Abstract
Aim of this paper is to find out the relationship between antioxidant activity of Abutilon indicum Linn and their phytochemical composition especially phenols and flavonols. Successive extractions were carried out for the Abutilon indicum plant with petroleum ether, chloroform, ethyl acetate, n-butanol, ethanol and water. All these extracts were evaluated for their antioxidant activities. Their antioxidant activities were correlated with their total phenol and flavonol content present in the plant. Ethyl acetate showed maximum free radical scavenging activity. IC50 value for various antioxidant methods for all extract showed no significance with total antioxidant capacity except IC50 value of LPO (r2 = 0.7273). Correlation between total antioxidant capacity and total phenolic content was not significant with r2 = 0.2554, P<0.3065. Total antioxidant capacity and total flavonol content showed similar correlation with r2 = 0.2554, P<0.0962.
Keywords: Abutilon indicum linn, antioxidant, DPPH, nitric oxide, nydrogen peroxide, ABTS, deoxyribose, lipid peroxidation, total antioxidant capacity
Phenols and flavonols, are considered to be responsible for the various biological activities[1,2]. Abutilon indicum Linn belongs to the family Malvaceae. It is distributed throughout India and Sri Lanka. In hindi it is called as Atibalaa, in English it is called country mallow and in Tamil it is called thutti. This plant contains mucilage, tannins, asparagines, gallic acid and sesquiterpenes. The juice of the plant is applied as an emollient to relieve soreness of the nates in young children. Leaves are cooked and eaten for bleeding piles. A decoction of the leaves is used as mouth wash in toothache and tender gums. The flowers are eaten raw as an application of boils and ulcers. The powdered flowers are eaten in ghee as a remedy in blood vomiting and in cough. The decoction of root is a nerve tonic, and antipyretic. An infusion of the root is considered a good cooling remedy in fevers and is also given in strangury and heamaturia. The seeds infusion in water forms a cool drink, useful in treating piles gonorrhea, gleet and chronic cystitis. A decoction of seed is given in dysentery, fistula and eyesores. Other biological uses of plant as following anthelmintic, antiinflammatory, laxative, anticancer, hypothermic activity, diuretic, astringent, demulcent and antiulcer activity[3].
2,2-Diphenyl-2-picryl hydrazyl (DPPH), 2,2’-azino-bis (3-ethylbenzothizoline-6-sulfonic acid) diammonium salts (ABTS) were obtained from Sigma/Aldrich, Co., St. Louis, USA. Phosphate buffer saline (PBS) from Gibco-BRL. Rutin from Across Organics, New Jersey, USA. Napthyl ethylene diamine dihydrochloride (NEDD) from Roch-Light Ltd, Suffolk, UK. Ascorbic acid, nitro blue tetrazolium (NBT) and butylated hydroxyl anisole (BHA), calcium chloride from S. D. fine Chemicals Ltd., Mumbai, India. Sodium nitroprusside, dimethyl sulphoxide (DMSO), potassium chloride and sodium chloride from Ranbaxy laboratories, Ltd., Mohali, India. Sulphanilic acid, sodium bicarbonate from E-Merck (India) Ltd, Mumbai, India. Disodium hydrogen phosphate was from Hi-media Lab Pvt, Ltd, Mumbai. All chemicals used were of analytical grade.
DPPH method was carried out in a 96 well micro titre plate[4–6]. To 200 μl of DPPH solution, 10 μl of each of the extract or the standard solution was added separately in wells of the microtitre plate. The final concentration of the test and standard solutions used are 1000 to 1.95 μg/ml. The plates were incubated at 37° for 20 min and the absorbance of the each well was measured at 490 nm, using ELISA reader against the corresponding test and standard blanks and the remaining DPPH was calculated. IC50 (Inhibitory concentration) is the concentration of the sample required to scavenge 50% of DPPH free radicals. Percentage of inhibition = [(Control-sample)/Control]×100.
Nitric oxide radical inhibition activity was carried out by earlier reported method[4–6]. The reaction mixture (6 ml) containing SNP (10 mM, 4 ml), PBS (1 ml) and 1 ml of the extract in DMSO were incubated at 25° for 150 min. After incubation, 0.5 ml of the reaction mixture containing nitrate was removed and 1 ml of sulphanilic acid reagent was added, mixed well and allowed to stand for 5 min for the completion of diazotization then 1 ml of NEDD was added, mixed and allowed to stand for 30 min in different light at room temperature. The absorbance of these solutions was measured at 540 nm using ELISA reader against corresponding blank solution. IC50 value obtained is the concentration of the sample required to inhibit 50% nitric oxide radical.
H2O2 scavenging activity was done using by p-NDA (p-nitrosodimethyl aniline) method[7,8]. The reaction mixture containing ferric chloride (0.1 mM, 0.5 ml), EDTA (0.1 mM 0.5 ml) ascorbic acid (0.1 mM, 0.5 ml), H2O2 (2 mM, 0.5 ml) and p- NDA (0.01 mM, 0.5 ml) were added to give a final volume of 3 ml. Sample blank was prepared by adding 0.5ml of sample and 2.5 ml of phosphate buffer pH 7.4. Absorbance was measured at 440 nm, percentage scavenging was calculated from the control, where no extract, instead DMSO was present.
For scavenging hydrogen peroxide radical, a solution of H2O2 (20 mM) was prepared in PBS, (pH 7.4). Various concentration of 1 ml of the extract or standard in methanol were added to 2 ml of H2O2 solution in PBS. The absorbance was measured at 230 nm, after 10 min against a blank solution that contained extracts in PBS without H2O2[4–6].
Superoxide radical scavenging was done by alkaline DMSO method. To the reaction mixture containing 1 ml of alkaline DMSO, 0.3 ml of the extract in DMSO at various concentrations were added to 0.1 ml of NDT (0.1 mg) to give a final volume of 1.4 ml. The absorbance was measured at 560 nm[6,8].
In ABTS radical scavengin method, 0.2 ml of various concentrations of the extract and standard was mixed with, 1 ml of distilled DMSO and 0.16 ml of ABTS solution to make a final volume of 1.36 ml. Absorbance was measured spectrophotometrically, after 20 min at 734 nm using ELISA reader. Blank is maintained without ABTS. IC50 value obtained is the concentration of the sample required to inhibit 50% ABTS radical mono cation[6,9].
Hydroxyl radical scavenging was done by deoxyribose method[10]. Various concentrations of the extracts, the compound and standard in DMSO (0.2 ml) were added to the reaction mixture containing deoxyribose (3 mM, 0.2 ml), ferric chloride (0.1 mM, 0.2 ml), EDTA (0.1 mM, 0.2 ml), ascorbic acid (0.1 mM, 0.2 ml) and hydrogen peroxide (2 mM,0.2 ml) in phosphate buffer PH 7.4 (20 mM) to give a total volume of 1.2 ml. The solutions were then incubated for 30 min at 37°. After incubation, ice cold TCA (0.2 ml, 15% w/v) and TBA (0.2 ml, 1% w/v) in 0.25 N HCl were added. The reaction mixture was kept in a boiling water bath for 30 min, cooled and the absorbance was measured at 532 nm.
For lipid peroxidation assay, the test sample (100 μl) of different concentrations were added to 1 ml of egg lecithin mixture, control was without test sample. Lipid peroxidation was induced by adding 10 μl FeCl3 (400 mM) and 10 μl L-ascorbic acid (200 mM). After incubation for 1 h at 37°, the reaction was stopped by adding 2 ml of 0.25 N HCl containing 15% TCA and 0.375% TBA and the reaction mixture was boiled for 15 min then cooled, centrifuged and absorbance of the supernatant was measured at 532 nm[4,5,11,12].
Total antioxidant capacity (TCA) was estimated by phosphomolybdenum method[5]. 100 μl of the extract is dissolved in 1 ml of TCA reagent. Blank is maintained with distilled water replacing the TCA reagent. Absorbance is seen at 695 nm.
The statistical analysis was carried out by one way (ANOVA). The values are represented as mean±SEM. Comparison of mean values of different groups treated with different dose levels of formulations and positive control with normal were estimated by Turkey's multiple comparison test. The correlations coefficient (r2) between the parameters established by regression analysis.
Percentage of yield for petroleum ether, chloroform, ethyl acetate, n-butanol, ethanol and water extracts were 2.18, 2.04, 1.38, 3.84, 5.81 and 6.12%, respectively and the results are tabulated in Table 1. Total Phenolic content petroleum ether, chloroform, ethyl acetate, n-butanol, ethanol and water were 16±0.57, 16.6±0.33, 2±0.57, 14±0.06, 64.6±1.2 and 5±0.57, respectively and the results are tabulated in Table 2. The differences in total Phenolic content of all samples were significant (P<0.05). The ethanol extract has the highest total phenol content followed by chloroform, petroleum ether, n-butanol, water and ethyl acetate. The differences between ethanol and ethyl acetate are almost 32 fold. Total flavonol content among different extract revealed that ethanol extract exhibited higher flavonol content of 85.3±0.8 mg/g of rutin followed by water 82±1.15, chloroform 41.3±0.8, pet ether 21±0.57, ethyl acetate 20±0.57 mg/g of ruin. n-butanol extract showed very low content of flavonol with 19±1.52 mg/g of rutin.
TABLE 1.
PERCENTAGE YIELD OF DIFFERENT EXTRACTS OF ABUTILON INDICUM LINN
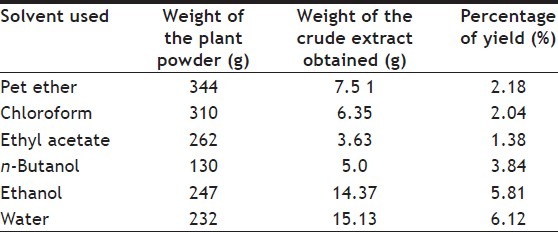
TABLE 2.
TOTAL PHENOL AND FLAVONOL ESTIMATION OF DIFFERENT EXTRACTS OF ABUTILON INDICUM LINN

DPPH scavenging activity of different extract of Abutilon indicum linn was shown in the Table 3. IC50 of various fractions was compared with total phenolic content. The antioxidant in DPPH not correlated well with phenolic content and total flavonol content.
TABLE 3.
ANTIOXIDANT ACTIVITY OF ABUTILON INDICUM LINN
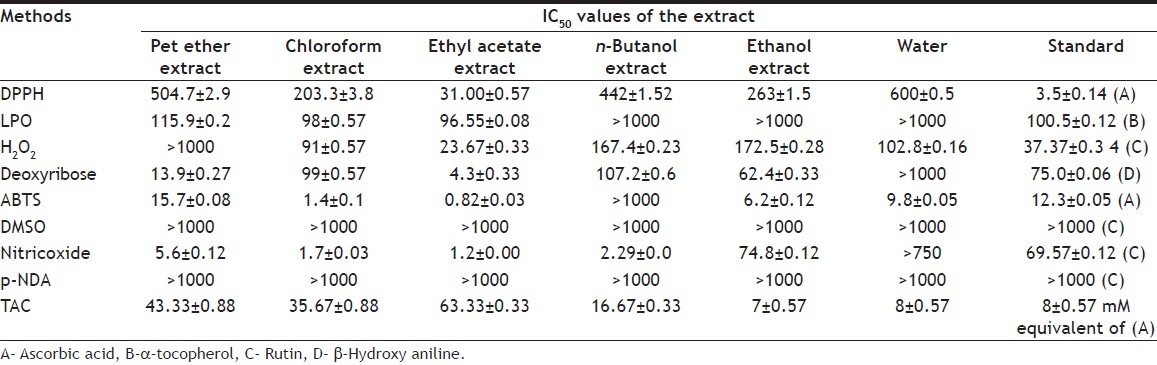
The behavior of all the extracts were monitored after inducing radical peroxidation in egg lecithin and the inhibitory activity displayed by ethyl acetate extract of Abutilon indicum is again noteworthy and comparable to standard α-tocopherol. The results were tabulated in Table 3. In this method correlation coefficient with Phenolic and flavonolic showed P<0.4732 and P<0.2025 respectively and it was not 95% confidence interval.
Hydrogen peroxide method itself is not very reactive, but it sometimes is toxic to cells. Since it may give rise to hydroxyl radicals inside the cell. Ethyl acetate was capable of scavenging hydrogen peroxide in a concentration dependent manner. There was no significant correlation with the total phenolic (P<0.9685, r2 = 0.0004) and flavonol content (P<0.5407, r2 = 0.1004). Ethyl acetate showed potent activity than the standard rutin.
Deoxy ribose was used as a detector molecule to detect the damage by OH radical in the presence or absence of EDTA. Ethyl acetate was not active on the degradation of hydroxyl radical. Correlation coefficient analysis showed no significant result with phenol (r2 = 0.08951) and flavonolic (r2 = 0.3586) content.
ABTS is an excellent tool for determining the antioxidant activity of hydrogen donating antioxidants and of chain braking antioxidants. Regression analysis showed no significant correlation with the phenol (P<0.8179 r2 = 0.01489), and flavonol content (P<0.4280, r2 = 0.1616).
Among the tested extracts, none of the extract showed active in this assay with the IC50 value above 1000 μg/ml in Scavenging of superoxide radical by alkaline DMSO method.
In nitric oxide radical inhibition activity, among the extracts, the ethyl acetate showed potent free radical scavenging activity against nitric oxide. IC50 value of ethyl acetate is 57.975 times potent than rutin standard.
In hydroxyl radical scavenging activity method by p-NDA method, almost all the extracts were found to be inactive with IC50 value above 1000 μg/ml. Rutin standard itself showed to be inactive in this assay.
Ethyl acetate extract showed maximum free radical scavenging activity. Total antioxidant capacity was compared with IC50 value of all antioxidant method and it was compared with total phenol and flavonol content. IC50 value for various antioxidant methods for all extract showed no significance with total antioxidant capacity except IC50 value of LPO (r2 = 0.7273). Correlation between total antioxidant capacity and total phenolic content was not significant with r2 = 0.2554, P<0.3065. Total antioxidant capacity and total flavonol content showed similar correlation with r2 = 0.2554, P<0.0962.
Several studies evaluated the relationship between antioxidant capacity of plant products and their chemical composition. Some authors found a correlation between the phenolic content, flavonolic content and antioxidant activity, while others found no relationship. It is reported that a high correspondence between total phenolic content and antioxidant activity in selected fruits, vegetables and grain products[13]. On the other hand, some reports showed that there is no correlation between antioxidant activity and phenolic content in plant extract containing phenolic compound[14].
In this study the correlation coefficient analysis found to have no relationship between antioxidant activity and total phenolic and flavonolic content. Among all the extract analyzed, a significant total phenolic content and total flavonolic content were found only in ethanol extract of Abutilon indicum but it did not show a higher antioxidant activity. On the other hand, ethyl acetate showed potent antioxidant activity in lipid peroxidation method, hydrogen peroxide scavenging activity. Deoxiribose, ABTS, nitric oxide and total antioxidant capacity but did not show high phenol and flavonolic content.
The relatively high antioxidant and free radical scavenging activity of extracts containing low phenolic content suggests that the type of phenolic compound is determined for the free radical or antioxidant activities rather than their amounts. This results agree with the report that the difference in antioxidant activities of the plant extract could be different qualitative and quantitative composition of their phenolic constituents from phenolic acids and their derivatives such as esters, depends on the number of hydroxyl groups in the molecules[14,15]. So it is worth to go for the isolation and identifications of the individual components from the extract and perform the same studies in order to find out the correlation of the individual components that is present in the extract with the antioxidant and biological activities.
Footnotes
Srividya, et al.: Antioxidant Activity of Abutilon Indicum Linn
REFERENCES
- 1.Mori A, Nishinoc Enoki N, Tavata S. Cytotoxicity of plant flavonoids against Hela cells. Phytochemistry. 1988;27:1017–20. [Google Scholar]
- 2.Coban T, Citoglu GS, Sever B, Iscan M. Antioxidant activity of plants used in Traditional medicine in turkey. Pharm Biol. 2003;41:608–13. [Google Scholar]
- 3.Khare CP. New York: Springer Science+Business Media; 2007. Indian Medicinal Plants: An Illustrated Dictionary. [Google Scholar]
- 4.Jayaprakasha GK, Jaganmohan Rao L, Sakariah KK. Antioxidant activities of flavidin in different in vitro model systems. Bioorg Med Chem. 2004;12:5141–6. doi: 10.1016/j.bmc.2004.07.028. [DOI] [PubMed] [Google Scholar]
- 5.Sithisarn P, Supabphot R, Gritsanapan W. Antioxidant activity of Siamese neem tree (VP-1209) J Ethnopharmacol. 2005;99:109–12. doi: 10.1016/j.jep.2005.02.008. [DOI] [PubMed] [Google Scholar]
- 6.Moreno S. Methods used to evaluate the free radical scavenging activity in food and biological systems. Food Sci Technol Inst. 2002;8:121–37. [Google Scholar]
- 7.Kunchandy E, Rao MN. Effect of Curcumin on hydroxyl radical generations through Fenton reactions. Int J Pharm. 1989;57:173–6. [Google Scholar]
- 8.Kunchandy E, Rao MN. Oxygen radical scavenging activity of curcumin. Int J Pharm. 1990;58:237–40. [Google Scholar]
- 9.Roberta R, Pellegrini N, Protefente A, Pannala A. Antioxidant activity applying an imporved ABTS radical cation decolorization assay. Free Radic Biol Med. 1999;26:1231–7. doi: 10.1016/s0891-5849(98)00315-3. [DOI] [PubMed] [Google Scholar]
- 10.Torel J, Collard J, Cillard P. Antioxidant activity of flavonoids and reactivity with peroxy radicals. Phytochemistry. 1985;25:383–5. [Google Scholar]
- 11.Duh PD, Yen GC, Yen WJ, Chang LW. Antioxidant effects of water extract from barley (Hordenum vulgare) prepared under different roasting temperatures. J Agric Food Chem. 2001;49:1455–63. doi: 10.1021/jf000882l. [DOI] [PubMed] [Google Scholar]
- 12.Cook NC, Samman S. Flavonoids-chemistry, metabolism, cardio protective effects and dietary sources. Nutr Biochem. 1996;7:66–76. [Google Scholar]
- 13.Velioglu YS, Mazza G, Gao L, Oomah BD. Antioxidant activity and total phenolic in selected fruits, vegetables and grain products. J Agric Food Chem. 1998;46:4113–7. [Google Scholar]
- 14.Kahkonen MP, Hopia AI, Vuorela HJ, Rauha JP, Philaja K, Kujala TS, et al. Antioxidant activity of plant extract containing phenolic compounds. J Agric Food Chem. 1999;47:3954–62. doi: 10.1021/jf990146l. [DOI] [PubMed] [Google Scholar]
- 15.Shahidi F, Marian N. 1st ed. Boca Raton: CRC Press; 2003. Phenolic in Food and Neutraceuticals. [Google Scholar]


