Abstract
The fruit fly Drosophila melanogaster is a good model to unravel the molecular mechanisms of innate immunity, and has led to some important discoveries on the sensing and signaling of microbial infections. The response of Drosophila to virus infections remains poorly characterized, and appears to involve two facets. On one hand RNA interference (RNAi) involves the recognition and processing of double stranded (ds) RNA into small interfering (si) RNAs by the host ribonuclease Dicer-2 (Dcr-2), whereas on the other hand an inducible response controlled by the evolutionarily conserved JAK-STAT pathway contributes to the antiviral host defense. In order to clarify the contribution of the siRNA and JAK-STAT pathways to the control of viral infections, we have compared the resistance of flies wild-type and mutant for Dcr-2 or the JAK kinase Hopscotch (Hop) to infections by seven RNA or DNA viruses belonging to different families. Our results reveal a unique susceptibility of hop mutant flies to infection by DCV and CrPV, two members of the Dicistroviridae family, which contrasts with the susceptibility of Dcr-2 mutant flies to many viruses, including the DNA virus IIV-6. Genome-wide microarray analysis confirmed that different sets of genes were induced following infection by DCV or by two unrelated RNA viruses, FHV and SINV. Overall, our data reveal that RNAi is an efficient antiviral mechanism, operating against a large range of viruses, including a DNA virus. By contrast, the antiviral contribution of the JAK-STAT pathway appears to be virus-specific.
INTRODUCTION
Viruses represent an important class of pathogens, causing serious concern for human health, as well as important economic losses in crops and animals. Because they replicate inside cells, and rely for the most part on host cell molecular machineries for their replication, viruses pose specific challenges to the immune system. Two major strategies of antiviral resistance have been described. In mammals, viral infection is first detected by pattern recognition receptors (PRRs) of the Toll- and RIG-I–like families that sense the viral nucleic acid and trigger the induction of interferons and other cytokines (1). These factors activate the production of antiviral molecules such as protein kinase R or oligo-2’, 5’- adenylate synthetase that contain the infection, and contribute to the activation of the adaptive immune response (2). In plants, viral nucleic acids are recognized by enzymes of the Dicer (Dcr) family, which produce small interfering (si) RNAs of 21-24 nucleotides (nt). These siRNAs are then loaded onto molecules of the Argonaute (AGO) family, and will guide them towards RNAs with complementary sequences: targeted RNAs are then either sliced by AGO or their translation is inhibited. This RNA interference (RNAi) mechanism provides efficient and sequence specific protection against viral infections (3).
RNAi also plays an important role in the control of viral infections in insects, as shown by the production of virus-derived siRNAs in infected flies, and the increased susceptibility to viral infection of Drosophila mutants for the genes Dcr-2 and AGO2 (3–6). In addition, several reports indicate that an inducible response also contributes to the control of viral infections (7–15). We previously showed that infection with Drosophila C virus (DCV), a member of the Dicistroviridae family, leads to induction of some 130 genes (11). Analysis of the regulation of one of these genes, vir-1, revealed the presence of functionally important binding sites for the transcription factor STAT in its promoter. The induction of vir-1, as well as several other DCV induced genes, was found to be dependent on the gene hopscotch (hop), which encodes the only JAK kinase in Drosophila. Furthermore, hop mutant flies succumb more rapidly than wild-type controls, with a higher viral load, to DCV infection (11). The Toll and IMD pathways, initially characterized for their role in the control of bacterial and fungal infections, were also proposed to play a role in the control of viral infections. Whereas the Toll pathway was associated with resistance to the Drosophila X virus (DXV) (15), the IMD pathway was implicated in the control of Sindbis virus (SINV) (7) and Cricket paralysis virus (CrPV) (9).
Altogether, the data in the present literature point to the involvement of both RNAi and an inducible expression of effector molecules to counter viral infections in insects (5, 16). However, whereas RNAi was shown to contribute to resistance to several RNA viruses (with either single stranded genomes of both polarities, or double stranded genomes), most studies on the inducible response have so far focused on a single virus. As a result, the global significance of the inducible response for the control of viral infections remains poorly understood. In particular, it is unclear at present if the JAK-STAT pathway is involved in a general antiviral response, providing broad antiviral immunity, or if it acts specifically on a critical step in the replication cycle of a specific virus or virus family. In order to address this important question, we have compared the resistance of a mutant for the JAK-STAT pathway to infection by seven RNA or DNA viruses. We find that hop mutant flies are more susceptible than wild-type controls to infections by the Dicistroviridae DCV and CrPV, but exhibit either no or a weak phenotype for other viruses, suggesting that the JAK-STAT pathway dependent inducible response is virus-specific. Genome-wide transcript profiling shows that infection by two other RNA viruses, Flock House Virus (FHV; Nodaviridae) and SINV (Alphaviridae) leads to upregulation of at least 400 genes, which only partially overlap with those induced by DCV. Overall, our data indicate that the siRNA pathway exerts broad antiviral activity and affects both RNA and DNA viruses, with virus-specific inducible responses contributing to the control of viral infections in Drosophila.
MATERIAL AND METHODS
Fly strains culture and infection
Oregon-R (OR) and yw were used as wild-type control flies. hopM38/msv1, Dcr-2L811fsX and Dcr-2R416X mutant flies were previously described (17–19). A genomic rescue of the Dcr 2 gene was established with the Fosmid FlyFos017074 (transgeneome.mpi-cbg.de) inserted at the landing site attP40 (2L), and the transgenic chromosome was recombined with the deficiency Df(2R)BSC45, which uncovers the Dcr-2 locus. For the rescue experiments, Dcr 2 mutants were crossed with the deficiency Df(2R)BSC45 or the Df(2R)BSC45-Dcr-2 rescue line. Flies were fed on standard cornmeal–agar medium at 25°C. All fly lines were tested for Wolbachia infection, and cured whenever necessary. Viral stocks were prepared in 10 mM Tris-HCl, pH 7.5 with the exception of VSV which was used directly from Vero cells culture supernatant (VSV 4×109 PFU/mL; DCV 5×1010 PFU/mL; CrPV 1×109 PFU/mL; FHV 5.5×109 PFU/mL; DXV 4.4×107 PFU/mL, IIV6 4.4×1011 PFU/mL and SINV 5×108 PFU/mL). Infections were performed with 4–6 days old adult flies by intrathoracic injection (Nanoject II apparatus, Drummond Scientific) with viral particles indicated in the figure legends. Injection of the same volume (4.6nL) of 10 mM Tris-HCl, pH 7.5, was used as a control. For bacterial infection, flies were pricked with a thin needle previously dipped in a concentrated overnight culture of Escherichia coli and Micrococcus luteus in LB medium. Infected flies were then incubated at room temperature, or at 29°C in the case of hopM38/msv1 and the corresponding control flies, and monitored daily for survival, or frozen for RNA isolation and virus titration at the indicated time points.
Cell culture and virus titration
Vero R cells were grown in Dulbecco's modified Eagle's medium (DMEM, Invitrogen) supplemented with 10% fetal calf serum (Biowest), Penicillin / Streptomycin (Invitrogen), non essential amino acid mix (Invitrogen), 10 mM pyruvate (Gibco) and 200 mM L-Glutamin (Invitrogen). Kc167 and S2 cells were grown in Schneider’s medium (Biowest) supplemented with 10% fetal calf serum, Glutamax (Invitrogen), Penicillin/Streptomycin (100× mix, 10 mg/mL/ 10000 U, Invitrogen). VSV and SINV were titrated from infected flies by plaque assay on Vero R cells. DCV, CrPV, FHV and IIV6 were titrated on Kc167 (DCV, CrPV and FHV) or S2 (IIV6) cells by the Reed-Muench method to calculate TCID50 and converted to PFU with a conversion factor of 0.7.
RNA analysis
Total RNA from infected flies was isolated using Trizol Reagent RT bromoanisole solution (MRC), according to the manufacturer’s instructions. 1µg total RNA was reverse transcribed using iScript™ cDNA synthesis kit (Biorad). The reverse transcription was run in the T3000 Thermocycler (Biometra) with the following PCR program: Step 1: 65°C for 5 min, step 2: 4°C for 5 min, step 3: 25°C for 10 min, step 4: 42°C for 60 min, step 5: 70°C for 15 min. 100 ng of cDNA was used for quantitative real time PCR, using the iQ™ Custom SYBR Green Supermix Kit (Biorad). The PCR was performed using the CFX384™ Real-Time System (Biorad) with the following program: step 1: 95°C for 3 min, step 2: 95°C for 10 sec, step 3: 55°C for 30 sec, repeated 39 times from step 2. Primers used for qPCR were as follows: RpL32 (forward 5'-GACGCTTCAAGGGACAGTATCTG-3'; reverse 5'-AAACGCGGTTCTGCATGAG-3'), vir-1 (forward 5'-GATCCCAATTTTCCCATCAA-3'; reverse 5'-GATTACAGCTGGGTGCACAA-3'), drosomycin (forward 5'-CGTGAGAACCTTTTCCAATATGATG-3'; reverse 5'-TCCCAGGACCACCAGCAT-3') and diptericin (forward 5'-GCTGCGCAATCGCTTCTACT-3'; reverse 5'-TGGTGGAGTGGGCTTCATG-3'). TotM, upd, upd2 and upd3 expression levels were quantified using the Brilliant II QRT-PCR Core Reagent Kit, 1-step (Stratagene). The reaction took place in a total volume of 20 µL using the Taq man gene expression assay (TotM (Dm02362087 s1), upd (os) (Dm01843792_g1), upd2 (Dm01844134 g1), upd3 (custom designed upd3exon2-ANY), RpL32 (Dm02151827 g1) all from Applied Biosystems). We used the 7500 Fast Real-Time PCR System (Applied Biosystems) with following PCR program: step 1: 45°C for 30 min, step2: 95°C for 10 min, step 3: 95°C for 15 sec, step 4: 60°C for 1 min, repeated 39 times from step 3. In all cases, gene expression was normalized to the ribosomal protein gene RpL32.
For IIV6, the expression of the annotated genes 206R, 224L, 244L and 261R was assessed by strand-specific RT-PCR. We used the Superscript III reverse transcriptase specifically adapted for gene specific priming and followed the manufacturer’s protocol (Invitrogen). Briefly, primer pairs were designed to amplify regions of the IIV6 genome exhibiting high density of small RNA reads or not. 1µg of total RNA extracted from infected S2 cells was reverse transcribed with 2 pmol of either forward (F) or reverse (R) primer and 200 units of SuperScript III reverse transcriptase. The reaction was then incubated for 1 hour at 55°C. 1µl of the resulting cDNA was then used to perform 25 cycles of PCR using Taq DNA polymerase (Invitrogen) and both F and R primers. The primer pairs were as follows: 206R (forward: 5’-AAGGAAAGTGGCGAGTACGA-3’, reverse 5’-AACAAACCCGTTTTCTTCCA -3’); 224L (forward: 5’-CCACCATCACATTGACCTTG-3’, reverse: 5’-ATAAGCGAACCCGAAATCA-3’); 244L (forward: 5’- TGGAAAAGAGTGGTCCCATTT-3’, reverse: 5’-TGTACCTCCCGGAAGATTT-3’); 261R (forward: 5’-CAGCCCCATCCGAATTACTA-3’, reverse: 5’-CTGCAACTGCAGAAATTTGA-3’). The PCR bands were sequenced to verify their viral origin.
Statistical analysis
An unpaired two-tailed Student's t-test was used for statistical analysis of data with GraphPad Prism (GraphPad Software). p values lower than 0.05 were considered statistically significant. Survival curves were plotted and analyzed by log-rank analysis (Kaplan-Meier method) using GraphPad Prism (GraphPad Software).
DNA microarray analysis
For each sample, Tris injected, DCV-infected (11), FHV and SINV-infected, three biologically independent samples comprising 45 male Oregon-R flies were used. RNA extraction, biotinylation and hybridization to Affymetrix Drosophila GeneChip microarrays (Affymetrix) were done as described (20). The Affymetrix Microarray Suite 5.0 (Affymetrix) or Excel (Microsoft) with a combination of built-in functions and custom formulae were used for data analysis. Raw data were sorted with the 'absent-marginal-present flags' generated by the Microarray Suite functions. Although an 'absent flag' might indicate that there was no mRNA of particular type present in a sample, 'marginal flags' and 'absent flags' may indicate problems with the hybridization; therefore, only data points marked as 'present' at least in one replicate were retained. The remaining data mass for each microarray was then normalized to itself, making the median of all the measurements one. A gene was considered as induced if at least present in one replicate, with a virus/Tris ratio higher then 2 for at least one of the time point. Classification of gene functions was analyzed by David Bioinformatics Resources 6.7 (21). The data set for FHV and SINV was submitted to the GEO database (Gene Expression Omnibus: http://www.ncbi.nlm.nih.gov/geo/), with the accession number GSE31542.
Assembly, sequencing and analysis of small RNA libraries
The small RNA library of S2 cells and whole flies were constructed as described (22) and sequenced by the Illumina 2G Analyzer. Reads were then aligned to a reference consisting of the IIV-6 genome from NCBI (accession code NC_003038) using the Bowtie program with standard parameters in genome assembly. Reads aligning to the IIV6 genome with a maximum of one mismatch were retained and analyzed using In-house Perl scripts and Excel. Sequences were submitted to the National Center for Biotechnology Information Small Read Archive (http://www.ncbi.nlm.nih.gov/Traces/sra/sra.cgi?) under the accession GSE41007.
RESULTS
RNAi provides broad antiviral protection in Drosophila
Several independent studies, including our own, have established that RNAi, and more precisely the siRNA pathway, serves as an efficient host defense against RNA viruses. These include viruses with single stranded genome of both (+) and (−) polarity and dsRNA viruses (23–30), and we confirmed that flies mutant for Dcr-2 die more rapidly than wild-type controls when they were infected with DCV, CrPV, FHV, SINV, Vesicular Stomatitis Virus (VSV; Rhabdoviridae) and DXV (Birnaviridae) (data not shown). Next, we addressed the question whether the siRNA pathway also participated in the control of a DNA virus infection, and infected wild-type and RNAi mutant flies with invertebrate iridescent virus 6 (IIV6; Iridoviridae). Infection of Dcr-2 mutant flies led to a more rapid and intense appearance of blue color, which is characteristic of the accumulation of iridescent viral particles, than in wild-type controls (Fig. 1A). Dcr 2−/− flies were significantly more susceptible to IIV6 infection than the corresponding wild-type (Fig. 1B). A fraction of Dcr-2−/− flies injected with buffer also died in the course of the experiment, confirming the increased sensitivity to stress associated with mutations of the siRNA pathway (31). The decreased survival time correlated with a 20-fold increased viral load in Dcr-2 mutant flies at 10 days post-infection (dpi) (Fig. 1C). Similar results were obtained when a different null allele of Dcr-2 was used, and the IIV6 susceptibility phenotype was rescued by a wild-type genomic Dcr-2 transgene (Fig. 1D). r2d2−/− and AGO2−/− null mutant flies also exhibited increased sensitivity to IIV6 (Fig. 1E). AGO2−/− flies contained more viral DNA than wild-type controls, confirming that this gene participates to the control of the infection (Fig. 1F).
FIGURE 1. Dcr-2 is involved in host-defense against the DNA virus IIV6.
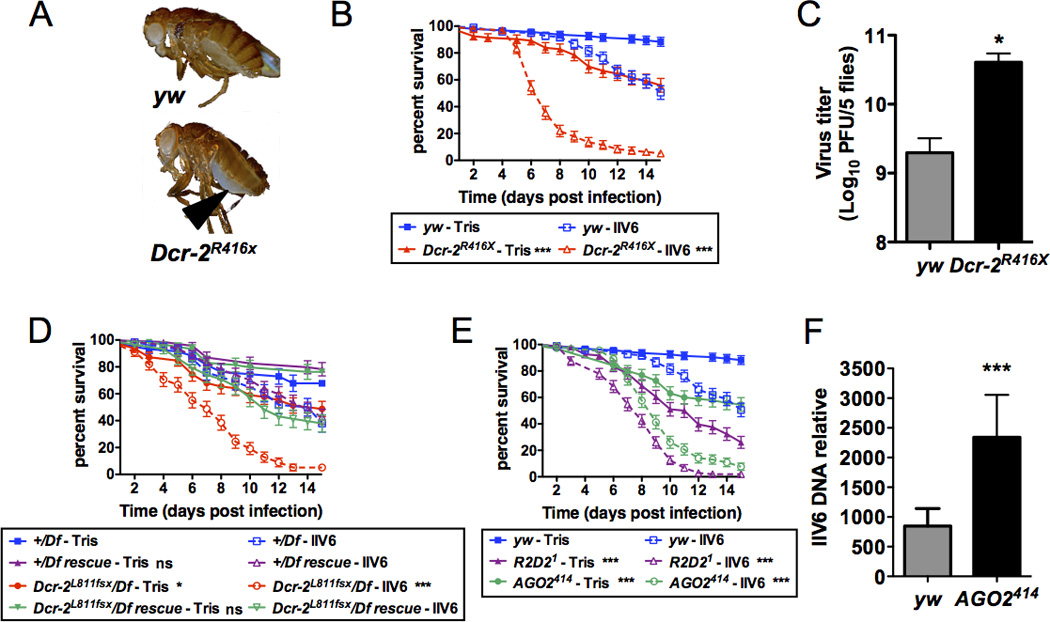
(A) Upon injection of IIV6 (5000PFU) in wild-type (yw) and Dcr-2R416X mutant flies, typical blue paracrystalline structures appeared earlier in the abdomen (arrow head) of the mutant flies. Representative individuals 10 dpi are shown. (B) Groups of 20 wild-type (yw) or Dcr 2R416X mutant flies were injected with IIV6 or Tris and survival was monitored daily. The difference between the wild-type and Dcr-2 mutant flies is statistically significant. (C) Viral titer in groups of 5 wild-type (yw) or Dcr-2R416X mutant flies was monitored 10 dpi. (D) Rescue of the hemizygous Dcr-2L811fsx for the IIV6 susceptibility phenotype by a transposon expressing a wild-type Dcr-2 transgene. Dcr-2L811fsx hemizygous flies (Dcr-2L811fsx/Df) are significantly more susceptible than Dcr-2L811fsx hemizygous flies complemented by a wild-type Dcr-2 transgene (Dcr-2L811fsx/Df rescue). Df is Df(2R)BSC45, a deficiency that fully uncovers the Dcr-2 locus. All control and genomic rescued flies are in CantonS background. (E) Survival rate of wild-type (yw), R2D21 and AGO2414 mutant flies upon IIV6 or Tris injection. (F) IIV6 DNA load was determined by qPCR in 4 groups of 6 flies of the indicated genotype at 10 dpi. For all panels, the data represent the mean and standard deviation of at least three independent experiments and the difference between controls and mutant flies is statistically significant (* p<0.05, ** p<0.01, *** p<0.001). All experiments are performed at 22°C (A, C, F) or 25°C (B, D and E).
We next sequenced small RNA libraries prepared from IIV6-infected S2 cells or adult flies. We observed several hundreds of thousand reads matching the IIV6 genome in both infected S2 cells and wild-type flies, but not in control non-infected S2 cells (Suppl. Table 1). The large majority of these reads had a size of 21 nt, which is characteristic for processing by Dcr 2. This peak was absent from the library prepared from infected Dcr-2−/− mutant flies (Fig. 2A). These data indicate that Dcr-2 generates 21nt IIV6-derived siRNAs in infected flies, and raise the question of the nature of the substrate used by Dcr-2 in the context of this infection. As previously reported for RNA viruses, the number of reads matching each strand of the viral genome was very similar (Suppl. Table 1). However, unlike RNA viruses, the virus-derived siRNAs were not uniformly distributed along the viral genome. Rather, several hotspots were observed, revealing that specific regions of the viral genome generate the siRNAs (Fig. 2B,C). These peaks do not correlate with the intensity of transcription of the viral genome, and some highly transcribed regions are located in areas not generating significant levels of siRNAs (32). The strong symmetry of the peaks observed in S2 cells and wild-type flies suggest that these regions are transcribed on both strands, and generate dsRNA. Indeed, we could detect bidirectional transcription in the areas of the viral genome covered by the peaks (Fig.2D). By contrast, transcription of only one strand of the DNA genome was detected for the locus 261R, which is located in a region that does not produce significant amount of siRNAs. Overall, these results indicate that the siRNA pathway in Drosophila can also protect against a DNA virus infection.
FIGURE 2. Virus-derived siRNAs in S2 cells and Drosophila adult flies infected by the DNA virus IIV6.
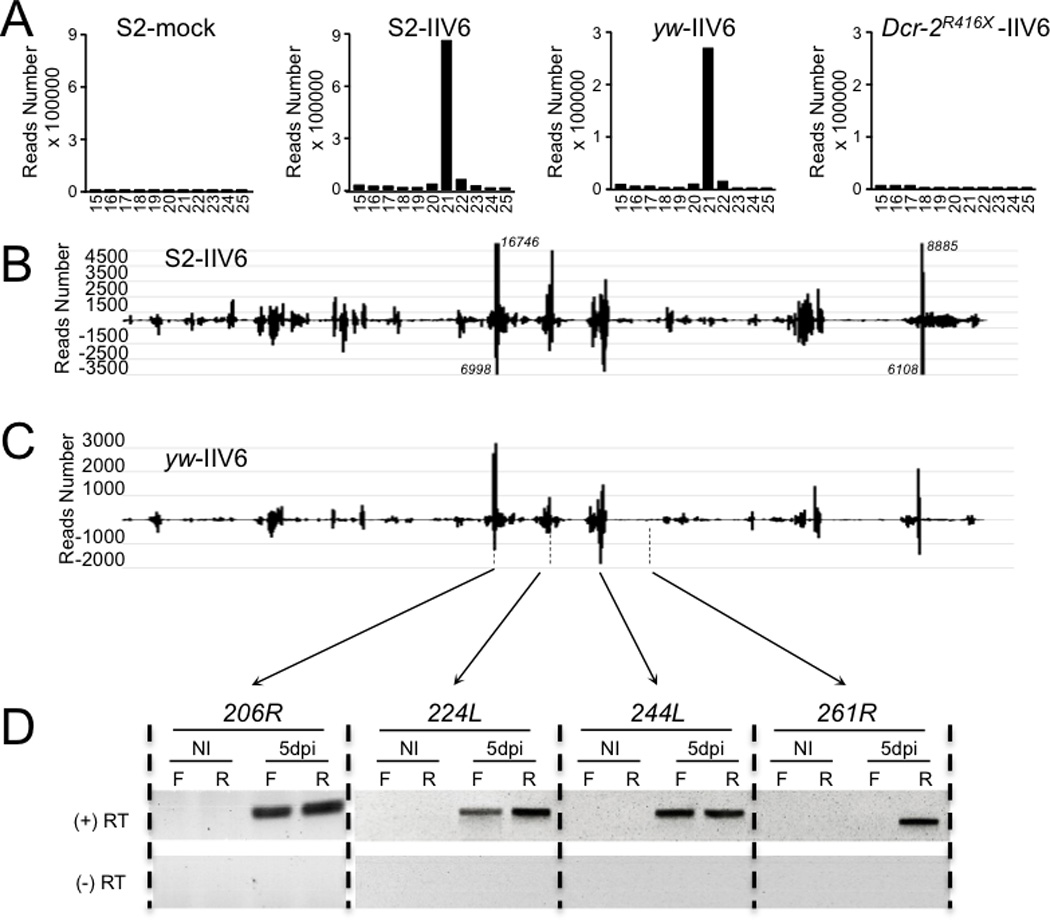
RNA was extracted 5 days post-infection (dpi) from S2 cells infected by IIV6 (MOI 0.01) and adult wild-type (yw) or mutant (Dcr-2R416X) flies injected with IIV6 (5000 PFU/fly). (A) Size distribution of the small RNAs matching the viral genome in S2 cells and adult flies of the indicated genotype. (B, C) Distribution of the 21nt siRNAs from the S2 cells (B) and yw adult flies (C) libraries along the IIV6 genome. Each IIV6-derived small RNA (vsiRNA) is represented by the position of its first nucleotide. The vsiRNAs matching the upper and lower strand of the DNA genome are respectively shown above (positive reads number) and below (negative reads number) the horizontal axis, which represents the genomic coordinates. In panel B, the number of reads for 4 peaks going off-scale is indicated next to them in italics. (D) Strand-specific RT-PCR with primers corresponding to the annotated viral genes 206R, 224L, 244L and 261R. The experiment was performed in the presence (+) or absence (−) of reverse transcriptase (RT). NI: non infected; F and R: forward and reverse strand primer used for reverse transcription.
The JAK Kinase Hopscotch does not confer broad antiviral immunity
To test the contribution of the JAK-STAT pathway in antiviral immunity in Drosophila, we injected loss-of-function mutants of the JAK kinase Hopscotch (hopM38/msv1) with different ssRNA, dsRNA and DNA viruses. As previously described, hopM38/msv1 mutant flies die more rapidly than wild-type controls following DCV infection, and contain about 10-fold more virus (Fig. 3A). By contrast, we did not observe significant differences in survival between wild-type and hopM38/msv1 mutant flies upon infection with the Alphavirus SINV (Fig. 4A), and the viral titers 2 dpi were not significantly different in wild-type and hopM38/msv1 mutant flies (data not shown), indicating that the JAK-STAT pathway does not contribute to the resistance to this virus. hopM38/msv1 mutant flies also resisted as well as wild-type flies to infections by the Rhabdovirus VSV and by the Nodavirus FHV (Fig. 4B, C). A slight reduction in the survival was observed in the case of the dsRNA virus DXV (Birnaviridae) and the DNA virus IIV6 (Fig. 4D, E). However, the difference between wild-type and hopM38/msv1 mutant flies was only statistically significant in the case of DXV infection. Furthermore, we did not observe statistically significant differences in the DXV and IIV6 viral titers in wild-type and hopM38/msv1 mutant flies in the format of our assays (data not shown).
FIGURE 3. The JAK kinase Hopscotch is involved in host-defense against DCV and CrPV.
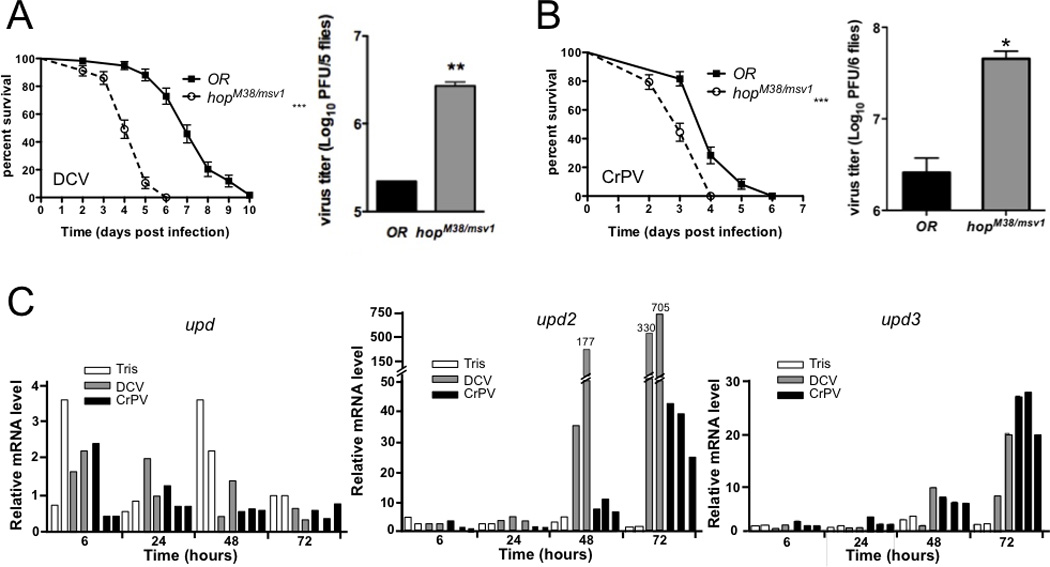
(A, B) Groups of 20 wild-type (OR) or hopscotch (hopM38/msv1) mutant flies were injected with DCV (500 PFU) (A) or CrPV (5 PFU) (B) and survival was monitored daily. The experiment was repeated three times and the data represent the mean and standard deviation. The difference between wild-type and hop mutant flies is statistically significant (*** p<0.001). In the right panel, viral titer was determined in groups of 5 flies 2 dpi for DCV (A) and 1dpi for CrPV (B). The data represent the mean and standard deviation of three independent experiments and the difference between wild-type and hop mutant flies is statistically significant (* p<0.05, ** p<0.01). (C) DCV and CrPV infection trigger induction of the genes upd2 and upd3, which encode cytokines activating the JAK/STAT pathway. Flies were infected with DCV or CrPV and expression of upd, upd2 and -3 was monitored in groups of 10 flies at the indicated time points by Taqman Q-PCR. The results of at least two independent experiments are shown.
FIGURE 4. Susceptibility of fly mutants for the JAK kinase Hopscotch to infection by SINV, VSV, FHV, DXV and IIV6.

Groups of 20 wild-type (OR) or hop mutant flies were injected with SINV (A), VSV (B), FHV (C), DXV (D) or IIV6 (E), and survival was monitored. For VSV and SINV the Tris buffer control injection is also shown, since hop mutant flies showed decreased survival at 29°C after day 16 upon both buffer and virus injection. Kaplan-Meier analysis of the results of at least two independent experiments reveal a statistically significant difference in survival between wild-type and hop mutant flies only in the case of DXV (* p<0.05). ns: not significant.
Overall, our data indicate that the JAK-STAT pathway is critical for the host defense against DCV, but plays a minor role for DXV and IIV6, and is essentially dispensable in the case of FHV, SINV and VSV. We therefore tested CrPV, another member of the Dicistroviridae family known to infect Drosophila. We observed a decrease in survival and a significant increase in viral titers compared to wild-type flies in CrPV infected hopM38/msv1 mutant flies (Fig. 3B). In conclusion, our data indicate that the JAK-STAT pathway in Drosophila confers protection against some viruses, in particular the Dicistroviridae, but does not provide broad antiviral immunity.
Inducible gene expression in FHV and SINV infected flies
The above results raised the question whether an inducible response contributes to the host-defense against viruses other than DCV and CrPV. We therefore conducted a genome-wide microarray analysis using Affymetrix DNA microarrays to monitor gene expression in flies infected by FHV (2 and 3 dpi) or SINV (4 and 8 dpi), and compared the data to those obtained for DCV infection (1 and 2 dpi). The time points were chosen for this analysis to take into account the different kinetics of replication and colonization of Drosophila by the different viruses (11, 24). For each virus, we observed a large overlap between the genes induced at the first and second time points. We then pursued our analysis focusing on the genes induced either at the first or the second time point. The microarray data revealed that 487 and 201 genes were induced or upregulated by a factor of at least 2 upon infection by FHV and SINV, respectively. When analyzed with the same criteria, 166 genes were induced by DCV (Fig. 5A and Suppl. Table 2).
FIGURE 5. Microarray analysis of gene induction following infection by DCV, FHV or SINV.
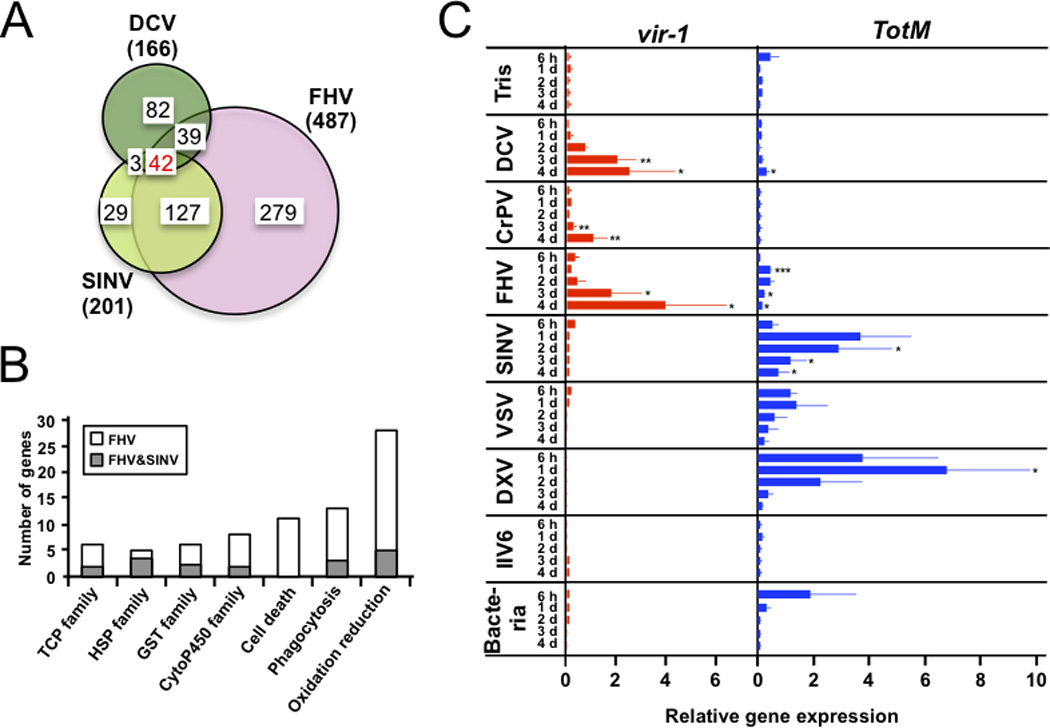
(A) Venn diagram showing the number of upregulated genes (by a factor of at least 2) following infection by the three viruses. The total number of genes regulated by each virus is indicated in parenthesis. (B) FHV and SINV induce members of the same gene families, but FHV triggers a stronger response. The number of genes belonging to seven gene ontology (GO) functional categories induced by both FHV and SINV or by FHV only are shown. (C) Expression of vir-1 and TotM by Q-PCR normalized for the expression of the housekeeping gene RpL32. Groups of 10 wild-type (OR) flies were injected with Tris buffer or the viruses DCV, CrPV, FHV, SINV, VSV, DXV, IIV6 or pricked with a needle dipped in a concentrated pellet of the Gram-positive bacteria M. luteus and the Gram-negative bacteria E. coli. RNA was extracted at 6h, 1d, 2d, 3d and 4d after challenge. The data represent the mean and the standard errors of at least two independent experiments. p-values were calculated for each time point individually versus the Tris injected control. *p<0.05, **p<0.01, ***p<0.001.
The data of this transcriptomic analysis call for two comments. First, we note that 42 genes were induced by all three viruses (Fig. 5A). We compared this set of genes with microarray studies performed on flies infected by fungi and bacteria (both extra- and intracellular) to identify a potential signature specific for viral infections (Suppl. Table 3). We observed that a number of genes, such as Frost, are upregulated similarly by all types of infections, suggesting that they are induced by the stress of the infection, rather than by recognition of specific characteristics of the infecting microorganism. Interestingly, other genes such as Vago, Obp99b, Mal-B1, Nmda1, CG8147, CG1572, l(2)gd1, CG14906, CG10911 and Tsp42EI appear to be only induced in response to viral infections, and may represent the core of an inducible antiviral gene expression program. The case of Obp99b is particularly striking, as this gene is strongly upregulated by the three viruses, but inhibited following the other types of infection. Clearly, the regulation and function of this molecule deserves further investigation. The genes CG4680, Eip75B, Sp7, CG10916, are induced both by the viruses and the intracellular bacterium Listeria (33), suggesting that they may participate in the defense against intracellular intruders (Suppl. Table 3).
A second comment is that the majority of upregulated genes are only induced by one or two of the viruses, revealing virus-specific responses. Interestingly, 84% of the genes upregulated by SINV are also induced by FHV, pointing to a strong similarity between the responses to the two viruses. FHV induced a higher number of genes than SINV, and only 34% of the genes induced by FHV are also induced by SINV (Fig. 5A). Interestingly though, many of the genes induced solely by FHV but not by SINV are members of the same gene families as the genes co-induced by both FHV and SINV. This peculiarity underlines the basic similarities between the transcriptional response to the two viruses. In addition, several genes associated with cell death are induced by FHV, but not SINV, which may reflect the higher virulence of FHV (Fig. 5B; Suppl. Tables 2 and 3). Only 22% and 16% of the genes induced by SINV and FHV, respectively, are also induced by DCV, indicating that DCV on one hand, and FHV and SINV on the other hand, trigger different inducible responses (Fig. 5A). We did not detect in our microarrays expression of the genes encoding the unpaired (Upd) cytokines, which activate the JAK-STAT pathway in Drosophila. However, QRT-PCR analysis revealed that upd2 and upd3, but not upd, are induced or upregulated following DCV and CrPV infection (Fig. 3C).
Virus-specific pattern of gene induction
To further characterize the transcriptional response triggered by different viruses, wild-type flies were injected with DCV, CrPV, FHV, SINV, VSV, DXV and IIV6 and gene induction was measured at 6 hours post-infection and 1, 2, 3 and 4 dpi. Gene expression was monitored by quantitative RT-PCR, which provides a more accurate quantification of gene expression than hybridization to short oligonucleotide probes on microarrays (34). We monitored expression of the DCV-induced gene vir-1 (11) and of Turandot M (TotM), which according to the microarrays is induced by FHV and SINV infection. We confirmed the induction of vir-1 by DCV and FHV (11) and detected a milder but significant induction of this gene by CrPV infection. By contrast, no induction of vir-1 by SINV, VSV, DXV and IIV6 was observed (Fig. 5C). For TotM, we confirmed the induction by FHV at different time points. Additionally we observed that TotM expression was significantly induced by DCV at late time points of infection (4 dpi). We note that induction of TotM by SINV, VSV and DXV was 10 –20 times stronger than the induction by FHV (Fig. 5C). The DNA virus IIV6 did not induce TotM at any measured time point. Interestingly we observed different profiles for vir-1 and TotM induction after viral challenge. Overall, the viruses that kill wild-type flies rapidly (within 10 days) such as DCV, CrPV and FHV were potent inducers of vir-1 whereas less pathogenic viruses such as SINV, VSV and DXV did not induce vir-1. The opposite trend was observed for TotM, which was most potently induced by SINV, VSV and DXV. The different pattern of induction of vir-1 and TotM suggests that the two genes may be regulated differently, even though both were previously shown to be regulated by the JAK-STAT pathway (11, 17). Indeed, the MAP3K MEKK1 and the Imd pathway are also known to contribute to the induction of TotM induction in some contexts (17, 35).
Some antimicrobial peptide genes were also upregulated according to the microarrays, suggesting an overlap between antiviral immunity and the antibacterial/antifungal defenses. We observed an enrichment for genes regulated by the Toll pathway (e.g. the cytokine Spaetzle (Spz) and the antifungal peptides Drosomycine (Drs) and Metchnikowine (Mtk)) in the DCV-specific set of genes (Suppl. Table 2). We also noted an enrichment of Imd pathway regulated genes, such as the antibacterial peptides Attacin-A and -C, Diptericin-B and the transcription factor Relish, in the genes upregulated by both DCV and FHV. However, when expression of diptericin and drosomycin, two markers of activation of the Imd and Toll pathways, respectively, was monitored by QRT-PCR, none of the viruses triggered an induction comparable to that of bacterial and fungal infections, although the wounding associated with the injection procedure clearly led to some expression of the genes (Suppl. Fig. 1).
DISCUSSION
We have investigated here the involvement of RNA interference and the evolutionarily conserved JAK-STAT signaling pathway in the resistance to a panel of seven viruses representing several important families, including the arboviruses SINV and VSV. Our data provide a contrasting picture, with on one hand a broad antiviral immunity based on RNA interference contributing to the defense against both RNA and DNA viruses, and on the other hand a virus-specific transcriptional response involving the JAK-STAT pathway but playing a critical role only in the case of Dicistroviridae infection.
RNA interference protects against a DNA virus infection
The present study extends work from several groups, including our own, showing that flies mutant for the siRNA pathway are more sensitive than wild-types to a large panel of RNA viruses, and shows that Dcr-2 is also required for the control of the DNA virus IIV-6. We note however that the increase of viral titer in siRNA pathway mutant flies is not as strong as in the case of some RNA viruses (e.g. VSV, (25)). This could reflect either the fact that only a portion of the viral genome is targeted by siRNAs, or the expression of a viral suppressor of RNAi by IIV6. Indeed, this virus encodes an RNaseIII enzyme, which could cleave siRNA duplexes, as previously reported in plants infected by the Sweet Potato Chlorotic Stunt Virus (36). The involvement of Dicer-mediated immune responses against DNA virus infections was previously reported in plants, where secondary structures in the transcribed viral RNAs, or dsRNAs formed from overlapping bidirectional transcripts, can be processed into siRNAs (37, 38). Production of dsRNA from DNA viruses also occurs in animal cells, as demonstrated by the critical role played by the dsRNA receptor TLR3 in the sensing of herpesvirus infection in mammals (39, 40). Our data are consistent with a model where dsRNA generated from convergent transcription of the IIV6 genome is processed by Dcr-2 and triggers RNAi. Thus, we conclude that RNAi provides an efficient and highly specific RNA-based defense against many types of viruses in Drosophila and probably other insects. This parallels the situation described in plants. The vertebrates, which largely rely on the induction of interferons to counter viral infections, appear to be the exception among multicellular organisms (1). Interestingly however, the DExD/H box helicase domains found in Dicer enzymes and RIG-I like receptors, which sense the presence of viral RNAs in cells infected by RNA and DNA viruses, are phylogenetically related (10). This suggests that an essential domain of a core molecule from the ancestral antiviral response, RNA silencing, was at some point recruited to sense viral RNAs in vertebrates and to subsequently activate a signaling pathway leading to production of interferons.
Virus-specific induced gene expression in Drosophila
Microarrays are powerful tools to monitor the global transcriptome of infected cells and compare the response to different infections. Despite its limitations for accurate measurements of the magnitude of expression changes, this technology provides useful information on changes in gene expression (34). Here, using whole genome Affymetrix microarrays to analyze the transcriptome of flies infected by DCV, FHV or SINV, we report the existence of virus-specific responses to infection. These results are in keeping with a previous study pointing to autophagy as an antiviral defense mechanism against VSV, but not DCV, infection (14). The three viruses we used belong to different families, and present different characteristics that make them valuable for the present study. For example, (i) DCV and FHV replicate rapidly and kill Drosophila upon injection, whereas SINV does not at the dose used (11, 24); (ii) DCV is a natural pathogen of Drosophila, whereas FHV and SINV have not been found in wild Drosophila populations (41); (iii) FHV and DCV possess respectively a strong and moderate viral suppressor of RNAi, whereas SINV presumably does not (28, 42, 43). The three viruses also have different tissue tropism, and may be associated with tissue specific modifications in the physiology of the infected host. For example, FHV was recently shown to be a cardiotropic virus, affected by potassium channels regulating heart function (44), whereas DCV infection causes intestinal obstruction (S.Chtarbanova and J.-L.I., in preparation).
The comparison of the transcriptomes of the flies infected by the three viruses revealed more similarities between FHV and SINV than between each of these and DCV. This may reflect the co-evolution of DCV with its host, and the fact that this virus may have learned to ward off the antiviral arsenal of its host. Indeed, DCV induces fewer genes than FHV even though the two viruses replicate with similar kinetics and lead to the rapid death of the flies. The genes induced by FHV and SINV encode chaperonins (Tcp or Hsp), glutathione transferases, cytochrome P450s, stress markers (Tot family), thioester containing proteins (TEPs) and cytoskeletal regulators, suggesting an involvement of oxidative stress and phagocytosis in the response to these viruses. The two viruses also upregulate the gene egghead (egh), which encodes a molecule involved in the uptake of dsRNA and antiviral immunity (27). In spite of the large overlap between the genes upregulated by FHV and SINV, the former induces a more intense transcriptional response than the latter. This may reflect the more aggressive replication of FHV in Drosophila. Indeed, the genes specifically induced by FHV include additional members of the families mentioned above (Hsp, Tcp, Gst, cytP450, TEPs), supporting the notion of a more intense response, but also genes associated with cell death. FHV also upregulates several molecules previously connected to innate immunity in Drosophila, such as Hel89B (45), POSH (46), MEKK1 (35), or molecules which may down-modulate the strong response to virus infection (e.g. the genes CG9311 and Pez, encoding tyrosine phosphatases). Finally, we note that FHV induced 8 genes encoding factors with RNA binding domains, including 4 DExD/H box helicases, which may participate in the sensing and neutralization of viral nucleic acids. This specificity may reflect a response of the host to counter the effect of the strong suppressor of RNAi B2, a dsRNA binding protein (47).
An intriguing aspect of the transcriptome of virus-infected flies is the upregulation of genes regulated by the Toll and IMD pathways. We observed an enrichment of Toll pathway target genes induced in flies infected by DCV, but not FHV or SINV, suggesting that DCV infection triggers this pathway. Among the genes induced by DCV, but not by the two other viruses, we also note the presence of Ect4, which encodes a TIR domain cytoplasmic molecule. The mammalian orthologue of this gene, SARM, was proposed to participate as a negative regulator of Toll-like receptor signalling in some antiviral defenses (48). Two other genes regulated by DCV and possibly establishing a connection between RNA silencing and the inducible response are worth mentioning: headcase was identified in a screen as a regulator of the siRNA pathway (49), while CG9925 encodes a protein with a Tudor domain, a characteristic of several components of the piRNA pathway (50).
Unlike the Toll-regulated genes, several genes regulated by IMD were induced in flies infected by DCV or FHV, although not by SINV. The Toll and IMD pathways play a well-characterized role in the regulation of bacterial and fungal infections, through the regulation of genes encoding antimicrobial peptides. These genes are also upregulated by viral infection, although not significantly compared to buffer injection. This low level of induction most likely explains our inability to detect antimicrobial peptides in the hemolymph of DCV infected flies (51). Although not formally establishing that the Toll and IMD pathways participate to the antiviral response, these results certainly do not rule out such a role (7, 9, 15). Alternatively, the induction of the antimicrobial genes may involve the transcription factor FOXO, a known regulator of stress resistance, and occur independently of the Toll and IMD pathways (52). Whatever the mechanism of induction, the biological significance of this weak induction of molecules normally active in the micromolar range is unclear. One possibility is that the Drosophila antimicrobial peptides carry additional functions, which do not require high-level expression. For example, some mammalian β-defensins play a dual role in innate immunity and, in addition to their antibacterial properties, interact with chemokine receptors with affinities in the nanomolar range, thus mediating chemoattraction of phagocytic cells (53).
Dicistroviridae-specific contribution of the JAK-STAT pathway to antiviral immunity
An unexpected finding reported here is that hopscotch mutant flies have a clear phenotype for DCV and CrPV but not for the other viruses tested. This indicates that the JAK-STAT pathway, in addition to RNAi, participates in the host-defense against members of the Dicistroviridae family. DCV infection leads to induction of the genes encoding the cytokines Upd2 and Upd3, which may subsequently activate the JAK-STAT pathway in non-infected cells, triggering an antiviral program of gene expression. Altogether, our results highlight that the contribution of the inducible response to the control of DCV is similar to that of RNAi, as flies mutant for either RNAi or the inducible JAK-STAT pathway succumb to infection two to three days before the controls, with an approximately 10-fold increase in the viral titer.
Interestingly, even though hop mutant flies appear to be specifically sensitive to Dicistroviridae, other viruses activate the JAK-STAT pathway. Indeed, we observed a slight increase in the lethality of hop mutant flies after infection with DXV and IIV6. In Aedes mosquitoes, the JAK/STAT pathway was also shown to activate a defense against Dengue, a member of the Flaviviridae family (54). We also note that the JAK-STAT pathway regulated gene vir-1 (11) is induced by DCV and CrPV, but also FHV, even though hop mutant flies resist to FHV infection much like wild-type flies. One hypothesis to explain this apparent paradox is that some genes may be induced in a JAK-STAT independent manner in the context of viral infections. For example, the gene TotM, which is induced by several viruses to which hop mutant flies resist normally, can be induced by the MEKK1 pathway, in addition to the JAK-STAT pathway (35). Indeed, we observed that TotM remains fully induced by FHV and SINV in hop mutant flies (C.D. and J.-L.I., unpublished observations). However, this hypothesis cannot account for the induction of vir-1 by FHV, because it is strongly reduced in hop mutant flies (C.D. and J.-L.I., unpublished observations). This suggests that some aspects of the JAK-STAT induced response may be redundant with other defenses for FHV, but not for DCV. The fact that FHV triggers a stronger transcriptional response than DCV (Fig. 5) is consistent with this hypothesis.
A key question pertains to the nature of the receptor detecting Dicistroviridae infection and triggering the JAK-STAT-dependent inducible response. Our data point to the induction of a specific subset of genes, including the JAK-STAT regulated gene vir-1 (11), by fast killing viruses such as DCV and CrPV, but also FHV, which replicate rapidly to high titers upon injection in the flies. Of note, vir-1 induction is not affected in flies expressing the dsRNA binding protein B2, or in Dcr-2 mutant flies, indicating that this gene is not induced following sensing of dsRNA (10). This suggests that sensing tissue damage and/or cell death could contribute to this inducible response, a hypothesis corroborated by the association of the JAK-STAT pathway with the cellular response to a variety of stresses (17, 55–57).
In conclusion, our data confirm that, beyond RNAi, an inducible response contributes to the control of some viral infections in Drosophila. However, this response is complex and great care should be exerted before generalizing the results obtained with one single virus species. This unexpected complexity probably reflects the intricate association of viruses with their host cells in different tissues, their different strategies of replication or protein expression, or their acquisition of suppressors of host-defense.
Supplementary Material
ACKNOWLEDGEMENTS
We thank Estelle Santiago and Miriam Yamba for excellent technical assistance; Phil Irving for help with the microarray experiments; Anette Schneeman (TSRI, La Jolla USA) for providing DXV and CrPV and Trevor Williams (Veracruz, Mexico) for providing IIV6; Stéphanie Blandin and Dominique Ferrandon for critical reading of the manuscript and helpful suggestions. The microarray analysis and the deep sequencing was performed at the Plateforme Biopuces et Séquençage, Institut de Génétique et de Biologie Moléculaire et Cellulaire (IGBMC).
Footnotes
This work was supported by the US National Institute of Health (PO1 AI070167), the Agence Nationale de la Recherche (ANR-09-MIEN-006-01), the Balzan Foundation (to J.A.H.), the European Research Council (ERC Starting Grant ncRNAVIR 260767, to S.P.), the Investissement d’Avenir program Laboratoire d’Excellence (NetRNA ANR-10-LABX-36) and the Centre National de la Recherche Scientifique.
REFERENCES
- 1.Beutler B, Eidenschenk C, Crozat K, Imler JL, Takeuchi O, Hoffmann JA, Akira S. Genetic analysis of resistance to viral infection. Nat Rev Immunol. 2007;7:753–766. doi: 10.1038/nri2174. [DOI] [PubMed] [Google Scholar]
- 2.Kasturi SP, Skountzou I, Albrecht RA, Koutsonanos D, Hua T, Nakaya HI, Ravindran R, Stewart S, Alam M, Kwissa M, Villinger F, Murthy N, Steel J, Jacob J, Hogan RJ, Garcia-Sastre A, Compans R, Pulendran B. Programming the magnitude and persistence of antibody responses with innate immunity. Nature. 2011;470:543–547. doi: 10.1038/nature09737. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 3.Ding SW. RNA-based antiviral immunity. Nat Rev Immunol. 2010;10:632–644. doi: 10.1038/nri2824. [DOI] [PubMed] [Google Scholar]
- 4.Kemp C, Imler JL. Antiviral immunity in drosophila. Curr Opin Immunol. 2009;21:3–9. doi: 10.1016/j.coi.2009.01.007. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 5.Sabin LR, Hanna SL, Cherry S. Innate antiviral immunity in Drosophila. Curr Opin Immunol. 2010;22:4–9. doi: 10.1016/j.coi.2010.01.007. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 6.van Rij RP, Berezikov E. Small RNAs and the control of transposons and viruses in drosophila. Trends in Microbiology. 2009;17:163–171. doi: 10.1016/j.tim.2009.01.003. [DOI] [PubMed] [Google Scholar]
- 7.Avadhanula V, Weasner BP, Hardy GG, Kumar JP, Hardy RW. A novel system for the launch of alphavirus RNA synthesis reveals a role for the Imd pathway in arthropod antiviral response. PLoS Pathog. 2009;5:e1000582. doi: 10.1371/journal.ppat.1000582. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 8.Carpenter J, Hutter S, Baines JF, Roller J, Saminadin-Peter SS, Parsch J, Jiggins FM. The transcriptional response of Drosophila melanogaster to infection with the sigma virus (Rhabdoviridae) PLoS ONE. 2009;4:e6838. doi: 10.1371/journal.pone.0006838. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 9.Costa A, Jan E, Sarnow P, Schneider D. The Imd pathway is involved in antiviral immune responses in Drosophila. PLoS ONE. 2009;4:e7436. doi: 10.1371/journal.pone.0007436. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 10.Deddouche S, Matt N, Budd A, Mueller S, Kemp C, Galiana-Arnoux D, Dostert C, Antoniewski C, Hoffmann JA, Imler JL. The DExD/H-box helicase Dicer-2 mediates the induction of antiviral activity in drosophila. Nat Immunol. 2008;9:1425–1432. doi: 10.1038/ni.1664. [DOI] [PubMed] [Google Scholar]
- 11.Dostert C, Jouanguy E, Irving P, Troxler L, Galiana-Arnoux D, Hetru C, Hoffmann JA, Imler JL. The Jak-STAT signaling pathway is required but not sufficient for the antiviral response of drosophila. Nat Immunol. 2005;6:946–953. doi: 10.1038/ni1237. [DOI] [PubMed] [Google Scholar]
- 12.Hedges LM, Johnson KN. Induction of host defence responses by Drosophila C virus. J Gen Virol. 2008;89:1497–1501. doi: 10.1099/vir.0.83684-0. [DOI] [PubMed] [Google Scholar]
- 13.Roxstrom-Lindquist K, Terenius O, Faye I. Parasite-specific immune response in adult Drosophila melanogaster: a genomic study. EMBO Rep. 2004;5:207–212. doi: 10.1038/sj.embor.7400073. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 14.Shelly S, Lukinova N, Bambina S, Berman A, Cherry S. Autophagy is an essential component of Drosophila immunity against vesicular stomatitis virus. Immunity. 2009;30:588–598. doi: 10.1016/j.immuni.2009.02.009. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 15.Zambon RA, Nandakumar M, Vakharia VN, Wu LP. The Toll pathway is important for an antiviral response in Drosophila. Proc Natl Acad Sci U S A. 2005;102:7257–7262. doi: 10.1073/pnas.0409181102. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 16.Fragkoudis R, Attarzadeh-Yazdi G, Nash AA, Fazakerley JK, Kohl A. Advances in dissecting mosquito innate immune responses to arbovirus infection. J Gen Virol. 2009;90:2061–2072. doi: 10.1099/vir.0.013201-0. [DOI] [PubMed] [Google Scholar]
- 17.Agaisse H, Petersen UM, Boutros M, Mathey-Prevot B, Perrimon N. Signaling Role of Hemocytes in Drosophila JAK/STAT-Dependent Response to Septic Injury. Dev Cell. 2003;5:441–450. doi: 10.1016/s1534-5807(03)00244-2. [DOI] [PubMed] [Google Scholar]
- 18.Lee YS, Nakahara K, Pham JW, Kim K, He Z, Sontheimer EJ, Carthew RW. Distinct roles for Drosophila Dicer-1 and Dicer-2 in the siRNA/miRNA silencing pathways. Cell. 2004;117:69–81. doi: 10.1016/s0092-8674(04)00261-2. [DOI] [PubMed] [Google Scholar]
- 19.Liu X, Jiang F, Kalidas S, Smith D, Liu Q. Dicer-2 and R2D2 coordinately bind siRNA to promote assembly of the siRISC complexes. Rna. 2006;12:1514–1520. doi: 10.1261/rna.101606. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 20.Irving P, Troxler L, Heuer TS, Belvin M, Kopczynski C, Reichhart JM, Hoffmann JA, Hetru C. A genome-wide analysis of immune responses in Drosophila. Proc Natl Acad Sci U S A. 2001;98:15119–15124. doi: 10.1073/pnas.261573998. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 21.Huang DW, Sherman BT, Lempicki RA. Systematic and integrative analysis of large gene lists using DAVID bioinformatics resources. Nature protocols. 2008;4:44–57. doi: 10.1038/nprot.2008.211. [DOI] [PubMed] [Google Scholar]
- 22.Pfeffer S. Identification of virally encoded microRNAs. Methods Enzymol. 2007;427:51–63. doi: 10.1016/S0076-6879(07)27003-X. [DOI] [PubMed] [Google Scholar]
- 23.Chotkowski HL, Ciota AT, Jia Y, Puig-Basagoiti F, Kramer LD, Shi PY, Glaser RL. West Nile virus infection of Drosophila melanogaster induces a protective RNAi response. Virology. 2008;377:197–206. doi: 10.1016/j.virol.2008.04.021. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 24.Galiana-Arnoux D, Dostert C, Schneemann A, Hoffmann JA, Imler JL. Essential function in vivo for Dicer-2 in host defense against RNA viruses in drosophila. Nat Immunol. 2006;7:590–597. doi: 10.1038/ni1335. [DOI] [PubMed] [Google Scholar]
- 25.Mueller S, Gausson V, Vodovar N, Deddouche S, Troxler L, Perot J, Pfeffer S, Hoffmann JA, Saleh MC, Imler JL. RNAi-mediated immunity provides strong protection against the negative-strand RNA vesicular stomatitis virus in Drosophila. Proc Natl Acad Sci U S A. 2010;107:19390–19395. doi: 10.1073/pnas.1014378107. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 26.Sabin LR, Zhou R, Gruber JJ, Lukinova N, Bambina S, Berman A, Lau CK, Thompson CB, Cherry S. Ars2 regulates both miRNA- and siRNA-dependent silencing and suppresses RNA virus infection in Drosophila. Cell. 2009;138:340–351. doi: 10.1016/j.cell.2009.04.045. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 27.Saleh MC, Tassetto M, van Rij RP, Goic B, Gausson V, Berry B, Jacquier C, Antoniewski C, Andino R. Antiviral immunity in Drosophila requires systemic RNA interference spread. Nature. 2009;458:346–350. doi: 10.1038/nature07712. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 28.van Rij RP, Saleh MC, Berry B, Foo C, Houk A, Antoniewski C, Andino R. The RNA silencing endonuclease Argonaute 2 mediates specific antiviral immunity in Drosophila melanogaster. Genes Dev. 2006;20:2985–2995. doi: 10.1101/gad.1482006. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 29.Wang XH, Aliyari R, Li WX, Li HW, Kim K, Carthew R, Atkinson P, Ding SW. RNA interference directs innate immunity against viruses in adult Drosophila. Science. 2006;312:452–454. doi: 10.1126/science.1125694. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 30.Zambon RA, Vakharia VN, Wu LP. RNAi is an antiviral immune response against a dsRNA virus in Drosophila melanogaster. Cell Microbiol. 2006;8:880–889. doi: 10.1111/j.1462-5822.2006.00688.x. [DOI] [PubMed] [Google Scholar]
- 31.Lim DH, Oh CT, Lee L, Hong JS, Noh SH, Hwang S, Kim S, Han SJ, Lee YS. The endogenous siRNA pathway in Drosophila impacts stress resistance and lifespan by regulating metabolic homeostasis. FEBS letters. 2011;585:3079–3085. doi: 10.1016/j.febslet.2011.08.034. [DOI] [PubMed] [Google Scholar]
- 32.D'Costa SM, Yao HJ, Bilimoria SL. Transcriptional mapping in Chilo iridescent virus infections. Arch Virol. 2004;149:723–742. doi: 10.1007/s00705-003-0232-4. [DOI] [PubMed] [Google Scholar]
- 33.Goto A, Yano T, Terashima J, Iwashita S, Oshima Y, Kurata S. Cooperative regulation of the induction of the novel antibacterial Listericin by peptidoglycan recognition protein LE and the JAK-STAT pathway. J Biol Chem. 2010;285:15731–15738. doi: 10.1074/jbc.M109.082115. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 34.Draghici S, Khatri P, Eklund AC, Szallasi Z. Reliability and reproducibility issues in DNA microarray measurements. Trends Genet. 2006;22:101–109. doi: 10.1016/j.tig.2005.12.005. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 35.Brun S, Vidal S, Spellman P, Takahashi K, Tricoire H, Lemaitre B. The MAPKKK Mekk1 regulates the expression of Turandot stress genes in response to septic injury in Drosophila. Genes Cells. 2006;11:397–407. doi: 10.1111/j.1365-2443.2006.00953.x. [DOI] [PubMed] [Google Scholar]
- 36.Cuellar WJ, Kreuze JF, Rajamaki ML, Cruzado KR, Untiveros M, Valkonen JP. Elimination of antiviral defense by viral RNase III. Proc Natl Acad Sci U S A. 2009;106:10354–10358. doi: 10.1073/pnas.0806042106. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 37.Moissiard G, Voinnet O. RNA silencing of host transcripts by cauliflower mosaic virus requires coordinated action of the four Arabidopsis Dicer-like proteins. Proc Natl Acad Sci U S A. 2006;103:19593–19598. doi: 10.1073/pnas.0604627103. [DOI] [PMC free article] [PubMed] [Google Scholar] [Retracted]
- 38.Blevins T, Rajeswaran R, Shivaprasad PV, Beknazariants D, Si-Ammour A, Park HS, Vazquez F, Robertson D, Meins F, Jr, Hohn T, Pooggin MM. Four plant Dicers mediate viral small RNA biogenesis and DNA virus induced silencing. Nucleic Acids Res. 2006;34:6233–6246. doi: 10.1093/nar/gkl886. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 39.Tabeta K, Georgel P, Janssen E, Du X, Hoebe K, Crozat K, Mudd S, Shamel L, Sovath S, Goode J, Alexopoulou L, Flavell RA, Beutler B. Toll-like receptors 9 and 3 as essential components of innate immune defense against mouse cytomegalovirus infection. Proc Natl Acad Sci U S A. 2004;101:3516–3521. doi: 10.1073/pnas.0400525101. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 40.Zhang SY, Jouanguy E, Ugolini S, Smahi A, Elain G, Romero P, Segal D, Sancho-Shimizu V, Lorenzo L, Puel A, Picard C, Chapgier A, Plancoulaine S, Titeux M, Cognet C, von Bernuth H, Ku CL, Casrouge A, Zhang XX, Barreiro L, Leonard J, Hamilton C, Lebon P, Heron B, Vallee L, Quintana-Murci L, Hovnanian A, Rozenberg F, Vivier E, Geissmann F, Tardieu M, Abel L, Casanova JL. TLR3 deficiency in patients with herpes simplex encephalitis. Science. 2007;317:1522–1527. doi: 10.1126/science.1139522. [DOI] [PubMed] [Google Scholar]
- 41.Plus N, Croizier G, Jousset FX, David J. Picornaviruses of laboratory and wild Drosophila melanogaster: geographical distribution and serotypic composition. Ann Microbiol (Paris) 1975;126:107–117. [PubMed] [Google Scholar]
- 42.Berry B, Deddouche S, Kirschner D, Imler JL, Antoniewski C. Viral suppressors of RNA silencing hinder exogenous and endogenous small RNA pathways in Drosophila. PLoS ONE. 2009;4:e5866. doi: 10.1371/journal.pone.0005866. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 43.Li H, Li WX, Ding SW. Induction and suppression of RNA silencing by an animal virus. Science. 2002;296:1319–1321. doi: 10.1126/science.1070948. [DOI] [PubMed] [Google Scholar]
- 44.Eleftherianos I, Won S, Chtarbanova S, Squiban B, Ocorr K, Bodmer R, Beutler B, Hoffmann JA, Imler JL. ATP-sensitive potassium channel (KATP)-dependent regulation of cardiotropic viral infections. Proc Natl Acad Sci U S A. 2011;108:12024–12029. doi: 10.1073/pnas.1108926108. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 45.Yagi Y, Ip YT. Helicase89B is a Mot1p/BTAF1 homologue that mediates an antimicrobial response in Drosophila. EMBO Rep. 2005;6:1088–1094. doi: 10.1038/sj.embor.7400542. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 46.Tsuda M, Langmann C, Harden N, Aigaki T. The RING-finger scaffold protein Plenty of SH3s targets TAK1 to control immunity signalling in Drosophila. EMBO Rep. 2005;6:1082–1087. doi: 10.1038/sj.embor.7400537. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 47.Chao JA, Lee JH, Chapados BR, Debler EW, Schneemann A, Williamson JR. Dual modes of RNA-silencing suppression by Flock House virus protein B2. Nat Struct Mol Biol. 2005;12:952–957. doi: 10.1038/nsmb1005. [DOI] [PubMed] [Google Scholar]
- 48.Carty M, Goodbody R, Schroder M, Stack J, Moynagh PN, Bowie AG. The human adaptor SARM negatively regulates adaptor protein TRIF-dependent Toll-like receptor signaling. Nat Immunol. 2006;7:1074–1081. doi: 10.1038/ni1382. [DOI] [PubMed] [Google Scholar]
- 49.Dorner S, Lum L, Kim M, Paro R, Beachy PA, Green R. A genomewide screen for components of the RNAi pathway in Drosophila cultured cells. Proc Natl Acad Sci U S A. 2006;103:11880–11885. doi: 10.1073/pnas.0605210103. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 50.Arkov AL, Ramos A. Building RNA-protein granules: insight from the germline. Trends Cell Biol. 2010;20:482–490. doi: 10.1016/j.tcb.2010.05.004. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 51.Sabatier L, Jouanguy E, Dostert C, Zachary D, Dimarcq JL, Bulet P, Imler JL. Pherokine-2 and-3: Two Drosophila molecules related to pheromone/odor-binding proteins induced by viral and bacterial infections. Eur J Biochem. 2003;270:3398–3407. doi: 10.1046/j.1432-1033.2003.03725.x. [DOI] [PubMed] [Google Scholar]
- 52.Becker T, Loch G, Beyer M, Zinke I, Aschenbrenner AC, Carrera P, Inhester T, Schultze JL, Hoch M. FOXO-dependent regulation of innate immune homeostasis. Nature. 2010;463:369–373. doi: 10.1038/nature08698. [DOI] [PubMed] [Google Scholar]
- 53.Ganz T. Defensins: antimicrobial peptides of innate immunity. Nat Rev Immunol. 2003;3:710–720. doi: 10.1038/nri1180. [DOI] [PubMed] [Google Scholar]
- 54.Souza-Neto JA, Sim S, Dimopoulos G. An evolutionary conserved function of the JAK-STAT pathway in anti-dengue defense. Proc Natl Acad Sci U S A. 2009;106:17841–17846. doi: 10.1073/pnas.0905006106. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 55.Buchon N, Broderick NA, Poidevin M, Pradervand S, Lemaitre B. Drosophila intestinal response to bacterial infection: activation of host defense and stem cell proliferation. Cell host & microbe. 2009;5:200–211. doi: 10.1016/j.chom.2009.01.003. [DOI] [PubMed] [Google Scholar]
- 56.Cronin SJ, Nehme NT, Limmer S, Liegeois S, Pospisilik JA, Schramek D, Leibbrandt A, Simoes Rde M, Gruber S, Puc U, Ebersberger I, Zoranovic T, Neely GG, von Haeseler A, Ferrandon D, Penninger JM. Genome-wide RNAi screen identifies genes involved in intestinal pathogenic bacterial infection. Science. 2009;325:340–343. doi: 10.1126/science.1173164. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 57.Jiang H, Patel PH, Kohlmaier A, Grenley MO, McEwen DG, Edgar BA. Cytokine/Jak/Stat signaling mediates regeneration and homeostasis in the Drosophila midgut. Cell. 2009;137:1343–1355. doi: 10.1016/j.cell.2009.05.014. [DOI] [PMC free article] [PubMed] [Google Scholar]
Associated Data
This section collects any data citations, data availability statements, or supplementary materials included in this article.


