Abstract
There is a growing interest in developing new limb salvage therapies for patients with severe peripheral artery disease who have no alternative to amputation. Cell and gene therapy studies are showing promise in controlling pain and minor ulceration in patients with significant critical limb ischemia. Among cardiovascular cell and molecular therapy programs, The Methodist Hospital is one of the leading centers in both gene and cell therapy for critical limb ischemia. Randomized controlled trials continue to be performed, and these experimental therapies will move from research to pharmacy within the decade. In conjunction with aggressive medical and surgical management, these emergent therapies may help patients with critical limb ischemia avoid a major amputation and are one of the foundations of any advanced limb salvage program.
Keywords: amputation, arteriogenesis, cell therapy, critical limb ischemia, fibroblast growth factor (FGF), gene therapy, hepatocyte growth factor (HGF), limb salvage, molecular therapy, therapeutic angiogenesis, vascular endothelial cell growth factor (VEGF), vasculogenesis

Introduction
Approximately 20–30% of patients with critical limb ischemia are not considered candidates for vascular or endovascular procedures, and amputation is often the only option available. This corresponds to 120,000 amputations annually in the United States.1 Leg amputation due to atherosclerotic critical limb ischemia (CLI) gives rise to an acute mortality rate of around 30% and a 5-year survival rate of less than 30%.2, 3 Poor prognosis and the increasing number of patients with CLI have created a growing need for new therapies to induce angiogenesis, with the most emphasis being placed on gene and cell therapy.
Therapeutic angiogenesis is the iatrogenic, induced formation of a capillary network in ischemic tissue by altering the local milieu to provide a proangiogenic environment. Physiologically, angiostatic mediators generally outweigh the angiogenic molecules, preventing the occurrence of angiogenesis. Introduction of a vector to force-express an angiogenic factor disrupts this equilibrium. Therapeutic angiogenesis can occur via two mechanisms: vasculogenesis and angiogenesis.4 Vasculogenesis is the in situ assembly of precursor cells, or angioblasts, into capillaries, whereas angiogenesis is the formation of new capillaries from preexisting vessels. Angiogenesis is initiated by vasodilatation and increased permeability of the vessel (Figure 1). The molecular, cellular, humoral, and mechanical factors result in destabilization of vessel wall homeostasis. This leads to endothelial cell proliferation, migration, and formation of a tube, which then matures with the arrival of pericytes and smooth muscle cells. Numerous soluble growth factors and inhibitors, cytokines and proteases, and extracellular matrix proteins and adhesion molecules strictly control this multi-step process. No influx of nontissue resident cells appears to be required.5 The resulting capillaries are small, with a diameter of about 10 to 20 μm, and cannot be considered sufficient to compensate or substitute for a large occluded transport artery. Originally, vasculogenesis and angiogenesis were considered independent events, with vasculogenesis occurring exclusively during embryogenesis and angiogenesis occurring in adults. It is now recognized that both mechanisms are involved in vessel formation within a single microenvironment.6, 7 Vasculogenesis has been reported to contribute between 3.5–25% to adult neovascularization.7
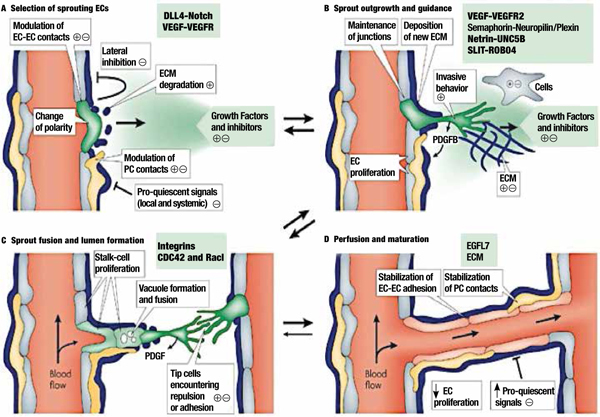
Figure 1.
Biology of Angiogenesis.32
Panel A. Angiogenic sprouting. (A) Sprouting is controlled by the balance between pro-angiogenic signals (+), such as vascular endothelial growth factor (VEGF), and factors that promote quiescence (–), such as tight pericyte (PC; yellow) contact, certain extracellular matrix (ECM) molecules, or VEGF inhibitors. In conditions that favor angiogenesis, some endothelial cells (ECs) can sprout (green), whereas others fail to respond (grey). Sprouting requires the flipping of apical–basal polarity, the induction of motile and invasive activity, the modulation of cell–cell contacts, and local matrix degradation. (B) The growing EC sprout is guided by VEGF gradients. Other signals may include attractive (+) or repulsive (–) matrix cues and guidepost cells in the tissue environment. Release of platelet-derived growth factor B (PDGFB) by the tip cells promotes the recruitment of PCs to new sprouts. EC–EC junctions need to be maintained after lumen formation to prevent excessive leakage. (C) Adhesive or repulsive interactions that occur when tip cells encounter each other regulate the fusion of adjacent sprouts and vessels. Lumen formation in stalk ECs involves the fusion of vacuoles, but other mechanisms may also contribute. (D) Fusion processes at the EC–EC interfaces establish a continuous lumen. Blood flow improves oxygen delivery and thereby reduces pro-angiogenic signals that are hypoxia-induced. Perfusion is also likely to promote maturation processes such as the stabilization of cell junctions, matrix deposition, and tight PC attachment. Growth factor withdrawal can trigger sprout retraction and endothelial apoptosis. DLL4: delta-like-4 ligand; EGFL7: epidermal growth factor ligand-7; ROBO4: roundabout homologue-4 (also known as magic roundabout); VEGFR2: VEGF receptor-2.
Panel B. Origin of endothelial cells and assembly of the vasculature. Mesodermal cells in the early embryo differentiate into endothelial precursor cells (EPCs, angioblasts) and form aggregates, known as blood islands (left). Fusion of blood islands leads to the vasculogenic formation of honeycomb-shaped primary capillary plexi in the yolk sac and embryo itself. Blood circulation is established and primary plexi are remodeled into a hierarchical network of arterioles and arteries (red), capillaries (grey), and venules and veins (blue). The dorsal aorta and cardinal vein are directly formed through the assembly of angioblasts. The vasculogenic incorporation of circulating EPCs into growing blood vessels may contribute to regenerative or pathological neovascularization in the adult. Vascular smooth-muscle cells (vSMCs) are associated with arteries and veins, whereas capillaries are covered by pericytes (yellow). The first lymphatic endothelial cells (LECs) sprout from the embryonic veins, then migrate and form lymphatic sacs. Further steps of lymphangiogenic growth involve sprouting, branching, proliferation, differentiation, and remodeling processes. The recruitment of lymphangioblasts from the adjacent mesenchyme has been speculated to be a further source of LECs. Blind-ending lymphatic capillaries (green) feed into collecting vessels and ducts. These larger lymphatics are sparsely covered by SMCs (purple) and contain valves that prevent backflow.
Panel C. Vasculogenic and intussusceptive growth of blood vessels. (A) Recruitment and incorporation of cells from blood circulation requires positive chemotactic signals and local factors that promote adhesion to endothelial cells (ECs). Transendothelial migration and/or the incorporation of precursor cells into the endothelium require the modulation of existing EC–EC contacts. Circulating precursor cells (angioblasts) have been suggested to differentiate into ECs and lead to an expansion of the vessel diameter (circumferential growth) or, alternatively, they may be recruited to new angiogenic sprouts. Circulation-derived cells do not necessarily acquire an endothelial fate but can instead promote the proliferation and sprouting of local ECs from a perivascular location. (B) Intussusception — the splitting of vessels through the insertion of tissue pillars — is a separate mechanism that leads to the expansion of blood vessels. Little is known about the function or regulation of intussusceptive growth, but the process is likely to involve EC proliferation, cell movement, the degradation of extracellular matrix (ECM), and the deposition of new ECM molecules. CXCR4: CXC-chemokine receptor-4; SDF1 (also known as CXCL12): stromal cell-derived factor-1; VEGF: vascular endothelial growth factor; VEGFR1: VEGF receptor-1.
Reprinted with permission.32


Distinct from angiogenesis or vasculogenesis, arteriogenesis may be the mechanism with the greatest clinical importance. Arteriogenesis is also known as collateralization. It does not involve the formation of new vessels; rather, it is the process by which pre-existing collateral vessels enlarge up to 25 times their original size to facilitate increases in blood supply.8 The resulting high-conductance vessels rapidly increase blood flow, unlike capillaries formed via angiogenesis or vasculogenesis. This process represents structural remodeling of existing vessels, driven in part by changes in vessel shear stress, and is not simply a result of permanent vasodilation.9 The increase in shear stress activates endothelial cells to release factors that recruit monocytes to the collaterals. These monocytes produce the multiple mediators of arteriogenesis and induce inflammation.9 The local inflammatory environment plays an important role in providing signals vital to the enlargement of the collaterals. Similarly, inflammatory foci in tissues have been characterized by uncontrolled angiogenesis, and angiogenesis is important in the spread of malignancy.10 Therapeutic angiogenesis for critical limb ischemia is delivered via gene vectors or cell implantation.
Gene Therapy
In vivo gene transfer techniques for vascular applications include 1) viral gene transfer: retrovirus, adenovirus, adeno-associated virus, or hemagglutinating virus of Japan (Sendai virus); 2) liposomal gene transfer using cationic liposomes; and 3) naked plasmid DNA transfer. Initially, single applications of therapy were the norm, but this has now changed to multiple applications over a 4- to 8-week interval to allow for continued priming of the area targeted for angiogenesis. Jeffrey Isner is credited with popularizing therapeutic angiogenesis with his group’s first trials using an isoform of vascular endothelial cell growth factor (VEGF165) on a plasmid. Table 1 shows the numerous patient series and controlled studies that have been performed in this area and the reported clinical outcomes demonstrating clinical efficacy for the treatment.11, 12 Since then, numerous angiogenic growth factors, such as VEGF121, VEGF-2, basic fibroblast growth factor (FGF), and hepatocyte growth factor (HGF) have been and continue to be tested in clinical trials (Table 1). In addition to intramuscular injection of naked plasmid DNA, adenoviral delivery of angiogenic growth factors has also been used in these trials.
Table 1.
Results of gene therapy for critical limb ischemia.
| Author | Year | Study Level | N | Vector | Product | Delivery | Clinical Outcome* |
| Isner | 1996 | patient series/uncontrolled trial | 1 | Plasmid | pVEGF 165 | Catheter mediated | |
| Baumgartner | 1998 | patient series/uncontrolled trial | 9 | Plasmid | pVEGF 165 | IM | Positive |
| Isner | 1998 | patient series/uncontrolled trial | 6 | Plasmid | phVEGF 165 | IM | Positive |
| Baumgartner | 2000 | patient series/uncontrolled trial | 62 | Plasmid | pVEGF 165 | IM | Equivocal |
| Rajagopalan | 2001 | patient series/uncontrolled trial | 5 | Adenovirus | VEGF 121 | IM | Equivocal |
| Comerota | 2002 | patient series/uncontrolled trial | 51 | Plasmid | NV1FGF | IM | Positive |
| Makinen | 2002 | controlled trial | 36 | Adenovirus | VEGF | Catheter mediated | Positive |
| Mohler | 2003 | patient series/uncontrolled trial | 21 | Adenovirus | VEGF 121 | IM | Equivocal |
| Shyu | 2003 | patient series/uncontrolled trial | 21 | Adenovirus | VEGF165 | IM | Positive |
| Morishita | 2004 | patient series/uncontrolled trial | 6 | Plasmid | HGF | IM | Positive |
| Kim | 2004 | patient series/uncontrolled trial | 9 | Plasmid | VEGF165 | IM | Equivocal |
| Matyas | 2005 | controlled trial | 13 | Adenovirus | FGF-4 | IM | Equivocal |
| Kusumanto | 2006 | controlled trial | 54 | Plasmid | phVEGF165 | IM | Equivocal |
| Marui | 2007 | patient series/uncontrolled trial | 7 | Plasmid | FGF-1 | IM | Positive |
| Rajagopalan | 2007 | controlled trial | 34 | Adenovirus | HIF-1alpha | IM | Equivocal |
| Powell | 2008 | controlled trial | 104 | Plasmid | HGF | IM | Equivocal |
| Nikol | 2008 | controlled trial | 125 | Plasmid | NV1FGF | IM | Positive |
| Baumgartner | 2009 | patient series/uncontrolled trial | 6 | Plasmid | FGF1 | IM | Equivocal |
| Shigematsu | 2010 | controlled trial | 44 | Plasmid | HGF | IM | Positive |
| Powell | 2010 | controlled trial | 27 | Plasmid | HGF | IM | Equivocal |
| Gu | 2011 | patient series/uncontrolled trial | 21 | Plasmid | HGF | IM | Positive |
| Henry | 2011 | patient series/uncontrolled trial | 12 | Plasmid | HGF | IM | Positive |
| Morishita | 2011 | patient series/uncontrolled trial | 22 | Plasmid | HGF | IM | Positive |
| Shigematsu | 2011 | patient series/uncontrolled trial | 10 | Plasmid | HGF | IM | Positive |
| Belch | 2011 | controlled trial | 525 | Plasmid | NV1FGF | IM | Negative |
| Niebuhr | 2012 | controlled trial | 72 | Plasmid | NV1FGF | IM | Negative |
| Makino | 2012 | patient series/uncontrolled trial | 22 | Plasmid | HGF | IM | Positive |
*Positive clinical outcome defined as improved perfusion, reduced pain, or decreased amputation. FGF=fibroblast growth factor; HGF=hepatocyte growth factor; HIF=hypoxia-inducible factor; IM=intramuscular; VEGF=vascular endothelial cell growth factor
TRAFFIC:
In the Therapeutic Angiogenesis with Recombinant Fibroblast Growth Factor-2 (rFGR-2) for Intermittent Claudication (TRAFFIC) study,13 one or two doses of intra-arterial rFGF-2 were infused in 190 patients with intermittent claudication. Of those 190 patients, 174 reached the 90-day outcome mark, and they demonstrated an increase in walking time of 0.60 minutes with placebo, 1.77 minutes with a single dose, and 1.54 minutes with a double dose (P = 0.075). Intra-arterial rFGF-2 resulted in a significant increase in peak walking time at 90 days.
RAVE:
The Regional Angiogenesis with Vascular Endothelial Growth Factor (RAVE) study — a phase 2, double-blind, placebo-controlled study — examined the efficacy and safety of intramuscular delivery of AdVEGF121 (adenoviral VEGF121) in 105 patients.14 The primary efficacy endpoint, change in peak walking time at 12 weeks, did not differ between the placebo, low-dose, and high-dose (1.5±3.1 minutes) groups. Secondary endpoints such as ankle-brachial index (ABI), claudication onset time, and quality-of-life measures were also similar among groups at 12 and 26 weeks. However, in these patients, AdVEGF121 administration was associated with increased peripheral edema.
TALISMAN 201:
This study evaluated the efficacy and safety of intramuscular administration of NV1FGF, a plasmid-based fibroblast growth factor 1, versus placebo in 125 CLI patients presenting with nonhealing ulcer(s).15 Patients were randomized to receive 8 intramuscular injections of placebo or vector on days 1, 15, 30, and 45. Both NV1FGF and placebo showed similar improvements in ulcer healing (19.6% vs. 14.3%, respectively; P = 0.514). However, the use of NV1FGF significantly reduced (by two fold) the risk of all amputations (hazard ratio [HR] 0.498; P = 0.015) and major amputations (HR 0.371 P = 0.015).
WALK:
The WALK trial tested whether intramuscular administration of Ad2/HIF-1α/VP16, an engineered recombinant type 2 adenovirus vector encoding constitutively active HIF-1α (hypoxia-inducible factor 1, alpha subunit), improved walking time in patients with claudication. In this randomized, placebo-controlled study, 289 patients received 20 intramuscular injections of HIF-1α to each leg and were followed for 12 months to determine changes in peak walking time from baseline. Median peak walking time increased by 0.82 minutes in the placebo group and by 0.82 minutes, 0.28 minutes, and 0.78 minutes, respectively, in the three groups with escalating doses of HIF-1α (2×109, 2×1010, and 2×1011) viral particle (P = NS between placebo and each HIF-1α treatment group). There were no significant differences in claudication onset time, ABI, or quality-of-life measurements between the placebo and each HIF-1α group.16
HGF-STAT:
In the Study to Assess the Safety of Intramuscular Injection of Hepatocyte Growth Factor Plasmid to Improve Limb Perfusion in Patients With Critical Limb Ischemia (HGF-STAT trial),17 105 patients received placebo or HGF-plasmid intramuscular injection as follows: 0.4 mg at days 0, 14, and 28 (low dose); 4.0 mg at days 0 and 28 (middle dose); or 4.0 mg at days 0, 14, and 28 (high dose). Adverse events occurred in 86% of the patients, and most were related to CLI or comorbid conditions and were not different between groups. Transcutaneous oxygen tension (TcPO2) increased at 6 months in the high-dose group compared with the placebo, low-dose, and middle-dose groups (ANCOVA P = 0.0015). There was no difference between groups in secondary endpoints, including ABI, toe-brachial index (TBI), pain relief, wound healing, or major amputation. In a follow-on study,18 patients were randomized to 3:1 HGF (n = 21) vs. placebo (n = 6). There was no difference in adverse events or serious adverse events. Change in TBI significantly improved from baseline at 6 months in the HGF-treated group compared with placebo. Change in rest pain assessment by visual analog scale from baseline at 6 months was also significantly improved in the HGF-treated group compared with placebo. Complete ulcer healing at 12 months occurred in 31% of the HGF group and 0% of the placebo (P = .28). At 12 months, there was no difference between groups in major amputation of the treated limb (29% in HGF group vs. 33% in placebo group) or mortality (19% in HGF group vs. 17% in placebo group).
VIROMED:
The purpose of this phase I clinical trial was to evaluate the safety, tolerability, and preliminary efficacy of naked DNA therapy expressing 2 isoforms of hepatocyte growth factor (pCK-HGF-X7) in 22 patients with CLI. Over a 3-month follow-up period, there was a significant reduction in pain observed, a significant increase in the mean ABI value, and a significant rise in the mean TcPO2 value on the dorsum of the foot and anterior and posterior calf. Wound healing improvement was observed in the 6 of 9 patients that had an ulcer at baseline.19
Summary:
A meta-analysis has shown the efficacy of therapeutic angiogenesis in critical ischemia (odds ratio [OR] = 2.20; 95% CI = 1.01-4.79; P = 0.046). There was a slightly significantly higher risk of potential nonserious adverse events (edema, hypotension, proteinuria) in treated patients (OR = 1.81; 95% CI = 1.01-3.38; P = 0.045). However, there were no differences in mortality from any cause, malignancy, or retinopathy.20
Cell Therapy
Recent evidence indicates that bone marrow mononuclear cells (BM-MNC) promote collateral vessel formation in patients with severe peripheral arterial disease (PAD). The BM-MNC from patients with CLI have evidence of an impaired phenotype and a lower number of endothelial progenitor cells compared to normal or those with Buerger’s Disease.21 Multiple strategies have been employed to mobilize and derive cells to improve the performance of cell therapy in CLI. Table 2 shows the numerous patient series and controlled studies that have been performed in this area and the successful reported clinical outcomes.
Table 2.
Results of cell therapy for critical limb ischemia.
| Author | Year | Study Level | N | Product | Clinical Outcome* |
| Tateishi-Yuyama | 2002 | randomized unblinded trial | 45 | BM-MNC | Positive |
| Esato | 2002 | patient series/uncontrolled trial | 8 | BM-MNC | Positive |
| Saigawa | 2004 | patient series/uncontrolled trial | 8 | BM-MNC | Positive |
| Higashi | 2004 | patient series/uncontrolled trial | 8 | BM-MNC | Positive |
| Miyamoto | 2004 | patient series/uncontrolled trial | 12 | BM-MNC | Positive |
| Nizankowski | 2005 | patient series/uncontrolled trial | 10 | BM-MNC | Positive |
| Durdu | 2006 | randomized unblinded trial | 28 | BM-MNC | Positive |
| Bartsch | 2006 | patient series/uncontrolled trial | 10 | BM-MNC | Positive |
| Miyamoto | 2006 | patient series/uncontrolled trial | 8 | BM-MNC | Positive |
| Kajiiguchi | 2007 | patient series/uncontrolled trial | 7 | BM-MNC | Equivocal |
| Huang | 2007 | controlled trial | 74 | BM-MNC | Positive |
| Hernandez | 2007 | patient series/uncontrolled trial | 12 | BM-MNC | Positive |
| Gu | 2008 | patient series/uncontrolled trial | 16 | BM-MNC | Positive |
| Chochola | 2008 | patient series/uncontrolled trial | 28 | BM-MNC | Positive |
| Wester | 2008 | 8 | BM-MNC | Positive | |
| VanTongeren | 2008 | patient series/uncontrolled trial | 27 | BM-MNC | Positive |
| DeVriese | 2008 | patient series/uncontrolled trial | 16 | BM-MNC | Equivocal |
| Amann | 2009 | patient series/uncontrolled trial | 51 | BM-MNC | Positive |
| Prohazka | 2009 | patient series/uncontrolled trial | 37 | BM-MNC | Positive |
| Huang | 2004 | patient series/uncontrolled trial | 5 | PB-MNC | Positive |
| Kawamura | 2005 | patient series/uncontrolled trial | 30 | PB-MNC | Positive |
| Lenk | 2005 | patient series/uncontrolled trial | 7 | PB-MNC | Positive |
| Huang | 2005 | controlled trial | 28 | PB-MNC | Positive |
| Ishida | 2005 | patient series/uncontrolled trial | 6 | PB-MNC | Positive |
| Kawamura | 2006 | patient series/uncontrolled trial | 75 | PB-MNC | Positive |
| Huang | 2007 | controlled trial | 76 | PB-MNC | Positive |
| Powell | 2011 | controlled trial | 86 | BM-TRC | Positive |
| Walter | 2011 | controlled trial | 40 | BM-MNC | Equivocal |
*Positive clinical outcome defined as improved perfusion, reduced pain, or decreased amputation. BM-MNC=bone marrow mononuclear cells; BM-TRC=bone marrow tissue repair cells; PB-MNC=peripheral blood mononuclear cells
INTRAMUSCULAR BM-MNC:
The first large report on the use of BM-MNC in limb ischemia was the Therapeutic Angiogenesis Using Cell Transplantation (TACT) study. Intramuscular injection of autologous bone marrow mononuclear cells resulted in a 3-year amputation-free rate of 60% (95% CI 46–74). There was significant improvement in the leg pain scale and ulcer size, and pain-free walking distance was maintained during at least 2 years after the therapy, although the ABI and TcPO2 value did not significantly change.22 In the Bone Marrow Outcomes Trial 1 (BONMOT-1) examining intramuscular injections of autologous bone marrow cells (BMC), 51 patients with impending major amputation due to severe CLI had a limb salvage rate of 59% at 6 months and 53% at last follow-up (mean 411±261 days, range 175–1186).23 Perfusion measured with ABI and TcPO2 at baseline and after 6 months increased in patients with consecutive limb salvage (ABI 0.33±0.18 to 0.46±0.15, TcPO2 12±12 mmHg to 25±15 mmHg) but did not change in patients eventually undergoing major amputation. Clinically most important, patients who did not require amputation saw an improvement in mean Rutherford category from a baseline of 4.9 to 3.3 at 6 months (P = 0.0001). Furthermore, analgesics consumption was reduced by 62%.23 In BONMOT-1 and the subsequent placebo-controlled, double-blind study (BONMOT-CLI), BM-MNC injections were placed along the axial line of the occluded native arteries; this maximizes efficacy because the density of preformed collaterals is highest in parallel orientation to the axial arteries, which is the location for collateral growth. In BONMOT-1 and BONMOT-CLI, the number of injections was also increased corresponding to the length of the arterial occlusion, from 40 injections for infra-popliteal disease only to 60 injections when femoral, popliteal, and infra-popliteal disease was present. In the RESTORE–CLI trial of 77 patients, Ixmyelocel-T treatment led to a significantly prolonged first occurrence of treatment failure (e.g., major amputation of injected leg, all-cause mortality, doubling of total wound surface area from baseline, de novo gangrene) (P = 0.0032, logrank test). Amputation-free survival (major amputation of injected leg; all-cause mortality) saw a 32% reduction, but this was not statistically significant (P = 0.3). Treatment effect in post hoc analyses of patients with baseline wounds was more pronounced.24 In the interim analysis of the HARVEST trial, the BMAC (bone marrow aspirate concentrate) group demonstrated trends toward improvement in amputation, pain, quality of life, Rutherford classification, and ABI when compared with controls.25
INTRA-ARTERIAL BM-MNC:
In the PROVASA (Intra-arterial Progenitor Cell Transplantation of Bone Marrow Mononuclear Cells for Induction of Neovascularization in Patients With Peripheral Arterial Occlusive Disease) study, 40 patients were randomized to intra-arterial delivery of either BM-MNC or placebo.26 There was a significant improvement in ulcer healing and significant rest pain reduction in subjects treated with BM-MNC versus placebo. The trial also demonstrated that multiple treatments of BM-MNC were associated with significantly greater improvements in ulcer healing and rest pain than a single treatment. However, patients with Rutherford class ischemia (gangrene or major tissue loss) at baseline did not respond to therapy. The major predictors of successful ulcer healing were total cell number delivered, repeated cell administration, and greater cell functionality measured by in vitro assays.
INTRA-ARTERIAL AND INTRAMUSCULAR BM-MNC:
Combined intra-arterial (IA) plus intramuscular (IM) BMC delivery may be more effective than exclusive intramuscular injections. In a study by Franz et al., a dual intramuscular and intra-arterial autologous BM-MNC implantation strategy was employed in nine patients for whom limb amputation was recommended. Following the procedure, there was no significant improvement in ABI. Three (33.3%) underwent major amputations. The remaining six patients demonstrated an improvement in rest pain. Complete wound healing was achieved within 3 months in all patients who had ulcers.27 In a study comparing exclusive IM (n = 12) versus combined IM plus IA (n = 15) delivery of autologous BMC, there were no adverse reactions related to injection of the cells.28 Two patients in the IA plus IM group required limb amputation because of ongoing critical ischemia versus seven patients in the IM group (P = 0.17). The remaining patients had a significant and sustained (>12 months) improvement in pain-free walking, mean ABI, and pain scores within 6 weeks follow-up.28 Similar findings were seen in the TAM-PAD study.29
Summary:
The Cochrane database included only two small studies (57 patients); these showed that the treatment groups experienced a greater reduction in rest pain (P < 0.001) and an increase in ABI with a statistically significant increase in pain-free walking distance (mean increase 306.4 m versus 78.6 m, P = 0.007) compared to the control groups. However, a smaller proportion of participants underwent amputation in the treatment group compared with the control group (0% versus 36%, P = 0.007).30 The authors stressed the need for further randomized controlled clinical trials to interpret the impact of cell therapy on clinical outcomes.
In a meta-analysis of autologous cell therapy to treat patients with critical limb ischemia, researchers identified 37 trials (controlled and noncontrolled, randomized and nonrandomized trials) using autologous bone marrow or granulocyte colony stimulating factor (G-CSF) mobilized peripheral blood cells.31 Autologous cell therapy was effective in improving surrogate indexes of ischemia, subjective symptoms, and hard endpoints (ulcer healing and amputation). On the contrary, G-CSF monotherapy was not associated with significant improvement in the same endpoints. Patients with thromboangiitis obliterans showed some larger benefits than patients with atherosclerotic CLI. The intramuscular route of administration and the use of bone marrow cells seemed somehow more effective than intra-arterial administration and the use of mobilized peripheral blood cells. This meta-analysis indicates that intramuscular autologous bone marrow cell therapy is a feasible, relatively safe, and potentially effective therapeutic strategy for PAD patients who are not candidates for traditional revascularization.
Cardiovascular Molecular and Cell Therapy Program at Methodist
The Cardiovascular Molecular and Cell Therapy Program at The Methodist Hospital is a multidisciplinary program that promotes clinical trials in cardiovascular disease. The program is concentrating on three molecular therapeutic areas in the cardiovascular arena: critical limb ischemia, congestive heart failure, and heart-lung transplantation. The multidisciplinary team is composed of surgeons (vascular, cardiac, and transplant), physicians (cardiologists and pulmonologists), and scientists (molecular, cell, and nano sciences), and it runs investigator-initiated trials and recruits subjects to national trials. The program is a leading recruiter in gene and cell therapy for limb salvage in CLI, and in cell therapy for patients with heart failure. Other interests are the use of cell therapy as an adjunctive in coronary artery bypass graft and left ventricular assist device surgeries, and the modulation of the biology of organ transplant prior to implantation using nanotechnology. Methodist is one of few sites to be involved in studies on the molecular therapeutic modulation of peripheral angioplasty sites in an attempt to locally influence restenosis rates. The Cardiovascular Molecular and Cell Therapy Program is a crucial element in advanced therapy for limb salvage and offers patients with no interventional options a biological alternative not available at many centers.
Conflict of Interest Disclosures: The author has completed and submitted the Methodist DeBakey Cardiovascular Journal Conflict of Interest Statement and none were reported.
Funding/Support: The author has no funding disclosures.
References
- 1.Lawall H, Bramlage P, Amann B. Stem cell and progenitor cell therapy in peripheral artery disease. A critical appraisal. Thromb Haemost. 2010;103:696–709. doi: 10.1160/TH09-10-0688. [DOI] [PubMed] [Google Scholar]
- 2.Adam DJ, Beard JD, Cleveland T, Bell J, Bradbury AW, Forbes JF. Bypass versus angioplasty in severe ischaemia of the leg (BASIL): multicentre randomised controlled trial. Lancet. 2005;366:1925–34. doi: 10.1016/S0140-6736(05)67704-5. [DOI] [PubMed] [Google Scholar]
- 3.Hirsch AT, Haskal ZJ, Hertzer NR, Bakal CW, Creager MA, Halperin JL. ACC/AHA 2005 Practice Guidelines for the management of patients with peripheral arterial disease (lower extremity renal, mesenteric, and abdominal aortic): a collaborative report from the American Association for Vascular Surgery/Society for Vascular Surgery, Society for Cardiovascular Angiography and Interventions, Society for Vascular Medicine and Biology, Society of Interventional Radiology, and the ACC/AHA Task Force on Practice Guidelines (Writing Committee to Develop Guidelines for the Management of Patients With Peripheral Arterial Disease): endorsed by the American Association of Cardiovascular and Pulmonary Rehabilitation; National Heart, Lung, and Blood Institute; Society for Vascular Nursing; TransAtlantic Inter-Society Consensus; and Vascular Disease Foundation. Circulation. 2006;113:e463–654. doi: 10.1161/CIRCULATIONAHA.106.174526. [DOI] [PubMed] [Google Scholar]
- 4.Risau W. Mechanisms of angiogenesis. Nature. 1997;386(6626):671–4. doi: 10.1038/386671a0. [DOI] [PubMed] [Google Scholar]
- 5.Hirota K, Semenza GL. Regulation of angiogenesis by hypoxiainducible factor 1. Crit Rev Oncol Hematol. 2006;59:15–26. doi: 10.1016/j.critrevonc.2005.12.003. [DOI] [PubMed] [Google Scholar]
- 6.Augustin H. Tubes branches, and pillars: The many ways of forming a new vasculature. Circ Res. 2001;89:645–7. [PubMed] [Google Scholar]
- 7.Hirschi K, Goodell M. Common origins of blood and blood vessels in adults? Differentiation. 2001;68(4-5):186–92. doi: 10.1046/j.1432-0436.2001.680406.x. [DOI] [PubMed] [Google Scholar]
- 8.Zhu S, Liu X, Li Y, Goldschmidt-Clermont PJ, Dong C. Aging in the atherosclerosis milieu may accelerate the consumption of bone marrow endothelial progenitor cells. Arterioscler Thromb Vasc Biol. 2007;27:113–9. doi: 10.1161/01.ATV.0000252035.12881.d0. [DOI] [PubMed] [Google Scholar]
- 9.van Royen N, Piek JJ, Buschmann I, Hoefer I, Voskuil M, Schaper W. Stimulation of arteriogenesis; a new concept for the treatment of arterial occlusive disease. Cardiovasc Res. 2001;49:543–53. doi: 10.1016/s0008-6363(00)00206-6. [DOI] [PubMed] [Google Scholar]
- 10.Asahara T, Masuda H, Takahashi T, Kalka C, Pastore C, Silver M, et al. Bone marrow origin of endothelial progenitor cells responsible for postnatal vasculogenesis in physiological and pathological neovascularization. Circ Res. 1999;85:221–8. doi: 10.1161/01.res.85.3.221. [DOI] [PubMed] [Google Scholar]
- 11.Baumgartner I, Pieczek A, Manor O, Blair R, Kearney M, Walsh K, et al. Constitutive expression of phVEGF165 after intramuscular gene transfer promotes collateral vessel development in patients with critical limb ischemia. Circulation. 1998;97(12):1114–23. doi: 10.1161/01.cir.97.12.1114. [DOI] [PubMed] [Google Scholar]
- 12.Isner JM. Arterial gene transfer of naked DNA for therapeutic angiogenesis: early clinical results. Adv Drug Deliv Rev. 1998;30(1-3):185–97. doi: 10.1016/s0169-409x(97)00115-4. [DOI] [PubMed] [Google Scholar]
- 13.Lederman RJ, Mendelsohn FO, Anderson RD, Saucedo JF, Tenaglia AN, Hermiller JB, et al. Therapeutic angiogenesis with recombinant fibroblast growth factor-2 for intermittent claudication (the TRAFFIC study): a randomised trial. Lancet. 2002;359(9323):2053–8. doi: 10.1016/s0140-6736(02)08937-7. [DOI] [PubMed] [Google Scholar]
- 14.Rajagopalan S, Mohler ER, 3rd, Lederman RJ, Mendelsohn FO, Saucedo JF, Goldman CK, et al. Regional angiogenesis with vascular endothelial growth factor in peripheral arterial disease: a phase II randomized, double-blind, controlled study of adenoviral delivery of vascular endothelial growth factor 121 in patients with disabling intermittent claudication. Circulation. 2003;108(16):1933–8. doi: 10.1161/01.CIR.0000093398.16124.29. [DOI] [PubMed] [Google Scholar]
- 15.Nikol S, Baumgartner I, Van Belle E, Diehm C, Visoná A, Capogrossi MC, et al. Therapeutic angiogenesis with intramuscular NV1FGF improves amputation-free survival in patients with critical limb ischemia. Mol Ther. 2008;16(5):972–8. doi: 10.1038/mt.2008.33. [DOI] [PubMed] [Google Scholar]
- 16.Creager MA, Olin JW, Belch JJ, Moneta GL, Henry TD, Rajagopalan S, et al. Effect of hypoxia-inducible factor-1alpha gene therapy on walking performance in patients with intermittent claudication. Circulation. 2011;124(16):1765–73. doi: 10.1161/CIRCULATIONAHA.110.009407. [DOI] [PubMed] [Google Scholar]
- 17.Powell RJ, Simons M, Mendelsohn FO, Daniel G, Henry TD, Koga M, et al. Results of a double-blind, placebo-controlled study to assess the safety of intramuscular injection of hepatocyte growth factor plasmid to improve limb perfusion in patients with critical limb ischemia. Circulation. 2008;118(1): 58–65. doi: 10.1161/CIRCULATIONAHA.107.727347. [DOI] [PubMed] [Google Scholar]
- 18.Powell RJ, Goodney P, Mendelsohn FO, Moen EK, Annex BH. HGF-0205 Trial Investigators. Safety and efficacy of patient specific intramuscular injection of HGF plasmid gene therapy on limb perfusion and wound healing in patients with ischemic lower extremity ulceration: results of the HGF-0205 trial. J Vasc Surg. 2010;52(6):525–30. doi: 10.1016/j.jvs.2010.07.044. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 19.Gu Y, Zhang J, Guo L, Cui S, Li X, Ding D, et al. A phase I clinical study of naked DNA expressing two isoforms of hepatocyte growth factor to treat patients with critical limb ischemia. J Gene Med. 2011;13(11):602–10. doi: 10.1002/jgm.1614. [DOI] [PubMed] [Google Scholar]
- 20.De Haro J, Acin F, Lopez-Quintana A, Florez A, Martinez-Aguilar E, Varela C. Meta-analysis of randomized controlled clinical trials in angiogenesis: gene and cell therapy in peripheral arterial disease. Heart Vessels. 2009;24(5):321–8. doi: 10.1007/s00380-008-1140-z. [DOI] [PubMed] [Google Scholar]
- 21.Idei N, Soga J, Hata T, Fujii Y, Fujimura N, Mikami S, et al. Autologous bone-marrow mononuclear cell implantation reduces long-term major amputation risk in patients with critical limb ischemia: a comparison of atherosclerotic peripheral arterial disease and Buerger disease. Circ Cardiovasc Interv. 2011;4:15–25. doi: 10.1161/CIRCINTERVENTIONS.110.955724. [DOI] [PubMed] [Google Scholar]
- 22.Matoba S, Tatsumi T, Murohara T, Imaizumi T, Katsuda Y, Ito M, et al. Long-term clinical outcome after intramuscular implantation of bone marrow mononuclear cells (Therapeutic Angiogenesis by Cell Transplantation [TACT] trial) in patients with chronic limb ischemia. Am Heart J. 2008;156(5):1010–8. doi: 10.1016/j.ahj.2008.06.025. [DOI] [PubMed] [Google Scholar]
- 23.Amann B, Luedemann C, Ratei R, Schmidt-Lucke JA. Autologous bone marrow cell transplantation increases leg perfusion and reduces amputations in patients with advanced critical limb ischemia due to peripheral artery disease. Cell Transplant. 2009;18(3):371–80. doi: 10.3727/096368909788534942. [DOI] [PubMed] [Google Scholar]
- 24.Powell RJ, Marston WA, Berceli SA, Guzman R, Henry TD, Longcore AT, et al. Cellular therapy with Ixmyelocel-T to treat critical limb ischemia: the randomized, double-blind, placebo-controlled RESTORE-CLI trial. Mol Ther. doi: 10.1038/mt.2012.52. 2012 Mar 27. doi: 10.1038/mt.2012.52. [Epub ahead of print] [DOI] [PMC free article] [PubMed] [Google Scholar]
- 25.Iafrati MD, Hallett JW, Geils G, Pearl G, Lumsden A, Peden E, et al. Early results and lessons learned from a multicenter, randomized, double-blind trial of bone marrow aspirate concentrate in critical limb ischemia. J Vasc Surg. 2011;54(6):1650–8. doi: 10.1016/j.jvs.2011.06.118. [DOI] [PubMed] [Google Scholar]
- 26.Walter DH, Krankenberg H, Balzer J, Kalka C, Baumgartner I, Schluter M, et al. Intraarterial administration of bone marrow mononuclear cells in patients with critical limb ischemia: a randomized-start, placebo-controlled pilot trial (PROVASA). Circ Cardiovas Interv. 2011;4:26–37. doi: 10.1161/CIRCINTERVENTIONS.110.958348. [DOI] [PubMed] [Google Scholar]
- 27.Franz RW, Parks A, Shah KJ, Hankins T, Hartman JF, Wright ML. Use of autologous bone marrow mononuclear cell implantation therapy as a limb salvage procedure in patients with severe peripheral arterial disease. J Vasc Surg. 2009;50(5):1378–90. doi: 10.1016/j.jvs.2009.07.113. [DOI] [PubMed] [Google Scholar]
- 28.Van Tongeren RB, Hamming JF, Fibbe WE, Van Weel V, Frerichs SJ, Stiggelbout AM, et al. Intramuscular or combined intramuscular/intra-arterial administration of bone marrow mononuclear cells: a clinical trial in patients with advanced limb ischemia. J Cardiovasc Surg (Torino). 2008;40(1):51–8. [PubMed] [Google Scholar]
- 29.Bartsch T, Brehm M, Zeus T, Kögler G, Wernet P, Strauer BE. Transplantation of autologous mononuclear bone marrow stem cells in patients with peripheral arterial disease (the TAM-PAD study). Clin Res Cardiol. 2007;96(12):891–9. doi: 10.1007/s00392-007-0569-x. [DOI] [PubMed] [Google Scholar]
- 30.Moazzami K, Majdzadeh R, Nedjat S. Local intramuscular transplantation of autologous mononuclear cells for critical lower limb ischaemia. Cochrane Database Syst Rev. 2011;Dec 7(12):CD008347. doi: 10.1002/14651858.CD008347.pub2. [DOI] [PubMed] [Google Scholar]
- 31.Fadini GP, Agostini C, Avogaro A. Autologous stem cell therapy for peripheral arterial disease meta-analysis and systematic review of the literature. Atherosclerosis. 2010;209(1):10–7. doi: 10.1016/j.atherosclerosis.2009.08.033. [DOI] [PubMed] [Google Scholar]
- 32.Adams RH, Alitalo K. Molecular recognition of angiogenesis and lymphangiogenesis. Nat Rev Mol Cell Biol. 2007 Jun;8(6):464–78. doi: 10.1038/nrm2183. [DOI] [PubMed] [Google Scholar]


