Abstract
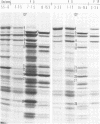
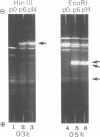
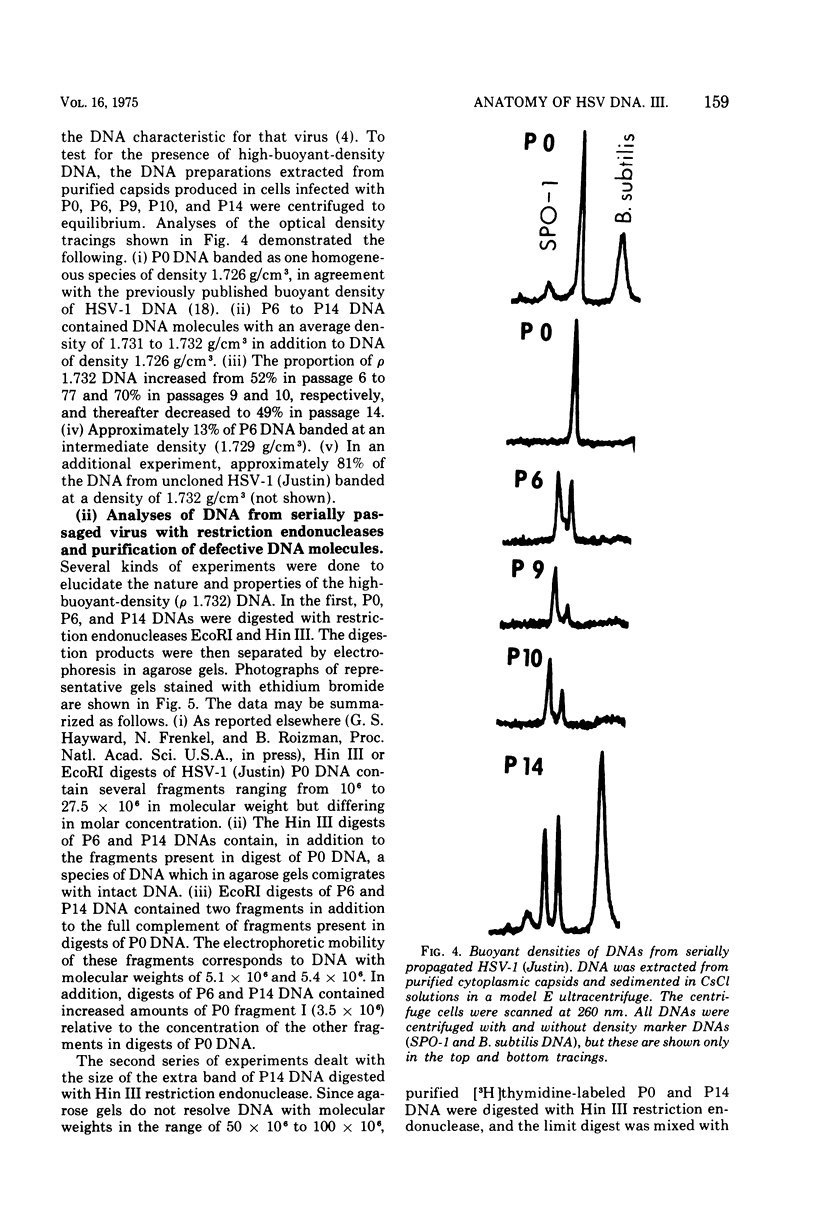
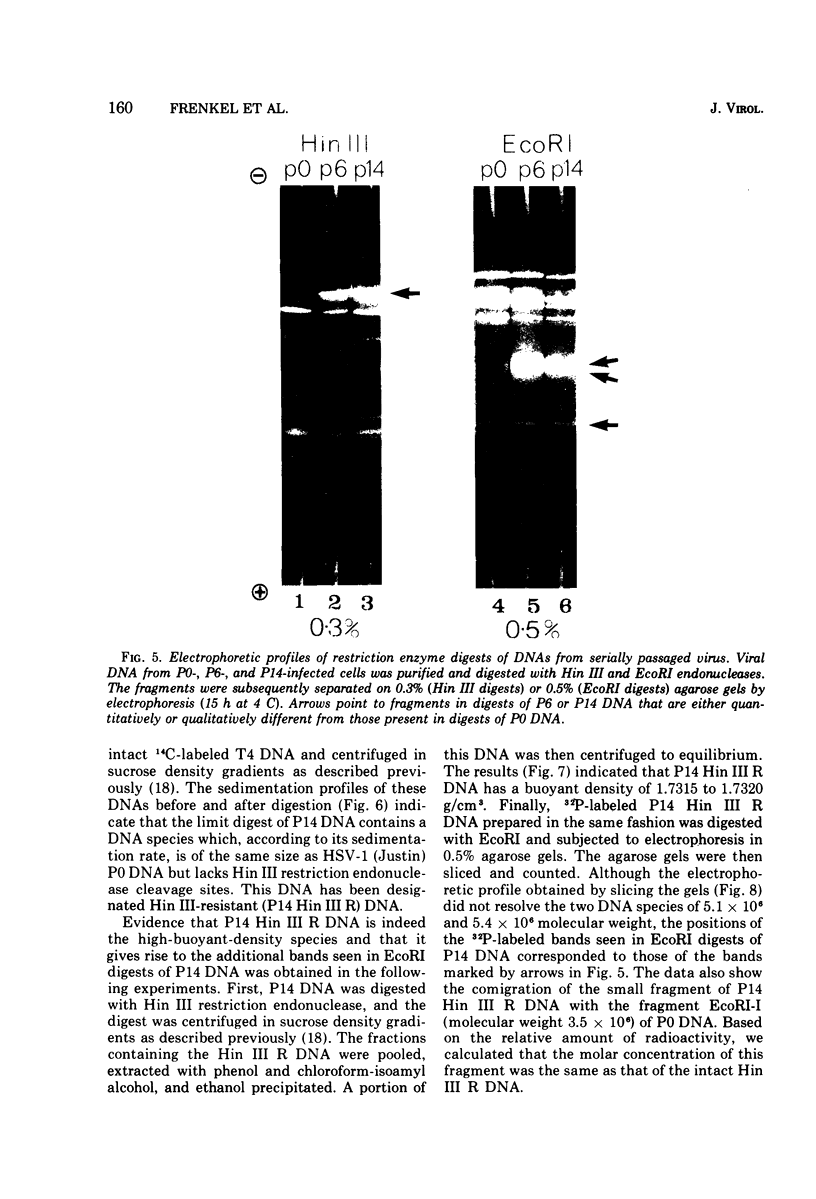
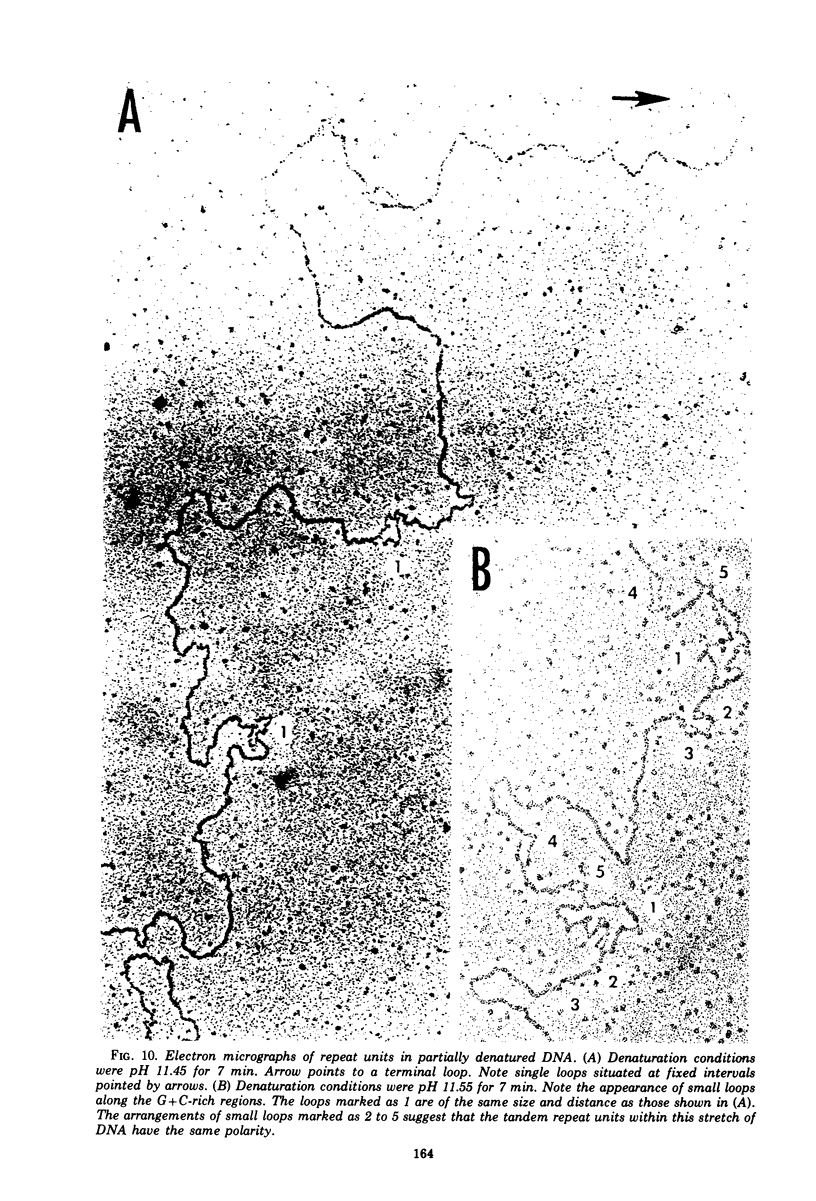
We have characterized the virus progeny and its DNA from plaque-purified and undiluted passages of herpes simplex virus 1 in HEp-2 cells. Secifically, (i) infectious virus yields declined progressively in passages 1 through 10 and gradually increased at passages 11 through 14. The yields correlated with PFU/particle ratios. (ii) In cells infected with virus from passages 6 through 10, there was an overproduction of an early viral polypeptide (no. 4) and a delay in the synthesis of late viral proteins. In addition, the virus in these passages interfered with the replication of a nondefective marker virus. Cells infected with passage 14 virus produced normal amounts of polypeptide 4 and, moreover, this virus showed minimal interfering capacity. (iii) In addition to DNA of density 1.726 g/cm-3, which was the sole component present in viral progeny of passage 0, passages 6 through 14 contained one additional species (p 1.732) and in some instances (passages 6 and 10) also DNA of an intermediate buoyant density. The ratio of p 1.732 to p 1.726 DNA increased to a maximum of 4 in passages 6 through 9 and gradually decreased to 1 in passages 10 through 14. (iv) p 1.732 DNA cannot be differentiated from p 1.726 DNA with respect to size; however, it has no Hin III restriction enzyme cleavage sites and yields only predominantly two kinds of fragments with molecular weights of 5.1 x 10-6 and 5.4 x 10-6 upon digestion with EcoRI enzyme. (v) Partial denaturation profiles of purified p 1.732 DNA from passage 14 revealed the presence of two types of tandemly repeated units corresponding roughly in size to the EcoRI fragments and situated in different molecules. (vi) In addition to the two kinds of p 1.732 molecules consisting of tandem repaeat units of different sizes, other evidence for the diversity of defective DNA molecules emerged from comparisons of specific infectivity and interfering capacity of the progeny from various passages. The data suggest that some of the particles with DNA of normal buoyant density (1.726) must also be defective since the capacity to interfere and to produce an excess of polypeptide 4 did not appear to be proportional to the amount of high-buoyant-density defective DNA. The data suggest that defective interfering particles are replaced by defective particles with diminished capacity to interfere and that more than one species of defective DNA molecules evolves on serial preparation of HSV.
Full text
PDF










Images in this article
Selected References
These references are in PubMed. This may not be the complete list of references from this article.
- Anker H. S. A solubilizable acrylamide gel for electrophoresis. FEBS Lett. 1970 Apr 16;7(3):293–293. doi: 10.1016/0014-5793(70)80185-5. [DOI] [PubMed] [Google Scholar]
- Ben-Porat T., Demarchi J. M., Kaplan A. S. Characterization of defective interfering viral particles present in a population of pseudorabies virions. Virology. 1974 Sep;61(1):29–37. doi: 10.1016/0042-6822(74)90239-6. [DOI] [PubMed] [Google Scholar]
- Brockman W. W., Nathans D. The isolation of simian virus 40 variants with specifically altered genomes. Proc Natl Acad Sci U S A. 1974 Mar;71(3):942–946. doi: 10.1073/pnas.71.3.942. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Bronson D. L., Dreesman G. R., Biswal N., Benyesh-Melnick M. Defective virions of herpes simplex viruses. Intervirology. 1973;1(3):141–153. doi: 10.1159/000148841. [DOI] [PubMed] [Google Scholar]
- Ejercito P. M., Kieff E. D., Roizman B. Characterization of herpes simplex virus strains differing in their effects on social behaviour of infected cells. J Gen Virol. 1968 May;2(3):357–364. doi: 10.1099/0022-1317-2-3-357. [DOI] [PubMed] [Google Scholar]
- Fareed G. C., Byrne J. C., Martin M. A. Triplication of a unique genetic segment in a simian virus 40-like virus of human origin and evolution of new viral genomes. J Mol Biol. 1974 Aug 5;87(2):275–288. doi: 10.1016/0022-2836(74)90149-1. [DOI] [PubMed] [Google Scholar]
- Frenkel N., Lavi S., Winocour E. The host DNA sequences in different populations of serially passaged SV40. Virology. 1974 Jul;60(1):9–20. doi: 10.1016/0042-6822(74)90360-2. [DOI] [PubMed] [Google Scholar]
- Gibson W., Roizman B. Proteins specified by herpes simplex virus. Staining and radiolabeling properties of B capsid and virion proteins in polyacrylamide gels. J Virol. 1974 Jan;13(1):155–165. doi: 10.1128/jvi.13.1.155-165.1974. [DOI] [PMC free article] [PubMed] [Google Scholar]
- HOGGAN M. D., ROIZMAN B. The isolation and properties of a variant of Herpes simplex producing multinucleated giant cells in monolayer cultures in the presence of antibody. Am J Hyg. 1959 Sep;70:208–219. doi: 10.1093/oxfordjournals.aje.a120071. [DOI] [PubMed] [Google Scholar]
- Hayward G. S. Unique double-stranded fragments of bacteriophage T5 DNA resulting from preferential shear-induced breakage at nicks. Proc Natl Acad Sci U S A. 1974 May;71(5):2108–2112. doi: 10.1073/pnas.71.5.2108. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Heine J. W., Honess R. W., Cassai E., Roizman B. Proteins specified by herpes simplex virus. XII. The virion polypeptides of type 1 strains. J Virol. 1974 Sep;14(3):640–651. doi: 10.1128/jvi.14.3.640-651.1974. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Honess R. W., Roizman B. Proteins specified by herpes simplex virus. XI. Identification and relative molar rates of synthesis of structural and nonstructural herpes virus polypeptides in the infected cell. J Virol. 1973 Dec;12(6):1347–1365. doi: 10.1128/jvi.12.6.1347-1365.1973. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Honess R. W., Roizman B. Regulation of herpesvirus macromolecular synthesis. I. Cascade regulation of the synthesis of three groups of viral proteins. J Virol. 1974 Jul;14(1):8–19. doi: 10.1128/jvi.14.1.8-19.1974. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Huang A. S. Defective interfering viruses. Annu Rev Microbiol. 1973;27:101–117. doi: 10.1146/annurev.mi.27.100173.000533. [DOI] [PubMed] [Google Scholar]
- Inman R. B., Schnös M. Partial denaturation of thymine- and 5-bromouracil-containing lambda DNA in alkali. J Mol Biol. 1970 Apr 14;49(1):93–98. doi: 10.1016/0022-2836(70)90378-5. [DOI] [PubMed] [Google Scholar]
- Kieff E. D., Bachenheimer S. L., Roizman B. Size, composition, and structure of the deoxyribonucleic acid of herpes simplex virus subtypes 1 and 2. J Virol. 1971 Aug;8(2):125–132. doi: 10.1128/jvi.8.2.125-132.1971. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Lang D., Mitani M. Simplified quantitative electron microscopy of biopolymers. Biopolymers. 1970;9(3):373–379. doi: 10.1002/bip.1970.360090310. [DOI] [PubMed] [Google Scholar]
- Lavi S., Rozenblatt S., Singer M. F., Winocour E. Acquisition of sequences homologous to host DNA by closed circular simian virus 40 DNA. II. Further studies on the serial passage of virus clones. J Virol. 1973 Sep;12(3):492–500. doi: 10.1128/jvi.12.3.492-500.1973. [DOI] [PMC free article] [PubMed] [Google Scholar]
- ROIZMAN B., ROANE P. R., Jr A physical difference between two strains of herpes simplex virus apparent on sedimentation in cesium chloride. Virology. 1961 Sep;15:75–79. doi: 10.1016/0042-6822(61)90079-4. [DOI] [PubMed] [Google Scholar]
- ROIZMAN B. The programming of herpes virus multiplication in doubly-infected and in puromycin-treated cells. Proc Natl Acad Sci U S A. 1963 Feb 15;49:165–171. doi: 10.1073/pnas.49.2.165. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Roizman B., Spear P. G. Preparation of herpes simplex virus of high titer. J Virol. 1968 Jan;2(1):83–84. doi: 10.1128/jvi.2.1.83-84.1968. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Sharp P. A., Sugden B., Sambrook J. Detection of two restriction endonuclease activities in Haemophilus parainfluenzae using analytical agarose--ethidium bromide electrophoresis. Biochemistry. 1973 Jul 31;12(16):3055–3063. doi: 10.1021/bi00740a018. [DOI] [PubMed] [Google Scholar]
- Smith H. O., Wilcox K. W. A restriction enzyme from Hemophilus influenzae. I. Purification and general properties. J Mol Biol. 1970 Jul 28;51(2):379–391. doi: 10.1016/0022-2836(70)90149-x. [DOI] [PubMed] [Google Scholar]
- Spear P. G., Roizman B. Proteins specified by herpes simplex virus. V. Purification and structural proteins of the herpesvirion. J Virol. 1972 Jan;9(1):143–159. doi: 10.1128/jvi.9.1.143-159.1972. [DOI] [PMC free article] [PubMed] [Google Scholar]
- VINOGRAD J., HEARST J. E. Equilibrium sedimentation of macromolecules and viruses in a density gradient. Fortschr Chem Org Naturst. 1962;20:373–422. [PubMed] [Google Scholar]
- WATSON D. H., RUSSELL W. C., WILDY P. Electron microscopic particle counts on herpes virus using the phosphotungstate negative staining technique. Virology. 1963 Mar;19:250–260. doi: 10.1016/0042-6822(63)90062-x. [DOI] [PubMed] [Google Scholar]
- Wadsworth S., Jacob R. J., Roizman B. Anatomy of herpes simplex virus DNA. II. Size, composition, and arrangement of inverted terminal repetitions. J Virol. 1975 Jun;15(6):1487–1497. doi: 10.1128/jvi.15.6.1487-1497.1975. [DOI] [PMC free article] [PubMed] [Google Scholar]