Summary
Cellular senescence is a defense mechanism in response to molecular damage which accumulates with aging. Correspondingly, the number of senescent cells has been reported to be greater in older than in younger subjects and furthermore associates with age-related pathologies. Inter-individual differences exist in the rate at which a person ages (biological age). Here, we studied whether younger biological age is related to fewer senescent cells in middle- aged individuals with the propensity for longevity, using p16INK4a as a marker for cellular senescence. We observed that a younger biological age associates with lower levels of p16INK4a positive cells in human skin.
Keywords: p16INK4a, biological age, familial longevity, cellular senescence
Increasing experimental evidence indicates that the accumulation of molecular damage underlies the aging process and age-related pathologies (Hamilton et al., 2001; Stadtman, 2001; Pamplona, 2008). Cellular defense mechanisms that occur in response to molecular damage include macromolecule repair, apoptosis, and cellular senescence. In tissues, the prevalence of senescent cells, that is, cells with a permanently arrested cell cycle, has been shown to increase with chronological age, both in animal models (Herbig et al., 2006; Janzen et al., 2006; Krishnamurthy et al., 2006; Molofsky et al., 2006) and in humans (Dimri et al., 1995; Ressler et al., 2006). Furthermore, increased numbers of senescent cells were found to associate with age-related pathologies such as atherosclerotic lesions (Minamino et al., 2002), diabetes (Sone & Kagawa, 2005), and renal disease (Melk et al., 2005; Sis et al., 2007). Higher levels of p16INK4a were associated as well with higher serum creatinine after renal transplantation (Koppelstaetter et al., 2008; McGlynn et al., 2009).
Within the Leiden Longevity Study (LLS), we have previously shown that middle-aged offspring from long-lived nonagenarian siblings are biologically younger than their partners, who are age- and environmentally matched controls (see Schoenmaker et al., 2006 for study design details). This is reflected in a lower mortality rate (Schoenmaker et al., 2006), a lower prevalence of cardiometabolic diseases (Westendorp et al., 2009), beneficial glucose and lipid metabolism, preservation of insulin sensitivity (Wijsman et al., 2011), and resistance to cellular stress in vitro (Dekker et al., 2009). The cyclin- dependent kinase inhibitor CDKN2A, commonly referred to as p16INK4a or p16, has been established as a general marker of cellular senescence as p16INK4a was observed to be expressed in most senescent cells in other studies (Krishnamurthy et al., 2004; Campisi Jd’Adda di, 2007; Baker et al., 2011; Rodier & Campisi, 2011). Here, we compared the frequency of p16INK4a positive cells in human skin biopsies from the upper inner arm of 89 middle-aged offspring with familial longevity (‘better agers’) with those of their 89 partners. We hypothesized that the younger biological age of the offspring would be reflected in lower numbers of p16INK4a positive cells when compared with their partners.
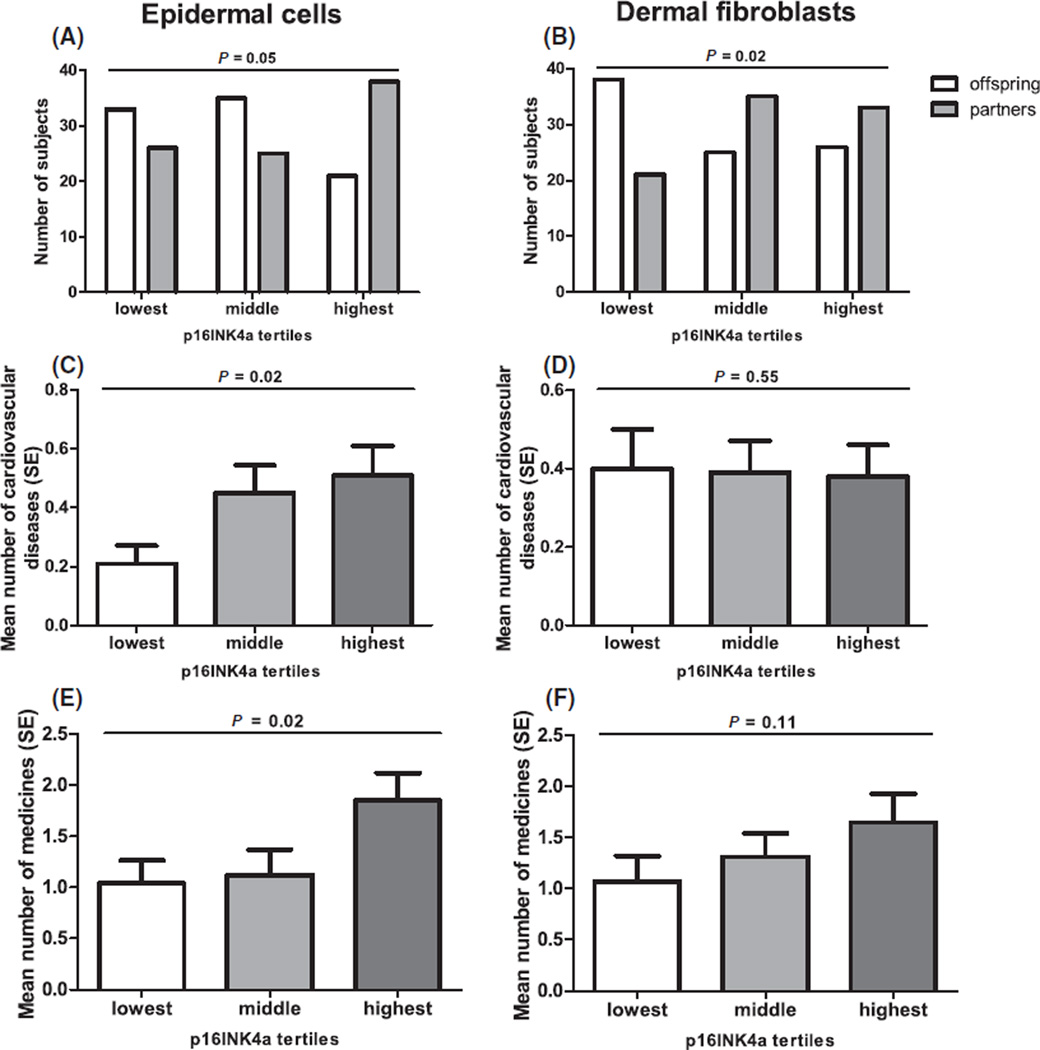
The age of the subjects varied from 46 to 81 years, with an average of 63 years. Further characteristics of the subjects are given in Table S1. Figure 1 shows representative figures of p16INK4a staining in both epidermis and dermis. The distribution of tertiles of p16INK4a positive cells differed considerably between offspring and partners in both epidermal cells and dermal fibroblasts. In the lowest tertile of p16INK4a positive cells, the offspring significantly outnumbered the partners, whereas in the highest tertile, the partners significantly outnumbered the offspring (Fig. 2). After adjustment for possible confounders such as age, gender, and smoking, these correlations remained essentially unaltered. Although these correlations were present in both epidermis and dermis, the correlation between the number of p16INK4a positive cells in the epidermis and those in the dermis was low (Pearson’s correlation coefficient 0.119, P = 0.115).
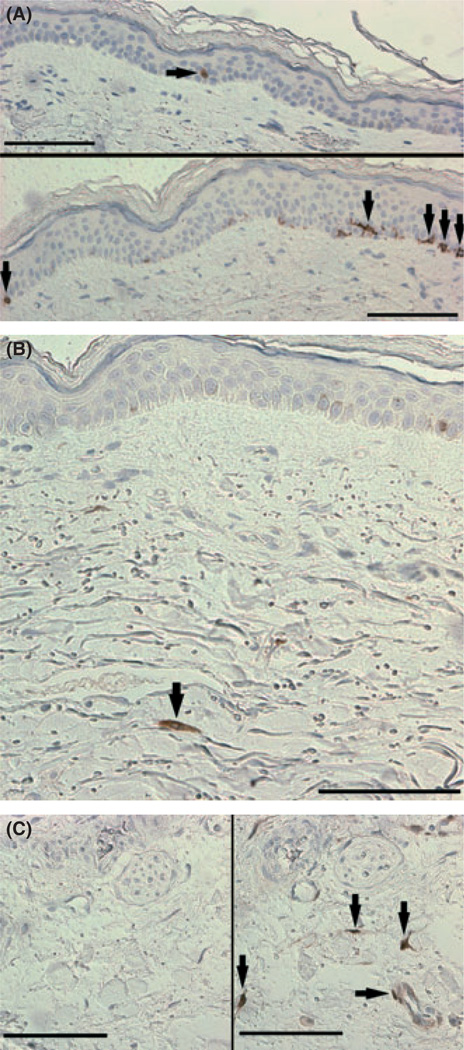
Fig. 1.
p16INK4a staining in human skin. (A) Representative p16INK4a staining of the epidermis from a subject in the middle tertile with one positive cell visible and an image of a section from a subject in the highest tertile (lower image with five positive cells visible). Epidermal staining was located along the basal membrane and mainly nuclear/perinuclear in nature although some cells displayed extensive cytoplasmic staining; note the dendritic nature of the fourth from right positive cell in the lower image characteristic of a melanocyte. (B) Representative p16INK4a staining of the dermis. (C) Negative control (no primary antibody, left image) and positive control (right image) of a skin sample used during all staining because of the consistent positive staining seen throughout the tissue. Line bars represent 100 µm (one dermal counting field was 315 by 315 µm) and black arrows the locations of positively stained cells.
Fig. 2.
Tertiles of p16INK4a positivity in human skin. Left column (A, C, E) presents epidermal cells, the right column (B, D, F) presents dermal fibroblasts. (A, B) A comparison of distribution of tertiles of p16INK4a positive cells between offspring from long-lived families and their partners, N = 178. (C, D) Average number ± standard error (SE) of cardiovascular diseases over tertiles of p16INK4a positive cells, N = 155. (E, F) Average number ± SE of medicines over tertiles of p16INK4a positive cells, N = 136. Tertiles of p16INK4a positive epidermal cells: lowest ≤ 0.30 (median = 0.00), middle 0.30–1.30 (median = 0.55), highest ≥ 1.30 (median = 3.09) cells per mm length of the epidermal–dermal junction. Tertiles of p16INK4a positive dermal fibroblasts: lowest ≤ 0.72 (median = 0.00), middle 0.72–2.05 (median = 1.29), highest ≥ 2.05 (median 3.20) cells per 1 mm2 dermis. P-values are adjusted for age, gender, and smoking in (A) and (B); for gender and smoking in (C), (D), (E) and (F).
Next, to confirm that the number of p16INK4a positive cells in human skin is a marker of biological age, we studied the relationship between the number of p16INK4a positive cells and age-related pathologies. The number of cardiovascular diseases (CVD) and medication use was significantly associated with tertiles of p16INK4a positive cells in epidermal cells (Fig. 2), also after adjustment for gender and smoking. There was no relationship between the tertiles of p16INK4a positive dermal fibroblasts and the number of CVD. However, a trend toward higher p16INK4a positivity in dermal fibroblasts and medication use was found.
For the first time, we have shown here that a marker of cellular senescence, p16INK4a, associates with familial longevity. In addition, we were able to reproduce earlier findings that cellular senescence in situ is associated with age-related pathologies. Although further evidence for the link between familial longevity and cellular senescence using other markers of senescence in skin or other tissues would strengthen these findings, the found association between p16INK4a and age-related disease is supported by a recent study. Clearance of p16INK4a positive cells in a mouse model was observed to delay the onset of age-related diseases (Baker et al., 2011). Smoking, a well-known risk factor for most age-related diseases, was previously also found to associate with expression of p16INK4a in peripheral blood T-cells (Liu et al., 2009). However, adjustment for smoking did not alter the results.
We have previously shown that skin fibroblasts from the middle-aged offspring respond to chemical stress in vitro with lower cellular senescence and higher apoptosis when compared with age-matched controls; this was comparable to the fibroblast response of young relative to old donors (Dekker et al., 2009). Thus, human familial longevity is not only associated with fewer p16INK4a positive skin cells in situ, and it is also associated with fewer senescent cells after a cellular stressor in vitro.
It is tempting to speculate based on the evidence presented here that the accumulation of senescent cells contributes to tissue failure and ill health. A reduced rate of cellular senescence appears to be a characteristic of offspring from long-lived families both in vitro and in situ, indicating a role for cellular senescence in the healthy phenotype of familial longevity. We hypothesize that offspring, enriched for genetic effects on longevity, carry also other molecular defense mechanisms (such as repair of damage and apoptosis) of better quality than their partners, resulting in less cellular senescence. Thus, the healthy phenotype of humans enriched for familiar longevity could be related to cellular senescence. However, further work is required to determine whether these findings are causative rather than associative in nature to better understand the role senescent cells have in vivo.
Supplementary Material
Acknowledgments
We thank N.M. Hudson for her help with skin tissue processing.
Funding
This work was funded by SenterNovem, IGE01014 and IGE5007, NGI/NWO; 05040202 and 050-060-810, Unilever PLC and FP6 036894.
Footnotes
Conflict of interest
Apart from the authors from Unilever, no funders had any role in the study design, analysis, or writing of this manuscript.
Supporting Information
Additional supporting information may be found in the online version of this article:
Table S1 Characteristics of subjects.
As a service to our authors and readers, this journal provides supporting information supplied by the authors. Such materials are peer-reviewed and may be re-organized for online delivery, but are not copy-edited or typeset. Technical support issues arising from supporting information (other than missing files) should be addressed to the authors.
References
- Baker DJ, Wijshake T, Tchkonia T, LeBrasseur NK, Childs BG, van de Sluis B, Kirkland JL, van Deursen JM. Clearance of p16Ink4a-positive senescent cells delays ageing-associated disorders. Nature. 2011;479:232–236. doi: 10.1038/nature10600. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Campisi Jd’Adda di FF. Cellular senescence: when bad things happen to good cells. Nat. Rev. Mol. Cell Biol. 2007;8:729–740. doi: 10.1038/nrm2233. [DOI] [PubMed] [Google Scholar]
- Dekker P, Maier AB, van Heemst D, de Koning-Treurniet C, Blom J, Dirks RW, Tanke HJ, Westendorp RG. Stress-induced responses of human skin fibroblasts in vitro reflect human longevity. Aging Cell. 2009;8:595–603. doi: 10.1111/j.1474-9726.2009.00506.x. [DOI] [PubMed] [Google Scholar]
- Dimri GP, Lee X, Basile G, Acosta M, Scott G, Roskelley C, Medrano EE, Linskens M, Rubelj I, Pereira-Smith O. A biomarker that identifies senescent human cells in culture and in aging skin in vivo. Proc. Natl. Acad. Sci. U.S.A. 1995;92:9363–9367. doi: 10.1073/pnas.92.20.9363. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Hamilton ML, Van Remmen H, Drake JA, Yang H, Guo ZM, Kewitt K, Walter CA, Richardson A. Does oxidative damage to DNA increase with age? Proc. Natl. Acad. Sci. U.S.A. 2001;98:10469–10474. doi: 10.1073/pnas.171202698. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Herbig U, Ferreira M, Condel L, Carey D, Sedivy JM. Cellular senescence in aging primates. Science. 2006;311:1257. doi: 10.1126/science.1122446. [DOI] [PubMed] [Google Scholar]
- Janzen V, Forkert R, Fleming HE, Saito Y, Waring MT, Dombkowski DM, Cheng T, DePinho RA, Sharpless NE, Scadden DT. Stem-cell ageing modified by the cyclin-dependent kinase inhibitor p16INK4a. Nature. 2006;443:421–426. doi: 10.1038/nature05159. [DOI] [PubMed] [Google Scholar]
- Koppelstaetter C, Schratzberger G, Perco P, Hofer J, Mark W, Ollinger R, Oberbauer R, Schwarz C, Mitterbauer C, Kainz A, Karkoszka H, Wiecek A, Mayer B, Mayer G. Markers of cellular senescence in zero hour biopsies predict outcome in renal transplantation. Aging Cell. 2008;7:491–497. doi: 10.1111/j.1474-9726.2008.00398.x. [DOI] [PubMed] [Google Scholar]
- Krishnamurthy J, Torrice C, Ramsey MR, Kovalev GI, Al-Regaiey K, Su L, Sharpless NE. Ink4a/Arf expression is a biomarker of aging. J. Clin. Invest. 2004;114:1299–1307. doi: 10.1172/JCI22475. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Krishnamurthy J, Ramsey MR, Ligon KL, Torrice C, Koh A, Bonner-Weir S, Sharpless NE. p16INK4a induces an age-dependent decline in islet regenerative potential. Nature. 2006;443:453–457. doi: 10.1038/nature05092. [DOI] [PubMed] [Google Scholar]
- Liu Y, Sanoff HK, Cho H, Burd CE, Torrice C, Ibrahim JG, Thomas NE, Sharpless NE. Expression of p16(INK4a) in peripheral blood T-cells is a biomarker of human aging. Aging Cell. 2009;8:439–448. doi: 10.1111/j.1474-9726.2009.00489.x. [DOI] [PMC free article] [PubMed] [Google Scholar]
- McGlynn LM, Stevenson K, Lamb K, Zino S, Brown M, Prina A, Kingsmore D, Shiels PG. Cellular senescence in pretransplant renal biopsies predicts postoperative organ function. Aging Cell. 2009;8:45–51. doi: 10.1111/j.1474-9726.2008.00447.x. [DOI] [PubMed] [Google Scholar]
- Melk A, Schmidt BM, Vongwiwatana A, Rayner DC, Halloran PF. Increased expression of senescence-associated cell cycle inhibitor p16INK4a in deteriorating renal transplants and diseased native kidney. Am. J. Transplant. 2005;5:1375–1382. doi: 10.1111/j.1600-6143.2005.00846.x. [DOI] [PubMed] [Google Scholar]
- Minamino T, Miyauchi H, Yoshida T, Ishida Y, Yoshida H, Komuro I. Endothelial cell senescence in human atherosclerosis: role of telomere in endothelial dysfunction. Circulation. 2002;105:1541–1544. doi: 10.1161/01.cir.0000013836.85741.17. [DOI] [PubMed] [Google Scholar]
- Molofsky AV, Slutsky SG, Joseph NM, He S, Pardal R, Krishnamurthy J, Sharpless NE, Morrison SJ. Increasing p16INK4a expression decreases forebrain progenitors and neurogenesis during ageing. Nature. 2006;443:448–452. doi: 10.1038/nature05091. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Pamplona R. Membrane phospholipids, lipoxidative damage and molecular integrity: a causal role in aging and longevity. Biochim. Biophys. Acta. 2008;1777:1249–1262. doi: 10.1016/j.bbabio.2008.07.003. [DOI] [PubMed] [Google Scholar]
- Ressler S, Bartkova J, Niederegger H, Bartek J, Scharffetter-Kochanek K, Jansen-Durr P, Wlaschek M. p16INK4A is a robust in vivo biomarker of cellular aging in human skin. Aging Cell. 2006;5:379–389. doi: 10.1111/j.1474-9726.2006.00231.x. [DOI] [PubMed] [Google Scholar]
- Rodier F, Campisi J. Four faces of cellular senescence. J. Cell Biol. 2011;192:547–556. doi: 10.1083/jcb.201009094. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Schoenmaker M, de Craen AJ, de Meijer PH, Beekman M, Blauw GJ, Slagboom PE, Westendorp RG. Evidence of genetic enrichment for exceptional survival using a family approach: the Leiden Longevity Study. Eur. J. Hum. Genet. 2006;14:79–84. doi: 10.1038/sj.ejhg.5201508. [DOI] [PubMed] [Google Scholar]
- Sis B, Tasanarong A, Khoshjou F, Dadras F, Solez K, Halloran PF. Accelerated expression of senescence associated cell cycle inhibitor p16INK4A in kidneys with glomerular disease. Kidney Int. 2007;71:218–226. doi: 10.1038/sj.ki.5002039. [DOI] [PubMed] [Google Scholar]
- Sone H, Kagawa Y. Pancreatic beta cell senescence contributes to the pathogenesis of type 2 diabetes in high-fat diet-induced diabetic mice. Diabetologia. 2005;48:58–67. doi: 10.1007/s00125-004-1605-2. [DOI] [PubMed] [Google Scholar]
- Stadtman ER. Protein oxidation in aging and age-related diseases. Ann N. Y. Acad. Sci. 2001;928:22–38. doi: 10.1111/j.1749-6632.2001.tb05632.x. [DOI] [PubMed] [Google Scholar]
- Westendorp RG, van HD, Rozing MP, Frolich M, Mooijaart SP, Blauw GJ, Beekman M, Heijmans BT, de Craen AJ, Slagboom PE. Nonagenarian siblings and their offspring display lower risk of mortality and morbidity than sporadic nonagenarians: The Leiden Longevity Study. J. Am. Geriatr. Soc. 2009;57:1634–1637. doi: 10.1111/j.1532-5415.2009.02381.x. [DOI] [PubMed] [Google Scholar]
- Wijsman CA, Rozing MP, Streefland TC, le CS, Mooijaart SP, Slagboom PE, Westendorp RG, Pijl H, van Heemst D. Familial longevity is marked by enhanced insulin sensitivity. Aging Cell. 2011;10:114–121. doi: 10.1111/j.1474-9726.2010.00650.x. [DOI] [PubMed] [Google Scholar]
Associated Data
This section collects any data citations, data availability statements, or supplementary materials included in this article.