Abstract
People experience the feeling of the missing body part long after it has been removed after amputation are known as phantom limb sensations. These sensations can be painful, sometimes becoming chronic and lasting for several years (or called phantom pain). Medical treatment for these individuals is limited. Recent neurobiological investigations of brain plasticity after amputation have revealed new insights into the changes in the brain that may cause phantom limb sensations and phantom pain. In this article, I review recent progresses of the cortical plasticity in the anterior cingulate cortex (ACC), a critical cortical area for pain sensation, and explore how they are related to abnormal sensory sensations such as phantom pain. An understanding of these alterations may guide future research into medical treatment for these disorders.
Keywords: anterior cingulate cortex, long-term potentiation, long-term depression, phantom pain, amputation, mice
INTRODUCTION
It is well known that adult somatosensory cortex is dynamic and plastic [1-4]. Cortical anatomic connections and functional representations can be modified by experience, and some of these modification can occur within a very short amount of time. At the synaptic level, it has been believed that use-dependent changes in synaptic strength, such as long-term potentiation (LTP) and long-term depression (LTD) may serve as key synaptic mechanisms of cortical plasticity [4-7].
Cortical changes not only occur during development and normal physiological conditions, but also under pathological conditions, such as tissue/nerve injury or the loss of a limb [see 8]. For example, it has been demonstrated that cortical reorganization occurs after limb or digit amputation [9-13]. Most human amputees experience phantom limb sensation or phantom pain [see 14 for review]. However, it is unclear if cortical reorganization directly contributes to phantom sensations and/or phantom pain. Furthermore, the molecular and cellular mechanisms contributing to amputation related cortical reorganization are still largely unknown. In this review, I shall focus on the ACC, a critical forebrain area for pain and cognition, and explore synaptic plastic changes after amputation, under both in vitro and in vivo conditions.
ACC AND NOCICEPTION/PAIN
The ACC forms a large region around the rostrum of the corpus callosum and is involved in emotional and attentive responses to internal and external stimulation [15, 16]. Neuroimaging and electrophysiological studies in humans have shown that somatosensory stimuli, including those causing pain, activate ACC neurons and other related limbic areas [17-21, see 4 for reviews]. Electrophysiological recordings demonstrate that ACC neurons respond to peripheral nociceptive stimulation in animals [22, 23]. Behavioral experiments in both rats and mice show that lesions of the medial frontal cortex, which includes the ACC, significantly reduces sensitivity to noxious heat applied to the hindpaw in the hot-plate test [24, 25], whereas electrical and chemical stimulation of regions within the ACC facilitates behavioral responses to noxious heat in the tail-flick test [26]. Consistent with these animal studies, the unpleasantness of pain is abolished in patients with frontal lobotomies or cingulotomies [see 4 for review]. As compared with sensory neurons in the spinal cord, ACC neurons show receptive fields receiving input from almost any part of the body surface [see 21 for review]. Furthermore, most ACC neurons are polymodal (e.g., responding to both non-noxious and noxious stimuli). In vivo recording from human patients show that some ACC neurons exhibit bilateral responses to noxious stimuli, although some of them show evidence of restricted receptive fields [19]. In monkeys, it has also been reported that some nociceptive ACC neurons were activated during anticipation of pain [22]. Recently, in adult mice, we performed in vivo whole-cell patch-clamp recordings from pyramidal neurons in adult mice ACC under urethane anesthetized conditions [27]. We found that peripheral noxious pinch stimuli induced evoked spike responses in all three types of ACC neurons, showing that ACC neurons are indeed nociceptive. Moreover, direct electrical stimulation of the ACC in freely moving mice generated fear memory, providing strong evidence that activation of the ACC neurons indeed cause fear or unpleasant feelings in animals [28].
IMAGING OF PHANTOM PAIN IN THE BRAIN
Human imaging studies have provided powerful tool for studying cortical involvement in chronic pain including phantom pain, since it is impossible to measure phantom pain in animals directly [see 14, 29 for reviews]. It has been reported that functional and anatomic changes in sensory and motor cortices are related to phantom pain [see 14 for review], although such imaging method may not be useful for detecting plastic change at synaptic level. The precise functional contribution of these cortical changes to phantom pain is still unclear. Stimulation of motor cortex where amputation also triggered long-term plastic changes has been reported to relief phantom pain [see 30 for review]. It is possible that some of these changes may even take place in local inhibitory circuits; and thus activation of these inhibitory circuits can help to reduce pain by indirectly affecting pain related cortical areas such as ACC and insular cortex. Alternatively, they may affect endogenous descending inhibitory systems to produce analgesic effects [4].
CORTICAL REORGANIZATION AND PHANTOM PAIN
Cortical reorganization within somatosensory systems following amputation has been reported in both animals and humans [see 31 for review]. For example, the representation of the forepaw in the primary somatosensory cortex of a raccoon that had lost a forearm had been completely reactivated by an expanded representation of the stump. Furthermore, evidence from anatomic, electrophysiological and behavioral approaches consistently indicates that extensive cortical changes occur in the adult brain after amputation [31]. Not only do such changes occur at cortical levels, but long-lasting changes could also occur at subcortical structures, including the thalamus and brainstem nuclei [31, 32]. For example, in the thalamus, it has been reported that neurons that normally respond to stimuli on the missing limb become responsive to touch on the stump of the missing limb [33]. It is proposed that the growth of new connections at both subcortical and cortical levels of the central nervous system, in combination with changes in connectional synaptic strengths at the same levels, mediate most reorganization in the cortex [9, 31]. Furthermore, it has been reported that there is a possible link between cortical reorganization and phantom pain. Flor et al. reported in human studies that the amount of cortical reorganization correlates with the extent of phantom pain [34]. More interestingly, recent studies using animal models of neuropathic pain showed that nerve injury (which can be caused by amputation as well) triggered long-lasting synaptic structural changes within the ACC or related prefrontal cortical areas [35-38].
DIGIT OR TAIL AMPUTATION AS ANIMAL MODEL FOR STUDYING BRAIN CHANGES
Due to the difficulty in using animal models for the study of phantom pain, one alternative approaches is to investigate possible synaptic changes in cortical areas that are important for pain perception after amputation or nerve injury. Recent studies using animal studies with distal tail or single digit amputation models revealed that amputation caused prolonged neuronal activation and rapid expression of three immediate early genes in the cortex, including the ACC and hippocampus [39-42]. Interestingly, changes in synaptic plasticity, such as LTP and LTD, have also been reported in both brain slices from amputated animals and recordings in vivo [39, 40, 43, 44]. In vivo intracellular recordings from anesthetized mice showed that peripheral digit amputation caused long-lasting changes in neuronal excitability. These findings provide strong physiological evidence that synaptic connections in the brain can be altered after amputation. While it is too early to speculate on the behavioral correlates of these synaptic changes, long-term changes in behavioral responses to noxious stimuli had been reported [41].
CORTICAL SYNAPTIC TRANSMISSION
Glutamate is the major fast excitatory transmitter in the ACC [39, 45]. Bath application of 6-cyano-7-nitroquinoxaline-2,3-dione (CNQX) completely abolishes fast excitatory postsynaptic currents (EPSCs) recorded in the ACC neurons. In addition to the alpha-amino-3-hydroxy-5-methyl-4-isoxazolepropionic acid (AMPA) receptor, postsynaptic glutamate kainate (KA) receptors contribute to fast excitatory synaptic transmission in the ACC. Single-focal stimulation could induce small KA receptor-mediated EPSCs in the presence of a selective AMPA receptor antagonist, GYKI 53655. Genetic deletion of the KA GluR6 and GluR5 subunits completely abolished KA EPSCs and KA-activated currents [45].
Gamma-aminobutyric acid (GABA) is the major inhibitory transmitter in the ACC and IC. The inhibitory postsynaptic currents (IPSCs) are mainly mediated by postsynaptic GABAA receptors [46] Bath application of picrotoxin completely abolishes spontaneous IPSCs and evoked IPSCs. GABAB receptors are also found in the ACC neurons, although the role of GABAB remains to be investigated. There are few studies that investigate the modulation of inhibitory transmission in the ACC. A recent study using KA knockout mice reported that inhibitory transmission in the ACC is under tonic modulation of KA GluR5 receptor [46].
CORTICAL DEPRESSION AND POTENTIATION
LTD and LTP are two major forms of synaptic plasticity in the central synapses. Both LTD and LTP have been reported in ACC synapses. For LTD, at least two forms of LTD have been reported: NMDA receptor-dependent and -independent LTD [47]. ACC LTD induced by presynaptic stimulation with postsynaptic depolarization is NMDA receptor-dependent. However, NMDA receptor-independent LTD has been reported using field potential recordings from adult ACC slices [39, 44]. Thus, it is likely that different induction protocols result in different forms of ACC LTD. Both NMDA receptor NR2A and NR2B contribute to ACC LTD [48 for review]. Paired-pulse facilitation (PPF) is not changed during LTD in the ACC [45], supporting the idea that induction of LTD may depend on postsynaptic mechanisms. Consistently, ACC LTD was abolished in GluR2 knockout mice [47].
While early studies failed to report robust LTP in the ACC neurons using tetanic stimulation, theta burst stimulation (TBS) and other LTP induction protocols induced reliable LTP in ACC synapses of adult animals [see 4, 7 for review] Genetic, pharmacological and electrophysiological approaches have been used to investigate the basic mechanisms for LTP in the ACC synapses. Different stimulation protocols can be used for inducing LTP in the ACC pyramidal cells. Pairing training protocol (synaptic activity paired with postsynaptic depolarization), the spike-excitatory postsynaptic potential (EPSP) pairing protocol, and TBS protocol all induce LTP in the ACC pyramidal neurons [49]. Unlike the field recordings induced by TBS, LTP induced by the pairing protocol is mainly triggered by the activation of NMDA receptors but not L-type voltage-gated calcium channels (L-VGCCs) [50]. Bath application of NR2A antagonist NVP-AAM077 and NR2B antagonist ifenprodil/Ro 25-6981 almost completely blocked NMDA receptor-mediated EPSCs as well as LTP. By contrast, the NR2A or NR2B antagonist alone only reduced LTP [49]. Activation of NMDA receptors leads to an increase in postsynaptic Ca2+ in dendritic spines. Ca2+ serves as an important intracellular signal for triggering a series of biochemical events that contribute to the expression of LTP. Ca2+ binds to calmodulin (CaM) and leads to activation of calcium-stimulated signaling pathways.
Ca2+-stimulated, neuron-specific adenylyl cyclase subtype 1 (AC1) is highly expressed in the ACC neurons, and LTP induced by TBS or pairing stimulation are abolished in AC1 knockout mice [50]. The importance of AC1 activity in the ACC LTP has been further confirmed by the use of a selective AC1 inhibitor [51]. Several other signaling proteins or protein kinases are found to be involved in ACC LTP, including Ca2+-calmodulin-dependent protein kinase IV (CaMKIV), early growth response gene 1 (egr1), mitogen-activated protein kinase (MAP kinase) and fragile X mental retardation protein (FMRP) [4]. We have recently investigated the roles of GluR1 and GluR2/3 using genetic and pharmacological approaches. We found that GluR1 subunit C-terminal peptide analog, Pep1-TGL, blocked the induction of ACC LTP. Thus, in the ACC, the interaction between the C-terminus of GluR1 and PDZ domain proteins is required for the induction of LTP. Synaptic delivery of the GluR1 subunit from extrasynaptic sites is the key mechanism underlying synaptic plasticity and GluR1-PDZ interactions play a critical role in this type of plasticity. Application of Philanthotoxin-433 (PhTx) 5 min after LTP induction reduced synaptic potentiation, while PhTx had no effect on basal AMPA receptor-mediated responses, suggesting that Ca2+-permeable GluR2-lacking receptors contribute to the maintenance of ACC LTP. ACC LTP is absent GluR1 knockout mice [52]. We also examined the role of GluR2 related peptides in synaptic potentiation in the ACC and found that the GluR2/3-PDZ interaction had no effect on ACC LTP, and the same interfering peptides inhibited ACC LTD. Recent studies found that PKMzeta (PKMζ) activity is critical for maintaining synaptic potentiation in the ACC [38].
CORTICAL NETWORK PLASTICITY
To explore plastic changes within cortical circuit, we recently used a 64-channel multi-electrode dish (MED64) system, a two-dimensional electric activity monitoring device, to characterize LTD in adult mouse ACC. The MED64 system allows us to detect the field excitatory postsynaptic potentials (fEPSP) at multiple sites in mouse ACC, which is difficult to achieve with conventional field recording systems. Within the ACC, we observed spatial distribution of excitatory synaptic transmission when stimulating deep layer V [44]. This was the only area we could detect more than 20 channels with inward responses. Stimulating other areas, such as layer I or II/III, induced at most 10 channels of inward responses. We found, however, that not every activated channel underwent LTD; the channels that are within a 300 µm radius of the stimulation site being the most likely to exhibit this form of synaptic plasticity [44]. It is believed that such multiple channel recording system will provide new information about cortical LTP and LTD at both synaptic and circuit levels in near future.
LOSS OF LTD AFTER AMPUTATION
One major hypothesis for central plastic changes after peripheral injury is enhanced central synaptic transmission and neuronal excitability. There are at least two possible mechanisms to achieve this at synaptic level: enhanced synaptic transmission, and the loss of the ability to undergo LTD. Indeed, our previous work found that amputation of a third hindpaw digit in an adult rat induced rapid expression of immediate early genes in the ACC bilaterally and caused a loss of LTD that persisted for at least 2 weeks [39]. Similar to previous studies in rats, we also found that a loss of LTD in mouse ACC after distal tail amputation [44]. Similarly, LTD induced by the pharmacological activation of mGluR1 was also lost following amputation. Biochemical data showed that the surface level of mGluR1 was not changed after amputation compared to the sham group, suggesting that amputation triggered loss of LTD is not due to reduction of postsynaptic membrane levels of mGluR1. The modification is thus likely to be downstream of the mGluRs. Future studies are clearly needed for revealing the exact signaling pathways.
CORTICAL NETWORKS IN PHANTOM PAIN
Most of recent work at molecular and cellular levels focused on synaptic changes after nerve injury or amputation [4, 7]. It is well known that pain-related cortical neurons are highly wired with other cortical regions as well as subcortical areas [see 21 for review]. Future studies are clearly needed to understand synaptic changes within these cortical-cortical and cortical-subcortical connections. For example, a recent study showed that in neuropathic pain conditions (Note: the injury to peripheral nerve happen in most cases of limb amputation) can trigger facilitated synaptic transmission between somatosensory cortex and ACC [53], in addition to well-known plastic changes within the ACC [see 4, 7]. Furthermore, our recent study found that cortical-spinal projection neurons in the ACC undergoing potentiation after nerve injury (Chen et al., unpublished data). It is thus likely that amputation may trigger wide-spreading plastic changes in sensory and motor related cortical networks.
OPIOID ANALGESIA AND AMPUTATION
Opioids have been commonly used for the treatment of phantom pain. It produces some analgesic effects in subpopulation of patients, while other patients did not respond to the treatment as those oftern reported in neuropathic pain [for example see 54 for review]. Few studies have been reported using animal models. To test possible changes in opioid produced analgesic effects, we used distal tail amputation mouse model for studying this. Amputation of a segment of the tail produced long-lasting changes in nociception and morphine-induced antinociception. Plastic changes in nociceptive transmission may occur at the spinal cord as well as supraspinal structures after tail amputation [55]. Acute hyperalgesia is detected at the remaining part of the tail as well as hindpaw. Morphine induced facilitation of the HP response at a low dose and a greater dose of morphine is required to produce complete inhibition of the HP response. Since these effects happen at five weeks after the surgery, tail amputation may serve as a mouse model for studying long-term plastic changes in morphine produced analgesia after amputation.
CONCLUSIONS AND FUTURE DIRECTIONS
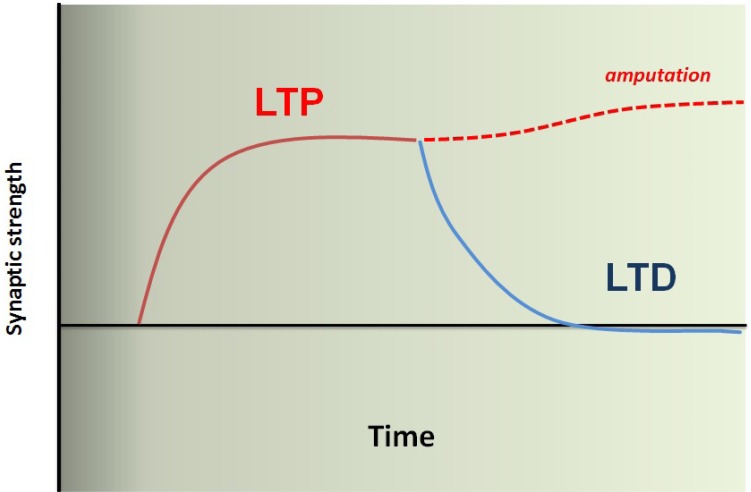
In conclusion, integrative physiological studies in genetically manipulated mice, have generated new and novel information regarding the basic mechanisms of chronic pain including phantom pain, particularly at the cortical levels. It is clear that nerve injuries or amputations trigger a series of plastic changes in pain-related cortical areas including the ACC. Loss of LTD as well as potentiation of synaptic transmission within the ACC and between cortical areas contributes to enhanced excitability of pain-processing and feeling cortical neurons (see Figure 1). Some of these cortical changes may not require persistent peripheral sensory inputs; thus will not respond to any medical treatment that targeted at lower subcortical levels. In addition to contribution to pain or phantom pain, such cortical plastic changes may also triggers a series of brain disorders such as emotional fear, anxiety, mood depression, and impairment of cognitive functions. Future studies designed to understand the relationship between pain and mood disorders, and identify novel molecular targets contributing to these events, will help us to find better pain medicine for patients including those with phantom pain.
Fig. 1.
A model for enhanced excitatory synaptic transmission in the pain-related ACC after amputation. Under normal physiological conditions, acute sensory experience may trigger short-lasting plastic changes in the ACC synapses. LTD provides an important mechanism to 'reset' those enhanced synapses. Thus, the cortical synapses and circuits are maintained at baseline levels, and allowed to process multiple sensory experiences. In case of amputation, abnormal neuronal activity from the periphery triggers long-lasting changes in cortical synapses, these may include changes at transcriptional and translational levels. Consequently, ACC synapses loss the ability to undergo LTD, or failure to reset the enhanced synapses. Enhanced cortical synapses may direct contribute to pain perception without additional sensory inputs or with the help of baseline brain activity (or noise brain activity) (or called central pain in some cases). It may also contribute to phantom limb sensations, as well as hyperalgesia or allodynia when the same synapses also receive peripheral noxious or non-noxious inputs. Resetting these enhanced synapses or recover the ability to undergo LTD may help us to control or reduce phantom pain.
ACKNOWLEDGEMENTS
I would like to give thanks for the funding supports from the EJLB-CIHR Michael Smith Chair in Neurosciences and Mental Health in Canada, Canada Research Chair, CIHR operating grants.
References
- 1.Buonomano DV, Merzenich MM. Cortical plasticity: from synapses to maps. Annu Rev Neurosci. 1998;21:149–186. doi: 10.1146/annurev.neuro.21.1.149. [DOI] [PubMed] [Google Scholar]
- 2.Kaas JH. Plasticity of sensory and motor maps in adult mammals. Annu Rev Neurosci. 1991;14:137–167. doi: 10.1146/annurev.ne.14.030191.001033. [DOI] [PubMed] [Google Scholar]
- 3.Ramachandran VS. Behavioral and magnetoencephalographic correlates of plasticity in the adult human brain. Proc Natl Acad Sci U S A. 1993;90:10413–10420. doi: 10.1073/pnas.90.22.10413. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 4.Zhuo M. Cortical excitation and chronic pain. Trends Neurosci. 2008;31:199–207. doi: 10.1016/j.tins.2008.01.003. [DOI] [PubMed] [Google Scholar]
- 5.Bliss TV, Collingridge GL. A synaptic model of memory: long-term potentiation in the hippocampus. Nature. 1993;361:31–39. doi: 10.1038/361031a0. [DOI] [PubMed] [Google Scholar]
- 6.Singer W. Development and plasticity of cortical processing architectures. Science. 1995;270:758–764. doi: 10.1126/science.270.5237.758. [DOI] [PubMed] [Google Scholar]
- 7.Zhuo M. Cortical plasticity as a new endpoint measurement for chronic pain. Mol Pain. 2011;7:54. doi: 10.1186/1744-8069-7-54. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 8.Bliss TV, Cooke SF. Long-term potentiation and long-term depression: a clinical perspective. Clinics (Sao Paulo) 2011;66(Suppl 1):3–17. doi: 10.1590/S1807-59322011001300002. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 9.Jones EG, Pons TP. Thalamic and brainstem contributions to large-scale plasticity of primate somatosensory cortex. Science. 1998;282:1121–1125. doi: 10.1126/science.282.5391.1121. [DOI] [PubMed] [Google Scholar]
- 10.Merzenich M. Long-term change of mind. Science. 1998;282:1062–1063. doi: 10.1126/science.282.5391.1062. [DOI] [PubMed] [Google Scholar]
- 11.Merzenich MM, Nelson RJ, Stryker MP, Cynader MS, Schoppmann A, Zook JM. Somatosensory cortical map changes following digit amputation in adult monkeys. J Comp Neurol. 1984;224:591–605. doi: 10.1002/cne.902240408. [DOI] [PubMed] [Google Scholar]
- 12.Merzenich MM, Sameshima K. Cortical plasticity and memory. Curr Opin Neurobiol. 1993;3:187–196. doi: 10.1016/0959-4388(93)90209-h. [DOI] [PubMed] [Google Scholar]
- 13.Pons TP, Garraghty PE, Ommaya AK, Kaas JH, Taub E, Mishkin M. Massive cortical reorganization after sensory deafferentation in adult macaques. Science. 1991;252:1857–1860. doi: 10.1126/science.1843843. [DOI] [PubMed] [Google Scholar]
- 14.Flor H, Nikolajsen L, Staehelin Jensen T. Phantom limb pain: a case of maladaptive CNS plasticity? Nat Rev Neurosci. 2006;7:873–881. doi: 10.1038/nrn1991. [DOI] [PubMed] [Google Scholar]
- 15.Devinsky O, Morrell MJ, Vogt BA. Contributions of anterior cingulate cortex to behaviour. Brain. 1995;118:279–306. doi: 10.1093/brain/118.1.279. [DOI] [PubMed] [Google Scholar]
- 16.Rainville P, Duncan GH, Price DD, Carrier B, Bushnell MC. Pain affect encoded in human anterior cingulate but not somatosensory cortex. Science. 1997;277:968–971. doi: 10.1126/science.277.5328.968. [DOI] [PubMed] [Google Scholar]
- 17.Davis KD, Taylor SJ, Crawley AP, Wood ML, Mikulis DJ. Functional MRI of pain- and attention-related activations in the human cingulate cortex. J Neurophysiol. 1997;77:3370–3380. doi: 10.1152/jn.1997.77.6.3370. [DOI] [PubMed] [Google Scholar]
- 18.Derbyshire SW, Vogt BA, Jones AK. Pain and Stroop interference tasks activate separate processing modules in anterior cingulate cortex. Exp Brain Res. 1998;118:52–60. doi: 10.1007/s002210050254. [DOI] [PubMed] [Google Scholar]
- 19.Hutchison WD, Davis KD, Lozano AM, Tasker RR, Dostrovsky JO. Pain-related neurons in the human cingulate cortex. Nat Neurosci. 1999;2:403–405. doi: 10.1038/8065. [DOI] [PubMed] [Google Scholar]
- 20.Lenz FA, Rios M, Zirh A, Chau D, Krauss G, Lesser RP. Painful stimuli evoke potentials recorded over the human anterior cingulate gyrus. J Neurophysiol. 1998;79:2231–2234. doi: 10.1152/jn.1998.79.4.2231. [DOI] [PubMed] [Google Scholar]
- 21.Vogt BA. Pain and emotion interactions in subregions of the cingulate gyrus. Nat Rev Neurosci. 2005;6:533–544. doi: 10.1038/nrn1704. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 22.Koyama T, Tanaka YZ, Mikami A. Nociceptive neurons in the macaque anterior cingulate activate during anticipation of pain. Neuroreport. 1998;9:2663–2667. doi: 10.1097/00001756-199808030-00044. [DOI] [PubMed] [Google Scholar]
- 23.Sikes RW, Vogt BA. Nociceptive neurons in area 24 of rabbit cingulate cortex. J Neurophysiol. 1992;68:1720–1732. doi: 10.1152/jn.1992.68.5.1720. [DOI] [PubMed] [Google Scholar]
- 24.Lee DE, Kim SJ, Zhuo M. Comparison of behavioral responses to noxious cold and heat in mice. Brain Res. 1999;845:117–121. doi: 10.1016/s0006-8993(99)01956-3. [DOI] [PubMed] [Google Scholar]
- 25.Pastoriza LN, Morrow TJ, Casey KL. Medial frontal cortex lesions selectively attenuate the hot plate response: possible nocifensive apraxia in the rat. Pain. 1996;64:11–17. doi: 10.1016/0304-3959(95)00070-4. [DOI] [PubMed] [Google Scholar]
- 26.Calejesan AA, Kim SJ, Zhuo M. Descending facilitatory modulation of a behavioral nociceptive response by stimulation in the adult rat anterior cingulate cortex. Eur J Pain. 2000;4:83–96. doi: 10.1053/eujp.1999.0158. [DOI] [PubMed] [Google Scholar]
- 27.Koga K, Li X, Chen T, Steenland HW, Descalzi G, Zhuo M. In vivo whole-cell patch-clamp recording of sensory synaptic responses of cingulate pyramidal neurons to noxious mechanical stimuli in adult mice. Mol Pain. 2010;6:62. doi: 10.1186/1744-8069-6-62. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 28.Tang J, Ko S, Ding HK, Qiu CS, Calejesan AA, Zhuo M. Pavlovian fear memory induced by activation in the anterior cingulate cortex. Mol Pain. 2005;1:6. doi: 10.1186/1744-8069-1-6. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 29.Apkarian AV, Bushnell MC, Treede RD, Zubieta JK. Human brain mechanisms of pain perception and regulation in health and disease. Eur J Pain. 2005;9:463–484. doi: 10.1016/j.ejpain.2004.11.001. [DOI] [PubMed] [Google Scholar]
- 30.Reilly KT, Sirigu A. The motor cortex and its role in phantom limb phenomena. Neuroscientist. 2008;14:195–202. doi: 10.1177/1073858407309466. [DOI] [PubMed] [Google Scholar]
- 31.Kaas JH, Florence SL, Jain N. Subcortical contributions to massive cortical reorganizations. Neuron. 1999;22:657–660. doi: 10.1016/s0896-6273(00)80725-4. [DOI] [PubMed] [Google Scholar]
- 32.Jain N, Florence SL, Qi HX, Kaas JH. Growth of new brainstem connections in adult monkeys with massive sensory loss. Proc Natl Acad Sci U S A. 2000;97:5546–5550. doi: 10.1073/pnas.090572597. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 33.Davis KD, Kiss ZH, Luo L, Tasker RR, Lozano AM, Dostrovsky JO. Phantom sensations generated by thalamic microstimulation. Nature. 1998;391:385–387. doi: 10.1038/34905. [DOI] [PubMed] [Google Scholar]
- 34.Flor H, Elbert T, Knecht S, Wienbruch C, Pantev C, Birbaumer N, Larbig W, Taub E. Phantom-limb pain as a perceptual correlate of cortical reorganization following arm amputation. Nature. 1995;375:482–484. doi: 10.1038/375482a0. [DOI] [PubMed] [Google Scholar]
- 35.Metz AE, Yau HJ, Centeno MV, Apkarian AV, Martina M. Morphological and functional reorganization of rat medial prefrontal cortex in neuropathic pain. Proc Natl Acad Sci U S A. 2009;106:2423–2428. doi: 10.1073/pnas.0809897106. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 36.Seminowicz DA, Laferriere AL, Millecamps M, Yu JS, Coderre TJ, Bushnell MC. MRI structural brain changes associated with sensory and emotional function in a rat model of long-term neuropathic pain. Neuroimage. 2009;47:1007–1014. doi: 10.1016/j.neuroimage.2009.05.068. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 37.Xu H, Wu LJ, Wang H, Zhang X, Vadakkan KI, Kim SS, Steenland HW, Zhuo M. Presynaptic and postsynaptic amplifications of neuropathic pain in the anterior cingulate cortex. J Neurosci. 2008;28:7445–7453. doi: 10.1523/JNEUROSCI.1812-08.2008. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 38.Li XY, Ko HG, Chen T, Descalzi G, Koga K, Wang H, Kim SS, Shang Y, Kwak C, Park SW, Shim J, Lee K, Collingridge GL, Kaang BK, Zhuo M. Alleviating neuropathic pain hypersensitivity by inhibiting PKMzeta in the anterior cingulate cortex. Science. 2010;330:1400–1404. doi: 10.1126/science.1191792. [DOI] [PubMed] [Google Scholar]
- 39.Wei F, Li P, Zhuo M. Loss of synaptic depression in mammalian anterior cingulate cortex after amputation. J Neurosci. 1999;19:9346–9354. doi: 10.1523/JNEUROSCI.19-21-09346.1999. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 40.Wei F, Xu ZC, Qu Z, Milbrandt J, Zhuo M. Role of EGR1 in hippocampal synaptic enhancement induced by tetanic stimulation and amputation. J Cell Biol. 2000;149:1325–1334. doi: 10.1083/jcb.149.7.1325. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 41.Zhuo M. NMDA receptor-dependent long term hyperalgesia after tail amputation in mice. Eur J Pharmacol. 1998;349:211–220. doi: 10.1016/s0014-2999(98)00197-6. [DOI] [PubMed] [Google Scholar]
- 42.Wu MF, Pang ZP, Zhuo M, Xu ZC. Prolonged membrane potential depolarization in cingulate pyramidal cells after digit amputation in adult rats. Mol Pain. 2005;1:23. doi: 10.1186/1744-8069-1-23. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 43.Wei F, Zhuo M. Potentiation of sensory responses in the anterior cingulate cortex following digit amputation in the anaesthetised rat. J Physiol. 2001;532:823–833. doi: 10.1111/j.1469-7793.2001.0823e.x. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 44.Kang SJ, Liu MG, Chen T, Ko HG, Baek GC, Lee HR, Lee K, Collingridge GL, Kaang BK, Zhuo M. Plasticity of metabotropic glutamate receptor-dependent long-term depression in the anterior cingulate cortex after amputation. J Neurosci. 2012;32:11318–11329. doi: 10.1523/JNEUROSCI.0146-12.2012. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 45.Wu LJ, Toyoda H, Zhao MG, Lee YS, Tang J, Ko SW, Jia YH, Shum FW, Zerbinatti CV, Bu G, Wei F, Xu TL, Muglia LJ, Chen ZF, Auberson YP, Kaang BK, Zhuo M. Upregulation of forebrain NMDA NR2B receptors contributes to behavioral sensitization after inflammation. J Neurosci. 2005;25:11107–11116. doi: 10.1523/JNEUROSCI.1678-05.2005. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 46.Wu LJ, Xu H, Ren M, Zhuo M. Genetic and pharmacological studies of GluR5 modulation of inhibitory synaptic transmission in the anterior cingulate cortex of adult mice. Dev Neurobiol. 2007;67:146–157. doi: 10.1002/dneu.20331. [DOI] [PubMed] [Google Scholar]
- 47.Toyoda H, Wu LJ, Zhao MG, Xu H, Jia Z, Zhuo M. Long-term depression requires postsynaptic AMPA GluR2 receptor in adult mouse cingulate cortex. J Cell Physiol. 2007;211:336–343. doi: 10.1002/jcp.20940. [DOI] [PubMed] [Google Scholar]
- 48.Toyoda H, Zhao MG, Zhuo M. NMDA receptor-dependent long-term depression in the anterior cingulate cortex. Rev Neurosci. 2006;17:403–413. doi: 10.1515/revneuro.2006.17.4.403. [DOI] [PubMed] [Google Scholar]
- 49.Zhao MG, Toyoda H, Lee YS, Wu LJ, Ko SW, Zhang XH, Jia Y, Shum F, Xu H, Li BM, Kaang BK, Zhuo M. Roles of NMDA NR2B subtype receptor in prefrontal long-term potentiation and contextual fear memory. Neuron. 2005;47:859–872. doi: 10.1016/j.neuron.2005.08.014. [DOI] [PubMed] [Google Scholar]
- 50.Liauw J, Wu LJ, Zhuo M. Calcium-stimulated adenylyl cyclases required for long-term potentiation in the anterior cingulate cortex. J Neurophysiol. 2005;94:878–882. doi: 10.1152/jn.01205.2004. [DOI] [PubMed] [Google Scholar]
- 51.Wang H, Xu H, Wu LJ, Kim SS, Chen T, Koga K, Descalzi G, Gong B, Vadakkan KI, Zhang X, Kaang BK, Zhuo M. Identification of an adenylyl cyclase inhibitor for treating neuropathic and inflammatory pain. Sci Transl Med. 2011;3:65ra3. doi: 10.1126/scitranslmed.3001269. [DOI] [PubMed] [Google Scholar]
- 52.Toyoda H, Wu LJ, Zhao MG, Xu H, Zhuo M. Time-dependent postsynaptic AMPA GluR1 receptor recruitment in the cingulate synaptic potentiation. Dev Neurobiol. 2007;67:498–509. doi: 10.1002/dneu.20380. [DOI] [PubMed] [Google Scholar]
- 53.Eto K, Wake H, Watanabe M, Ishibashi H, Noda M, Yanagawa Y, Nabekura J. Inter-regional contribution of enhanced activity of the primary somatosensory cortex to the anterior cingulate cortex accelerates chronic pain behavior. J Neurosci. 2011;31:7631–7636. doi: 10.1523/JNEUROSCI.0946-11.2011. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 54.Huse E, Larbig W, Flor H, Birbaumer N. The effect of opioids on phantom limb pain and cortical reorganization. Pain. 2001;90:47–55. doi: 10.1016/s0304-3959(00)00385-7. [DOI] [PubMed] [Google Scholar]
- 55.Kim SJ, Zhuo M. Biphasic modulation of behavioral nociceptive responses by morphine in adult mice after amputation. Sheng Li Xue Bao. 2004;56:436–443. [PubMed] [Google Scholar]