be used in a real-time manner to decipher the molecular support of any outbreak and/or MDR bacteria as recently exemplified with whole-genome sequencing as a rapid and powerful tool to elucidate the origin of the huge outbreak of Escherichia coli responsible for hemolytic-uremic syndrome in Germany (5) or the Haitian cholera outbreak (6). Genomic sequences of ABIsac_ColiS and ABIsac_ColiR were sequenced using both paired-end pyrosequencing strategy on the 454-Titanium instrument and with an additive shotgun for ABIsac_ColiR (454 Life Sciences, Branford, CT) (7) and SOLiD version 4 paired-end sequencing technology (Applied Biosystems, Foster City, CA) (8). For genome annotation, all contigs from these two strains were submitted to an online bioserver, the RAST server (RAST stands for Rapid Annotation using Subsystems Technology) (http://www.theseed.org) (9) to predict protein-encoding genes, rRNA and tRNA sequences, and assign function to these genes. Predicted open reading frames (ORFs) by RAST server were confirmed by BLASTP (E value 10E−8; identity ≥ 30%; coverage ≥ 50%) against no redundant protein (nr) and clusters of orthologous groups of proteins (COG) databases of the National Center for Biotechnology Information (NCBI) (http://www.ncbi.nlm.nih.gov). tRNA and rRNA genes were also verified on tRNAscan-SE Search Server (http://lowelab.ucsc.edu/tRNAscan-SE) and RFAM (http://rfam.sanger.ac.uk), respectively. Genome comparison was performed by “in silico” DNA-DNA hybridization using BLASTN analysis on a local bioserver to determine the full-length alignment between two genome sequences and the coverage percentage using the cutoff stringency of an E value at 1.00e−5. Genome alignment of both A. baumannii ABIsac_ColiS and ABIsac_ColiR strains was performed using Mauve alignment software (10). All antimicrobial resistance genes and mutated genes involved in antibiotic resistance were retrieved from this functional annotation.
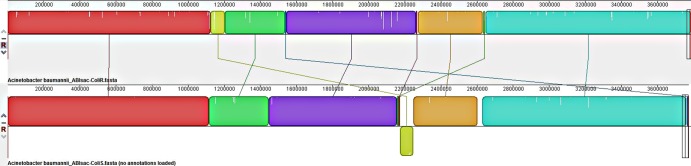
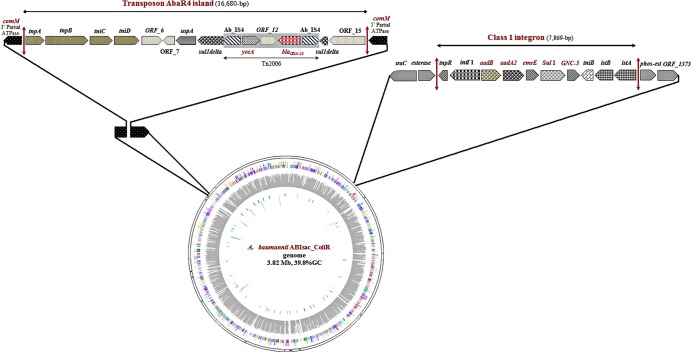
The assembly of the paired-end and shotgun sequences from A. baumannii ABIsac_ColiS gave a chromosome size of 3,771,873 bp with 38.77% GC content assembled into 275 contigs with the length of the contigs ranging from 902 bp to 98,458 bp and two plasmids of 68,612 bp (pABIsac_A) and 9,893 bp (pABIsac_B). With paired-end and shotgun sequences from A. baumannii ABIsac_ColiR, the assembly process gave a chromosome size of 3,785,453 bp with 38.84% GC content assembled into 108 contigs with the length of the contigs ranging from 1,077 bp to 192,975 bp and two plasmids of 68,347 bp (pABIsac_C) and 9.879 bp (pABIsac_D) (Table 1). The two plasmids have 96% sequence identity (small plasmids) and 99% sequence identity (large plasmids) to plasmids p2ABTCDC0715 and p1ABTCDC0715 previously reported in A. baumannii in Taiwan (11). “In silico” DNA-DNA hybridization and Mauve alignment demonstrate that these two strains, ABIsac_ColiS and ABIsac_ColiR, were likely the same clone (Fig. 1 and Table 1) as previously suggested using pulsed-field gel electrophoresis (PFGE) and multilocus sequence typing (MLST) analysis (12). Differences between the two strains consist mainly of the loss of a prophage in A. baumannii ABIsac_ColiR compared to ABIsac_ColiS. The loss of the prophage in ABIsac_ColiR may explain the impaired virulence of this strain (4) as recently demonstrated for Pseudomonas aeruginosa in the context of the Liverpool epidemic strain in cystic fibrosis patients (13). Table 2 lists the antibiotic resistance-encoding genes found in the two A. baumannii genomes. Resistance to sulfonamides (sul1 gene) and aminoglycosides (aadB and aadA2 genes) were located on the chromosome within a 7.8-kb class 1 integron (Fig. 2). Interestingly, the chromosomal blaOXA23-like gene was located in transposon Tn2006 in a 16.7-kb genomic island (Fig. 2) that completely replaced the 86-kb genomic region previously reported in A. baumannii strain AYE (14) within the comM gene. Finally, resistance to colistin, rifampin, and fluoroquinolones in ABIsac_ColiR were mediated by point mutation on target genes (Table 2). Rifampin resistance was likely due to a D525Y mutation in the rifampin resistance-determining region (RRDR) of the rpoB gene (Table 2). The same mutation responsible for rifampin resistance has recently been found in an A. baumannii strain isolated in Italy (15). This mutation has also been reported in Mycobacterium tuberculosis rifampin-resistant isolates (16, 17). Finally, resistance to colistin in strain ABIsac_ColiR was likely due to mutations in the pmrA gene with E changed to D at position 8 [pmrA(E8D)] (Table 2). These two proteins constitute a two-component system (PmrAB) involved in the modification of lipid A, the major constituent of the lipopolysaccharide (LPS) membrane, and mutations in the PmrAB two-component system have been reported recently in in vitro-selected A. baumannii strains (18). Mutations or disruption of the A. baumannii lipid A biosynthesis genes lpxA and lpxC by insertion sequence ISAba11 resulting in complete loss of lipopolysaccharide production has also been shown to be responsible for colistin resistance in vitro (19, 20) but was not found in our clinical isolate. To the best of our knowledge, the mutation in the PmrAB (18) two-component system is novel and reported for the first time in a clinical isolate that was likely selected because of colistin therapy.
Table 1.
Genome features of A. baumannii ABIsac_ColiS and ABIsac_ColiR strains compared to other A. baumannii strains
| A. baumannii strain | Bacterial chromosome or plasmid | EMBL or GenBank accession no. | Size (bp) | % GC content | No. of CDSa | No. of tRNAs | 5S-23S-16S operons | Full alignment length (bp) with cutoff E value of 1.00e−5 (% Cov)b |
|---|---|---|---|---|---|---|---|---|
| Our strains | ||||||||
| ABIsac_ColiS | Chromosome | CAKA01000001 to CAKA01000275 | 3,771,873 | 38.77 | 3,581 | 63 | 4 | 3,785,394 (99.99) |
| pABIsac_A | 68,612 | 33.19 | 97 | 0 | 0 | |||
| pABIsac_B | 9,893 | 36.91 | 12 | 0 | 0 | |||
| ABIsac_ColiR | Chromosome | CAKB01000001 to CAKB01000108 | 3,785,453 | 38.84 | 3,624 | 65 | 4 | |
| pABIsac_C | 68,347 | 33.18 | 99 | 0 | 0 | |||
| pABIsac_D | 9,879 | 36.92 | 13 | 0 | 0 | |||
| Other strains | ||||||||
| TCDC-AB0715 | Chromosome | CP002522 | 4,138,388 | 39 | 3,851 | 42 | 4 | 3,710,086 (98.01) |
| AYE | Chromosome | CU459141 | 3,936,291 | 39.4 | 3,607 | 72 | 6 | 3,470,222 (91.67) |
| ATCC 17978 | Chromosome | CP000521 | 3,976,747 | 38.9 | 3,351 | 69 | 5 | 3,429,245 (90.59) |
| SDF | Chromosome | CU468230 | 3,421,954 | 39.2 | 2,913 | 72 | 5 | 2,665,462 (70.41) |
CDS, coding sequences.
In silico DNA-DNA hybridization of A. baumannii ABIsac_ColiR genome with respect to other A. baumannii genomes. % Cov, percent coverage.
Fig 1.
Mauve alignment of A. baumannii ABIsac_ColiS and ABIsac_ColiR genomes.
Table 2.
Antibiotic resistance genes in A. baumannii ABIsac_ColiR genome
| Antibiotic class | Gene | Size (aa)a | Functionb | Organism with the best BLAST hit in GenBank | % aa identity | E value |
|---|---|---|---|---|---|---|
| Beta-lactams | ampC | 432 | Class C beta-lactamase (transpeptidase superfamily) | A. baumannii ACICU | 100 | 0 |
| 350 | Predicted Zn-dependent hydrolase of the beta-lactamase fold | A. baumannii ACICU | 100 | 0 | ||
| 309 | Metallo-beta-lactamase domain protein | A. baumannii 6014059 | 100 | 1e−177 | ||
| blaOXA-23 | 273 | Class D carbapenemase OXA-23 | A. baumannii TCDC-AB0715 | 100 | 1e−159 | |
| 414 | Beta-lactamase class A | A. baumannii ACICU | 100 | 0 | ||
| blaOXA-82(blaOXA-51-like) | 274 | Class D carbapenemase OXA-82 | A. baumannii ABNIH3 | 100 | 1e−159 | |
| 288 | Metallo-beta-lactamase domain protein | A. baumannii ACICU | 100.00 | 1e−171 | ||
| ampC | 384 | Beta-lactamase/d-alanine carboxypeptidase (transpeptidase superfamily) | A. baumannii MDR-ZJ06 | 99.00 | 0 | |
| 810 | Predicted hydrolase of the metallo-beta-lactamase superfamily (class C beta-lactamase) | A. baumannii TCDC-AB0715 | 99.00 | 0 | ||
| 227 | Putative metallo-beta-lactamase | A. baumannii AB056 | 99 | 1e−168 | ||
| Aminoglycosides | adeT | 335 | RND-type efflux pump involved in aminoglycoside resistance/substrate-binding protein, aliphatic sulfonate family | A. baumannii SDF | 100 | 0 |
| adeT | 355 | RND-type efflux pump involved in aminoglycoside resistance/TRAP-type C-4-dicarboxylate transport system, periplasmic component | A. baumannii 1656-2 | 99 | 0 | |
| 334 | RND-type efflux pump involved in aminoglycoside resistance/transporter | A. baumannii AYE | 99 | 0 | ||
| aphA6 | 259 | Aminoglycoside 3′-phosphotransferase/kanamycin resistance protein | A. baumannii AB058 | 99.60 | 1e−143 | |
| aadB | 198 | Aminoglycoside-2″-adenylyltransferase/gentamicin resistance protein | Salmonella enterica serovar Typhimurium | 100 | 1e−111 | |
| aadA2 | 259 | Aminoglycoside adenylyltransferase/streptomycin adenylyltransferase | Yersinia pestis biovar Orientalis IP275 | 99.614 | 1e−147 | |
| 329 | Aminoglycoside phosphotransferase | A. baumannii ACICU | 100 | 0 | ||
| Macrolides | macB | 664 | Macrolide-specific efflux protein MacB/ABC transporter permease | A. baumannii ACICU | 100 | 0 |
| macA | 446 | Macrolide-specific efflux protein MacA/membrane fusion protein | A. baumannii ACICU | 100 | 0 | |
| Sulfonamide | sul1 | 279 | Dihydropteroate synthase | E. coli FVEC1412 | 100 | 0 |
| Bicyclomycin | 514 | Bicyclomycin resistance protein | A. baumannii AYE | 100 | 1e−139 | |
| Chloramphenicol | cmr | 409 | Major facilitator superfamily multidrug/chloramphenicol efflux transporter | A. baumannii AYE | 99.76 | 0 |
| catB2 | 210 | Chloramphenicol acetyltransferase | A. baumannii AYE | 100 | 1e−122 | |
| Colistin | pmrA mutated (E8D) | 224 | Transcriptional regulatory protein/polymyxin resistance protein | A. baumannii AYE | 99.55 | 1e−126 |
| Rifampin | rpoB mutated (D525Y) | 1362 | DNA-directed RNA polymerase subunit beta | A. baumannii AYE | 99.93 | 0 |
| Fluoroquinolones | gyrA mutated (S83L, G145D, S218G, L644P, T872A) | 905 | DNA gyrase, A subunit/type IIA topoisomerase | A. baumannii AYE | 99 | 0 |
| parC mutated (S84L, E208G, S467G, A661V) | 740 | DNA topoisomerase IV subunit A/ParC | A. baumannii AYE | 99 | 0 |
aa, amino acids.
RND, resistance-nodulation-cell division; TRAP transporter, tripartite ATP-independent periplasmic transporter.
Fig 2.
Circular representation of A. baumannii ABIsac_ColiR and ABIsac_ColiS chromosomes and antibiotic resistance determinants. The transposon AbaR4 island and class 1 integron were identical in the chromosomes of both strains, whereas pmrA, pmrB, and rpoB mutations were found only in the ABIsac_ColiR chromosome. The genes and the proteins they encode follow: comM, competence protein ComM disrupted/Mg2+ chelatase-like protein; tnpA, transposase protein A; tnpB, transposase protein B; tniC, transposition helper protein C; tniD, probable transposition protein; ORF_6, hypothetical protein; ORF_7, hypothetical protein; uspA, universal stress protein; suldelta, sul1delta fusion protein/sulfate permease interrupted by Tn2006; Ab_IS4, IS4 family transposase ORF 1; yeeA, DNA methylase; ORF_12, hypothetical protein; blaOXA-23, OXA-23 carbapenemase; Ab_IS4, IS4 family transposase ORF 1; suldelta, sul1delta fusion protein/sulfate permease, interrupted by Tn2006; ORF_15, hypothetical protein; comM, competence protein ComM disrupted/Mg2+ chelatase-like protein; araC, transcriptional regulator of the AraC family; tnpR, transposon Tn21 resolvase; intI1, IntI1 integrase; aadB, aminoglycoside-2′-adenylyltransferase; aadA2, aminoglycoside adenyltransferase A2; emrE, ethidium bromide-methyl viologen resistance protein EmrE; sul1, dihydropteroate synthase; GCN-5, GCN5-like N-acetyltransferase; tniB, nucleoside triphosphate (NTP)-binding protein; istB, transposon NTP-binding protein; istA, transposase IstA protein; pcaR, regulon regulatory protein; ORF-1373, 4-hydroxybenzoate 3-monooxygenase.
In conclusion, the present work demonstrated that real-time whole-genome sequence comparison is a powerful tool to decipher all antibiotic resistance determinants in clinical microbiology when outbreak and/or novel MDR bacteria are isolated from clinical specimens.
Nucleotide sequence accession numbers.
All contig and plasmid sequences of these two MDR isolates of A. baumannii have been submitted to EMBL database under accession numbers CAKA01000001 to CAKA01000275 for A. baumannii ABIsac_ColiS and accession numbers CAKB01000001 to CAKB01000108 for A. baumannii ABIsac_ColiR.
ACKNOWLEDGMENTS
We thank Linda Hadjadj, Ti Tien Nguyen, and Romain Rivet for technical assistance. We thank American Journal Experts for correcting the English.
This work was funded by the French Centre National de la Recherche Scientifique (CNRS).
We declare that we have no conflicts of interest.
Footnotes
Published ahead of print 15 October 2012
REFERENCES
- 1. Kempf M, Rolain JM. 2012. Emergence of resistance to carbapenems in Acinetobacter baumannii in Europe: clinical impact and therapeutic options. Int. J. Antimicrob. Agents 39:105–114 [DOI] [PubMed] [Google Scholar]
- 2. Peleg AY, Seifert H, Paterson DL. 2008. Acinetobacter baumannii: emergence of a successful pathogen. Clin. Microbiol. Rev. 21:538–582 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 3. Lopez-Rojas R, Dominguez-Herrera J, McConnell MJ, Docobo-Perez F, Smani Y, Fernandez-Reyes M, Rivas L, Pachon J. 2011. Impaired virulence and in vivo fitness of colistin-resistant Acinetobacter baumannii. J. Infect. Dis. 203:545–548 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 4. Rolain JM, Roch A, Castanier M, Papazian L, Raoult D. 2011. Acinetobacter baumannii resistant to colistin with impaired virulence: a case report from France. J. Infect. Dis. 204:1146–1147 [DOI] [PubMed] [Google Scholar]
- 5. Rasko DA, Webster DR, Sahl JW, Bashir A, Boisen N, Scheutz F, Paxinos EE, Sebra R, Chin CS, Iliopoulos D, Klammer A, Peluso P, Lee L, Kislyuk AO, Bullard J, Kasarskis A, Wang S, Eid J, Rank D, Redman JC, Steyert SR, Frimodt-Moller J, Struve C, Petersen AM, Krogfelt KA, Nataro JP, Schadt EE, Waldor MK. 2011. Origins of the E. coli strain causing an outbreak of hemolytic-uremic syndrome in Germany. N. Engl. J. Med. 365:709–717 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 6. Chin CS, Sorenson J, Harris JB, Robins WP, Charles RC, Jean-Charles RR, Bullard J, Webster DR, Kasarskis A, Peluso P, Paxinos EE, Yamaichi Y, Calderwood SB, Mekalanos JJ, Schadt EE, Waldor MK. 2011. The origin of the Haitian cholera outbreak strain. N. Engl. J. Med. 364:33–42 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 7. Margulies M, Egholm M, Altman WE, Attiya S, Bader JS, Bemben LA, Berka J, Braverman MS, Chen YJ, Chen Z, Dewell SB, Du L, Fierro JM, Gomes XV, Godwin BC, He W, Helgesen S, Ho CH, Irzyk GP, Jando SC, Alenquer ML, Jarvie TP, Jirage KB, Kim JB, Knight JR, Lanza JR, Leamon JH, Lefkowitz SM, Lei M, Li J, Lohman KL, Lu H, Makhijani VB, McDade KE, McKenna MP, Myers EW, Nickerson E, Nobile JR, Plant R, Puc BP, Ronan MT, Roth GT, Sarkis GJ, Simons JF, Simpson JW, Srinivasan M, Tartaro KR, Tomasz A, Vogt KA, Volkmer GA, Wang SH, Wang Y, Weiner MP, Yu P, Begley RF, Rothberg JM. 2005. Genome sequencing in microfabricated high-density picolitre reactors. Nature 437:376–380 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 8. Shendure J, Porreca GJ, Reppas NB, Lin X, McCutcheon JP, Rosenbaum AM, Wang MD, Zhang K, Mitra RD, Church GM. 2005. Accurate multiplex polony sequencing of an evolved bacterial genome. Science 309:1728–1732 [DOI] [PubMed] [Google Scholar]
- 9. Aziz RK, Bartels D, Best AA, DeJongh M, Disz T, Edwards RA, Formsma K, Gerdes S, Glass EM, Kubal M, Meyer F, Olsen GJ, Olson R, Osterman AL, Overbeek RA, McNeil LK, Paarmann D, Paczian T, Parrello B, Pusch GD, Reich C, Stevens R, Vassieva O, Vonstein V, Wilke A, Zagnitko O. 2008. The RAST Server: rapid annotations using subsystems technology. BMC Genomics 9:75 doi:10.1186/1471-2164-9-75 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 10. Darling AC, Mau B, Blattner FR, Perna NT. 2004. Mauve: multiple alignment of conserved genomic sequence with rearrangements. Genome Res. 14:1394–1403 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 11. Chen CC, Lin YC, Sheng WH, Chen YC, Chang SC, Hsia KC, Liao MH, Li SY. 2011. Genome sequence of a dominant, multidrug-resistant Acinetobacter baumannii strain, TCDC-AB0715. J. Bacteriol. 193:2361–2362 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 12. Kempf M, Rolain JM, Azza S, Diene S, Joly-Guillou ML, Dubourg G, Colson P, Papazian L, Richet H, Fournier PE, Ribeiro A, Raoult D. 28 June 2012. Investigation of Acinetobacter baumannii resistance to carbapenems in Marseille hospitals, south of France: a transition from an epidemic to an endemic situation. APMIS [Epub ahead of print.] doi:10.1111/j.1600-0463.2012.02935.x [DOI] [PubMed] [Google Scholar]
- 13. Winstanley C, Langille MG, Fothergill JL, Kukavica-Ibrulj I, Paradis-Bleau C, Sanschagrin F, Thomson NR, Winsor GL, Quail MA, Lennard N, Bignell A, Clarke L, Seeger K, Saunders D, Harris D, Parkhill J, Hancock RE, Brinkman FS, Levesque RC. 2009. Newly introduced genomic prophage islands are critical determinants of in vivo competitiveness in the Liverpool epidemic strain of Pseudomonas aeruginosa. Genome Res. 19:12–23 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 14. Fournier PE, Vallenet D, Barbe V, Audic S, Ogata H, Poirel L, Richet H, Robert C, Mangenot S, Abergel C, Nordmann P, Weissenbach J, Raoult D, Claverie JM. 2006. Comparative genomics of multidrug resistance in Acinetobacter baumannii. PLoS Genet. 2:e7 doi:10.1371/journal.pgen.0020007 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 15. Giannouli M, Di Popolo A, Durante-Mangoni E, Bernardo M, Cuccurullo S, Amato G, Tripodi MF, Triassi M, Utili R, Zarrilli R. 2012. Molecular epidemiology and mechanisms of rifampicin resistance in Acinetobacter baumannii isolates from Italy. Int. J. Antimicrob. Agents 39:58–63 [DOI] [PubMed] [Google Scholar]
- 16. Cavusoglu C, Hilmioglu S, Guneri S, Bilgic A. 2002. Characterization of rpoB mutations in rifampin-resistant clinical isolates of Mycobacterium tuberculosis from Turkey by DNA sequencing and line probe assay. J. Clin. Microbiol. 40:4435–4438 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 17. Herrera L, Jimenez S, Valverde A, Garcia-Aranda MA, Saez-Nieto JA. 2003. Molecular analysis of rifampicin-resistant Mycobacterium tuberculosis isolated in Spain (1996–2001). Description of new mutations in the rpoB gene and review of the literature. Int. J. Antimicrob. Agents 21:403–408 [DOI] [PubMed] [Google Scholar]
- 18. Adams MD, Nickel GC, Bajaksouzian S, Lavender H, Murthy AR, Jacobs MR, Bonomo RA. 2009. Resistance to colistin in Acinetobacter baumannii associated with mutations in the PmrAB two-component system. Antimicrob. Agents Chemother. 53:3628–3634 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 19. Moffatt JH, Harper M, Adler B, Nation RL, Li J, Boyce JD. 2011. Insertion sequence ISAba11 is involved in colistin resistance and loss of lipopolysaccharide in Acinetobacter baumannii. Antimicrob. Agents Chemother. 55:3022–3024 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 20. Moffatt JH, Harper M, Harrison P, Hale JD, Vinogradov E, Seemann T, Henry R, Crane B, St. Michael F, Cox AD, Adler B, Nation RL, Li J, Boyce JD. 2010. Colistin resistance in Acinetobacter baumannii is mediated by complete loss of lipopolysaccharide production. Antimicrob. Agents Chemother. 54:4971–4977 [DOI] [PMC free article] [PubMed] [Google Scholar]