Abstract
Burkholderia cenocepacia H111, a strain isolated from a cystic fibrosis patient, has been shown to effectively kill the nematode Caenorhabditis elegans. We used the C. elegans model of infection to screen a mini-Tn5 mutant library of B. cenocepacia H111 for attenuated virulence. Of the approximately 5,500 B. cenocepacia H111 random mini-Tn5 insertion mutants that were screened, 22 showed attenuated virulence in C. elegans. Except for the quorum-sensing regulator cepR, none of the mutated genes coded for the biosynthesis of classical virulence factors such as extracellular proteases or siderophores. Instead, the mutants contained insertions in metabolic and regulatory genes. Mutants attenuated in virulence in the C. elegans infection model were also tested in the Drosophila melanogaster pricking model, and those also attenuated in this model were further tested in Galleria mellonella. Six of the 22 mutants were attenuated in D. melanogaster, and five of these were less pathogenic in the G. mellonella model. We show that genes encoding enzymes of the purine, pyrimidine, and shikimate biosynthesis pathways are critical for virulence in multiple host models of infection.
INTRODUCTION
The Burkholderia cepacia complex (Bcc) consists of 17 closely related, but phenotypically diverse, bacterial species. Strains of the Bcc have been isolated from soil, the rhizospheres of plants, groundwater, industrial settings, hospital environments, and infected humans and animals. Some strains could be valuable for biotechnology, as they can be used for bioremediation, as biological pest control agents, and for plant growth promotion. Conversely, some strains from the Bcc can act as problematic opportunistic pathogens in patients suffering from cystic fibrosis (CF) and also in immunocompromised individuals (1–4). B. cenocepacia and Burkholderia multivorans are currently the Bcc species most frequently isolated from clinical samples (1, 4). In CF patients these species can cause serious infections, leading to rapid decline in lung function and an often fatal pneumonia (known as “cepacia syndrome”) (5, 6).
Over the past decade, substantial progress has been made in identifying and characterizing the virulence determinants and infection mechanisms of Bcc strains (7). These studies used different Bcc strains and various infection models. Since strain-to-strain variability and the infection model used have a tremendous effect on the outcome of pathogenicity assays, knowledge of the importance of individual virulence factors in different infection hosts is sparse (7, 8).
In this study, we screened a Burkholderia cenocepacia H111 random insertion mutant bank for attenuated virulence in Caenorhabditis elegans. To exclude experimental and host-specific factors, other nonmammalian animal models were used to test those mutants which showed attenuation in pathogenicity to C. elegans. The first was the fruit fly, Drosophila melanogaster, into which bacteria were injected by pricking with a needle (9, 10). Those mutants that were also attenuated in D. melanogaster were further tested using the greater wax moth larva Galleria mellonella, which allows injection of a precise dosage of bacteria (8, 11). We checked mutants for the production of AidA, which is known to play a role in pathogenicity to C. elegans (8, 12), as well as for other previously identified virulence factors, including production of acyl-homoserine lactone (AHL) signal molecules, siderophores, proteases, and polysaccharides.
MATERIALS AND METHODS
Organisms and culture conditions.
Bacterial strains and plasmids used in this study are listed in Table 1. Unless otherwise stated, bacteria were grown at 37°C in modified Luria-Bertani (LB) broth (13) containing 5 g NaCl liter−1. Solid media contained agar to a final concentration of 1.5%. Antibiotics were added as required at final concentrations of 20 μg ml−1 gentamicin, 50 μg ml−1 kanamycin, 10 μg ml−1 tetracycline, 50 μg ml−1 spectinomycin, 25 μg ml−1 streptomycin, and 10 μg ml−1 chloramphenicol. Growth of liquid cultures was monitored by measurement of optical density at 600 nm (OD600) using an Ultraspec 3100 pro (Amersham Bioscience). Supplements were added to media to the following concentrations: l-histidine, l-cysteine, dl-tryptophan, and adenosine, 500 μM; guanine, adenine, adenosine, and inosine, 20 μg liter−1. Killing assays were performed using Caenorhabditis elegans strain Bristol N2, which was obtained from the Caenorhabditis Genetics Center (University of Minnesota, St. Paul, MN). Nematodes were maintained on NG agar (14) at 20°C with Escherichia coli strain OP50 as a food source (15). For the D. melanogaster pricking assay, the D. melanogaster Canton-S wild-type strain was used. The flies were kept at room temperature and fed with standard sucrose cornmeal (http://flystocks.bio.indiana.edu/Fly_Work/media-recipes/harvardfood.htm). G. mellonella was bought from Fischerei Brumann, Zurich, Switzerland, and kept in an incubator at 14°C. The larvae were used within 3 weeks of purchase.
Table 1.
Bacterial strains and plasmids used in this study
| Strain or plasmid | Characteristics | Reference or source |
|---|---|---|
| E. coli | ||
| OP 50 | Food source strain for C. elegans, uracil auxotrophic | 15 |
| XL1-Blue | recA1 endA1 gyrA96 thi-1 hsdR17 supE44 relA1 lac [F′ proAB lacIq ZΔM15 Tn10 (Tetr)] | Stratagene |
| MT102 | araD139 (ara-leu)7679 Δlac thi hsdR | Laboratory collection |
| Pseudomonas putida F117(pAS-C8) | Bioluminescent AHL biosensor, F117 with pBBR1MCS-5 carrying PcepI::gfp(ASV) Plac::cepR; Gmr | 77 |
| Burkholderia cenocepacia | ||
| H111 | CF isolate, Germany | 78 |
| H111-R | Kmr; cepR::Km mutant of H111 | 79 |
| H111 purD | Kmr; purD::Km mutant of H111 | This study |
| H111 purF | Kmr; purF::Km mutant of H111 | This study |
| H111 purL | Kmr; purL::Km mutant of H111 | This study |
| H111 purA | Kmr; purA::Km mutant of H111 | This study |
| H111 aroK | Kmr; aroK::Km mutant of H111 | This study |
| H111 ilvC | Kmr; ilvC::Km mutant of H111 | This study |
| H111 cysI | Kmr; cysI::Km mutant of H111 | This study |
| H111 cysB | Kmr; cysB::Km mutant of H111 | This study |
| H111 ahcY | Kmr; ahcY::Km mutant of H111 | This study |
| H111 hisG | Kmr; hisG::Km mutant of H111 | This study |
| H111 hisH | Kmr; hisH::Km mutant of H111 | This study |
| H111 trpA | Kmr; trpA::Km mutant of H111 | This study |
| H111 trpB | Kmr; trpB::Km mutant of H111 | This study |
| H111 trpF | Kmr; trpF::Km mutant of H111 | This study |
| H111 gatA | Kmr; gatA::Km mutant of H111 | This study |
| H111 lon | Kmr; lon::Km mutant of H111 | This study |
| H111 rsaM | Kmr; rsaM::Km mutant of H111 | This study |
| H111 pyrD | Kmr; pyrD::Km mutant of H111 | This study |
| H111 rsuA | Kmr; rsuA::Km mutant of H111 | 43 |
| G. mellonella | Fischerei Brumann, Zürich | |
| C. elegans Bristol N2 | Caenorhabditis Genetics Center, University of Minnesota, Minneapolis | |
| D. melanogaster Canton-S | ||
| Plasmids | ||
| pRK600 | Cmr oriColE1 RK2-Mob+ RK2-Tra+, helper plasmid in triparental conjugations | 21 |
| pSB403 | Tcr, luxRI′::luxCDABE transcriptional fusion, bioluminescent AHL sensor plasmid | 80 |
| pGEM-3Zf(+) | Apr, multicopy cloning vector | Promega |
| pUTmini-Tn5 Km2-luxCDABE | Kmr, delivery vector for mini-Tn5 Km2-luxCDABE | 81 |
| pCR2.1-TOPO | Apr, Kmr, cloning vector | Invitrogen |
| pRG930 | 16.1-kb cosmid, derived from pGV910, Smr/Spr | 20 |
| pRG930-purD | pRG930 containing purD gene, Smr/Spr | This study |
| pRG930-purF | pRG930 containing purF gene, Smr/Spr | This study |
| pRG930-purL | pRG930 containing purL gene, Smr/Spr | This study |
| pRG930-pyrD | pRG930 containing pyrD gene, Smr/Spr | This study |
| pRG930-aroK | pRG930 containing aroK gene, Smr/Spr | This study |
| pUC19 | Cloning vector, Apr, lacZ | Invitrogen, Carlsbad, CA |
Transposon insertion mutagenesis.
The hybrid transposon (Table 1) was randomly inserted into the genome of B. cenocepacia H111 by triparental mating as described previously (24). Transconjugants were selected on LB medium containing kanamycin and tetracycline. These random insertion mutants were picked and grown in 150 μl LB medium in polypropylene microtiter plates (Nunc). For storage, 75 μl of 50% (vol/vol) glycerol was added and the plates were frozen at −80°C.
DNA manipulations and sequence analysis of Tn5 mutants.
Cloning, restriction enzyme analysis, and transformation of E. coli were performed essentially as described previously (16). PCR was performed using TaKaRa rTaq DNA polymerase (TaKaRa Shuzo), as per the manufacturer's instructions. Plasmid DNA was isolated with the QIAprep Spin Miniprep kit (Qiagen), and chromosomal DNA from B. cenocepacia was purified with the DNeasy tissue kit (Qiagen). DNA fragments were purified from agarose gels using the QIAquick gel extraction kit (Qiagen).
To ensure that the attenuated mutants bore only one transposon, Southern blotting was employed. Genomic DNA was digested with SphI. The fragments were electrophoresed through a 1% agarose gel and transferred using a blot apparatus (Stratagene, Heidelberg, Germany) to a positively charged nylon membrane (Hybond-N+; Amersham, Cleveland, OH). The transferred DNA was UV fixed, and the blots were hybridized with a digoxigenin (DIG)-coupled probe. The probe was prepared and detected using DIG High-Prime DNA labeling and detection starter kit I (Roche, Mannheim, Germany) according the manufacturer's instructions. Probes for the detection of the transposons were constructed by PCR amplification of the Tn5 kanamycin cassette using Kan res-v and Kan res-r primers (17) (Table 2).
Table 2.
Primers used in this study
| Primer | Sequence (5′→3′) | Description |
|---|---|---|
| pRG930_seq_rv | ACG TTT CAG TTT GCT CAT GG | Sequencing primer, pRG930 |
| pRG930_seq_fw | GGT CCT GCA ACT TTA TCC G | Sequencing primer, pRG930 |
| Kan res-v | ACC GAG GCA GTT CCA TAG G | Mini-Tn5 Km2-luxCDABE |
| Kan res-r | ATG AGC CAT ATT CAA CGG G | Mini-Tn5 Km2-luxCDABE |
| M13v | GTA AAA CGA CGG CCA G | Sequencing primer/pCR2.1TOPO |
| M13r | CAG GAA ACA GCT ATG AC | Sequencing primer/pCR2.1TOPO |
| ARB2 | GGC CAC GCG TCG ACT AGT AC | Arbitrary PCR, round 2 |
| ARB6 | GGC CAC GCG TCG ACT AGT CAN NNN NNN NNN ACG CC | Arbitrary PCR, round 1 |
| luxCext2 | AGT CAT TCA ATA TTG GCA GG | Arbitrary PCR, round 1 |
| luxCint2 | GGA TTG CAC TAA ATC ATC CAC | Arbitrary PCR, round 2 |
| Seq_O | CAC TTG TGT ATA AGA GTC AG | Sequencing primer, o end of the mini-Tn5 Km2-luxCDABE transposon |
The insertion position of the transposon was determined by two different techniques, SphI cloning and arbitrary PCR. DNA sequences flanking transposon insertions were determined by arbitrary PCR as described previously (18), with some modification. Briefly, we performed two rounds of PCR amplification using a degenerate arbitrary primer to anneal to the chromosome and one specific to the mini-Tn5 transposon. Primers used in the first round were ARB6 and luxCext2 (Table 2). First-round reaction conditions were as follows: (i) 5 min at 95°C; (ii) 6 cycles of 30 s at 95°C, 30 s at 30°C, and 1 min at 72°C; (iii) 30 cycles of 30 s at 95°C, 30 s at 45°C, and 1 min at 72°C; and (iv) 5 min at 72°C. The second round of PCR amplification used 5 μl purified first-round PCR product as the template and primers ARB2 and luxCint2 (Table 2). Second-round reaction conditions were as follows: (i) 30 cycles of 30 s at 95°C, 30 s at 45°C, and 1 min at 72°C, and (ii) 5 min at 72°C. The PCR products were purified from an agarose gel and ligated into the vector pCR 2.1-TOPO.
Sequencing reactions were carried out to determine the transposon integration sites using the Seq_O primer (Table 2), which binds to the O end of the mini-Tn5 transposon. Integration sites were determined using the online BLAST search engine (http://www.ncbi.nlm.nih.gov./).
For the SphI cloning, genomic DNA was digested with SphI. This cleaved the i end of the transposon, including the kanamycin resistance gene. The digested DNA was cloned into pUC19 and transferred into E. coli DH5α. Bacteria containing a transposon fragment were selected on LB plates containing kanamycin. The sequence of the inserted DNA was found by sequencing with standard M13 primers, Kan res-v and Kan res-r (Table 2).
Complementation of the Tn5 mutants using a B. cenocepacia H111 cosmid library.
Chromosomal DNA was extracted from B. cenocepacia H111 using a standard protocol (19). The DNA was partially digested with EcoRI and ligated into cosmid pRG930 (20). Packaging and of the cosmid into E. coli HB101 cells and titer determination were carried out using the Gigapack III Gold packaging reaction kit from Invitrogen as described in the instruction manual but with the omission of the dilution step of the titer determination reaction. The whole cosmid library was pooled and introduced into each transposon mutant by triparental mating (21). Complemented mutants were selected on ABC minimal plates containing streptomycin and spectinomycin. Cosmids were then extracted using a miniprep kit (Qiagen) and the cosmid ends sequenced with the primers pRG930_seq_rv and pRG930_seq_fw (Table 2) to determine the extent of the genomic insert.
Pathogenicity screen with C. elegans.
Screening was carried out on NG agar plates as described previously (22, 23). H111 mutants were grown at 37°C overnight in LB broth in microtiter dishes (Nunc). Fifty microliters of the respective cultures was spread on NG agar plates in 24-well multiplates (Greiner, Germany). Plates containing bacteria were incubated overnight at 37°C. After a brief incubation at room temperature to allow cooling, plates were inoculated with 20 to 30 synchronized L4 worms of the C. elegans Bristol N2 wild-type strain (obtained from the Caenorhabditis Genetics Center, University of Minnesota, Minneapolis, MN) (24). The plates were then incubated at 20°C, and live worms were counted after 2 days. In the initial screen approximately 230 24-well plates were tested in a single replicate. All mutants identified in the initial screen were retested in five independent replicates.
G. mellonella killing assays.
The G. mellonella infection assay was performed as described previously (8). Modifications were made to the protocol as follows. To prevent contamination, the Tn5 mutants' growth medium was supplemented with kanamycin. For hemolymph extraction, larvae were first sterilized by brief immersion in absolute ethanol. Hemolymph was removed using a syringe with a 22-gauge needle. Approximately 100 μl was obtained per larva. Dilutions were plated for bacterial enumeration. Caterpillars in the final larval stage were purchased from Fischerei Brumann, Zurich, Switzerland.
D. melanogaster pricking assays.
D. melanogaster Canton-S wild-type flies were used for the pricking procedure. The flies were bred on standard cornmeal sucrose medium and kept at room temperature. The assay was performed as described previously (10). Modifications were made to this protocol as follows. Two- to 9-day-old male and female flies were used in this experiment. For anesthetization, ether was used instead of a CO2 pad. For each trial, duplicate sets of 15 flies were used and the experiment was performed in triplicate. The negative-control flies were inoculated with a 10 mM MgSO4 solution. If more than 5 flies died due to pricking injury, the experiment was repeated. The infected flies were incubated at 26°C.
AHL quantification.
Strains were grown to an OD600 of 3.0. Bacteria were collected by centrifugation at 6,500 rpm for 5 min and the supernatant filter sterilized. One hundred microliters of supernatant was mixed with 100 μl exponential-growth-phase Pseudomonas putida F117(pAS-C8-Gmr). This strain is highly specific for C8 homoserine lactone (HSL) detection. Commercial C8 HSLs were used as a standard. The cells were incubated in FluoroNunc Polysorp microtiter plates (Nunc Roskilde) for 6 h in the dark at 30°C. Fluorescence was measured with a Lamda Fluoro 320 Plus reader (Bio-Tek Instruments, Winooski, VT), and the relative fluorescence units (RFU) were determined.
Determination of siderophore production and extraction of pyochelin.
Siderophore activity of the H111 mutants was tested on CAS agar as described previously (25). CAS plates were inoculated with 5 μl culture at an OD600 of 1.0, allowed to dry, and incubated at 37°C for ∼48 h. Siderophore activity was determined by comparison of halo diameter (a larger halo indicates higher siderophore production) and color (25, 26).
For pyochelin extraction, mutants were grown overnight in 200 ml NG medium to reflect the growth conditions used for the initial identification of mutants attenuated in virulence to C. elegans. This ensured both the growth of the mutants and the relevance of the results to pathogenicity in the C. elegans model. Bacteria were collected by centrifugation, and the pH of the spent culture supernatants was adjusted to ∼2.0 by the addition of HCl. The siderophores were extracted twice by addition of 0.4 volume dichloromethane. The organic phase was removed by rotary evaporation, and the residue was resuspended in 200 to 250 μl methanol.
The extracts were analyzed by thin-layer chromatography on silica 60 plates (VWR) with chloroform-acetic acid-ethanol at 90:5:2.5 (vol/vol) as the solvent (27). Plates were dried after development and siderophores detected by UV light. Pyochelin was identified by the presence of two yellow-green fluorescent bands corresponding to the two pyochelin stereoisomers, pyochelins I and II (Rfs, 0.35 and 0.37, respectively), and salicylate was visualized as a blue fluorescent band (Rf, 0.74) (28, 29).
Determination of protease activity and EPS production.
Protease activity was determined on skimmed milk agar as described previously (30). Five microliters of overnight culture from each strain to be tested (diluted to an OD600 of 1) was dropped onto the plate and allowed to dry. The plate was incubated at 37°C for 48 h. Extracellular polysaccharides (EPS) production was determined on YEM agar supplemented with Congo red (31). EPS-positive strains showed a slimy, slightly red colony morphology after overnight incubation at 37°C, whereas EPS-negative strains turned red as a result of integration of the dye.
Production of AidA.
Detection of AidA was performed by Western blotting. Whole-cell proteins were separated on a 15% SDS-polyacrylamide gel and then transferred to a polyvinylidene difluoride (PVDF) membrane (Immobilon-P; Millipore, Eschborn, Germany). Anti-AidA antibodies (12) were used to probe the membrane. Detection reactions were performed with alkaline phosphatase-conjugated anti-rabbit immunoglobulin G (Sigma, Steinheim, Germany) according to the recommendations of the manufacturer (Roche, Mannheim, Germany).
Growth on minimal media.
The growth of the transposon mutants was tested using ABC or ABG minimal medium (32). For growth tests, 5 ml of ABC or ABG solution was inoculated with bacteria to an OD600 of 0.01 and incubated at 37°C with shaking. For the functional complementation (supplementation) of the purine mutants, 20 mg liter−1 adenine, inosine, or guanine was added to ABC minimal medium.
RESULTS
Identification of B. cenocepacia H111 mutants attenuated in C. elegans and their phenotypic characterization.
It has previously been shown that on NG medium the CF isolate B. cenocepacia H111 kills C. elegans N2 within 3 days. We employed this “slow-killing” assay to screen a collection of approximately 5,500 random mini-Tn5 insertion mutants of B. cenocepacia H111 for attenuated virulence. In total, 23 mutants that reproducibly showed reduced killing after 48 h compared with the wild type were isolated (Fig. 1; Table 3). Previous work has identified several factors that contribute to the pathogenicity of B. cenocepacia (7), and we therefore tested the mutants for the production of some of these factors, including siderophores, extracellular polysaccharides (EPS), proteases, AHL quorum-sensing (QS) signaling molecules, and AidA, a protein required for nematode pathogenicity (Table 3).
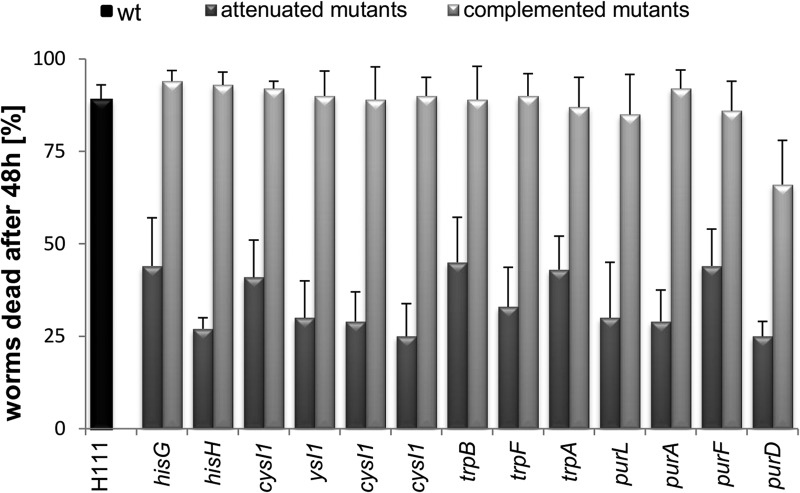
Fig 1.
Virulence of wild-type B. cenocepacia H111 and auxotrophic mutants in the C. elegans model. C. elegans were counted 48 h after spreading the worms on medium covered with a lawn of the respective bacterial strain. Each experiment consisted of three independent replicates. Black bar represents the wild type (wt) (H111), dark gray bars represent mutants grown on NG medium, and light gray bars represent mutants grown on nutritionally supplemented NG medium. Nutritional supplementation was as follows: hisG and hisH, histidine; cysI and cysB, cysteine, trpA, trpB, and trpF, dl-tryptophan; purD, purF, purL, and purA, adenosine.
Table 3.
B. cenocepacia H111 mutants attenuated in C. elegans
| Strain | Gene disrupted | H111 gene ID | Mean (SD) % dead C. elegans larvae after 48 ha | D. melanogaster survival time (h) | Mean (SD) % surviving G. mellonella after 48 h | AHL productiona | Protease activitya | EPS productiona | Siderophore productiona | Expression of AidAa | Growth on ABCa |
|---|---|---|---|---|---|---|---|---|---|---|---|
| HIII | None (wt) | 89 (4) | <72 | 10 (10) | + | + | + | + | + | + | |
| A4 | purD | 358071918 | 24 (4) | >336 | 72 (18) | < | < | − | − | + | − |
| D9 | purF | 358073772 | 44 (10) | >336 | 29 (8) | + | < | < | + | + | − |
| C2-1 | purL | 358073038 | 30 (8) | >336 | 50 (20) | < | + | − | − | + | − |
| F8 | purA | 358073158 | 29 (8) | <72 | 0 (0) | + | + | < | < | + | − |
| A11 | aroK | 358076029 | 25 (4) | <288 | 93 (12) | + | + | − | +c | + | − |
| H2 | ilvC | 358071882 | 21 (9) | >336 | 27 (25) | + | + | < | − | + | − |
| C5 | cysI | 358073512 | 29 (8) | <72 | NTd | + | + | < | +c | + | − |
| E10 | cysI | 358073512 | 41 (10) | <72 | NT | + | + | < | +c | + | − |
| F7 | cysI | 358073512 | 30 (16) | <72 | NT | + | + | < | +c | + | − |
| B1 | cysI | 358073512 | 25 (9) | <72 | NT | + | + | < | +c | + | − |
| G11 | cysB | 358073513 | 62 (8) | <72 | NT | + | + | + | +c | + | + |
| E11 | ahcY | 358071424 | 11 (9) | <72 | NT | + | + | − | + | + | − |
| E8 | hisG | 358075994 | 44 (12) | <72 | NT | + | + | < | + | + | − |
| G5-1 | hisH | 358075989 | 27 (3) | <72 | NT | + | + | < | < | + | < |
| D1 | trpA | 358073777 | 43 (9) | <72 | NT | + | + | − | + | − | − |
| G5-2 | trpB | 358073779 | 45 (12) | <72 | NT | + | + | + | + | < | − |
| B8 | trpF | 358073780 | 33 (10) | <72 | NT | < | + | − | + | + | − |
| C2-2 | gatA | 358076086 | 63 (15) | <72 | NT | + | < | + | + | + | + |
| E5 | rsuA | 358074812 | 29 (9) | <72 | NT | < | − | + | − | < | + |
| G3 | lon | 358073031 | 46 (10) | <72 | NT | < | − | + | + | + | + |
| E12-2 | rsaM | 358072205 | 17 (8) | <72 | NT | + | < | + | < | < | + |
| D6b | pyrD | 358074754 | 23 (7) | >336 | 87 (15) | < | − | − | − | + | − |
| G1 | cepR | 358072206 | 7 (5) | <72 | 0 (0) | − | − | < | + | + | + |
+, wt level; <, less than wt level; −, absence.
This strain showed a reduced growth rate in LB medium (see Fig. S1 in the supplemental material).
Halo on CAS agar of normal size but pink color absent.
NT, not tested.
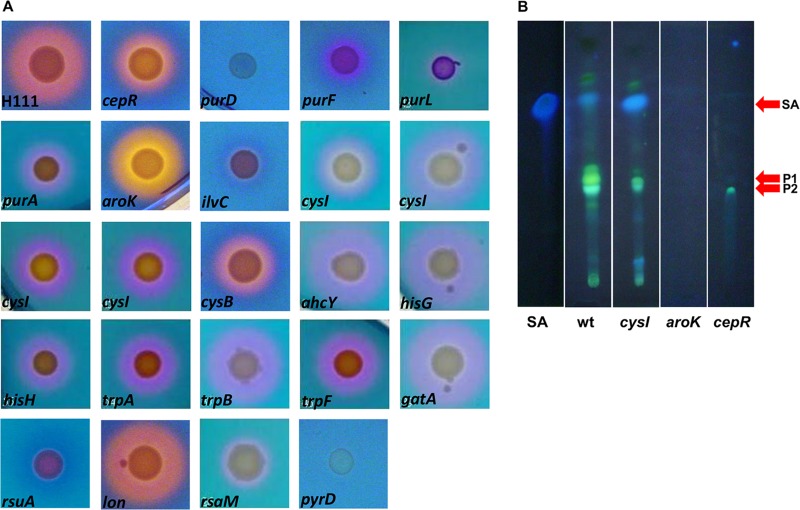
B. cenocepacia H111 produces the two siderophores ornibactin and pyochelin, which were previously shown to be important for virulence in mammals, C. elegans, and larvae of the greater wax moth G. mellonella but not alfalfa (8, 33, 34). Some of the mutants showed reduced siderophore production (Table 3). In addition, we observed that some of the mutants formed a yellow rather than a pink halo on CAS agar (Fig. 2A), which is indicative of a loss of pyochelin production (35). Extraction of culture supernatant and analysis by thin-layer chromatography confirmed that these mutants produced no or reduced amounts of pyochelin (Fig. 2B).
Fig 2.
Siderophore production of attenuated B. cenocepacia H111 Tn5 mutants. (A) Mutants were inoculated on CAS plates and incubated at 37°C for 48 h. The halo diameter corresponds to siderophore activity. A pink-tinted halo is indicative of pyochelin production. (B) Production of pyochelin as determined by thin-layer chromatography. Bands corresponding to pyochelins I and II (P1/P2) and salicylate (SA) are indicated. Extracts were as follows: SA, salicylate standard; wt, wild-type H111; cysI, H111 cysI; aroK, H111 aroK; cepR, H111 cepR.
The production of extracellular polysaccharides (EPS) is considered to enhance persistence of Bcc species in the lung, similar to alginate in Pseudomonas aeruginosa (36, 37). We tested the attenuated mutants for EPS production on YEM agar, which stimulates the production of the major Bcc polysaccharide cepacian (38, 39). Many of the attenuated mutants showed reduced EPS production (Table 3). However, a defined cepacian-deficient mutant of B. cenocepacia K56-2 was found to be as pathogenic as the wild type in C. elegans and in G. mellonella (data not shown), suggesting that the lack of EPS production is not responsible for the reduced virulence of our mutants. It should be noted that cepacian production has been shown to be important for pathogenicity in murine infection models (40, 41). Furthermore, our phenotypic characterization revealed that several of the mutants produced no or reduced amounts of AHL signal molecules, proteases, or AidA (Table 3).
Mutant D6 grew slower in LB medium, whereas the growth rates of the other mutants were indistinguishable from that of the wild type (see Fig. S1 in the supplemental material). In contrast to the wild type, however, many of the mutants showed poor or no growth on minimal medium supplemented with either glucose or citrate as a carbon source and thus were auxotrophic mutants.
Locations of transposon insertions within the attenuated mutants.
The locations of the transposon insertions were determined by sequencing the DNA regions flanking the transposon (Table 3). To our surprise, only one of the disrupted genes coded for a previously described virulence factor (the QS regulator CepR), while 19 of the mutations were in metabolic genes, three mutations were in regulatory genes, and three mutants were found to have lost an entire replicon (R12, R33, and R40). The latter three mutants have been described elsewhere (42).
Four of the genes identified in the transposon screen (cepR, lon, rsuA, and rsaM) code for regulatory proteins. The isolation of a cepR mutant is in full agreement with previous work that has demonstrated that the QS regulator CepR is essential for virulence in multiple infection hosts (8). One mutant was found to carry the transposon insertion in the rsuA gene. An rsuA mutant was previously identified in a screen for biofilm-defective mutants, and this gene (previously known as yciL) has been characterized as a higher-level quorum-sensing (QS) regulator (43). The transposon insertion site of another mutant that was affected in QS was mapped to the intergenic region between the divergent rsaM and cepR genes, such that expression of both genes is affected (data not shown). Finally, the lon gene codes for an ATP-dependent protease which belongs to the AAA+ (ATPases associated with a variety of cellular activities) superfamily of enzymes (44). This is a widespread family of enzymes (45), responsible for diverse functions, including protein unfolding, DNA replication, and recombination (44). Disruption of the lon gene has been shown to reduce the pathogenicity of Salmonella enterica (46), Pseudomonas syringae (47), Campylobacter jejuni (48), Agrobacterium tumefaciens (49), and Brucella abortus (50). This appears to be due to the role of Lon in the upregulation of type three secretion systems (44, 51, 52).
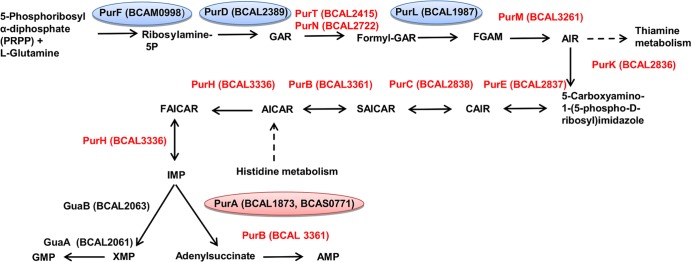
The 19 mutants defective in metabolic functions had mutations that fell within three major pathways: the purine biosynthetic pathway, the shikimate pathway, and the pyrimidine biosynthetic pathway. Four of the attenuated mutants identified during the C. elegans screen carried insertions within genes of the purine biosynthesis pathway (purA, purF, purL, and purD) (Fig. 3), suggesting that de novo purine biosynthesis plays an important role in the nematode pathogenicity of B. cenocepacia H111. The purF, purD, and purL genes are all positioned in the initial part of the purine biosynthetic pathway (Fig. 3), and upon inactivation of any of them, one would expect complete abrogation of purine production. The purA gene, however, is positioned later in the pathway and would not be necessary for the production of guanine- and xanthine-related purines. IMP is the first molecule containing the purine double-ring system to be formed in the purine pathway. This molecule can be used in the production of both adenine and guanine by the cell (53, 54) (Fig. 3). Growth of the purD, purF, and purL mutants could be successfully restored by the addition of inosine to ABC minimal medium (Fig. 4C, D, and E). Supplementation with adenine, but not with guanine, restored growth to these mutants in ABC minimal medium (Fig. 4C, D, and E). Adenine can be converted to guanine via the purine metabolic pathway, but the reciprocal conversion is not possible via this pathway. The addition of inosine, adenine, and guanine to ABC medium did not affect the growth of wild-type H111 (Fig. 4A).
Fig 3.
The B. cenocepacia purine biosynthetic pathway. Mutants with attenuated virulence isolated in this study are circled.
Fig 4.
Growth of purine mutants in ABC medium supplemented with various metabolites. Tubes: 1, unsupplemented medium; 2, supplementation with inosine; 3, supplementation with adenine; 4, supplementation with guanine; 5, supplementation with adenine and guanine. (A) B. cenocepacia HIII; (B) purA mutant; (C) purD mutant; (D) purF mutant; (E) purL mutant.
The pyrD and aroK mutants showed very similar phenotypic characteristics (Table 3). The pyrD gene codes for a class 2 dihydroorotate dehydrogenase, which is a key component in pyrimidine metabolism. The aroK gene encodes a component of the shikimate pathway, through which chorismate, an important precursor in the production of aromatic amino acids, is produced. Another mutant was disrupted in ilvC, a ketol-acid reductoisomerase, which plays a role in valine, leucine, and coenzyme A biosynthesis.
Of the remaining metabolic mutants, 10 had defects in amino acid biosynthesis. Four cysteine pathway mutants were all disrupted in the cysI gene (which codes for the beta subunit of a sulfide reductase), and one mutant bore an insertion in the cysB gene (a potential transcriptional regulator of the cys regulon). Two additional genes that play a role in the histidine metabolic pathway, hisG (encoding an ATP-phosphoribosyltransferase), and hisH (encoding a glutamine-amidotransferase), were found to be important for pathogenicity in the C. elegans model, as were the tryptophan biosynthetic pathway genes trpA, trpB, and trpF [encoding tryptophan-synthase alpha and beta chains and N-(5′-phosphoribosyl)anthranilate-isomerase, respectively].
The final two metabolic mutants bore insertions in genes not directly involved in amino acid biosynthesis; the ahcY (S-adenosylhomocysteine hydrolase) gene, the product of which acts as a coenzyme in cysteine and methionine metabolism, and the gatA (glutamyl-tRNA amidotransferase subunit A) gene, which plays a role in protein synthesis.
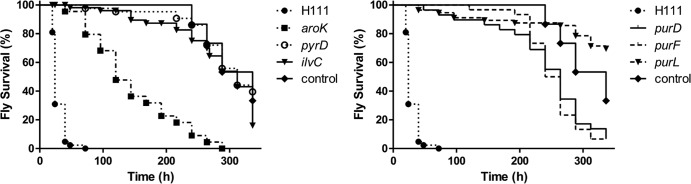
Five of the mutants attenuated in C. elegans are also less virulent in D. melanogaster and G. mellonella.
Previous work has shown that some virulence factors are host specific, while other factors are important for pathogenicity in multiple infection models (8). We tested the 23 mutants showing attenuation in the C. elegans pathogenicity assay using the D. melanogaster infection model to discern host-specific and general factors. The fruit fly D. melanogaster has been shown to be a useful nonmammalian infection host for the determination of the pathogenicities of different Bcc strains (9). Of the 23 mutants with reduced C. elegans virulence, six were also strongly attenuated in the D. melanogaster infection model (Fig. 5). These six mutants were further tested for virulence using larvae of the greater wax moth G. mellonella as a host in order to validate their function as general virulence factors (Fig. 6). Five of the six mutants were attenuated compared to the wild type in this model, namely, the purD, purF, and purL purine biosynthesis mutants, the pyrD pyrimidine biosynthesis mutant, and the aroK mutant, which is defective in aromatic amino acid biosynthesis. The ilvC mutant showed attenuation in C. elegans and D. melanogaster but not in G. mellonella. It is therefore tempting to speculate that in G. mellonella but not in the other infection hosts tested, sufficient amounts of certain metabolites are available to rescue the defects of the ilvC mutant. In contrast to the purD, purF, and purL mutants, the purA mutant exhibited wild-type pathogenicity in both the D. melanogaster and the G. mellonella infection models (Table 3). In silico analysis of the B. cenocepacia H111 genome revealed a homologous gene product (GeneID 358069869) with 51.5% identity to H111 PurA. It is possible that this gene is functionally similar to purA and is able to at least partially rescue the production of adenine-related purines in D. melanogaster and G. mellonella. The quorum-sensing regulator CepR was previously shown to be important for pathogenicity to C. elegans but not to G. mellonella (8), and our results show that this regulator is also not essential for pathogenicity to D. melanogaster (Table 3).
Fig 5.
Virulence of wild-type B. cenocepacia H111 and attenuated auxotrophic Tn5 mutants in the D. melanogaster infection model. Fifteen flies were inoculated with bacterial culture per experiment and incubated at 26°C. Live flies were counted every 24 h postinfection. Data are based on three independent experiments and were analyzed using survival curves generated by the Kaplan-Meier statistical method. The significance of the difference between results for the wild type and the mutants was determined using the log rank (Mantel-Cox) test. For all the tested strains the P value was <0.0001.
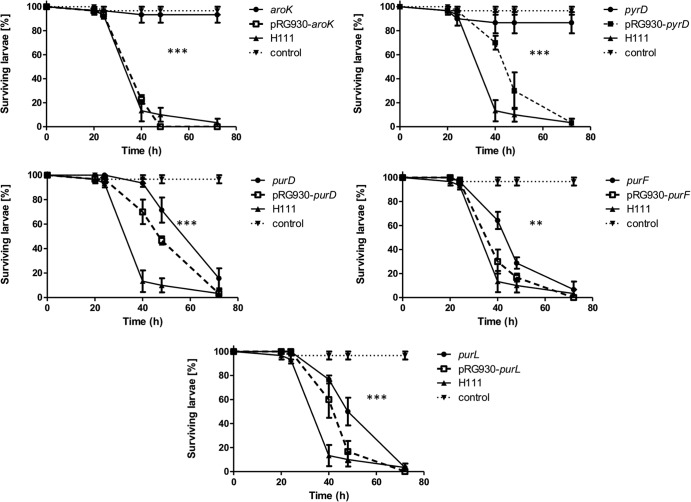
Fig 6.
Virulence of wild-type B. cenocepacia H111type, attenuated auxotrophic Tn5 mutants, and complemented mutant strains in the G. mellonella infection model. G. mellonella larvae were infected with approximately 2 × 105 bacteria and incubated at 30°C in the dark. Live and dead larvae were counted after 20, 24, 40, 48, and 72 h postinfection. The curves were calculated using three independent replicates. The significance of the difference between results for the wild type and the Tn5 mutants was determined using the log rank (Mantel-Cox) test and is indicated as follows: *, 0.01 ≤ P ≤ 0.05; **, 0.001 ≤ P ≤ 0.01; ***, P < 0.001.
In summary, five mutants were identified which were attenuated in all three infection hosts used. The genes inactivated in these strains (aroK, pyrD, purD, purF, and purL) may therefore encode factors that are universally important for virulence, whereas the genes inactivated in the other mutants were specific virulence factors for C. elegans and/or D. melanogaster.
Complementation and supplementation of the Tn5 metabolic mutants.
The aroK, pyrD, purD, purF, and purL mutants, which showed attenuation in all the animal models tested, were complemented using a B. cenocepacia H111 cosmid library. All complemented mutants were able to grow at the wild-type rate on minimal medium with citrate as a carbon source (see Fig. S2 in the supplemental material). Furthermore, pathogenicity of the complemented mutants was at least partially restored (Fig. 6). Supplementation of nematode growth medium with histidine, cysteine, or adenosine as appropriate restored virulence to wild-type levels in the C. elegans model in all metabolic mutants except the purD mutant, which showed an intermediate level of virulence (Fig. 1; Table 3).
DISCUSSION
Over the past few years, nonmammalian infection models have been established as attractive alternatives to traditional animal models because of their practical advantages, particularly the possibility of performing high-throughput screens. In this study, we used the nematode C. elegans as an infection host to screen a B. cenocepacia H111 mutant library for attenuated strains.
Previous work has shown that the CepIR QS system is crucial for B. cenocepacia virulence in C. elegans, as it controls the expression of factors that contribute to pathogenicity in this infection model (8, 22). The finding that in one of the attenuated mutants the transposon had inactivated cepR and in two other mutants (the rsuA and lon mutants) it had affected the QS circuitry therefore validates the screening strategy. One of the QS-regulated virulence factors that has been shown to be important for killing of C. elegans is the protein AidA, although its mode of action remains to be elucidated (12). Interestingly, AidA has not been found to play a role in any other infection host tested so far, and it thus appears to be a specific virulence factor required for infection of nematodes (8). The finding that the trpA and trpB mutants produce no or greatly reduced amounts of AidA may contribute to the specific attenuation of these strains in the C. elegans model (Table 3). Supplementing the medium with tryptophan restored AidA production as well as virulence of the two mutants (Fig. 1 and data not shown), indicating that the attenuation of the two mutants is not a consequence of a secondary mutation. However, at present it is unclear how trpA and trpB, which are required for tryptophan biosynthesis, affect expression of AidA.
Other QS-regulated virulence factors of B. cenocepacia include the ZmpA and ZmpB proteases and the siderophore pyochelin (8). Proteolytic activity was shown to be important for pathogenicity in mammals but not in invertebrates or alfalfa, likely because they specifically modulate the host immune response of mammals by degrading specific tissue components such as collagen and fibronectin and by obstructing immune proteins (55–57). Hence, the abolished or lowered proteolytic activity observed for several of the mutants (Table 3) does not account for their lowered virulence in the infection models used. However, one would expect that these mutants would be attenuated in mammals. Pyochelin production in B. cenocepacia has been shown to be dependent on the availability of its precursors, salicylic acid and cysteine (58). As expected, all five mutants with insertions in the genes of the cysteine biosynthetic pathway showed a reduction in pyochelin production (Fig. 2) and an increase in the accumulation of its precursor salicylic acid. Likewise, the aroK mutant, which is defective in the biosynthesis of aromatic metabolites, including the pyochelin precursor salicylic acid, did not produce pyochelin.
Our mutant screen identified a few genes that were essential for virulence in all three nonmammalian infection models used in this study. Rather than coding for typical virulence factors, these genes were purF, purD, and purL from the purine biosynthesis pathway, pyrD from the pyrimidine synthesis pathway, and aroK from the shikimate pathway. Given that B. cenocepacia produces a battery of virulence factors (7), the inactivation of just one of these may not greatly affect the overall pathogenicity of the organism, explaining why such factors were not identified in our screen. The genes identified encode enzymes that are critical for essential anabolic pathways, and consequently these mutants were unable to grow in minimal medium. Importantly, auxotrophy per se is not the reason for the reduced virulence of these strains, as we determined that approximately 8% of the mutants in the transposon insertion library were unable to grow in minimal medium. This suggests that it is the lack or shortage of specific metabolites in each infection host that causes attenuation. In agreement with this hypothesis, we observed that nematode pathogenicity of the mutants could be restored to wild-type levels by supplementing the medium with appropriate metabolites (Fig. 1). It is important to note, however, that on NG medium, which was used for the C. elegans assays, the mutants showed no growth defects, suggesting that the reduced virulence is not a just a consequence of a lowered infection dose. In the case of the aroK mutant, we tested whether the strain could persist within infected G. mellonella larvae. At 8 days postinfection the animals did not show any disease symptoms, yet we were able to isolate the mutant from the hemolymph of the larvae (approximately 550 bacteria per μl), demonstrating that the bacteria were capable of in vivo survival. We hypothesize that the nutritional environment in the infection host supports persistence of the mutant but neither significant growth nor energy-consuming virulence factor production.
Although it remains to be determined whether our B. cenocepacia mutants are also attenuated in a mammalian infection host, it is notable that the same metabolic pathways have been identified as key systems in virulence in murine models. De novo purine biosynthesis has been shown to be essential for the virulence of a variety of pathogens, including Francisella tularensis (59), Salmonella enterica serovar Typhimurium (60), Staphylococcus aureus (61), Streptococcus pneumoniae (62), Yersinia pestis (63), Vibrio vulnificus (64), Bacillus anthracis (54, 65), Brucella melitensis (66), and Brucella abortus (67). Previous work has also shown that in V. vulnificus, pyrimidine biosynthetic genes are preferentially expressed during infection, and a pyrH mutant was attenuated in virulence (64, 68). In Listeria monocytogenes, both purine and pyrimidine biosynthetic genes were found to be upregulated in infected mammalian cells (69). In a recent study, it was demonstrated that de novo nucleotide biosynthesis is critical for survival and growth of bacteria in human serum, and therefore the purine and pyrimidine biosynthetic pathways are essential for proliferation of bacterial pathogens in the bloodstream (70). Finally, the shikimate pathway, of which the AroK protein is a part, has been demonstrated to be required for virulence of S. enterica serovar Typhimurium (71), P. aeruginosa (72), and L. monocytogenes (73). Most interestingly in the context of this study is the finding that inactivation of aroB, which is required for shikimate biosynthesis in Burkholderia pseudomallei, the causative agent of melioidosis, renders the organism avirulent. Moreover, murine challenge studies revealed partial protection in BALB/c mice vaccinated with an aroB mutant (74). Given that the shikimate pathway is crucial to bacteria but missing in mammals (75), the enzymes involved in this pathway have been considered particularly interesting drug targets for developing nontoxic antimicrobial agents (76).
In summary, we have identified several genes in B. cenocepacia that are critical for pathogenicity in multiple nonmammalian infection hosts. These results not only further our understanding of the virulence mechanisms used by this opportunistic pathogen but also have led to the identification of some potential targets for the development of novel antibacterial drugs. Some of the strongly attenuated mutants identified in this study could also be of interest for the development of live vaccines.
Supplementary Material
ACKNOWLEDGMENTS
This work has been supported by the Swiss National Science Foundation (projects 31003A-122013 and 143773) and the Danish Council for Strategic Research (CAR project).
Footnotes
Published ahead of print 22 October 2012
Supplemental material for this article may be found at http://dx.doi.org/10.1128/IAI.00768-12.
REFERENCES
- 1. Coenye T, Vandamme P. 2003. Diversity and significance of Burkholderia species occupying diverse ecological niches. Environ. Microbiol. 5:719–729 [DOI] [PubMed] [Google Scholar]
- 2. Eberl L, Tummler B. 2004. Pseudomonas aeruginosa and Burkholderia cepacia in cystic fibrosis: genome evolution, interactions and adaptation. Int. J. Med. Microbiol. 294:123–131 [DOI] [PubMed] [Google Scholar]
- 3. Mahenthiralingam E, Baldwin A, Dowson CG. 2008. Burkholderia cepacia complex bacteria: opportunistic pathogens with important natural biology. J. Appl. Microbiol. 104:1539–1551 [DOI] [PubMed] [Google Scholar]
- 4. Mahenthiralingam E, Urban TA, Goldberg JB. 2005. The multifarious, multireplicon Burkholderia cepacia complex. Nat. Rev. Microbiol. 3:144–156 [DOI] [PubMed] [Google Scholar]
- 5. Isles A, Maclusky I, Corey M, Gold R, Prober C, Fleming P, Levison H. 1984. Pseudomonas cepacia infection in cystic-fibrosis—an emerging problem. J. Pediatr. 104:206–210 [DOI] [PubMed] [Google Scholar]
- 6. Baldwin A, Sokol PA, Parkhill J, Mahenthiralingam E. 2004. The Burkholderia cepacia epidemic strain marker is part of a novel genomic island encoding both virulence and metabolism-associated genes in Burkholderia cenocepacia. Infect. Immun. 72:1537–1547 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 7. Loutet SA, Valvano MA. 2010. A decade of Burkholderia cenocepacia virulence determinant research. Infect. Immun. 78:4088–4100 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 8. Uehlinger S, Schwager S, Bernier SP, Riedel K, Nguyen DT, Sokol PA, Eberl L. 2009. Identification of specific and universal virulence factors in Burkholderia cenocepacia strains by using multiple infection hosts. Infect. Immun. 77:4102–4110 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 9. Castonguay-Vanier J, Vial L, Tremblay J, Deziel E. 2010. Drosophila melanogaster as a model host for the Burkholderia cepacia complex. PLoS One 5:e11467 doi:10.1371/journal.pone.0011467 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 10. Apidianakis Y, Rahme LG. 2009. Drosophila melanogaster as a model host for studying Pseudomonas aeruginosa infection. Nat. Protoc. 4:1285–1294 [DOI] [PubMed] [Google Scholar]
- 11. Seed KD, Dennis JJ. 2008. Development of Galleria mellonella as an alternative infection model for the Burkholderia cepacia complex. Infect. Immun. 76:1267–1275 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 12. Huber B, Feldmann F, Kothe M, Vandamme P, Wopperer J, Riedel K, Eberl L. 2004. Identification of a novel virulence factor in Burkholderia cenocepacia H111 required for efficient slow killing of Caenorhabditis elegans. Infect. Immun. 72:7220–7230 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 13. Bertani G. 1951. Studies on lysogenesis. 1. The mode of phage liberation by lysogenic Escherichia coli. J. Bacteriol. 62:293–300 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 14. Tan MW, Rahme LG, Sternberg JA, Tompkins RG, Ausubel FM. 1999. Pseudomonas aeruginosa killing of Caenorhabditis elegans used to identify P. aeruginosa virulence factors. Proc. Natl. Acad. Sci. U. S. A. 96:2408–2413 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 15. Brenner S. 1974. Genetics of Caenorhabditis elegans. Genetics 77:71–94 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 16. Sambrook J, Fritsch EF, Maniatis T. 1989. Molecular cloning: a laboratory manual, 2nd ed Cold Spring Harbor Laboratory Press, Cold Spring Harbor, NY [Google Scholar]
- 17. Southern EM. 1975. Detection of specific sequences among DNA fragments separated by gel-electrophoresis. J. Mol. Biol. 98:503–517 [DOI] [PubMed] [Google Scholar]
- 18. O'Toole GA, Kolter R. 1998. Initiation of biofilm formation in Pseudomonas fluorescens WCS365 proceeds via multiple, convergent signalling pathways: a genetic analysis. Mol. Microbiol. 28:449–461 [DOI] [PubMed] [Google Scholar]
- 19. Better M, Lewis B, Corbin D, Ditta G, Helinski DR. 1983. Structural relationships among Rhizobium meliloti symbiotic promoters. Cell 35:479–485 [DOI] [PubMed] [Google Scholar]
- 20. Vandeneede G, Deblaere R, Goethals K, Vanmontagu M, Holsters M. 1992. Broad host range and promoter selection vectors for bacteria that interact with plants. Mol. Plant Microbe Interact. 5:228–234 [DOI] [PubMed] [Google Scholar]
- 21. de Lorenzo V, Timmis KN. 1994. Analysis and construction of stable phenotypes in gram-negative bacteria with Tn5-derived and Tn10-derived minitransposons. Methods Enzymol. 235:386–405 [DOI] [PubMed] [Google Scholar]
- 22. Köthe M, Antl M, Huber B, Stoecker K, Ebrecht D, Steinmetz I, Eberl L. 2003. Killing of Caenorhabditis elegans by Burkholderia cepacia is controlled by the cep quorum-sensing system. Cell. Microbiol. 5:343–351 [DOI] [PubMed] [Google Scholar]
- 23. Cardona ST, Wopperer J, Eberl L, Valvano MA. 2005. Diverse pathogenicity of Burkholderia cepacia complex strains in the Caenorhabditis elegans host model. FEMS Microbiol. Lett. 250:97–104 [DOI] [PubMed] [Google Scholar]
- 24. Stiernagle T, Hope IA. (ed). 1999. C. elegans: a practical approach. Oxford University Press, New York, NY [Google Scholar]
- 25. Schwyn B, Neilands JB. 1987. Universal chemical-assay for the detection and determination of siderophores. Anal. Biochem. 160:47–56 [DOI] [PubMed] [Google Scholar]
- 26. Lewenza S, Conway B, Greenberg EP, Sokol PA. 1999. Quorum sensing in Burkholderia cepacia: identification of the LuxRI homologs CepRI. J. Bacteriol. 181:748–756 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 27. Visca P, Ciervo A, Sanfilippo V, Orsi N. 1993. Iron-regulated salicylate synthesis by Pseudomonas spp. J. Gen. Microbiol. 139:1995–2001 [DOI] [PubMed] [Google Scholar]
- 28. Rinehart KL, Staley AL, Wilson SR, Ankenbauer RG, Cox CD. 1995. Stereochemical assignment of the pyochelins. J. Org. Chem. 60:2786–2791 [Google Scholar]
- 29. Ankenbauer RG, Toyokuni T, Staley A, Rinehart KL, Jr, Cox CD. 1988. Synthesis and biological activity of pyochelin, a siderophore of Pseudomonas aeruginosa. J. Bacteriol. 170:5344–5351 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 30. Gerhardt P, Murray RGE, Costilow RN, Nester EW, Wood WA, Krieg NR, Phillips GB. (ed). 1981. Manual of methods for general bacteriology. American Society for Microbiology, Washington, DC [Google Scholar]
- 31. Sage A, Linker A, Evans LR, Lessie TG. 1990. Hexose phosphate-metabolism and exopolysaccharide formation in Pseudomonas cepacia. Curr. Microbiol. 20:191–198 [Google Scholar]
- 32. Clark DJ, Maaloe O. 1967. DNA replication and division cycle in Escherichia coli. J. Mol. Biol. 23:99–112 [Google Scholar]
- 33. Sokol PA, Darling P, Woods DE, Mahenthiralingam E, Kooi C. 1999. Role of ornibactin biosynthesis in the virulence of Burkholderia cepacia: characterization of pvdA, the gene encoding l-ornithine N-5-oxygenase. Infect. Immun. 67:4443–4455 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 34. Visser MB, Majumdar S, Hani E, Sokol PA. 2004. Importance of the ornibactin and pyochelin siderophore transport systems in Burkholderia cenocepacia lung infections. Infect. Immun. 72:2850–2857 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 35. Malott RJ, O'Grady EP, Toller J, Inhuelsen S, Eberl L, Sokol PA. 2009. A Burkholderia cenocepacia orphan LuxR homolog is involved in quorum-sensing regulation. J. Bacteriol. 191:2447–2460 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 36. Chung JW, Altman E, Beveridge TJ, Speert DP. 2003. Colonial morphology of Burkholderia cepacia complex genomovar III: implications in exopolysaccharide production, pilus expression, and persistence in the mouse. Infect. Immun. 71:904–909 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 37. Cunha MV, Sousa SA, Leitao JH, Moreira LM, Videira PA, SA-Correia I. 2004. Studies on the involvement of the exopolysaccharide produced by cystic fibrosis-associated isolates of the Burkholderia cepacia complex in biofilm formation and in persistence of respiratory infections. J. Clin. Microbiol. 42:3052–3058 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 38. Bartholdson SJ, Brown AR, Mewburn BR, Clarke DJ, Fry SC, Campopiano DJ, Govan JR. 2008. Plant host and sugar alcohol induced exopolysaccharide biosynthesis in the Burkholderia cepacia complex. Microbiology 154:2513–2521 [DOI] [PubMed] [Google Scholar]
- 39. Ferreira AS, Leitao JH, Silva IN, Pinheiro PF, Sousa SA, Ramos CG, Moreira LM. 2010. Distribution of cepacian biosynthesis genes among environmental and clinical Burkholderia strains and role of cepacian exopolysaccharide in resistance to stress conditions. Appl. Environ. Microbiol. 76:441–450 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 40. Conway BA, Chu KK, Bylund J, Altman E, Speert DP. 2004. Production of exopolysaccharide by Burkholderia cenocepacia results in altered cell-surface interactions and altered bacterial clearance in mice. J. Infect. Dis. 190:957–966 [DOI] [PubMed] [Google Scholar]
- 41. Sousa SA, Ulrich M, Bragonzi A, Burke M, Worlitzsch D, Leitao JH, Meisner C, Eberl L, SA-Correia I, Doring G. 2007. Virulence of Burkholderia cepacia complex strains in gp91phox−/− mice. Cell. Microbiol. 9:2817–2825 [DOI] [PubMed] [Google Scholar]
- 42. Agnoli K, Schwager S, Uehlinger S, Vergunst A, Viteri DF, Nguyen DT, Sokol PA, Carlier A, Eberl L. 2012. Exposing the third chromosome of Burkholderia cepacia complex strains as a virulence plasmid. Mol. Microbiol. 83:362–378 [DOI] [PubMed] [Google Scholar]
- 43. Huber B, Riedel K, Kothe M, Givskov M, Molin S, Eberl L. 2002. Genetic analysis of functions involved in the late stages of biofilm development in Burkholderia cepacia H111. Mol. Microbiol. 46:411–426 [DOI] [PubMed] [Google Scholar]
- 44. Van Melderen L, Aertsen A. 2009. Regulation and quality control by Lon-dependent proteolysis. Res. Microbiol. 160:645–651 [DOI] [PubMed] [Google Scholar]
- 45. Snider J, Thibault G, Houry WA. 2008. The AAA plus superfamily of functionally diverse proteins. Genome Biology 9:216. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 46. Takaya A, Suzuki M, Matsui H, Tomoyasu T, Sashinami H, Nakane A, Yamamoto T. 2003. Lon, a stress-induced ATP-dependent protease, is critically important for systemic Salmonella enterica serovar Typhimurium infection of mice. Infect. Immun. 71:690–696 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 47. Lan L, Deng X, Xiao Y, Zhou J-M, Tang X. 2007. Mutation of Lon protease differentially affects the expression of Pseudomonas syringae type III secretion system genes in rich and minimal media and reduces pathogenicity. Mol. Plant Microbe Interact. 20:682–696 [DOI] [PubMed] [Google Scholar]
- 48. Cohn MT, Ingmer H, Mulholland F, Jorgensen K, Wells JM, Brondsted L. 2007. Contribution of conserved ATP-dependent proteases of Campylobacter jejuni to stress tolerance and virulence. Appl. Environ. Microbiol. 73:7803–7813 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 49. Su SC, Stephens BB, Alexandre G, Farrand SK. 2006. Lon protease of the alpha-proteobacterium Agrobacterium tumefaciens is required for normal growth, cellular morphology and full virulence. Microbiology 152:1197–1207 [DOI] [PubMed] [Google Scholar]
- 50. Robertson GT, Kovach ME, Allen CA, Ficht TA, Roop RM. 2000. The Brucella abortus Lon functions as a generalized stress response protease and is required for wild-type virulence in BALB/c mice. Mol. Microbiol. 35:577–588 [DOI] [PubMed] [Google Scholar]
- 51. Butler SM, Festa RA, Pearce MJ, Darwin KH. 2006. Self-compartmentalized bacterial proteases and pathogenesis. Mol. Microbiol. 60:553–562 [DOI] [PubMed] [Google Scholar]
- 52. Tsilibaris V, Maenhaut-Michel G, Van Melderen L. 2006. Biological roles of the Lon ATP-dependent protease. Res. Microbiol. 157:701–713 [DOI] [PubMed] [Google Scholar]
- 53. Zhang Y, Morar M, Ealick SE. 2008. Structural biology of the purine biosynthetic pathway. Cell. Mol. Life Sci. 65:3699–3724 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 54. Jenkins A, Cote C, Twenhafel N, Merkel T, Bozue J, Welkos S. 2011. Role of purine biosynthesis in Bacillus anthracis pathogenesis and virulence. Infect. Immun. 79:153–166 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 55. Corbett CR, Burtnick MN, Kooi C, Woods DE, Sokol PA. 2003. An extracellular zinc metalloprotease gene of Burkholderia cepacia. Microbiology 149:2263–2271 [DOI] [PubMed] [Google Scholar]
- 56. Kooi C, Subsin B, Chen R, Pohorelic B, Sokol PA. 2006. Burkholderia cenocepacia ZmpB is a broad-specificity zinc metalloprotease involved in virulence. Infect. Immun. 74:4083–4093 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 57. Kooi C, Corbett CR, Sokol PA. 2005. Functional analysis of the Burkholderia cenocepacia ZmpA metalloprotease. J. Bacteriol. 187:4421–4429 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 58. Farmer KL, Thomas MS. 2004. Isolation and characterization of Burkholderia cenocepacia mutants deficient in pyochelin production: pyochelin biosynthesis is sensitive to sulfur availability. J. Bacteriol. 186:270–277 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 59. Meibom KL, Charbit A. 2010. Francisella tularensis metabolism and its relation to virulence. Front. Microbiol. 1:140. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 60. Hoiseth SK, Stocker BAD. 1981. Aromatic-dependent Salmonella typhimurium are non-virulent and effective as live vaccines. Nature 291:238–239 [DOI] [PubMed] [Google Scholar]
- 61. Mei JM, Nourbakhsh F, Ford CW, Holden DW. 1997. Identification of Staphylococcus aureus virulence genes in a murine model of bacteraemia using signature-tagged mutagenesis. Mol. Microbiol. 26:399–407 [DOI] [PubMed] [Google Scholar]
- 62. Polissi A, Pontiggia A, Feger G, Altieri M, Mottl H, Ferrari L, Simon D. 1998. Large-scale identification of virulence genes from Streptococcus pneumoniae. Infect. Immun. 66:5620–5629 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 63. Brubaker RR. 1970. Interconversion of purine mononucleotides in Pasteurella pestis. Infect. Immun. 1:446–454 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 64. Kim YR, Lee SE, Kim CM, Kim SY, Shin EK, Shin DH, Chung SS, Choy HE, Progulske-Fox A, Hillman JD, Handfield M, Rhee JH. 2003. Characterization and pathogenic significance of Vibrio vulnificus antigens preferentially expressed in septicemic patients. Infect. Immun. 71:5461–5471 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 65. Ivanovics G, Marjai E, Dobozy A. 1968. The growth of purine mutants of Bacillus anthracis in the body of the mouse. J. Gen. Microbiol. 53:147–162 [DOI] [PubMed] [Google Scholar]
- 66. Crawford RM, Van De Verg L, Yuan L, Hadfield TL, Warren RL, Drazek ES, Houng HH, Hammack C, Sasala K, Polsinelli T, Thompson J, Hoover DL. 1996. Deletion of purE attenuates Brucella melitensis infection in mice. Infect. Immun. 64:2188–2192 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 67. Alcantara RB, Read RDA, Valderas MW, Brown TD, Roop RM. 2004. Intact purine biosynthesis pathways are required for wild-type virulence of Brucella abortus 2308 in the BALB/c mouse model. Infect. Immun. 72:4911–4917 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 68. Lee SE, Kim SY, Kim CM, Kim MK, Kim YR, Jeong K, Ryu HJ, Lee YS, Chung SS, Choy HE, Rhee JH. 2007. The pyrH gene of Vibrio vulnificus is an essential in vivo survival factor. Infect. Immun. 75:2795–2801 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 69. Klarsfeld AD, Goossens PL, Cossart P. 1994. Five Listeria monocytogenes genes preferentially expressed in infected mammalian cells: plcA, purH, purD, pyrE and an arginine ABC transporter gene, arpJ. Mol. Microbiol. 13:585–597 [DOI] [PubMed] [Google Scholar]
- 70. Samant S, Lee H, Ghassemi M, Chen J, Cook JL, Mankin AS, Neyfakh AA. 2008. Nucleotide biosynthesis is critical for growth of bacteria in human blood. PLoS Pathog. 4:e37 doi:10.1371/journal.ppat.0040037 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 71. Gunel-Ozcan A, Brown KA, Allen AG, Maskell DJ. 1997. Salmonella typhimurium aroB mutants are attentuated in BALB/c mice. Microb. Pathog. 23:311–316 [DOI] [PubMed] [Google Scholar]
- 72. Priebe GP, Meluleni GJ, Coleman FT, Goldberg JB, Pier GB. 2003. Protection against fatal Pseudomonas aeruginosa pneumonia in mice after nasal immunization with a live, attenuated aroA deletion mutant. Infect. Immun. 71:1453–1461 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 73. Stritzker J, Janda J, Schoen C, Taupp M, Pilgrim S, Gentschev I, Schreier P, Geginat G, Goebel W. 2004. Growth, virulence, and immunogenicity of Listeria monocytogenes aro mutants. Infect. Immun. 72:5622–5629 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 74. Cuccui J, Easton A, Chu KK, Bancroft GJ, Oyston PCF, Titball RW, Wren BW. 2007. Development of signature-tagged mutagenesis in Burkholderia pseudomallei to identify genes important in survival and pathogenesis. Infect. Immun. 75:1186–1195 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 75. Roberts F, Roberts CW, Johnson JJ, Kyle DE, Krell T, Coggins JR, Coombs GH, Milhous WK, Tzipori S, Ferguson DJP, Chakrabarti D, McLeod R. 1998. Evidence for the shikimate pathway in apicomplexan parasites. Nature 393:801–805 [DOI] [PubMed] [Google Scholar]
- 76. Coggins JR, Abell C, Evan LB, Frederickson M, Robinson DA, Roszak AW, Lapthorn AP. 2003. Experiences with the shikimate-pathway enzymes as targets for rational drug design. Biochem. Soc. Trans. 31:548–552 [DOI] [PubMed] [Google Scholar]
- 77. Steidle A, Sigl K, Schuhegger R, Ihring A, Schmid M, Gantner S, Stoffels M, Riedel K, Givskov M, Hartmann A, Langebartels C, Eberl L. 2001. Visualization of N-acylhomoserine lactone-mediated cell-cell communication between bacteria colonizing the tomato rhizosphere. Appl. Environ. Microbiol. 67:5761–5770 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 78. Romling U, Fiedler B, Bosshammer J, Grothues D, Greipel J, von der Hardt H, Tummler B. 1994. Epidemiology of chronic Pseudomonas aeruginosa infections in cystic fibrosis. J. Infect. Dis. 170:1616–1621 [DOI] [PubMed] [Google Scholar]
- 79. Huber B, Riedel K, Hentzer M, Heydorn A, Gotschlich A, Givskov M, Molin S, Eberl L. 2001. The cep quorum-sensing system of Burkholderia cepacia H111 controls biofilm formation and swarming motility. Microbiology 147:2517–2528 [DOI] [PubMed] [Google Scholar]
- 80. Winson MK, Swift S, Fish L, Throup JP, Jorgensen F, Chhabra SR, Bycroft BW, Williams P, Stewart G. 1998. Construction and analysis of luxCDABE-based plasmid sensors for investigating N-acyl homoserine lactone-mediated quorum sensing. FEMS Microbiol. Lett. 163:185–192 [DOI] [PubMed] [Google Scholar]
- 81. Winson MK, Swift S, Hill PJ, Sims CM, Griesmayr G, Bycroft BW, Williams P, Stewart G. 1998. Engineering the luxCDABE genes from Photorhabdus luminescens to provide a bioluminescent reporter for constitutive and promoter probe plasmids and mini-Tn5 constructs. FEMS Microbiol. Lett. 163:193–202 [DOI] [PubMed] [Google Scholar]
Associated Data
This section collects any data citations, data availability statements, or supplementary materials included in this article.