Abstract
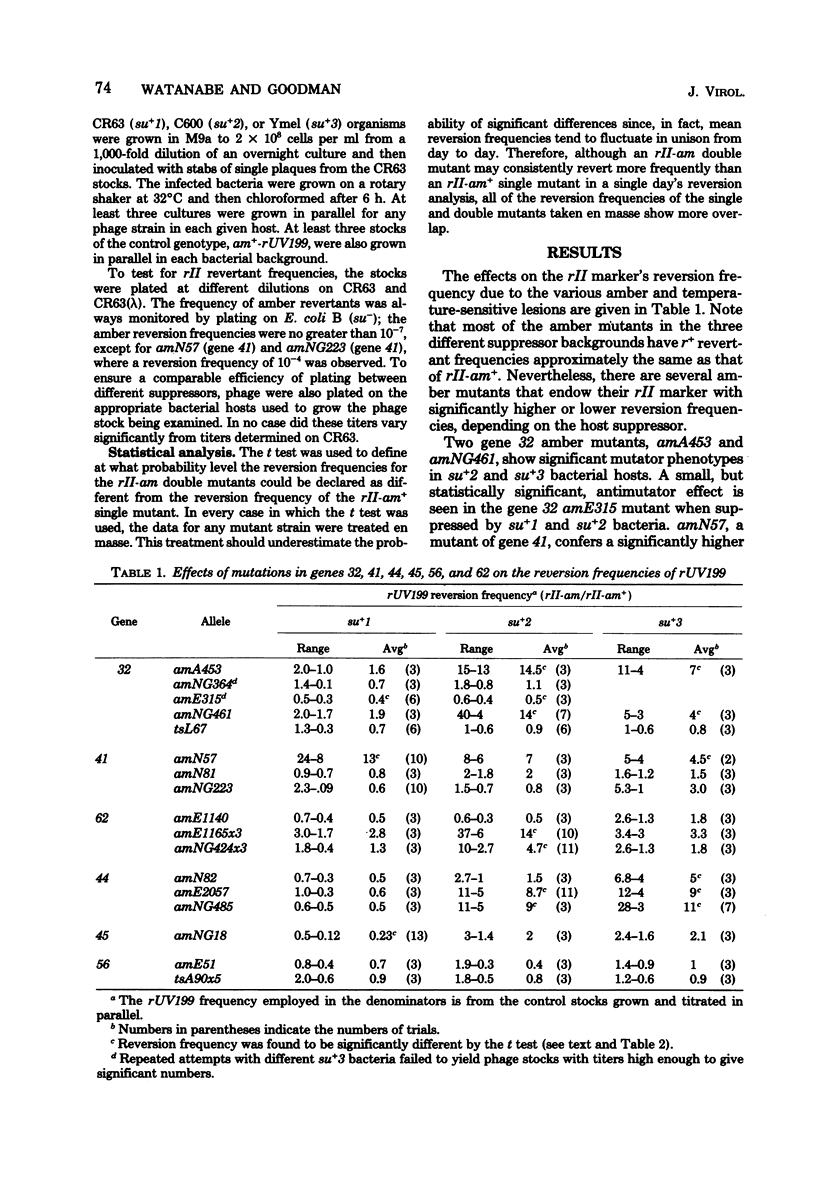
Bacteriophage T4 genes 32, 41, 44, 45, 56, and 62 are essential to DNA replication. Amber mutants (suppressed by su+1, su+2, or su+3 bacteria) in these genes were examined for any mutator or antimutator effects on the reversion of a transition mutation. In every case except for mutations in gene 56, elevated or lowered error frequencies were observed. These results indicate the importance of all of the replicative proteins in the determination of error frequency.
Full text
PDF



Selected References
These references are in PubMed. This may not be the complete list of references from this article.
- Alberts B. M., Frey L. T4 bacteriophage gene 32: a structural protein in the replication and recombination of DNA. Nature. 1970 Sep 26;227(5265):1313–1318. doi: 10.1038/2271313a0. [DOI] [PubMed] [Google Scholar]
- Bernstein C., Bernstein H., Mufti S., Strom B. Stimulation of mutation in phage T 4 by lesions in gene 32 and by thymidine imbalance. Mutat Res. 1972 Oct;16(2):113–119. doi: 10.1016/0027-5107(72)90171-6. [DOI] [PubMed] [Google Scholar]
- Bernstein H. Reversion of frameshift mutations stimulated by lesions in early function genes of bacteriophage T4. J Virol. 1971 Apr;7(4):460–466. doi: 10.1128/jvi.7.4.460-466.1971. [DOI] [PMC free article] [PubMed] [Google Scholar]
- CHAMPE S. P., BENZER S. Reversal of mutant phenotypes by 5-fluorouracil: an approach to nucleotide sequences in messenger-RNA. Proc Natl Acad Sci U S A. 1962 Apr 15;48:532–546. doi: 10.1073/pnas.48.4.532. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Campbell D. A., Rowe L. B. Enhanced recovery of rII mutations in the absence of selection in a ligase-negative mutant of bacteriophage T 4 . Mutat Res. 1972 Aug;15(4):361–369. doi: 10.1016/0027-5107(72)90001-2. [DOI] [PubMed] [Google Scholar]
- Carroll R. B., Neet K. E., Goldthwait D. A. Self-association of gene-32 protein of bacteriophage T4. Proc Natl Acad Sci U S A. 1972 Sep;69(9):2741–2744. doi: 10.1073/pnas.69.9.2741. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Carroll R. B., Neet K., Goldthwait D. A. Studies of the self-association of bacteriophage T4 gene 32 protein by equilibrium sedimentation. J Mol Biol. 1975 Jan 25;91(3):275–291. doi: 10.1016/0022-2836(75)90380-0. [DOI] [PubMed] [Google Scholar]
- Chiu C. S., Greenberg G. R. Mutagenic effect of temperature-sensitive mutants of gene 42 (dCMP hydroxymethylase) of bacteriophage T4. J Virol. 1973 Jul;12(1):199–201. doi: 10.1128/jvi.12.1.199-201.1973. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Drake J. W., Allen E. F. Antimutagenic DNA polymerases of bacteriophage T4. Cold Spring Harb Symp Quant Biol. 1968;33:339–344. doi: 10.1101/sqb.1968.033.01.039. [DOI] [PubMed] [Google Scholar]
- Drake J. W., Allen E. F., Forsberg S. A., Preparata R. M., Greening E. O. Genetic control of mutation rates in bacteriophageT4. Nature. 1969 Mar 22;221(5186):1128–1132. [PubMed] [Google Scholar]
- Drake J. W. The genetic control of spontaneous and induced mutation rates in bacteriophage T4. Genetics. 1973 Apr;73(Suppl):45–64. [PubMed] [Google Scholar]
- Hopfield J. J. Kinetic proofreading: a new mechanism for reducing errors in biosynthetic processes requiring high specificity. Proc Natl Acad Sci U S A. 1974 Oct;71(10):4135–4139. doi: 10.1073/pnas.71.10.4135. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Huberman J. A., Kornberg A., Alberts B. M. Stimulation of T4 bacteriophage DNA polymerase by the protein product of T4 gene 32. J Mol Biol. 1971 Nov 28;62(1):39–52. doi: 10.1016/0022-2836(71)90129-x. [DOI] [PubMed] [Google Scholar]
- Koch R. E., Drake J. W. Ligase-defective bacteriophage T4. I. Effects on mutation rates. J Virol. 1973 Jan;11(1):35–40. doi: 10.1128/jvi.11.1.35-40.1973. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Koch R. E., McGaw M. K., Drake J. W. Mutator mutations in bacteriophage T4 gene 32 (DNA unwinding protein). J Virol. 1976 Aug;19(2):490–494. doi: 10.1128/jvi.19.2.490-494.1976. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Manoil C., Sinha N., Alberts B. Intracellular DNA-protein complexes from bacteriophage T4-infected cells isolated by a rapid two-step procedure. Characterization and identification of the protein components. J Biol Chem. 1977 Apr 25;252(8):2734–2741. [PubMed] [Google Scholar]
- Moise H., Hosoda J. T4 gene 32 protein model for control of activity at replication fork. Nature. 1976 Feb 12;259(5543):455–458. doi: 10.1038/259455a0. [DOI] [PubMed] [Google Scholar]
- Molineux I. J., Gefter M. L. Properties of the Escherichia coli in DNA binding (unwinding) protein: interaction with DNA polymerase and DNA. Proc Natl Acad Sci U S A. 1974 Oct;71(10):3858–3862. doi: 10.1073/pnas.71.10.3858. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Morris C. F., Sinha N. K., Alberts B. M. Reconstruction of bacteriophage T4 DNA replication apparatus from purified components: rolling circle replication following de novo chain initiation on a single-stranded circular DNA template. Proc Natl Acad Sci U S A. 1975 Dec;72(12):4800–4804. doi: 10.1073/pnas.72.12.4800. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Muzyczka N., Poland R. L., Bessman M. J. Studies on the biochemical basis of spontaneous mutation. I. A comparison of the deoxyribonucleic acid polymerases of mutator, antimutator, and wild type strains of bacteriophage T4. J Biol Chem. 1972 Nov 25;247(22):7116–7122. [PubMed] [Google Scholar]
- Sigal N., Delius H., Kornberg T., Gefter M. L., Alberts B. A DNA-unwinding protein isolated from Escherichia coli: its interaction with DNA and with DNA polymerases. Proc Natl Acad Sci U S A. 1972 Dec;69(12):3537–3541. doi: 10.1073/pnas.69.12.3537. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Speyer J. F. Mutagenic DNA polymerase. Biochem Biophys Res Commun. 1965 Oct 8;21(1):6–8. doi: 10.1016/0006-291x(65)90417-1. [DOI] [PubMed] [Google Scholar]
- Williams W. E., Drake J. W. Mutator mutations in bacteriophage T4 gene 42 (dHMC hydroxymethylase). Genetics. 1977 Jul;86(3):501–511. doi: 10.1093/genetics/86.3.501. [DOI] [PMC free article] [PubMed] [Google Scholar]



