Abstract
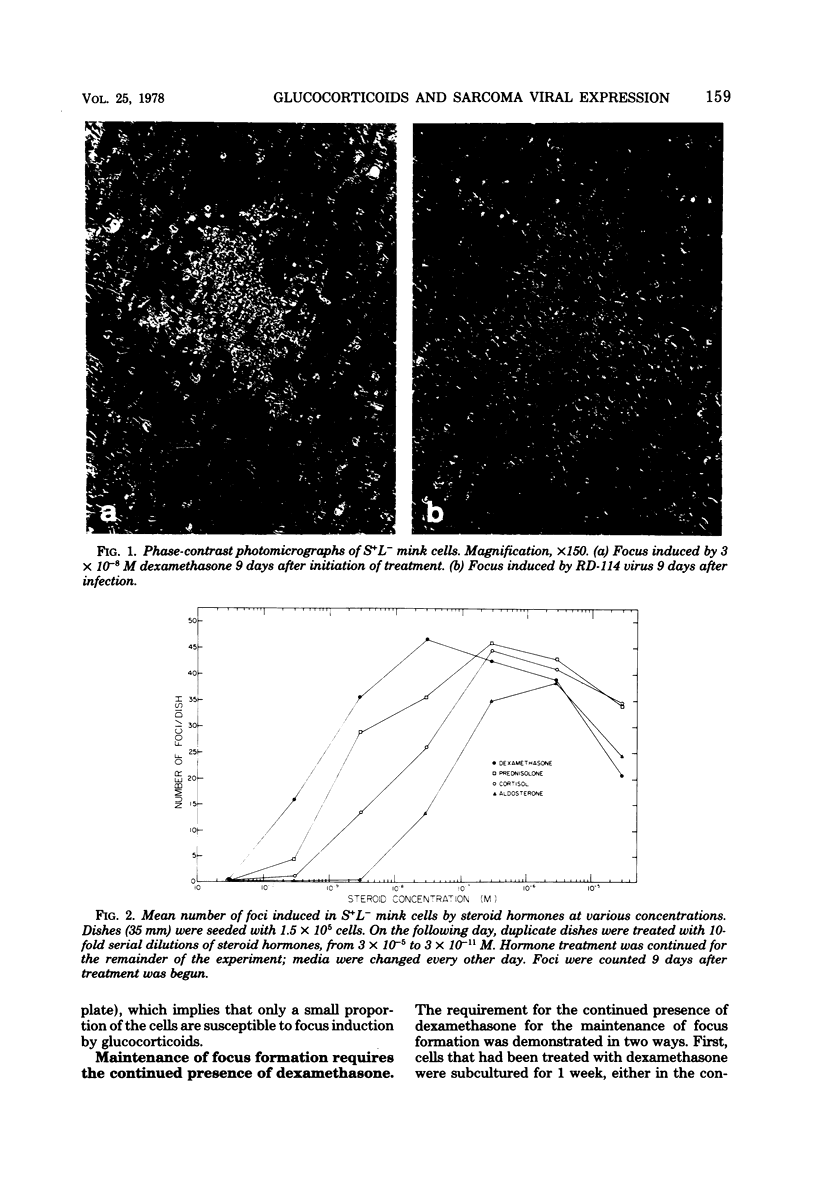
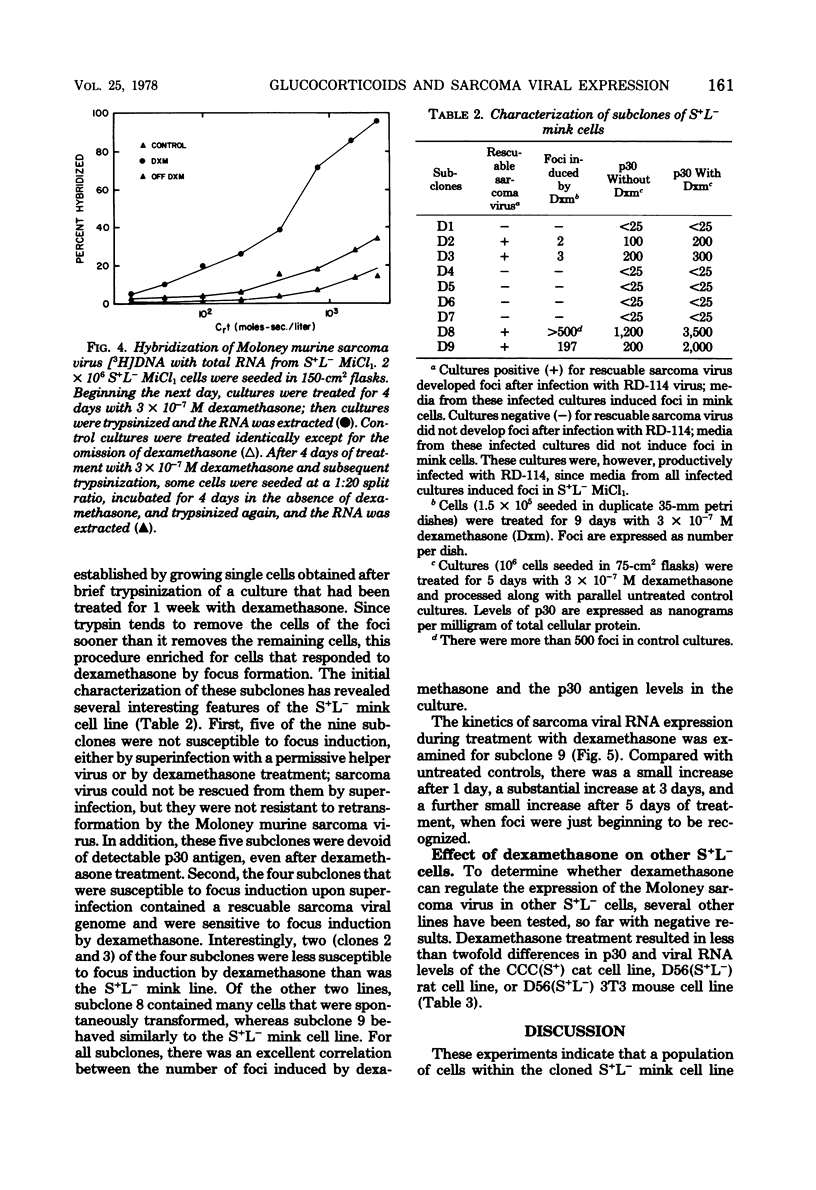
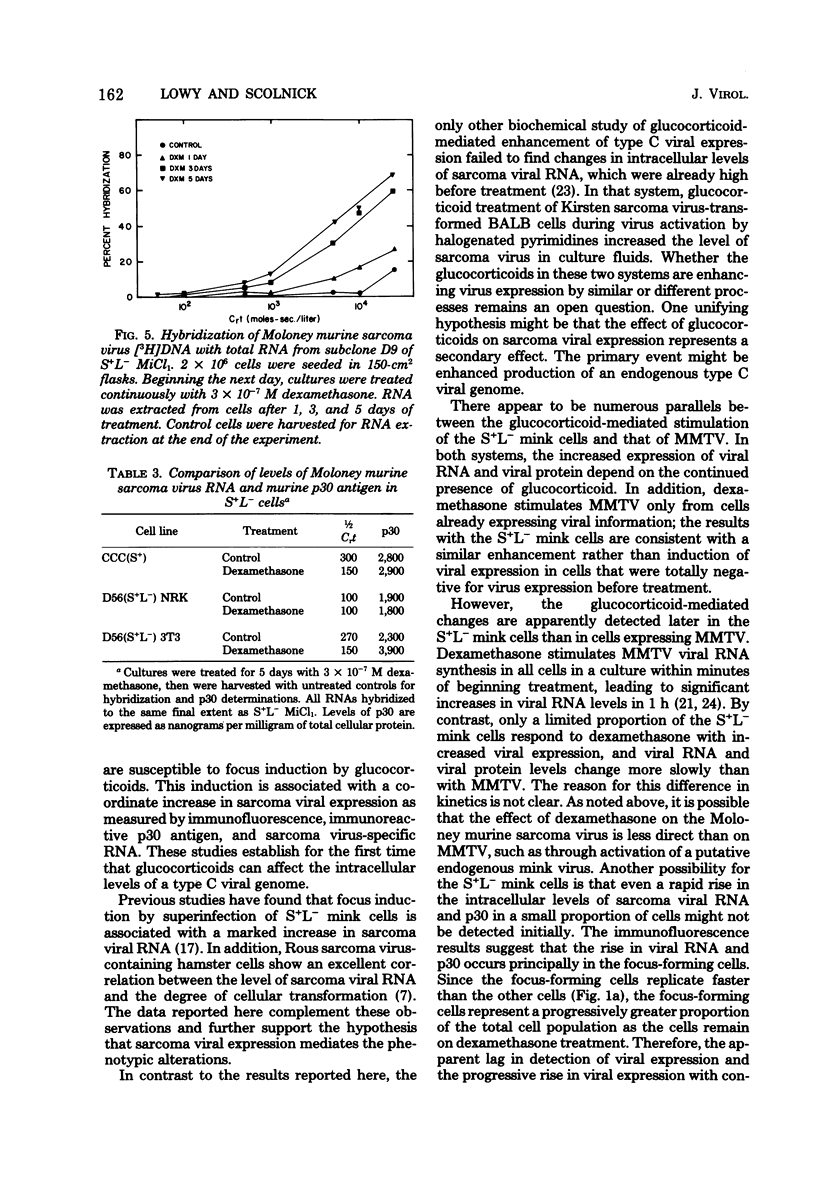
Dexamethasone (3 X 10(-10) to 3 X 10(-6) M) induced foci of morphologically transformed cells in a small proportion of a mink cell line that contains the Moloney murine sarcoma viral genome (S+L-). The induction was glucocorticoid specific, since other steroids with glucocorticoid activity (prednisolone, cortisol, and aldosterone) induced foci with an efficiency that paralleled their glucocorticoid activity, and steroids lacking glucocorticoid activity (17B-estradiol, testosterone, and progesterone) failed to induce foci. Viral antigen, as measured by specific immunofluorescence, was localized to the foci. The induction of foci by dexamethasone (3 X 10(-7)) was accompanied by an approximately 10-fold increase in intracellular Moloney murine sarcoma virus-specific RNA and viral p30 antigen. Removal of dexamethasone was followed by the disappearance of foci and a decrease in viral RNA and p30. In this cell system, therefore, glucocorticoids can affect the intracellular levels of type C viral RNA and protein.
Full text
PDF



Images in this article
Selected References
These references are in PubMed. This may not be the complete list of references from this article.
- Bassin R. H., Phillips L. A., Kramer M. J., Haapala D. K., Peebles P. T., Nomura S., Fischinger P. J. Transformation of mouse 3T3 cells by murine sarcoma virus: release of virus-like particles in the absence of replicating murine leukemia helper virus. Proc Natl Acad Sci U S A. 1971 Jul;68(7):1520–1524. doi: 10.1073/pnas.68.7.1520. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Bassin R. H., Tuttle N., Fischinger P. J. Isolation of murine sarcoma virus-transformed mouse cells which are negative for leukemia virus from agar suspension cultures. Int J Cancer. 1970 Jul 15;6(1):95–107. doi: 10.1002/ijc.2910060114. [DOI] [PubMed] [Google Scholar]
- Benveniste R. E., Scolnick E. M. RNA in mammalian sarcoma virus transformed nonproducer cells homologous to murine leukemia virus RNA. Virology. 1973 Feb;51(2):370–382. doi: 10.1016/0042-6822(73)90436-4. [DOI] [PubMed] [Google Scholar]
- Berliner J. A., Gerschenson L. E. The effects of a glucocorticoid on the cell surface of RLC-GAI cells. J Cell Physiol. 1975 Dec;86(3 Pt 1):523–531. doi: 10.1002/jcp.1040860309. [DOI] [PubMed] [Google Scholar]
- Birnstiel M. L., Sells B. H., Purdom I. F. Kinetic complexity of RNA molecules. J Mol Biol. 1972 Jan 14;63(1):21–39. doi: 10.1016/0022-2836(72)90519-0. [DOI] [PubMed] [Google Scholar]
- Britten R. J., Graham D. E., Neufeld B. R. Analysis of repeating DNA sequences by reassociation. Methods Enzymol. 1974;29:363–418. doi: 10.1016/0076-6879(74)29033-5. [DOI] [PubMed] [Google Scholar]
- Deng C. T., Boettiger D., Macpherson I., Varmus H. E. The persistence and expression of virus-specific DNA in revertants of Rous sarcoma virus-transformed BHK-21 cells. Virology. 1974 Dec;62(2):512–521. doi: 10.1016/0042-6822(74)90411-5. [DOI] [PubMed] [Google Scholar]
- Fischinger P. J., Blevins C. S., Nomura S. Simple, quantitative assay for both xenotropic murine leukemia and ecotropic feline leukemia viruses. J Virol. 1974 Jul;14(1):177–179. doi: 10.1128/jvi.14.1.177-179.1974. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Fischinger P. J., Nomura S., Peebles P. T., Haapala D. K., Bassin R. H. Reversion of murine sarcoma virus transformed mouse cells: variants without a rescuable sarcoma virus. Science. 1972 Jun 2;176(4038):1033–1035. doi: 10.1126/science.176.4038.1033. [DOI] [PubMed] [Google Scholar]
- Frankel A. S., Haapala D. K., Neubauer R. L., Fischinger P. J. Elimination of the sarcoma genome from murine sarcoma virus transformed cat cells. Science. 1976 Mar 26;191(4233):1264–1266. doi: 10.1126/science.1257745. [DOI] [PubMed] [Google Scholar]
- McAllister R. M., Nicolson M., Gardner M. B., Rongey R. W., Rasheed S., Sarma P. S., Huebner R. J., Hatanaka M., Oroszlan S., Gilden R. V. C-type virus released from cultured human rhabdomyosarcoma cells. Nat New Biol. 1972 Jan 5;235(53):3–6. doi: 10.1038/newbio235003a0. [DOI] [PubMed] [Google Scholar]
- Paran M., Gallo R. C., Richardson L. S., Wu A. M. Adrenal corticosteroids enhance production of type-C virus induced by 5-iodo-2'-deoxyuridine from cultured mouse fibroblasts. Proc Natl Acad Sci U S A. 1973 Aug;70(8):2391–2395. doi: 10.1073/pnas.70.8.2391. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Parks W. P., Noon M. C., Gilden R., Scolnick E. M. Serological studies with low-molecular-weight polypeptides from the Moloney strain of murine leukemia virus. J Virol. 1975 Jun;15(6):1385–1395. doi: 10.1128/jvi.15.6.1385-1395.1975. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Parks W. P., Scolnick E. M., Kozikowski E. H. Dexamethasone stimulation of murine mammary tumor virus expression: a tissue culture source of virus. Science. 1974 Apr 12;184(4133):158–160. doi: 10.1126/science.184.4133.158. [DOI] [PubMed] [Google Scholar]
- Peebles P. T. An in vitro focus-induction assay for xenotropic murine leukemia virus, feline leukemia virus C, and the feline--primate viruses RD-114/CCC/M-7. Virology. 1975 Sep;67(1):288–291. doi: 10.1016/0042-6822(75)90427-4. [DOI] [PubMed] [Google Scholar]
- Peebles P. T., Scolnick E. M., Howk R. S. Increased sarcoma virus RNA in cells transformed by leukemia viruses: model for leukemogenesis. Science. 1976 Jun 11;192(4244):1143–1145. doi: 10.1126/science.179144. [DOI] [PubMed] [Google Scholar]
- Peeples P. T., Gerwin B. I., Papageorge A. G., Smith S. G. Murine sarcoma virus defectiveness. Viral polymerase expression murine and nonmurine host cells transformed by S+L-type murine sarcoma virus. Virology. 1975 Oct;67(2):344–355. doi: 10.1016/0042-6822(75)90436-5. [DOI] [PubMed] [Google Scholar]
- Ringold G. M., Yamamoto K. R., Tomkins G. M., Bishop M., Varmus H. E. Dexamethasone-mediated induction of mouse mammary tumor virus RNA: a system for studying glucocorticoid action. Cell. 1975 Nov;6(3):299–305. doi: 10.1016/0092-8674(75)90181-6. [DOI] [PubMed] [Google Scholar]
- Ringold G., Lasfargues E. Y., Bishop J. M., Varmus H. E. Production of mouse mammary tumor virus by cultured cells in the absence and presence of hormones: assay by molecular hybridization. Virology. 1975 May;65(1):135–147. doi: 10.1016/0042-6822(75)90014-8. [DOI] [PubMed] [Google Scholar]
- Scolnick E. M., Howk R. S., Anisowicz A., Peebles P. T., Scher C. D., Parks W. P. Separation of sarcoma virus-specific and leukemia virus-specific genetic sequences of Moloney sarcoma virus. Proc Natl Acad Sci U S A. 1975 Nov;72(11):4650–4654. doi: 10.1073/pnas.72.11.4650. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Scolnick E. M., Young H. A., Parks W. P. Biochemical and physiological mechanisms in glucocorticoid hormone induction of mouse mammary tumor virus. Virology. 1976 Jan;69(1):148–156. doi: 10.1016/0042-6822(76)90202-6. [DOI] [PubMed] [Google Scholar]
- Wu A. M., Reitz M. S., Paran M., Gallo R. C. Mechanism of stimulation of murine type-C RNA tumor virus production by glucocorticoids: post-transcriptional effects. J Virol. 1974 Oct;14(4):802–812. doi: 10.1128/jvi.14.4.802-812.1974. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Young H. A., Shih T. Y., Scolnick E. M., Parks W. P. Steroid induction of mouse mammary tumor virus: effect upon synthesis and degradation of viral RNA. J Virol. 1977 Jan;21(1):139–146. doi: 10.1128/jvi.21.1.139-146.1977. [DOI] [PMC free article] [PubMed] [Google Scholar]