Abstract
Background
The micropropagation is a powerful tool to scale up plants of economical and agronomical importance, enhancing crop productivity. However, a small but growing body of evidence suggests that epigenetic mechanisms, such as DNA methylation and histone modifications, can be affected under the in vitro conditions characteristic of micropropagation. Here, we tested whether the adaptation to different in vitro systems (Magenta boxes and Bioreactors) modified epigenetically different clones of Agave fourcroydes and A. angustifolia. Furthermore, we assessed whether these epigenetic changes affect the regulatory expression of KNOTTED1-like HOMEOBOX (KNOX) transcription factors.
Results
To gain a better understanding of epigenetic changes during in vitro and ex vitro conditions in Agave fourcroydes and A. angustifolia, we analyzed global DNA methylation, as well as different histone modification marks, in two different systems: semisolid in Magenta boxes (M) and temporary immersion in modular Bioreactors (B). No significant difference was found in DNA methylation in A. fourcroydes grown in either M or B. However, when A. fourcroydes was compared with A. angustifolia, there was a two-fold difference in DNA methylation between the species, independent of the in vitro system used. Furthermore, we detected an absence or a low amount of the repressive mark H3K9me2 in ex vitro conditions in plants that were cultured earlier either in M or B. Moreover, the expression of AtqKNOX1 and AtqKNOX2, on A. fourcroydes and A. angustifolia clones, is affected during in vitro conditions. Therefore, we used Chromatin ImmunoPrecipitation (ChIP) to know whether these genes were epigenetically regulated. In the case of AtqKNOX1, the H3K4me3 and H3K9me2 were affected during in vitro conditions in comparison with AtqKNOX2.
Conclusions
Agave clones plants with higher DNA methylation during in vitro conditions were better adapted to ex vitro conditions. In addition, A. fourcroydes and A. angustifolia clones displayed differential expression of the KNOX1 gene during in vitro conditions, which is epigenetically regulated by the H3K4me3 and H3K9me2 marks. The finding of an epigenetic regulation in key developmental genes will make it important in future studies to identify factors that help to find climate-resistant micropropagated plants.
Keywords: Epigenetics, In vitro, Histone methylation, Agave, KNOX genes
Background
DNA methylation and histone modifications are important epigenetic mechanisms for gene regulation in eukaryotes [1-3]. In plants, epigenetic mechanisms play an important role in development [4,5], flowering [6], pathogen recognition [7], senescence [8] and somaclonal variation [9-11]. It has been found that DNA methylation/demethylation is affected by exogenous and endogenous factors in both in vivo and in vitro conditions, such as auxin concentration, temperature and aging [5,12-14]. DNA methylation patterns can also change, depending on the method of plant propagation [15].
The use of in vitro plant propagation techniques allows the scale up of crops of agronomic importance [16,17], maintaining genetic stability among clones. Although clonal plants are usually very stable at the genetic level, epigenetic modifications in DNA [12,18,19], as well as histones [20-24], mainly by methylation in lysine 9 and 4 of the histone H3, could be affected and, therefore, induce somaclonal variation [11]. There is evidence of epigenetic changes, although some can be quite stable with time [25], occurring during in vitro culture. For instance, Valledor et al.[5] found that an increase in plant vigor and rejuvenation is due to DNA methylation. On the other hand, it has been found that the decrease in organogenesis capability of Pinus radiata was related to low levels of acetylation in histone H4 and high levels of DNA methylation [26]. It has been proposed that the changes in DNA methylation patterns in maize and apple are induced by tissue culture during in vitro conditions [27-29], and these methylation patterns play an important role during plant development [4,5]. Furthermore, not only methylation [30,31] but also demethylation in the DNA could cause epigenetic alterations that provoke abnormalities during the in vitro process [32,33]. One of the factors involved in epigenetic changes during in vitro conditions is the exposure to growth regulators, which are widely used in plant tissue culture [34,35] to promote multiplication and growth.
Plant growth regulators have been involved in the expression of several genes, and some studies have even suggested reciprocal links between growth regulators and homeobox genes [36]. One of the most studied homeobox genes regulated by plant growth regulators is the KNOTTED1-like HOMEOBOX (KNOX) transcription factor group [37-39]. In Arabidopsis thaliana, there are eight KNOX genes that have been divided into two classes. STM, KNAT1, KNAT2 and KNAT6 belong to class I, while KNAT3, KNAT4, KNAT5 and KNAT7 fall into class II [40]. The expression of two, KNAT1 and KNAT6, is altered by cytokinins and auxins [41-43]. Rupp et al.[41] found an increase in the transcription level of KNAT1 in the cytokinin-overproducing mutant amp1, which might occur by acting through the activation of KNAT1. Furthermore, Dean et al.[43] found in A. thaliana that exogenous auxin treatments alter the promoter activity of the gene KNAT6. In the same way, Montero-Cortes et al.[39] found that in somatic embryos from micropropagated coconut plants, the expression of CnKNOX1 was stimulated by gibberellic acid, while in CnKNOX2 the hormone produced a decrease in its expression. Taking all these findings as a whole, it is clear that plant growth regulators have an important impact on KNOX genes.
Studies done in Agave tequilana have shown that KNOX genes are associated with organogenesis during bulbil formation [44]. According to Abraham-Juarez et al.[44], AtqKNOX1, homologous to KNAT1, and AtqKNOX2, homologous to KNAT6, presented an increase in the levels of expression as bulbils mature. The regulation of KNOX genes in Agave has not been clearly understood, but studies in Arabidopsis and maize suggest that chromatin configuration could be an important factor in the regulation of these genes [40,45,46]. In humans, homeobox genes, which encode transcription factors evolutionarily conserved, are epigenetically regulated [47]. Therefore, it is possible that, as in animals, plant homeobox genes, such as KNOX, can also be epigenetically regulated.
Although the epigenome is highly dependent on the surrounding environment [48,49], the epigenetic changes that occur under in vitro conditions are still unknown, as is how these changes impact plants’ development in the field. In order to understand the effect of in vitro conditions on the epigenetic regulation of AtqKNOX1 (KNAT1) and AtqKNOX2 (KNAT6), in this study we provide a detailed epigenetic analysis comparing different Agave plants cultured under different in vitro and ex vitro conditions.
Results
In order to assess the epigenetic and molecular differences that might be occurring in Agave plants cultured in different in vitro systems (see Methods; Figure 1), as well as in ex vitro conditions, we used three different Agave fourcroydes clones (P20, P21 and P159) and one A. angustifolia clone (BM26) (Figure 1A). Sixteen-week-old clones were cultured in vitro (T0), as described in Methods, and transferred for five weeks to two different in vitro growth systems: semisolid in Magenta boxes (M) and temporary immersion in modular Bioreactors (B). After that treatment, the plants from each in vitro growth system (M and B) were transplanted to soil (S), where they stayed for two months ex vitro (Figure 1B). In total, five culture conditions (T0, M, B, SM and SB) were evaluated for each clone (P20, P21, P159 and BM26).
Figure 1.
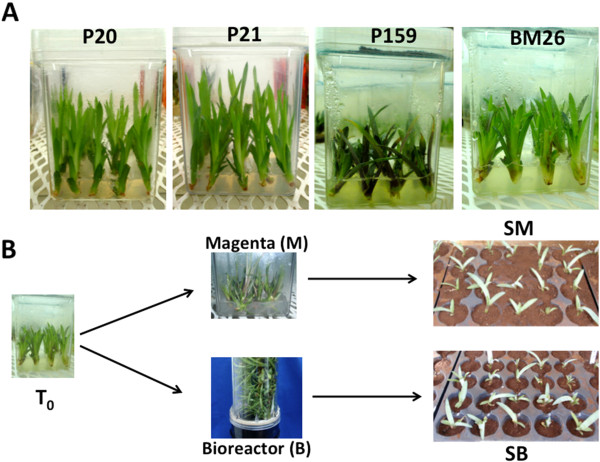
In vitro culture of Agave fourcroydes clones P20, P21 and P159 and A. angustifolia clone BM26. (A) Ten-week-old Agave plants grown under photoperiod conditions were considered as T0. (B) Experimental design for the culture of the plantlets cultured ten weeks in multiplication medium (T0). After ten weeks, the plantlets were moved to a semisolid growth medium in Magenta boxes (M) and temporary immersion in modular Bioreactors (B), where they stayed for five weeks. After that, the plantlets were transferred to soil previously cultured in M (SM) and to soil previously cultured in B (SB), where they stayed eight more weeks.
As previously reported by Robert et al.[50], it was observed that after five weeks in in vitro, the A. fourcroydes plants under Bioreactor conditions developed better than those grown in Magenta boxes; they were shorter, more vigorous and presented a more intense coloration (Figure 2). A similar situation was observed in the A. angustifolia clone (BM26), which also produced more leaves when cultured in the Bioreactor. These phenotypic characteristics were not conserved once the BM26 plants were evaluated eight weeks later in ex vitro conditions (SM and SB). Many of the BM26 plants did not survive after a couple of weeks ex vitro, and the few plants that did survive were small, pale and had only a few roots. In contrast, more than 90% of the A. fourcroydes plants (P20, P21 and P159) survived. They grew rapidly, produced many strong roots and the leaves developed the characteristic purple color in (data not shown).
Figure 2.

In vitro and ex vitro culture of Agave fourcroydes clones P20, P21 and P159 and A. angustifolia clone BM26. Plantlets were grown in two different systems: a semisolid growth medium in Magenta boxes (M) and temporary immersion in modular Bioreactors (B) for five weeks under in vitro conditions and for two months under ex vitro conditions (in soil; SM and SB).
Transfer to ex vitro conditions increases DNA methylation in Agave angustifolia
To gain insight into DNA methylation from plants grown in two different systems during in vitro (M and B) and ex vitro (SM and SB) conditions, three different A. fourcroydes clones (P20, P21 and P159) and one A. angustifolia clone (BM26) were chosen as starting material (Figures 1 and 3). Global DNA methylation rates were analyzed by HPLC (see Methods) from plants collected at T0, M, B, SM and SB. The global quantification of 5-methyl-deoxycitidine has shown that DNA methylation in A. fourcroydes (P20, P21 and P159) and A. angustifolia (BM26) cultured either in M or B is not significantly different (Figure 3). However, there is a two-fold difference in the DNA methylation rate of A. angustifolia in comparison with A. fourcroydes. Under ex vitro conditions (SM and SB), it was observed that BM26 increases its methylation rate two-fold in comparison with the in vitro conditions, while that of P20, P21 and P159 showed an increase of 1 to 3% from in vitro to ex vitro. Only one clone, P21, presented 1% less 5mdC under ex vitro versus in vitro conditions, showing 34% in the plants that were cultured in M in comparison with 33% for those cultured in SM. This suggests that ex vitro conditions could change the methylation behavior in different clones from the same species, potentially resulting in a change in a plant’s performance once it is in the field. On the other hand, we cannot rule out, at least for the case of A. fourcroydes, that the slight increase in DNA methylation observed during ex vitro in comparison with in vitro conditions (SM vs M and SB vs B) could be due to plant development.
Figure 3.
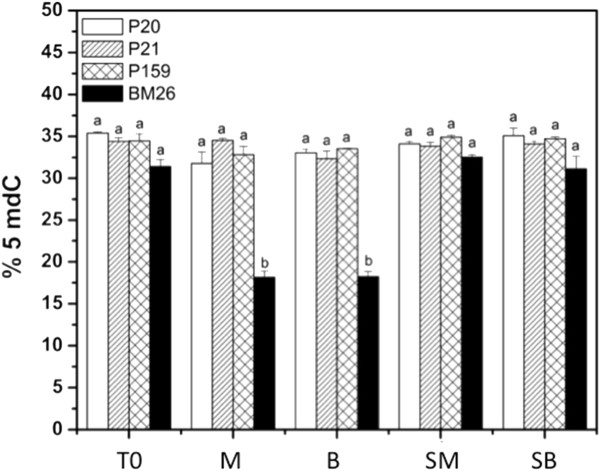
Methylation quantification in genomic DNA from Agave fourcroydes clones P20, P21 and P159 and A. angustifolia clone BM26. Plantlets from multiplication medium (T0), semisolid growth medium in Magenta boxes (M), temporary immersion in modular Bioreactors (B), soil previously cultured in M (SM) and soil previously cultured in B (SB) were used for the analysis. Each bar corresponds to the mean with its standard error (n = 15). Different letters in columns represent the statistical significance of mean differences at a given time by the Tukey test (P ≤ 0.01).
In vitro conditions induce methylation in H3K9
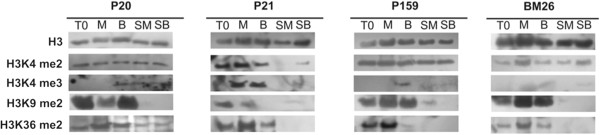
The Western blot analysis using antibodies against the H3K4 di- and tri-methylated isoforms, as well as for the H3K9me2 and H3K36me2 marks (see Methods), showed important changes in histone methylation patterns in all the clones during ex vitro conditions in comparison with the in vitro ones. In all ex vitro samples (SM and SB), we detected an absence or a low amount of the repressive mark H3K9me2, in spite of the fact that this mark had accumulated noticeably only during in vitro conditions (Figure 4). This suggests the possible formation of heterochromatin-like structures, as a result of the in vitro conditions. Marks H3K4me2, H3K4me3 and H3K36me2, which are implicated in the activation of transcription [51], were differentially detected among the clones and between in vitro and ex vitro conditions. For instance, H3K4me2 was present in all samples and all conditions except P21, for which this mark was absent in SM and present at a very low amount in SB. Furthermore, H3K36me2 was found in all samples in vitro but absent ex vitro, except for P20, where this mark was also found. In contrast, H3K4me3 presented a different pattern in each clone that may lead to the formation of eurochromatic structures as a stress mechanism provoked by in vitro conditions in different plants. Therefore, although the clones are genetically very similar, the response of each clone to the in vitro and ex vitro conditions is different, giving a specific epigenetic identity to each. For instance, in P20, H3K4me3 accumulated in B, SM and SB, in P21 this mark accumulated in M and B, and in P159 it accumulated only in B. In BM26, the H3K4me3 mark was slightly accumulated in B and SB.
Figure 4.
Immunoblot analyses of histone modification in Agave fourcroydes clones P20, P21 and P159 and A. angustifolia clone BM26. Analyses were carried out from plantlets cultured in multiplication medium (T0), semisolid growth medium in Magenta boxes (M), temporary immersion in modular Bioreactors (B), soil previously cultured in M (SM) and soil previously cultured in B (SB). Similar amounts of histone H3 were loaded as confirmed with an H3-specific antibody detecting the unmodified C-terminal part of H3.
KNOX expression is induced by in vitro conditions
The effect of in vitro conditions on the expression of the KNOX genes was analyzed by qRT-PCR assays of P20, P21, P159 and BM26 in T0, M, B, SM and SB (Figure 5). We found that AtqKNOX1 showed expression only in P21 and P159 at T0; and P20 at M. However, the highest expression detected was in P20 at B. In the case of ex vitro conditions, this gene was slightly expressed in P21 and P159 in SM and in P21 and BM26 in SB. Therefore, AtqKNOX1 is differentially expressed in the Agave clones during in vitro conditions. On the other hand, the expression of AtqKNOX2 was low or absent under all culture conditions in the BM26 clone. In the case of P159, the expression of AtqKNOX2 was almost the same while the clone was in vitro. However, once the plants were in ex vitro conditions, the expression of this gene was almost undetectable. It is worth mentioning that in the case of P21, the expression of AtqKNOX2 was low in all culture conditions. However, P20 presented the highest expression of AtqKNOX2 in B; while in T0 and M, the expression of this gene was very similar to P21 and P159, respectively. Furthermore, under ex vitro conditions (SM and SB), AtqKNOX2 expression in P20 was as low as that of P21.
Figure 5.
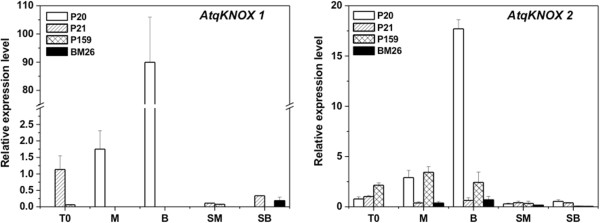
Real-time PCR expression of AtqKNOX expression in Agave fourcroydes clones P20, P21 and P159 and A. angustifolia clone BM26. Expression was performed from plantlets cultured in multiplication medium (T0), semisolid growth medium in Magenta boxes (M), temporary immersion in modular Bioreactors (B), soil previously cultured in M (SM) and soil previously cultured in B (SB). Total RNAs used as the templates for qRT-PCR were isolated from all conditions and clones. Each bar corresponds to the mean with its standard error (n = 3). Relative transcript abundances of AtqKNOX1 and AtqKNOX2 were normalized to the constitutive gene UBIQUITIN11. The means and standard deviation of biological replicates are shown.
AtqKNOX1 is epigenetically regulated
Since in vitro conditions show a differential expression of AtqKNOX1 and AtqKNOX2 in A. fourcroydes and A. angustifolia clones (Figure 5), we examined the molecular events accompanying the epigenetically induced modification of these genes by Chromatin ImmunoPrecipitation (ChIP) in two clones, one of A. fourcroydes (P20) and one of A. angustifolia (BM26) from T0, M, B, SM and SB (Figure 6). Normalization of histone H3 immunoprecipitated from all samples to the corresponding input fraction revealed changes in H3K4me3 and H3K9me2. We observed an enrichment of the H3K4me3 mark in P20 at the B condition in comparison to the input of the AtqKNOX1 gene (Figure 6), which correlated with its expression (Figure 5).
Figure 6.
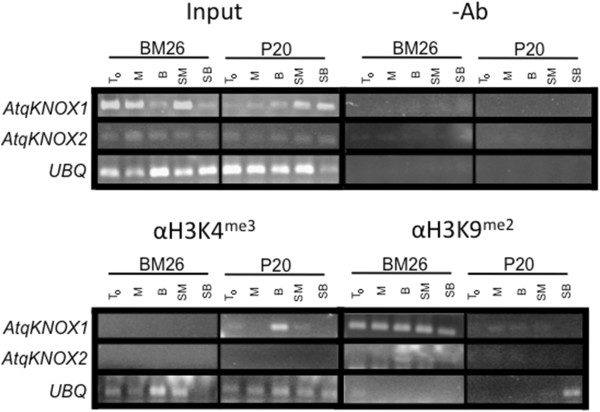
Effects of in vitro conditions upon the histone H3-methylation patterns of Agave fourcroydes clone P20 and A. angustifolia clone BM26 using Chromatin ImmunoPrecipitation (ChIP). Samples were collected from plants cultured in multiplication medium (T0), semisolid growth medium in Magenta boxes (M), temporary immersion in modular bioreactors (B), soil previously cultured in M (SM) and soil previously cultured in B (SB). The plant samples were examined for the Histone H3-tail methylation patterns the AtqKNOX1 and AtqKNOX2 genes. Input (input DNA), tenfold-diluted samples were used as templates for the input lanes. Negative controls (−Ab) with no antibody samples were treated in the same way as immunoprecipitated chromatins with H3K4me3 and H3K9me2. Amplified UBIQUITIN11 with specific primers was used as a control for the quality of the samples.
The H3K4me3 methylation levels in AtqKNOX1 in BM26 were not detected in any condition, which also correlates with its lack of expression found in Figure 5. During in vitro conditions in the Bioreactor, the H3K4me3 levels increased, favoring the expression due to epigenetic regulation of AtqKNOX1 in the clone P20. Considering these results and the ones obtained with gene expression studies, it appears that the AtqKNOX1 expression (Figure 5) is directly correlated with the H3K4me3 levels. Furthermore, we determined the levels of H3K9me2, a mark related to heterochromatin and repressive gene regions, of all analyzed genes (Figure 6). We found that only in BM26 was there an increase in H3K9me2 in AtqKNOX1 in all culture conditions. This result is consistent with the lack of expression of this gene in BM26.
These results could help to develop new strategies to optimize the use of more efficient in vitro conditions in order to guarantee the epigenetic stability of the cultures in the field.
Discussion
Plant tissue culture has been used for many years to propagate elite plants and for genetic breeding [52,53]. In Mexico, clonal propagation has been successfully employed to improve revigorization and juvenility in commercial plantations of Agave [16,50,54,55]. It is known that Agave plants cultivated under in vitro conditions for several generations do not contribute to genetic variation among clones [15], but some phenotypic variations have still been found. One of the explanations for these phenotypic changes under in vitro culture could be epigenetic regulation.
There is small but increasing evidence describing the epigenetic changes during in vitro culture. It has been found that not only the environment during the in vitro culture can change the epigenetic profile of the plant [27-29], but also the epigenetic status of the donor plants; even the organs within the donor plants can determine the later behavior of the explants [26]. For instance, it has been found that in vitro conditions change DNA methylation [27-29] and even this epigenetic mechanism has been related to plant development and rejuvenation [4,5]. Therefore, the DNA hypomethylation found in BM26 when this clone was changed from T0 to either the Magenta box or the Bioreactor (Figure 3) could be due to stress [9], which might be related to the increase in the mortality rate observed when these plants were transplanted to soil. Another explanation is that rejuvenation is occurring in this clone during its time in the Magenta boxes or the Bioreactor. Valledor et al.[5] found that there is a relationship between DNA methylation and aging-revigoration in plants, such that aging increases as DNA global methylation increases. In plants, DNA methylation usually increases with aging [26,56], while in mammals it decreases with time [57,58]. Therefore, the decrease in DNA methylation observed in A. angustifolia during in vitro culture (Figure 3) could be a mechanism for rejuvenation.
Li et al.[28] reported that several physiological changes related to in vitro culture, such as leaf structural changes, modifications in plant water content and changes in photosynthetic systems, are related to the stress provoked by in vitro conditions, and the stress seems to be related to the content of global DNA methylation. We observed in Agave that the semisolid system in the Magenta boxes generates longer leaves in comparison with the plants cultured in the temporary immersion of the modular Bioreactors (Figure 2). Although we did not observe a significant difference in DNA methylation between Magenta boxes and Bioreactors in the same clone (Figure 3), we observed a difference in histone methylation patterns between plants grown in these two in vitro systems (Figure 4). The genetic expression provoked by stress in plants depends on histone postranslational modifications and DNA methylation [59]. In the case of histone methylation, there are no reports that explain or suggest either the somaclonal variation or the genes affected epigenetically. Although there is information about the role of DNA methylation during in vitro culture, the histone modifications and the changes in chromatin modulation are still unknown. We found that clones genetically and even phenotypically alike have different epigenetic responses to in vitro culture (Figure 4). Moreover, there are no reports of the epigenetic stability of the micropropagated plants once they are ex vitro; so far, it is unknown whether histone modifications are involved. It is known that the epigenetics of an organism can change depending on development [60], biotic [7] or abiotic interactions [61], and even stress exposure [62]. Therefore, the mechanism of stress response due to the exposure to growth regulators during in vitro conditions could be one of the candidates for regulation by epigenetic factors. We found that during in vitro conditions, epigenetic modifications in histones (Figure 4), mainly through H3K9me2, which is very important in the initiation and maintenance of heterochromatin silencing [2] and in the control of DNA methylation [63], are affected. However, once the plants were transferred to ex vitro conditions in the field, this histone mark was absent or in present in low amounts, suggesting that plants can change the epigenome-phenotype.
In other in vitro systems, such as the potato, the DNA methylation variation associated with tissue culture protocols has been investigated [64]. It was found that DNA methylation changes occurring among the tissue types are an essential factor contributing to developmental stage differences, as well as tissue-culture-induced variation. Therefore, the hypermethylation found in P159 at SM could be induced by elements of the tissue culture media such as plant hormones, which have been shown to induce methylation changes in plant tissue cultures [12,19,65]. There is evidence that the use of the auxin 2,4-D in maize cultures generates changes in the DNA methylation pattern, depending on the concentration [12]. Plant hormones regulate growth and development in plants by controlling the expression of genes involved in these processes.
KNOX genes have been implicated in plant hormone metabolism [66,67]. Hay et al.[42] found that auxins repress the KNAT1 gene, promoting leaf development in Arabidopsis. Furthermore, it has been proposed that alterations in auxin gradients could result in a failure to down-regulate KNOX expression [37]. Different epigenetic mechanisms have been suggested for the regulation of KNOX genes during organogenesis [68,69]. In this study, we showed that the AtqKNOX1 gene is epigenetically regulated by H3K4me3 and H3K9me2 (Figure 6). Histone modification is a very complex epigenetic mechanism that so far has not been decoded [70-72]. However, studies in Arabidopsis have revealed that histone H3K9 methylation exists predominately as mono- and di-methylation, while trimethylation in H3K9 is quite rare [73]. There is evidence showing that in plants H3K27me3, H3K9me3 and H3K4me2 are euchromatic marks, while H3K9me2 is more associated with the repression of the transcription [63,74]. Chromatin changes have become an important key element for development in plants [75] and histone modification is essential [76]. Changes in KNOX1 gene expression among species could be due to different factors such as diversification of repressors of these genes [67]. Among the main roles of KNOX are the formation of auxin maxima, which provide feedback to repress KNOX expression, allowing leaflet outgrowth [67,77].
It will be interesting to study the methylation patterns from different generations exposed to in vitro conditions compared to those that were not, to determine whether the plants remember the in vitro exposure through epigenetic marks.
Conclusions
DNA methylation and histone modifications are very important epigenetic mechanisms that can be affected by in vitro conditions. Our studies indicate that under in vitro conditions, DNA methylation is affected in A. angustifolia, but not in A. fourcroydes. In addition, A. fourcroydes presented differential expression of AtqKNOX1 and AtqKNOX2, depending on the in vitro system used. Furthermore, the regulatory expression of AtqKNOX1 was related to the H3K4me3 and H3K9me2 marks. We propose that in vitro conditions change key genes by epigenetic regulation, which could be an important tool to find plants better adapted to overcome climate challenges.
Methods
Plant material and growth conditions
Three different in vitro-propagated Agave fourcroydes clones (P20, P21 and P159) and one Agave angustifolia clone (BM26) were used. The media used for plant induction, multiplication and growth of the plants was Murashige and Skoog [78], at pH 5.7, with some modifications as reported by Robert et al. [16,55]. Briefly, the plants from each clone were kept for six weeks in Magenta containers filled with 50mL of Murashige and Skoog media with reduced nitrogen, solidified with 1.75g/L of Gelrite (semisolid media) and without growth regulators. All plantlets were then transferred to and maintained in multiplication media supplemented with 10 mg/L BAP and 0.025 mg/L 2,4-D for ten weeks. Sixteen-week-old plants of the same size from each clone were divided as follows: 25 were sampled for analysis (T0) and 100 were cultured in growth medium supplemented with 1mg/L BAP and 0.025 mg/L 2,4-D. At this growing stage, two different systems were used: 50 plantlets were maintained in semisolid growth media in Magenta boxes [Magenta (M)] supplemented with 10 g/L of Agar, and 50 plantlets were cultured in liquid growth medium under temporary immersion in modular Bioreactors [Bioreactor (B)], as described by Robert et al.[50]. After five weeks, 25 plantlets from both in vitro systems (M and B) and from each clone (P20, P21, P159 and BM26) were sampled, and the remaining 25 from M and B were transferred to soil (SM and SB), where they grew for another eight weeks before they were also evaluated (Figure 1B).
Histone isolation and Western blots
Histones from Agave spp. clones (P20, P21, P159 and BM26) were isolated from 0.5 grams of leaf tissue from T0, M, B, SM and SB using sulfuric acid extraction of nuclei proteins followed by acetone precipitation, according to Jackson et al.[79]. Ten micrograms of isolated histones per sample were used for Western blots. The proteins were transferred to nitrocellulose membrane (0.45μm) by electrophoresis for four hours at 265mA. Membranes were blocked with 5% milk and 0.5% Tween in PBS, and probed with various antibodies, as follows: dimethyl-Histone H3 [Lys-4] (Upstate, cat. #07–030), trimethyl-Histone H3 [Lys-4] (Upstate, cat. #04–745), dimethyl-Histone H3 [Lys-9], (Upstate, cat. #07–441) and anti-dimethyl-Histone H3 [Lys-36] (Upstate, cat. #07–274). Di-(m2/H3) and tri-(m3/H3) methylated levels were measured and compared in histones isolated from different samples. The amount of loaded histone H3 in each sample was determined from Western blots using antibodies specific to non-methylated H3 (Upstate, cat. #06-755). Signals from bands obtained with methylation-specific antibodies were normalized against the respective histone H3 amounts (measured as signal intensities of Western-blot bands obtained with anti-histone H3-antibodies). All blots were stripped and reprobed with the histone H3 antibody to demonstrate equal loading. Data from four independent measurements consistently gave the same results.
DNA methylation
DNA extraction was done following the method described by Echevarria-Machado et al.[80]. DNA digestion was performed as described Santoyo et al.[81], with slight modifications. Five μg of DNA from P20, P21, P159 and BM26 at T0, M, B, SM and SB were dissolved in 42μL of ultra pure water and mixed with 5μL of 10 X DNA digestion buffer (200 mM acetic acid, 200mM glycine, 50mM magnesium chloride, 5mM zinc acetate, 2 mM calcium chloride adjusted with sodium hydroxide to pH 5.3). The mixture was hydrolyzed with 2μL of DNase I (D2821-Sigma, 10U/μL) and 1μL of Nuclease P1 (N8630-Sigma, 1.25U/μL) overnight at 37°C and then frozen for 10–15 min at 0°C and then incubated at 100°C for five min. Samples were mixed with 5μL of 100 mM NaOH and 2μL Calf intestine alkaline phosphatase (P4879-Sigma, 1U/μL) and incubated for 3.5 h at 37°C and then mixed with 100μl of water and 50μl mobile phase D (see below). Samples were centrifuged at 18,000 × g for 10 min at 4°C, and the supernatant was transferred to a new tube and stored at −20°C until analysis. Forty µl of sample was injected to liquid chromatographic system (HPLC, Agilent series 1200), and the bases were separated on a chromatographic column, Luna C18 (250 × 4.6mm, 5μm from Phenomenex) at 40°C. The absorbance was measured using a diode array detector at 286 nm. The separation was realized according to the method described by Lopez Torres et al.[82] with some modifications. Four mobile phases were used: A, deionized water; B, acetonitrile; C, methanol; and D, 50mM ammonium phosphate dibasic, 15mM ammonium acetate adjusted with phosphoric acid to pH 4.1. The gradient program was as follows: 0 to 4 min 80% A, 20% D; 4 to 11 min 78% A, 2% C, 20% D; 11 to 15 min 77% A, 3% C, 20% D; 15 to 15.8 min 35% A, 20% B, 25% C, 20% D; 15.8 to 16 min 30% A, 25% B, 25% C, 20% D at a total flow rate of 1 mL/min. The percentage of global DNA methylation was calculated as follows: concentration of 5-methyl-2’-deoxycytosine (5mdC)/ [concentration of 5mdC + concentration of 2’-deoxycytosine (dC)] × 100. All the analysis was achieved with three biological replicates from different DNA extractions. Statistical comparison was performed by one-way analysis of variance (ANOVA). The significance grade was determined by the test of several means of Tukey (P ≤ 0.01).
Gene expression
Total RNA was extracted from 0.2g leaf tissue of P20, P21, P159 and BM26 from T0, M, B, SM and SB by using the BRL Trizol reagent (Invitrogen) and re-purified with the Qiagen RNeasy Mini Kit, following the manufacturer’s instructions. Reverse transcriptase (RT) reactions were performed in a 20-μl volume containing 2μg of total RNA and 200 units of the M-MLV Reverse Transcriptase (Invitrogen), following the manufacturer’s instructions. cDNA templates for qRT-PCR amplification were prepared from three individual plants for each condition. Each reaction contained 100 ng of cDNA template, 10 pM of each primer and 1× EXPRESS SYBR® GreenERTM pPCR SuperMix Universal (11784-200-Invitrogen). Real-time PCR assays were performed in a Step OneTM Real Time PCR System (Applied Biosystems) under the following conditions: 5 min at 95°C, followed by 35 cycles of 95°C for 40 sec, 62°C for 40 sec and 72°C for 90 sec, and a final cycle of 72°C for 5 min. Transcript levels of AtqKNOX1 and AtqKNOX2 in the samples were normalized to the level of UBIQUITIN (UBQ11) and the data are expressed as the relative expression level. The specificity of the PCR product amplifications was determined by a melting curve analysis. Data obtained from Real-time PCR were used to calculate the relative quantification of the target gene expression and compared to the expression of the UBQ11 using the 2-∆∆ct method [83]. We used the primers reported by Abraham-Juarez et al.[44] to determine gene expression in Agave: AtqKNOX1 (GenBank Accession No. GU980050) forward 5’-gagggcagttcataggtgat -3’, reverse 5’-ttcccacaggagtaggtctc -3’ (190bp); AtqKNOX2 (GenBank Accession No. GU980051) forward 5’- gaatggtggactgctcacta-3’, reverse 5’-cctcagtcgtcgtcatagaa-3’ (225bp) (Additional file 1: Figure S1); and UBQ11 was used as a control 5’-gacgggcgcacccttgcggatta-3’, 5’-tcctggatcttcgccttgacatt-3’ (211bp). Statistical comparison was performed by one-way analysis of variance (ANOVA).
Chromatin immunoprecipitation (ChIP) assay
ChIP assays were performed as described by De-la-Peña et al.[7]. The antibodies used were anti-dimethyl Histone H3 [Lys9] (Upstate #07-441) and anti-trimethyl Histone H3 [Lys4] (Upstate #05-745). For all ChIP experiments, chromatin was isolated from leaves of P20 and BM26 from T0, M, B, SM and SB conditions. Each immunoprecipitation experiment was independently performed three times with separately isolated biological samples. All PCR reactions were done in 25μl: 5min at 95°C, followed by 38 cycles of 95°C 30 sec, 56°C 30 sec, 72°C 2 min, and 72°C 5 min. Intensities were normalized versus the input sample representing 15% of the DNA used as template. The ChIP primer sequences used were as follows: AtqKNOX1 forward 5’-gagggcagttcataggtgat-3’, reverse 5’-ttcccacaggagtaggtctc -3’; AtqKNOX2 forward 5’- gaatggtggactgctcacta-3’, reverse 5’-cctcagtcgtcgtcatagaa-3’; and UBQ11 was used as a control 5’-gacgggcgcacccttgcggatta-3’, 5’-tcctggatcttcgccttgacatt-3’.
Abbreviations
B: Bioreactor; ChIP: Chromatin immuno precipitation; dC: 2’-deoxycytosine; 2,4-D: 2,4-Dichlorophenoxyacetic acid; HPLC: High-performance liquid chromatography; KNOX: KNOTTED1-like HOMEOBOX; K: Lysine; M: Magenta; me: Methylation; 5mdC: 5-methyl-deoxycitidine; RT-PCR: Reverse transcription polymerase chain reaction; UBQ11: UBIQUITIN11.
Competing interests
The authors declare that they have no competing interests.
Authors’ contributions
GN carried out the biochemical and molecular studies. GO and JH carried out the induction and propagations of the Agave cultures. MR participated in the design of the study and drafted the manuscript. KW and AL carried out the DNA methylation. CD conceived of the study, participated in its design and coordination, carried out the epigenetic studies and drafted the manuscript. All authors read and approved the final manuscript.
Supplementary Material
Figure S1. A) Comparison of the AqtKNOX1 nucleotide sequences with KNAT1 (At4g08150) Arabidopsis sequences; B) Comparison of the AtqKNOX2 nucleotide sequences with KNAT2 (At1g23389) Arabidopsis sequences. Alignment was performed using Blast [84]. The * indicates the conserved residues between the Agave with Arabidopsis. The : indicates that at least one residue is different between Agave and Arabidopsis. Names of the genes are indicated on the left. The squares indicate the primers that were used for RT-PCR and ChIP.
Contributor Information
Clelia De-la-Peña, Email: clelia@cicy.mx.
Geovanny Nic-Can, Email: nic_can82@hotmail.com.
Gabriel Ojeda, Email: g_220178@hotmail.com.
José L Herrera-Herrera, Email: herrera@cicy.mx.
Adolfo López-Torres, Email: adolfo_lopez_torres@hotmail.com.
Kazimierz Wrobel, Email: szlembez1@yahoo.com.
Manuel L Robert-Díaz, Email: robert@cicy.mx.
Acknowledgements
These studies were supported by the Consejo Nacional de Ciencia y Tecnología (Grants No. 121768 and CB-2012/178149 to CD).
References
- Salozhin SV, Prokhorchuk EB, Georgiev GP. Methylation of DNA- One of the major epigenetic markers. Biochemistry. 2005;70:525–532. doi: 10.1007/s10541-005-0146-8. [DOI] [PubMed] [Google Scholar]
- Shilatifard A. Chromatin modifications by methylation and ubiquitination: implications in the regulation of gene expression. Annu Rev Biochem. 2006;75:243–269. doi: 10.1146/annurev.biochem.75.103004.142422. [DOI] [PubMed] [Google Scholar]
- Vanyushin B. DNA methylation and epigenetics. Rus J Genet. 2006;42:985–997. doi: 10.1134/S1022795406090055. [DOI] [PubMed] [Google Scholar]
- Ruiz-Garcia L, Cervera MT, Martinez-Zapater JM. DNA methylation increases throughout Arabidopsis development. Planta. 2005;222:301–306. doi: 10.1007/s00425-005-1524-6. [DOI] [PubMed] [Google Scholar]
- Valledor L, Hasbún R, Meijón M, Rodríguez J, Santamaría E, Viejo M, Berdasco M, Feito I, Fraga M, Cañal MJ. et al. Involvement of DNA methylation in tree development and micropropagation. Plant Cell Tiss Organ Cult. 2007;91:75–86. doi: 10.1007/s11240-007-9262-z. [DOI] [Google Scholar]
- Dennis ES, Peacock WJ. Epigenetic regulation of flowering. Curr Opin Plant Biol. 2007;10:520–527. doi: 10.1016/j.pbi.2007.06.009. [DOI] [PubMed] [Google Scholar]
- De-la-Peña C, Rangel-Cano A, Alvarez-Venegas R. Regulation of disease-responsive genes mediated by epigenetic factors: interaction of Arabidopsis–Pseudomonas. Mol Plant Pathol. 2012;13:388–398. doi: 10.1111/j.1364-3703.2011.00757.x. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Ay N, Irmler K, Fisher A, Uhlemann R, Reuter G, Humbeck K. Epigenetic programming via histone methylation at WRKY53 controls leaf senescence in Arabidopsis thaliana. Plant J. 2009;58:333–346. doi: 10.1111/j.0960-7412.2009.03782.x. [DOI] [PubMed] [Google Scholar]
- Baranek M, Krizan B, Ondrusikova E, Pidra M. DNA-methylation changes in grapevine somaclones following in vitro culture and thermotherapy. Plant Cell Tiss Organ Cult. 2010;101:11–22. doi: 10.1007/s11240-009-9656-1. [DOI] [Google Scholar]
- Jaligot E, Rival A, Beulé T, Dussert S, Verdeil JL. Somaclonal variation in oil palm (Elaeis guineensis Jacq.): the DNA methylation hypothesis. Plant Cell Rep. 2000;19:684–690. doi: 10.1007/s002999900177. [DOI] [PubMed] [Google Scholar]
- Miguel C, Marum L. An epigenetic view of plant cells cultured in vitro: somaclonal variation and beyond. J Exp Bot. 2011;62:3713–3725. doi: 10.1093/jxb/err155. [DOI] [PubMed] [Google Scholar]
- LoSchiavo F, Pitto L, Giuliano G, Torti G, Nuti-Ronchi V, Marazziti D, Vergara R, Orselli S, Terzi M. DNA methylation of embryogenic carrot cell cultures and its variations as caused by mutation, differentiation, hormones and hypomethylating drugs. Theor Appl Genet. 1989;77:325–331. doi: 10.1007/BF00305823. [DOI] [PubMed] [Google Scholar]
- Vergara R, Verde F, Pitto L, LoSchiavo F, Terzi M. Reversible variations in the methylation pattern of carrot DNA during somatic embryogenesis. Plant Cell Rep. 1990;8:697–700. doi: 10.1007/BF00272097. [DOI] [PubMed] [Google Scholar]
- Trap-Gentil M-V, Hébrard C, Lafon-Placette C, Delaunay A, Hagége D, Joseph C, Brignolas F, Lefebvre M, Barnes S, Maury S. Time course and amplitude of DNA methylation in the shoot apical meristem are critical points for bolting induction in sugar beet and bolting tolerance between genotypes. J Exp Bot. 2011;62:2585–2597. doi: 10.1093/jxb/erq433. [DOI] [PubMed] [Google Scholar]
- Díaz-Martínez M, Nava-Cedillo A, Guzmán-López JA, Escobar-Guzmán R, Simpson J. Polymorphism and methylation patterns in Agave tequilana Weber var. ‘Azul’ plants propagated asexually by three different methods. Plant Sci. 2012;185-186:321–330. doi: 10.1016/j.plantsci.2012.01.004. [DOI] [PubMed] [Google Scholar]
- Robert ML, Herrera JL, Chan JL, Contreras F. In: Biotechnology in Agriculture and Forestry. Bajaj JPY, editor. Berlin: Springer- Verlag; 1992. Micropropagation of Agave spp; pp. 306–329. [Google Scholar]
- Quiroz-Figueroa F, Rojas-Herrera R, Galaz-Avalos R, Loyola-Vargas V. Embryo production through somatic embryogenesis can be used to study cell differentiation in plants. Plant Cell Tiss Organ Cult. 2006;86:285–301. doi: 10.1007/s11240-006-9139-6. [DOI] [Google Scholar]
- Park S, Murthy H, Chakrabarthy D, Paek K. Detection of epigenetic variation in tissue-culture-derived plants of Doritaenopsis by methylation-sensitive amplification polymorphism (MSAP) analysis. In Vitro Cell Dev Biol - Plant. 2009;45:104–108. doi: 10.1007/s11627-008-9166-6. [DOI] [Google Scholar]
- Peraza-Echeverria S, Herrera-Valencia VA, James-Kay A. Detection of DNA methylation changes in micropropagated banana plants using methylation-sensitive amplification polymorphism (MSAP) Plant Sci. 2001;161:359–367. doi: 10.1016/S0168-9452(01)00421-6. [DOI] [PubMed] [Google Scholar]
- Williams L, Zhao J, Morozova N, Li Y, Avivi Y, Grafi G. Chromatin reorganization accompanying cellular dedifferentiation is associated with modifications of histone H3, redistribution of HP1, and activation of E2F-target genes. Dev Dynam. 2003;228:113–120. doi: 10.1002/dvdy.10348. [DOI] [PubMed] [Google Scholar]
- Alatzas A, Foundouli A. Distribution of ubiquitinated histone H2A during plant cell differentiation in maize root and dedifferentiation in callus culture. Plant Sci. 2006;171:481–487. doi: 10.1016/j.plantsci.2006.05.008. [DOI] [PubMed] [Google Scholar]
- Berdasco M, Alcázar R, García-Ortiz MV, Ballestar E, Fernández AF, Roldán-Arjona T, Tiburcio AF, Altabella T, Buisine N, Quesneville H. et al. Promoter DNA hypermethylation and gene repression in undifferentiated Arabidopsis cells. PLoS One. 2008;3:e3306. doi: 10.1371/journal.pone.0003306. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Law RD, Suttle JC. Chromatin remodeling in plant cell culture: patterns of DNA methylation and histone H3 and H4 acetylation vary during growth of asynchronous potato cell suspensions. Plant Physiol Bioch. 2005;43:527–534. doi: 10.1016/j.plaphy.2005.03.014. [DOI] [PubMed] [Google Scholar]
- Tanurdzic M, Vaughn MW, Jiang H, Lee TJ, Slotkin RK, Sosinski B, Thompson WF, Doerge RW, Martienssen RA. Epigenomic consequences of immortalized plant cell suspension culture. PLoS Biol. 2008;6:2880–2895. doi: 10.1371/journal.pbio.0060302. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Smykal P, Valledor L, Rodriguez R, Griga M. Assessment of genetic and epigenetic stability in long-term in vitro shoot culture of pea (Pisum sativum L.) Plant Cell Rep. 2007;26:1985–1998. doi: 10.1007/s00299-007-0413-9. [DOI] [PubMed] [Google Scholar]
- Valledor L, Meijón M, Hasbún R, Cañal MJ, Rodríguez R. Variations in DNA methylation, acetylated histone H4, and methylated histone H3 during Pinus radiata needle maturation in relation to the loss of in vitro organogenic capability. J Plant Physiol. 2010;167:351–357. doi: 10.1016/j.jplph.2009.09.018. [DOI] [PubMed] [Google Scholar]
- Kaeppler SM, Phillips RL. Tissue culture-induced DNA methylation variation in maize. Proc Natl Acad Sci USA. 1993;90:8773–8776. doi: 10.1073/pnas.90.19.8773. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Li X, Xu M, Korban SS. DNA methylation profiles differ between field and in vitro-grown leaves of apple. J Plant Physiol. 2002;159:1229–1234. doi: 10.1078/0176-1617-00899. [DOI] [Google Scholar]
- Phillips RL, Kaeppler SM, Peschke VM. In: Progress in plant cellular and molecular biology. Nijkamp HJJ, VanDerPlas LHW, Van Aartrijk J, editor. Dordrecht: Klumer Academic Publishing; 1990. Do we understand somaclonal variation? pp. 131–141. [Google Scholar]
- Smulders MJM, Kortekass WR, Vosman B. Tissue culture-induced DNA methylation polymorphism in repetitive DNA of tomato calli and regenerated plants. Theor Appl Genet. 1995;91:1257–1264. doi: 10.1007/BF00220938. [DOI] [PubMed] [Google Scholar]
- Smulders M, de Klerk G. Epigenetics in plant tissue culture. Plant Growth Regul. 2011;63:137–146. doi: 10.1007/s10725-010-9531-4. [DOI] [Google Scholar]
- Peredo EL, Arroyo-Garcia R, Reed BM, Revilla MA. Genetic and epigenetic stability of cryopreserved and cold-stored hops (Humulus lupulus L.) Cryobiology. 2008;57:234–241. doi: 10.1016/j.cryobiol.2008.09.002. [DOI] [PubMed] [Google Scholar]
- Peredo EL, Arroyo-Garcia R, Revilla MA. Epigenetic changes detected in micropropagated hop plants. J Plant Physiol. 2009;166:1101–1111. doi: 10.1016/j.jplph.2008.12.015. [DOI] [PubMed] [Google Scholar]
- Varga A, Thoma LH, Bruinsma J. Effects of auxins and cytokinins on epigenetic instability of callus-propagated Kalanchoe blossfeldiana Poelln. Plant Cell Tiss Organ Cult. 1988;15:223–231. doi: 10.1007/BF00033646. [DOI] [Google Scholar]
- Morcillo F, Gagneur C, Adam H, Richaud F, Singh R, Cheah SC, Rival A, Duval Y, Tregear JW. Somaclonal variation in micropropagated oil palm. Characterization of two novel genes with enhanced expression in epigenetically abnormal cell lines and in response to auxin. Tree Physiol. 2006;26:585–594. doi: 10.1093/treephys/26.5.585. [DOI] [PubMed] [Google Scholar]
- Frugis G, Giannino D, Mele G, Nicolodi C, Innocenti AM, Chiappetta A, Bitonti MB, Dewitte W, Van Onckelen H, Mariotti D. Are homeobox Knotted-like genes and cytokinins the leaf architects? Plant Physiol. 1999;119:371–374. doi: 10.1104/pp.119.2.371. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Hay A, Craft J, Tsiantis M. Plant hormones and homeoboxes: bridging the gap? BioEssays. 2004;26(4):395–404. doi: 10.1002/bies.20016. [DOI] [PubMed] [Google Scholar]
- Jasinski S, Piazza P, Craft J, Hay A, Woolley L, Rieu I, Phillips A, Hedden P, Tsiantis M. KNOX action in Arabidopsis is mediated by coordinate regulation of cytokinin and gibberellin activities. Curr Biol. 2005;15:1560–1565. doi: 10.1016/j.cub.2005.07.023. [DOI] [PubMed] [Google Scholar]
- Montero-Córtes M, Sáenz L, Córdova I, Quiroz A, Verdeil JL, Oropeza C. GA3 stimulates the formation and germination of somatic embryos and the expression of a KNOTTED-like homeobox gene of Cocos nucifera (L.) Plant Cell Rep. 2010;29:1049–1059. doi: 10.1007/s00299-010-0890-0. [DOI] [PubMed] [Google Scholar]
- Hake S, Smith HMS, Holtan H, Magnani E, Mele G, Ramirez J. The role of KNOX genes in plant development. Annu Rev Cell Dev Biol. 2004;20:125–151. doi: 10.1146/annurev.cellbio.20.031803.093824. [DOI] [PubMed] [Google Scholar]
- Rupp H-M, Frank M, Werner T, Strnad M, Schmülling T. Increased steady state mRNA levels of the STM and KNAT1 homeobox genes in cytokinin overproducing Arabidopsis thaliana indicate a role for cytokinins in the shoot apical meristem. Plant J. 1999;18:557–563. doi: 10.1046/j.1365-313X.1999.00472.x. [DOI] [PubMed] [Google Scholar]
- Hay A, Barkoulas M, Tsiantis M. ASYMMETRIC LEAVES1 and auxin activities converge to repress BREVIPEDICELLUS expression and promote leaf development in Arabidopsis. Development. 2006;133:3955–3961. doi: 10.1242/dev.02545. [DOI] [PubMed] [Google Scholar]
- Dean G, Casson S, Lindsey K. KNAT6 gene of Arabidopsis is expressed in roots and is required for correct lateral root formation. Plant Mol Biol. 2004;54:71–84. doi: 10.1023/B:PLAN.0000028772.22892.2d. [DOI] [PubMed] [Google Scholar]
- Abraham-Juarez MJ, Martinez-Hernandez A, Leyva-Gonzalez MA, Herrera-Estrella L, Simpson J. Class I KNOX genes are associated with organogenesis during bulbil formation inAgave tequilanaJ. Exp Bot. 2010;61:4055–4067. doi: 10.1093/jxb/erq215. [DOI] [PubMed] [Google Scholar]
- Ori N, Eshed Y, Chuck G, Bowman JL, Hake S. Mechanisms that control KNOX gene expression in the Arabidopsis shoot. Development. 2000;127:5523–5532. doi: 10.1242/dev.127.24.5523. [DOI] [PubMed] [Google Scholar]
- Greene B, Walko R, Hake S. Mutator insertions in an intron of the Maize knotted1 gene result in dominant suppressible mutations. Genetics. 1994;138:1275–1285. doi: 10.1093/genetics/138.4.1275. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Bae NS, Swanson MJ, Vassilev A, Howard BH. Human histone deacetylase SIRT2 interacts with the homeobox transcription factor HOXA10. J Biochem. 2004;135:695–700. doi: 10.1093/jb/mvh084. [DOI] [PubMed] [Google Scholar]
- Lira-Medeiros CF, Parisod C, Fernandes RA, Mata CS, Cardoso MA, Gomes Ferreira PC. Epigenetic variation in mangrove plants occurring in contrasting natural environment. PLoS One. 2010;5:e10326. doi: 10.1371/journal.pone.0010326. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Vaillant I, Paszkowski J. Role of histone and DNA methylation in gene regulation. Curr Opin Plant Biol. 2007;10:528–533. doi: 10.1016/j.pbi.2007.06.008. [DOI] [PubMed] [Google Scholar]
- Robert ML, Herrera-Herrera JL, Herrera-Herrera G, Herrera-Alamillo MA, Fuentes-Carrillo P. In: Plant Cell Culture Protocols. 2. Loyola-Vargas VM, Vázquez-Flota F, editor. New Jersey: Humana Press; 2006. A new temporary immersion bioreactor system for micropropagation; pp. 121–129. [DOI] [PubMed] [Google Scholar]
- Kouzarides T. Chromatin modifications and their function. Cell. 2007;128:693–705. doi: 10.1016/j.cell.2007.02.005. [DOI] [PubMed] [Google Scholar]
- Xiao Y, Niu G, Kozai T. Development and application of photoautotrophic micropropagation plant system. Plant Cell Tiss Organ Cult. 2011;105:149–158. doi: 10.1007/s11240-010-9863-9. [DOI] [Google Scholar]
- Bonga J, Klimaszewska K, Von Aderkas P. Recalcitrance in clonal propagation, in particular of conifers. Plant Cell Tiss Organ Cult. 2010;100:241–254. doi: 10.1007/s11240-009-9647-2. [DOI] [Google Scholar]
- Robert ML, Herrera-Herrera JL, Herrera-Alamillo MA, Quijano A, Balám U. Manual for the in vitro culture of Agaves. United Nations Industrial Development Organization. Vienna: Common Fund for Commodities; 2004. Technical paper. [Google Scholar]
- Robert ML, Herrera JL, Contreras F, Scorer KN. In vitro propagation of Agave fourcroydes Lem. (Henequen) Plant Cell Tiss Organ Cult. 1987;8:37–48. doi: 10.1007/BF00040731. [DOI] [Google Scholar]
- Fraga M, Rodriguez R, Ca§al MJ. Genomic DNA methylation-demethylation during ageing-reinvigoration of Pinus radiata. Tree Physiol. 2002;22:813–816. doi: 10.1093/treephys/22.11.813. [DOI] [PubMed] [Google Scholar]
- Gonzalo S. Epigenetic alterations in aging. J Appl Physiol. 2010;109:586–597. doi: 10.1152/japplphysiol.00238.2010. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Wilson VL, Smith RA, Ma S, Cutler RG. Genomic 5-methyldeoxycytidine decreases with age. J Biol Chem. 1987;262:9948–9951. [PubMed] [Google Scholar]
- Chinnusamy V, Zhu JK. Epigenetic regulation of stress responses in plants. Curr Opin Plant Biol. 2009;12:133–139. doi: 10.1016/j.pbi.2008.12.006. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Kaufmann K, Pajoro A, Angenent GC. Regulation of transcription in plants: mechanisms controlling developmental switches. Nat Rev Genet. 2010;11:830–842. doi: 10.1038/nrg2885. [DOI] [PubMed] [Google Scholar]
- Kim JM, To TK, Nishioka T, Seki M. Chromatin regulation functions in plant abiotic stress responses. Plant Cell Environ. 2010;33:604–611. doi: 10.1111/j.1365-3040.2009.02076.x. [DOI] [PubMed] [Google Scholar]
- Gutzat R, Mittelsten Scheid O. Epigenetic responses to stress: triple defense? Curr Opin Plant Biol. 2012. In press. doi:dpi.org/10.1016/j.pbi.2012.08.007. [DOI] [PMC free article] [PubMed]
- Mathieu O, Probst AV, Paszkowski J. Distinct regulation of histone H3 methylation at lysines 27 and 9 by CpG methylation in Arabidopsis. EMBO J. 2005;24:2783–2791. doi: 10.1038/sj.emboj.7600743. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Joyce SM, Cassells AC. Variation in potato microplant morphology in vitro and DNA methylation. Plant Cell Tiss Organ Cult. 2002;70:125–137. doi: 10.1023/A:1016312303320. [DOI] [Google Scholar]
- Arnholdt-Schmitt B, Herterich S, Neumann KH. Physiological aspects of genome variability in tissue culture .1. Growth phase-dependent differential DNA methylation of the carrot genome (Daucus carota L.) during primary culture. Theor Appl Genet. 1995;91:809–815. doi: 10.1007/BF00220964. [DOI] [PubMed] [Google Scholar]
- Kusaba S, Kano-Murakami Y, Matsuoka M, Tamaoki M, Sakamoto T, Yamaguchi I, Fukumoto M. Alteration of hormone levels in transgenic tobacco plants overexpressing the rice homeobox gene OSH1. Plant Physiol. 1998;116:471–476. doi: 10.1104/pp.116.2.471. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Hay A, Tsiantis M. KNOX genes: versatile regulators of plant development and diversity. Development. 2010;137:3153–3165. doi: 10.1242/dev.030049. [DOI] [PubMed] [Google Scholar]
- Phelps-Durr TL, Thomas J, Vahab P, Timmermans MCP. Maize rough sheath2 and Its Arabidopsis orthologue ASYMMETRIC LEAVES1 interact with HIRA, a predicted histone chaperone, to maintain knox gene silencing and determinacy during organogenesis. Plant Cell. 2005;17:2886–2898. doi: 10.1105/tpc.105.035477. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Li H, He Z, Lu G, Lee SC, Alonso J, Ecker JR, Luan S. A WD40 domain cyclophilin interacts with histone H3 and functions in gene repression and organogenesis in Arabidopsis. Plant Cell. 2007;19:2403–2416. doi: 10.1105/tpc.107.053579. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Cosgrove MS. Writers and readers: deconvoluting the harmonic complexity of the histone code. Nat Struct Mol Biol. 2012;19:739–740. doi: 10.1038/nsmb.2350. [DOI] [PubMed] [Google Scholar]
- Strahl BD, Allis CD. The language of covalent histone modifications. Nature. 2000;403:41–45. doi: 10.1038/47412. [DOI] [PubMed] [Google Scholar]
- Jenuwein T, Allis CD. Translating the histone code. Science. 2001;293:1074–1080. doi: 10.1126/science.1063127. [DOI] [PubMed] [Google Scholar]
- Johnson L, Mollah S, Garcia BA, Muratore TL, Shabanowitz J, Hunt DF, Jacobsen SE. Mass spectrometry analysis of Arabidopsis histone H3 reveals distinct combinations of post-translational modifications. Nucleic Acids Res. 2004;32:6511–6518. doi: 10.1093/nar/gkh992. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Liu C, Lu F, Cui X, Cao X. Histone methylation in higher plants. Annu Rev Plant Biol. 2010;61:395–420. doi: 10.1146/annurev.arplant.043008.091939. [DOI] [PubMed] [Google Scholar]
- Berger F, Gaudin V. Chromatin dynamics and Arabidopsis development. Chromosome Res. 2003;11:277–304. doi: 10.1023/A:1022844127716. [DOI] [PubMed] [Google Scholar]
- Kouzarides T. Histone methylation in transcriptional control. Curr Opin Genet Dev. 2002;12:198–209. doi: 10.1016/S0959-437X(02)00287-3. [DOI] [PubMed] [Google Scholar]
- Barkoulas M, Hay A, Kougioumoutzi E, Tsiantis M. A developmental framework for dissected leaf formation in the Arabidopsis relative Cardamine hirsuta. Nat Genet. 2008;40:1136–1141. doi: 10.1038/ng.189. [DOI] [PubMed] [Google Scholar]
- Murashige T, Skoog F. A revised medium for rapid growth and bio assays with tobacco tissue cultures. Physiol Plant. 1962;15:473–497. doi: 10.1111/j.1399-3054.1962.tb08052.x. [DOI] [Google Scholar]
- Jackson JP, Jonhson L, Jasencakova Z, Zhang X, PerezBurgos L, Singh PB, Cheng X, Schubert I, Jenuwein T, Jacobsen SE. Dimethylation of histone H3 lysine 9 is a critical mark for DNA methylation and gene silencing in Arabidopsis thaliana. Chromosoma. 2004;112:308–315. doi: 10.1007/s00412-004-0275-7. [DOI] [PubMed] [Google Scholar]
- Echevarría-Machado I, Sánchez-Cach L, Hernández-Zepeda C, Rivera-Madrid R, Moreno-Valenzuela O. A simple and efficient method for isolation of DNA in high mucilaginous plant tissues. Mol Biotech. 2005;31:129–135. doi: 10.1385/MB:31:2:129. [DOI] [PubMed] [Google Scholar]
- Santoyo MM, Flores CR, Torres AL, Wrobel K. Global DNA methylation in earthworms: A candidate biomarker of epigenetic risks related to the presence of metals/metalloids in terrestrial environments. Environ Pollut. 2011;159:2387–2392. doi: 10.1016/j.envpol.2011.06.041. [DOI] [PubMed] [Google Scholar]
- Lopez-Torres A, Yanez Barrientos E, Wrobel K, Wrobel K. Selective derivatization of cytosine and methylcytosine moieties with 2-bromoacetophenone for submicrogram DNA methylation analysis by reversed phase HPLC with spectrofluorimetric detection. Anal Chem. 2011;83:7999–8005. doi: 10.1021/ac2020799. [DOI] [PubMed] [Google Scholar]
- Livak KJ, Schmittgen TD. Analysis of relative gene expression data using Real-time quantitative PCR and the 2 − ΔΔCT method. Methods. 2001;25:402–408. doi: 10.1006/meth.2001.1262. [DOI] [PubMed] [Google Scholar]
- Altschul SF, Madden TL, Schaffer AA, Zhang J, Zhang Z, Miller W, Lipman DJ. Gapped BLAST and PSI-BLAST: a new generation of protein database search programs. Nucleic Acids Res. 1997;25:3389–3402. doi: 10.1093/nar/25.17.3389. [DOI] [PMC free article] [PubMed] [Google Scholar]
Associated Data
This section collects any data citations, data availability statements, or supplementary materials included in this article.
Supplementary Materials
Figure S1. A) Comparison of the AqtKNOX1 nucleotide sequences with KNAT1 (At4g08150) Arabidopsis sequences; B) Comparison of the AtqKNOX2 nucleotide sequences with KNAT2 (At1g23389) Arabidopsis sequences. Alignment was performed using Blast [84]. The * indicates the conserved residues between the Agave with Arabidopsis. The : indicates that at least one residue is different between Agave and Arabidopsis. Names of the genes are indicated on the left. The squares indicate the primers that were used for RT-PCR and ChIP.