Abstract
Background
Arcobacter spp. (family Campylobacteraceae) are ubiquitous zoonotic bacteria that are being increasingly recognised as a threat to human health. A previously published 16S rRNA-RFLP Arcobacter spp. identification method produced specific RFLP patterns for the six species described at that time, using a single endonuclease (MseI). The number of characterised Arcobacter species has since risen to 17. The aim of the present study was to update the 16S rRNA-RFLP identification method to include all currently characterised species of Arcobacter.
Results
Digestion of the 16S rRNA gene with the endonuclease MseI produced clear, distinctive patterns for 10 of the 17 species, while the remaining species shared a common or very similar RFLP pattern. Subsequent digestion of the 16S rRNA gene from these species with the endonucleases MnlI and/or BfaI generated species-specific RFLP patterns.
Conclusions
16S rRNA-RFLP analysis identified 17 Arcobacter spp. using either polyacrylamide or agarose gel electrophoresis. Microheterogeneities within the 16S rRNA gene, which interfered with the RFLP identification, were also documented for the first time in this genus, particularly in strains of Arcobacter cryaerophilus isolated from animal faeces and aborted foetuses.
Keywords: Arcobacter, Identification, Agarose, Polyacrylamide, 16S rRNA-RFLP, 16S rRNA gene mutations
Background
The genus Arcobacter, included in the family Campylobacteraceae, has expanded rapidly since it was first recognised in 1991 [1], and currently includes 17 species. Some of these species are considered enteropathogenic to humans and animals, as well as important zoonotic agents. Arcobacter species negatively impact the food industry, as many meat products are frequently contaminated with these bacteria, and multiple species have been described from shellfish [2-6]. In addition, the International Commission on Microbiological Specification for Foods classified A. butzleri as a serious hazard to human health [7]. However, the true incidence of Arcobacter species in environmental and clinical samples is thought to be underestimated because specific detection and identification methods are not normally applied and can be inaccurate [2,8].
A 16S rRNA restriction fragment length polymorphism (RFLP) method for the identification of Arcobacter species has previously been described [9]. The method involved a single digestion with the MseI endonuclease and discriminated all Arcobacter species that had been described up to 2008, i.e. A. butzleri, A. cryaerophilus, A. cibarius, A. skirrowii, A. nitrofigilis and A. halophilus[9]. Further molecular methods for the identification of Arcobacter species have been reviewed elsewhere [2,9]. Most of the methods described target only the most common species i.e. A. butzleri[10,11], A. cryaerophilus[12] and/or A. skirrowii[13,14]. Even the most recently proposed identification method, m-PCR, described by Douidah et al. [15] in 2010, only targeted five species: A. butzleri, A. cryaerophilus, A. skirrowii, A. cibarius and A. thereius. Furthermore, using this method, the species A. defluvii, A. ellisii, A. venerupis and A. butzleri produced an identical and therefore uninformative amplicon [2,5,6].
The limitations of the current methods have arisen because of the limited testing of certain species, as well as the identification of novel species [2,4-6]. Douidah et al.[15] suggested that the reliance of the currently-available 16S rRNA-RFLP method on polyacrylamide gel electrophoresis was a major disadvantage for its routine use. Furthermore, the recently described species A. thereius, isolated from aborted pig foetuses [16], and A. trophiarum, which was recovered from porcine faecal matter [17], produce the same RFLP pattern as A. butzleri[2]. Additionally, the new species A. venerupis, from clams, produces a pattern that is very similar to A. marinus[6,18].
The aim of the present study was to update the 16S rRNA-RFLP identification method to include all the currently characterised species of Arcobacter, and to provide protocols for both polyacrylamide and agarose gel electrophoresis so that the method can easily be adapted.
Results
MseI digestion can discriminate 10 of the 17 currently described Arcobacter species
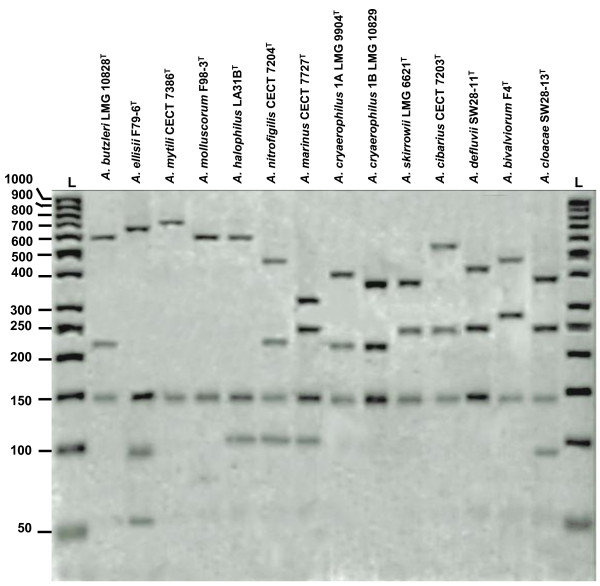
Following digestion with the endonuclease MseI, species-specific differential RFLP patterns were obtained for 47 of the 121 strains (38.8%), representing 12 of the 17 species that make up the Arcobacter genus (A. nitrofigilis, A. cryaerophilus, A. skirrowii, A. cibarius, A. halophilus, A. mytili, A. marinus, A. molluscorum, A. ellisii, A. bivalviorum and A. venerupis), including the new described species A. cloacae (Figure 1 and Table 1). However, A. venerupis produced a pattern very similar to that of A. marinus, with only a single 141 bp band distinguishing the two species (Figure 4 and Additional file 1: Table S1). In addition, the new species A. suis (F41) showed the same banding pattern as A. defluvii, while the characteristic A. butzleri pattern (Figure 4 and Additional file 1: Table S1) was also observed following MseI digestion of A. thereius and A. trophiarum and 11 of the 19 (57.9%) A. cryaerophilus strains. Of these, nine strains (MICV1-1, MICV3-2, FE4, FE5, FE6, FE9, FE11, FE13 and FE14) were isolated from animal faeces in Valdivia, Chile, and two strains were isolated in Ireland (LMG 9863 and LMG 9871) from aborted ovine and bovine foetuses, respectively. The RFLP results for these 11 strains were discordant with those of m-PCR and their identity was confirmed by sequencing the 16S rRNA and rpoB genes.
Figure 1.
16S rRNA-RFLP patterns (agarose gel 3.5%) obtained for Arcobacter spp. using the endonuclease MseI. Lanes: L, 50 bp ladder, Fermentas. The obtained patterns agree with those expected from the computer simulation (Additional file 1: Table S1). Species that share an identical or similar pattern (Additional file 1: Table S1) were: A. butzleri, that produced a pattern identical to those of A. trophiarum, A. thereius and atypical strains (n=11) of A. cryaerophilus; A. marinus CECT 7727T with a pattern very similar to the one of A. venerupis CECT 7836T and A. defluvii with an identical pattern to the one of A. suis strain F41. The identification of these species required additional digestions with other enzymes (Figures 2 – 4, Additional file 2: Table S2 and Additional file 3: Table S3).
Table 1.
Arcobacter spp. strains used in this study
| SPECIES | STRAIN | SOURCE |
|---|---|---|
|
A. butzleri |
LMG 10828T,¶,Ω, LMG 11118Ω |
Human faeces |
| |
W24-2-1, W24-05-1, W07-01-8, W03-03-6, W26-02-2, W03-02-7, W21-05-1, W2105-3, W21-05-7, W24-01-1, W10-01-1 |
Sea water |
| |
SWDS1-3-2 |
Sewage |
| |
F42, F46Ω, F49, F51 |
Pork meat |
| |
F15, F22, F23, F24, F25 |
Turkey meat |
| |
F44, F47, F52 |
Chicken meat |
| |
F43, F50Ω, F53 |
Beef meat |
| |
F1, F2, F29, F30, F38, F98-1, SAN600-1,SAN600-6, SAN512-1, SAN547-10, SAN548-8, SAN582-1, SAN582-6 |
Mussels |
| |
T62 |
Soil |
|
A. trophiarum |
LMG 25534T,¶,Ω, LMG 25535¶,Ω |
Pig faeces |
| |
CECT 7650Ω |
Chicken cloacae |
|
A. thereius |
LMG 24486T,¶,Ω, LMG 24487¶,Ω |
Porcine abortion foetus |
| |
SW24Ω |
Sewage |
| |
F61-1Ω |
Pork meat |
| |
F89-4 |
Mussels |
| |
F93-4Ω |
Clams |
|
A. cryaerophilus |
LMG 9904T,¶,Ω, LMG 9871¶,Ω |
Bovine abortion foetus |
| |
LMG 9865¶,Ω, LMG 10241¶,Ω, LMG 6622, LMG 10229¶,Ω |
Porcine abortion |
| |
LMG 7537¶, LMG 9863¶,Ω |
Ovine abortion foetus |
| |
LMG 10829¶ |
Human blood |
| |
LMG 9861¶,Ω |
Bovine abortion foetus |
| |
FE4Ω, FE5¶,Ω, FE6¶,Ω, FE9¶,Ω, FE11Ω, FE13Ω |
Chicken cloacal swabs |
| |
FE14Ω |
Ovine faeces |
| |
MICV1-1¶,Ω, MICV3-2¶,Ω |
Cow faeces |
|
A. nitrofigilis |
CECT 7204T,¶,Ω, LMG 7547Ω |
Roots of Spartina alterniflora |
| |
F39Ω, F40¶, F72Ω |
Mussels |
|
A. skirrowii |
LMG 6621T,¶,Ω |
Lamb faeces |
| |
LMG 9911 |
Porcine abortion |
| |
Houf 989¶,Ω, Houf 994Ω |
Cow faeces |
| |
S7Ω |
Sludge |
| |
F94-1Ω |
Clams |
| |
F125-1Ω |
Mussels |
| |
ArcoEΩ, ArcoFΩ |
|
|
A. cibarius |
CECT 7203T,¶,Ω |
Chicken meat |
| |
NC81Ω, NC88Ω |
Piggery effluent |
| |
H742, H743Ω, H745, H746Ω, H748 |
Poultry carcasses |
|
A.halophilus |
LA31BT,¶,Ω |
Hypersaline lagoon |
|
A.mytili |
CECT 7386T,¶,Ω, CECT 7385¶,Ω |
Mussels |
| |
T234Ω |
Brackish water |
|
A.marinus |
CECT 7727T,¶,Ω |
Seawater/starfish |
|
A.defluvii |
CECT 7697T,¶,Ω, SW28-7¶,Ω, SW28-8, SW28-9, SW28-10, SW30-2¶,Ω, SW30-7, SW30-8 |
Sewage |
| |
MICCC4-2Ω |
Pig faeces |
| |
SAN599-9Ω |
Mussels |
|
A.molluscorum |
CECT 7696T,¶,Ω, F91¶,Ω, F101-1¶,Ω |
Mussels |
|
A.ellisii |
F79-6T,¶,Ω, F79-2¶,Ω, F79-7¶,Ω |
Mussels |
|
A.bivalviorum |
F4T,¶,Ω, F118-2¶,Ω, F118-4¶,Ω |
Mussels |
|
A.venerupis |
F67-11T,¶,Ω |
Clams |
|
A.suis |
F41T,¶,Ω |
Pork meat |
|
A.cloacae |
SW28-13T,¶,Ω |
Sewage |
| F26¶,Ω | Mussels |
ATCC American Type Culture Collection, LMG Belgian Co-ordinated Collection of Micro-organisms, CECT Colección Española de Cultivos Tipo.
¶ Sequenced 16S rRNA gene.
Ω Sequenced rpoB gene.
Figure 4.
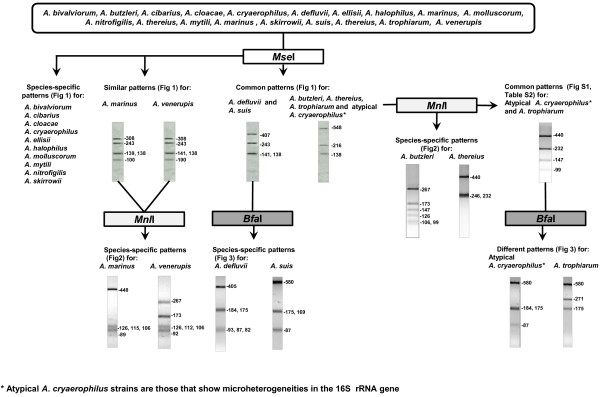
Flow chart illustrating the proposed order of restriction endonuclease digestions for the 16S rRNA–RFLP analysis for the identification of Acrobacter spp.
Microhetergeneities in A. cryaerophilus strains interfere with RFLP identification
The chromatograms of the 16S rRNA gene sequences (1405 bp) of seven of the 11 unresolved A. cryaerophilus strains (MIC V1-1, MICV3-2, FE5, FE6, FE9, LMG 9863 and LMG 9871) showed mutations (i.e. microheterogeneities) at positions 192 (T→C) and 205 (A→G), which were within the target region (TTAA) of the MseI endonuclease (Additional file 4: Figure S1).
Digestion with MnlI and/or BfaI resolves the remaining species
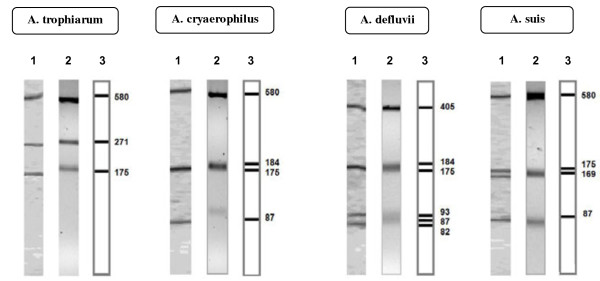
A second restriction digest using MnlI (Fermentas) was then carried out for those strains with common or similar RFLP patterns following MseI digestion (Additional file 1: Table S1 and Additional file 2: Table S2). MnlI generated a species-specific pattern for A. butzleri, A. thereius, A. marinus and A. venerupis, and a common pattern for A. trophiarum and the atypical strains of A. cryaerophilus (Figures 2 and 4). A further restriction digest step using FspBI (Fermentas), an isoschizomer of BfaI, produced species-specific RFLP patterns for the separation of A. defluvii from A. suis (F41), and A. trophiarum from the atypical A. cryaerophilus strains (Figure 3 and Additional file 3: Table S3). After carrying out 16S rRNA gene restriction digests as illustrated in Figure 4, all of the 121 strains were correctly identified.
Figure 2.
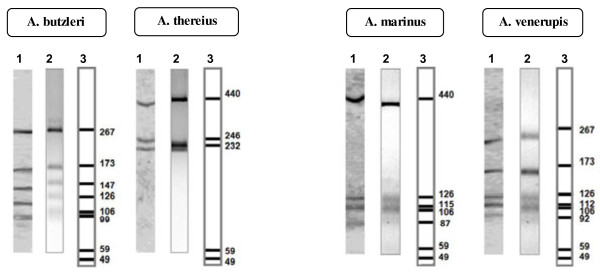
Species-specific 16S rRNA-RFLP patterns for species A.butzleri,A.thereius,A.marinus and A. venerupis, obtained using endonuclease Mnll. 1, polyacrylamide gel 15%; 2, agarose gel 3.5% and 3, computer simulation.
Figure 3.
Species-specific 16S rRNA-RFLP patterns obtained using endonuclease BfaI for A.trophiarum,A.cryaerophilus,A.defluvii and the recently described species A.suis. 1, polyacrylamide gel 15%; 2, agarose gel 3.5% and 3, computer simulation.
Discussion
The proposed 16S rRNA-RFLP method described here used an initial digestion with MseI endonuclease, as in the original method [9], which enabled 10 of the 17 accepted species, including the recently described species A. cloacae, to be identified. Further digestion was necessary to resolve species that showed the MseI digestion pattern of A. butzleri (also common to A. thereius, A. trophiarum and to the atypical strains of A. cryaerophilus with 16S rRNA gene microheterogeneities). Computer simulation revealed that two endonucleases, MnlI and TasI, produced discriminative patterns between the species A. butzleri and A. thereius (Figure 2 and Additional file 5: Figure S2). Furthermore, these two enzymes also produced discriminative patterns between A. marinus and A. venerupis (Figure 2), which showed distinctive but very similar patterns following MseI digestion (Figure 4 and Additional file 1: Table S1). MnlI was selected because it generated more distinctive banding patterns, enabling easier discrimination than TasI (Additional file 5: Figure S2). Considering that A. butzleri is a very common species [2,8,19-21], the identification of the majority of strains will normally be obtained with this second (MnlI) endonuclease reaction (Figures 1, 2, 4). In fact, 79.3% of the strains (96/121) included in the current study were correctly identified with this second digestion step.
However, a third digestion, using the enzyme BfaI, was required to distinguish between A. defluvii and the recently described species A. suis and for distinguishing A. trophiarum from the atypical A. cryaerophilus strains following MnlI digestion (Figures 3,4 and Additional file 3: Table S3). The proposed method enables reliable and fast species identification for a large collection of isolates, requiring, at most, digestion of the PCR-amplified 16S rRNA gene (1026 bp) with three restriction endonucleases (MseI, MnlI and/or BfaI).
The original 16S rRNA-RFLP method [9] has been used to identify more than 800 Arcobacter strains recovered from meat products, shellfish and water in various studies [3-6,19-22]. The existing method has also helped to discover new species on the basis of novel RFLP patterns, including A. mytili[3], A. molluscorum[4], A. ellisii[5], A. bivalviorum, A. venerupis[6] and A. cloacae[23]. Furthermore, as well as identifying the more common Arcobacter species, this technique has confirmed the presence of other rare species in atypical habitats, such A. nitrofigilis in mussels and A. thereius in pork meat [20]. The updated technique described here is likely to supersede the current method in all of these areas.
The use of the 16S rRNA-RFLP method in parallel with the more commonly used molecular identification method, m-PCR [13], as well as the fact that strains with incongruent results were sequenced (rpoB and/or 16S rRNA gene sequencing), ensured accurate species identification, and highlighted the limitations of both identification methods [2,4-6,23]. The presence of microheterogeneities in the 16S rRNA gene, as in the case of the 11 atypical A. cryaerophilus strains, had not previously been observed. These strains produced the m-PCR amplicon expected for A. cryaerophilus, which targets the 23S rRNA gene [13], but showed the A. butzleri 16S rRNA-RFLP pattern [9]. However, rpoB and 16S rRNA gene sequencing results confirmed these strains as A. cryaerophilus. 16S rRNA-RFLP patterns that differ from those described here can be expected for any newly discovered Arcobacter species [3-6,9,23]. Nevertheless, intra-species nucleotide diversity (i.e. mutations or microheterogeneities in the operon copies of the 16S rRNA gene) at the endonuclease cleavage sites can also generate a novel RFLP pattern for a given isolate, or result in a pattern identical to another species [9,24,25]. In the latter situation, misidentifications may occur, as described here.
Conclusions
In conclusion, the 16S rRNA-RFLP protocols described here for the identification of Arcobacter spp. can be carried out using either agarose or polyacrylamide gel electrophoresis (Figures 1–3, Additional file 1: Table S1, Additional file 2: Table S2, Additional file 3: Table S3), depending on the requirements of an individual laboratory. It is important, however, to carry out the 16S rRNA gene digestions in the order illustrated in the flow chart (Figure 4).
The method provided in this study is reproducible, reliable, simple, fast, and reasonably inexpensive, and can be carried out efficiently in any laboratory. The technique is highly applicable for investigations of the prevalence of arcobacters in a variety of food products, water, wastewater or other environmental samples. It will enable investigators to determine the true incidence of the recently described species A. mytili, A. marinus, A. trophiarum, A. molluscorum, A. defluvii, A. ellisii, A. bivalviorum, A. venerupis, A. cloacae and A. suis clarifying their prevalence and epidemiology.
Methods
Bacterial strains and culture conditions
A group of 121 Arcobacter strains isolated from diverse origins were used in this study, including the type strains of the 17 Arcobacter species, as well as strains included in the original descriptions of all species (Table 1). Strains belonging to the most recently described Arcobacter species (A. cloacae, n=2, and A. suis, n=1) [23] were also included in the analysis.
All Arcobacter strains were cultured in TSA supplemented with 5% sheep blood at 30°C under aerobic conditions for 48 h in preparation for DNA extraction.
Strain identification by RFLP
All strains were identified in parallel using the 16S rRNA-RFLP method described by Figueras et al. [9] and the m-PCR method of Houf et al.[13]. Furthermore, the identities of some strains, especially those that gave either an unknown RFLP pattern, or contradictory results between the two methods (16S rRNA-RFLP and m-PCR), were confirmed by sequencing the 16S rRNA and/or the rpoB genes (Table 1) using primers and conditions described previously [3,26].
For the RFLP identification, total genomic DNA was extracted from all strains and used as template for the PCR amplification of a 1026 bp region of the 16S rRNA gene, as previously described [9,27]. 16S rRNA amplicons were digested with TruI (Fermentas, Vilnius, Lithuania), an isoschizomer of MseI, in a 30 μl final volume containing 10 μl of the amplification product, 10 U of the enzyme, 2 μl of 10× buffer, and distilled water. The reaction mixture was incubated at 65°C for 4 h. To separate the restriction fragments, the digested products were electrophoresed on 15% polyacrylamide gels (ProtoGel, Hessle, United Kingdom) at 350 V for 5 h [9], and on 3.5% agarose gels at 100 V for 90 min. In both cases, gels were prepared in 1× Tris-Borate-EDTA (TBE) buffer, and 50 bp ladder (Fermentas) was used as a molecular weight marker. Gels were stained with either SYBR Safe (Invitrogen, Carlsbad, CA, USA) or Red Safe (Ecogen, Barcelona, Spain) DNA gel stains, according to the manufacturers’ instructions, and then photographed on a UV transilluminator Vilber Lourmat Model TFX-35C (Marne-la-Vallée, France).
Determination of restriction endonuclease recognition sites
Restriction endonuclease recognitions sites within the 16S rRNA sequences of all strains included in this study (Table 1 and Additional file 1: Table S1, Additional file 2: Table S2, Additional file 3: Table S3) were identified using NEBcutter V 2.0 software [28], which is available online (http://tools.neb.com/NEBcutter2/index.php). Experimental validation of the selected enzymes was carried out following the manufacturers’ instructions, under the conditions described above.
Competing interests
The authors declare that they have no competing interests.
Authors’ contributions
MJF designed the research project, evaluated results and was principal author. LC isolated the nine strains of A. cryaerophilus in Chile and carried out the speciation and 16S rRNA gene mutation analyses. AL carried out the computer simulations, the experimental digestions and participated in the drafting of manuscript under the supervision of LC and MJF. All authors read and approved the final manuscript.
Supplementary Material
Table S1. Computer simulated profiles of Arcobacter spp. 16S rRNA gene (1026 bp) digestion with MseI endonuclease. Species with specific RFLP patterns are in bold.
Figure S1. Microheterogeneities (or mutations) in the 16S rRNA gene of seven atypical A. cryaerophilus strains in relation to the type strain (LMG 9904T), strain LMG 10829 (A. cryaerophilus subgroup 1B) and the type strain ofA. butzleri (LMG 10828T). Sequence alignment of the 16S rRNA gene (positions 190–207 in relation to Escherichia coli) of seven atypical A. cryaerophilus strains showing mutations at positions 192 (T→C) and 205 (A→G), which alter the MseI restriction enzyme recognition site (TTAA). IUPAC code, Y = Pyrimidine (C or T); R = Purine (A or G).
Figure S2. Agarose gel (3.5%) comparing the 16S rRNA-RFLP patterns obtained using endonucleases a\) TasI and b) MnlI for species A. butzleri,A. thereius and A. trophiarum. Lanes 1 and 14, 50 bp ladder (Fermentas); 2, A. butzleri LMG 10828T; 3, A. butzleri F42; 4, A. butzleri F43; 5, A. butzleri F44; 6, A. butzleri F50; 7, A. butzleri LMG 11118; 8, A. thereius LMG 24486T; 9, A. thereius SW24; 10, A. thereius F89-4; 11, A.thereius F93-4 y 12, A.thereius LMG 24487; 13, A. trophiarum CECT 7650 (identical pattern to that of the 11 atypical strains of A. cryaerophilus, Additional file 2: Table S2). MnlI was selected because it produced more distinctive patterns among the species than TasI.
Table S2. Computer simulated profiles of Arcobacter spp.16S rRNA gene (1026 bp) digestion with MnlI endonuclease. Species in bold are those that show a specific RFLP pattern that was not distinguished with MseI.
Table S3. Computer simulated profiles of Arcobacter spp. 16S rRNA gene (1026 bp) digestion with BfaI endonuclease. Species in bold are those that now show a specific RFLP pattern that was not distinguished previously with MseI or MnlI.
Contributor Information
María José Figueras, Email: mariajose.figueras@urv.cat.
Arturo Levican, Email: aalevican@gmail.com.
Luis Collado, Email: luiscollado@uach.cl.
Acknowledgments
The authors thank Dr. Maqsudul Alam (University of Hawaii, Manoa, HI), Dr. Kurt Houf (Ghent University, Belgium), Dr. Nalini Chinivasagam (Animal Research Institute, Queensland, Australia) and Dr. Robert Madden (Queen’s University Belfast, Ireland) for kindly providing Arcobacter strains. AL is thankful to Universitat Rovira i Virgili for a doctoral grant and to CONICYT, Chile, for financial support through Becas Chile. This work was supported in part by the project with reference AGL2011-30461-C02-02 from the Ministerio de Ciencia e Innovación (Spain).
References
- Vandamme P, Falsen E, Rossau R, Hoste B, Segers P, Tytgat R, De Ley J. Revision of Campylobacter, Helicobacter, and Wolinella taxonomy: emendation of generic descriptions and proposal of Arcobacter gen. nov. Int J Syst Bacterio. 1991;41:88–103. doi: 10.1099/00207713-41-1-88. [DOI] [PubMed] [Google Scholar]
- Collado L, Figueras MJ. Taxonomy, epidemiology and clinical relevance of the genus Arcobacter. Clin Microbiol Rev. 2011;24:174–192. doi: 10.1128/CMR.00034-10. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Collado L, Cleenwerck I, Van Trappen S, De Vos P, Figueras MJ. Arcobacter mytili sp. nov., an indoxyl acetate-hydrolysis-negative bacterium isolated from mussels. Int J Syst Evol Microbiol. 2009;59:1391–1396. doi: 10.1099/ijs.0.003749-0. [DOI] [PubMed] [Google Scholar]
- Figueras MJ, Collado L, Levican A, Perez J, Solsona MJ, Yustes C. Arcobacter molluscorum sp. nov., new species isolated from shellfish. Syst Appl Microbiol. 2011;34:105–109. doi: 10.1016/j.syapm.2010.10.001. [DOI] [PubMed] [Google Scholar]
- Figueras MJ, Levican A, Collado L, Inza MI, Yustes C. Arcobacter ellisii sp. nov., isolated from mussels. Syst Appl Microbiol. 2011;34:414–418. doi: 10.1016/j.syapm.2011.04.004. [DOI] [PubMed] [Google Scholar]
- Levican A, Collado L, Aguilar C, Yustes C, Diéguez AL, Romalde JL, Figueras MJ. Arcobacter bivalviorum sp. nov. and Arcobacter venerupis sp. nov., new species isolated from shellfish. Syst Appl Microbiol. 2012;35:133–138. doi: 10.1016/j.syapm.2012.01.002. [DOI] [PubMed] [Google Scholar]
- International Commission on Microbiological Specifications for Foods. Microorganisms in foods 7. Microbiological testing in food safety management. New York, NY: Kluwer Academic/Plenum Publishers; 2002. [Google Scholar]
- Vandenberg O, Dediste A, Houf K, Ibekwem S, Souayah H, Cadranel S, Douat N, Zissis G, Butzler JP, Vandamme P. Arcobacter species in humans. Emerg Infect Dis. 2004;10:1863–1867. doi: 10.3201/eid1010.040241. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Figueras MJ, Collado L, Guarro J. A new 16S rDNA-RFLP method for the discrimination of the accepted species of Arcobacter. Diagn Microbiol Infect Dis. 2008;62:11–15. doi: 10.1016/j.diagmicrobio.2007.09.019. [DOI] [PubMed] [Google Scholar]
- Kärenlampi RI, Tolvanen TP, Hanninen ML. Phylogenetic analysis and PCR-restriction fragment length polymorphism identification of Campylobacter species based on partial groEL gene sequences. J Clin Microbiol. 2004;42:5731–5738. doi: 10.1128/JCM.42.12.5731-5738.2004. [DOI] [PMC free article] [PubMed] [Google Scholar]
- González A, Moreno Y, Gonzalez R, Hernández J, Ferrus MA. Development of a simple and rapid method based on polymerase chain reaction-based restriction fragment length polymorphism analysis to differentiate Helicobacter, Campylobacter, and Arcobacter species. Curr Microbiol. 2006;53:416–421. doi: 10.1007/s00284-006-0168-5. [DOI] [PubMed] [Google Scholar]
- Brightwell G, Mowat E, Clemens R, Boerema J, Pulford DJ, On S. Development of a multiplex and real time PCR assay for the specific detection of Arcobacter butzleri and Arcobacter cryaerophilus. J Microbiol Methods. 2007;68:318–325. doi: 10.1016/j.mimet.2006.09.008. [DOI] [PubMed] [Google Scholar]
- Houf K, Tutenel A, De Zutter L, Van Hoof J, Vandamme P. Development of a multiplex PCR assay for the simultaneous detection and identification of Arcobacter butzleri, Arcobacter cryaerophilus and Arcobacter skirrowii. FEMS Microbiol Lett. 2000;193:89–94. doi: 10.1111/j.1574-6968.2000.tb09407.x. [DOI] [PubMed] [Google Scholar]
- Kabeya H, Kobayashi Y, Maruyama S, Mikami T. Distribution of Arcobacter species among livestock in Japan. Vet Microbiol. 2003;93:153–158. doi: 10.1016/S0378-1135(02)00312-7. [DOI] [PubMed] [Google Scholar]
- Douidah L, De Zutter L, Vandamme P, Houf K. Identification of five human and mammal associated Arcobacter species by a novel multiplex-PCR assay. J Microbiol Methods. 2010;80:281–286. doi: 10.1016/j.mimet.2010.01.009. [DOI] [PubMed] [Google Scholar]
- Houf K, On S, Coenye T, Debruyne L, De Smet S, Vandamme P. Arcobacter thereius sp. nov., isolated from pigs and ducks. Int J Syst Evol Microbiol. 2009;59:2599–2604. doi: 10.1099/ijs.0.006650-0. [DOI] [PubMed] [Google Scholar]
- De Smet S, Vandamme P, De Zutter L, On S, Douidah L, Houf K. Arcobacter trophiarum sp. nov. isolated from fattening pigs. Int J Syst Evol Microbiol. 2011;63:356–36118. doi: 10.1099/ijs.0.022665-0. [DOI] [PubMed] [Google Scholar]
- Kim HM, Hwang CY, Cho BC. Arcobacter marinus sp. nov. Int J Syst Evol Microbiol. 2010;60:531–536. doi: 10.1099/ijs.0.007740-0. [DOI] [PubMed] [Google Scholar]
- Collado L, Inza I, Guarro J, Figueras MJ. Presence of Arcobacter spp. in environmental waters correlates with high levels of fecal pollution. Environ Microbiol. 2008;10:1635–1640. doi: 10.1111/j.1462-2920.2007.01555.x. [DOI] [PubMed] [Google Scholar]
- Collado L, Guarro J, Figueras MJ. Prevalence of Arcobacter in meat and shellfish. J Food Prot. 2009;72:1102–1106. doi: 10.4315/0362-028x-72.5.1102. [DOI] [PubMed] [Google Scholar]
- Collado L, Kasimir G, Perez U, Bosch A, Pinto R, Saucedo G, Huguet JM, Figueras JM. Occurrence and diversity of Arcobacter spp. along the Llobregat river catchment, at sewage effluents and in a drinking water treatment plant. Water Res. 2010;44:3696–3702. doi: 10.1016/j.watres.2010.04.002. [DOI] [PubMed] [Google Scholar]
- Collado L, Levican A, Perez J, Figueras MJ. Arcobacter defluvii sp. nov., isolated from sewage. Int J Syst Evol Microbiol. 2011;61:1895–1901. doi: 10.1099/ijs.0.025668-0. [DOI] [PubMed] [Google Scholar]
- Levican A, Collado L, Figueras MJ. Arcobacter cloacae sp. nov. and Arcobacter suis sp. nov., two new species isolated from food and sewage. Syst Appl Microbiol. in press. [DOI] [PubMed]
- Figueras MJ, Soler L, Chacón MR, Guarro J, Martínez-Murcia AJ. Use of restriction fragment length polymorphism of the PCR-amplified 16S rRNA gene for the identification of Aeromonas spp. J Clin Microbiol. 2000;38:2023–2025. doi: 10.1128/jcm.38.5.2023-2025.2000. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Alperi A, Figueras MJ, Inza I, Martinez-Murcia AJ. Analysis of 16S rRNA gene mutations in a subset of Aeromonas strains and their impact in species delineation. Int Microbiol. 2008;11:185–194. [PubMed] [Google Scholar]
- Martínez-Murcia AJ, Benlloch S, Collins MD. Phylogenetic interrelationships of members of the genera Aeromonas and Plesiomonas as determined by 16S ribosomal DNA sequencing: lack of congruence with results of DNA-DNA hybridizations. Int J Syst Bacteriol. 1992;42:412–421. doi: 10.1099/00207713-42-3-412. [DOI] [PubMed] [Google Scholar]
- Marshall SM, Melito PL, Woodward DL, Johnson WM, Rodgers FG, Mulvey R. Rapid identification of Campylobacter, Arcobacter, and Helicobacter isolates by PCR-restriction fragment length polymorphism analysis of the 16S rRNA gene. J Clin Microbiol. 1999;37:4158–4160. doi: 10.1128/jcm.37.12.4158-4160.1999. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Vincze T, Posfai J, Roberts RJ. NEBcutter: a program to cleave DNA with restriction enzymes. Nucleic Acids Res. 2003;31:3688–3691. doi: 10.1093/nar/gkg526. http://tools.neb.com/NEBcutter2/index.php. [DOI] [PMC free article] [PubMed] [Google Scholar]
Associated Data
This section collects any data citations, data availability statements, or supplementary materials included in this article.
Supplementary Materials
Table S1. Computer simulated profiles of Arcobacter spp. 16S rRNA gene (1026 bp) digestion with MseI endonuclease. Species with specific RFLP patterns are in bold.
Figure S1. Microheterogeneities (or mutations) in the 16S rRNA gene of seven atypical A. cryaerophilus strains in relation to the type strain (LMG 9904T), strain LMG 10829 (A. cryaerophilus subgroup 1B) and the type strain ofA. butzleri (LMG 10828T). Sequence alignment of the 16S rRNA gene (positions 190–207 in relation to Escherichia coli) of seven atypical A. cryaerophilus strains showing mutations at positions 192 (T→C) and 205 (A→G), which alter the MseI restriction enzyme recognition site (TTAA). IUPAC code, Y = Pyrimidine (C or T); R = Purine (A or G).
Figure S2. Agarose gel (3.5%) comparing the 16S rRNA-RFLP patterns obtained using endonucleases a\) TasI and b) MnlI for species A. butzleri,A. thereius and A. trophiarum. Lanes 1 and 14, 50 bp ladder (Fermentas); 2, A. butzleri LMG 10828T; 3, A. butzleri F42; 4, A. butzleri F43; 5, A. butzleri F44; 6, A. butzleri F50; 7, A. butzleri LMG 11118; 8, A. thereius LMG 24486T; 9, A. thereius SW24; 10, A. thereius F89-4; 11, A.thereius F93-4 y 12, A.thereius LMG 24487; 13, A. trophiarum CECT 7650 (identical pattern to that of the 11 atypical strains of A. cryaerophilus, Additional file 2: Table S2). MnlI was selected because it produced more distinctive patterns among the species than TasI.
Table S2. Computer simulated profiles of Arcobacter spp.16S rRNA gene (1026 bp) digestion with MnlI endonuclease. Species in bold are those that show a specific RFLP pattern that was not distinguished with MseI.
Table S3. Computer simulated profiles of Arcobacter spp. 16S rRNA gene (1026 bp) digestion with BfaI endonuclease. Species in bold are those that now show a specific RFLP pattern that was not distinguished previously with MseI or MnlI.