Summary
YscU, a component of the Yersinia type III secretion machine, promotes auto-cleavage at asparagine 263 (N263). Mutants with an alanine substitution at yscU codon 263 displayed secretion defects for some substrates (LcrV, YopB and YopD), however transport of effector proteins into host cells (YopE, YopH, YopM) continued to occur. Two yscU mutations were isolated that, unlike N263A, completely abolished type III secretion; YscUG127D promoted auto-cleavage at N263, whereas YscUG270N did not. When fused to glutathione-S-transferase, the YscU C-terminal cytoplasmic domain promoted auto-cleavage and Gst-YscUC also exerted a dominant-negative phenotype by blocking type III secretion. Gst-YscUC/N263A caused a similar blockade and Gst-YscUC/G270N reduced secretion. Gst-YscUC and Gst-YscUC/N263A bound YscL, the regulator of the ATPase YscN, whereas Gst-YscUC/G270N did not. When isolated from Yersinia, Gst-YscUC and Gst-YscUC/N263A associated with YscK-YscL-YscQ; however, Gst-YscUC/G270N interacted predominantly with the machine component YscO, but not with YscK-YscL-YscQ. A model is proposed whereby YscU auto-cleavage promotes interaction with YscL and recruitment of ATPase complexes that initiate type III secretion.
Introduction
During infection, pathogenic Yersinia spp. (Y. pestis, Y. pseudotuberculosis and Y. enterocolitica) use type III machines to inject effector Yops into immune cells in an effort to escape phagocytic killing (Cornelis, 2006; Hueck, 1998). The proximal portion of the type III secretion apparatus is formed by membrane proteins, which are translocated via the Sec pathway and assembled to generate a structure with rotational symmetry in the bacterial envelope (Blocker et al., 2001; Galan and Wolf-Watz, 2006; Marlovits et al., 2004; Michiels et al., 1991). Assembly of the distal portion of the apparatus involves secretion of early type III substrates such as YscF, a polypeptide that polymerizes to form a hollow needle and extension of the secretion machine (Blocker et al., 2001; Hoiczyk and Blobel, 2001; Marenne et al., 2003; Marlovits et al., 2004; Mueller et al., 2005). YscH (YopR) and YscP represent additional early secretion substrates, one of which (YscP) controls needle length (Agrain et al., 2005; Allaoui et al., 1995; Journet et al., 2003; Sorg et al., 2006). Three proteins are thought to comprise middle substrates. LcrV is deposited at the tip of needles, whereas YopB and YopD are inserted into host cell membranes and thereby complete assembly of the type III pathway between Yersinia and host targets cells (Broz et al., 2007; Lee et al., 2000; Neyt and Cornelis, 1999; Sarker et al., 1998). Once activated by low calcium signals that are thought to be derived from the cytosol of host cells, type III machines enable transport of late substrates (effectors YopE, YopH, YopM, YopN, YopO, YopP, YopQ and YopT) into host cells (Cornelis et al., 1998; Lee et al., 2001; Marketon et al., 2005).
There is no hierarchy in the synthesis of Yersinia machine components or secretion substrates. Recent work suggests that the type III apparatus switches its substrate specificity during morphogenesis from early to middle and, upon completion of the entire conduit, to late substrates (Edqvist et al., 2003; Sorg et al., 2007). If so, what are the secretion signals that define the different classes of substrates and what are the mechanisms that allow type III machines to distinguish between these proteins and switch their specificity? The characterization of secretion signals typically requires translational fusions between substrate and reporter genes, thereby generating hybrid proteins capable of traveling the pathway in a manner similar to other substrates in this class (Anderson and Schneewind, 1997; Cheng et al., 1997; Lee and Schneewind, 2002; Michiels and Cornelis, 1991; Schesser et al., 1996; Sorg et al., 2005a; Sorg et al., 2006; Sory et al., 1995). Although this has been tested for many substrates and reporters, only some substrates can be studied in this manner. Those that do not fulfill the aforementioned criteria (most of the early and middle substrates) are often required for stepwise assembly of the type III pathway and/or lose their substrate properties when transplanted onto a reporter protein (DeBord et al., 2001).
To date, YopR (early substrate) and YopE (late substrate) represent the best studied examples. Secretion of YopR occurs early and leads to transport into the extracellular medium (Lee and Schneewind, 1999), whereas YopE is only recognized by type III machines once the entire pathway has been completed, permitting YopE transport into HeLa cells and immune cells (Cheng and Schneewind, 1999; Rosqvist et al., 1991). The minimal secretion signal of YopR encompasses the first 11 residues of the 165 amino acid polypeptide (Sorg et al., 2006). In contrast to YopE, YopR is not known to require binding to a chaperone for substrate recognition. The SycE chaperone binds with high affinity to YopE residues 15–100, an interaction that is absolutely essential for type III injection of YopE into immune cells (Cheng and Schneewind, 1999; Lee et al., 1998; Sory et al., 1995; Woestyn et al., 1996). Other chaperones promote type III transport of other effectors, however, in all cases examined, chaperones are displaced from their cargo prior to initiation of the substrate into the type III machine (Thomas et al., 2004). Fusion of YopR to a reporter (ubiquitin, β-galactosidase, glutathione-S-transferase or dihydrofolate reductase) that folds rapidly and assumes a structure that cannot pass through the machine (Lee and Schneewind, 2002) causes a secretion blockade for all other substrates in the type III pathway (Sorg et al., 2006). On the other hand, fusion of YopE to an impassable reporter generates hybrids that can be rejected from the type III pathway without interfering with the secretion of other substrates (Sorg et al., 2005a; Sorg et al., 2005b).
In the flagellar assembly pathway, different classes of substrates can be derived from the precise order of secretion, the position of the assembled product within basal body-hook-filaments or from the assembly defects caused by mutations in specific substrate genes (Blocker et al., 2003; Ferris and Minamino, 2006; Hughes et al., 1993; Macnab, 2004; Williams et al., 1996). Informed by these strategies, Macnab and colleagues identified machine components with affinity for specific substrates and/or FliI, the secretion ATPase (McMurry et al., 2004; Minamino and Macnab, 2000a; Minamino et al., 2003; Zhu et al., 2002). FlhB, the flagellar secretion machine component and homolog of YscU, encompasses an N-terminal membrane domain and a C-terminal cytoplasmic domain that promotes auto-cleavage between N269 and P270 of its NPTH signature motif (Ferris et al., 2005; Minamino and Macnab, 2000b). Salmonella flhB mutants with alanine substitutions at codon 269 (N269A) or 270 (P270A) assemble polyhook structures that cannot be extended into flagellar filaments or promote bacterial motility, presumably because variant flhB products cannot switch the specificity between different classes of secretion substrates (Ferris et al., 2005; Fraser et al., 2003). The C-terminal cytoplasmic domain of the switch, FlhBC, acts as a dominant-negative by blocking both flagellar assembly and motility (Fraser et al., 2003). FlhBC promotes auto-cleavage and, when purified, can bind to other machine components (FliI, FliH, FliJ, and the cytoplasmic domain of FlhA) as well as several substrates, i.e. rod and hook components (FlgE, FlgD), the hook length control protein FliK and even flagellin (FliC) (McMurry et al., 2004; Minamino and Macnab, 2000a; Minamino et al., 2004; van Arnam et al., 2004; Zhu et al., 2002). In agreement with the conjecture that substrate recognition of all type III machines (flagellar and virulence systems) may occur by universal mechanisms, Yersinia yscU mutants with alanine substitution at the auto-cleavage site (N263A) were unable to deposit LcrV at the tip of type III needles or to secrete the other two middle substrates (YopB and YopD) (Allaoui et al., 1994; Edqvist et al., 2003; Lavander et al., 2002; Sorg et al., 2007). As type III needles are nonfunctional unless their tip has been decorated with LcrV, it has been suggested that Yersinia harboring yscU N263A cannot transport effectors into mammalian cells, albeit that N263A mutants can secrete late substrates under low calcium conditions in laboratory media (Sorg et al., 2007).
To further characterize the role of YscU in substrate recognition, we isolated mutations in its structural gene and studied their phenotype. Additional work examined the role of YscUC as a dominant-negative for type III secretion. Auto-cleavage generated the expected fragments, YscUCN and YscUCC. When purified from the cytoplasm of Yersinia, YscUCN/CC bound to YscL-YscK-YscQ, but lacked physical interactions with the ATPase YscN or the early (YscP) and middle substrates (LcrV). YscUC/G270N, a mutant that was completely defective in auto-cleavage or substrate recognition, lacked the ability to bind YscL (the regulator of YscN) and may thereby cause a critical defect in recruiting the ATPase to YscU and promoting secretion.
Results
yscU mutants that cannot promote type III secretion
Upon chelation of calcium ions with sodium oxalate, Y. pestis cannot form colonies on agar medium at 37°C unless mutations in type III machine genes relieve the low-calcium growth restriction (LCR) that is caused by the massive secretion of Yops under such conditions (Fig. 1A) (Yother and Goguen, 1985). As expected, Mu insertions in yscU allowed variants of Y. pestis KIM D27 biovar mediaevalis (pCD1+, pMT1+, pgm−), to form colonies at 37°C on HIA agar with chelated calcium, whereas yscU mutants expressing plasmid-encoded wild-type yscU displayed growth-restriction (Fig. 1A). The yscU N263A mutation, generated via site-directed mutagenesis of plasmid encoded yscU, caused a small improvement in growth of mutant Yersinia at 37°C, indicating that the partial blockade in substrate recognition cannot fully relieve the LCR imposed by activation of the type III pathway (Fig. 1A).
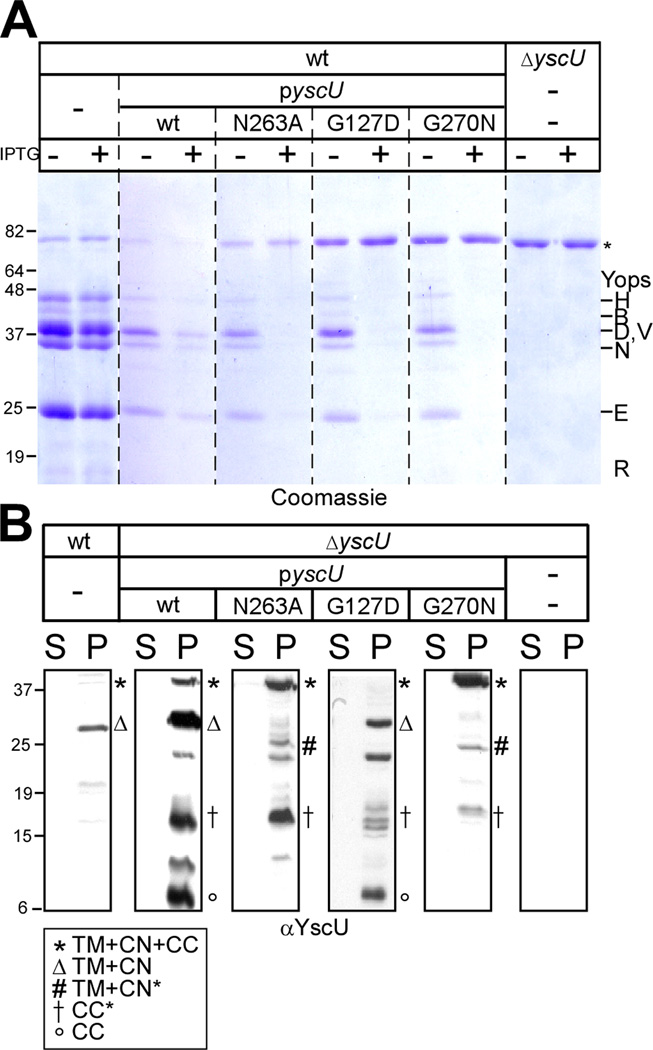
Fig. 1.
Phenotypic evaluation of hydroxylamine–generated mutants in the TTS gene yscU. (A) Y. pestis KIM D27 with chloramphenicol cassette inserted into the yopQ gene (MEL24) and ΔyscU (CHI110) Y. pestis containing vector control, pyscU (wt), pyscU N263A, pyscU G127D, and pyscU G270N on HIA plates containing 35 µg/mL chloramphenicol, 20 mM MgCl2, 20 mM oxalic acid, and 1 mM IPTG to induce type III secretion in the absence of calcium. Plates were grown at 37°C for 2 days to examine the LCR phenotype of indicated strains. (B) Diagram of the YscU protein indicating the four transmembrane helices and its hypothesized location in reference to the type III translocon. Mutations studied further are denoted with a red star. (C) Amino acid and corresponding DNA sequence of Y. enterocolitica W22703 yscU. Transmembrane domains are highlighted in light blue and the XNPTH cleavage motif highlighted in purple. All nonsense mutations obtained during hydroxylamine mutagenesis are denoted in green and missense mutations that generated phenotypes (G127D, G270N, N263A) are indicated in red. (D) Y. enterocolitica strains W22703, ΔyscU (CT-132), and ΔyscU containing pyscU (wt), pyscU N263A, pyscU G127D, and pyscU G270N were induced for type III secretion in TSB containing 35 µg/mL of chloramphenicol when needed for plasmid maintenance. Strains were grown in the absence of calcium (Ca2+) (5 mM EGTA) in the absence (−) or presence (+) of the inducer IPTG; and following centrifugation of culture aliquots, proteins in the supernatant (S) were separated by 12% SDS-PAGE. Gels were visualized with Coomassie blue staining; molecular mass markers (in kilodaltons) are indicated to the left of the gel while secreted Yop effectors are indicated to the right. An 80kD non-specific band common to all supernatants is indicated by an asterisk (*). (E) Supernatants generated in D were immunoblotted with polyclonal antisera specific to YopR, LcrV, YopB, YopD and YopE. The percent secretion of YopR, LcrV, YopB, YopD and YopE was calculated by comparing the secretion in each of the mutant strains to the amount of protein secreted in Y. enterocolitca ΔyscU containing pyscU (wt). Values are reported below the respective immunoblots. Secretion of YopR, LcrV, YopB, YopD, and YopE in Y. enterocolitca ΔyscU containing pyscU (wt) was set at 100%.
We sought to isolate point mutations in yscU that completely blocked type III secretion. Purified pyscU plasmid was subjected to hydroxylamine mutagenesis and, following electroporation of DNA into yscU mutant yersiniae, transformants were selected on agar at 26°C, streaked on HIA with sodium oxalate and incubated at 37°C to score LCR. Plasmids from LCR mutants were isolated, genetic linkage to episomal DNA verified via repeat transformation into yscU mutants, and mutations identified by DNA sequencing. Most plasmids harbored mutations that introduced non-sense codons into the yscU open reading frame (highlighted in green in Fig. 1C), thereby truncating the yscU open reading frame and generating protein products that could not be detected by immunoblotting with YscU specific antisera (data not shown). Two plasmids carried a missense mutation, G127D and G270N, respectively (highlighted in red in Fig. 1C). The G127D substitution is positioned in a predicted cytoplasmic loop, connecting the second and third transmembrane domain, whereas G270N is positioned seven residues downstream from the YscU auto-cleavage site in the C-terminal domain of YscU (Fig. 1B).
Wild-type and yscU mutant Y. enterocolitica harboring either no plasmid or low-copy-number plasmids with yscU or its mutant alleles under control of the IPTG-inducible tac promoter were grown in TSB medium and incubated at 37°C without calcium. Coomassie-stained SDS-PAGE of culture supernatant samples revealed type III secretion of Yop proteins by wild-type yersiniae, but not by its isogenic yscU variant (Fig. 1D). Plasmid encoded wild-type yscU restored the ability of yscU mutants to promote type III secretion, even in the absence of IPTG (Fig. 1D). This can be explained by the leaky expression of yscU from the tac promoter and is in agreement with the conjecture that small amounts of YscU suffice to promote Yop secretion (Lavander et al., 2002). The yscU N263A mutant displayed a unique phenotype (Fig. 1D). In agreement with earlier observations by Cornelis and colleagues, LcrV, YopB, and YopD (middle substrates) could not be detected by Coomassie-staining of supernatant samples, whereas YopE, YopN, and YopH were detectable, albeit in reduced amounts (Fig. 1D, data not shown). Immunoblotting with specific antisera revealed that the yscU N263A mutant indeed failed to secrete LcrV, as already reported by Cornelis and co-workers (Fig. 1E)(Sorg et al., 2007). Nevertheless, small amounts of YopB and YopD could be detected in the culture supernatant of the yscU N263A mutant (24% and 5% of wild-type, respectively), and the reduced secretion was similar to that measured for YopE (28% of wild-type) and YopR (8% of wild-type) (Fig. 1E) as well as YopH and YopN (data not shown). Thus, while the yscU N263A mutant clearly displays the reported secretion defect of LcrV, close examination of Yops suggests that it does not specifically affect all middle substrates, but rather reduces the secretion of several early, middle, and late substrates. These observations are in agreement with the intermediate LCR phenotype of yscU N263A mutants (Fig. 1A) and the type III injection phenotype during HeLa cell infection (vide infra). In contrast, the yscU mutant G270N was completely defective in type III secretion, while the G127D mutant secreted only very small amounts of YopB but none of the other substrates (Fig.1E). Yersinia yscU mutants harboring various plasmids were examined for their ability to synthesize secretion substrates. All five strains synthesized YopR, YopB, LcrV, YopD and YopE, (Fig. S1). In agreement with earlier reports, synthesis of secretion substrates was decreased in the ΔyscU mutant as well as the ΔyscU strains harboring pyscUG127D and pyscUG270N (Lavander et al., 2002), plasmids that failed to complement the yscU deletion mutant. Expression of pyscUN263A in the ΔyscU mutant strain restored synthesis of secretion substrates to near wild-type levels (Fig. S1).
Both the yscU N263A and yscU G270N mutations were generated on the pYV virulence plasmid. Strains were grown in the presence and absence of calcium and induced for type III secretion. Immunoblots revealed that Y. enterocolitica pYV yscU N263A and yscU G270N displayed the same secretion phenotypes as the ΔyscU strains harboring pyscUN263A or pyscUG270N (Fig. S2A). Y. enterocolitica pYV yscU G270N was unable to secrete any type III substrates, whereas Y. enterocolitica pYV yscU N263A was defective in the secretion of early and middle substrates (YopR, LcrV, and YopD), but able to secrete the effectors (YopE, Fig. S2A).
yscU alleles unable to support Yersinia type III injection
Low calcium induced secretion of Yops by bacteria grown in laboratory media is not a measure for the physiological sequence of Yersinia type III assembly and transport reactions, but rather represents a convenient high-throughput assay for the analysis of secretion phenotypes. To test secretion phenotypes of yscU alleles under physiological conditions, HeLa cells were infected with wild-type or yscU mutant Y. enterocolitica (Fig. 2). The effect of the type III pathway on these cells was viewed in microscopy experiments that interrogated overall cellular morphology as well as type III-mediated actin cable rearrangements by fluorescence staining with rhodamine-labeled phalloidin (Fig. 2B) (Rosqvist et al., 1991; Rosqvist et al., 1994; Sorg et al., 2005a). As expected, wild-type Y. enterocolitica caused rounding and contraction of HeLa cells as well as actin cable rearrangements, whereas an isogenic yscU deletion variant did not (Fig. 2B). Complementation of the yscU mutant with plasmid-encoded wild-type yscU restored both phenotypes (Fig. 2B). Plasmids encoding yscU G127D or yscU G270N each failed to restore Yersinia type III injection into HeLa cells, whereas yscU N263A at least in part restored cell rounding and actin cable rearrangements (Fig. 2B). This result was surprising due to the fact that LcrV secretion, a prerequisite for type III injection of effector Yops into host cells, could not be detected in the N263A mutant (Fig. 1 and Fig. S1). It is possible, however, that the amount of LcrV secreted by the N263A mutant is below detection, but still sufficient to promote type III injection of HeLa cells. This conjecture is in agreement with the recent report that only minor amounts of secreted LcrV are necessary to promote type III injection and rounding of host cells (Broms et al., 2007).
Fig. 2.
yscU N263A, but not G127D or G270N alleles support Y. enterocolitica type III injection into HeLa cells. (A) Y. enterocolitica strains W22703, ΔyscU (CT-132), and ΔyscU containing pyscU (wt), pyscU N263A, pyscU G127D, and pyscU G270N were used to infected HeLa tissue culture cells at an MOI of 10. IPTG was added to the medium to induce expression of various yscU alleles. The infection medium (M) was removed and centrifuged, separating the supernatant (S) and pellet (P). Digitonin was added to tissue culture cells with adherent bacteria. Digitonin extracted samples (D) were centrifuged, separating the supernatant (S) and pellet (P). All samples were precipitated with methanol/chloroform. Proteins were separated by 15% SDS-PAGE and immunoblotted with polyclonal antisera specific for YopR, YopE, YopH, YopN, YopB, YopD, LcrV, IκB, and RpoA. (B) HeLa tissue culture cells, 3 × 105 cells/flask, were infected at an MOI of 10 with Y. enterocolitica strains W22703, ΔyscU (CT-132), and ΔyscU containing pyscU (wt), pyscU N263A, pyscU G127D, and pyscU G270N. Cytotoxicity of Y. enterocolitica infected HeLa cells was visualized by staining F-actin with rhodamine-conjugated phalloidin. Wild-type infected HeLa cells are shown as a control for type III injection.
To quantify Yersinia type III injection, infected HeLa cells were analyzed by biochemical fractionation. The culture medium (M) was decanted and centrifuged to separate extracellular medium in the supernatant (S) from non-adherent bacteria in the pellet (P). Tissue culture cells were then extracted with digitonin (D), a detergent that disrupts the cholesterol containing plasma membrane of HeLa cells, but not the bacterial envelope. Digitonin extracts were centrifuged to separate cytosolic contents in the supernatant (S) from bacterial sediment and large cellular organelles in the pellet (P) (Lee et al., 1998). After precipitation with chloroform/methanol, proteins were separated on SDS-PAGE and analyzed by immunoblotting with specific antisera (Fig. 2A). As expected, wild-type Y. enterocolitica injected YopE, YopH, and YopN into the cytoplasm of HeLa cells, whereas the yscU mutant did not (Fig. 2A). Complementation of the yscU mutant with plasmid encoded wild-type yscU restored type III injection of YopE, YopH, and YopN, however plasmids encoding yscU G127D or yscU G270N failed to do so (Fig. 2A). Plasmid encoded yscU N263A restored Yersinia type III injection of YopE, YopH, and YopN (Fig. 2A). Unlike wild-type yscU, the N263A mutant had in part lost the ability to specifically target Yops into HeLa cells, as small amounts of YopE, YopH, and YopN were found in the extracellular medium, similar to class I mutant yersiniae (Fig. 2A) (Anderson et al., 2002; Cheng et al., 2001). Y. enterocolitica pYV yscU N263A and yscU G270N displayed similar injection phenotypes as the ΔyscU strains harboring pyscUN263A and pyscUG270N (Fig. S2BC).
Auto-cleavage of YscU and its variants
To examine yscU mutants for their ability to promote YscU auto-cleavage, wild-type or yscU mutant Y. enterocolitica were grown in TSB without calcium at 37°C for 3 hours. Following centrifugation, bacterial sediment and culture supernatant were separated and protein in both fractions was analyzed by immunoblotting with rabbit antisera raised against purified YscUC. A prominent immunoreactive species, YscUTM+CN (the N-terminal product of auto-cleavage at N263) was detected in wild-type bacteria (Fig. 3). Small amounts of full-length polypeptide (YscUTM+CN+CC) were also detectable (Fig. 3B). Over-expression of plasmid encoded wild-type yscU from the tac promoter in Y. enterocolitica W22703 produced a greater abundance of immune-reactive YscU species, including full-length YscUTM+CN+CC (both catalyst and substrate of auto-cleavage), YscU TM+CN, YscUCC, as well as less abundant species that likely represent proteolytic degradation products (Fig. 3). In agreement with previous observations of Cornelis and colleagues, yscU N263A mutant yersiniae accumulated YscUTM+CN+CC and aberrant proteolysis fragments, but not YscU TM+CN and YscUCC, indicating that YscUN263A is defective in auto-cleavage (Fig. 3B)(Sorg et al., 2007). The yscU G270N mutant displayed the same phenotype, whereas yscU G127D behaved similar to wild-type, accumulating YscUTM+CN and YscUCC (Fig. 3B). IPTG-induced over-expression of any yscU allele (wt, N263A, G127D, and G270N) abolished type III secretion of wild-type yersiniae (Fig. 3A). These dominant-negative phenotypes required the presence of IPTG in growth media (Fig. 3A), whereas complementation of type III secretion defects in yscU mutants by plasmid encoded yscU under control of the same promoter occurred even in the absence of IPTG (Fig. 1D). From this, we surmise that YscU, YscUN263A, YscUG127D and YscUG270N exert biological activities that perturb proper function of type III machines once these proteins reach a critical concentration.
Fig. 3.
Expression of pyscU N263A, G127D, and G270N in wild-type Y. enterocolitica shows dominant negative activity. (A) W22703 Y. enterocolitica strains expressing pyscU (wt), pyscU N263A, pyscU G127D, and pyscU G270N were induced for type III secretion (−Ca2+) in the absence (−) or presence (+) of IPTG. Following centrifugation of culture aliquots, proteins in the supernatant (S) were separated by 12% SDS-PAGE. Gels were visualized with Coomassie blue staining; molecular mass markers (in kilodaltons) are indicated to the left of the gel while secreted Yop effectors are indicated to the right. (B) Y. enterocolitica wild-type, ΔyscU containing pyscU (wt), pyscU N263A, pyscU G127D, and pyscU G270N, and ΔyscU were grown in the absence of calcium (Ca2+); and following centrifugation of culture aliquots, proteins in the supernatant (S) and bacterial pellet (P) were separated by SDS-PAGE and immunoblotted with polyclonal antisera specific to YscU (α-YscU). Several different species of the YscU protein were identified in the pellet fraction as denoted in the key.
Dominant-negative properties of YscUC and its variants
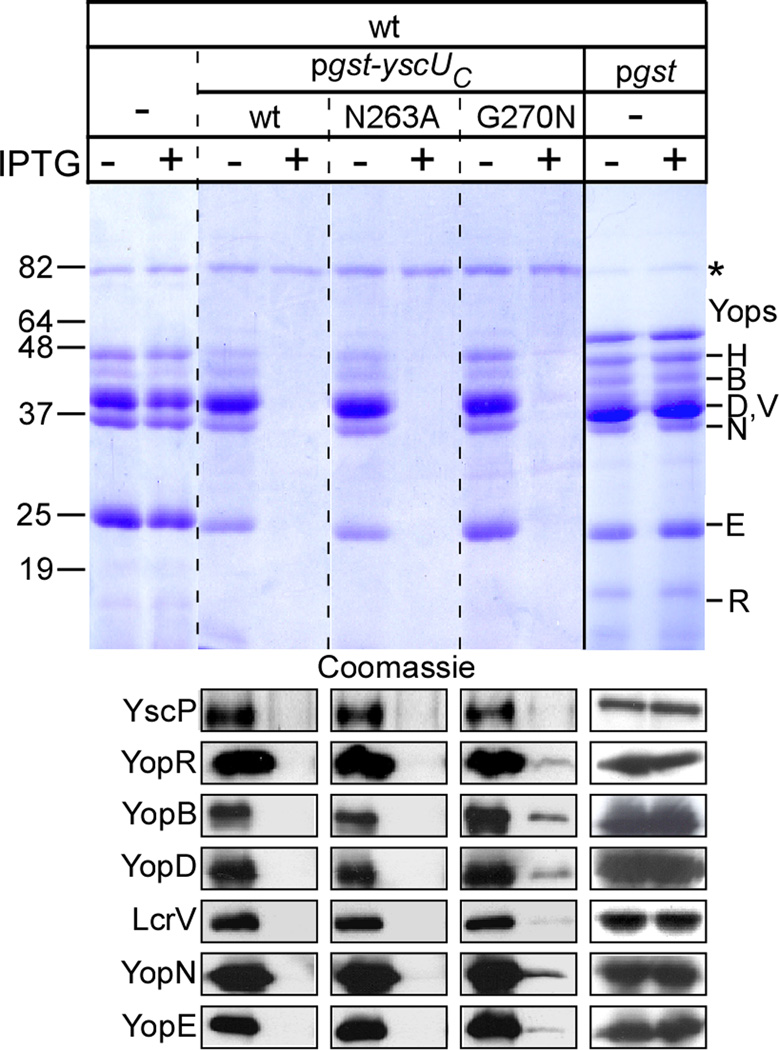
Macnab and colleagues observed that the C-terminal cytoplasmic domain of FlhB, which also catalyzes auto-cleavage at the conserved asparagine-proline bond, inhibits Salmonella flagellar assembly, secretion of flagellin, as well as bacterial motility (Ferris et al., 2005; Fraser et al., 2003; Minamino and Macnab, 2000b). We wondered whether the C-terminal domain of YscU (YscUC, residues 205–354), when fused to the C-terminal end of glutathione S-transferase (Gst), exhibited a similar property for the Yersinia type III pathway. GST-YscUC was first expressed in E. coli, purified using affinity chromatography, and was found by mass spectrometry analysis to harbor the same cleavage site (N263/P264) as the flagellar protein FlhB (Fig. S3). GST-YscUC was processed in the absence of any of the type III secretion system components from yersiniae supporting the hypothesis that the C-terminal domain of YscU undergoes auto-cleavage (Fig. S3). In yersiniae, IPTG-inducible expression of plasmid encoded gst-yscUC from the tac promoter failed to restore type III secretion and the LCR phenotype of yscU mutant yersiniae (data not shown), however expression of this hybrid in wild-type Y. enterocolitica blocked type III secretion of Yops as measured by Coomassie stained SDS-PAGE or immunoblotting (Fig. 4). This phenotype was due to the YscUC portion of the fusion protein, as expression of Gst alone did not block secretion of Yops (Fig. 4). IPTG-inducible expression of Gst-YscUC/N263A similarly blocked secretion, whereas expression of Gst-YscUC/G270N dramatically reduced the abundance of Yop proteins, YscP, and LcrV (Fig. 4). Together, these results suggest that all three hybrid proteins, Gst-YscUC (wild-type), Gst-YscUC/N263A, and Gst-YscUC/G270N function as dominant-negatives for the Yersinia type III pathway.
Fig. 4.
Expression of wild-type Gst-YscUC and the N263A mutant produce dominant negative effects on type III secretion in Y. enterocolitica W22703. Y. enterocolitica W22703 containing plasmid-encoded gst-yscUC (pKER25), gst-yscUC N263A (pKER64), gst-yscUC (pKER68), and gst (pDA259) were induced for type III secretion in the absence (−) and presence (+) of IPTG. Following centrifugation of culture aliquots, proteins in the supernatant (S) were separated by 12% SDS-PAGE, analyzed by Coomassie blue staining, and immunoblotted with polyclonal antisera specific to YscP, YopR, YopB, YopD, LcrV, YopN, and YopE. Molecular mass markers (in kilodaltons) are indicated to the left of the gel and secreted Yop effectors are indicated to the right.
Interaction of YscUC and its variants with Yersinia type III machines
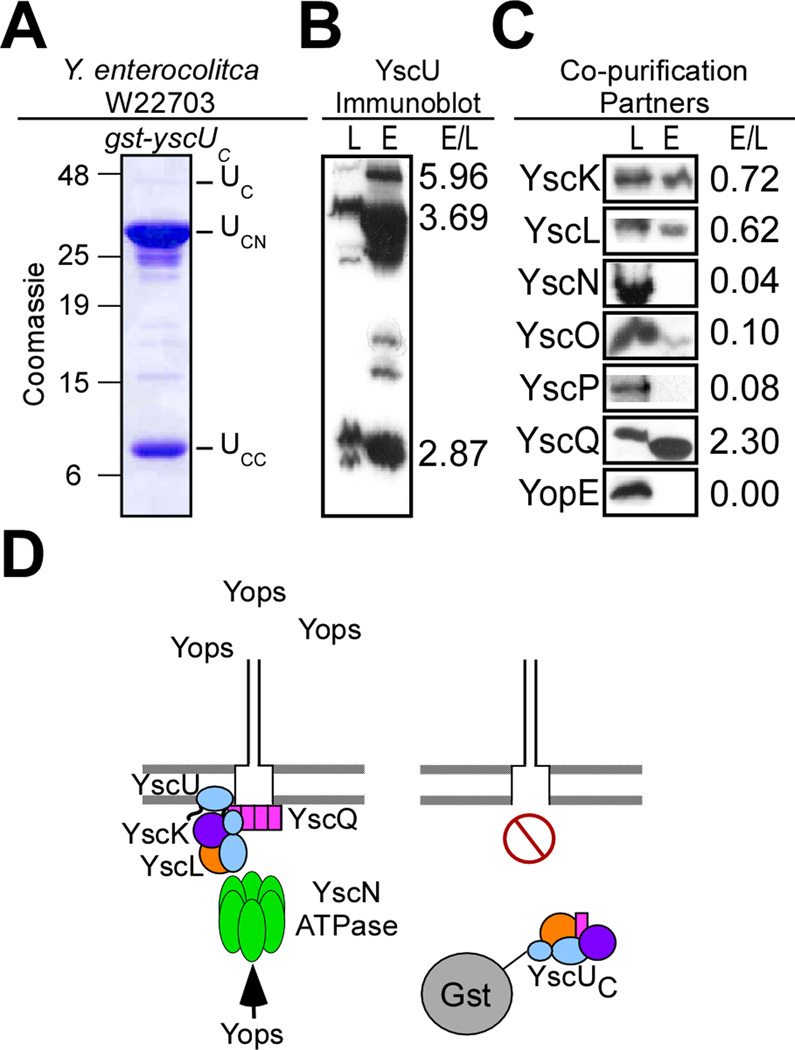
Assuming that specific interactions between YscUC and the type III machine are responsible for blocking secretion, we examined Yersinia lysates harboring the hybrid protein. Crude lysates, generated by French pressure cell disruption of bacteria followed by slow speed removal of unbroken Yersinia, were subjected to sedimentation by ultracentrifugation of bacterial membranes at 100,000 ×g. Cleared lysates, containing the bacterial cytoplasm, were separated from the membrane sediment and protein in both fractions were analyzed for the presence of Gst-YscUC; the hybrid was only detected in the soluble (cytoplasmic) fraction, suggesting that the polypeptide does not interact with type III needle complexes that have assembled in Yersinia membranes (data not shown). Cleared lysates were subjected to affinity chromatography on glutathione S-sepharose (Fig. 5). Coomassie stained SDS-PAGE of eluate revealed three relevant species, full-length Gst-YscUC as well as its auto-cleavage fragments Gst-YscUCN and Gst-YscUCC (Fig. 5AB). The identity of each of these species and their auto-cleavage at N263 were confirmed by mass spectrometry of gel-excised polypeptide species (Fig. S3).
Fig. 5.
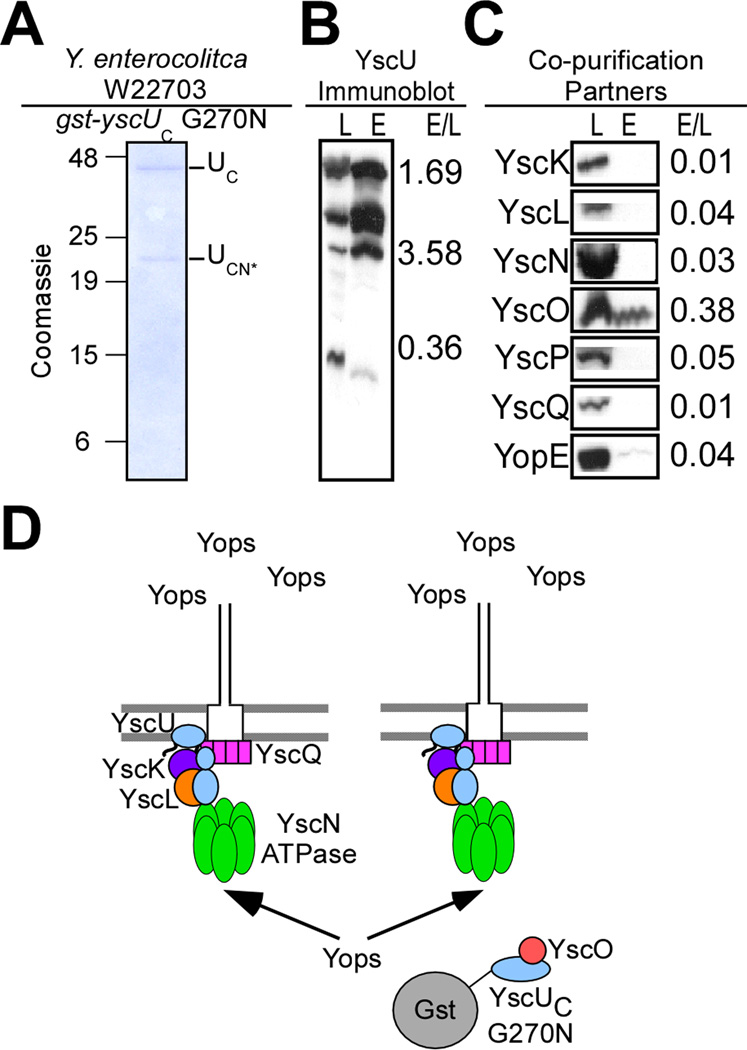
Gst-YscUC is cleaved at N263 and co-purifies in vivo with YscK, YscL, YscQ, and small amounts of YscO. (A) Y. enterocolitica W22703 expressing gst-yscUC (pKER25) under control of the tac promoter was induced for type III secretion in the absence of calcium. Cells were harvested by centrifugation and lysed via a French pressure cell. Samples were centrifuged at 100,000 ×g and separated into lysate supernatant (S) and pellet (P). The lysate supernatant (S) was subject to affinity chromatography on glutathione sepharose. A sample representing the eluate fraction was analyzed by 15% SDS-PAGE and Coomassie blue staining. Three major species in the elution (E) fractions at 42kD, 32kD, and 10kD were analyzed by mass spectrometry and identified as Gst-YscUC (aa 205–354) (UC), Gst-YscUCN (aa205–263) (UCN), and YscUCC (aa264–354) (UCC) respectively. (B) The lysate supernatant (L) and eluate fractions (E) were separated by SDS-PAGE and immunoblotted with antisera specific to YscU. Three species of YscU were detected via immunoblot. (C) For co-purification partners, the lysate supernatant (L) and eluate fractions (E) were separated by SDS-PAGE and immunoblotted with antisera specific to YscK, YscL, YscN, YscO, YscP, YscQ, and YopE. The relative intensity of immunoreactive signals in the lysate and eluate fractions (E/L) is expressed as a ratio to the right of each blot. (D) A model for the capture of machinery components and subsequent dominant negative effect of Gst-YscUC when expressed in wild-type Y. enterocolitica.
Immunoblotting of cleared lysate and eluate fractions for the presence of soluble type III machine components revealed co-purification of YscK, YscL, YscQ, and of very small amounts of YscO (Fig. 5C). Yops, including the translocator class of substrates, did not co-purify with Gst-YscUC and its associated machine components (Fig. 5CD, data not shown). FlhBC interacts directly with the Salmonella hook-length control protein FliK (which presumably does not require a specific chaperone for secretion) and with FliH-FliI, i.e. the homologs of YscL-YscN (Minamino and Macnab, 2000a, b; Minamino et al., 2004). Surprisingly, Gst-YscUC in Yersinia lysates failed to co-purify with either YscP, the needle-length control protein predicted to bind to the C-terminus of YscU or with YscN, the ATPase universally present in all type III machines (Fig. 5C)(Blaylock et al., 2006; Edqvist et al., 2003; Payne and Straley, 1998, 1999; Woestyn et al., 1994).
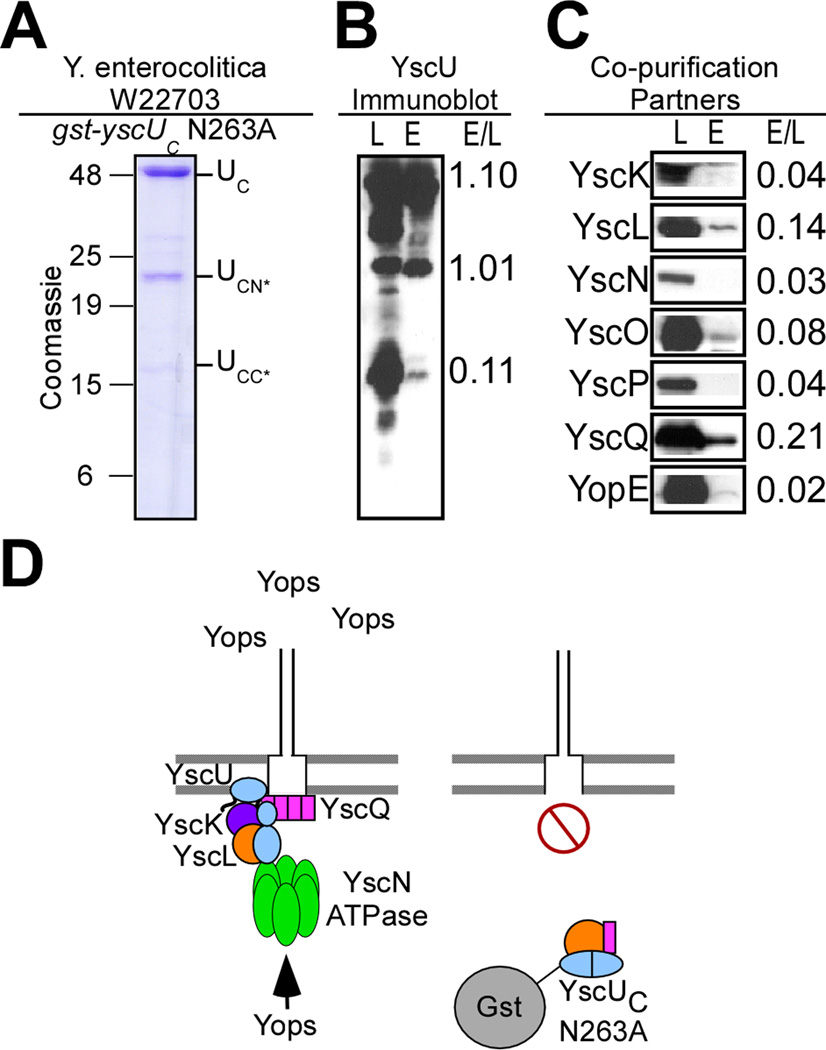
Affinity chromatography of Yersinia cleared lysates harboring Gst-YscUC/N263A revealed purification of the full-length hybrid protein as well as several aberrant degradation products, however Gst-YscUCN/N263A and Gst-YscUCC/N263A were not observed (Fig. 6AB). Some GST-YscUC/N263A was insoluble, and less of this fusion protein purified as compared to GST-YscUC. Despite the decrease in fusion protein purification, affinity chromatography of Gst-YscUC/N263A and immunoblotting of load and eluate fractions revealed an association of small amounts of YscL, YscQ, and YscO (Fig. 6C). Unlike wild-type Gst-YscUC, YscK did not co-purify with Gst-YscUC/N263A (Fig. 6CD).
Fig. 6.
Purification of Gst-YscUC N263A reveals a unique cleavage product of 16kD (YscUCC*) and captures YscL, YscO, and YscQ, but not YscK in vivo. (A) Y. enterocolitica W22703 expressing gst-yscUC N263A (pKER64) under control of the tac promoter was induced for type III secretion in the absence of calcium. Cells were harvested by centrifugation and lysed via a French pressure cell. Samples were centrifuged at 100,000 ×g and separated into lysate supernatant (S) and pellet (P). The lysate supernatant (S) was subject to affinity chromatography on glutathione sepharose. A sample representing the eluate fraction was analyzed by 15% SDS-PAGE and Coomassie blue staining. Three major species in the elution (E) fractions at 42kD, 22kD, and 16kD were present and identified as Gst-YscUC (aa 205–354) (UC), Gst-YscUCN* (UCN*), and YscUCC* (UCC*) respectively. (B) The lysate supernatant (L) and eluate fractions (E) were separated by SDS-PAGE and immunoblotted with antisera specific to YscU. Two new species of YscU were detected via immunoblot. (C) The lysate supernatant (L) and eluate fractions (E) were separated by SDS-PAGE and immunoblotted with antisera specific to YscK, YscL, YscN, YscO, YscP, YscQ, and YopE. The relative intensity of immunoreactive signals in the lysate and eluate fractions (E/L) is expressed as a ratio to the right of each blot. (D) A model for the capture of machinery components and the subsequent dominant negative effect of Gst-YscUC N263A when expressed in wild-type Y. enterocolitica.
Finally, affinity chromatography of Gst-YscUC/G270N revealed the presence of full-length polypeptide as well as degradation products including Gst-YscUCN*/G270N and YscUCC*/G270N (Fig. 7AB). Gst-YscUCN/G270N and Gst-YscUCC/G270N were not observed (Fig. 7AB). Purification was hindered as the majority of this polypeptide was insoluble (data not shown). This suggests a role for amino acid G270 not only in cleavage of YscU but also in solubility and perhaps proper folding of the C-terminal domain. Despite its low yield, affinity chromatography of Gst-YscUC/G270N revealed co-purification with YscO (Fig. 7C). In contrast to Gst-YscUC or Gst-YscUC/N263A, an association of the hybrid with YscK, YscL, and YscQ was not observed (Fig. 7C). As a control, affinity chromatography of Gst alone did not yield co-purification of Yops or Yersinia type III machinery components (Fig. S4).
Fig. 7.
Gst-YscUC G270N co-purifies in vivo with YscO, but not YscK, YscL or YscQ. (A) Y. enterocolitica W22703 expressing gst-yscUC G270N (pKER68) under control of the tac promoter was induced for type III secretion in the absence of calcium. Cells were harvested by centrifugation and lysed via a French pressure cell. Samples were centrifuged at 100,000 ×g and separated into lysate supernatant (S) and pellet (P). The lysate supernatant (S) was subject to affinity chromatography on glutathione sepharose. A sample representing the eluate (E) fraction was collected and analyzed by 15% SDS-PAGE and Coomassie blue staining. Two major species in the elution (E) fractions at 42kD and 23kD were identified as Gst-YscUC (aa 205–354) (UC) and Gst-YscUCN* (UCN*). (B) The lysate supernatant (L) and eluate fractions (E) were separated by SDS-PAGE and immunoblotted with antisera specific to YscU. Three species of YscU were detected on immunoblot. (C) The lysate supernatant (L) and eluate fractions (E) were separated by SDS-PAGE and immunoblotted with antisera specific to YscK, YscL, YscN, YscO, YscP, YscQ, and YopE. The relative intensity of immunoreactive signals in the lysate and eluate fractions (E/L) is expressed as a ratio to the right of each blot. (D) A model for the capture of YscO by Gst-YscUC G270N when expressed in wild-type Y. enterocolitica.
G270N abolishes YscUC binding to YscL
In accordance with the widely accepted view that YscU represents a switch for secretion from early to middle and then late substrates, we presume that physical association of type III machine components with YscU may be a dynamic process that could be impacted by YscU auto-cleavage (Edqvist et al., 2003; Sorg et al., 2007). Affinity chromatography of Gst-YscUC from Yersinia cleared lysates revealed small amounts of full-length polypeptide as well as the auto-cleavage products, Gst-YscUCN and Gst-YscUCC. We wondered whether YscK, YscL, YscQ, and YscO, i.e. the type III components that co-purified with Gst-YscUC, bound to the full-length hybrid or its auto-cleavage products, thus enabling the examination of machine dynamics at different stages of substrate recognition. However, attempts to separate Gst-YscUC from Gst-YscUCN/CC were unsuccessful as the full-length species promotes auto-cleavage. We therefore examined Gst-YscUC (both full-length hybrid and Gst-YscUCN/CC) for its properties of in vitro binding to purified type III machine components and compared this with Gst-YscUC/N263A and Gst-YscUC/G270N, which accumulate variant full length hybrids. His-tagged type III machine components were purified by affinity chromatography (Fig. 8A) and added to glutathione-S-sepharose beads that had been pre-charged with Gst-YscUC, Gst-YscUC/N263A, or Gst-YscUC/G270N. As a control, incubation of glutathione-sepharose beads pre-charged with Gst alone with YscK, YscL, YscN, YscQ, LcrV, and YopE did not cause co-sedimentation of secretion machinery components (Fig. 8B). Analysis of His-YscO, revealed that this protein bound non-specifically to beads charged with Gst alone; we were therefore unable to examine the association of YscU and YscO in vitro (data not shown). Gst-YscUC, i.e. a mixture of full-length and Gst-YscUCN/CC, co-sedimented with each of the three type III machine components (YscK, YscL, and YscQ) that had been identified as in vivo ligands for the wild-type hybrid (Fig. 8B). A similar result was obtained for Gst-YscUC/N263A (Fig. 8B). In contrast, Gst-YscUC/G270N lost the ability to bind YscL, as only trace amounts of this machine component could be detected in the glutatione-sepharose sediment (Fig. 8B). Nevertheless, Gst-YscUC/G270N retained the property of binding YscK and YscQ (Fig. 8B). As His-YscL was the only protein with differential binding properties, we wondered whether purified His-YscL was functional in the context of the Yersinia type III secretion pathway. pKER154, expressing His-yscL, was able to complement the Y. enterocolitica ΔyscL mutant for secretion of early, middle, and late substrates even in the absence of induction by IPTG (Fig. S5).
Fig. 8.
Unlike Gst-YscUC wild-type and N263A, the G270N mutant does not capture His-YscL in vitro. (A) E. coli expressing His-yscK, His-yscL, His-yscN, His-yscQ, His-lcrV, and yopE-His were induced with 1 mM IPTG for expression of the fusion proteins. Cells were harvested by centrifugation and lysed via a French pressure cell. Samples were centrifuged at 100,000 ×g and separated into lysate supernatant (S) and pellet (P). Histidine tagged proteins were purified under denaturing conditions, separated by 15% SDS-PAGE and detected with Coomassie blue staining. Full length products are denoted by an arrow. The positions of molecular weight markers (in kilodaltons) are indicated to the left of the gel. (B) Wild-type, N263A, and G270N Gst-YscUC protein and Gst alone (10 µg charged to glutathione S-sepharose beads) were incubated with 10 µg each of His-YscK, His-YscL, His-YscN, His-YscQ, His-LcrV, and YopE-His. Samples were centrifuged and the supernatant precipitated with methanol-chloroform (unbound, U). Beads were washed three times with PBS and sedimented (bound, B). Both fractions were separated and analyzed by SDS-PAGE followed by immunoblot with polyclonal antisera to YscK, YscL, YscN, YscQ, LcrV, and YopE. Percent bound (%B) was calculated to be the relative intensity of immunoreactive signals in the bound fraction as compared to the total amount of immunoreactive protein (B/B+U) and is expressed as a percentage to the right of each blot.
Macnab and colleagues reported that purified FlhBC binds to both purified FliI and FliH, however our in vivo affinity chromatography approach failed to detect association of YscN (the Yersinia homolog of FliI) with complexes formed on Gst-YscUC (Fig. 5C) (Blaylock et al., 2006; Minamino and Macnab, 2000a, b; Minamino et al., 2003). To test whether isolated YscN binds directly to YscU, we used purified His-YscN and incubated the protein with pre-charged glutathione S-sepharose beads. YscN bound Gst-YscUC, Gst-YscUC/N263A and Gst-YscUC/G270N (Fig. 8B). One simple explanation for this observation is that the in vivo association of YscN with Gst-YscUC may be hindered by the formation of type III machine complexes that include YscK, YscL, and YscQ (Jackson and Plano, 2000). Of note, His-YscN has been previously demonstrated to complement ΔyscN mutant Y. enterocolitica for type III secretion (Blaylock et al., 2006).
Discussion
Assembly and function of the bacterial basal-body and flagellar filament involves a large number of genes and a wide spectrum of biological mechanisms, ranging from gene expression, protein secretion and assembly, to morphogenetic events. Nine polypeptides of the basal body / flagellar type III machine complex represent homologs of bacterial type III secretion machines; their needle complexes are comprised of 5–6 proteins and function solely as a conduit for the transport of effector proteins into host cells (Blocker et al., 2001; Blocker et al., 2003; Hueck, 1998; Kubori et al., 1998). Most secretion substrates of the basal body complex represent proteins that are ultimately incorporated into a supra-molecular filament that functions as a rotary motor (Macnab, 2004). The precise location of these substrate proteins can be discerned by electron microscopy of flagella derived from specific mutants (Ferris et al., 2005; Minamino et al., 1999; Williams et al., 1996). Importantly, waves of regulated gene expression control the availability of proteins as substrates for secretion and assembly (Karlinsey et al., 2000; Macnab, 2004). Such regulatory control likely directs the flagellar machine to distinguish between different substrates. Notably, once activated by Yersinia contact with host cells, expression of type III secretion machine components and substrates does not appear to be regulated. Type III needles are assembled and operated in a non-synchronous manner, suggesting that substrate recognition of each machine complex must be controlled by specific morphogenetic events at each individual needle complex (Sorg et al., 2006).
FliK, the hook length control protein, binds to FlhBC, the cytoplasmic domain of the switch protein (Minamino and Macnab, 2000b; Minamino et al., 2004; Moriya et al., 2006). A molecular mechanism that activates FlhBC auto-cleavage has thus far not been revealed, however current models suggest that the physical interaction between FliK and FlhBC, perhaps the secretion of FliK or the severance of FliK interaction with FlhBC play important roles in flipping the switch (Ferris and Minamino, 2006; Minamino et al., 2004; Minamino et al., 2006; Moriya et al., 2006). Prior to switching, FlhBC is thought to direct rod and hook substrates into the type III pathway. Once these structures have been assembled and the switch has been flipped, FlhBC promotes the secretion of flagellin (Fraser et al., 2003; Minamino and Macnab, 2000b). Previous work also revealed physical association between FlhBC, various rod, hook, or flagellin substrates as well as the FliI ATPase complex, including the regulator FliH and the general chaperone FliJ (Minamino et al., 2003; Zhu et al., 2002). These biochemical studies could not establish which associations between FlhB and machine or substrate components occur before and which occur after FlhB auto-cleavage. Thus, there is still uncertainty whether FlhB auto-cleavage represents a collateral phenomenon of the switch or whether auto-cleavage is the morphogenetic event and substrate specificity switch of type III machines. Substrate switching of flagellar type III machines in Escherichia coli and Salmonella typhimurium also involves Flk, a membrane protein that controls secretion of the transcription factor FlgM, however similar complexity does not seem to exist in Yersinia as these species do not express flk homologs (Aldridge et al., 2006; Karlinsey et al., 1997; Karlinsey et al., 1998).
Recent work in the field of Yersinia type III machines led to new advances in the general appreciation of the aforementioned switch. Similar to its FlhB homolog, YscU also promotes auto-cleavage (Edqvist et al., 2003; Lavander et al., 2002; Sorg et al., 2007). Substitutions of the conserved residues at the cleavage site of YscU affect substrate recognition and assembly of needle structures, which also lack LcrV at their tip (Sorg et al., 2007). Although previous work suggested that yscU N263A mutants are unable to insert needles into red blood cells, data presented here indicate that the N263A mutant, while defective in its ability to secrete large amounts of Yops, is still capable of effector transport into HeLa cells (a process that requires the secretion of LcrV, YopB and YopD)(Sorg et al., 2007). Although impaired in auto-cleavage, the YscU N263A variant must be able to secrete substrates and enable parts of the physiological sequence of transport reactions. Thus, in contrast to FlhBN269A, which promotes only assembly of poly-hook structures, YscUN263A can be viewed as being partially defective for switching substrate specificity.
We sought to identify yscU mutants that were completely defective in their ability to promote type III secretion and isolated two alleles, G127D and G270N. One of the mutants, G127D, promoted auto-cleavage, while the other (G270N) did not. We also observed that Gst-YscUC, similar to FlhBC, functions as a dominant-negative for type III secretion (Ferris et al., 2005; Minamino and Macnab, 2000b). Informed by affinity chromatography and immune detection of machine components, we were able to identify molecules that physically interact with YscU before and after its auto-cleavage.
We report that Gst-YscUC (carrying the G270N substitution) binds YscO. YscO is a secreted machinery component (Payne and Straley, 1998) that may be involved in the secretion of YscP (Payne and Straley, 1999), an early substrate (K. Riordan, unpublished observation). Preparations of wild-type Gst-YscUC, which are largely comprised of Gst-YscUCN/CC (and also contain small amounts of full-length Gst-YscUC) bound to YscL and to the ATPase regulatory complex, YscK-YscL-YscQ (Blaylock et al., 2006; Jackson and Plano, 2000). Gst-YscUC/G270N was unable to bind YscL in vivo or in vitro. Gst-YscUC/G270N also failed to associate with YscK-YscL-YscQ. The observation that Gst-YscUC/G270N binds purified YscK, YscN, and YscQ in vitro (but not to YscK-YscL-YscQ in vivo) may be due to formation of pre-assembled complexes that are offered to this machine component. Informed by these results, we currently entertain a model whereby association of full-length YscU with YscO leads to recruitment of YscP and to assembly of needles with proper length. Once needle assembly has been completed, which appears to involve YscP and YscO secretion, YscU may promote auto-cleavage and association with YscL. Recruitment of YscK-YscL-YscQ presumably allows the YscN ATPase to fuel the secretion of middle and late substrates. Although this is not demonstrated by data presented here, we speculate that YscU auto-cleavage may prevent association of the switch protein with YscO. Future work will need to test the molecular predictions that can be derived from this model (Fig. 9).
Fig. 9.
A model for the role of YscU in the substrate specificity switch of Y. enterocolitica type III secretion machines. Uncleaved YscU is represented by two blue ovals signifying the YscUTM domain and uncleaved YscUC. Cleaved YscU is represented by three ovals signifying YscUTM, YscUCN, and YscUCC. It is proposed that YscO, binds to uncleaved YscUC allowing for secretion of early substrates, including the needle protein YscF, YscP, and YopR (YscH). Once needle assembly is completed and the needle sized by the YscP protein, YscU is signaled to undergo auto-cleavage at which point YscO dissociates from YscUC and may be secreted. Cleaved YscUCN+CC then recruits YscL to the base of the needle apparatus. YscL serves as the tether between YscUCN+CC and the YscK-YscQ-YscN ATPase complex.
Experimental Procedures
Bacterial strains, media, and manipulations
Y. enterocolitica W22703 (Cornelis and Colson, 1975) was propagated in tryptic soy broth or agar (TSB/TSA) at 26°C. Antibiotics were added when appropriate to a final concentration of 35 µg/mL chloramphenicol or 35 µg/mL kanamycin. Escherichia coli strains were propagated at 37°C in Luria-Bertani (LB) broth or agar supplemented with 35 µg/mL chloramphenicol or 35 µg/mL kanamycin with the exception of E. coli strain CR202, which was grown at 30°C (Roncero et al., 1990). Y. pestis KIM D27, a KIM5 (non-pigmented) variant of Y. pestis biovar mediaevalis KIM lacking the 102 kb pgm locus, was grown in heart infusion broth or agar (HIB/HIA) at 26°C (Brubaker, 1969). Media were supplemented with 20 mM MgCl2 and 20 mM oxalic acid (HIA/MgOx) for low calcium conditions and 35 µg/mL chloramphenicol for plasmid maintenance. Standard methods for transformation of plasmids into E. coli, Y. pestis, and Y. enterocolitica were employed.
Construction of Y. enterocolitica yscU mutant (∆51–205)
The yscU in-frame deletion strain, CT-132, was created as follows. The pYV227 plasmid, isolated from Y. enterocolitica W22703, was used to amplify 1 kb fragments upstream and downstream of the yscU gene using the following primers: YscU del 1kb up F (5`-AAGAGCTCGGTGCTAGTCCTTTCAATGC-3`), YscU del 1kb up R (5`-AAAGATCTGAAATAGTAGTCAGAAAGC-3`), YscU del 1kb down F (5`-AAAGATCTGAATACTATCAATATATTAAGGAACT-3`), and YscU del 1kb down R (5`-AAGTCGACTTATAACATTTCGGAATGTTGTTTCTC-3`) (restriction sites denoted in bold). The 1 kb upstream and downstream fragments were ligated into pLC28 previously digested with SacI and SalI and transformed into E. coli mating strain S17.1 λpir+ (Cheng et al., 1997). Following the allelic exchange approach previously described, the resulting strain, CT-132, resulted in an in-frame deletion of the yscU gene that retains amino acids 1–50 and 206–354 with an intervening BglII site (Cheng et al., 1997). CT-132 does not express a functional yscU gene product.
Plasmids
For the construction of pCT-143, the yscU allele was amplified from the virulence plasmid pYV227 of Y. enterocolitica W22703 with primers (5`-AAACATATGAGCGGAGAAAAGACAGAGCA-3`) and (5`-AAAGGATCCTAACATTTCGGAATGCTGTTTCTCGAT-3`) and cloned into pCR2.1 (Invitrogen). The resulting plasmid was digested with NdeI and BamHI and the fragment ligated into pDA37, a derivative of pHSG575 (Takeshita et al., 1987), which was modified to include the tac promoter prior to an engineered NdeI-BamHI cloning site (Anderson and Schneewind, 1999) yielding a yscU allele that was IPTG (Isopropyl β-D-1-thiogalactopyranoside) inducible. For the construction of pKER25 expressing gst-yscUC, amino acids 205–354 of yscU were amplified with primers BamHI-YscUc F (5`-AAGGATCCTTTGAATACTATCAATATATTAAGG-3`) and YscUc-EcoRI R (5`-AAGAATTCTTATAACATTTCGGAATGCTGTT-3`). This fragment was ligated into pCR2.1 (Invitrogen) and digested with BamHI and EcoRI, as was pKER17 (Blaylock et al., 2006), a modified version of pGEX-2TK (GE Healthcare) that contains the neomycin phosphotransferase (npt) gene for kanamycin resistance. The resulting plasmid, pKER25, expressed gst-yscUC. For the construction pgst, glutathione S-transferase (gst) was amplified from pGEX-2TK using primers that were flanked by NdeI and BamHI restriction sites. gst was ligated into pHSG576 (a low copy, pHSG576/pSC101 derivative modified to contain the tac promoter), yielding pDA259 encoding for IPTG inducible expression of gst (Takeshita et al., 1987). All DNA sequences of mutant alleles were confirmed by the University of Chicago Cancer Research Center DNA sequencing facility.
For the construction of His-YscK, the yscK allele was amplified from the virulence plasmid of pYV227 Y. enterocolitica W22703 with primers (5`-AACATATGATGATGGAAAATTATATTACCTCT-3`) and (5`- AAGGATCCTTATTTGAACAAATGGCTGCAT-3`) and cloned into pCR2.1. The resulting plasmid was digested with NdeI and BamHI and the fragment ligated into pET16b (Novagen) to create pKER21. For the construction of His-YscQ, the yscQ allele was amplified from the virulence plasmid of pYV227 Y. enterocolitica W22703 with primers (5`- AACATATGATGAGTTTGTTAACCTTGCCA-3`) and (5`- AAGAATCCTCATGAAATCGTAACCTCTGT-3`) and cloned into pCR2.1. The resulting plasmid was digested with NdeI and BamHI and the fragment ligated into pET16b (Novagen) to create pKER26. For the construction of His-YscO, the yscO allele was amplified from the virulence plasmid of pYV227 Y. enterocolitica W22703 with primers (5`- AACATATGATACGCCGCCTGCACCG-3`) and (5`- AAGGATCCTTAGGCGTTCCTGTGATGCTGT -3`) and cloned into pCR2.1. The resulting plasmid was digested with NdeI and BamHI and the fragment ligated into pET16b (Novagen) to create pKER120. Construction of YopE-His, His-LcrV, and His-YscN was previously reported (Blaylock et al., 2006; Cheng and Schneewind, 1999; Overheim et al., 2005).
Site-Directed Mutagenesis
To introduce the N263A mutation into the plasmid expressing full length yscU, pCT-143 expressing yscU was used as a template. Site-directed mutagenesis was carried out according to the QuikChange ® Site-Directed Mutagenesis Kit (Stratagene). The primers used to make pyscU N263A were: (5`-TCATCAGTGGTGGTAGCTGCGCCGACCCATATTGCTATT-3`) and (5`- AATAGCAATATGGGTCGGCGCAGCTACCACCACTGATGA-3`). To introduce mutations N263A and G270N into the C-terminal domain of yscU, pKER25 expressing gst-yscUC was used as a template. The primers used to make the mutations to generate pKER64 (gst-yscUC N263A) were (5`-TCATCAGTGGTGGTAGCTGCGCCGACCCATATTGCTATT-3`) and (5`-AATAGCAATATGGGTCGGCGCAGCTACCACCACTGATGA-3`) and to generate pKER68 (gst-yscUC G270N) were (5`-CCGACCCATATTGCTATTAATATTCTTTATAAGCGA-3`) and (5`-TCGCTTATAAAGAATATTAATAGCAATATGGGTCGG-3`) (mutation sites underlined). All sequences were confirmed by the University of Chicago Cancer Research Center DNA sequencing facility.
Creation of Y. pestis KIM D27 ∆yscU strain CHI110
E. coli strain CR202 containing transposon Mucts62pAp5hp1 (also called MuAphP1, a Mu-P1 hybrid) (Roncero et al., 1990) was grown overnight in Luria Broth (LB) containing 50 µg/mL ampicillin. To obtain lysates, CR202 was diluted 1:50 into HIB supplemented with 2.5 mM of both MgCl2 and CaCl2 and grown at 30°C for 1 hour 45 minutes. E. coli CR202 was then shifted to 42°C for 2 hours to induce lysis. This lysate was centrifuged, sterilized by passage through a 0.2 µm filter, and stored on ice. Overnight Y. pestis KIM D27 culture was diluted 1:25 into HIB containing 2.5 mM of both MgCl2 and CaCl2 and grown for 3 hours at 26°C. Y. pestis KIM D27 was harvested by centrifugation, suspended in 10 mM MgSO4, and incubated at room temperature for 20 minutes. For infection, 5 mL of E. coli CR202 Mu lysate was added to 2.5 mL of Y. pestis KIM D27 and incubated at room temperature for 20 minutes. The cells were harvested by centrifugation, suspended in 10 mM MgSO4, and plated on blood base (BB) agar containing 20 mM MgCl2, 20 mM oxalic acid, and 50 µg/mL ampicillin (BB/MgOx agar). Plates were grown at 33°C (determined to be the optimal temperature) to select for Y. pestis KIM D27 LCR- mutants which grow at 37°C in the absence of calcium. Genomic DNA was harvested from each mutant and digested with either TaqαI or BspHI. Inverse PCR was performed using primers complementary to the left (BfaI-LT, BfaI-RT) and right (TaqI-LT, TaqI-RT) ends of Mu: BfaI-LT (5`-TGCACTACAGGCTTGCAAGCCCCAC-3`), BfaI-RT (5’-AGCTGGTTCATGGTTGTTATACGGC-3’), TaqI-LT (5`-CCGTAAATACACGCCTCGTTGTCCA-3`), and TaqI-RT (5’-CCATACCCTAAGTGATCCCCATGTA-3`). PCR products were sequenced by the University of Chicago Cancer Research Center DNA sequencing facility. CHI110 contains the Mu transposon inserted after the 99th nucleotide (position 13308) of the yscU gene (13209–14273) on pCD1 which disrupts the function of the yscU gene rendering CHI110 functionally deficient in type III secretion.
Screen for non-functional yscU mutations
pCT-143 containing the yscU gene under the control of the IPTG inducible tac promoter was subjected to hydroxylamine (HA) mutagenesis. Fresh 1 M NH2OH (hydroxylamine) pH 6.0 was prepared and mixed in a 1:1 v/v ratio with freshly harvested pCT-143. The solution was incubated at 68°C for 2 hours after which time the DNA was harvested using QIAquick PCR purification (Qiagen). 10 µL of HA-treated pCT-143 was transformed into Y. pestis KIM D27 that had been diluted 1:20 into 20 mL of HIB and grown for 3 hours at 26°C. Transformants were recovered at 26°C for 2 hours and plated on HIA containing 35 µg/mL chloramphenicol for selection. After 2 days, colonies were selected and patched on HIA/MgOx plates containing 35 µg/mL of chloramphenicol and grown at 26°C and 37°C. Colonies that grew at 37°C (phenotype LCR−) were selected. The yscU gene was amplified using primers (5-AAACATATGAGCGGAGAAAAGACAGAGCA-3`) and (5`- AAAGGATCCTAACATTTCGGAATGCTGTTTCTCGAT-3`) and purified using QIAquick PCR purification. PCR products were sequenced at the University of Chicago Cancer Research Center DNA sequencing facility and mutations confirmed by comparison with the native yscU sequence.
Digitonin Fractionation of Yersinia-Infected HeLa Cells
HeLa cells were grown in Dulbecco’s Modified Eagle Medium (DMEM) supplemented with 10% fetal bovine serum (FBS) and 2 mM glutamine at 37°C in 5% CO2. Two hours prior to infection, overnight yersinia cultures were diluted 1:50 into fresh TSB, containing 35 µg/mL chloramphenicol when appropriate. Ten minutes prior to infection, 1 mM IPTG was added to induce expression of indicated genes. One hour prior to infection, 90% confluent HeLa cells were washed twice with 1 × PBS and 10 mL of Optimem medium and 1 mM IPTG were added. HeLa cells were infected with indicated yersinia strains at an MOI of 10. Three hours following infection, the tissue culture medium (M) was decanted and centrifuged at 15,000 rpm for 15 minutes. Proteins in 7 mL of supernatant (S) were precipitated with methanol/chloroform and the rest of the supernatant discarded. Pellets (P) were suspended in 10 mL of 1% SDS in 1 × PBS and 7 mL of the pellet (P) fraction was precipitated with methanol/chloroform. 1% digitonin (D) in 1 × PBS was added to the tissue cultures cells and their attached bacteria for 20 minutes at room temperature to disrupt HeLa cell plasma membranes. Following HeLa cell disruption, cell remnants were detached from the flasks using a cell scraper and digitonin lysates (D) were centrifuged and proteins in the supernatant (S) and pellet (P) processed as described above. Proteins were separated on 15% SDS PAGE gels, transferred to PVDF membranes, and immuno-blotted using specific polyclonal anti-sera to YopR, YopE, YopH, YopN, YopB, YopD, LcrV, IκB, and RpoA.
Cytotoxicity assay
HeLa cell tissue cultures, 3 ×105 cells, were grown in 12 well tissue culture plates and infected as described above. After three hours of infection, the media was removed and the cells fixed for 20 minutes in a solution of 3.7% formaldehyde in 1 × PBS. The fixation was quenched with 0.1 M glycine in PBS. Cells were permeabilized with 0.1% Triton-X 100 in 1 × PBS for 30 minutes at 4°C. Cells were washed and blocked for 15 minutes with 1 × PBS containing 0.05% Tween-20 and 5% skim milk. Actin filaments were labeled with 99 nM (3U) of Rhodamine-phalloidin (Flexa) for 20 minutes at room temperature. The labeling solution was removed and each well washed four times with 1 mL of 1 × PBS. Cells were visualized with a Nikon TE2000-U inverted microscope. Rhodamine visualization was achieved through excitation at 591 nm and emission at 608 nm. Images were captured with a Cascade 1K CCD camera.
Type III secretion
Overnight Yersinia cultures were diluted 50 fold into fresh TSB supplemented with antibiotic, 5 mM EDTA (−Ca2+) or 5 mM CaCl2 (+Ca2+) and grown for 2–3 hours at 26°C. Cultures were then shifted to 37°C for 3 hours to induce type III secretion. Where indicated, IPTG was added to cultures to induce expression of protein products. Secretion assays were conducted as described elsewhere (Cheng et al., 1997).
Protein electrophoresis and immunodetection
Proteins were resolved by electrophoresis on 12% or 15% sodium dodecyl sulfate (SDS)-polyacrylamide gels (PAGE). For immunoblots, proteins were transferred to polyvinylidene difluoride membranes (Millipore) and probed with anti-sera as described previously (Cheng et al., 1997).
Protein purification
For purification of Gst-YscUC, Gst- YscUC N263A, Gst- YscUC G270N, and Gst Y. enterocolitica W22703 containing pKER25, pKER64, pKER69, and pDA259, respectively, were grown overnight in TSB media containing 35 µg/mL kanamycin or chloramphenicol. Overnight cultures were diluted 50 fold into 1 L of M9 minimal media containing 5 mM EDTA and 35 µg/mL kanamycin or chloramphenicol and grown at 26°C for 3 hours (OD600nm 0.6). Upon shift to 37°C for 3 hours, 1 mM IPTG was added to induce expression of hybrid proteins. The cultures were harvested by centrifugation at 7000 ×g for 10 min and the bacterial sediment was suspended in 20 mL of phosphate-buffered saline (PBS). Samples were passed through a French pressure cell at 16,000 lbs/in2 twice and the lysate clarified by ultracentrifugation at 100,000 ×g for 30 minutes. Clarified lysate was loaded onto a 1 mL glutathione sepharose column (GE Healthcare) pre-equilibrated with PBS. The column was then washed with 10 column volumes of PBS and bound proteins were eluted with 10 mM glutathione in PBS. Concentrations of the GST-YscUC proteins bound to the beads were determined by removing a small aliquot of beads and boiling in 10% SDS. Samples were then centrifuged at 13,000 ×g for 5 minutes, the supernatant removed and protein concentrations determined using the DC Protein Assay (Bio-Rad).
For purification of His-YscK, His-YscL, His-YscN, His-YscO, His-YscQ, His-LcrV, and YopE-His, E. coli BL21(DE3) containing plasmids pKER21 (His-YscK), pKER14 (His-YscL), pBB7 (His-YscN), pKER120 (His-YscO), pKER26 (His-YscQ), prLcrV (His-LcrV) (Overheim et al., 2005), or pLC164 (YopE-His) (Cheng and Schneewind, 1999) were inoculated in LB containing 100 µg/mL ampicillin. Overnight cultures were diluted 50 fold into 1 L of LB containing 100 µg/mL ampicillin and grown at 37°C for 3 hours (OD600nm 0.6). 1 mM IPTG was then added and the cultures grown for an additional 3 hours to induce expression of hybrid proteins. Bacteria were harvested by centrifugation of cultures at 7000 ×g for 10 min and bacterial sediment was suspended in 20 mL lysis buffer (6 M urea, 150 mM NaCl, 2 mM CaCl2, 50 mM Tris-HCl, pH8.0). All proteins were purified from E. coli BL21(DE3) under denaturing conditions and refolded according to a previously established protocol (Blaylock et al., 2006). Protein concentrations of all histidine tagged proteins were determined using the DC Protein Assay (Bio-Rad).
In vitro binding assay
For the in vitro binding assay, E. coli CA8000 containing all three GST-YscUC fusion proteins as well as a Gst control was grown in 1 L LB containing 35 µg/mL of kanamycin or chloramphenicol and prepared as stated above for protein purification. Each was loaded onto a 1 mL glutathione-S-sepharose column and washed with 10 column volumes of PBS. Following the wash step, the beads were harvested and stored for further use. Following the determination of the concentration of each protein on the beads, charged beads were mixed with uncharged beads to ensure a final concentration of 10 µg of Gst-tagged protein. 500 µL of bead volume containing 10 µg of Gst-tagged protein was then mixed with 500 µL of 1% TritonX-100 in PBS and rotated at 4°C for 1 hour. These beads were harvested following centrifugation at 13,000 ×g for 15 minutes. 10 µg of Gst-YscUC, Gst-YscUC N263A, Gst-YscUC G270N, or Gst was mixed with 10 µg of purified His-YscK, His-YscL, His-YscQ, His-YscO, His-YscN, His-LcrV, or YopE-His and incubated in a total volume of 1 mL with 1%Triton X-100 in PBS. Samples were rotated at 4°C for 1 hour at which point they were centrifuged at 13,000 ×g for 15 minutes. The supernatant was removed (unbound, U) and precipitated using a methanol-chloroform extraction protocol (Marraffini and Schneewind, 2005). Supernatants were then suspended in 200 µL of sample buffer (1 M Tris-HCl pH 6.8, 10% SDS, 20% glycerol, 4% β-mercaptoethanol, and bromophenol blue). The sedimented pellets (Bound, B) were washed 3 times with PBS followed by a final centrifugation at 13,000 ×g for 15 minutes. 100 µL of sample buffer was added to equal volume of the beads and the unbound (U) and bound (B) fractions were visualized by SDS-PAGE followed by immunoblot with anti-sera to YscK, YscL, YscN, YscQ, LcrV, and YopE respectively.
Supplementary Material
Acknowledgements
We thank Bill Blaylock and Joseph Sorg for advice, and members of our laboratory for critical comments on this manuscript. We thank Luisa Cheng, Melanie Marketon, and Christina Tam for ΔyscU mutants in Y. enterocolitca and Y. pestis and for plasmids. We appreciate the assistance of Ross Tomaino and the Taplin Mass Spectrometry Facility at Harvard University with mass spectrometry experiments. This work has been supported by a grant from the National Institute of Allergy and Infectious Diseases, Infectious Diseases Branch AI42797 (to O.S.). K.E.R. was a trainee of the NIH Medical Scientist Training Program at The University of Chicago (GM07281). O.S. acknowledges membership within and support from the Region V “Great Lakes” Regional Center of Excellence in Biodefense and Emerging Infectious Diseases Consortium (GLRCE, National Institute of Allergy and Infectious Diseases Award 1-U54-AI-057153).
References
- Agrain C, Sorg I, Paroz C, Cornelis GR. Secretion of YscP from Yersinia enterocolitica is essential to control the length of the injectisome needle but not to change the type III secretion substrate specificity. Mol. Microbiol. 2005;57:1415–1427. doi: 10.1111/j.1365-2958.2005.04758.x. [DOI] [PubMed] [Google Scholar]
- Aldridge P, Karlinsey JE, Becker E, Chevance FF, Hughes KT. Flk prevents premature secretion of the anti-sigma factor FlgM into the periplasm. Mol. Microbiol. 2006;60:630–643. doi: 10.1111/j.1365-2958.2006.05135.x. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Allaoui A, Woestyn S, Sluiters C, Cornelis G. YscU, a Yersinia enterocolitica inner membrane protein involved in Yop secretion. J. Bacteriol. 1994;176:4534–4542. doi: 10.1128/jb.176.15.4534-4542.1994. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Allaoui A, Schulte R, Cornelis GR. Mutational analysis of the Yersinia enterocolitica virC operon: characterization of yscE, F, G, I, J, K required for Yop secretion and yscH encoding YopR. Mol. Microbiol. 1995;18:343–355. doi: 10.1111/j.1365-2958.1995.mmi_18020343.x. [DOI] [PubMed] [Google Scholar]
- Anderson DM, Schneewind O. A mRNA signal for the type III secretion of Yop proteins by Yersinia enterocolitica. Science. 1997;278:1140–1143. doi: 10.1126/science.278.5340.1140. [DOI] [PubMed] [Google Scholar]
- Anderson DM, Schneewind O. Yersinia enterocolitica type III secretion: an mRNA signal that couples translation and secretion of YopQ. Mol. Microbiol. 1999;31:1139–1148. doi: 10.1046/j.1365-2958.1999.01254.x. [DOI] [PubMed] [Google Scholar]
- Anderson DM, Ramamurthi KS, Tam C, Schneewind O. YopD and LcrH regulate the expression of Yersinia enterocolitica YopQ at a post-transcriptional step and bind to yopQ mRNA. J. Bacteriol. 2002;184:1287–1295. doi: 10.1128/JB.184.5.1287-1295.2002. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Blaylock B, Riordan KE, Missiakas DM, Schneewind O. Characterization of the Yersinia enterocolitica type III secretion ATPase YscN and its regulator, YscL. J. Bacteriol. 2006;188:3525–3534. doi: 10.1128/JB.188.10.3525-3534.2006. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Blocker A, Jouihri N, Larquet E, Gounon P, Ebel F, Parsot C, Sansonetti P, Allaoui A. Structure and composition of the Shigella flexneri 'needle complex', a part of its type III secreton. Mol. Microbiol. 2001;39:652–663. doi: 10.1046/j.1365-2958.2001.02200.x. [DOI] [PubMed] [Google Scholar]
- Blocker A, Komoriya K, Aizawa S. Type III secretion systems and bacterial flagella: insights into their function from structural similarities. Proc. Natl. Acad. Sci. USA. 2003;100:3027–3030. doi: 10.1073/pnas.0535335100. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Broms JE, Francis MS, Forsberg A. Diminished LcrV secretion attenuates Yersinia pseudotuberculosis virulence. J. Bacteriol. 2007;189:8417–8429. doi: 10.1128/JB.00936-07. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Broz P, Mueller CA, Muller SA, Phillipsen A, Sorg I, Engel A, Cornelis GR. Function and molecular architecture of the Yersinia injectisome tip complex. Mol. Microbiol. 2007;65:1311–1320. doi: 10.1111/j.1365-2958.2007.05871.x. [DOI] [PubMed] [Google Scholar]
- Brubaker RR. Mutation rate to non-pigmentation in Pasteurella pestis. J. Bacteriol. 1969;98:1404–1406. doi: 10.1128/jb.98.3.1404-1406.1969. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Cheng LW, Anderson DM, Schneewind O. Two independent type III secretion mechanisms for YopE in Yersinia enterocolitica. Mol. Microbiol. 1997;24:757–765. doi: 10.1046/j.1365-2958.1997.3831750.x. [DOI] [PubMed] [Google Scholar]
- Cheng LW, Schneewind O. Yersinia enterocolitica type III secretion: On the role of SycE in targeting YopE into HeLa cells. J. Biol. Chem. 1999;274:22102–22108. doi: 10.1074/jbc.274.31.22102. [DOI] [PubMed] [Google Scholar]
- Cheng LW, Kay O, Schneewind O. Regulated secretion of YopN by the type III machinery of Yersinia enterocolitica. J. Bacteriol. 2001;183:5293–5301. doi: 10.1128/JB.183.18.5293-5301.2001. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Cornelis GR, Colson C. Restriction of DNA in Yersinia enterocolitica detected by the recipient ability for a derepressed R factor from Escherichia coli. J. Gen. Microbiol. 1975;87:285–291. doi: 10.1099/00221287-87-2-285. [DOI] [PubMed] [Google Scholar]
- Cornelis GR, Boland A, Boyd AP, Geuijen C, Iriarte M, Neyt C, Sory M-P, Stainier I. The virulence plasmid of Yersinia an antihost genome. Microbiol. Mol. Biol. Rev. 1998;62:1315–1352. doi: 10.1128/mmbr.62.4.1315-1352.1998. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Cornelis GR. The type III injectisome. Nat. Rev. Microbiol. 2006;4:811–825. doi: 10.1038/nrmicro1526. [DOI] [PubMed] [Google Scholar]
- DeBord K, Lee VT, Schneewind O. On the Role of LcrG and LcrV during the type III targeting of effector Yops by Yersinia enterocolitica. J. Bacteriol. 2001;183:4588–4598. doi: 10.1128/JB.183.15.4588-4598.2001. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Edqvist PJ, Olsson J, Lavander M, Sundberg L, Forsberg A, Wolf-Watz H, Lloyd SA. YscP and YscU regulate substrate specificity of the Yersinia type III secretion system. J. Bacteriol. 2003;185:2259–2266. doi: 10.1128/JB.185.7.2259-2266.2003. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Ferris HU, Furukawa Y, Minamino T, Kroetz MB, Kihara M, Namba K, Macnab RM. FlhB regulates ordered export of flagellar components via autocleavage mechanism. J. Biol. Chem. 2005;280:41236–41242. doi: 10.1074/jbc.M509438200. [DOI] [PubMed] [Google Scholar]
- Ferris HU, Minamino T. Flipping the switch: bringing order to flagellar assembly. Trends Microbiol. 2006;14:519–526. doi: 10.1016/j.tim.2006.10.006. [DOI] [PubMed] [Google Scholar]
- Fraser GM, Hirano T, Ferris HU, Devgan LL, Kihara M, Macnab RM. Substrate specificity of type III flagellar protein export in Salmonella is controlled by subdomain interactions in FlhB. Mol. Microbiol. 2003;48:1043–1057. doi: 10.1046/j.1365-2958.2003.03487.x. [DOI] [PubMed] [Google Scholar]
- Galan JE, Wolf-Watz H. Protein delivery into eukaryotic cells by type III secretion machines. Nature. 2006;444:567–573. doi: 10.1038/nature05272. [DOI] [PubMed] [Google Scholar]
- Hoiczyk E, Blobel G. Polymerization of a single protein of the pathogen Yersinia enterocolitica into needles punctures eukaryotic cells. Proc. Natl. Acad. Sci. USA. 2001;98:4669–4674. doi: 10.1073/pnas.071065798. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Hueck CJ. Type III protein secretion in bacterial pathogens of animals and plants. Microbiol. Mol. Biol. Rev. 1998;62:379–433. doi: 10.1128/mmbr.62.2.379-433.1998. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Hughes KT, Gillen KL, Semon MJ, Karlinsey JE. Sensing structural intermediates in bacterial flagellar assembly by export of a negative regulator. Science. 1993;262:1277–1280. doi: 10.1126/science.8235660. [DOI] [PubMed] [Google Scholar]
- Jackson MW, Plano GV. Interactions between type III secretion apparatus components from Yersinia pestis detected using the yeast two-hybrid system. FEMS Microbiol. Lett. 2000;186:85–90. doi: 10.1111/j.1574-6968.2000.tb09086.x. [DOI] [PubMed] [Google Scholar]
- Journet L, Agrain C, Broz P, Cornelis GR. The needle length of bacterial injectisomes is determined by a molecular ruler. Science. 2003;302:1757–1760. doi: 10.1126/science.1091422. [DOI] [PubMed] [Google Scholar]
- Karlinsey JE, Pease AJ, Winkler ME, Bailey JL, Hughes KT. The flk gene of Salmonella typhimurium couples flagellar P- and L-ring assembly to flagellar morphogenesis. J. Bacteriol. 1997;179:2389–2400. doi: 10.1128/jb.179.7.2389-2400.1997. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Karlinsey JE, Tsui HCT, Winkler ME, Hughes KT. Flk couples flgM translation to flagellar ring assembly in Salmonella typhimurium. J. Bacteriol. 1998;180:5384–5397. doi: 10.1128/jb.180.20.5384-5397.1998. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Karlinsey JE, Lonner J, Brown KL, Hughes KT. Translation/secretion coupling by type III secretion systems. Cell. 2000;102:487–497. doi: 10.1016/s0092-8674(00)00053-2. [DOI] [PubMed] [Google Scholar]
- Kubori T, Matsushima Y, Nakamura D, Uralil J, Lara-Tejero M, Sukhan A, Galan JE, Aizawa S-I. Supermolecular structure of the Salmonella typhimurium type III protein secretion system. Science. 1998;280:602–605. doi: 10.1126/science.280.5363.602. [DOI] [PubMed] [Google Scholar]
- Lavander M, Sundberg L, Edqvist PJ, Lloyd SA, Wolf-Watz H, Forsberg A. Proteolytic cleavage of the FlhB homologue YscU of Yersinia pseudotuberculosis is essential for bacterial survival but not for type III secretion. J. Bacteriol. 2002;184:4500–4509. doi: 10.1128/JB.184.16.4500-4509.2002. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Lee VT, Anderson DM, Schneewind O. Targeting of Yersinia Yop proteins into the cytosol of HeLa cells: one-step translocation of YopE across bacterial and eukaryotic membranes is dependent on SycE chaperone. Mol. Microbiol. 1998;28:593–601. doi: 10.1046/j.1365-2958.1998.00822.x. [DOI] [PubMed] [Google Scholar]
- Lee VT, Schneewind O. Type III machines of pathogenic yersiniae secrete virulence factors into the extracellular milieu. Mol. Microbiol. 1999;31:1619–1629. doi: 10.1046/j.1365-2958.1999.01270.x. [DOI] [PubMed] [Google Scholar]
- Lee VT, Tam C, Schneewind O. LcrV, a substrate for Yersinia enterocolitica type III secretion, is required for toxin targeting into the cytosol of HeLa cells. J. Biol. Chem. 2000;275:36869–36875. doi: 10.1074/jbc.M002467200. [DOI] [PubMed] [Google Scholar]
- Lee VT, Mazmanian SK, Schneewind O. A program of Yersinia enterocolitica type III secretion reactions is triggered by specific host signals. J. Bacteriol. 2001;183:4970–4978. doi: 10.1128/JB.183.17.4970-4978.2001. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Lee VT, Schneewind O. Yop fusions to tightly folded protein domains and their effects on Yersinia enterocolitica type III secretion. J. Bacteriol. 2002;184:3740–3745. doi: 10.1128/JB.184.13.3740-3745.2002. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Macnab RM. Type III flagellar protein export and flagellar assembly. Biochim. Biophys. Acta. 2004;1694:207–217. doi: 10.1016/j.bbamcr.2004.04.005. [DOI] [PubMed] [Google Scholar]
- Marenne MN, Journet L, Mota LJ, Cornelis GR. Genetic analysis of the formation of the Ysc-Yop translocation pore in macrophages by Yersinia enterocolitica : role of LcrV, YscF, and YopN. Microb. Pathog. 2003;35:243–258. doi: 10.1016/s0882-4010(03)00154-2. [DOI] [PubMed] [Google Scholar]
- Marketon MM, DePaolo RW, DeBord KL, Jabri B, Schneewind O. Plague bacteria target immune cells during infection. Science. 2005;309:1739–1741. doi: 10.1126/science.1114580. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Marlovits TC, Kubori T, Sukhan A, Thomas DR, Galan JE, Unger VM. Structural insights into the assembly of the type III secretion needle complex. Science. 2004;306:1040–1042. doi: 10.1126/science.1102610. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Marraffini LA, Schneewind O. Anchor structure of staphylococcal surface proteins. V. Anchor structure of the sortase B substrate IsdC. J. Biol. Chem. 2005;280:16263–16271. doi: 10.1074/jbc.M500071200. [DOI] [PubMed] [Google Scholar]
- McMurry JL, van Arnam JS, Kihara M, Macnab RM. Analysis of the cytoplasmic domains of Salmonella FlhA and interactions with components of the flagellar export machinery. J. Bacteriol. 2004;186:7586–7592. doi: 10.1128/JB.186.22.7586-7592.2004. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Michiels T, Cornelis GR. Secretion of hybrid proteins by the Yersinia Yop export system. J. Bacteriol. 1991;173:1677–1685. doi: 10.1128/jb.173.5.1677-1685.1991. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Michiels T, Vanooteghem J-C, Lambert de Rouvroit C, China B, Gustin A, Boudry P, Cornelis GR. Analysis of virC an operon involved in the secretion of Yop proteins by Yersinia enterocolitica. J. Bacteriol. 1991;173:4994–5009. doi: 10.1128/jb.173.16.4994-5009.1991. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Minamino T, Gonzalez-Pedrajo B, Yamaguchi K, Aizawa SI, Macnab RM. FliK, the protein responsible for flagellar hook length control in Salmonella is exported during hook assembly. Mol. Microbiol. 1999;34:295–304. doi: 10.1046/j.1365-2958.1999.01597.x. [DOI] [PubMed] [Google Scholar]
- Minamino T, Macnab RM. Interactions among components of the Salmonella flagellar export apparatus and its substrates. Mol. Microbiol. 2000a;35:1052–1064. doi: 10.1046/j.1365-2958.2000.01771.x. [DOI] [PubMed] [Google Scholar]
- Minamino T, Macnab RM. Domain structure of Salmonella FlhB, a flagellar export component responsible for substrate specificity switching. J. Bacteriol. 2000b;182:4906–4914. doi: 10.1128/jb.182.17.4906-4914.2000. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Minamino T, Gonzalez-Pedrajo B, Kihara M, Namba K, Macnab RM. The ATPase FliI can interact with the type III flagellar protein export apparatus in the absence of its regulator, FliH. J. Bacteriol. 2003;185:3983–3988. doi: 10.1128/JB.185.13.3983-3988.2003. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Minamino T, Saijo-Hamano Y, Furukawa Y, Gonzalez-Pedrajo B, Macnab RM, Namba K. Domain organization and function od Salmonella FliK, a flagellar hook-length control protein. J. Mol. Biol. 2004;341:491–502. doi: 10.1016/j.jmb.2004.06.012. [DOI] [PubMed] [Google Scholar]
- Minamino T, Ferris HU, Moriya N, Kihara M, Namba K. Two parts of the T3S4 domain of the hook-length control protein FliK are essential for teh substrate specificity switching of teh flagellar type III export apparatus. J. Mol. Biol. 2006;362:1148–1158. doi: 10.1016/j.jmb.2006.08.004. [DOI] [PubMed] [Google Scholar]
- Moriya N, Minamino T, Hughes KT, Macnab RM, Namba K. The type III flagellar export specificity switch is dependent on FliK ruler and a molecular clock. J. Mol. Biol. 2006;359:466–477. doi: 10.1016/j.jmb.2006.03.025. [DOI] [PubMed] [Google Scholar]
- Mueller CA, Broz P, Muller SA, Ringler P, Erne-Brand F, Sorg I, Kuhn M, Engel A, Cornelis GR. The V-antigen of Yersinia forms a distinct structure at the tip of injectisome needles. Science. 2005;310:674–676. doi: 10.1126/science.1118476. [DOI] [PubMed] [Google Scholar]
- Neyt C, Cornelis GR. Insertion of a Yop translocation pore into the macrophage plasma membrane by Yersinia enterocolitica : requirement for translocators YopB and YopD, but not LcrG. Mol. Microbiol. 1999;33:971–981. doi: 10.1046/j.1365-2958.1999.01537.x. [DOI] [PubMed] [Google Scholar]
- Overheim KA, Depaolo RW, Debord KL, Morrin EM, Anderson DM, Green NM, Brubaker RR, Jabri B, Schneewind O. LcrV plague vaccine with altered immunomodulatory properties. Infect. Immun. 2005;73:5152–5159. doi: 10.1128/IAI.73.8.5152-5159.2005. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Payne PL, Straley SC. YscO of Yersinia pestis is a mobile component of the Yop secretion system. J. Bacteriol. 1998;180:3882–3890. doi: 10.1128/jb.180.15.3882-3890.1998. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Payne PL, Straley SC. YscP of Yersinia pestis is a secreted component of the Yop secretion system. J. Bacteriol. 1999;181:2852–2862. doi: 10.1128/jb.181.9.2852-2862.1999. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Roncero C, Darzins A, Casadaban MJ. Pseudomonas aeruginosa transposable bacteriophages D3112 and B3 require pili and surface growth for adsorption. J. Bacteriol. 1990;172:1899–1904. doi: 10.1128/jb.172.4.1899-1904.1990. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Rosqvist R, Forsberg A, Wolf-Watz H. Intracellular targeting of the Yersinia YopE cytotoxin in mammalian cells induces actin microfilament disruption. Infect. Immun. 1991;59:4562–4569. doi: 10.1128/iai.59.12.4562-4569.1991. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Rosqvist R, Magnusson K-E, Wolf-Watz H. Target cell contact triggers expression and polarized transfer of Yersinia YopE cytotoxin into mammalian cells. EMBO J. 1994;13:964–972. doi: 10.1002/j.1460-2075.1994.tb06341.x. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Sarker MR, Neyt C, Stainier I, Cornelis GR. The Yersinia yop virulon: LcrV is required for extrusion of the translocators YopB and YopD. J. Bacteriol. 1998;180:1207–1214. doi: 10.1128/jb.180.5.1207-1214.1998. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Schesser K, Fritzh-Lindsten E, Wolf-Watz H. Delineation and mutational analysis of the Yersinia pseudotuberculosis YopE domains which mediate translocation across bacterial and eukaryotic cellular membranes. J. Bacteriol. 1996;178:7227–7233. doi: 10.1128/jb.178.24.7227-7233.1996. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Sorg I, Wagner S, Amstutz M, Muller SA, Broz P, Lussi Y, Engel A, Cornelis GR. YscU recognizes translocators as export substrates of the Yersinia injectisome. EMBO J. 2007;26:3015–3024. doi: 10.1038/sj.emboj.7601731. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Sorg JA, Miller NC, Marketon MM, Schneewind O. Rejection of impassable substrates by Yersinia type III secretion machines. J. Bacteriol. 2005a;187:7090–7102. doi: 10.1128/JB.187.20.7090-7102.2005. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Sorg JA, Miller NC, Schneewind O. Substrate recognition of type III secretion machines - testing the RNA signal hypothesis. Cell. Microbiol. 2005b;7:1217–1225. doi: 10.1111/j.1462-5822.2005.00563.x. [DOI] [PubMed] [Google Scholar]
- Sorg JA, Blaylock B, Schneewind O. Secretion signal recognition by YscN, the Yersinia type III secretion ATPase. Proc. Nat. Acad. Sci. USA. 2006;103:16490–16495. doi: 10.1073/pnas.0605974103. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Sory M-P, Boland A, Lambermont I, Cornelis GR. Identification of the YopE and YopH domains required for secretion and internalization into the cytosol of macrophages, using the cyaA gene fusion approach. Proc. Natl. Acad. Sci. USA. 1995;92:11998–12002. doi: 10.1073/pnas.92.26.11998. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Takeshita S, Sato M, Toba M, Masahashi W, Hashimoto-Gotoh T. High-copy-number and low-copy-number plasmid vectors for LacZ alpha-complementation and chloramphenicol- or kanamycin-resistance selection. Gene. 1987;61:63–74. doi: 10.1016/0378-1119(87)90365-9. [DOI] [PubMed] [Google Scholar]
- Thomas J, Stafford GP, Hughes C. Docking of cytosolic chaperone-substrate complexes at the membrane ATPase during flagellar type III protein export. Proc. Nat. Acad. Sci. USA. 2004;101:3945–3950. doi: 10.1073/pnas.0307223101. [DOI] [PMC free article] [PubMed] [Google Scholar]
- van Arnam JS, McMurry JL, Kihara M, Macnab RM. Analysis of an engineered Salmonella flagellar fusion protein, FliR-FlhB. J. Bacteriol. 2004;186:2495–2498. doi: 10.1128/JB.186.8.2495-2498.2004. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Williams AW, Yamagushi S, Togashi F, Aizawa SC, Kawagashi I, MacNab RM. Mutations in fliK and flhB affecting flagellar hook and filament assembly in Salmonella typhimurium. J. Bacteriol. 1996;178:2960–2970. doi: 10.1128/jb.178.10.2960-2970.1996. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Woestyn S, Allaoui A, Wattiau P, Cornelis G. YscN, the putative energizer of the Yersinia Yop secretion machinery. J. Bacteriol. 1994;176:1561–1569. doi: 10.1128/jb.176.6.1561-1569.1994. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Woestyn S, Sory M-P, Boland A, Lequenne O, Cornelis GR. The cytosolic SycE and SycH chaperones of Yersinia protect the region of YopE and YopH involved in translocation across eukaryotic cell membranes. Mol. Microbiol. 1996;20:1261–1271. doi: 10.1111/j.1365-2958.1996.tb02645.x. [DOI] [PubMed] [Google Scholar]
- Yother J, Goguen JD. Isolation and characterization of Ca2+-blind mutants of Yersinia pestis. J. Bacteriol. 1985;164:704–711. doi: 10.1128/jb.164.2.704-711.1985. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Zhu K, Gonzalez-Pedrajo B, Macnab RM. Interactions among membrane and soluble components of the flagellar export apparatus of Salmonella. Biochem. J. 2002;41:9516–9524. doi: 10.1021/bi0203280. [DOI] [PubMed] [Google Scholar]
Associated Data
This section collects any data citations, data availability statements, or supplementary materials included in this article.