Abstract
Background
Nitric oxide (NO), a potent signaling molecule, is known to inhibit platelet function in vivo. We investigated how the levels of NO and its metabolites change during routine platelet storage. We also tested whether the material of platelet storage containers affects nitrite content since many plastic materials are known to contain and release nitrite.
Study design and methods
For nitrite and nitrate measurement, leukoreduced apheresis platelets (PLT) and concurrent plasma (CP) were collected from healthy donors using the Trima Accel. Sixty mL aliquots of PLT or CP were stored in CLX or PL120 Teflon containers at 20–24°C with agitation and daily samples were processed to yield PLT pellet and supernatant. In a separate experiment, PLT was stored in PL120 Teflon to measure NO generation using electron paramagnetic resonance (EPR).
Results
Nitrite level increased markedly in both PLT supernatant and CP stored in CLX containers at a rate of 58 nM/day and 31 nM/day respectively. However, there was a decrease in nitrite level in PLT stored in PL120 Teflon containers. Nitrite was found to leach from CLX containers and this appears to compensate for nitrite consumption in these preparations. Nitrate level did not significantly change during storage.
Conclusion
Platelets stored at 20–24°C maintain measurable levels of nitrite and nitrate. Nitrite decline in non-leachable Teflon containers in contrast to increases in CLX containers which leach nitrite, suggests that it is consumed by platelets, residual leukocytes or erythrocytes. These results suggest NO-related metabolic changes occur in platelet units during storage.
Keywords: nitric oxide, nitrite, platelet storage, transfusion
Introduction
Nitric Oxide (NO) is the endothelium-derived free radical that regulates vascular tone and other function by stimulating soluble guanylyl cyclase (sGC) in smooth muscle cells 1–3 and also plays an important role in the regulation of thrombosis and hemostasis by modulating platelet function 4,5. NO generated in the endothelium by endothelial nitric oxide synthase (eNOS) can diffuse into the lumen of the vessel and traverse the platelet membrane. NO binding to intracellular sGC leads to the generation of cyclic guanosine monophosphate (cGMP) in platelets and initiation of various downstream signals including reduction of Ca2+ mobilization 6–8, shape change by cytoskeleton rearrangement 9,10, and decreased adhesion and aggregation 10,11. While the effect of endothelium-derived NO on platelet function is well established by numerous studies, the presence of an eNOS signaling pathway endogenous to platelets is still under debate. It was first reported in 1990 by Radomski et al. that platelets have a functional eNOS signaling pathway 12, however recent studies suggested that platelets do not express detectable amount of eNOS 13–15. Gambaryan et al. suggested that previous studies might have used impure platelets which contained other cell types and/or inappropriate antibodies against eNOS 14.
In addition to eNOS-derived NO production, an alternative pathway for NO generation was recently established showing that the inorganic anions nitrate (NO3−) and nitrite (NO2−), once considered inert end products from NO generation, can be reduced to NO by non-enzymatic as well as enzymatic pathways 16–19. Nitrate from dietary or endogenous sources is first converted to nitrite by commensal bacteria in the oral cavity 19,20, nitrite then can be further reduced to NO by several pathways including deoxyhemoglobin 21,22, deoxymyoglobin 23,24, xanthine oxidoreductase 25–27, or ascorbate 28. Recent studies on human subjects show that nitrate from dietary source inhibits platelet aggregation in response to platelet agonists such as collagen and ADP 29 and inorganic nitrite inhibits platelet aggregation in the presence of erythrocytes 30 suggesting a NOS-independent antiplatelet effect of nitrite metabolites.
Since there is increasing demand for platelet transfusion, it is of potential value to understand platelet physiology and biochemical changes which occur during storage to improve platelet quality. Platelet storage is currently restricted to 5 days because of the risk of bacterial contamination and because of well characterized decrements in PLT quality during storage, leading to a decline in platelet storage properties as well as apoptotic changes including externalization of phosphatidylserine and mitochondrial damage 31–36. There is no published study so far on the levels of NO and its metabolites as a function of platelet storage time. Considering the possibility that nitrite and nitrate can be converted back to NO and NO inhibits many aspects of platelet function, we studied NO metabolism in platelets by monitoring levels of nitrite, nitrate and NO during storage of apheresis platelet units and concurrent plasma.
Materials and Methods
Apheresis platelets collection and storage
Apheresis platelets were collected from healthy donors using a Trima Accel cell separator (Software version 5.1, CaridianBCT Inc., Lakewood, CO) with a targeted yield of 3.5×1011 platelets. Aliquots of 60 mL of apheresis PLT were transferred, using a sterile connecting device (TSCD, model SC-201A, Terumo Medical Corp., Elkton, MD), into CLX (Pall Corporation, East Hills, NY) or PermaLife PL120 Teflon (OriGen Biomedical, Austin TX) containers. Similarly, 60 mL aliquots of CP were sterilely transferred into CLX or PL120 Teflon containers. PLT and CP aliquots were stored in a PLT incubator (Helmer, Nobelsville, IN) at 20–24°C with constant reciprocal agitation.
Nitrite, nitrate and nitrosothiol analysis
Nitrite and nitrate levels in the apheresis platelet units were measured using a standard chemiluminescence method 37. For nitrite analysis, 1mL samples of PLT or CP were first centrifuged at 3,000 xg and the resulting supernatant and pellets were separately mixed with nitrite preserving solution (K3Fe(CN6N-ethylmaleimide, water, Nondiet P-40) to maintain nitrite levels and kept frozen at −70°C until analysis. Samples were deproteinized by dilution with the same amount of methanol, then centrifuged for 5 min at 13,000 rpm (AccuSpinMicroR, Fisher Scientific, Pittsburgh, PA) and the supernatant was immediately injected into the chemiluminescent nitric oxide analyzer (NOA, Sievers, Model 280 NO analyzer, Boulder, CO) using helium as the carrier gas for chemiluminiscence determination of NO. For nitrate analysis, 5% Triton X-100 in deionized water was added to PLT supernatant, pellets or CP instead of nitrite preserving solution at a 1:9 ratio between the sample and 5% Triton X-100. At the time of sample analysis, a 3:1 dilution of ethanol and thawed sample was centrifuged, and the supernatant was analyzed with NOA using the vanadium(III) chloride (VCl3) and chemiluminescence assay. For nitrosothiol (SNO) analysis, 4 parts of a thiol-stabilization solution (NEM-DPTA; K3Fe(CN6N-ethylmaleimide, Diethylenetriaminepenta acetic acid, Nondiet P-40, water) were mixed with one part of samples. A 9:1 dilution of sample and 5% acid sulfanilamide (AS) was incubated for 5 min; one half was injected into the NOA (I3− assay) to give combined SNO and iron-nitrosyl complexes. The other half was incubated with 50 mM HgCl2then incubated again with 5% AS, and injected into the NOA to give iron-nitrosyl complex levels 38.
NO measurement with EPR
Sixty mL of PLT or 45 ml of CP were stored in PL120 Teflon containers. Samples were taken on each day of storage and mixed with cPTIO (2-(4-carboxyphenyl)-4,4,5,5-tetramethylimidazoline-1-oxyl-3-oxide, 200µM final concentration) and frozen until EPR analysis. The cPTIO can be used as NO spin trap 39, as it specifically reacts with NO to yield a non-paramagnetic product. Therefore, the EPR line heights are inversely proportional to amount of NO in solution. In our study we used only the central peak height for NO detection purposes. No attempt was made to estimate the actual NO concentrations, as the stoichiometry of cPTIO reaction with NO is too complex 40. For the clarity of presentation, we plotted inverted central peak heights, so the values are directly proportional to the amount of NO present.
Aggregation measurement and analysis of CD62 positive cells
PLT aggregation was measured at 37°C using a light transmittance instrument (Chronolog, Havertown, PA). PLT samples were diluted with CP to yield a PLT count of 3.0×105/µL. ADP and collagen were added together at final concentrations of 10 µmol/L and 10 µg/mL, respectively, to initiate aggregation. The extent of aggregation (percent amplitude) was measured to describe the aggregation process.
The percent of PLTs expressing membrane-bound CD62P (p-selectin) was measured by flow cytometry (FACSCalibur, BD Biosciences, San Jose, CA). PLT samples were diluted to 1x106 PLTs/mL with PBS supplemented with 0.1% human albumin. Sample aliquots were incubated with fluorescein isothiocyanate (FITC)-conjugated CD61 monoclonal antibodies and with phycoerythrin (PE)-conjugated CD62P (eBioscience Inc., San Diego, CA) at saturating concentrations for 15 min at 22°C. Mouse IgG1 PE and FITC PLT isotype control (BD Biosciences) were used at saturating concentrations as negative controls.
Results
Measurement of NO metabolites in PLT and CP
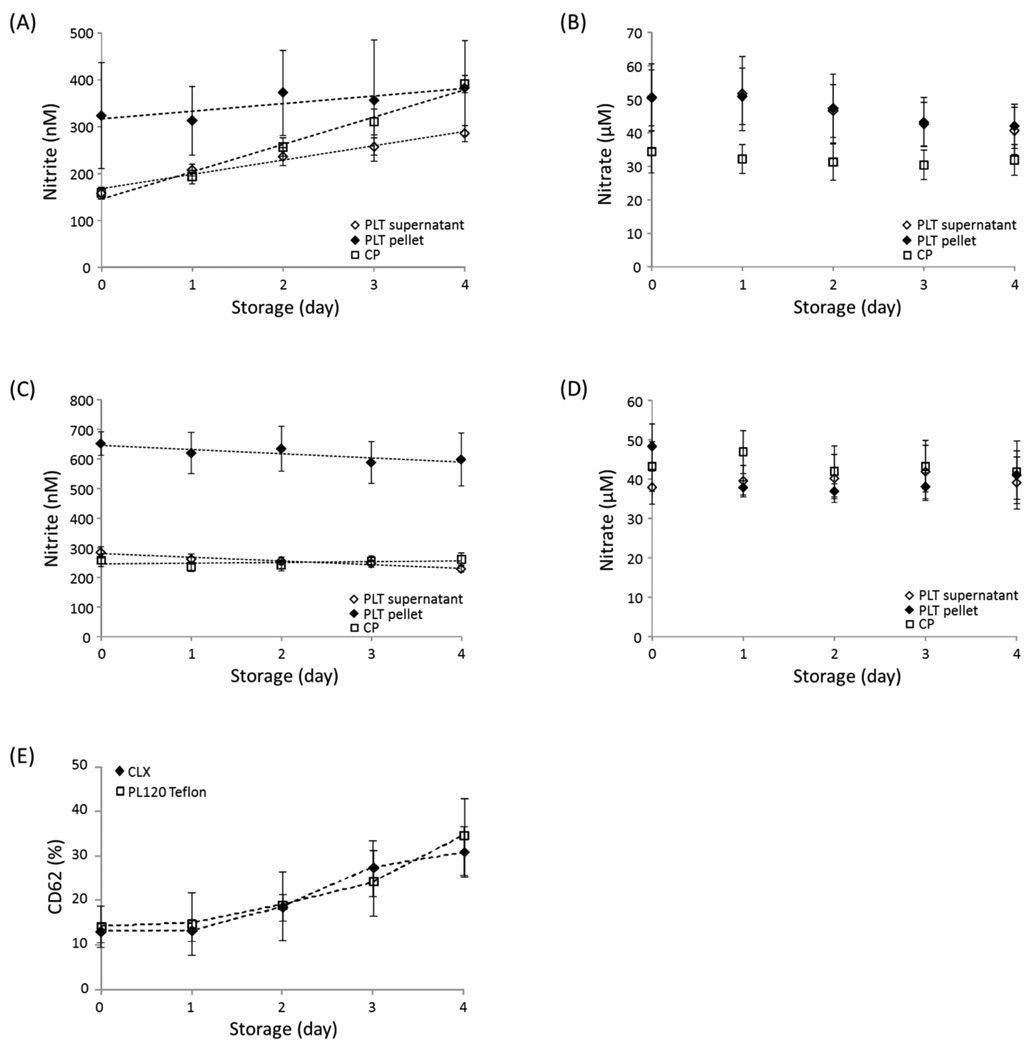
PLT and CP were stored in CLX or PL120 Teflon containers with continuous agitation and the levels of NO metabolites were analyzed. Nitrite, nitrate and nitrosothiol (SNO) contents were analyzed using a chemiluminescence method. We could not detect any noticeable amount of SNO (data not shown). Nitrite levels in both PLT supernatant and CP stored in CLX containers increased greatly during 4 days of storage at a rate of 31 nM/day and 58 nM/day, respectively, from an initial level of about 160 nM (Fig. 1A) and this increase was maintained until day 7 in a linear fashion (data not shown). In contrast, nitrite levels of the supernatant of PLT aliquots stored in PL120 Teflon containers slightly decreased from 285 nM to 230 nM while that of CP did not show any change (Fig. 1C). The levels of CD62-positive platelets stored in CLX and PL120 Teflon bags were very similar throughout storage period showing about 30% at day 4 (Fig. 1E) implying increased nitrite levels in PLTs stored in CLX did not affect platelet activation during storage.
Figure 1. Changes in nitrite content during storage.
Leukoreduced (< 2×104 leukocytes/60 mL) apheresis platelets were collected from healthy donors using the Trima Accel. Sixty mL of PLT or CP was stored in CLX (A and B, n=5) or PL120 Teflon (C and D, n=4) containers at 20–24°C with agitation. Nitrite (A and C) and nitrate (B and D) were measured in the PLT supernatant, pellets and CP using a chemiluminescence method. The percentage of CD62 positive cell was measured by flow cytometry (E, n=3). Data are means ± SEM.
Nitrate levels during storage of PLT aliquots in CLX containers were initially about 50 µM and decreased slightly over 4 days to 41 µM. No significant changes in nitrate levels were observed with the PLT stored in PL120 Teflon and CP aliquots stored in CLX or PL120 Teflon containers (Fig. 1B and D).
Analysis of nitrite leaching from platelet storage containers
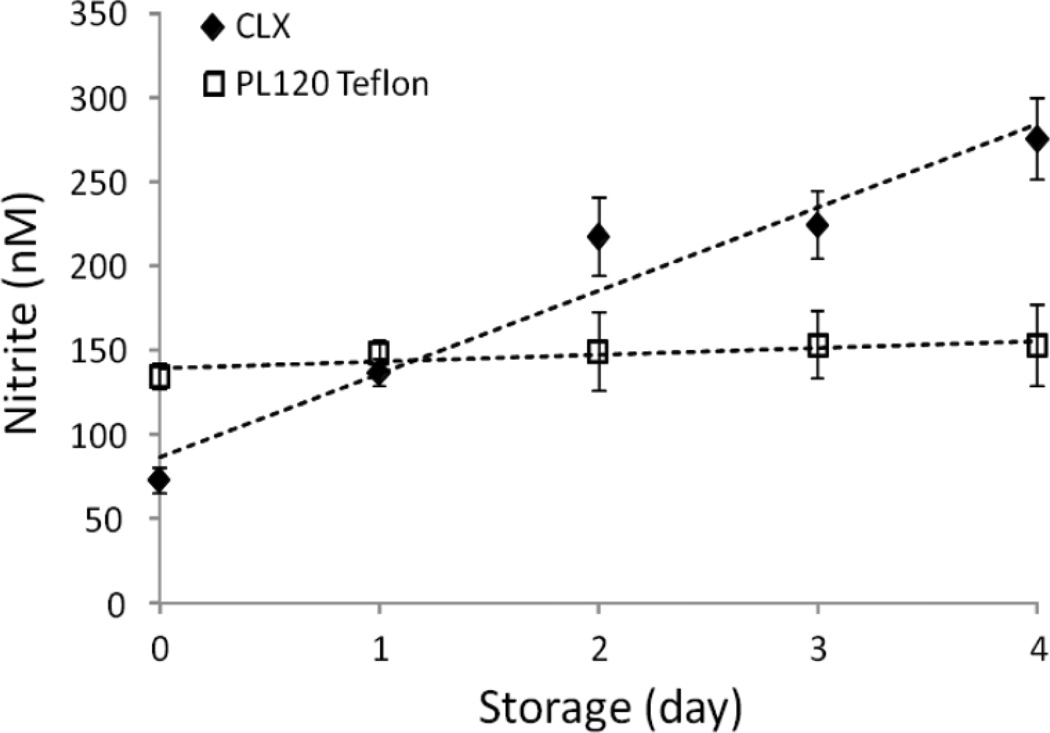
To test whether platelet storage containers leach nitrite during storage, plasma was stored in CLX or PL120 Teflon containers and the nitrite content was measured. The CLX container, which is being used widely for platelet storage for transfusion, is made of polyvinyl chloride (PVC) plastics and triethylhexyl trimellitate (TEHTM) plasticizer; the PL120 Teflon container is made of polytetrafluoroethylene (PTFE) plastics and contains no plasticizer. Nitrite levels in plasma stored in CLX containers showed a linear increase at a rate of 45 nM/day, while plasma stored in PL120 Teflon container did not show any change in nitrite level over the 4 days (Fig. 2). There was no significant difference in nitrite leaching levels of CLX container between room air storage and anoxic argon chamber storage (data not shown), thus gas exchange with air does not seem related to nitrite leaching. Nitrate levels in plasma stored in CLX container did not show significant changes during the same period (data not shown).
Figure 2. Nitrite leaching out of CLX container.
Sixty ml of plasma was stored in CLX containers (n=4) or in PL120 Teflon containers (n=4) at 20–24°C and aliquots from each container were taken every day to measure nitrite content with triiodide (I3−) ozone-based chemiluminescence. Data are means ± SEM.
Although we confirmed that nitrite leached from the CLX containers during storage, the fact that increase rate of nitrite is smaller in PLT supernatant than in CP (Fig. 1A) suggests that the platelets and/or other types of residual cells in the PLT utilized nitrite during the 4 days of storage.
Measurement of NO in PLT and CP during storage in PL120 Teflon
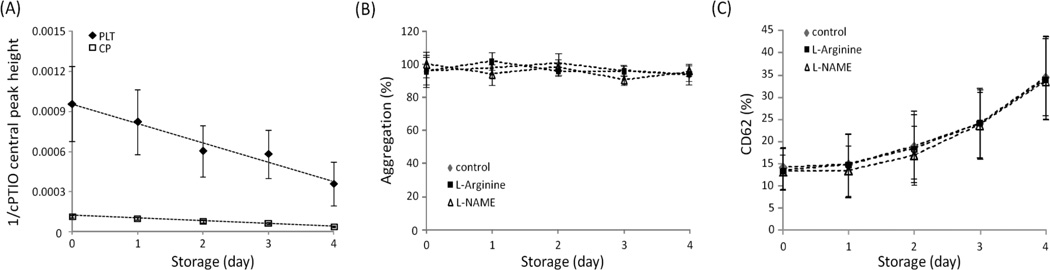
Using NO probe cPTIO, NO production was analyzed with EPR during storage in PL120 Teflon container (Fig. 3A). The amount of NO generated at each day in PLT was substantially greater than in CP; however NO production decreased gradually over time both in PLT aliquots and CP suggesting a decline in NO producing activity during storage. However this method was not amenable to quantitate analysis due to complicated kinetics of cPTIO reaction with NO.
Figure 3. NO measurement during storage.
PLT or CP aliquots were stored in PL120 Teflon containers at 20–24°C with agitation. Samples were taken every day and mixed with cPTIO (200 µM) to analyze NO level with EPR. Inverted central peak heights were shown, so the values are directly proportional to the amount of NO present (A). Aggregation (B) was measured by PLT response to two agonists (ADP and collagen, 10 µM and 10 µg/ml, respectively) via changes in light transmittance and the percentage of CD62 positive PLT (C) was measured by flow cytometry. Data are means ± SEM.
In an additional series of experiments, L-Arginine (NOS substrate, 300µM) or L-NAME (NG-nitro-L-arginine methyl ester, NOS inhibitor, 300µM) was added to the PLT at day 0 to determine if detected NO was enzymatically produced by NOS in platelet preparations during storage. There were no apparent changes in NO level after the addition of L-Arginine or L-NAME compared to control (data not shown). The possible explanations are that: 1) NOS contribution to NO level change in apheresis platelets is trivial or absent since platelet might not even express any NOS as others suggested 13–15; or 2) changes in NO level were under the detection limit of EPR instrument we used. We also confirmed that addition of L-Arginine and L-NAME did not affect PLT aggregating property and the percentage of CD62 positive cells during storage (Fig. 3B and C). We then added nitrite (1µM) to the PLT at day 0 to check if additional nitrite would cause an increase in NO level during storage through its reduction to NO, however we did not observe any clear increase in NO level after the addition of nitrite (data not shown). It seems that endogenous level of nitrite in the PLT (~ 300nM) might be enough to create pseudo first order kinetics in NO generation reaction.
Discussion
The inhibitory role of endothelium-derived NO in platelet function has been known since the early 1980’s 41–43. NO exerts its inhibitory role on platelets by stimulating cGMP production via sGC activation in platelets. cGMP then activates protein kinase G (PKG) which phosphorylates numerous proteins related to platelet function resulting in inhibition of platelet responses to its agonist. However, biological effects of NO generated from the serial reduction of nitrate or nitrite on platelet function are not well studied yet. Although nitrite and nitrate are the major oxidation products of NO metabolism, the reduction pathway of nitrate and nitrite to NO has been recognized only in the last decade. Our diet is a major source for nitrite and nitrate since one serving (~ 300g) of a nitrate-rich vegetable such as spinach or beetroot is known to contain more nitrate than what is produced endogenously over a day by all three NOS enzymes44,45. Absorbed nitrate can be reduced to nitrite by bacterial nitrate reductases in the oral cavity and nitrite can be further reduced to NO in blood and tissues by several pathways 46 including deoxyhemoglobin, deoxymyoglobin, xanthine oxidase, mitochondrial respiratory chain enzymes, cytochrome P-450 and non-enzymatic reduction in the presence of protons or vitamin C. Webb et al. recently showed that oral intake of nitrate rich food such as beetroot juice decreased platelet aggregation 29 suggesting NO from exogenous sources might have a great effect on platelet function. Considering the potential effect of nitrite and nitrate on platelet function, it is important to study NO metabolism during platelet storage since changes in levels of nitrite and nitrate during storage could modulate platelet function and/or affect post-transfusion viability.
To better understand platelet physiology during storage, in this study, we examined the levels of NO derivatives, nitrite and nitrate, under routine storage conditions. We stored apheresis platelet aliquots in standard CLX containers, with the same surface to volume ratio and similar platelet counts as those in whole blood derived PLTs. CLX plastic is composed of PVC and the plasticizer triethylhexyl trimellitate 47. It has been known that plastic materials of storage containers are of great importance for regulation of gas exchange and maintenance of in vitro and in vivo platelet properties 48. Nitrite or other NO precursors are used in the processes of synthesizing polymeric matrices 49–51 and are known to be slowly released from the final products. Therefore we examined whether nitrite is also released from platelet storage containers. As shown in Fig. 2, nitrite leaching was prominent in CLX, but absent in PL120 Teflon container. CLX and PL120 containers are made of different plastics, PVC and PTFE respectively, and PL120 does not contain any plasticizer. It seems that the chemical composition of each plastic material affects nitrite leaching levels.
There is no published information available on the levels of NO metabolites during platelet storage. In Fig. 1A, we observed an increase in nitrite level in both PLT supernatant and CP at a rate of 31 nM/day and 58 nM/day, respectively. We believe that this increase is caused by nitrite leaching from the material of the CLX containers during storage, based on our result in Fig. 2. However, the increase rate is much smaller in PLT supernatant (31 nM/day) than in CP (58 nM/day). In addition, we observed that nitrite level slightly decreased at a rate of 12.5 nM/day in the supernatant of PLT aliquots stored in PL120 Teflon container which does not leach nitrite (Fig. 1C). These results imply that active platelets, residual leukocytes and/or contaminating erythrocytes in PLT utilized nitrite during storage. Although erythrocyte and leukocyte counts were significantly reduced in PLT preparations compared to those of whole blood, there were 1.3×106 erythrocytes and 2.0×104 leukocytes present in 60ml of 7.8×1010 PLT.
Srihirun et al. showed that nitrite could inhibit platelet aggregation and activation measured by changes in impedance and CD62-positive platelets, respectively, in the presence of 20% Hct erythrocyte through its reduction to NO and this was promoted by deoxygenation 30. In our study, it is not likely that deoxyhemoglobin in the residual erythrocytes of the PLT is responsible for the reduction of nitrite to NO because pO2 was maintained constantly high throughout the entire storage period with values of 148 ± 11 mmHg at day 0 and 147 ± 26 mmHg at day 4. The reaction between nitrite and oxyhemoglobin which generates nitrate and methemoglobin is possible and this might have contributed to nitrite decrease in PLT during storage. Although Arora et al. reported that cytochrome P-450 reductase plays a role as a nitrite reductase in human platelets 52, they used non-physiological concentrations of nitrite (~ 100 µM) in order to observe NO generation and we observed no evidence that higher concentrations of nitrite than those normally present in plasma affected NO production in PLT. Therefore there is no substantial evidence so far that human platelets or leukocytes express specific nitrite reductases under physiological conditions. Further study should be performed to clarify the mechanism for the nitrite utilization in apheresis platelet units. The small amount of reduction of nitrate levels in PLT stored in CLX container is also at present unexplained but this may be a part of serial reduction pathways converting nitrate to other NO metabolites.
Although we could not quantitate the low levels of NO that appear to be produced by the stored PLT, our present study shows a clear difference between PLT and CP in NO levels during storage in PL120 Teflon containers (Fig. 3A). These likely reflect the above mentioned reductive pathways rather than an enzymatic production of NO by NOS. Based on previous reports on the absence of active NOS in platelets 13–15 and our result suggesting no effect of L-Arginine and L-NAME on NO production (data not shown), contribution of endogenous NOS to NO production in PLT during storage seems to be very little.
Collectively, our current study shows that: 1) NO metabolite levels are measurable in apheresis platelet units as shown by nitrite consumption by PLT stored in Teflon containers where nitrite decreased over storage time 2) platelet preparations are unintentionally enriched by nitrite during routine storage in CLX containers by nitrite release from the plastic materials but this enrichment does not appear to be detrimental to PLT. However, NO which may be generated in vivo after platelet transfusion by the reduction of nitrite that was leached from the platelet container could modify the eventual function of transfused and endogenous platelets.
Acknowledgements
The authors thank Dr Gary Moroff for his thoughtful comments and suggestions about this study.
Research support from Molecular Medicine Branch, NIDDK, NIH and Blood Components Department, American Red Cross
Abbreviations
- PLT(s)
apheresis platelet(s)
- CP
concurrent plasma
- NO
nitric oxide
- NOS
nitric oxide synthase
- EPR
electron paramagnetic resonance
Footnotes
Disclaimers: There are no disclaimers to share.
Conflict of Interest: A.N. Schechter is a co-inventor of a patent issued to NIH for the use of sodium nitrite in the treatment of cardiovascular diseases. The authors declare that they have no conflicts of interest relevant to the manuscript submitted to TRANSFUSION.
References
- 1.Ignarro LJ, Byrns RE, Buga GM, Wood KS. Endothelium-derived relaxing factor from pulmonary artery and vein possesses pharmacologic and chemical properties identical to those of nitric oxide radical. Circ Res. 1987;61:866–879. doi: 10.1161/01.res.61.6.866. [DOI] [PubMed] [Google Scholar]
- 2.Palmer RM, Ferrige AG, Moncada S. Nitric oxide release accounts for the biological activity of endothelium-derived relaxing factor. Nature. 1987;327:524–526. doi: 10.1038/327524a0. [DOI] [PubMed] [Google Scholar]
- 3.Palmer RM, Ashton DS, Moncada S. Vascular endothelial cells synthesize nitric oxide from L-arginine. Nature. 1988;333:664–666. doi: 10.1038/333664a0. [DOI] [PubMed] [Google Scholar]
- 4.Azuma H, Ishikawa M, Sekizaki S. Endothelium-dependent inhibition of platelet aggregation. Br J Pharmacol. 1986;88:411–415. doi: 10.1111/j.1476-5381.1986.tb10218.x. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 5.Gkaliagkousi E, Ferro A. Nitric oxide signalling in the regulation of cardiovascular and platelet function. Front Biosci. 2011;16:1873–1897. doi: 10.2741/3828. [DOI] [PubMed] [Google Scholar]
- 6.Antl M, von Bruhl ML, Eiglsperger C, Werner M, Konrad I, Kocher T, Wilm M, Hofmann F, Massberg S, Schlossmann J. IRAG mediates NO/cGMP-dependent inhibition of platelet aggregation and thrombus formation. Blood. 2007;109:552–559. doi: 10.1182/blood-2005-10-026294. [DOI] [PubMed] [Google Scholar]
- 7.Cavallini L, Coassin M, Borean A, Alexandre A. Prostacyclin and sodium nitroprusside inhibit the activity of the platelet inositol 1,4,5-trisphosphate receptor and promote its phosphorylation. J Biol Chem. 1996;271:5545–5551. doi: 10.1074/jbc.271.10.5545. [DOI] [PubMed] [Google Scholar]
- 8.Trepakova ES, Cohen RA, Bolotina VM. Nitric oxide inhibits capacitative cation influx in human platelets by promoting sarcoplasmic/endoplasmic reticulum Ca2+-ATPase-dependent refilling of Ca2+ stores. Circ Res. 1999;84:201–209. doi: 10.1161/01.res.84.2.201. [DOI] [PubMed] [Google Scholar]
- 9.Hauser W, Knobeloch KP, Eigenthaler M, Gambaryan S, Krenn V, Geiger J, Glazova M, Rohde E, Horak I, Walter U, Zimmer M. Megakaryocyte hyperplasia and enhanced agonist-induced platelet activation in vasodilator-stimulated phosphoprotein knockout mice. Proc Natl Acad Sci U S A. 1999;96:8120–8125. doi: 10.1073/pnas.96.14.8120. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 10.Roberts W, Michno A, Aburima A, Naseem KM. Nitric oxide inhibits von Willebrand factor-mediated platelet adhesion and spreading through regulation of integrin alpha(IIb)beta(3) and myosin light chain. J Thromb Haemost. 2009;7:2106–2115. doi: 10.1111/j.1538-7836.2009.03619.x. [DOI] [PubMed] [Google Scholar]
- 11.de Graaf JC, Banga JD, Moncada S, Palmer RM, de Groot PG, Sixma JJ. Nitric oxide functions as an inhibitor of platelet adhesion under flow conditions. Circulation. 1992;85:2284–2290. doi: 10.1161/01.cir.85.6.2284. [DOI] [PubMed] [Google Scholar]
- 12.Radomski MW, Palmer RM, Moncada S. An L-arginine/nitric oxide pathway present in human platelets regulates aggregation. Proc Natl Acad Sci U S A. 1990;87:5193–5197. doi: 10.1073/pnas.87.13.5193. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 13.Ozuyaman B, Godecke A, Kusters S, Kirchhoff E, Scharf RE, Schrader J. Endothelial nitric oxide synthase plays a minor role in inhibition of arterial thrombus formation. Thromb Haemost. 2005;93:1161–1167. doi: 10.1160/TH03-09-0588. [DOI] [PubMed] [Google Scholar]
- 14.Gambaryan S, Kobsar A, Hartmann S, Birschmann I, Kuhlencordt PJ, Muller-Esterl W, Lohmann SM, Walter U. NO-synthase-/NO-independent regulation of human and murine platelet soluble guanylyl cyclase activity. J Thromb Haemost. 2008;6:1376–1384. doi: 10.1111/j.1538-7836.2008.03014.x. [DOI] [PubMed] [Google Scholar]
- 15.Tymvios C, Moore C, Jones S, Solomon A, Sanz-Rosa D, Emerson M. Platelet aggregation responses are critically regulated in vivo by endogenous nitric oxide but not by endothelial nitric oxide synthase. Br J Pharmacol. 2009;158:1735–1742. doi: 10.1111/j.1476-5381.2009.00408.x. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 16.Weitzberg E, Lundberg JO. Nonenzymatic nitric oxide production in humans. Nitric Oxide. 1998;2:1–7. doi: 10.1006/niox.1997.0162. [DOI] [PubMed] [Google Scholar]
- 17.Zweier JL, Samouilov A, Kuppusamy P. Non-enzymatic nitric oxide synthesis in biological systems. Biochim Biophys Acta. 1999;1411:250–262. doi: 10.1016/s0005-2728(99)00018-3. [DOI] [PubMed] [Google Scholar]
- 18.Gladwin MT, Schechter AN, Kim-Shapiro DB, Patel RP, Hogg N, Shiva S, Cannon RO, 3rd, Kelm M, Wink DA, Espey MG, Oldfield EH, Pluta RM, Freeman BA, Lancaster JR, Jr, Feelisch M, Lundberg JO. The emerging biology of the nitrite anion. Nat Chem Biol. 2005;1:308–314. doi: 10.1038/nchembio1105-308. [DOI] [PubMed] [Google Scholar]
- 19.Lundberg JO, Weitzberg E, Gladwin MT. The nitrate-nitrite-nitric oxide pathway in physiology and therapeutics. Nat Rev Drug Discov. 2008;7:156–167. doi: 10.1038/nrd2466. [DOI] [PubMed] [Google Scholar]
- 20.Duncan C, Dougall H, Johnston P, Green S, Brogan R, Leifert C, Smith L, Golden M, Benjamin N. Chemical generation of nitric oxide in the mouth from the enterosalivary circulation of dietary nitrate. Nat Med. 1995;1:546–551. doi: 10.1038/nm0695-546. [DOI] [PubMed] [Google Scholar]
- 21.Cosby K, Partovi KS, Crawford JH, Patel RP, Reiter CD, Martyr S, Yang BK, Waclawiw MA, Zalos G, Xu X, Huang KT, Shields H, Kim-Shapiro DB, Schechter AN, Cannon RO, 3rd, Gladwin MT. Nitrite reduction to nitric oxide by deoxyhemoglobin vasodilates the human circulation. Nat Med. 2003;9:1498–1505. doi: 10.1038/nm954. [DOI] [PubMed] [Google Scholar]
- 22.Nagababu E, Ramasamy S, Abernethy DR, Rifkind JM. Active nitric oxide produced in the red cell under hypoxic conditions by deoxyhemoglobin-mediated nitrite reduction. J Biol Chem. 2003;278:46349–46356. doi: 10.1074/jbc.M307572200. [DOI] [PubMed] [Google Scholar]
- 23.Shiva S, Huang Z, Grubina R, Sun J, Ringwood LA, MacArthur PH, Xu X, Murphy E, Darley-Usmar VM, Gladwin MT. Deoxymyoglobin is a nitrite reductase that generates nitric oxide and regulates mitochondrial respiration. Circ Res. 2007;100:654–661. doi: 10.1161/01.RES.0000260171.52224.6b. [DOI] [PubMed] [Google Scholar]
- 24.Rassaf T, Flogel U, Drexhage C, Hendgen-Cotta U, Kelm M, Schrader J. Nitrite reductase function of deoxymyoglobin: oxygen sensor and regulator of cardiac energetics and function. Circ Res. 2007;100:1749–1754. doi: 10.1161/CIRCRESAHA.107.152488. [DOI] [PubMed] [Google Scholar]
- 25.Zhang Z, Naughton DP, Blake DR, Benjamin N, Stevens CR, Winyard PG, Symons MC, Harrison R. Human xanthine oxidase converts nitrite ions into nitric oxide (NO) Biochem Soc Trans. 1997;25:524S. doi: 10.1042/bst025524s. [DOI] [PubMed] [Google Scholar]
- 26.Millar TM, Stevens CR, Benjamin N, Eisenthal R, Harrison R, Blake DR. Xanthine oxidoreductase catalyses the reduction of nitrates and nitrite to nitric oxide under hypoxic conditions. FEBS Lett. 1998;427:225–228. doi: 10.1016/s0014-5793(98)00430-x. [DOI] [PubMed] [Google Scholar]
- 27.Godber BL, Doel JJ, Sapkota GP, Blake DR, Stevens CR, Eisenthal R, Harrison R. Reduction of nitrite to nitric oxide catalyzed by xanthine oxidoreductase. J Biol Chem. 2000;275:7757–7763. doi: 10.1074/jbc.275.11.7757. [DOI] [PubMed] [Google Scholar]
- 28.Carlsson S, Wiklund NP, Engstrand L, Weitzberg E, Lundberg JO. Effects of pH, nitrite, and ascorbic acid on nonenzymatic nitric oxide generation and bacterial growth in urine. Nitric Oxide. 2001;5:580–586. doi: 10.1006/niox.2001.0371. [DOI] [PubMed] [Google Scholar]
- 29.Webb AJ, Patel N, Loukogeorgakis S, Okorie M, Aboud Z, Misra S, Rashid R, Miall P, Deanfield J, Benjamin N, MacAllister R, Hobbs AJ, Ahluwalia A. Acute blood pressure lowering, vasoprotective, and antiplatelet properties of dietary nitrate via bioconversion to nitrite. Hypertension. 2008;51:784–790. doi: 10.1161/HYPERTENSIONAHA.107.103523. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 30.Srihirun S, Sriwantana T, Unchern S, Kittikool D, Noulsri E, Pattanapanyasat K, Fucharoen S, Piknova B, Schechter AN, Sibmooh N. Platelet inhibition by nitrite is dependent on erythrocytes and deoxygenation. PLoS One. 2012 doi: 10.1371/journal.pone.0030380. In Press. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 31.Seghatchian J, Krailadsiri P. The platelet storage lesion. Transfus Med Rev. 1997;11:130–144. doi: 10.1053/tm.1997.0110130. [DOI] [PubMed] [Google Scholar]
- 32.Holme S. Storage and quality assessment of platelets. Vox Sang. 1998;74(Suppl 2):207–216. doi: 10.1111/j.1423-0410.1998.tb05422.x. [DOI] [PubMed] [Google Scholar]
- 33.Holme S, Moroff G, Murphy S. A multi-laboratory evaluation of in vitro platelet assays: the tests for extent of shape change and response to hypotonic shock. Biomedical Excellence for Safer Transfusion Working Party of the International Society of Blood Transfusion. Transfusion. 1998;38:31–40. doi: 10.1046/j.1537-2995.1998.38198141495.x. [DOI] [PubMed] [Google Scholar]
- 34.Perrotta PL, Perrotta CL, Snyder EL. Apoptotic activity in stored human platelets. Transfusion. 2003;43:526–535. doi: 10.1046/j.1537-2995.2003.00349.x. [DOI] [PubMed] [Google Scholar]
- 35.Ohto H, Nollet KE. Overview on platelet preservation: better controls over storage lesion. Transfus Apher Sci. 2011;44:321–325. doi: 10.1016/j.transci.2011.03.008. [DOI] [PubMed] [Google Scholar]
- 36.Klinger MH. The storage lesion of platelets: ultrastructural and functional aspects. Ann Hematol. 1996;73:103–112. doi: 10.1007/s002770050210. [DOI] [PubMed] [Google Scholar]
- 37.Yang BK, Vivas EX, Reiter CD, Gladwin MT. Methodologies for the sensitive and specific measurement of S-nitrosothiols, iron-nitrosyls, and nitrite in biological samples. Free Radic Res. 2003;37:1–10. doi: 10.1080/1071576021000033112. [DOI] [PubMed] [Google Scholar]
- 38.Basu S, Wang X, Gladwin MT, Kim-Shapiro DB. Chemiluminescent detection of S-nitrosated proteins: comparison of tri-iodide, copper/CO/cysteine, and modified copper/cysteine methods. Methods Enzymol. 2008;440:137–156. doi: 10.1016/S0076-6879(07)00808-7. [DOI] [PubMed] [Google Scholar]
- 39.Joseph J, Kalyanaraman B, Hyde JS. Trapping of nitric oxide by nitronyl nitroxides: an electron spin resonance investigation. Biochem Biophys Res Commun. 1993;192:926–934. doi: 10.1006/bbrc.1993.1504. [DOI] [PubMed] [Google Scholar]
- 40.Hogg N, Singh RJ, Joseph J, Neese F, Kalyanaraman B. Reactions of nitric oxide with nitronyl nitroxides and oxygen: prediction of nitrite and nitrate formation by kinetic simulation. Free Radic Res. 1995;22:47–56. doi: 10.3109/10715769509147527. [DOI] [PubMed] [Google Scholar]
- 41.Mellion BT, Ignarro LJ, Ohlstein EH, Pontecorvo EG, Hyman AL, Kadowitz PJ. Evidence for the inhibitory role of guanosine 3', 5'-monophosphate in ADP-induced human platelet aggregation in the presence of nitric oxide and related vasodilators. Blood. 1981;57:946–955. [PubMed] [Google Scholar]
- 42.Schafer AI, Alexander RW, Handin RI. Inhibition of platelet function by organic nitrate vasodilators. Blood. 1980;55:649–654. [PubMed] [Google Scholar]
- 43.Radomski MW, Palmer RM, Moncada S. Comparative pharmacology of endothelium-derived relaxing factor, nitric oxide and prostacyclin in platelets. Br J Pharmacol. 1987;92:181–187. doi: 10.1111/j.1476-5381.1987.tb11310.x. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 44.Lundberg JO, Weitzberg E. The biological role of nitrate and nitrite: the times they are a-changin'. Nitric Oxide. 2010;22:61–63. doi: 10.1016/j.niox.2009.11.004. [DOI] [PubMed] [Google Scholar]
- 45.Govoni M, Jansson EA, Weitzberg E, Lundberg JO. The increase in plasma nitrite after a dietary nitrate load is markedly attenuated by an antibacterial mouthwash. Nitric Oxide. 2008;19:333–337. doi: 10.1016/j.niox.2008.08.003. [DOI] [PubMed] [Google Scholar]
- 46.Lundberg JO, Weitzberg E. NO generation from inorganic nitrate and nitrite: Role in physiology, nutrition and therapeutics. Arch Pharm Res. 2009;32:1119–1126. doi: 10.1007/s12272-009-1803-z. [DOI] [PubMed] [Google Scholar]
- 47.Carmen R. The selection of plastic materials for blood bags. Transfus Med Rev. 1993;7:1–10. doi: 10.1016/s0887-7963(93)70027-9. [DOI] [PubMed] [Google Scholar]
- 48.Murphy S. Platelet storage for transfusion. Semin Hematol. 1985;22:165–177. [PubMed] [Google Scholar]
- 49.Mowery KA, Schoenfisch MH, Saavedra JE, Keefer LK, Meyerhoff ME. Preparation and characterization of hydrophobic polymeric films that are thromboresistant via nitric oxide release. Biomaterials. 2000;21:9–21. doi: 10.1016/s0142-9612(99)00127-1. [DOI] [PubMed] [Google Scholar]
- 50.Bicak N, Sherrington DC, Bulbul H. Vinylamine polymer via chemical modification of PVC. European Polymer Journal. 2001;37:801–805. [Google Scholar]
- 51.Zhang H, Annich GM, Miskulin J, Osterholzer K, Merz SI, Bartlett RH, Meyerhoff ME. Nitric oxide releasing silicone rubbers with improved blood compatibility: preparation, characterization, and in vivo evaluation. Biomaterials. 2002;23:1485–1494. doi: 10.1016/s0142-9612(01)00274-5. [DOI] [PubMed] [Google Scholar]
- 52.Arora S, Tyagi YK, Kumar A, Majumder S, Saluja D, Raj HG, Chatterjee S, Saso L, Prasad AK, Parmar VS. The role of calreticulin transacetylase in the activation of human platelet nitrite reductase by polyphenolic acetates. Biol Pharm Bull. 2009;32:161–165. doi: 10.1248/bpb.32.161. [DOI] [PubMed] [Google Scholar]
- 53.Schedel A, Rolf N. Genome-wide platelet RNA profiling in clinical samples. Methods Mol Biol. 2009;496:273–283. doi: 10.1007/978-1-59745-553-4_17. [DOI] [PubMed] [Google Scholar]