Abstract
The population epidemiology of AKI is not well described. Here, we analyzed data from the Nationwide Inpatient Sample, a nationally representative dataset, to identify cases of dialysis-requiring AKI using validated International Classification of Diseases, Ninth Revision (ICD-9) codes. From 2000 to 2009, the incidence of dialysis-requiring AKI increased from 222 to 533 cases per million person-years, averaging a 10% increase per year (incidence rate ratio=1.10, 95% CI=1.10–1.11 per year). Older age, male sex, and black race associated with higher incidence of dialysis-requiring AKI. The rapid increase in incidence was evident in all age, sex, and race subgroups examined. Temporal changes in the population distribution of age, race, and sex as well as trends of sepsis, acute heart failure, and receipt of cardiac catheterization and mechanical ventilation accounted for about one third of the observed increase in dialysis-requiring AKI among hospitalized patients. The total number of deaths associated with dialysis-requiring AKI rose from 18,000 in 2000 to nearly 39,000 in 2009. In conclusion, the incidence of dialysis-requiring AKI increased rapidly in all patient subgroups in the past decade in the United States, and the number of deaths associated with dialysis-requiring AKI more than doubled.
AKI is one of the most common and serious complications of hospitalized patients.1 Those patients suffering the most severe form of AKI—requiring acute dialysis—have particularly high rates of adverse outcomes during hospitalization and after discharge.2–4 Despite the importance of AKI, there have been relatively few studies about its population epidemiology.
Prior studies have expressed AKI incidence as cases per hospitalization or per intensive care unit stay,4–6 making comparisons across regions and over time difficult to interpret, because thresholds for admission may vary. Few studies have studied AKI incidence as case per underlying population, and recent temporal trends in the US population incidence are not well defined.
In this study, we used a nationally representative sample of hospitalizations to estimate the US population incidence of dialysis-requiring AKI and identify specific demographic subgroups with increased propensity for dialysis-requiring AKI. We explored whether the change in disease burden is associated with changing demographics or secular trends in conditions or interventions that are known to predispose to AKI.
From 2000 to 2009, there were 1.09 million hospitalizations (95% confidence interval [CI]=1.04–1.15 million) with dialysis-requiring AKI in the United States (patient characteristics are shown in Supplemental Table 1). Hospitalized patients with dialysis-requiring AKI were older than their counterparts without dialysis-requiring AKI (63.4 versus 47.6 years), were more likely to be male (57.3% versus 41.1%), were more likely to be black (15.6% versus 10.2%), were more likely to have sepsis (27.7% versus 2.6%), were more likely to have heart failure (6.2% versus 2.7%), and were more likely to undergo cardiac catheterization (5.2% versus 4.4%) and mechanical ventilation (29.9% versus 2.4%).
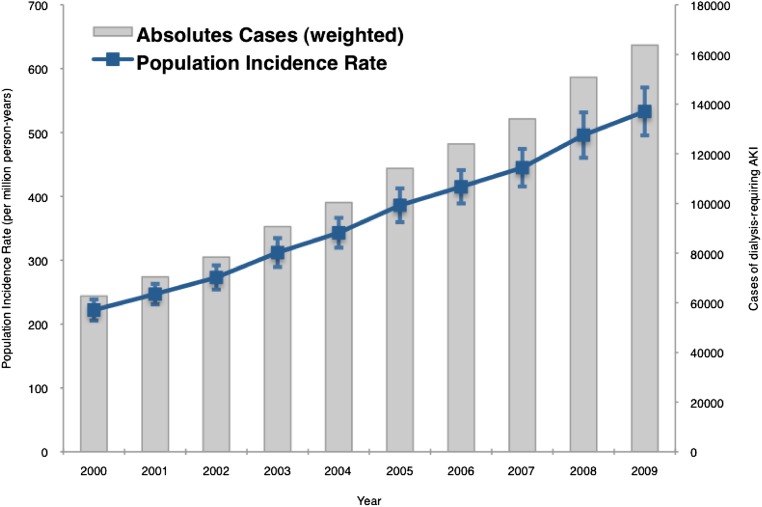
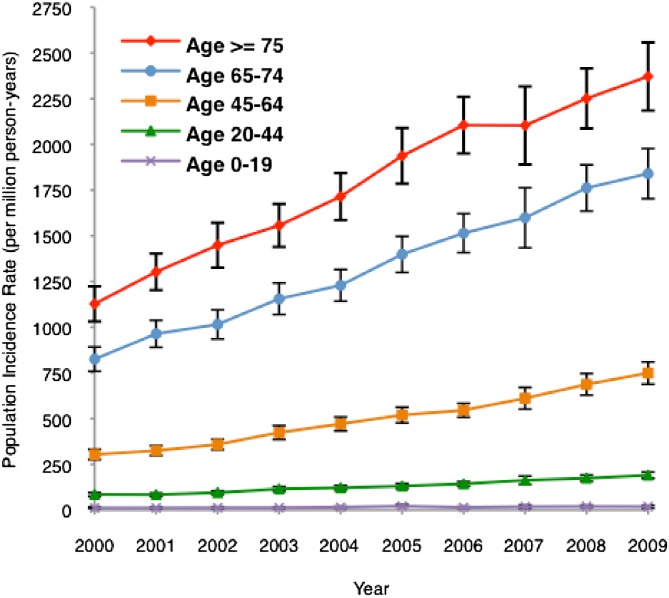
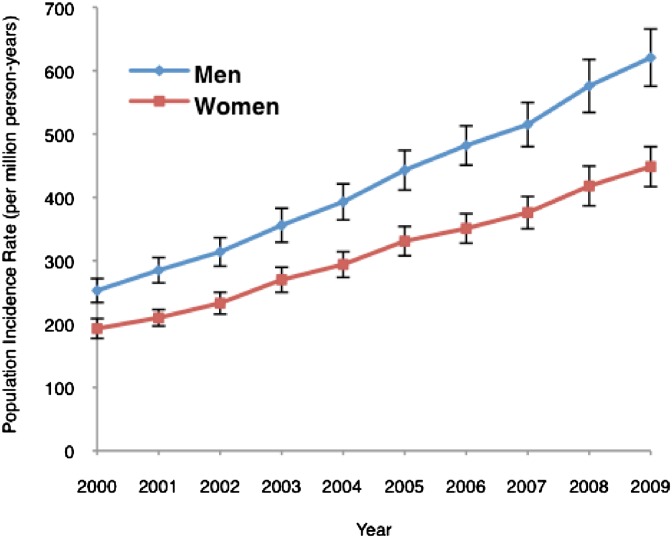
The number of hospitalizations with dialysis-requiring AKI increased from 63,000 cases in 2000 to almost 164,000 cases in 2009 (Figure 1). The population incidence rate of dialysis-requiring AKI increased from 222 to 533 cases/million person-years, a yearly increase of 10% per year (incidence rate ratio=1.10, 95% CI=1.10–1.11 per year). In subgroup analyses, we found that dialysis-requiring AKI was more common in the elderly (versus younger age groups), men (versus women), and non-Hispanic blacks (versus non-Hispanic whites). All age, sex, and racial–ethnic subgroups showed a similar pattern of increase in disease incidence over the study period (Figures 2 and 3 and Supplemental Figure 1).
Figure 1.
Population incidence of dialysis-requiring AKI in the United States from 2000 to 2009 (absolute count and incidence rate per million person-years). I bars represent 95% CIs for incidence rates. The number of cases of dialysis-requiring AKI increased from 63,000 in 2000 to almost 164,000 in 2009; the population incidence increased at 10% per year from 222 to 533 cases/million person-years.
Figure 2.
Population incidence of dialysis-requiring AKI in the United States by age groups from 2000 to 2009. I bars represent 95% CIs for incidence rates. All age groups showed a similar pattern of increase in incidence.
Figure 3.
Population incidence of dialysis-requiring AKI in the United States by sex from 2000 to 2009. I bars represent 95% CIs for incidence rates. Both groups showed a similar pattern of increase in incidence.
The inpatient case fatality rate associated with a single episode of dialysis-requiring AKI declined from 29.1% in 2000 to 23.5% in 2009. However, owing to the rapid increase in the absolute number of cases, the total number of inpatient deaths associated with dialysis-requiring AKI more than doubled from 18,000 in 2000 to almost 39,000 in 2009.
In univariate logistic regression, the odds of developing dialysis-requiring AKI among all hospitalized patients increased annually by 10.1% (odds ratio [OR]=1.10, 95% CI=1.09–1.11, P<0.001). Adjustment for demographics, sepsis, acute heart failure, cardiac catheterization, and mechanical ventilation reduced the OR per year to 1.07 (95% CI=1.06–1.07, P<0.001) (Table 1). Therefore, changing demographics, increased incidence of sepsis and acute heart failure, or increased use of cardiac catheterization and mechanical ventilation accounted for about one third of the increased risk of dialysis-requiring AKI among hospitalized patients over time. Similar results were observed in all subgroups examined (Table 1).
Table 1.
Temporal changes in risk of dialysis-requiring AKI among hospitalized patients in the United States from 2000 to 2009
| Population at Risk (2000–2009) | Crude OR/yr (95% CI) | Model 1a Adjusted OR/yr (95% CI) | Model 2b Adjusted OR/yr (95% CI) |
|---|---|---|---|
| Overall | 1.10 (1.09–1.11) | 1.09 (1.08–1.10) | 1.07 (1.06–1.07) |
| Age strata (yr) | |||
| 0–19 | 1.08 | 1.08 | 1.07 |
| 20–44 | 1.10 | 1.10 | 1.08 |
| 45–64 | 1.10 | 1.10 | 1.06 |
| 65–74 | 1.11 | 1.10 | 1.08 |
| ≥75 | 1.08 | 1.08 | 1.06 |
| Sex | |||
| Male | 1.10 | 1.10 | 1.07 |
| Female | 1.10 | 1.09 | 1.06 |
Note that all models for trend analysis showed P<0.001.
Adjusted for age, sex, and race.
Adjusted for age, sex, race, sepsis, acute heart failure, cardiac catheterization, and mechanical ventilation.
In summary, we found that population incidence of dialysis-requiring AKI has increased rapidly at 10% annually using a nationally representative sample of hospitalizations. Our findings are consistent with and extend on older studies showing an increase in disease incidence in the United States over time.7,8 There have also been scattered reports of rising population incidence of AKI treated with renal replacement in assorted regions around the world.9 These prior studies did not include more recent years, analyze important subgroup differences, or explore potential reasons for the temporal trend.
Using a nationally representative sample, we quantified the increased propensity for dialysis-requiring AKI with age.
We found increased risk of dialysis-requiring AKI among men. Prior studies have reported men to have higher,4,5 same,10 or lower11 risk of AKI than women. Across the spectrum of renal disease, sex disparities are also not uniform. Men are more likely than women to develop incident ESRD, but men do not have higher prevalence of CKD than women.12,13 Some animal models showed that differences in sex hormone production increase male susceptibility to ischemic renal injury.14
We found that non-Hispanic blacks have a higher incidence of dialysis-requiring AKI compared with non-Hispanic whites. Although racial–ethnic disparities in ESRD have been well established,13 racial differences in AKI are less well defined. Some4,5 but not all6 prior studies have shown that blacks are at higher risk for AKI. Blacks do not seem to have higher CKD prevalence than whites.12 Recently discovered genetic polymorphisms have been postulated to explain much of the black–white difference in the risk of ESRD, but whether the same genetic factors play a role in susceptibility to AKI has not been explored.15
Our findings highlight the public health importance of AKI. Traditionally, much of the focus on kidney disease epidemiology had been on ESRD. This past decade has also witnessed considerable growth in CKD epidemiology, allowing us to identify a large vulnerable population at increased risk for ESRD and cardiovascular disease.12,16 Driven by these data, policy discussions related to kidney disease have mostly addressed ESRD and CKD.17,18
Our findings underscore the importance of AKI in the overall burden of kidney disease across its spectrum. The incidence of dialysis-requiring AKI in the United States is now higher than the incidence of ESRD, which has increased only modestly over the past several years (from 354 cases/million person-years in 2005 to 371 cases/million person-years in 2009).13 Because the number of nondialysis-requiring AKI cases is approximately 10-fold higher than the number of dialysis-requiring AKI cases19 and because even small acute changes in serum creatinine are associated with increased morbidity and mortality,20 our data likely represent only the tip of the iceberg in terms of the public health burden of AKI.
We were unable to entirely explain the rising incidence of dialysis-requiring AKI. It is plausible that factors not captured by our analyses are responsible, such as contrast procedures performed outside of the cardiac catheterization laboratory, because radiology procedures, such as computed tomography scans, could not be reliably captured by discharge codes. We did not include preexisting conditions, such as CKD and diabetes mellitus, as predictors, because discharge codes for such chronic conditions may have limited accuracy.21,22 In addition, it is doubtful that temporal trends in CKD prevalence could explain the observed increase in dialysis-requiring AKI. Although increases in CKD prevalence have been reported previously, those studies are confounded by calibration problems with serum creatinine.12 When renal function was assessed using cystatin C, there was scant evidence of secular increase in CKD prevalence.23 Even the most liberal creatinine-based estimates put the increase in CKD prevalence at 3% annually,12,24 much smaller than the 10% annual increase in AKI incidence that we observed.
Another potential explanation for our observations is more liberal use of acute dialysis over time. To our knowledge, only one prior study has directly measured, in the same population, the community incidence of both dialysis-requiring and nondialysis-requiring AKI when the latter was defined using actual observed inpatient serum creatinine measurements. A parallel increase in the population incidence of both dialysis-requiring and nondialysis-requiring AKI was seen from 1996 to 2003 (the former from 195 to 295 cases/million person-years, and the latter from 3227 to 5224 cases/million person-years).7 This finding argues strongly that earlier and more liberal use of acute dialysis is not a major contributor to the observed trend.
Those data are also inconsistent with the hypothesis that our results are caused by a code creep artifact. Although diagnostic codes for AKI without specifying dialysis may be subject to increased coding over time (because of increased disease awareness or changes in consensus definitions), procedure codes for acute dialysis are unlikely to be inflated, because they are tied to reimbursement.
Our study has several strengths, most notably the use of a large nationally representative sample, which allowed for accurate estimates of national incidence rates, excellent power, and generalizability. We also recognize several limitations, the primary limitation being the reliance on administrative data without clinical data, such as serum creatinine. We relied on the best validated set of diagnostic and procedure codes to define our primary outcome of dialysis-requiring AKI. Although the performance characteristics of AKI diagnostic codes vary in different reports,21 codes for dialysis-requiring AKI have better validity than codes for nondialysis-requiring AKI.25 Hence, we decided not to examine the latter as an outcome in our study. Administrative codes also did not allow us to discriminate between continuous versus intermittent renal replacement therapy and de novo AKI versus acute-on-chronic renal disease. Because data were completely deidentified, we could not identify the individuals who had recurrent episodes of dialysis-requiring AKI, although this occurrence is a very uncommon.
Accurately defining the population incidence of AKI allows the medical and lay communities to appreciate the true public health burden of AKI. More research is needed to address reasons for underlying disparities among sex, age, and racial groups and causes behind the rapid increase in the incidence of dialysis-requiring AKI.
Concise Methods
We extracted 2000–2009 data from the Nationwide Inpatient Sample (NIS), a US nationally representative administrative database of hospitalizations.26 The NIS is the largest all-payer publicly available national hospital database containing a 20% stratified sample of all short-term, nonfederal, nonrehabilitation hospitals. Hospitals are sampled according to geographic region, location (urban/rural), teaching status, ownership, and bed size; all discharges from the sampled hospitals are included in the database. Each data entry represents an individual hospitalization and includes demographic variables, discharge diagnoses, procedures, hospital charges, insurance payers, and length of stay. The NIS was developed as part of the Healthcare Cost and Utilization Project sponsored by the Agency for Healthcare Research and Quality. Because of the sampling strategy, NIS data can be reinflated with appropriate weights (provided by Healthcare Cost and Utilization Project) to represent hospitalizations at a national level. NIS has been used extensively to study national trends in hospital care. The Committee on Human Research of the University of California, San Francisco approved the study protocol.
Cases of dialysis-requiring AKI were identified using validated International Classification of Diseases, Ninth Revision, Clinical Modification codes.25 Dialysis-requiring AKI was defined as the presence of any of the following diagnostic codes (in any position): 584.5 (acute renal failure [ARF] with lesion of tubular necrosis), 584.6 (ARF with lesion of renal cortical necrosis), 584.7 (ARF with lesion of renal medullary necrosis), 584.8 (ARF with other specified pathologic lesion in kidney), or 584.9 (ARF unspecified); and the presence of any of the following procedure codes (in any position): 39.95 (hemodialysis), V45.1 (renal dialysis status), V56.0 (extracorporeal dialysis), or V56.1 (fitting and adjustment of extracorporeal dialysis catheter). To avoid inclusion of patients admitted for initiation of chronic maintenance hemodialysis, we excluded the cases with procedure codes for arteriovenous fistula creation or revision (39.27, 39.42, 39.43, and 39.93). This algorithm has been shown to produce high sensitivity, specificity, and positive and negative predictive values (all ≥90%).25
Data were analyzed using Stata/SE 11.2. We estimated the total (weighted) number of hospitalizations with dialysis-requiring AKI for each calendar year, applying appropriate discharge-level sampling weights to account for the NIS sampling scheme. We divided the number of discharges with dialysis-requiring AKI by the total population (obtained from the US Census Bureau) to calculate population incidence rates. All results presented are weighted estimates unless otherwise specified.
Subgroup analyses for age and sex subgroups were performed in a similar fashion, normalizing the denominator to the corresponding subgroup population totals from the US Census Bureau. Because the NIS includes race–ethnicity in mutually exclusive categories of white, black, and Hispanic, we used only non-Hispanic white and non-Hispanic black population estimates as the population denominators. Owing to the large number of states with missing race data, especially earlier in our study period, we undertook two approaches to estimate race-specific incidence rates. First, we used all available data in the NIS and race-specific population estimates from the entire US population. This data use would likely underestimate disease incidence, because the numerator, but not the denominator, had missing data. Second, we performed an analysis using only data from the 16 states that provided complete race data from 2000 to 2009 (California, Colorado, Connecticut, Florida, Hawaii, Iowa, Kansas, Maryland, Massachusetts, Missouri, New Jersey, New York, South Carolina, Tennessee, Utah, and Wisconsin). These incidence rates would not be expected to be nationally representative.
Estimating the percent yearly increase in nationally estimated rates required use of aggregated census data, from which we derived CIs and SEMs for the yearly national rates. To estimate the yearly increase, we fit a generalized linear model with a log link, with weights inversely proportional to the variability of the estimated yearly rates.
To explore potential reasons for temporal changes in risk of dialysis-requiring AKI among hospitalized patients, we created a logistic regression model using dialysis-requiring AKI as the outcome and calendar year as the predictor. Given the large number of patient-level observations in the NIS (5–8 million for each year), we took a random 10% subsample of each year's data to facilitate regression analysis, because the analysis required all 10 years of data to be aggregated.
Additional patient level covariates were then entered into the model to determine the degree that they explained temporal trends. We analyzed four a priori selected conditions and interventions that are known AKI risk factors to study, because these data have become more common in recent years: sepsis, acute heart failure, cardiac catheterization, and mechanical ventilation.27–29 Sepsis was defined by the presence of any of the following diagnostic codes: 038.x (septicemia), 020.0 (septicemic), 790.7 (bacteremia), 117.9 (disseminated fungal infection), 112.5 (disseminated Candida infection), or 112.81 (disseminated fungal endocarditis).27 Acute heart failure was defined by the presence of 428.x as the primary diagnosis.30 Cardiac catheterizations were captured by any of the following procedure codes: 36.0x (removal of coronary artery obstruction and insertion of stents), 00.66 (percutaneous transluminal coronary angioplasty or coronary atherectomy), 37.22 (left heart cardiac catheterization), 37.23 (combined right and left heart catheterization), or 88.53–57 (angiography using contrast material).28 Mechanical ventilation was defined by the presence of procedure code 96.7x (mechanical ventilation).29
Disclosures
None.
Supplementary Material
Acknowledgments
This study was supported by National Institute of Diabetes and Digestive and Kidney Diseases of the National Institutes of Health Grants F32DK093212 (to R.K.H.), T32DK07219 (to R.K.H.), and K24DK92291 (to C.-y.H.).
The opinions, results, and conclusions reported in this paper are the authors, and they are independent from the funding source.
Footnotes
Published online ahead of print. Publication date available at www.jasn.org.
See related editorial, “The Inexorable Rise of AKI: Can We Bend the Growth Curve?,” on pages 3–5.
This article contains supplemental material online at http://jasn.asnjournals.org/lookup/suppl/doi:10.1681/ASN.2012080800/-/DCSupplemental.
References
- 1.Lameire N, Van Biesen W, Vanholder R: Acute kidney injury. Lancet 372: 1863–1865, 2008 [DOI] [PubMed] [Google Scholar]
- 2.Hsu CY, Chertow GM, McCulloch CE, Fan D, Ordoñez JD, Go AS: Nonrecovery of kidney function and death after acute on chronic renal failure. Clin J Am Soc Nephrol 4: 891–898, 2009 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 3.Lo LJ, Go AS, Chertow GM, McCulloch CE, Fan D, Ordoñez JD, Hsu CY: Dialysis-requiring acute renal failure increases the risk of progressive chronic kidney disease. Kidney Int 76: 893–899, 2009 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 4.Liangos O, Wald R, O’Bell JW, Price L, Pereira BJ, Jaber BL: Epidemiology and outcomes of acute renal failure in hospitalized patients: A national survey. Clin J Am Soc Nephrol 1: 43–51, 2006 [DOI] [PubMed] [Google Scholar]
- 5.Xue JL, Daniels F, Star RA, Kimmel PL, Eggers PW, Molitoris BA, Himmelfarb J, Collins AJ: Incidence and mortality of acute renal failure in Medicare beneficiaries, 1992 to 2001. J Am Soc Nephrol 17: 1135–1142, 2006 [DOI] [PubMed] [Google Scholar]
- 6.Nash K, Hafeez A, Hou S: Hospital-acquired renal insufficiency. Am J Kidney Dis 39: 930–936, 2002 [DOI] [PubMed] [Google Scholar]
- 7.Hsu CY, McCulloch CE, Fan D, Ordoñez JD, Chertow GM, Go AS: Community-based incidence of acute renal failure. Kidney Int 72: 208–212, 2007 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 8.Waikar SS, Curhan GC, Wald R, McCarthy EP, Chertow GM: Declining mortality in patients with acute renal failure, 1988 to 2002. J Am Soc Nephrol 17: 1143–1150, 2006 [DOI] [PubMed] [Google Scholar]
- 9.Hoste EA, Schurgers M: Epidemiology of acute kidney injury: How big is the problem? Crit Care Med 36[Suppl]: S146–S151, 2008 [DOI] [PubMed] [Google Scholar]
- 10.Bagshaw SM, George C, Bellomo R, ANZICS Database Management Committee : Changes in the incidence and outcome for early acute kidney injury in a cohort of Australian intensive care units. Crit Care 11: R68, 2007 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 11.Thakar CV, Liangos O, Yared JP, Nelson D, Piedmonte MR, Hariachar S, Paganini EP: ARF after open-heart surgery: Influence of gender and race. Am J Kidney Dis 41: 742–751, 2003 [DOI] [PubMed] [Google Scholar]
- 12.Coresh J, Selvin E, Stevens LA, Manzi J, Kusek JW, Eggers P, Van Lente F, Levey AS: Prevalence of chronic kidney disease in the United States. JAMA 298: 2038–2047, 2007 [DOI] [PubMed] [Google Scholar]
- 13.Collins AJ, Foley RN, Chavers B, Gilbertson D, Herzog C, Johansen K, Kasiske B, Kutner N, Liu J, St Peter W, Guo H, Gustafson S, Heubner B, Lamb K, Li S, Li S, Peng Y, Qiu Y, Roberts T, Skeans M, Snyder J, Solid C, Thompson B, Wang C, Weinhandl E, Zaun D, Arko C, Chen SC, Daniels F, Ebben J, Frazier E, Hanzlik C, Johnson R, Sheets D, Wang X, Forrest B, Constantini E, Everson S, Eggers P, Agodoa L: United States Renal Data System 2011 Annual Data Report: Atlas of chronic kidney disease and end-stage renal disease in the United States. Am J Kidney Dis 59[Suppl 1]: A7, e1–420, 2012 [DOI] [PubMed] [Google Scholar]
- 14.Robert R, Ghazali DA, Favreau F, Mauco G, Hauet T, Goujon JM: Gender difference and sex hormone production in rodent renal ischemia reperfusion injury and repair. J Inflamm (Lond) 8: 14, 2011 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 15.Tzur S, Rosset S, Shemer R, Yudkovsky G, Selig S, Tarekegn A, Bekele E, Bradman N, Wasser WG, Behar DM, Skorecki K: Missense mutations in the APOL1 gene are highly associated with end stage kidney disease risk previously attributed to the MYH9 gene. Hum Genet 128: 345–350, 2010 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 16.Go AS, Chertow GM, Fan D, McCulloch CE, Hsu CY: Chronic kidney disease and the risks of death, cardiovascular events, and hospitalization. N Engl J Med 351: 1296–1305, 2004 [DOI] [PubMed] [Google Scholar]
- 17.US Department of Health and Human Services : Healthy People 2010, Atlanta, GA, CDC Office of Disease Prevention and Health Promotion, 2000 [Google Scholar]
- 18.Couser WG, Riella MC, Joint International Society of Nephrology and International Federation of Kidney Foundations World Kidney Day 2011 Steering Committee : World Kidney Day 2011: Protect your kidneys, save your heart. Clin J Am Soc Nephrol 6: 235–238, 2011 [DOI] [PubMed] [Google Scholar]
- 19.James MT, Hemmelgarn BR, Wiebe N, Pannu N, Manns BJ, Klarenbach SW, Tonelli M, Alberta Kidney Disease Network : Glomerular filtration rate, proteinuria, and the incidence and consequences of acute kidney injury: A cohort study. Lancet 376: 2096–2103, 2010 [DOI] [PubMed] [Google Scholar]
- 20.Chertow GM, Burdick E, Honour M, Bonventre JV, Bates DW: Acute kidney injury, mortality, length of stay, and costs in hospitalized patients. J Am Soc Nephrol 16: 3365–3370, 2005 [DOI] [PubMed] [Google Scholar]
- 21.Grams ME, Plantinga LC, Hedgeman E, Saran R, Myers GL, Williams DE, Powe NR, CDC CKD Surveillance Team : Validation of CKD and related conditions in existing data sets: A systematic review. Am J Kidney Dis 57: 44–54, 2011 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 22.Quan H, Parsons GA, Ghali WA: Validity of information on comorbidity derived rom ICD-9-CCM administrative data. Med Care 40: 675–685, 2002 [DOI] [PubMed] [Google Scholar]
- 23.Foley RN, Wang C, Snyder JJ, Collins AJ: Cystatin C levels in U.S. adults, 1988-1994 versus 1999-2002: NHANES. Clin J Am Soc Nephrol 4: 965–972, 2009 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 24.Hsu CY: Where is the epidemic in kidney disease? J Am Soc Nephrol 21: 1607–1611, 2010 [DOI] [PubMed] [Google Scholar]
- 25.Waikar SS, Wald R, Chertow GM, Curhan GC, Winkelmayer WC, Liangos O, Sosa MA, Jaber BL: Validity of International Classification of Diseases, Ninth Revision, Clinical Modification Codes for Acute Renal Failure. J Am Soc Nephrol 17: 1688–1694, 2006 [DOI] [PubMed] [Google Scholar]
- 26.Healthcare Cost and Utilization Project (HCUP) : HCUP Nationwide Inpatient Sample (NIS) 2000–2009, Rockville, MD, Agency for Healthcare Research and Quality; [PubMed] [Google Scholar]
- 27.Martin GS, Mannino DM, Eaton S, Moss M: The epidemiology of sepsis in the United States from 1979 through 2000. N Engl J Med 348: 1546–1554, 2003 [DOI] [PubMed] [Google Scholar]
- 28.Lloyd-Jones D, Adams RJ, Brown TM, Carnethon M, Dai S, De Simone G, Ferguson TB, Ford E, Furie K, Gillespie C, Go A, Greenlund K, Haase N, Hailpern S, Ho PM, Howard V, Kissela B, Kittner S, Lackland D, Lisabeth L, Marelli A, McDermott MM, Meigs J, Mozaffarian D, Mussolino M, Nichol G, Roger VL, Rosamond W, Sacco R, Sorlie P, Stafford R, Thom T, Wasserthiel-Smoller S, Wong ND, Wylie-Rosett J, American Heart Association Statistics Committee and Stroke Statistics Subcommittee : Executive summary: Heart disease and stroke statistics—2010 update: A report from the American Heart Association. Circulation 121: 948–954, 2010 [DOI] [PubMed] [Google Scholar]
- 29.Wunsch H, Linde-Zwirble WT, Angus DC, Hartman ME, Milbrandt EB, Kahn JM: The epidemiology of mechanical ventilation use in the United States. Crit Care Med 38: 1947–1953, 2010 [DOI] [PubMed] [Google Scholar]
- 30.Lee DS, Donovan L, Austin PC, Gong Y, Liu PP, Rouleau JL, Tu JV: Comparison of coding of heart failure and comorbidities in administrative and clinical data for use in outcomes research. Med Care 43: 182–188, 2005 [DOI] [PubMed] [Google Scholar]
Associated Data
This section collects any data citations, data availability statements, or supplementary materials included in this article.