Abstract
Meningiomas constitute ~34% of primary intracranial tumors and are associated with increased mortality in NF2 patients. To evaluate potential medical therapies for these tumors, we have established a quantifiable orthotopic model for NF2-deficient meningiomas. We showed that telomerase-immortalized Ben-Men-1 benign meningioma cells harbored a single nucleotide deletion in NF2 exon 7 and did not express the NF2 protein, merlin. We also demonstrated that AR-42, a pan-histone deacetylase inhibitor, inhibited proliferation of both Ben-Men-1 and normal meningeal cells by increasing expression of p16INK4A, p21CIP1/WAF1, and p27KIP1. Also, AR-42 increased pro-apoptotic Bim expression and decreased anti-apoptotic BclXL levels. However, AR-42 predominantly arrested Ben-Men-1 cells at G2/M, while inducing cell-cycle arrest at G1 in meningeal cells. Consistently, AR-42 substantially decreased the levels of cyclin D1, E, and A, and PCNA in meningeal cells while significantly reducing the expression of cyclin B, important for progression through G2, in Ben-Men-1 cells. In addition, AR-42 decreased Aurora A and B expression. To compare the in vivo efficacies of AR-42 and AR-12, a PDK1 inhibitor, we generated and used luciferase-expressing Ben-Men-1-LucB cells to establish intracranial xenografts that grew over time. While AR-12 treatment moderately slowed tumor growth, AR-42 caused regression of Ben-Men-1-LucB tumors. Importantly, AR-42-treated tumors showed minimal regrowth when xenograft-bearing mice were switched to normal diet. Together, these results suggest that AR-42 is a potential therapy for meningiomas. The differential effect of AR-42 on cell-cycle progression of normal meningeal and meningioma cells may have implications for why AR-42 is well-tolerated while it potently inhibits tumor growth.
Keywords: Meningioma, neurofibromatosis type 2 (NF2), histone deacetylase inhibitor (HDACi), AR-42, AR-12
Introduction
Meningiomas are tumors originating from the meningothelial cells of the arachnoid layer lining the brain and can occur at the convexity, the skull base, and along the spine (1). About 80% of meningiomas are benign (WHO grade I), whereas the remaining are atypical (grade II) and anaplastic (grade III). These tumors cause significant morbidity, including cranial nerve palsy, seizures, and brainstem compression, which may lead to paralysis, aspiration pneumonia, and death. Surgical resection and radiation are current treatment options; however, complete resection of tumors is often difficult, especially for those located along the skull base. Approximately 20% of benign meningiomas recur over ten years, while grade II and grade III tumors possess greater rates of recurrence. Meningiomas can occur sporadically or in patients with neurofibromatosis type 2 (NF2), a genetic disorder characterized by the development of multiple nervous system tumors, including meningiomas and vestibular schwannomas (2). Meningiomas in NF2 patients are associated with disease severity and increased risk of mortality. NF2 patients often have multiple tumors, and their treatment is challenging. Consequently, the development of novel and effective medical therapeutics to treat meningiomas is urgently needed.
NF2 is caused by mutations in the Neurofibromatosis 2 (NF2) gene, which encodes the tumor suppressor protein merlin (3,4). Most NF2-associated meningiomas and about 50–60% of sporadic meningiomas contain NF2 mutations (2). The observation that NF2 mutations are present in both benign and malignant meningiomas suggests that NF2 inactivation may be an early tumorigenic event and that loss of merlin disrupts important signaling pathways, ultimately leading to tumorigenesis. Merlin-deficient meningioma cells exhibit specific characteristics, such as cytoskeletal and cell contact defects, altered cell morphology and growth properties, and susceptibility to senescence (5). In addition, Nf2 inactivation in arachnoidal cells or meningeal precursor cells leads to meningioma formation in mice, further corroborating merlin’s role in tumorigenesis (6,7). While these mouse models await further characterization, additional models that closely mimic the clinical presentation of NF2-deficient benign meningiomas and facilitate efficient quantitation of intracranial tumor growth will enhance therapeutic testing.
Meningioma cell lines are valuable tools to study tumor biology and potential treatments. Several patient-derived cell lines, such as IOMM-Lee and KT21-MG1, have been established from malignant meningiomas, which exhibit complex genetic changes and aggressive features (8–10). Using IOMM-Lee cells and primary meningioma cultures, McCutcheon et al. (11) generated orthotopic xenografts; however, tumor growth was not monitored over time, and primary meningioma cells did not uniformly produce tumors. Baia et al. (12) established intracranial xenografts using luciferase-expressing IOMM-Lee cells and used bioluminescence imaging (BLI) to quantify tumor growth. However, a quantifiable NF2-deficient, benign meningioma model has not been described. While several benign meningioma cell lines have been developed utilizing the HPV E6/E7 or SV40 T-antigen (13,14), transformation by viral oncogenes alters growth signaling and behavior of these cells. Püttmann et al. (15) generated a benign meningioma cell line, Ben-Men-1, from a grade I meningioma using telomerase. Ben-Men-1 cells exhibit characteristics of meningothelial differentiation and lack one copy of chromosome 22, which harbors the NF2 gene. However, the status of the second NF2 allele in Ben-Men-1 cells is not known.
NF2 inactivation in meningiomas and vestibular schwannomas perturbs several signaling pathways, including the AKT pathway (16–20). Previously, we showed that a small-molecule inhibitor of the AKT pathway, AR-12 (formerly OSU-03012), effectively inhibits the growth of NF2-deficient schwannoma cells (21). We also demonstrated that AR-42 (HDAC-42), a pan-histone deacetylase inhibitor (HDACi), potently inhibits schwannoma and meningioma cell proliferation (22,23). In addition to inhibiting the activities of histone deacetylases, which are frequently over-expressed in human cancers (24), AR-42 can also reduce AKT phosphorylation by disrupting the interaction between HDAC6 and protein phosphatase-1 (PP1), enabling free PP1 to dephosphorylate AKT (25). However, the efficacies of these compounds have not been evaluated in an NF2-deficient, benign meningioma model in vivo. Further, the effect of AR-42 in normal meningeal cells has not been examined.
Here, we showed that Ben-Men-1 cells are NF2-deficient. Using luciferase-expressing Ben-Men-1 cells, we established a quantifiable intracranial meningioma model to evaluate AR-42 and AR-12 as potential therapies. Also, we found that AR-42 differentially inhibited cell-cycle progression of normal meningeal and meningioma cells.
Materials and Methods
Cell cultures and drugs
Ben-Men-1 benign meningioma cells have been described (15). Malignant meningioma KT21-MG1 and IOMM-Lee cells (8,9) were kindly provided by Dr. Anita Lal, University of California, San Francisco. HE1-1 is an adenovirus E1-transformed human embryonic kidney cell line (unpublished), and HMS-97 is a human malignant schwannoma cell line (26). All cell lines and primary human meningeal cells (ScienCell) were grown in Dulbecco’s modified eagle medium with 10% fetal bovine serum (Invitrogen). AR-42 and AR-12 were supplied by Arno Therapeutics and formulated into rodent chow (Research Diets) to deliver about 25mg/kg/day and 100mg/kg/day, respectively (21–23). Also, AR-42 was dissolved in dimethylsulfoxide (DMSO) for in vitro experiments.
Mutational analysis
Genomic DNA was extracted from Ben-Men-1 cells using the PureGene® DNA isolation kit (Qiagen). NF2 exons were amplified by polymerase chain reaction (PCR) using Takara ExTaq™ DNA polymerase and primer pairs flanking each exon (27). PCR products were purified using the Qiagen Gel Extraction kit and sequenced from both 5′ and 3′ directions via automated DNA sequencing. The results were confirmed by sequencing PCR products obtained using PfuUltra High-fidelity DNA polymerase (Stratagene).
Cell proliferation assay and flow cytometry
Cells were plated at 7,500 cells/well in 96-well plates overnight and treated with various concentrations of AR-42 for 72hrs. Cell proliferation was measured by resazurin assay (28), and the 50% inhibitory concentration (IC50) was calculated (21). For cell-cycle analysis, subconfluent cells were treated with 1μM AR-42 or DMSO as a control for two days. For mitotic block, nocodazole (100ng/ml) was added to drug-treated cells for another 24hr prior to harvesting. Following treatment, floating and adherent cells were collected, washed, and fixed in 75% ethanol (29). Fixed cells were incubated with 0.2M phosphate-citrate buffer, pH7.8 to extract low-molecular-weight DNA, stained in propidium iodide (50μg/ml) and RNase A (100μg/ml), and analyzed using a Calibur fluorescence-activated cell sorter (Becton Dickinson) (29). Data analysis was performed using FlowJo software (Tree Star).
Western blot
Subconfluent cells were treated with the indicated concentrations of AR-42 for one or two days. Treated cells were harvested and lysed in cold RIPA buffer supplemented with protease inhibitor cocktail (Sigma). Equal amount of protein (20μg) in each lysate was run on an SDS-polyacrylamide gel, and Western blot was performed as described previously (21).
Lentiviral transduction
Ben-Men-1 cells were transduced with Lenti-CMV-Luc lentiviruses (Qiagen) in the presence of hexadimethrine bromide (8μg/ml) at 37°C overnight. Transduced cells were passaged at 1:5 dilution and then selected for puromycin resistance. Luciferase activity was detected using the Luciferase Reporter Assay System (Promega) (30), and the clone with the highest activity, designated Ben-Men-1-LucB (data not shown), was used subsequently.
Establishment of a skull-base meningioma model
The Institutional Animal Care and Use Committee at Nationwide Children’s Hospital approved this animal study. Ben-Men-1-LucB cells were harvested, washed, and resuspended in phosphate buffered saline (0.5–1×106 cells/mouse in 3μl). Six-to-eight week-old SCID C.B17 mice were anesthetized with 5% isoflurane (Baxter), and their heads were stabilized in a Kopf Small-Animal Stereotaxic Instrument. A midline sagittal incision was made on the cranial skin, and a burr hole was drilled in the skull 1.5mm anterior and 1.5mm to the right of the bregma. A 26-gauge needle attached to a 10-μl Hamilton syringe loaded with cells was slowly inserted through the burr hole and downward about 5mm to the skull base. The cell suspension was injected at a rate of 1.5μl/min via an automatic microinjection unit, and the needle was left in place for 1min before being withdrawn slowly. The incision was closed using 3M VetBond™ Tissue Adhesive. After recovery, mice were monitored for tumor growth and any neurological deficits.
BLI and small-animal magnetic resonance imaging (MRI)
To verify tumor establishment, mice stereotactically inoculated with Ben-Men-1-LucB cells were imaged using a Xenogen IVIS® Spectrum (Caliper) two and four weeks after injection. Briefly, mice were injected intraperitoneally with D-luciferin (150mg/kg body weight; Caliper) and anesthetized with isoflurane. Kinetic analysis of luciferase activity was performed to identify the time at which the peak luminescent signal was emitted. All subsequent BLI was conducted at this peak time. Once tumors had established, mice were divided into three groups and fed normal diet, AR-42-formulated diet, or AR-12-containing diet ad libitum. Tumor growth was monitored monthly by BLI. Photon emission was quantified by region of interest analysis using Living Image software (Caliper). Due to variation in initial tumor size, the luminescence for each mouse was normalized to the signal measured prior to treatment and expressed as the mean normalized luminescence ± standard deviation for each treatment group (n = 5 per group). Also, photon emission from a representative group of mice measured together for each indicated time point was displayed as total flux. A cohort of mice treated with AR-42 for six months were switched to normal diet for an additional six months and imaged monthly to assess the extent of tumor re-growth.
For MRI, mice were anesthetized with isoflurane, injected intraperitoneally with Magnevist® contrast agent (0.1mM/kg), and scanned using a BioSpec 94/30 USR 9.4 T microMRI (Bruker). Tumor volumes were calculated from T1 images as described previously (22,26).
Immunohistochemistry
Mice were injected intraperitoneally with BrdU (0.5mg per 10g body weight) 2hr prior to euthanasia. Heads were dissected and fixed in 10% phosphate-buffered formalin. Following washing in running water for 2hr, heads were treated with 0.1M Tris-HCl, pH7.2 overnight and decalcified in 0.35M EDTA in 0.1M Tris-HCl, pH6.95 for 10–14 days. After optimal decalcification (31), samples were cut in half, incubated in 10% sucrose in 0.1M Tris-HCl, pH7.2 overnight, and embedded in paraffin. Sections (5μm) were obtained, deparaffinized, and stained with hematoxylin and eosin. For immunostaining, deparaffinized sections were heated in 1mM citric acid, pH6.0 in a steamer for 20min and sequentially treated with 3% hydrogen peroxide, Super Block, and a primary antibody overnight (Supplementary Methods), followed by incubation with an HRP-conjugated secondary antibody and color development with AEC chromogen (ScyTek). Hematoxylin was used as a counterstain. Stained sections were mounted and visualized under a Zeiss Axioskop® microscope. Negative controls were treated with the same procedure but without the primary antibody.
Results
Ben-Men-1 benign meningioma cells are NF2-deficient
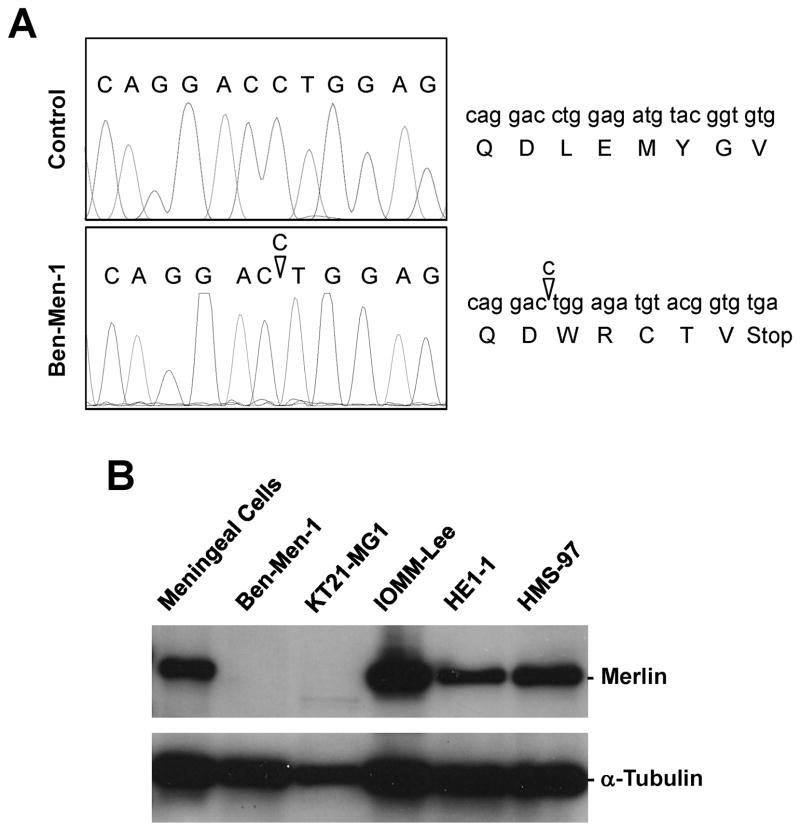
As Ben-Men-1 cells lack one copy of chromosome 22, which contains the NF2 gene (15), we determined the status of the remaining NF2 allele by scanning the 17 NF2 exons for mutations. We detected a deletion of a cytosine (nucleotide #640 relative to the major transcription initiation site designated as +1; [30]) in exon 7 in Ben-Men-1 cells, resulting in a premature stop codon five amino acids downstream of the mutation (Figure 1A). Western blot analysis revealed that Ben-Men-1 cells did not express merlin protein, in contrast to meningeal cells, IOMM-Lee malignant meningioma cells, and HMS-97 malignant schwannoma cells (Figure 1B). While the frameshift mutation predicts a truncated product of 218 amino acids, a protein of this size was not detected in Ben-Men-1 cells (Supplementary Figure S1). In addition, we did not detect merlin protein in KT21-MG1 malignant meningioma cells. These results indicate that Ben-Men-1 and KT21-MG1 cells are NF2-deficient.
Figure 1. Ben-Men-1 cells contain a mutation in the NF2 gene and do not express merlin.
(A) Ben-Men-1 cells carry a deletion of a cytosine residue (del640C; marked with an inverted triangle) in NF2 exon 7, resulting in premature termination. The 17 NF2 exons were obtained from Ben-Men-1 and control DNA by PCR and sequenced according to Materials and Methods. (B) Western blot analysis showed that Ben-Men-1 benign meningioma cells and KT21-MG1 malignant meningioma cells did not express merlin protein, in contrast to normal meningeal cells, IOMM-Lee malignant meningioma cells, HE1-1 embryonic kidney cells, and HMS-97 malignant schwannoma cells. α-tubulin was used as a loading control.
AR-42 differentially affects cell-cycle progression of normal meningeal and Ben-Men-1 meningioma cells
Previously, we showed that AR-42 decreased Ben-Men-1 cell growth with an IC50 of ~1μM (22). Intriguingly, AR-42 also inhibited proliferation of meningeal cells at a similar IC50 (Supplementary Figure S2). Consistent with its action as an HDACi (32), AR-42 treatment increased global acetylation of intracellular proteins, including histone H2B, in both meningeal and Ben-Men-1 cells (Supplementary Figures S3A and S3B). Also, AR-42 decreased expression of p-AKT and two downstream targets of the AKT/mTOR pathway, p-S6 ribosomal protein and p-4E-BP1, in both cell types. These results indicate that AR-42 exerts similar effects on protein acetylation and the AKT pathway in meningeal and meningioma cells.
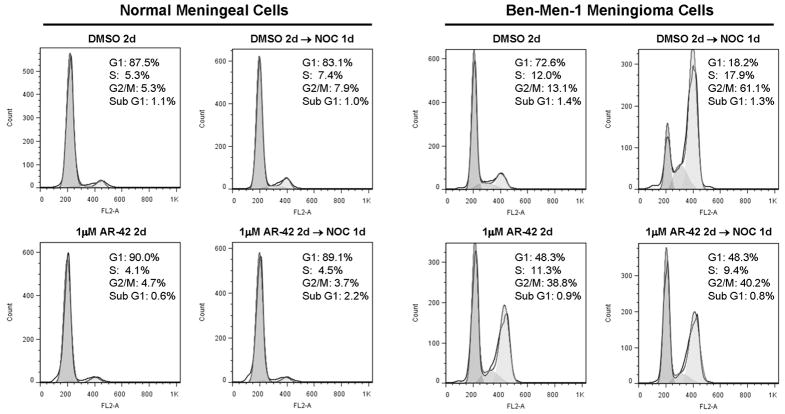
To compare the effect of AR-42 on cell-cycle progression, we performed flow cytometry analysis on meningeal and Ben-Men-1 cells treated with 1 μM AR-42 for two days. Consistent with our previous observation (22), AR-42 treatment substantially increased the number of Ben-Men-1 cells in G2/M from 13.1% to 38.8% (Figure 2). Intriguingly, AR-42 did not affect the percentage of meningeal cells in G2/M but increased the G1 population from 87.5% to 90%, suggesting G1 arrest. To enhance detection of cells arrested in G1, nocodazole, an agent that disrupts microtubule polymerization, was added to AR-42-treated cells for another day (from 48 to 72hr) to block cells in M phase. Note that the doubling time for Ben-Men-1 cells is about two days (15), and meningeal cells double about every three days (Supplementary Figure S4). Addition of nocodazole to AR-42-treated meningeal cells significantly increased the G1 population compared to the nocodazole/DMSO-treated control (from 83.1% to 89.1%; Figure 2), confirming that AR-42 induced G1 arrest in meningeal cells. As expected, nocodazole substantially increased the percentage of DMSO-treated Ben-Men-1 cells in G2/M from 13.1% to 61.1% while decreasing the G1 fraction from 72.6% to 18.2%. However, addition of nocodazole to AR-42-treated Ben-Men-1 cells only slightly increased the G2/M population compared to AR-42-treated cells in the absence of nocodazole (from 38.8% to 40.2%), suggesting that AR-42 also impeded G1 progression of Ben-Men-1 cells. Collectively, these results demonstrated that AR-42 inhibited cell-cycle progression of Ben-Men-1 meningioma cells in both G2/M and G1, while it arrested meningeal cells in G1.
Figure 2. Differential effect of AR-42 on cell-cycle progression of normal meningeal and Ben-Men-1-LucB cells.
Meningeal and Ben-Men-1-LucB cells were treated with 1μM AR-42 or DMSO for two days. To block cells in mitosis, nocodazole (NOC) was added to AR-42-treated cells for another day. After fixation, cells were stained with propidium iodide and analyzed by flow cytometry. AR-42 treatment increased the fraction of Ben-Men-1 cells in G2/M, while increasing the number of meningeal cells in the G1 population. Addition of nocodazole further increased the percentage of meningeal cells in G1.
AR-42 modulates the expression of cell-cycle regulators
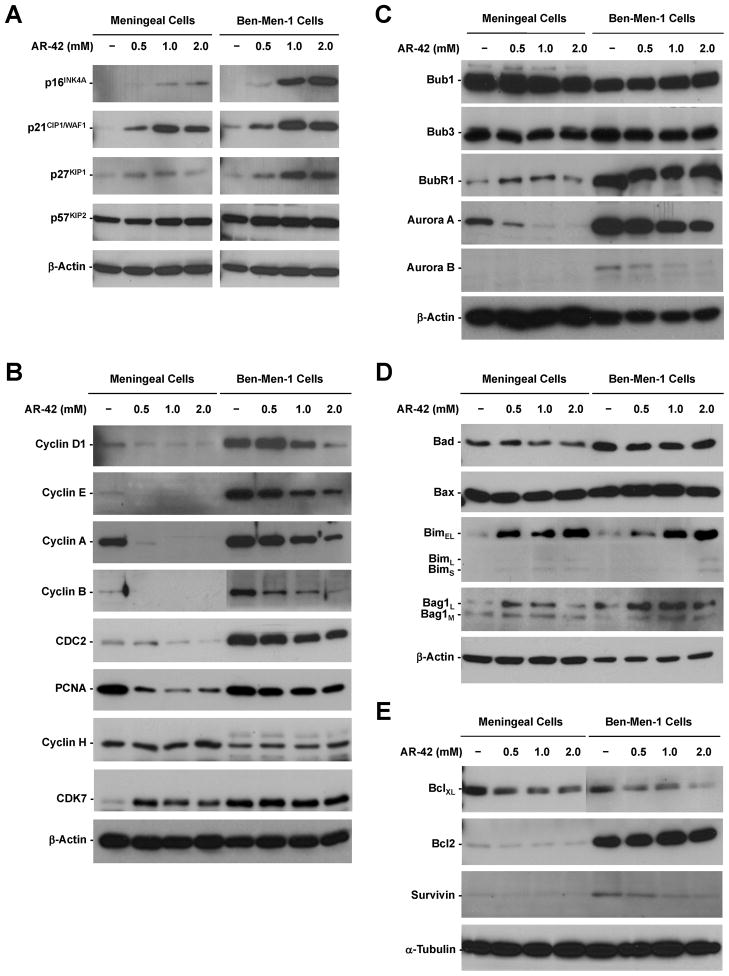
To investigate the mechanism by which AR-42 differentially induces cell-cycle arrest in meningeal and Ben-Men-1 cells, we analyzed the expression of various CDK inhibitors and cyclins. Both meningeal and Ben-Men-1 cells expressed low levels of p16INK4A, p21CIP1/WAF1, and p27KIP1. Treatment with AR-42 induced the expression of these CDK inhibitors in a dose-dependent manner in both cell types (Figure 3A). However, AR-42 did not affect p57KIP2 expression. These results show that inhibition of cell-cycle progression by AR-42 in meningeal and Ben-Men-1 cells is mediated, in part, by induction of multiple CDK inhibitors.
Figure 3. The effects of AR-42 on the expression of various cell-cycle and apoptotic regulators.
Western blot analysis showed that the levels of p16INK4A, p21CIP1/WAF1, and p27KIP1 increased in meningeal and Ben-Men-1 cells treated with AR-42 for 24hr (A). While AR-42 attenuated the expression of cyclin D1, cyclin E, cyclin B, and CDC2, a marked reduction in the cyclin A and PCNA levels was observed in meningeal cells (B). However, AR-42 only modestly decreased PCNA expression but significantly reduced the cyclin B level in Ben-Men-1 cells. AR-42 decreased the levels of Aurora A and Aurora B, whereas Bub1, Bub3, and BubR1 expression were unaffected (C). AR-42 increased the expression of Bim isoforms (BimEL, BimL, and BimS) and Bag1 isoforms (Bag1L and Bag1M) (D), while decreasing the levels of BclXL and survivin in meningeal and Ben-Men-1 cells (E). β-actin and α-tubulin were used as loading controls.
Mitogen stimulation induces the expression of cyclin D, which activates CDK4/6 at early G1, followed by increasing cyclin E and A levels during late G1. CDK2, which binds to cyclin E and A, and CDC2, which binds to cyclin E, promote the G1-to-S transition. In addition, cyclin B and CDC2 are important for progression through G2 (33). While Ben-Men-1 cells expressed higher basal levels of cyclin D1 and E than meningeal cells, AR-42 treatment attenuated the expression of these cyclins and markedly decreased the cyclin A level in meningeal cells (Figure 3B). The decreased expression of these cyclins corroborated the observation that AR-42 induced G1 arrest in meningeal cells. Consistently, AR-42 notably reduced the level of PCNA, a protein induced in S-phase, and further decreased cyclin B and CDC2 expression in meningeal cells. Similarly, AR-42 treatment lowered the expression of cyclin D1, E, and A in Ben-Men-1 cells, albeit the decrease was less dramatic. Consistent with G2/M arrest, AR-42 only modestly decreased PCNA expression but significantly reduced the cyclin B level in Ben-Men-1 cells.
Taken together, these results suggest that AR-42 differentially affects cell-cycle progression of normal meningeal and Ben-Men-1 meningioma cells by regulating the expression of various cyclins and CDK inhibitors.
AR-42 decreases the expression of Aurora kinases and modulates the levels of apoptotic factors
We also analyzed the effect of AR-42 on the expression of various mitotic spindle assembly checkpoint kinases (34). Both meningeal and Ben-Men-1 cells strongly expressed Bub1 and Bub3; however, Ben-Men-1 cells exhibited higher levels of BubR1, Aurora A, and Aurora B than meningeal cells (Figure 3C). While the Bub1, Bub3, and BubR1 levels were not affected by AR-42 treatment, Aurora A expression decreased substantially in both meningeal and Ben-Men-1 cells treated with AR-42. Also, the level of Aurora B decreased in AR-42-treated Ben-Men-1 cells. The reduced expression of Aurora kinases may further reinforce the effect of AR-42 on G2/M arrest in Ben-Men-1 cells.
AR-42 induces apoptosis in several types of tumor cells (22,32,35,36); however, its effects on normal and tumor cells have not been compared. AR-42 treatment increased the expression of pro-apoptotic Bim in a dose-dependent manner but did not affect the levels of Bad and Bax in both meningeal and Ben-Men-1 cells (Figure 3D). Similarly, AR-42 increased the Bag1 level; however, the increase was not as pronounced at higher concentrations (e.g., 2 μM). Conversely, AR-42 decreased the expression of anti-apoptotic BclXL and survivin in both cell types (Figure 3E). While Ben-Men-1 cells expressed higher levels of Bcl2 and survivin than meningeal cells, the expression of Bcl2 was not affected by AR-42 treatment. These results indicate that AR-42 affects the expression of specific pro- and anti-apoptotic regulators.
AR-42 and AR-12 inhibited tumor growth in a quantifiable intracranial benign meningioma model
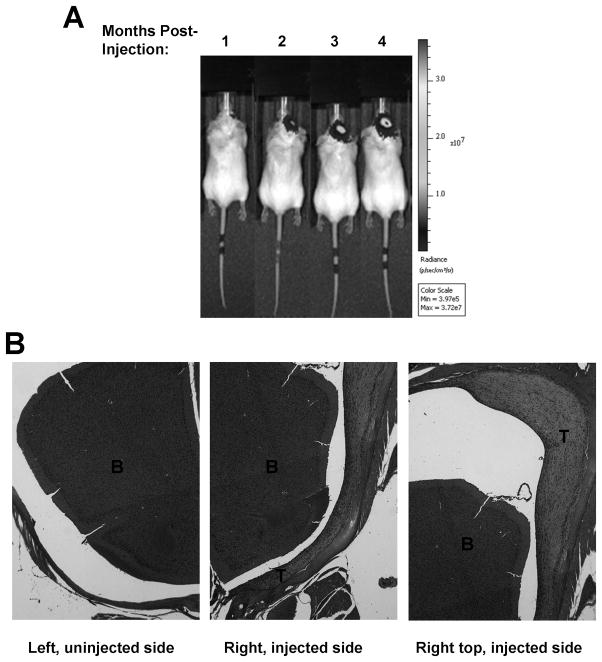
To establish a quantifiable model for NF2-deficient benign meningiomas, we stereotactically injected luciferase-expressing Ben-Men-1-LucB cells, which exhibited a similar sensitivity to AR-42 as their parental Ben-Men-1 cells (Supplementary Figure S5), to the skull base of SCID mice and monitored tumor growth by BLI. Ben-Men-1-LucB tumors grew slowly over time (Figure 4A). Unlike KT21-MG1 malignant meningioma xenografts, which invaded the brain tissue (Supplementary Figure S6), Ben-Men-1-LucB tumors grew from the site of injection along the side of the brain (Figure 4B). As in the original tumor, which expressed epithelial membrane antigen and vimentin, markers commonly used for meningiomas (15), we detected strong staining for vimentin in Ben-Men-1-LucB tumors (Supplementary Figure S7). These results demonstrated a quantifiable, orthotopic NF2-deficient benign meningioma model in which tumor growth could be easily monitored.
Figure 4. Ben-Men-1-LucB cells established intracranial xenograft tumors that grew slowly along the skull over time.
(A) About 5×105 Ben-Men-1-LucB cells were stereotactically injected at the skull base and tumor growth was monitored by BLI according to Materials and Methods. BLI detected increased bioluminescent signal in Ben-Men-1-LucB xenograft-bearing mice with each month after injection. (B) Hematoxylin and eosin staining of the head region of a xenograft-bearing mouse four months after implantation showed that Ben-Men-1-LucB tumors grew from the site of injection along the skull to the top of the head region and did not invade the adjacent brain tissue. B, brain; T, tumor.
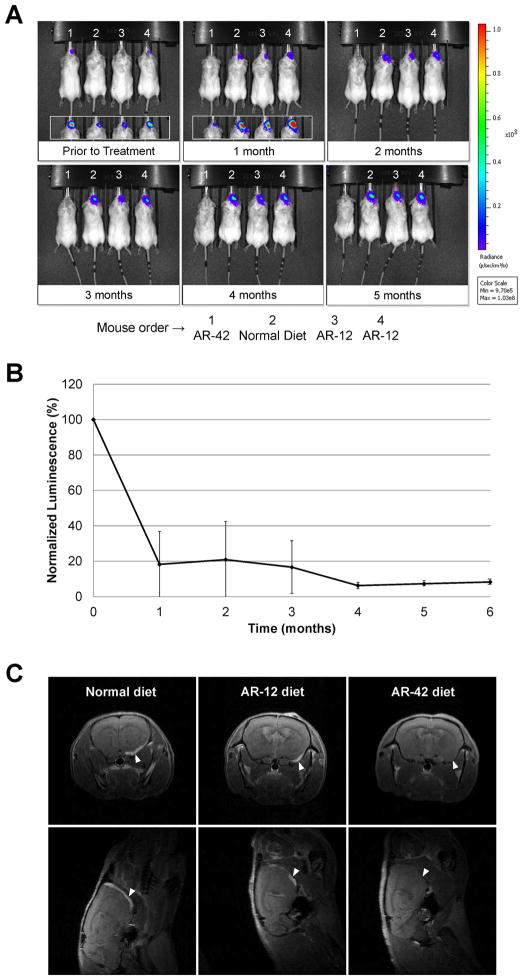
Using this model, we compared the in vivo efficacies of AR-42 and AR-12. Mice with established Ben-Men-1-LucB tumors were fed normal diet or diet containing AR-42 or AR-12 for six months. BLI showed that the tumors in mice fed normal diet grew steadily as demonstrated by an increase in bioluminescence signal over time (Figure 5A and Supplementary Figure S8A and S8B). The tumors in mice fed AR-12 diet also continued to increase in size; however, the growth was slower than those in mice fed normal diet by an average of ~54% after six months of treatment (Supplementary Figure S8B). In contrast, the tumors in mice fed AR-42 regressed, as evident by a sharp decrease in bioluminescence signal after one month, and remained small in subsequent months (Figures 5A and 5B). AR-42 treatment reduced the bioluminescence signal by ~82% after one month and ~92% after six months (Figure 5B).
Figure 5. AR-42 potently suppressed the growth of Ben-Men-1-LucB tumor xenografts.
Mice bearing Ben-Men-1-LucB tumors were established as described in Materials and Methods and fed either normal diet (mouse #2) or chow formulated with AR-42 (mouse #1) or AR-12 (mouse #3 and #4) for the indicated times (n = 5). Tumor growth was monitored by BLI. (A) A representative image shows that while the intensity of bioluminescent signal increased over time in the mouse fed normal diet (mouse #2), the signal in the mouse fed AR-42 diet (mouse #1) decreased sharply after one month of treatment and remained low in subsequent months. The bioluminescent signal in mice treated with AR-12 (mouse #3 and #4) increased over time but at a slower rate than that in the mouse fed normal diet (mouse #2). Insets in the images of mice prior to and one month after treatment are displayed using a lower threshold of bioluminescence detection. (B) Quantitation of the bioluminescent signals emitted from tumors in mice fed AR-42-containing diet was performed, and the relative bioluminescence signal is denoted as the percentage of total flux after each month of treatment relative to the total flux prior to treatment designated as a 100%. The data are shown as mean ± standard deviation and reflect the BLI measurements from five animals per group imaged for six months. (C) Following BLI, tumor-bearing mice fed normal diet or diet containing AR-42 or AR-12 were imaged with gadolinium contrast using a small-animal MRI. Coronal (top) and axial (bottom) T1 MR images showed that the AR-12-treated mouse exhibited a smaller tumor than the mouse fed normal diet. Also, a tumor was not detected in the AR-42-treated mouse after four months of treatment. Arrowheads point to the site of injection at the skull base.
MRI confirmed inhibition of tumor growth by AR-42 and AR-12. While tumors were observed in mice fed normal diet and AR-12-containing chow, the tumor volumes in AR-12-treated mice were smaller than those in mice fed normal diet (Figure 5C and Supplementary Table 1). In contrast, tumors were not detected in mice treated with AR-42 for four to six months. A small tumor was found in a mouse treated with AR-42 for a shorter duration (Supplementary Figure S9). It should be noted that the BLI detected a greater attenuation of tumor growth in AR-12-treated mice (compared to normal diet) than MRI (Supplementary Table 1). Nonetheless, both methods confirmed that AR-12 attenuated tumor growth.
Collectively, these results indicate that AR-42 causes tumor regression, whereas AR-12 moderately slows the growth of tumors over time.
Minimal regrowth of Ben-Men-1-LucB tumors was observed after removal from AR-42 treatment
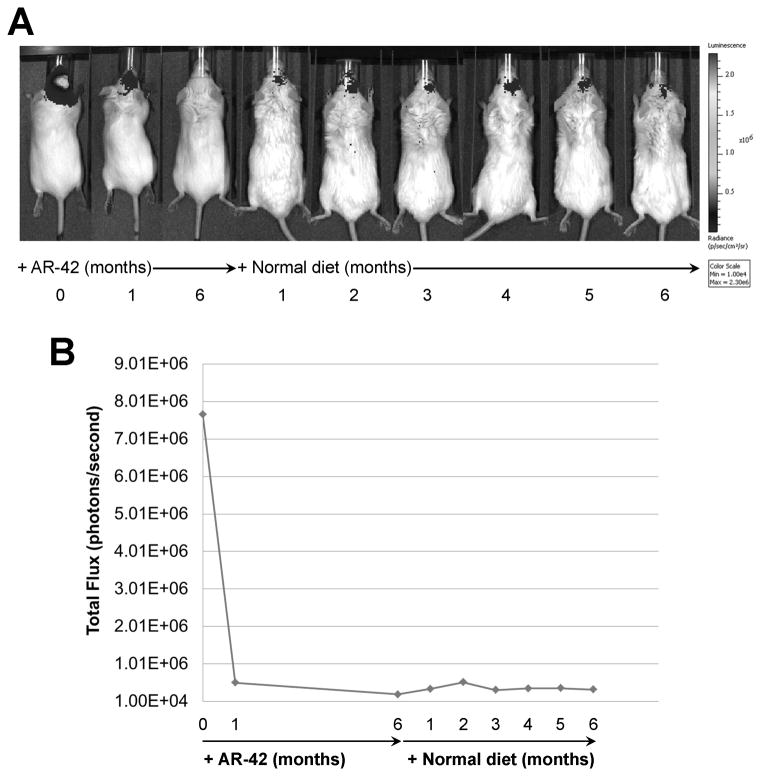
To evaluate potential tumor regrowth following AR-42 treatment, we fed an AR-42-treated mouse normal diet for an additional six months and monitored tumor growth monthly by BLI. While AR-42 treatment substantially decreased the tumor size as demonstrated by the marked decrease of bioluminescence signal after six months (from 7.7 × 106 to 2.0 × 105 photons/sec), the tumor remained small with minimal regrowth (about two-fold increase in bioluminescence signal) over six months after removal from AR-42-containing diet (Figures 6). Also, the tumor remained too small to be detected by MRI (Supplementary Figure S10).
Figure 6. AR-42-treated tumors exhibited minimal regrowth.
A Ben-Men-1-LucB xenograft-bearing mouse was fed AR-42 diet for six months and then switched to normal diet for an additional six months. (A) The bioluminescence signal emitted from the tumor markedly decreased following AR-42 treatment and remained low with minimal regrowth after removal from AR-42 diet. (B) Quantitation of the bioluminescence intensity confirmed minimal tumor regrowth in the AR-42-treated mouse after switching to normal diet for six months.
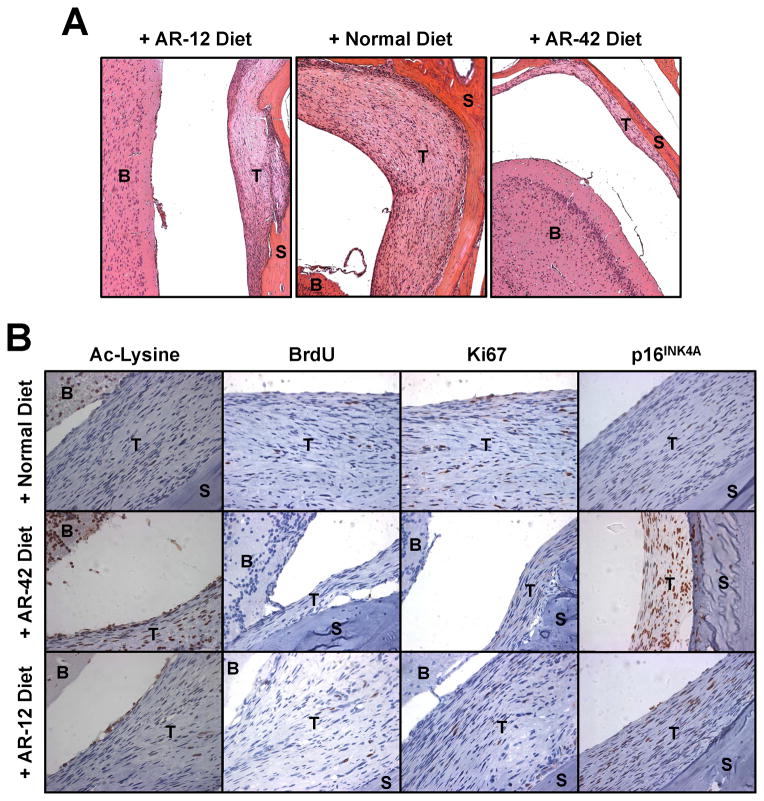
Histological analysis corroborated that the tumors in mice treated with AR-12 for six months were smaller than those fed normal diet (Figure 7A). Also, a very small tumor was found in a mouse treated with AR-42 for three months, consistent with the MRI result. AR-42-treated tumors exhibited increased levels of acetylated proteins compared to mice fed normal diet or diet containing AR-12 (Figure 7B). As observed in cultured cells (Figure 3A), a higher level of p16INK4A was detected in the tumors of AR-42-treated mice than in those of mice fed normal diet (Figure 8B). Consistent with the potent growth-inhibitory activity of AR-42, few or no BrdU- or Ki67-positive cells were seen in AR-42-treated tumors. Together with our previous observation that AR-42 induces apoptosis in vivo (22), these results indicate that AR-42 efficiently inhibits tumor growth in the Ben-Men-1-LucB benign meningioma model.
Figure 7. AR-42-treated tumors showed high levels of acetylated proteins and p16INK4A.
(A) Images shown are hematoxylin-eosin stained sections from tumor-bearing mice fed normal diet or diet containing AR-12 for six months or fed AR-42-containing diet for three months. (B) Sections were immunostained for acetylated lysine (Ac-Lysine), BrdU, Ki67, and p16INK4A. B, brain; T, tumor; S, skull.
Discussion
Development of novel therapeutics for meningiomas, particularly those associated with NF2, is urgently needed and requires disease-specific models. In the present study, we generated a quantifiable orthotopic NF2-deficient benign meningioma model using luciferase-expressing Ben-Men-1-LucB cells. Using this model, we compared the in vivo efficacies of AR-42, a pan-HDACi (25), and AR-12, a PDK1 inhibitor (37), and showed that AR-42 causes tumor regression, whereas AR-12 moderately slows the growth of tumors over time.
Previously, we reported that AR-42 efficiently inhibits schwannoma and meningioma cell proliferation by arresting cells at G2/M and inducing apoptosis (22). Intriguingly, AR-42 inhibited proliferation of normal meningeal cells at about the same IC50 as that for Ben-Men-1 meningioma cells. Although AR-42 exerted similar effects on protein acetylation and AKT phosphorylation in both normal and tumor cells, it arrested meningeal cells at G1 but inhibited Ben-Men-1 cells at both G1 and G2/M. Cell-cycle progression is modulated by CDKs, which are bound and activated by one or more cyclin proteins whose expression oscillates during specific phases of the cell cycle (33). In addition, several regulatory checkpoints exist throughout the cell cycle to maintain genomic stability. Activation of the G1 and G2 checkpoints increases the expression of CDK inhibitors and arrests cells in these phases, providing them time to repair any damage before continuing the cell cycle (38). The INK4 family of CDK inhibitors inhibits cyclin D-associated CDK4/6 at G1, whereas the CIP/KIP family regulates a wider range of cyclin-CDK complexes throughout the cell cycle. HDACi’s have been shown to induce G1 and/or G2/M arrest in tumor cells, and their action is mediated, in part, by altering the levels of various CDK inhibitors (39). HDACi’s can affect transcription of the p16INK4A and p21CIP1/WAF1 genes and alter the p27KIP1 level by a post-translational mechanism. Consistently, AR-42 induced the expression of both the INK (p16INK4A) and CIP/KIP (p21CIP1/WAF1 and p27KIP1) members. However, the induction of these CDK inhibitors by AR-42 in both meningeal and Ben-Men-1 meningioma cells implies that additional mechanisms contribute to the differential effects on cell-cycle progression in these cells.
Tumor cells frequently exhibit an abnormal G1 checkpoint as evident by alteration of the G1 cyclin-CDKs/the INK family/the retinoblastoma protein/E2F cascade (33,38). Consistent with this notion, Ben-Men-1 cells expressed higher basal levels of the G1 cyclins, D1 and E, than meningeal cells. While AR-42 treatment decreased cyclin E and A expression, significant amounts of these cyclins and PCNA were still detected even in the presence of 2 μM AR-42, suggesting that a fraction of AR-42-treated meningioma cells enter S phase. It is possible that the increased expression of multiple CDK inhibitors, together with the decreased levels of G1 cyclins, led to the growth inhibition at G1 observed in AR-42-treated Ben-Men-1 cells. However, the reduction of cyclin B expression by AR-42 may further inhibit the Ben-Men-1 cells that had progressed through S phase and arrest them at G2. In contrast, AR-42 treatment blocked the expression of cyclin D1, E, and A in meningeal cells, and this inhibition, together with the elevated levels of CDK inhibitors, could explain the G1 arrest observed in these cells. Consistently, only a small amount of PCNA and little cyclin B were detected in AR-42-treated meningeal cells. HDACi’s can affect cyclin D, E, and B expression through a transcriptional mechanism or increase acetylation of cyclin A, which facilitates its degradation (39–42). It will be interesting to investigate why AR-42 treatment caused a more pronounced reduction of the cyclin A level in meningeal cells than Ben-Men-1 cells. Nevertheless, the differential effect of AR-42 on cell-cycle progression of meningeal and meningioma cells involves modulation of the expression of multiple cell-cycle regulators.
The mitotic spindle assembly checkpoint, which protects the integrity of cell division, is frequently altered in human tumors and regulated by several kinases, including Bub1, Bub3, and BubR1 (34). In addition, Aurora kinases A and B are important for spindle assembly, chromosome segregation, and cytokinesis (43). Curiously, Ben-Men-1 cells expressed higher levels of Aurora A, Aurora B, and BubR1 than meningeal cells, suggesting deregulation of the mitotic spindle checkpoint in meningioma cells. Importantly, AR-42 decreased the expression of Aurora A and B, further corroborating G2/M arrest in Ben-Men-1 cells. HDACi’s can decrease transcription of Aurora A and B or promote their degradation in different cell types (44–45). However, the mechanism by which AR-42 reduces the expression of Aurora kinases in meningeal and meningioma cells is not known. As Aurora A and B are frequently amplified or over-expressed in a variety of solid and hematological malignancies (34,43), AR-42 may also be a potential therapeutic agent for these tumors.
The members of the Bcl2 family, which consists of pro-apoptotic and anti-apoptotic factors, interact with each other in a delicate balance that governs whether a cell will undergo apoptosis. Our observation that AR-42 increased the expression of Bim and Bag1 and decreased the level of BclXL coincides with previous findings that AR-42 induced apoptosis in tumor cells (22,32,35,36). Also, HDACi’s have been shown to induce the expression of Bcl2-family members, including Bim and BclXL (35,36,46). Together, these results suggest that AR-42- induced apoptosis is mediated, in part, through modulation of pro-apoptotic and anti-apoptotic factors.
Previously, we (21) showed that AR-12, a derivative of celecoxib that lacks cyclooxygenase 2-inhibitory activity (37), decreased AKT phosphorylation and efficiently inhibited schwannoma growth in vitro and in vivo. As in schwannoma xenografts (21), AR-12 exhibited moderate anti-tumor activity in Ben-Men-1-LucB meningiomas. Interestingly, AR-42 gave rise to more profound growth inhibition than AR-12 and caused tumor regression, suggesting that inhibition of the AKT pathway is not sufficient to account for the anti-tumor potency of AR-42. Also, the residual tumors in AR-42-treated mice showed minimal regrowth after switching to normal diet. As AR-42 also potently inhibits schwannoma growth and is well-tolerated in mice (22,23), these results suggest that it is a promising therapeutic agent for NF2-deficient tumors.
BLI is a sensitive and efficient way to non-invasively monitor tumor growth, particularly for longitudinal studies of benign tumors. Using BLI, we detected bioluminescence signal emitted from small luciferase-expressing tumors in AR-42-treated mice while MRI could not identify tumors in the same mice. This observation is consistent with a previous report that MRI efficiently identifies macroscopic tumors but is less effective at detecting small tumors in mice compared with BLI (47). Also, MRI measured a less pronounced reduction in tumor size in AR-12-treated mice than BLI. It is possible that BLI may quantify the differences in cell density in benign tumors. Treatment may affect peritumoral edema of intracranial meningiomas (48), and fluid surrounding the tumors may obscure the tumor margin used in volumetric MRI measurement. Nevertheless, MRI and BLI were complementary tools to quantify the effects of AR-42 and AR-12 on tumor growth.
Over-expressed or sustained HDAC activity has been observed in many solid and hematological malignancies, suggesting therapeutic potential of HDACi’s (24,49). Several HDACi’s are currently in clinical trials for various types of cancer. Among them, Zolinza (vorinostat) and Romidepsin (depsipeptide) have been approved by the FDA for the treatment of cutaneous T-cell lymphomas. AR-42 possesses growth-inhibitory activity that is comparable to or more potent than vorinostat in several tumor models (32,35,36,50). It is orally bioavailable, penetrates the blood-brain barrier, and exhibits low toxicity. Mice treated with AR-42 showed slight weight loss, leukopenia, anemia, liver hypertrophy, and testicular degeneration, and these effects are reversible after removal from treatment (23,50). AR-42 is currently being evaluated in a phase I/IIa clinical trial for relapsed and refractory multiple myeloma, chronic lymphocytic leukemia, and lymphoma. Our observation that AR-42 differentially affects cell-cycle progression of normal meningeal and meningioma cells may have implications for why it is well-tolerated while potently inhibiting tumor growth as prolonged G2 arrest may lead to apoptosis in proliferating tumor cells. Collectively, our results suggest that AR-42 merits a clinical trial for meningiomas and schwannomas.
Supplementary Material
Acknowledgments
Financial supports: This study was supported by grants from the Children’s Tumor Foundation, NIDCD (DC005985), US Department of Defense (NF080021), NF Midwest, and Advocure NF2 to DBW and LSC.
We sincerely thank Anita Lal for KT21-MG1 and IOMM-Lee cells, Kimerly Powell and the Small-Animal Imaging Core for MRI, and Allen Qian for technical assistance, and Beth Miles-Markley and Peter Houghton for critical reading of the manuscript.
Footnotes
Disclosure of Potential Conflicts of Interest: No potential conflicts of interest were disclosed.
Literature Cited
- 1.Wiemels J, Wrensch M, Claus EB. Epidemiology and etiology of meningioma. J Neurooncol. 2010;99:307–314. doi: 10.1007/s11060-010-0386-3. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 2.Goutagny S, Kalamarides M. Meningiomas and neurofibromatosis. J Neurooncol. 2010;99:341–47. doi: 10.1007/s11060-010-0339-x. [DOI] [PubMed] [Google Scholar]
- 3.Rouleau GA, Merel P, Lutchman M, Sanson M, Zucman J, Marineau C, et al. Alteration in a new gene encoding a putative membrane-organizing protein causes neuro-fibromatosis type 2. Nature. 1993;363:515–21. doi: 10.1038/363515a0. [DOI] [PubMed] [Google Scholar]
- 4.Trofatter JA, MacCollin MM, Rutter JL, Murrell JR, Duyao MP, Parry DM, et al. A novel moesin-, ezrin-, radixin-like gene is a candidate for the neurofibromatosis 2 tumor suppressor. Cell. 1993;72:791–800. doi: 10.1016/0092-8674(93)90406-g. [DOI] [PubMed] [Google Scholar]
- 5.James MF, Han S, Polizzano C, Plotkin SR, Manning BD, Stemmer-Rachamimov AO, et al. NF2/merlin is a novel negative regulator of mTOR complex 1, and activation of mTORC1 is associated with meningioma and schwannoma growth. Mol Cell Biol. 2009;29:4250–61. doi: 10.1128/MCB.01581-08. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 6.Kalamarides M, Niwa-Kawakita M, Leblois H, Abramowski V, Perricaudet M, Janin A, et al. Nf2 gene inactivation in arachnoidal cells is rate-limiting for meningioma development in the mouse. Genes Dev. 2002;16:1060–65. doi: 10.1101/gad.226302. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 7.Kalamarides M, Stemmer-Rachamimov AO, Niwa-Kawakita M, Chareyre F, Taranchon E, Han ZY, et al. Identification of a progenitor cell of origin capable of generating diverse meningioma histological subtypes. Oncogene. 2011;30:2333–44. doi: 10.1038/onc.2010.609. [DOI] [PubMed] [Google Scholar]
- 8.Lee WH. Characterization of a newly established malignant meningioma cell line of the human brain: IOMM-Lee. Neurosurgery. 1990;27:389–95. [PubMed] [Google Scholar]
- 9.Tanaka K, Sato C, Maeda Y, Koike M, Matsutani M, Yamada K, et al. Establishment of a human malignant meningioma cell line with amplified c-myc oncogene. Cancer. 1989;64:2243–49. doi: 10.1002/1097-0142(19891201)64:11<2243::aid-cncr2820641110>3.0.co;2-s. [DOI] [PubMed] [Google Scholar]
- 10.Ragel BT, Elam IL, Gillespie DL, Flynn JR, Kelly DA, Mabey D, et al. A novel model of intracranial meningioma in mice using luciferase-expressing meningioma cells. J Neurosurg. 2008;108:304–10. doi: 10.3171/JNS/2008/108/2/0304. [DOI] [PubMed] [Google Scholar]
- 11.McCutcheon IE, Friend KE, Gerdes TM, Zhang BM, Wildrick DM, Fuller GN. Intracranial injection of human meningioma cells in athymic mice: an orthotopic model for meningioma growth. J Neurosurg. 2000;92:306–14. doi: 10.3171/jns.2000.92.2.0306. [DOI] [PubMed] [Google Scholar]
- 12.Baia GS, Dinca EB, Ozawa T, Kimura ET, McDermott MW, James CD, et al. An orthotopic skull base model of malignant meningioma. Brain Pathol. 2008;18:172–79. doi: 10.1111/j.1750-3639.2007.00109.x. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 13.Cargioli TG, Ugur HC, Ramakrishna N, Chan J, Black PM, Carroll RS. Establishment of an in vivo meningioma model with human telomerase reverse transcriptase. Neurosurgery. 2007;60:750–59. doi: 10.1227/01.NEU.0000255397.00410.8F. [DOI] [PubMed] [Google Scholar]
- 14.Baia GS, Slocum AL, Hyer JD, Misra A, Sehati N, VandenBerg SR, et al. A genetic strategy to overcome the senescence of primary meningioma cell cultures. J Neurooncol. 2006;78:113–21. doi: 10.1007/s11060-005-9076-y. [DOI] [PubMed] [Google Scholar]
- 15.Püttmann S, Senner V, Braune V, Hillmann B, Exeler R, Rickert C, et al. Establishment of a benign meningioma cell line by hTERT-mediated immortalization. Lab Invest. 2005;85:1163–71. doi: 10.1038/labinvest.3700307. [DOI] [PubMed] [Google Scholar]
- 16.Watson MA, Gutmann DH, Peterson K, Chicoine MR, Kleinschmidt-DeMasters BK, Brown HG, et al. Molecular characterization of human meningiomas by gene expression profiling using high density oligonucleotide microarrays. Am J Pathol. 2002;161:665–72. doi: 10.1016/S0002-9440(10)64222-8. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 17.Welling DB, Lasak JM, Akhmametyeva E, Ghaheri B, Chang LS. cDNA microarray analysis of vestibular schwannomas. Otol Neurotol. 2002;23:736–48. doi: 10.1097/00129492-200209000-00022. [DOI] [PubMed] [Google Scholar]
- 18.Cuevas IC, Slocum AL, Jun P, Costello JF, Bollen AW, Riggins GJ, et al. Meningioma transcript profiles reveal deregulated Notch signaling pathway. Cancer Res. 2005;65:5070–75. doi: 10.1158/0008-5472.CAN-05-0240. [DOI] [PubMed] [Google Scholar]
- 19.Hanemann CO, Bartelt-Kirbach B, Diebold R, Kämpchen K, Langmesser S, Utermark T. Differential gene expression between human schwannoma and control Schwann cells. Neuropathol Appl Neurobiol. 2006;32:605e14. doi: 10.1111/j.1365-2990.2006.00769.x. [DOI] [PubMed] [Google Scholar]
- 20.Jacob A, Lee TX, Neff BA, Miller S, Welling B, Chang L-S. Phosphatidylinositol 3-kinase/AKT pathway activation in human vestibular schwannoma. Otol Neurotol. 2008;29:58–68. doi: 10.1097/mao.0b013e31816021f7. [DOI] [PubMed] [Google Scholar]
- 21.Lee TX, Packer MD, Huang J, Akhmametyeva EM, Kulp SK, Chen C-S, et al. Growth inhibitory and anti-tumor activities of OSU-03012, a novel PDK-1 inhibitor, on vestibular schwannoma and malignant schwannoma cells. Eur J Cancer. 2009;45:1709–20. doi: 10.1016/j.ejca.2009.03.013. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 22.Bush ML, Oblinger J, Brendel V, Santarelli G, Huang J, Akhmametyeva EM, et al. AR42, a novel histone deacetylase inhibitor, as a potential therapy for vestibular schwannomas and meningiomas. Neuro Oncol. 2011;13:983–99. doi: 10.1093/neuonc/nor072. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 23.Jacob A, Oblinger J, Bush ML, Brendel V, Santarelli G, Ray-Chaudhury A, et al. Preclinical validation of AR42, a novel histone deacetylase inhibitor, as treatment for vestibular schwannomas. Laryngoscope. 2012;122:174–89. doi: 10.1002/lary.22392. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 24.Minucci S, Pelicci PG. Histone deacetylase inhibitors and the promise of epigenetic (and more) treatments for cancer. Nat Rev Cancer. 2006;6:38–51. doi: 10.1038/nrc1779. [DOI] [PubMed] [Google Scholar]
- 25.Chen CS, Weng SC, Tseng PH, Lin HP, Chen C-S. Histone acetylation-independent effect of histone deacetylase inhibitors on Akt through the reshuffling of protein phosphatase 1 complexes. J Biol Chem. 2005;280:38879–87. doi: 10.1074/jbc.M505733200. [DOI] [PubMed] [Google Scholar]
- 26.Chang L-S, Jacob A, Lorenz M, Rock J, Akhmametyeva EM, Mihai G, et al. Growth of benign and malignant schwannoma xenografts in severe combined immunodeficiency mice. Laryngoscope. 2006;116:2018–26. doi: 10.1097/01.mlg.0000240185.14224.7d. [DOI] [PubMed] [Google Scholar]
- 27.Chang L-S, Welling DB. Molecular biology of vestibular schwannomas. Methods Mol Biol. 2009;493:163–77. doi: 10.1007/978-1-59745-523-7_10. [DOI] [PubMed] [Google Scholar]
- 28.Fields RD, Lancaster MV. Dual attribute continuous monitoring of cell proliferation/cytotoxicity. Am Biotechnol Lab. 1993;11:48–50. [PubMed] [Google Scholar]
- 29.Darzynkiewicz Z, Juan G. Current Protocols in Flow Cytometry. Unit 7.5. John Wiley & Sons, Inc; Somerset, NJ: 1997. DNA content measurement for DNA ploidy and cell cycle analysis. [Google Scholar]
- 30.Chang L-S, Akhmametyeva EM, Wu Y, Zhu L, Welling DB. Multiple transcription initiation sites, alternative splicing, and differential polyadenylation contribute to the complexity of human neurofibromatosis 2 transcripts. Genomics. 2002;79:63–76. doi: 10.1006/geno.2001.6672. [DOI] [PubMed] [Google Scholar]
- 31.Seilly DJ. A chemical test to determine the end point of EDTA decalcification. Med Lab Sci. 1982;39:71–73. [PubMed] [Google Scholar]
- 32.Yang YT, Balch C, Kulp SK, Mand MR, Nephew KP, Chen CS. A rationally designed histone deacetylase inhibitor with distinct antitumor activity against ovarian cancer. Neoplasia. 2009;11:552–63. doi: 10.1593/neo.09204. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 33.Hochegger H, Takeda S, Hunt T. Cyclin-dependent kinases and cell-cycle transitions: does one fit all. Nat Rev Mol Cell Biol. 2008;9:910–16. doi: 10.1038/nrm2510. [DOI] [PubMed] [Google Scholar]
- 34.Kops GJ, Weaver BA, Cleveland DW. On the road to cancer: aneuploidy and the mitotic checkpoint. Nat Rev Cancer. 2005;5:773–85. doi: 10.1038/nrc1714. [DOI] [PubMed] [Google Scholar]
- 35.Kulp SK, Chen CS, Wang DS, Chen CY, Chen CS. Antitumor effects of a novel phenylbutyrate-based histone deacetylase inhibitor, (S)-HDAC-42, in prostate cancer. Clin Cancer Res. 2006;12:5199–206. doi: 10.1158/1078-0432.CCR-06-0429. [DOI] [PubMed] [Google Scholar]
- 36.Lu YS, Kashida Y, Kulp SK, Wang YC, Wang D, Hung JH, et al. Efficacy of a novel histone deacetylase inhibitor in murine models of hepatocellular carcinoma. Hepatology. 2007;46:1119–30. doi: 10.1002/hep.21804. [DOI] [PubMed] [Google Scholar]
- 37.Zhu J, Huang JW, Tseng PH, Yang YT, Fowble J, Shiau CW, et al. From the cyclooxygenase-2 inhibitor celecoxib to a novel class of 3-phosphoinositide-dependent protein kinase-1 inhibitors. Cancer Res. 2004;64:4309–18. doi: 10.1158/0008-5472.CAN-03-4063. [DOI] [PubMed] [Google Scholar]
- 38.Sherr CJ, Roberts JM. CDK inhibitors: positive and negative regulators of G1-phase progression. Genes Dev. 1999;13:1501–12. doi: 10.1101/gad.13.12.1501. [DOI] [PubMed] [Google Scholar]
- 39.Mork CN, Faller DV, Spanjaard RA. A mechanistic approach to anticancer therapy: targeting the cell cycle with histone deacetylase inhibitors. Curr Pharm Des. 2005;11:1091–104. doi: 10.2174/1381612053507567. [DOI] [PubMed] [Google Scholar]
- 40.Nair AR, Boersma LJ, Schiltz L, Chaudhry MA, Muschel RJ. Paradoxical effects of trichostatin A: inhibition of NF-Y-associated histone acetyltransferase activity, phosphorylation of hGCN5 and downregulation of cyclin A and B1 mRNA. Cancer Lett. 2001;166:55–64. doi: 10.1016/s0304-3835(01)00418-9. [DOI] [PubMed] [Google Scholar]
- 41.Sambucetti LC, Fischer DD, Zabludoff S, Kwon PO, Chamberlin H, Trogani N, et al. Histone deacetylase inhibition selectively alters the activity and expression of cell cycle proteins leading to specific chromatin acetylation and antiproliferative effects. J Biol Chem. 1999;274:34940–47. doi: 10.1074/jbc.274.49.34940. [DOI] [PubMed] [Google Scholar]
- 42.Mateo F, Vidal-Laliena M, Canela N, Busino L, Martinez-Balbas MA, Pagano M, et al. Degradation of cyclin A is regulated by acetylation. Oncogene. 2009;28:2654–66. doi: 10.1038/onc.2009.127. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 43.Keen N, Taylor S. Aurora-kinase inhibitors as anticancer agents. Nat Rev Cancer. 2004;4:927–36. doi: 10.1038/nrc1502. [DOI] [PubMed] [Google Scholar]
- 44.Li Y, Kao GD, Garcia BA, Shabanowitz J, Hunt DF, Qin J, et al. A novel histone deacetylase pathway regulates mitosis by modulating Aurora B kinase activity. Genes Dev. 2006;20:2566–79. doi: 10.1101/gad.1455006. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 45.Cha TL, Chuang MJ, Wu ST, Sun GH, Chang SY, Yu DS, et al. Dual degradation of aurora A and B kinases by the histone deacetylase inhibitor LBH589 induces G2-M arrest and apoptosis of renal cancer cells. Clin Cancer Res. 2009;15:840–50. doi: 10.1158/1078-0432.CCR-08-1918. [DOI] [PubMed] [Google Scholar]
- 46.Zhao Y, Tan J, Zhuang L, Jiang X, Liu ET, Yu Q. Inhibitors of histone deacetylases target the Rb-E2F1 pathway for apoptosis induction through activation of proapoptotic protein Bim. Proc Natl Acad Sci USA. 2005;102:16090–95. doi: 10.1073/pnas.0505585102. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 47.Puaux AL, Ong LC, Jin Y, Teh I, Hong M, Chow PK, et al. A comparison of imaging techniques to monitor tumor growth and cancer progression in living animals. Int J Mol Imaging. 2011:e321538. doi: 10.1155/2011/321538. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 48.Nakano T, Asano K, Miura H, Itoh S, Suzuki S. Meningiomas with brain edema: radiological characteristics on MRI and review of the literature. Clin Imaging. 2002;26:243–49. doi: 10.1016/s0899-7071(02)00433-3. [DOI] [PubMed] [Google Scholar]
- 49.Bolden JE, Peart MJ, Johnstone RW. Anticancer activities of histone deacetylase inhibitors. Nat Rev Drug Discov. 2006;5:769–84. doi: 10.1038/nrd2133. [DOI] [PubMed] [Google Scholar]
- 50.Sargeant AM, Rengel RC, Kulp SK, Klein RD, Clinton SK, Wang YC, et al. OSU-HDAC42, a histone deacetylase inhibitor, blocks prostate tumor progression in the transgenic adenocarcinoma of the mouse prostate model. Cancer Res. 2008;68:3999–4009. doi: 10.1158/0008-5472.CAN-08-0203. [DOI] [PubMed] [Google Scholar]
Associated Data
This section collects any data citations, data availability statements, or supplementary materials included in this article.