Abstract
HIV-patients have excess of pneumococcal infection. We immunized 40 HIV-patients twice with pneumococcal conjugate vaccine (Prevnar, Pfizer) +/− a TLR9 agonist (CPG 7909). Peripheral blood mononuclear cells were stimulated with pneumococcal polysaccharides and cytokine concentrations measured. The CPG 7909 adjuvant group had significantly higher relative cytokine responses than the placebo group for IL-1β, IL-2R, IL-6, IFN-γ and MIP-β, which, did not correlate with IgG antibody responses. These findings suggests that CPG 7909 as adjuvant to pneumococcal conjugate vaccine induces cellular memory to pneumococcal polysaccharides in HIV-patients, independently of the humoral response.
Keywords: pneumococcal vaccine, cellular memory, CpG-ODN, adjuvants, HIV, TLR9
Streptococcus pneumoniae (pneumococci) are one of the leading global causes of otitis media, pneumonia and meningitis. Children, elderly and people with immunodefiencies are at higher risk of invasive pneumococcal disease. Among patients infected with HIV (human immunodeficiency virus) pneumococci are the most frequent cause of pneumonia and invasive bacterial infection.1 Even in countries with widespread use of highly active antiretroviral treatment (HAART), a significant higher risk of pneumonia persists among HIV-patients compared with the background.2 Therefore, development of more immunogenic pneumococcal vaccines for immunocompromised individuals, such as HIV-patients, is of great importance.
Anti-pneumococcal antibodies are considered as most important to prevent invasive pneumococcal disease. Although cellular immunity is believed to protect against pneumococcal colonization,3 present knowledge of polysaccharide-directed cellular immunity and its clinical significance is limited.
Toll-like receptors (TLRs) are part of the innate immune system. TLR9 senses unmethylated CpG motifs present in genomic DNA from bacteria and viruses. In humans, TLR9 is expressed on B cells and plasmacytoid dendritic cells.4 Stimulation of TLR9 activates the innate immune system and leads to release of inflammatory cytokines. Using CpG oligodeoxynucleotides as a vaccine adjuvant during immunization enhances a Th1-response, and may lead to IgG class switch.5
We have recently shown that both the quantity and quality of pneumococcal polysaccharide (PS) specific antibodies are enhanced when adjuvanting the heptavalent pneumococcal conjugate vaccine (PCV7) with CPG 7909.6 We hypothesized, that the use of a TLR9 agonist as an adjuvant with PCV7 would induce cellular memory to pneumococcal PS (PnPS).
Patients and Samples
Forty HIV-infected individuals enrolled in a double-blind, placebo-controlled phase 1b/2a trial previously described,6 were randomized to receive a double standard dose of PCV7 (Prevnar®, Pfizer) + 1 mg CPG 7909 or placebo (PBS similar in color and viscosity) at 0 and 3 mo. All patients gave written informed consent. Blood samples were collected at 0 and 4 mo to isolate sera (-80°C) and peripheral blood mononuclear cells (PBMC) (-170°C).
Antibody Measurements
Total IgG, IgG1, and IgG2, specific to capsular pneumococcal PS antigens for the serotypes 6B and 14, was measured by enzyme-linked immunosorbent assay (ELISA) using WHO guidelines,7 with some minor modifications. Briefly, polystyrene MaxiSorp™ microtiter plates (Nunc) were coated with purified pneumococcal PS (American Type Culture Collection, LGC Standards) serotype 6B and 14 (2.5 μg/ml in sterile PBS). Plates were incubated at 37°C in a humidified chamber (5% CO2) for 5 h and stored at 4°C. Human sera were adsorbed for 30 min with CWPS Multi (cell wall polysaccharide) (Statens Serum Institut) (10 µg/ml). The 89-SF US Reference Pneumococcal antiserum (kindly provided by Dr Milan S. Blake, FDA) was adsorbed with C-PS (Statens Serum Institut) (5 µg/ml) as recommended. The plates were washed 5 times with washing buffer (1X TBS /0.1% Brij solution). Sera was added in six-fold serial dilutions (50 µl/well) in duplicates and incubated for 18 h at room temperature. After incubation, plates were washed and polyclonal secondary antibody conjugate was added as described below.
For determination of IgG and IgG2 levels, secondary alkaline phosphatase-conjugated mouse monoclonal anti-human IgG (1:2,000) or IgG2 (1:500) antibody (Zymed Laboratories, Inc.) was added to the plates. After 2 h, plates were washed and 100 µl p-NitroPhenylPhosphate (Kem-En-Tec Diagnostics) was added to all wells and incubated for 15–30 min. The reaction was stopped with 100 µl of 1.0 M NaOH.
For determination of IgG1 levels, a secondary mouse monoclonal anti-human IgG1 biotin-conjugated antibody (Sigma) was diluted (1:1,000) and added to the plates. After 2 h plates were washed and streptavidin-HRP (R&D Systems) was added for 30 min. After washing, 100 µl of TMB-plus substrate (3,3′, 5, 5′- tetramethyl-benzidine) (Kem-En-Tec Diagnostics, Copenhagen, Denmark) was added. The reaction was stopped with 1.2 M H2SO4.
For IgG and IgG2 the ELISA plates optical density was evaluated spectrophotometrically at 405 nm with 690 nm as references. For IgG1 measurements were done at 450 nm with 540 nm as references. Measurements were performed on a FLUOstar Omega microplate reader (BMG LABTECH GmbH, Offenburg, Germany). Antibody concentrations were calculated using the 89SF standard serum as reference using 4-parameter fit. Duplicates that deviated by more than 10% were discarded. To account for inter-plate variation, we obtained a standardized pneumococcal quality control serum (kindly provided by Dr. David Goldblatt, National Institute of Biological Standards and Control, England).
Cytokine Measurements
Frozen PBMCs were quickly thawed and suspended in 9 ml warm (37°C) phosphate buffered saline (PBS) (pH = 7.4). The cells were centrifuged at 800 rpm (110 x g) for 10 min and the pellet re-suspended in 5 ml warm RPMI 1640 tissue culture medium (Gibco BRL, Gaithersburg, Md.) supplemented with 10% heat-inactivated fetal calf serum and 100 U/ml penicillin and 100 µg/ml streptomycin. After centrifugation the cells were tested for viability using Trypan Blue and re-suspended in warm RPMI to 1 × 106 cells/ml. A total of 150 µl (1.5 × 105 cells/well) were transferred to a 96-well flat-bottomed tissue culture plate (Sarstedt. Inc.). The cells were incubated overnight at 37°C and then stimulated with 20 µl of pneumococcal PS serotypes 6B and 14, respectively (final concentration 20 µg/ml) or 20 µl of LPS (lipopolysaccharide) (final concentration 100 ng/ml) or 20 µl of media. After incubation for 48 h, cell culture media was collected and stored at -80°C. Cytokine concentrations were measured by a multiplex bead assay (Invitrogen) on the Luminex platform (Luminex 100, Luminex Corp.). For baseline and post vaccination respectively, we calculated a stimulation index (SI) as follows: SI = [PnPS stimulated/unstimulated]. The relative cytokine response (RR) was calculated as a ratio between post vaccination and baseline SI: RR = (SIpost vaccination/SIbaseline).
Statistics
All statistical analyses were made on log-transformed data. For Gaussian distributed data we used an unpaired t test, otherwise Mann-Whitney test was used. Correlations were made using Pearson’s r coefficient for normally distributed data - otherwise Spearman’s rank. We used Stata software, version 9.2 (StataCorp, TX) for statistical analyses.
Study Population
The two groups were similar in age, sex, HAART-treatment, CD4-level, and viral load, at time of inclusion (Table 1).
Table 1. Baseline characteristics and antibody response.
| Baseline characteristics | |||
|---|---|---|---|
| |
|
CpG group, n (%) |
Placebo group, n (%) |
| |
|
n = 20 (50) |
n = 20 (50) |
| Sex |
|
|
|
| |
Male |
16 (80) |
16 (80) |
| |
Female |
4 (20) |
4 (20) |
| Median age, years (IQR) |
|
45.5 (40–56.5) |
47.5 (41.5–54) |
| Median CD4+ cell count, cells/µL (IQR) |
470 (375–775) |
510 (430–830) |
|
| Median log HIV RNA |
On HAART |
1.60 |
1.60 |
| Median log HIV RNA |
No HAART (IQR) |
3.37–4.68 |
3.72–5.03 |
| On HAART |
|
10 (50) |
10 (50) |
|
IgG subclass response (serotypes 6B+14) |
|
|
|
| |
|
CpG group |
Placebo group |
| IgG1 GMC |
Pre immunization |
0.14 (0.10–0.21) |
0.14 (0.10–0.22) |
| |
Post immunization |
0.71 (0.43–1.18) |
0.69 (0.38–1.23) |
| |
Ratio post/pre |
5.07 |
4.93 |
| IgG2 GMC |
Pre immunization |
0.48 (0.25–0.92) |
0.45 (0.22–0.94) |
| |
Post immunization |
4.68 (2.44–8.97) |
4.54 (2.11–9.75) |
| |
Ratio post/pre |
9.75 |
10.03 |
| IgG2/IgG1 Ratio |
Pre immunization |
3.43 |
3.21 |
| |
Post immunization |
6.59 |
6.58 |
| Ratio post/pre | 1.92 | 2.05 | |
CpG Does Not Affect Antibody Class Switch
The IgG1 and IgG2 antibody titers for the serotypes 6B and 14 were calculated as geometric mean concentrations (GMC) (Table 1). There were no differences in pre-vaccination and post-vaccination GMCs between the CPG 7909 group and the placebo group when comparing IgG1 and IgG2 levels or comparing the relative IgG1 and IgG2 response. Further, both pre- and post-vaccination IgG2/IgG1 ratios were similar between the two groups. Thus, adjuvanting Prevnar with CPG 7909 does not affect antibody class switch.
Increased Proinflammatory Cytokines Response to PnPS in CpG-Adjuvanted Group
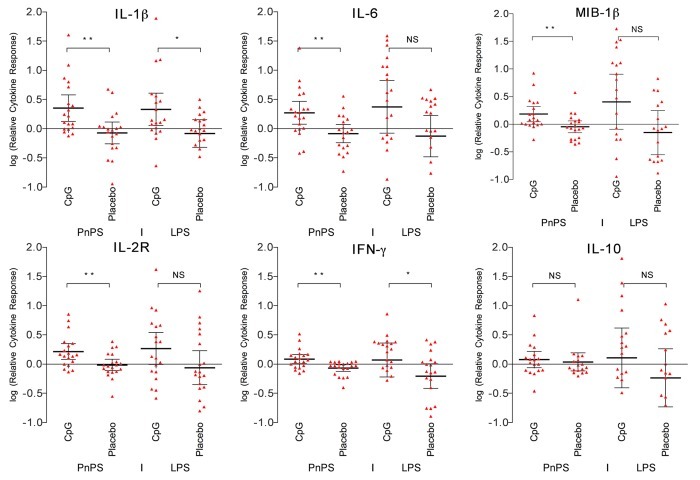
One month after the second vaccination, PBMCs from persons in the CPG 7909 group elicited an overall higher relative cytokine response to PnPS stimulation compared with the placebo group (Fig. 1). The response was statistically significant for the cytokines IL-1β (p = 0.0046), IL-6 (p = 0.0051), IFN-γ (p = 0.0047), the chemokine MIP-β (CCL4) (p = 0.0086), as well as the soluble receptor IL-2R (p = 0.0062). However, for the anti-inflammatory cytokine IL-10 we did not find a difference between the two groups (p = 0.37).
Figure 1. Cytokine response to PnPS and LPS. Relative cytokine response between baseline response and response after two doses of PCV7. The CPG 7909 group elicited an overall higher response to PnPS stimulation compared with the placebo group. IL-1β: p = 0.0046; IL-6: p = 0.0051; IFN-γ: p = 0.0047; MIP-β: p = 0.0086; IL-2R: p = 0.0062; IL-10: p = 0.37. NS = not significant (p > 0.05). * p < 0.05, ** p < 0.01.
We did not observe any influence of neither CD4 count nor viral load on PS induced relative cytokine responses (data not shown).
Evaluation of LPS-Response in CpG-Adjuvanted and Non-Adjuvanted Groups
As control we stimulated the PBMCs with LPS. A higher relative response in favor of the CpG group for two cytokines was observed. The difference was statistically significant for IL-1β (p = 0.038) and IFN-γ (p = 0.019) (Fig. 1).
Correlation of LPS and PnPS for IFN-γ and IL-1β
IFN-γ: CpG r = 0.31, p = NS vs. placebo r = 0.27, p = NS. IL-1β: CpG r = 0.68, p = < 0.01 vs. placebo r = 0.46, p = < 0.05.
No Correlation of Humoral and Cellular Response
We tested whether there were any correlations between the PS specific antibody response and the cellular response (Fig. 2). We found no correlations between the humoral and cellular immune components.
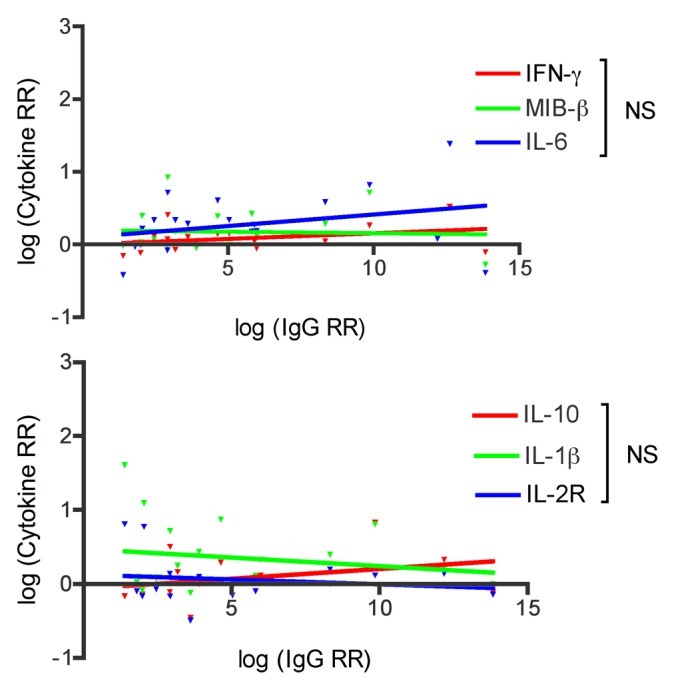
Figure 2. Correlation between IgG relative response (RR) and cytokine RR to PnPS serotypes 6B and 14. CpG group only showed. No correlations between the humoral and cellular immune response is observed: IFN-γ: r = 0.35, p = 0.13; MIB-β: r = 0.20, p = 0.40; IL-6: r = 0.29, p = 0.21; IL-10: r = 0.45, p = 0.06; IL-1b: r = -0.02, p = 0.93; IL-2R: r = -0.005, p = 0.99. NS = not significant (p > 0.05)
Discussion
In this study, we found that a TLR9 agonist adjuvant (CPG 7909) increases pro-inflammatory cytokine release in response to PnPS stimulation of PBMCs from adults with HIV infection following a two-dose PCV7 vaccination regimen (Fig. 1). The CPG 7909-induced effect on cytokine release did not correlate with the IgG response. This finding may indicate that TLR9 agonist adjuvants could be used to enhance antibody-independent cellular immunity to pneumococcal PS.
Recently, we described that adjuvanting PCV7 with CPG 7909 results in higher anti-pneumococcal PS antibody response compared with the non-adjuvanted group.6 Here we show that the immune-modulatory effect of this adjuvant, however, does not seem to affect the overall IgG1 and IgG2 subclass distribution (Table 1).
In contrast, we demonstrate the ability of human PBMCs to recognize purified pneumococcal PS after immunization with a conjugate pneumococcal PS vaccine. The cytokine responses demonstrate the in vivo effect of CPG 7909. The data are corrected for preexisting immunological memory, as the cytokine response was defined as a relative response, taking into consideration the baseline response. Therefore, we argue that the results shown reflect the immunological memory to serotype specific pneumococcal PS induced by immunization. Furthermore, the PBMCs were stimulated with highly purified polysaccharides, and not the conjugated polysaccharides contained in the vaccine. This rules out that the cytokine response could be caused by recognition of the carrier protein CRM197, which supports that the cellular recognition is polysaccharide specific.
Overall, the results showed an increase in antigen-induced cytokine production in PBMCs from persons in the CPG 7909 group, which were highly significant for the Th1-cytokine IFN-γ, the inflammatory markers IL-1β and IL-6, the chemokine MIP-1β, as well as soluble IL-2 receptor (Fig. 1). IFN-γ is considered a Th1 cytokine secreted primarily by T cells and natural killer cells, and the IFN-γ enzyme-linked immunosorbent spot assay is widely used as a correlate of T cell memory. B cells responds poorly to purified pneumococcal PS in vitro,8 and although IFN-γ can be produced by B cells, we find it unlikely that recognition by the memory B cell receptor solely could lead to the data shown in this study. Thus, other leukocytes are likely to be involved in the observed immunological response.
It is believed, that the polysaccharide-specific B cell internalizes and processes the carrier protein from the protein–polysaccharide conjugate, and presents the peptides to specific T cells, which then supplies T cell help for the differentiation of plasma cells and memory B cells.9 However, it has been shown that the polysaccharide portion of the conjugate vaccine enters the antigen-presenting cell, and later co-localizes with MHC II on the cell surface, still attached to the carrier protein CRM197.10 This important finding makes it plausible that the pneumococcal PS, and not just the carrier protein, could indeed be presented to T cells and/or other cells of the immune system, independently of B cells. The fact, that the humoral and cellular response in our study is not correlated supports this theory. Recently Avci FY et al. described how processed glycoconjugates leads to presentation of carbohydrate epitopes, which are able to stimulate carbohydrate-specific CD4+ T cells.11 Based on our data, we find it likely that CpG could help facilitate the induction of carbohydrate-specific T cells—although a tetramer staining, if possible, would have provided more substantial evidence.
Another possible contributing factor explaining increased cellular memory to PnPS could be recognition via C-type lectins, such as e.g., dendritic cell-specific intercellular adhesion molecule-3-Grabbing Non-integrin (DC-SIGN). C-type lectins are an important part of the innate host response against glycoepitopes, such as pneumococcal capsular PS or LPS. Some of the most invasive pneumococcal serotypes are not recognized by C-type lectins in the lungs, emphasizing the importance of these receptors in pathogen protection.12 Triggering DC-SIGN by an antigen leads to acetylation of nuclear factor kappa B (NF-κB) and cytokine production, but only after NF-κB is activated by a TLR facilitated signal.13 Hence, TLR9 signaling may be the key factor in the induction of the type of memory reported in our study.
Some of the most effective empirically derived vaccines are characterized by their ability to stimulate multiple innate microbe sensors, including TLRs.14 In our study, cellular memory was observed only in the CpG group, which may indicate that a dual signal is required to induce cellular memory. The PCV7 has been shown to activate TLR2.15 So when adding CpG as adjuvant to the PCV7, TLR9 and TLR2 are activated respectively. The immune system might interpret this as a live S. pneumoniae, which interestingly signals through both TLR2 and TLR9.16
We find it plausible that adjuvanting a conjugated polysaccharide vaccine with CpG could induce a kind of sub-phenotypic maturation of dendritic cells, and thereby represent some sort of complex innate memory—a linkage between innate and adaptive immunity. Further, a sub-phenotypic maturation of dendritic cells could also explain why we observe the enhanced IFN-γ and IL-1β responses to LPS stimulation, since LPS and pneumococcal PS can have shared epitopes.17 We examined the correlation between LPS and PNPS, and found none for IFN-γ. However, for IL-1β we observed a positive correlation for both the CpG group and the placebo group, which indeed could indicate increased innate recognition, not facilitated by CpG. This might slightly affect the interpretation of the PnPS induced IL-1β response, which should be kept in mind considering our data.
Prevnar includes aluminum phosphate. In animal models Alum has been shown to significantly improve the efficiency of CpG ODN as adjuvant,18 and the aluminum phosphate in Prevnar is probably necessary for the CpG-induced memory shown in this setting. The exact mechanism by which Alum enhances the immunostimulatory effects of CpG ODNs is yet to be determined. However, it is believed to be important for the CpG to be bound to the Alum to elicit its effect.19
In conclusion, our findings indicate the potential in inducing cellular memory to pneumococcal polysaccharides and may have important implications for the development of new pneumococcal vaccines. To our knowledge, this is the first human study to address pneumococcal polysaccharide-specific cellular memory following CpG-adjuvanted immunization. The fact that the memory seems to be antibody independent is to us very interesting. Antibodies can be correlated with protection against invasive pneumococcal disease in children, but not in adults—further, to date there exists no correlate of protection for non-invasive pneumococcal disease at all. Finding these immune correlates of protection is crucial to assist vaccine development, since they potentially could be used to supersede efficacy trials. We believe our results will contribute to the discussion of how our immune system reacts to glycoconjugate vaccines, and ask the question if we perhaps should start looking for correlates of protection in another part of the immune system?
Acknowledgments
We wish to thank D. Milan S. Blake (FDA, Bethesda, USA) for providing the antipneumo- coccal US standard reference serum 89-SF, and Dr David Goldblatt (National Institute of Biological Standards and Control, England) for providing the standardized pneumococcal quality control serum. Clinical trials registration: http://clinicaltrials.gov/ct2/show/NCT00562939
Glossary
Abbreviations:
- HIV
human immunodeficiency virus
- HAART
highly active antiretroviral treatment
- TLR
toll-like receptor
- PS
polysaccharide
- PnPS
pneumococcal polysaccharide
- PCV7
heptavalent pneumococcal conjugate vaccine
- PBMC
peripheral blood mononuclear cell
- ELISA
enzyme-linked immunosorbent assay
- LPS
lipopolysaccharide
- GMC
geometric mean concentrations
- DC-SIGN
Dendritic Cell-Specific Intercellular adhesion molecule-3-Grabbing Non-integrin
- NF-κB
nuclear factor kappa B
Disclosure of Potential Conflicts of Interest
All authors state no conflicts of interest.
Presented in part
Cellular and Cytokine Interactions in Health and Disease, Lisbon, Portugal, October 17–21, 2009.
Financial support
The Scandinavian Society for Antimicrobial Chemotherapy Foundation, the Aase and Ejnar Danielsen’s Foundation, the L. F. Foght’s Foundation, the King Christian X Foundation, and the Beckett Foundation. RO was recipient of a scholarship from the Danish Medical Research Council.
Footnotes
Previously published online: www.landesbioscience.com/journals/vaccines/article/20707
References
- 1.López-Palomo C, Martín-Zamorano M, Benítez E, Fernández-Gutiérrez C, Guerrero F, Rodríguez-Iglesias M, et al. Pneumonia in HIV-infected patients in the HAART era: incidence, risk, and impact of the pneumococcal vaccination. J Med Virol. 2004;72:517–24. doi: 10.1002/jmv.20045. [DOI] [PubMed] [Google Scholar]
- 2.Sogaard OS, Lohse N, Gerstoft J, Kronborg G, Ostergaard L, Pedersen C, et al. Hospitalization for pneumonia among individuals with and without HIV infection, 1995-2007: a Danish population-based, nationwide cohort study. Clin Infect Dis. 2008;47:1345–53. doi: 10.1086/592692. [DOI] [PubMed] [Google Scholar]
- 3.Moffitt KL, Malley R. Next generation pneumococcal vaccines. Curr Opin Immunol. 2011;23:407–13. doi: 10.1016/j.coi.2011.04.002. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 4.McCluskie MJ, Krieg AM. Enhancement of infectious disease vaccines through TLR9-dependent recognition of CpG DNA. Curr Top Microbiol Immunol. 2006;311:155–78. doi: 10.1007/3-540-32636-7_6. [DOI] [PubMed] [Google Scholar]
- 5.Krieg AM. Therapeutic potential of Toll-like receptor 9 activation. Nat Rev Drug Discov. 2006;5:471–84. doi: 10.1038/nrd2059. [DOI] [PubMed] [Google Scholar]
- 6.Søgaard OS, Lohse N, Harboe ZB, Offersen R, Bukh AR, Davis HL, et al. Improving the immunogenicity of pneumococcal conjugate vaccine in HIV-infected adults with a toll-like receptor 9 agonist adjuvant: a randomized, controlled trial. Clin Infect Dis. 2010;51:42–50. doi: 10.1086/653112. [DOI] [PubMed] [Google Scholar]
- 7.Bacterial Respiratory Pathogen Reference Laboratory. Training manual for Enzyme linked immunosorbent assay for the quantitation of Streptococcus pneumoniae serotype specific IgG. Available at: http://www.vaccine.uab.edu/ELISA%20Protocol.pdf Accessed 13 April 2010.
- 8.Lucas AH, Apicella MA, Taylor CE. Carbohydrate moieties as vaccine candidates. Clin Infect Dis. 2005;41:705–12. doi: 10.1086/432582. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 9.Pollard AJ, Perrett KP, Beverley PC. Maintaining protection against invasive bacteria with protein-polysaccharide conjugate vaccines. Nat Rev Immunol. 2009;9:213–20. doi: 10.1038/nri2494. [DOI] [PubMed] [Google Scholar]
- 10.Lai Z, Schreiber JR. Antigen processing of glycoconjugate vaccines; the polysaccharide portion of the pneumococcal CRM(197) conjugate vaccine co-localizes with MHC II on the antigen processing cell surface. Vaccine. 2009;27:3137–44. doi: 10.1016/j.vaccine.2009.03.064. [DOI] [PubMed] [Google Scholar]
- 11.Avci FY, Li X, Tsuji M, Kasper DL. A mechanism for glycoconjugate vaccine activation of the adaptive immune system and its implications for vaccine design. Nat Med. 2011;17:1602–9. doi: 10.1038/nm.2535. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 12.Sahly H, Keisari Y, Crouch E, Sharon N, Ofek I. Recognition of bacterial surface polysaccharides by lectins of the innate immune system and its contribution to defense against infection: the case of pulmonary pathogens. Infect Immun. 2008;76:1322–32. doi: 10.1128/IAI.00910-07. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 13.Gringhuis SI, den Dunnen J, Litjens M, van Het Hof B, van Kooyk Y, Geijtenbeek TB. C-type lectin DC-SIGN modulates Toll-like receptor signaling via Raf-1 kinase-dependent acetylation of transcription factor NF-kappaB. Immunity. 2007;26:605–16. doi: 10.1016/j.immuni.2007.03.012. [DOI] [PubMed] [Google Scholar]
- 14.Manicassamy S, Pulendran B. Modulation of adaptive immunity with Toll-like receptors. Semin Immunol. 2009;21:185–93. doi: 10.1016/j.smim.2009.05.005. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 15.Sen G, Khan AQ, Chen Q, Snapper CM. In vivo humoral immune responses to isolated pneumococcal polysaccharides are dependent on the presence of associated TLR ligands. J Immunol. 2005;175:3084–91. doi: 10.4049/jimmunol.175.5.3084. [DOI] [PubMed] [Google Scholar]
- 16.Mogensen TH, Paludan SR, Kilian M, Ostergaard L. Live Streptococcus pneumoniae, Haemophilus influenzae, and Neisseria meningitidis activate the inflammatory response through Toll-like receptors 2, 4, and 9 in species-specific patterns. J Leukoc Biol. 2006;80:267–77. doi: 10.1189/jlb.1105626. [DOI] [PubMed] [Google Scholar]
- 17.Heidelberger M, Horton D, Haskell TH. Cross-reactions of lipopolysaccharides of Pseudomonas aeruginosa in antipneumococcal and other antisera. Infect Immun. 1986;54:928–30. doi: 10.1128/iai.54.3.928-930.1986. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 18.Linghua Z, Xingshan T, Fengzhen Z. The efficacy of CpG oligodinucleotides, in combination with conventional adjuvants, as immunological adjuvants to swine streptococcic septicemia vaccine in piglets in vivo. Int Immunopharmacol. 2006;6:1267–76. doi: 10.1016/j.intimp.2006.03.009. [DOI] [PubMed] [Google Scholar]
- 19.Aebig JA, Mullen GE, Dobrescu G, Rausch K, Lambert L, Ajose-Popoola O, et al. Formulation of vaccines containing CpG oligonucleotides and alum. J Immunol Methods. 2007;323:139–46. doi: 10.1016/j.jim.2007.04.003. [DOI] [PMC free article] [PubMed] [Google Scholar]