Abstract
Trichomonas vaginalis infection in males has been largely uncharacterized. Past reports indicated increased susceptibility to other sexually transmitted infection (STI) agents such as human immunodeficiency virus and Neisseria gonorrhoeae with concurrent T. vaginalis infection. This warrants a more thorough review of male T. vaginalis incidence. A retrospective 3-year investigation of transcription-mediated amplification (TMA)-based urethral swab and first-void urine screening for T. vaginalis within a regional health care system was performed to address T. vaginalis prevalence in males. Of 622 total samples tested, 6.6% were positive for T. vaginalis. Delineation of all specimens by ZIP code of patient residence revealed 11 predominant ZIP codes with respect to testing volume and detection rates. Within these 11 ZIP codes, representing 78.3% of total testing volume, urine was the preferred specimen source compared to urethral swabs. Seven of these 11 ZIP codes contained majority African American populations. The aggregate T. vaginalis detection rate trended higher than that of the remaining four ZIP codes, which were comprised primarily of Caucasian populations (8.9% versus 5.0%, respectively; P = 0.15). The average age of a T. vaginalis-infected male (39.9 years) was significantly greater than those for Chlamydia trachomatis or N. gonorrhoeae (27.6 and 25.9 years, respectively; P < 0.001). Given the significant rate of T. vaginalis detection, with age distribution analogous to that reported in females, TMA-based detection of T. vaginalis can be a routine constituent within a comprehensive STI screening panel for males in high-prevalence STI communities.
INTRODUCTION
Past literature has estimated the worldwide incidence of trichomoniasis at 180 million cases, with 3 to 5 million cases occurring annually in the United States (1). Trichomonas vaginalis infection also plays an important role in the acquisition (2, 3) and transmission (4, 5) of human immunodeficiency virus. Moreover, codetection of Neisseria gonorrhoeae with T. vaginalis is a common occurrence (6). These data were largely generated using T. vaginalis diagnostic modalities (typically, vaginal saline suspension microscopy [wet mount] and culture) which have proven to be less sensitive than nucleic acid amplification testing (7–9).
Little clinical information is available about asymptomatic trichomoniasis in males (10). Molecular diagnostic techniques may enhance our ability to detect trichomoniasis. The advent of noncommercial PCR testing has increased T. vaginalis detection in males compared to culture techniques (11, 12). Prevalence rates in males presenting to sexually transmitted infection (STI) clinics have exceeded 10% (11, 12), with limited data suggesting increased distribution in older males (13). Furthermore, studies in females utilizing transcription-mediated amplification (TMA) demonstrated significant codetection of N. gonorrhoeae with T. vaginalis and further association with Chlamydia trachomatis (8, 14). These methods lend themselves to a true assessment of T. vaginalis in male populations. We hereby present data on the prevalence of trichomoniasis from a large male population presenting for nonemergent care within a metropolitan area of high STI prevalence.
(Results of this work were previously presented, in part, at the 111th General Meeting of the American Society for Microbiology, New Orleans, LA, 21 to 24 May 2011.)
MATERIALS AND METHODS
Setting.
U.S. metropolitan statistical area (MSA) data document a long-standing trend of high STI prevalence in the Milwaukee (Wisconsin) metropolitan area (15). The Milwaukee-Waukesha-West Allis MSA gonorrhea rate was 219.6 per 100,000 population in 2010. This was the second highest in the United States and 92.8% higher than the national cumulative MSA rate of 113.9 per 100,000 population. This MSA also had the second-highest chlamydia rate (738.1 per 100,000 population). The national MSA chlamydia rate was 452.6 per 100,000 population. Testing was performed at Wheaton Franciscan Laboratory, a centralized laboratory serving five Milwaukee metropolitan hospitals and an approximately 70-clinic outpatient physician group. The TMA-based T. vaginalis analyte-specific reagent (ASR) was offered to clinicians as a stand-alone assay or in conjunction with C. trachomatis and N. gonorrhoeae molecular screening.
Specimen collection.
Primary urethral specimens were deposited into Aptima swab specimen transport tubes per the Aptima Combo 2 assay (Gen-Probe, Incorporated, San Diego, CA) package insert protocol. Alternatively, approximate 2-ml aliquots of first-void male urine were added to Aptima urine specimen transport tubes within 24 h of specimen procurement.
Primary molecular screening assays.
Nonautomated T. vaginalis ASR, detecting organism-specific 16S rRNA via target capture, TMA, and chemiluminescent hybridization protection, was previously validated in-house on male urethral and urine specimens in conjunction with a proprietary alternative target TMA-based confirmatory assay (Gen-Probe). Hardick et al. (16) reported >96% sensitivity and specificity of a TMA-based assay for detection of T. vaginalis from males. Relative light unit values (RLU) of ≥50,000, as generated by stand-alone luminometry, were interpreted as positive. A similar method, the Aptima Combo 2 assay, was used for detection of N. gonorrhoeae-specific 16S rRNA and Chlamydia trachomatis-specific 23S rRNA (17).
Study.
The Wheaton Franciscan Healthcare institutional review board approved this 3-year study of all male primary clinical specimens subjected to T. vaginalis ASR. Greater than 99% of all specimens were also screened for C. trachomatis and N. gonorrhoeae. Specimen source, screening result, age of patient, race/ethnicity of patient (when available), and ZIP code of patient residence were collected. Five-digit ZIP code tabulation areas were accessed through the 2000 United States Census database to provide racial/ethnicity distribution. The STI phenotype (permutations of T. vaginalis, N. gonorrhoeae, and/or C. trachomatis TMA-based detection) was computed from any health care encounter that resulted in detection of at least one STI.
Statistical analysis.
The significance test of proportions (two-proportion Z-ratio) determined if differences in either rates of positive screening results, proportion of specimen source, or STI phenotype were significant. The t test for independent samples determined if differences in mean patient age associated with positive results were significant between STIs. The alpha level was set at 0.05 before the investigations commenced, and all P values are two-tailed.
RESULTS
STI profile in males.
The T. vaginalis detection rate (6.6%) from 622 specimens showed no significant difference versus that of C. trachomatis (8.8%; P = 0.15). Less N. gonorrhoeae (1.9%) was detected than T. vaginalis (P < 0.0002). Six hundred seventeen of the specimens came from health care encounters that resulted in TMA-based screening for all three STIs; 98 (15.9%) health care encounters resulted in detection of at least one STI. Instances of sole T. vaginalis detection (36.7% in a combined first-void urine and urethral swab data set; Table 1) nearly equaled those of sole C. trachomatis detection (39.8%). In terms of individual STI agents, Table 1 reveals that STI phenotype differences between first-void urine and urethral collections were minimal (P ≥ 0.05).
Table 1.
STI phenotype, determined by TMA-based assays specific for Trichomonas vaginalis, Chlamydia trachomatis, and Neisseria gonorrhoeae among screened males positive for at least one STI
| STI phenotypea |
% of patient encounters by specimen source |
||||
|---|---|---|---|---|---|
| Trichomonas vaginalis | Chlamydia trachomatis | Neisseria gonorrhoeae | First-void urineb | Urethrac | Totald |
| + | − | − | 39.7 | 28.0 | 36.7 |
| + | + | − | 2.7 | 4.0 | 3.1 |
| + | + | + | 1.4 | 0.0 | 1.0 |
| + | − | + | 1.4 | 0.0 | 1.0 |
| − | + | − | 34.2 | 56.0 | 39.8 |
| − | − | + | 6.8 | 8.0 | 7.1 |
| − | + | + | 13.7 | 4.0 | 11.2 |
+, positive TMA screen; −, negative TMA screen.
Seventy-three (15.5%) encounters had at least one STI.
Twenty-five (17.2%) encounters had at least one STI.
Ninety-eight (15.9%) encounters had at least one STI.
Demographics of males with T. vaginalis.
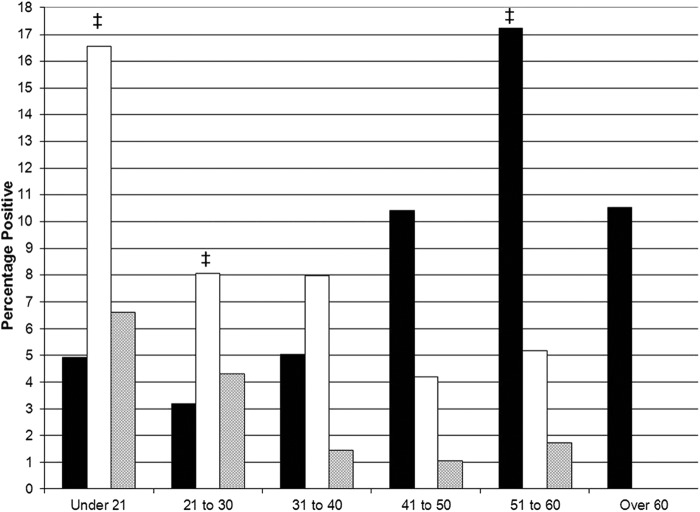
Mean and median ages of males tested were 33 and 31, respectively (range, 13 to 87 years). Detection of T. vaginalis was more common in older men (Fig. 1) and exceeded detection rates of the other STIs in this demographic. The age range of males with T. vaginalis was 14 to 67 years (median, 45); the mean age of 39.9 was greater than those associated with C. trachomatis and N. gonorrhoeae (27.6 and 25.9, respectively; P < 0.0001). T. vaginalis was the only STI agent detected in men over 60 years.
Fig 1.
Prevalence of Trichomonas vaginalis (solid bars), Chlamydia trachomatis (open bars), and Neisseria gonorrhoeae (shaded bars) among 622 males screened by transcription-mediated amplification-based methods, delineated by age (x axis). ‡, comparisons between T. vaginalis and C. trachomatis detection rates in which P values are <0.05.
For males with detectable T. vaginalis from whom race/ethnicity data were available (n = 29), 89.7% were African American. Eleven Milwaukee County ZIP code tabulation areas yielded more than 78% of all male T. vaginalis ASR screens. Characteristics of this data subset, in terms of STI phenotype and specimen submission ratio, were representative of the overall data set described in this report (data not illustrated). In spite of seven ZIP code tabulation areas having an African American majority (Table 2), the T. vaginalis detection rate was not significantly higher in this aggregate than in a four-ZIP code compilation that exhibited a Caucasian majority (P = 0.15).
Table 2.
Delineation of 11 Milwaukee County ZIP code tabulation areas responsible for significant T. vaginalis male screening into geographical areas with majority African American or Caucasian populations, with characterization of the two subgroups
| Parameter | Geographical region race majority |
||
|---|---|---|---|
| African Americana | Caucasianb | P value | |
| Percentage of urine submissionsc | 78.4 | 72.3 | 0.16 |
| Mean no. of screenings per ZIP code | 49.7 | 34.8 | 0.26 |
| Percentage detection ofd: | |||
| Trichomonas vaginalis | 8.9 | 5.0 | 0.15 |
| Chlamydia trachomatis | 8.4 | 9.4 | 0.74 |
| Neisseria gonorrhoeae | 3.7 | 4.3 | 0.78 |
The mean difference between African American and Caucasian populations was 43.9% per ZIP code tabulation area (n = 7).
The mean difference between Caucasian and African American populations was 32.6% per ZIP code tabulation area (n = 4).
Percentage of all submissions.
Combined urethral and male urine submissions.
DISCUSSION
Past diagnostic modalities for male STI screening, including those based on molecular diagnostics, have largely been limited to assays specific for C. trachomatis and N. gonorrhoeae. Introduction of the T. vaginalis ASR has enhanced screening for T. vaginalis, especially in females (18). However, T. vaginalis screening in males has been largely uncharacterized. Wet mount and culture diagnostics, traditionally implemented in female populations, were not utilized in our study set. One study with a limited number of positive patients (16) reported >96% sensitivity of TMA for detection of T. vaginalis in male urine. In our investigation, 6.6% of males within a subacute care population were positive for T. vaginalis. We also show that older men may be a reservoir for transmission of the protozoan. In addition, the T. vaginalis detection rate did not differ from that of C. trachomatis (P = 0.15) but exceeded that of N. gonorrhoeae (P < 0.0002).
Our study provides a more accurate description of STI in the male community. In contrast to female populations that exhibit increased T. vaginalis detection versus C. trachomatis (19, 20), T. vaginalis detection in our male population was not significantly less than C. trachomatis. This finding is relatively novel due to a perceived lack of laboratory diagnostic options for T. vaginalis in males. The male STI phenotype for T. vaginalis, C. trachomatis, and N. gonorrhoeae, although exhibiting no significant difference between first-void urine and urethral specimen sources (Table 1), differed markedly from traditional female STI phenotype data (20–22). In general, a 17 to 24% increase in sole detection of T. vaginalis over C. trachomatis was observed in female populations with STI versus the ∼5% difference noted in this male population. Sole detection of C. trachomatis in female populations with STI was ∼10% less than that observed in this male population.
The age distribution of males with T. vaginalis mimics those previously presented for females (19, 20). To summarize, T. vaginalis detection rates increased with patient age (Fig. 1). These data included an increased rate of T. vaginalis detection over that of C. trachomatis for the 51- to 60-year-old population within this high STI prevalence region (P = 0.04). In addition, a 10.5% detection rate was observed in men over age 60. Such findings now extend to both genders (20) and support the paradigm of T. vaginalis being a more-relevant marker of risky sexual behavior among multiple age groups, including older men. Krashin et al. (23) reported on a risk factor for T. vaginalis infection in a female adolescent population, i.e., having a male sexual partner ≥5 years older. The authors raise the possibility of older males being a conduit between an older female reservoir of trichomoniasis and female adolescents. T. vaginalis ASR presents a viable option for screening these male populations.
A great majority of males with detectable T. vaginalis in this study were African American. One inherent limitation was that not all laboratory requisitions provided race/ethnicity data (data were available for 97% of patients yielding a positive T. vaginalis result). Subsequent translation of patient residence ZIP code into general race/ethnicity representations of local environments via census data suggested widespread organism distribution among both majority Caucasian and majority African American geographical regions (Table 2). While secondary limitations (residence migration during census intervals or sexual partners in other ZIP code tabulation areas) can confound this approach, the apparent community-wide distribution of T. vaginalis itself may result from enhanced analytical sensitivity of the T. vaginalis ASR. Moreover, this finding may have significant clinical and public health ramifications. Additional studies are needed to determine the prevalence of trichomoniasis in the general population.
Our findings warrant additional large-scale investigations. Studies addressing prevalence of T. vaginalis in Caucasian men have documented 11 to 23% T. vaginalis seroprevalence rates (24, 25). However, these studies were conducted in health professional cohorts. Second, when our group first evaluated the T. vaginalis ASR in a metropolitan female population (8), the patients were predominately African American. We had limited access to a Caucasian population. Finally, preliminary data attempting to portray an increased risk of prostate cancer with antecedent T. vaginalis infection (24–26) were inconclusive. However, the report from Stark et al. (24) stated that T. vaginalis-seropositive patients with lethal prostate cancer were 6.4 times more likely to have a documented history of T. vaginalis infection than case controls. Highly sensitive T. vaginalis ASR could provide additional data on the association of T. vaginalis with prostate cancer in all populations.
In conclusion, evidence is provided for another utility of TMA-based testing. In a general sense, T. vaginalis ASR presents the opportunity, in a commercialized format (27), to satisfy a largely unmet clinical and public health need. Increased analytical sensitivity of TMA versus those of other nucleic acid amplification methodologies (28–30) can augment this benefit. This enhanced diagnostic option may ultimately provide epidemiologists the ability to define a more accurate picture of trichomoniasis in the male community. In turn, this may influence screening practices in the realm of targeted populations and chronic disease prevention.
ACKNOWLEDGMENTS
The authors express sincere appreciation to Deb Hamer and Pam Reiss for expert technical assistance.
T.K., C.M., K.L.M., M.N., and E.M. have received travel assistance from Gen-Probe, Incorporated.
Footnotes
Published ahead of print 24 October 2012
REFERENCES
- 1.Weinstock H, Berman S, Cates W., Jr 2004. Sexually transmitted diseases among American youth: incidence and prevalence estimates. Perspect. Sex. Reprod. Health 36:6–10 [DOI] [PubMed] [Google Scholar]
- 2.Chesson HW, Blandford JM, Pinkerton SD. 2004. Estimates of the annual number and cost of new HIV infections among women attributable to trichomoniasis in the United States. Sex. Transm. Dis. 31:547–551 [DOI] [PubMed] [Google Scholar]
- 3.Guenthner PC, Secor WE, Dezzutti CS. 2005. Trichomonas vaginalis-induced epithelial monolayer disruption and human immunodeficiency virus type 1 (HIV-1) replication: implications for the sexual transmission of HIV-1. Infect. Immun. 73:4155–4160 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 4.Hobbs MM, Kazembe P, Reed AW, Miller WC, Nkata E, Zimba D, Daly CC, Chakraborty H, Cohen MS, Hoffman I. 1999. Trichomonas vaginalis as a cause of urethritis in Malawian men. Sex. Transm. Dis. 26:381–387 [DOI] [PubMed] [Google Scholar]
- 5.Kissinger P, Amedee A, Clark RA, Dumestre J, Theall KP, Myers L, Hagensee ME, Farley TA, Martin DH. 2009. Trichomonas vaginalis treatment reduces vaginal HIV-1 shedding. Sex. Transm. Dis. 36:11–16 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 6.Heine P, McGregor JA. 1993. Trichomonas vaginalis: a reemerging pathogen. Clin. Obstet. Gynecol. 36:137–144 [DOI] [PubMed] [Google Scholar]
- 7.Huppert JS, Hesse E, Kim G, Kim M, Agreda P, Quinn N, Gaydos C. 2010. Adolescent women can perform a point-of-care test for trichomoniasis as accurately as clinicians. Sex. Transm. Infect. 86:514–519 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 8.Munson E, Napierala M, Olson R, Endes T, Block T, Hryciuk JE, Schell RF. 2008. Impact of Trichomonas vaginalis transcription-mediated amplification-based analyte-specific-reagent testing in a metropolitan setting of high sexually transmitted disease prevalence. J. Clin. Microbiol. 46:3368–3374 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 9.Nye MB, Schwebke JR, Body BA. 2009. Comparison of APTIMA Trichomonas vaginalis transcription-mediated amplification to wet mount microscopy, culture, and polymerase chain reaction for diagnosis of trichomoniasis in men and women. Am. J. Obstet. Gynecol. 200:188.e1-7. [DOI] [PubMed] [Google Scholar]
- 10.Napierala M, Munson E, Munson KL, Kramme T, Miller C, Burtch J, Olson R, Hryciuk JE. 2011. Three-year history of transcription-mediated amplification-based Trichomonas vaginalis analyte-specific reagent testing in a subacute care patient population. J. Clin. Microbiol. 49:4190–4194 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 11.Schwebke JR, Lawing LF. 2002. Improved detection by DNA amplification of Trichomonas vaginalis in males. J. Clin. Microbiol. 40:3681–3683 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 12.Wendel KA, Erbelding EJ, Gaydos CA, Rompalo AM. 2003. Use of urine polymerase chain reaction to define the prevalence and clinical presentation of Trichomonas vaginalis in men attending an STD clinic. Sex. Transm. Infect. 79:151–153 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 13.Miller WC, Swygard H, Hobbs MM, Ford CA, Handcock MS, Morris M, Schmitz JL, Cohen MS, Harris KM, Udry JR. 2005. The prevalence of trichomoniasis in young adults in the United States. Sex. Transm. Dis. 32:593–598 [DOI] [PubMed] [Google Scholar]
- 14.Huppert JS, Mortensen JE, Reed JL, Kahn JA, Rich KD, Miller WC, Hobbs MM. 2007. Rapid antigen testing compares favorably with transcription-mediated amplification assay for the detection of T. vaginalis in young women. Clin. Infect. Dis. 45:194–198 [DOI] [PubMed] [Google Scholar]
- 15.Centers for Disease Control and Prevention 2011. Sexually transmitted disease surveillance, 2010 US Department of Health and Human Services, Atlanta, GA [Google Scholar]
- 16.Hardick A, Hardick J, Wood BJ, Gaydos C. 2006. Comparison between the Gen-Probe transcription-mediated amplification Trichomonas vaginalis research assay and real-time PCR for Trichomonas vaginalis detection using a Roche LightCycler instrument with female self-obtained vaginal swab samples and male urine samples. J. Clin. Microbiol. 44:4197–4199 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 17.Gaydos CA, Quinn TC, Willis D, Weissfeld A, Hook EW, Martin DH, Ferrero DV, Schachter J. 2003. Performance of the APTIMA Combo 2 assay for detection of Chlamydia trachomatis and Neisseria gonorrhoeae in female urine and endocervical swab specimens. J. Clin. Microbiol. 41:304–309 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 18.Munson E, Miller C, Napierala M, Kramme T, Olson R, Munson KL, Olson S, Hryciuk JE, Schell RF. 2012. Assessment of screening practices in a subacute clinical setting following introduction of Trichomonas vaginalis nucleic acid amplification testing. Wisc. Med. J. 111:233–236 [PubMed] [Google Scholar]
- 19.Andrea SB, Chapin KC. 2011. Comparison of Aptima Trichomonas vaginalis transcription-mediated amplification assay and BD Affirm VPIII for detection of T. vaginalis in symptomatic women: performance parameters and epidemiological implications. J. Clin. Microbiol. 49:866–869 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 20.Munson E, Kramme T, Napierala M, Munson KL, Miller C, Hryciuk JE. 2012. Female epidemiology of transcription-mediated amplification-based Trichomonas vaginalis detection in a metropolitan setting with a high prevalence of sexually transmitted infection. J. Clin. Microbiol. 50:3927–3931 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 21.Munson E, Firmani MA. 2009. Molecular diagnosis of Neisseria gonorrhoeae infection in the United States. Expert Opin. Med. Diagn. 3:327–343 [DOI] [PubMed] [Google Scholar]
- 22.Munson E, Napierala M, Basile J, Miller C, Burtch J, Hryciuk JE, Schell RF. 2010. Trichomonas vaginalis transcription-mediated amplification-based analyte-specific reagent and alternative target testing of primary clinical vaginal saline suspensions. Diagn. Microbiol. Infect. Dis. 68:66–72 [DOI] [PubMed] [Google Scholar]
- 23.Krashin JW, Koumans EH, Bradshaw-Sydnor AC, Braxton JR, Evan Secor W, Sawyer MK, Markowitz LE. 2010. Trichomonas vaginalis prevalence, incidence, risk factors and antibiotic-resistance in an adolescent population. Sex. Transm. Dis. 37:440–444 [DOI] [PubMed] [Google Scholar]
- 24.Stark JR, Judson G, Alderete JF, Mundodi V, Kucknoor AS, Giovannucci EL, Platz EA, Sutcliffe S, Fall K, Kurth T, Ma J, Stampfer MJ, Mucci LA. 2009. Prospective study of T. vaginalis infection and prostate cancer incidence and mortality: physicians' health study. J. Natl. Cancer Inst. 101:1406–1411 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 25.Sutcliffe S, Giovannucci E, Alderete JF, Chang TH, Gaydos CA, Zenilman JM, De Marzo AM, Willett WC, Platz EA. 2006. Plasma antibodies against Trichomonas vaginalis and subsequent risk of prostate cancer. Cancer Epidemiol. Biomarkers Prev. 15:939–945 [DOI] [PubMed] [Google Scholar]
- 26.Sutcliffe S, Alderete JF, Till C, Goodman PJ, Hsing AW, Zenilman JM, De Marzo AM, Platz EA. 2009. Trichomonosis and subsequent risk of prostate cancer in the prostate cancer prevention trial. Int. J. Cancer 124:2082–2087 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 27.Schwebke JR, Hobbs MM, Taylor SN, Sena AC, Catania MG, Weinbaum BS, Johnson AD, Getman DK, Gaydos CA. 2011. Molecular testing for Trichomonas vaginalis in women: results from a prospective U.S. clinical trial. J. Clin. Microbiol. 49:4106–4111 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 28.Chernesky M, Jang D, Luinstra K, Chong S, Smieja M, Cai W, Hayhoe B, Portillo E, MacRitchie C, Main C, Ewert R. 2006. High analytical sensitivity and low rates of inhibition may contribute to detection of Chlamydia trachomatis in significantly more women by the APTIMA Combo 2 assay. J. Clin. Microbiol. 44:400–405 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 29.Ikeda-Dantsuji Y, Konomi I, Nagayama A. 2005. In vitro assessment of the APTIMA Combo 2 assay for the detection of Chlamydia trachomatis using highly purified elementary bodies. J. Med. Microbiol. 54:357–360 [DOI] [PubMed] [Google Scholar]
- 30.Schachter J, Chow JM, Howard H, Bolan G, Moncada J. 2006. Detection of Chlamydia trachomatis by nucleic acid amplification testing: our evaluation suggests that CDC-recommended approaches for confirmatory testing are ill-advised. J. Clin. Microbiol. 44:2512–2517 [DOI] [PMC free article] [PubMed] [Google Scholar]