Abstract
Background
In the cochlea patterning of the organ of Corti is tightly regulated to produce a single row of sound detecting inner hair cells and three rows of outer hair cells, which amplify and refine the signal. The recently identified R-Spondin family of signaling molecules usually act as co-activators of Wnt signaling; it is thought that they regulate turnover of Wnt receptors at the membrane. We sought to test whether R-Spondins function in the developing cochlea.
Results
Expression analysis of all four members of the R-Spondin family showed that only R-Spondin2 (Rspo2) is expressed in the cochlea during development of the sensory epithelium. Examination of an Rspo2 −/− mouse showed that loss of Rspo2 results in an additional single row of outer hair cells and disruption of peripheral innervation pattern. Addition of Rspo2 recombinant protein to organotypic cochlear cultures resulted in a small but significant decrease in the number of outer hair cells.
Conclusions
Rspo2 is required to limit the number of outer hair cells to three rows and for optimal arrangement of peripheral nerve fibers. The Rspo2 gain and loss-of-function studies show that in the ear, Rspo2 function is not consistent with its assigned role as a Wnt potentiator.
Keywords: ear, cochlea, Wnt, R-spondin, Rspo2, hair cells
Introduction
R-Spondin2 (roof plate-specific spondin, Rspo2) is one of the four known members of the R-Spondin family (Rspo1, 2, 3 and 4). R-Spondins are secreted proteins containing a hydrophilic secretion peptide signal, a cysteine-rich furin-like domain and a thrombospondin type 1 repeat domain (Yoon and Lee, 2012). Interestingly, the R-spondin family of molecules is only found in vertebrates and primitive chordates; screens in invertebrates such as Drosophila and Caenorhabditis have not identified any R-Spondin homologues (de Lau et al., 2012).
While their signaling pathways and mechanisms remain to be fully elucidated, all four R-Spondins have been shown to function as positive regulators of canonical Wnt signaling and Rspo3 has been shown to function in Wnt planar cell polarity (PCP) signaling (Kazanskaya et al., 2004; Nam et al., 2006; Binnerts et al., 2007; Chassot et al., 2008; Kim et al., 2008; Lu et al., 2008; Carmon et al., 2011; de Lau et al., 2011; Glinka et al., 2011; Ohkawara et al., 2011). It is likely that R-Spondins prolong a cell’s ability to respond to Wnt signaling by regulating Wnt receptor turnover at the membrane (Binnerts et al., 2007; Carmon et al., 2011; de Lau et al., 2011; Glinka et al., 2011; Ohkawara et al., 2011; Hao et al., 2012). R-Spondins are a relatively novel family of signaling molecules and at present most research into their function has focused on Wnt signaling; however, there are some indications that R-Spondins might allow cross-talk between signaling pathways or operate independently of Wnt signaling (Mathew et al., 2008; Friedman et al., 2009).
Developmental functions of Rspo2 include roles in myogenesis (Kazanskaya et al., 2004; Han et al., 2011), craniofacial morphogenesis (Yamada et al., 2009; Jin et al., 2011), keratinocyte proliferation (Chua et al., 2011), osteoblast maturation (Friedman et al., 2009), lung development (Bell et al., 2008) and limb development (Nam et al., 2007a; Aoki et al., 2008; Bell et al., 2008). In each case, Rspo2 is presumed to enhance Wnt signaling by its ability to synergize with Wnts to increase levels of stabilized β-catenin in the cytoplasm, Rspo2 cannot initiate the cascade in the absence of Wnt (Kim et al., 2008).
Various cell-signaling pathways are involved in shaping the cochlea and patterning the organ of Corti - recently reviewed in detail by (Groves and Fekete, 2012). Precise patterning of the sensory epithelium, an essential requirement for optimal hearing, is tightly regulated to produce a single row of inner hair cells (IHCs) and three rows of outer hair cells (OHCs) accompanied by supporting cells (SCs): two rows of phalangeal cells, two rows of pillar cells and three rows of Dieters’ cells. Early in cochlear development, Notch signaling is required to specify the field of prosensory cells (Hartman et al., 2010), while Wnt signaling is implicated in expansion of the prosensory domain (Jacques et al., 2012). BMP, Fgf and Hedgehog signaling then define the lateral boundaries of the presumptive organ of Corti (Driver et al., 2008; Hayashi et al., 2008; Ohyama et al., 2010). Once the sensory epithelium is defined, Notch signaling initiates segregation of cells into those fated to become hair cells and those fated to become supporting cells (Lanford et al., 1999). Fgf and BMP signaling contribute to cell fate decisions (Mueller et al., 2002; Hayashi et al., 2007; Puligilla et al., 2007; Hwang et al., 2010; Liu et al., 2011) and Wnt signaling is required for hair cell differentiation (Jacques et al., 2012).
Given that Wnt activity is required for hair cell differentiation, and that Rspo2 has a variety of developmental roles, we investigated the potential role of Rspo2 in cochlear development. Surprisingly we find that during development of the cochlea, Rspo2 does not enhance Wnt signaling.
RESULTS AND DISCUSSION
R-Spondin 2 is expressed dynamically in the developing cochlea
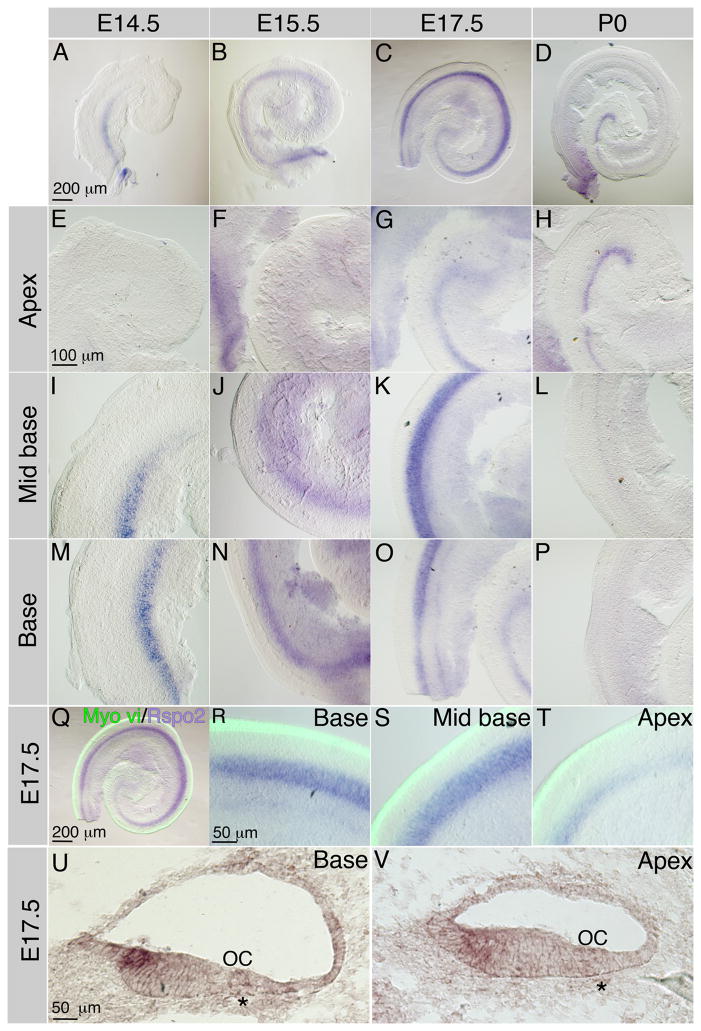
We began by investigating whether R-Spondin family members are expressed in the cochlea during development by performing RTPCR on cochlear tissue collected at Embryonic day (E) 13.25, E15.5, E17.25 and Post natal day (P) 0. Rspo1 and Rspo4 were not expressed at any stage between E13.25 and P0 (Table 1). Rspo2 was expressed between E13.25 and P0 (Table 1). Rspo3 was detected in very small amounts at E17.5 and P0; however, this expression initiated late in cell fate determination, and is unlikely to have an effect on cell fate in the sensory epithelium (Table 1). Having discerned that Rspo2 expression coincided with development of the organ of Corti, we used in situ hybridization to determine spatial distribution of Rspo2 mRNA. Examination of cochleae harvested from E13.5 (data not shown), E14.5, E15.5, E17.5 and P0 mice showed that Rspo2 mRNA was first detected by in situ hybridization in the basal greater epithelial ridge (GER) at E14.5 (Figure 1A, E, I, M). By E15.5, Rspo2 expression extended from the base to the mid base (around 65% of the length of the cochlea) (Figure 1B, F, J, N). At E17.5, the domain of Rspo2 expression had expanded to the apex (Figure 1C, G), was maintained in the mid base (Figure 1G) and began to down regulate in the base (Figure 1K). By P0, Rspo2 expression was restricted to the apex and apical portion of the mid base (Figure 1D, H, L, O). Examination of the Rspo2 expression domain in relation to the hair cells (marked by Myosin vi (Myo vi)) showed that Rspo2 expression was localized to a region around 75 μm medial to the organ of Corti in the greater epithelial ridge (Figure 1Q, R, S, T). Transverse sections through the temporal bone at E17.5 show that Rspo2 was expressed in cells of the inner sulcus (Figure 1U, V); in relation to the organ of Corti as marked by ‘OC’, it appeared that in the most mature region of the developing cochlea, the base (Figure 1, U), the distance between the Rspo2 expression domain and the hair cells was reduced in comparison to at the least mature point (the apex) (Figure 1V). Having shown that Rspo2 was expressed concordant with development of the organ of Corti, we sought to investigate the potential role of Rspo2 in development.
Table 1.
Expression of R-spondin family members during development of the cochlea. Tabulated results of an RT PCR assay showing that only Rspo2 is expressed throughout development. Trace amounts of Rspo3 were detected at E17.5 and P0 - a time after cell fate specification has occurred in the organ of Corti. Rspo1 and 4 were not detected at any stage. Positive and negative controls (kidney, lung or digits) were chosen based on previous expression analysis screens (Nam et al. 2007b).
| E14.5 Kidney | E14.5 Lung | E15.5 Digits | E13.25 cochlea | E15.5 cochlea | E17.5 cochlea | P0 cochlea | |
|---|---|---|---|---|---|---|---|
| Rspo1 | Yes | No | No | No | No | No | |
| Rspo2 | No | Yes | Yes | Yes | Yes | Yes | |
| Rspo3 | Yes | No | No | No | Trace | Trace | |
| Rspo4 | No | Yes | No | No | No | No |
Figure 1.
Spatial and temporal expression of Rspo2 in the developing cochlea. Low magnification views of whole mount in situ hybridization for Rspo2 at E14.5 (A), E15.5 (B), E17.5 (C) and P0 (D) show that Rspo2 is first expressed in the base at E14.5 (A), extended to the mid base by E15.5 (B), and the apex by E17.5 (C). At P0, expression of Rspo2 is maintained in the apex but was down regulated in the base. E–H high magnification views of the least developmentally advanced region of the cochlea, the apex, at E14.5 (E), E15.5 (F), E17.5 (G) and P0 (H) show that Rspo2 was not detected in the apex until E17.5, relatively late in cochlear development. I–L show high magnification views of the mid base of the cochlea at E14.5 (I), E15.5 (J), E17.5 (K) and P0 (L) show that the Rspo2 expression domain rapidly extends along the mid base from E14.5 (I), peaking at E17.5 (K), but is beginning to be down regulated by P0. M–P show high magnification views of the most mature region of the cochlea, the base, at E14.5 (M), E15.5 (N), E17.5 (O) and P0 (P). Rspo2 expression was detected strongly in the base at E14.5 (M) and E15.5 (N) but by E17.5, relative to its expression in the mid base, Rspo2 expression was starting to be down regulated. By P0, there was no Rspo2 expression in the base. Q The whole mount E17.5 cochlea shown in panels C, G, K and O showing in situ hybridization for Rspo2 (purple) and immunofluorescence for Myosin vi (green). R–T show higher magnifications of the base ©, mid base (S) and Apex (T) of the cochlea shown in Q/C. In the base and mid base, The Rspo2 domain was situated relatively far from the organ of Corti (labeled with Myosin vi) (Q, R, S). In the apex, Myosin vi labeling showed that the inner hair cells began to differentiate before Rspo2 expression reached levels detectable by in situ hybridization. U. In situ hybridization for Rspo2 on a transverse section through the basal region of an E17.5 cochlea shows Rspo2 expression in the luminal surface cells of the most lateral region of the GER. V. In situ hybridization for Rspo2 on a transverse section through the apical region of an E17.5 cochlea shows that in a less mature region of the cochlea, expression spans the depth of the GER and is more diffuse. * indicate the spiral vessel – a marker for the location of the developing organ of Corti (OC).
Loss of R-spondin 2 results in an extra row of outer hair cells
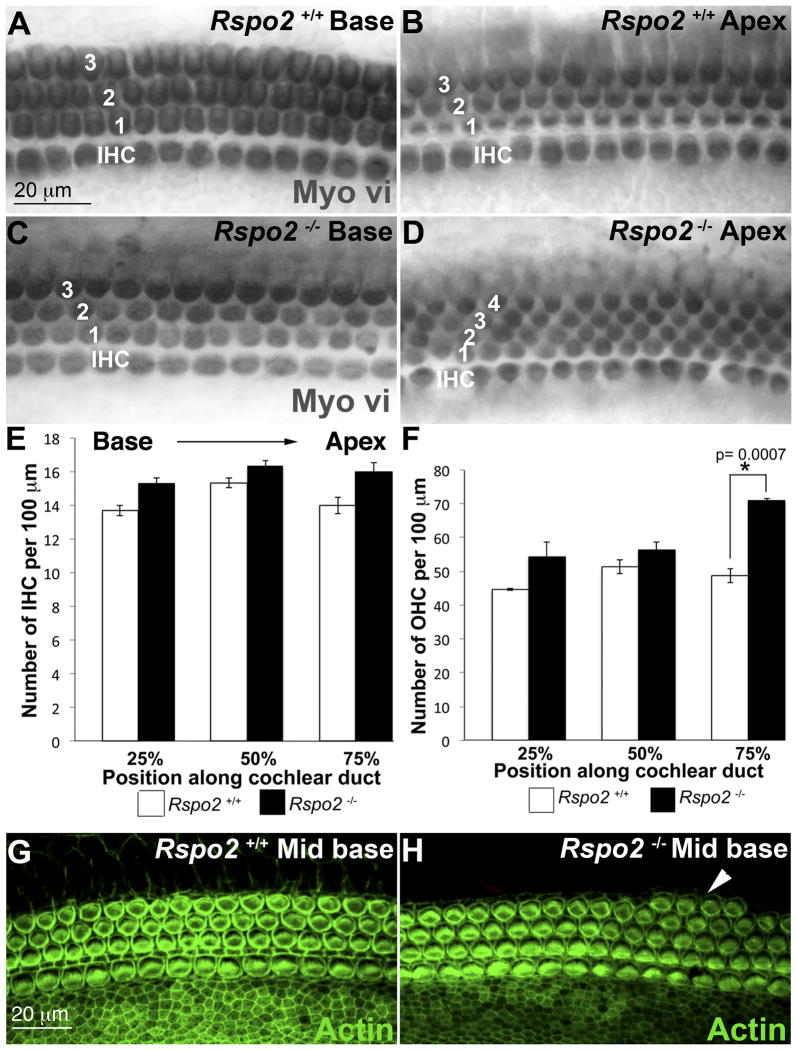
Examination of E18.5 Rspo2 homozygous null cochleae revealed a continuous fourth row of outer hair cells extending from the mid base (around 60% along the length from the base to the apex) to the apex (Figure 2A–D). Quantification of hair cells at distances of 25%, 50% and 75% from the most basal point of the cochlea, showed that there was no significant change in the number of inner hair cells (Figure 2E). Quantification of outer hair cells (Figure 2F) at the same points along the length of the cochlea gave no significant increase in the number of hair cells at 25% or 50%, but at 75% there was a significant increase (p ≤ 0.001). Counts were taken from 100 μm regions at each distance using three mutant-wild type sibling pairs across three different litters.
Figure 2.
Rspo2 homozygous null animals had an ectopic row of outer hair cells in the apical half of the cochlea. A–B Surface views of the base (A) and apex (B) or a wild type cochlea at E18.5. Immunostaining with the hair cell marker Myosin vi (Myo vi) showed that in the base (A) and the apex (B) there were the normal one row of inner hair cells and three rows of outer hair cells. C–D Surface views of the base (C) and apex (D) of an E18.5 Rspo2 null cochlea. C. At the base of the cochlea there was one row of inner hair cells and three rows of outer hair cells. D. At the apex there were one row of inner hair cells and four rows of outer hair cells. E. Counting inner hair cells within a 100 μm length of the sensory epithelium at the positions 25%, 50% and 75% along each cochlea (measured from the base) shows no significant difference between the number of inner hair cells in the wild type and the null (n=3). F. Counting outer hair cells within a 100 μm at the positions 25%, 50% and 75% along each cochlea shows no significant difference between the number of outer hair cells in the wild type and the null at 25% and 50%, but a significant increase in outer hair cells in the null at 75% (n=3). Bars on the graph represent standard deviation. G–H. Phalloidin labeling of actin in the mid base of a wild type (G) and Rspo2 homozygous null (H) cochlea at E18.5. Comparison of the orientation of the stereociliary bundles in G and H indicates that in the mutant cochlea the bundles are orientated normally. Arrowhead marks a patch of ectopic outer hair cells. IHC. Inner hair cell, OHC. Outer hair cell.
Relatively little is known of Rspo2 function during development, as yet it has only been identified as a canonical Wnt potentiator, but Rspo3 has recently been shown to function in Wnt PCP signaling (Glinka et al., 2011; Ohkawara et al., 2011). We investigated whether Rspo2 might also have a dual role in Wnt signaling. We examined mutant cochleae for phenotypes derived from disrupted planar cell polarity. Typically, in cochleae exhibiting Wnt PCP defects, the stereociliary bundles are misorientated and often, additional inner hair cells are observed due to failure of the duct to extend completely, disrupting the underlying organization of the hair cells (Montcouquiol et al., 2003). In the mid base of the mutant (Figure 2H), the stereociliary bundles were orientated no differently to those of the wild type and the inner hair cells occurred as a single continuous row (Figure 2G). We saw no disorganization of hair cell bundles, no disruption of inner hair cell organization and no significant difference in the length of the duct (data not shown). This implies that Rspo2 is not a necessary component of Wnt PCP signaling in the cochlea.
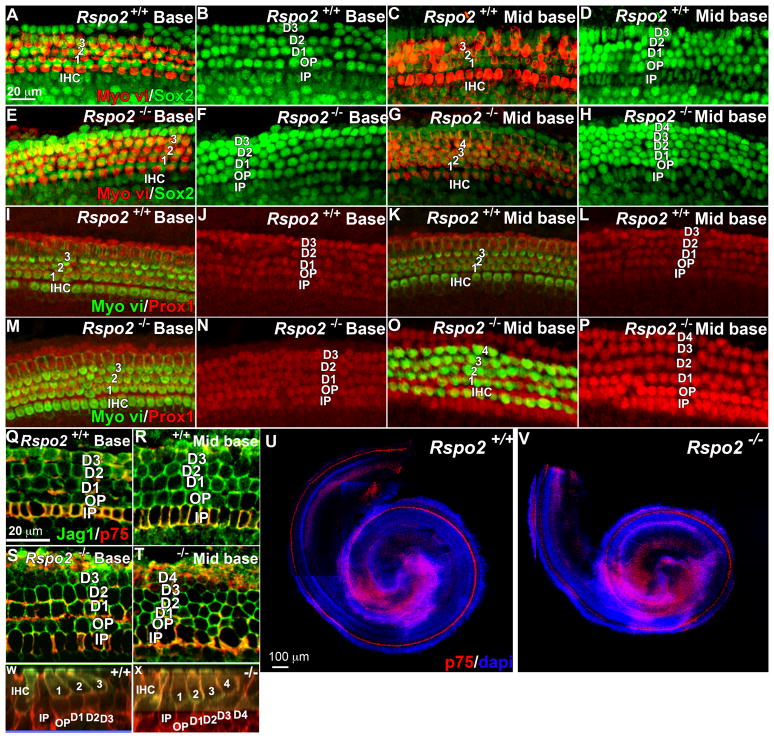
Having shown that Rspo2 mutant animals have one extra row of outer hair cells, we examined the support cells. It is common to see an increase in support cells with an increase in outer hair cells, however this is not always the case. Loss of Fgfr3 results in a row of ectopic outer hair cells at the expense of outer pillar cells (Colvin et al., 1996; Hayashi et al., 2007; Puligilla et al., 2007). To rule out a fate switch from support cell to hair cell, we examined the support cells with markers of specification, Sox2 (Kiernan et al., 2005; Hume et al., 2007; Dabdoub et al., 2008), Jagged1 (Morrison et al., 1999), and differentiation, Prox1 (Bermingham-McDonogh et al., 2006}. In the base where there are three rows of outer hair cells in both the mutant and the wild type, we saw two rows of Pillar cells and three rows of Deiters’ cells marked by Sox2 (figure 3A, B, E, F), Prox1 (Figure 3I, J, M, N) and Jagged1 (Figure 3Q, S). Sox2 also marks cells of the GER (Figure 3A, -H), this labeling is unaffected by loss of Rspo2. In the mid base of the mutant, where we observed 4 rows of outer hair cells, we found one extra row of support cells marked by Sox2, Prox1 or Jagged1 (Figure 3C, D, G, H, K, L, O, P, R, T). While Fgfr3 mutants lack outer Pillar cells, they have an additional Deiters’ cell, so we also tested whether the Pillar cells had fully differentiated. In the Rspo2 mutant, both Pillar cells were present (marked by Sox2, Prox1 and Jagged1), immunofluorescence for p75NTR showed that the Pillar cells had fully differentiated (Figure 3Q–V) and actin staining showed that the inner pillar cells had formed projections (Figure 3W, X).
Figure 3.
Ectopic hair cells in the apical half of the Rspo2 null cochlea were accompanied by an extra row of support cells. A–D. Immunofluorescence for Myo vi (red) and Sox2 (green) in the base (A, B) and mid base (C, D) of a wild type E18.5 cochlea. In both the base and the mid base, there were one row of inner hair cells (IHC) and three rows of outer hair cells (1,2,3), Sox2 marks cells of the inner sulcus and five rows of support cells (IP, OP, 1, 2, 3) (A, B, C, D). E–H. Immunofluorescence for Myo vi (red) and Sox2 (green) in the base (E, F) and mid base (G, H) of an E18.5 Rspo2 homozygous null cochlea. In the base there are one row of inner hair cells (IHC), three rows of outer hair cells (1,2,3) and five rows of Sox2 positive support cells (IP, OP, D1, D2, D3) and inner sulcus cells (E, F), but starting from the mid base there is an additional row of outer hair cells (4) (G) and an additional row of Sox2 positive support cells (4) (H). I–L. Immunofluorescence for Myo vi (green) and Prox1 (red) in the base (I, J) and mid base (K, L) of a wild type E18.5 cochlea. M–P. Immunofluorescence for Myo vi (green) and Prox1 (red) in the base (M, N) and mid base (O, P) of an Rspo2 homozygous null E18.5 cochlea. In the base there are one row of inner hair cells (IHC), three rows of outer hair cells (1,2,3) (I) and five rows of support cells (IP, OP, 1, 2, 3) (J), but in the mid base there is an extra row of outer hair cells (4) (K) and an extra row of Deiters’ cells (4) (L). Q and R. Immunofluorescence for Jagged1 (green) and p75 (red) in the base (Q) and mid base (R) of an E18.5 wild type cochlea. S and T. Immunofluorescence for Jagged1 (green) and p75 (red) in the base (S) and mid base (T) of an Rspo2 homozygous null E18.5 cochlea. In both the null and the wild type animals the pillar cells are marked by p75 and all support cells including the pillar cells by Jagged1. U and V low magnification views of p75 immunofluorescence (red) and nuclear staining with DAPI (blue) of a wild type (U) and an Rspo2 knock out (V) E18.5 cochlea shows that p75 positive cells extend to the apex in both the knock out and the wild type. W and X Orthogonal views of Z-stacks taken through the apical mid base showing that in both the wild type (W) and the mutant (X), phalloidin staining of actin (red) shows that the inner pillar cells have extended between the inner hair cell and first outer hair cell (labeled with Myo vi in green). IHC., Inner hair cell. OHC., Outer hair cell., IP., Inner Pillar cell. OP., Outer Pillar cell. 1–4., Deiters’ cell 1–4
Cochleae exposed to ectopic Rspo2 have fewer hair cells
Next, we used gain of function assays to test whether additional Rspo2 disrupts hair cell development. We established organotypic cochlear explant cultures at E13.5, a developmental stage where the hair cells in the base have begun to develop, but the hair cells in the apex have yet to arise. We applied 5 μg/ml of recombinant Rspo2 protein to the explants at the time of plating and cultured them for six days, by which time the program of hair cell differentiation was complete. Examination of the hair cells using Myosin vi immunofluorescence showed no change in the number of inner hair cells (Figure 4A), but a very slight decrease in the number of outer hair cells in the mid base and apex when compared to untreated control explants (Figure 4B). This decrease was significant (control n= 17, 5 μg/ml Rspo2 n= 15, mid base p= 0.016, apex p= 0.019) and was recapitulated, but not enhanced, by raising the concentration to 20 μg/ml. Previous work from our lab showed that activation of Wnt/β-Catenin signaling results in an increase in the number of hair cells and support cells, and inhibiting Wnt signaling prevents hair cells from differentiating (Jacques et al., 2012). When Rspo2 is deleted, or we introduce ectopic Rspo2, we see the opposite. Loss of Rspo2 results in ectopic outer hair cells and ectopic Rspo2 activity results in a decrease in the number of outer hair cells. In order to test whether the decrease in outer hair cells is an effect specific to Rspo2, we added Rspo1 recombinant protein to E13.5 cultures. Rspo1 is sometimes used as an alternative to Wnt3a to activate Wnt signaling (Ootani et al., 2009; Chai et al., 2012). Addition of Rspo1 recombinant protein resulted in an increase in the number of hair cells, consistent with activation of Wnt signaling (data not shown) (Jacques et al., 2012). This implies that Rspo2 is not able to transduce canonical Wnt signaling in the cochlea and that the effect of reducing the number of hair cells is specific to Rspo2.
Figure 4.
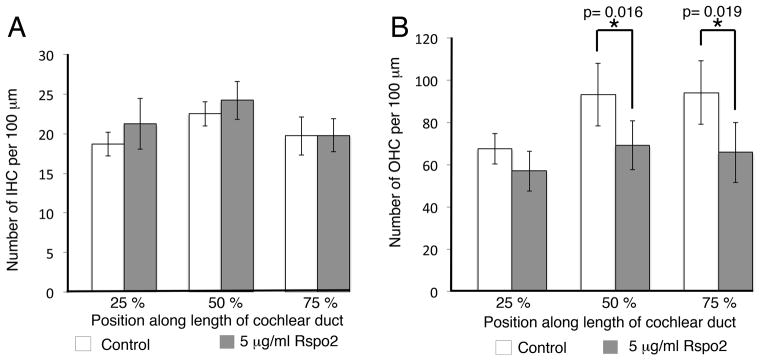
Rspo2 inhibits hair cell differentiation in vitro. A. Addition of 5μg/ml Rspo2 recombinant protein to E13.5 cochlear explants results in no change in the number of inner hair cells when compared to controls. B. Addition of 5 μg/ml Rspo2 recombinant protein results in a significant decrease in the number of outer hair cells in the mid base (50% along the length of the explant) and apex (75% along the length of the explant) when compared to untreated control cultures. Control n= 17, Rspo2 treated n= 15, 50% point p= 0.016, 75% point p= 0.019, two tailed student’s T test. Bars represent standard deviation.
So far, Rspo2 has been shown only to enhance Wnt signaling, the phenotype arising from deletion of Rspo2 and addition of Rspo2 recombinant protein is the opposite of that achieved by activation and inhibition of Wnt signaling implying other functions of Rspo2 in addition to its role in canonical Wnt signaling. It could be that in some circumstances Rspo2 can antagonize Wnt signaling, or it may be that Rspo2 does not always function within the Wnt cascade. It has been reported that ectopic BMP4 signaling results in additional hair cells (Puligilla et al., 2007), and loss of BMP antagonist Noggin results in ectopic inner hair cells and one ectopic row of outer hair cells (Hwang et al., 2010). The Rspo2 null phenotype is similar to what might be expected if inhibition of BMP4 activity were lifted, and is reminiscent of deletion of Noggin. Expression of Rspo2 is restricted to the GER, but the Rspo2 null phenotype manifests in the outer hair cells. This might be because Rspo2 expression lags behind specification of the inner hair cells (Figure 1R, S, T).
During early Xenopus development Rspo2 can antagonize Tgf-β signaling, independently of Wnt it antagonizes Activin and Xnr mediated induction of XBra and BMP4 mediated induction of Xvent2, although it is unclear whether this inhibitory effect on BMP4 is independent of Wnt (Kazanskaya et al., 2004). So far no direct link between Rspo2 signaling activity and BMP signaling has been identified, other than that Rspo2 mediated signaling is upstream of BMP4 transcription (Aoki et al., 2008; Bell et al., 2008; Jin et al., 2011).
Loss of R-spondin 2 results in disrupted innervation of the outer hair cells
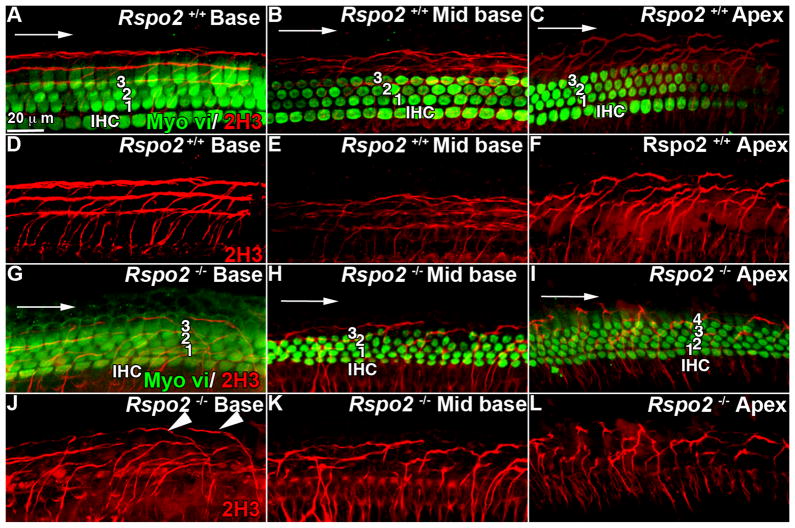
In addition to the hair cell phenotype we also observed some disruption of innervation of the organ of Corti. In the cochlea, there are two types of neuron that extend into the sensory epithelium, type I fibers that interact with inner hair cells, and type II (peripheral) fibers, that connect with the outer hair cells. In wild type animals, staining fibers with marker 2H3, we observed that as type I nerve fibers enter the organ of Corti they meet the inner hair cells, and as type II fibers invade the organ of Corti they uniformly turned towards the base and formed three bundled tracks running parallel between the Deiters’ cells as previously shown (Yang et al., 2011) (Figure 5A, D, G). In the mid base, where development has not progressed so far, the fibers had infiltrated the sensory epithelium and the type II fibers have started to turn towards the base, but had not yet formed bundles (Figure 5B, E, H). In the still more immature apex, the peripheral fibers entered the organ of Corti, but had only begun to turn (Figure 5C, F, I). In Rspo2 homozygous null animals, in the base of the cochlea where there were three rows of outer hair cells (Figure 5M), type I dendrites appear to behave normally, whereas type II fibers penetrated the sensory epithelium and began to turn towards the base, but did not form the clearly defined tracks that we saw in the wild type base (Figure 5J, M, P). The lack of organization looked similar to that of the wild type mid base, suggesting a developmental delay; however, some fibers appeared to turn towards the apex (Figure 5P, marked by arrow heads). In the mid base where there were four rows of outer hair cells (Figure 5N), the fibers invaded the sensory epithelium, but seemed to lack directionality although the distribution of the fibers looked similar to that of the wild type apex (Figure 5K, Q). It might be that the lack of organization here was a secondary effect to the presence of a fourth row of outer hair cells. In the apex where there were four rows of outer hair cells (Figure 5O) the effect was more pronounced with clear disorganization of innervation (Figure 5L, R). The presence of an extra row of hair cells was likely to contribute to the phenotype in the mid base and apex; however, it is unclear why the orientation of the fibers is affected in the base. It is possible that the phenotype observed at E18.5 was due to a developmental delay that is self-correcting, however perinatal lethality of the Rspo2 mutation precludes investigation. It may be that Rspo2 has a role in chemo attraction/repulsion during axon guidance processes. Little is known about the signaling processes underlying the arrangement and orientation of the fibers once they cross the habenula perforata (Yang et al., 2011). Although Wnt signaling has been implicated in axon guidance (Salinas, 2012), as yet no evidence that canonical or non-canonical Wnt signaling affect organization of type II fibers has been reported. Loss of Prox1 results in misdirected fiber extension and failure of bundling (Fritzsch et al., 2010); however, we saw normal distribution of Prox1 in Rspo2 −/− cochleae. Disruption of Ephrin or Fgf signaling appears to have a similar phenotype to Rspo2 deletion, fibers cross into the organ of Corti, but they fail to orientate properly and do not form bundles (Puligilla et al., 2007; Coate et al., 2012). No evidence has been presented that Rspo2 is implicated in either Ephrin or Fgf signaling. Intriguingly, it has been shown in vitro that BMP4 has a shortening effect on spiral ganglia derived neurites (Whitlon et al., 2007). It has been speculated that BMP4 has a repulsive effect on spiral ganglia (Pan et al., 2011), but because BMP4 is essential for early patterning events in the cochlea, it has not been possible to examine the effect of BMP4 deletion on type II fibers in vivo.
Figure 5.
Innervation of the outer hair cells was disrupted in Rspo2 knock out mice. A–F Immunofluorescence for Myo vi (green) and neurofilament (2H3) (red) in the base (A, D) mid base (B, E) and apex (C, F) of a wild type E18.5 cochlea. At all points there are one row of inner hair cells (IHC) and three rows of outer hair cells (1,2,3). In the base (A, D) the nerve fibers enter the sensory epithelium and uniformly turn towards the base, forming three clearly defined tracks. In the mid base (B, E) the fibers have entered the sensory epithelium and turned towards the base and are in the process of forming bundles. In the apex (C, F) the fibers have entered the sensory epithelium and turned towards the base but have not yet started to form bundles. G–L. Immunofluorescence for Myo vi (green) and 2H3 (red) in the base (G, J), mid base (H, K) and apex (I, L) of an E18.5 Rspo2 homozygous null cochlea. In the base there are one row of inner hair cells (IHC) and three rows of outer hair cells (1,2,3) (G) but in the mid base (H) and apex (I) there are one row of inner hair cells (IHC) and four rows of outer hair cells (OHC). In the base (G, J) the fibers have entered the sensory epithelium, but in contrast to the wild type (D), the fibers have not formed tracks and some appear to be turning towards the apex (marked with an arrow head). In the mid base (H, K) and apex (I, L) the fibers are disorganized. IHC., Inner hair cell. Arrows point in the direction of the base.
In summary, we have identified Rspo2 as a necessary component of the signal transduction network required for normal patterning of the inner ear. In the absence of Rspo2, an extra row of outer hair cells is generated and innervation of the sensory epithelium is disrupted. In this developmental context, we cannot easily place Rspo2 within the Wnt signaling pathway; however, as knowledge of R-Spondin function expands over time, subsequent work on the links between R-Spondins and other signaling networks may allow us to conduct new studies and draw more conclusions.
Experimental Procedures
Animals
Mutant tissue was obtained from mice containing a Neomycin insertion in Exon2 of the Rspo2 coding region (Yamada et al., 2009). Mice were bred and genotyped as previously described (Yamada et al., 2009). Rspo2 heterozygous null mice were backcrossed into the C57BL/6 background for over 10 generations, and then intercrossed to generate homozygotes, minimizing the possibility of an unrelated phenotype. Tissue for in situ hybridization and RT PCR was harvested from CD-1, ICR mice maintained and euthanized in accordance with NIH and UCSD guidelines for the care and use of laboratory animals. Three pairs of mice each were used to examine markers for hair cell differentiation, supporting cell subtypes, stereocilia orientation and innervation. Within each study the sibling pairs were collected from independent litters.
Reverse Transcriptase PCR
RNA from dissected cochleae at stages E13.25, E15.5, E17.25 and P0, E14 kidneys, E14 lungs, E15.5 distal phalanges and E13.5 bodies, was collected using Trizol, and cDNA transcribed from this using the TaqMan cDNA reverse transcription kit (Invitrogen). Each cochlear RNA prep contained RNA from 8 litter mates. Rspo 1, 2, 3 and 4 were amplified using annealing temperature 56° C over 35 cycles using primers: Rspo1: Forward TGTGAAATGAGCGAGTGGTCC, Reverse TCTCCCAGATGCTCCAGTTCT. Rspo2: Forward TTGCATAGAGGCCGCTGCTTT, Reverse CTGGTCAGAGGATCAGGAATG. Rspo3: Forward GTACACTGTGAGGCCAGTGAA, Reverse ATGGCTAGAACACCTGTCCTG. Rspo4: Forward CTGGACTCCCTGCATACACAA, Reverse CACGGGGAGAAGGAAAGTTTC.
Whole mount In situ Hybridization
Dig labeled probes were prepared from the plasmid provided as a gift to us by J.K Yoon (Nam et al., 2007b). The plasmid was linearized with mlu1 and transcribed with T3 Polymerase for anti sense transcripts.
Temporal bones were collected at E13.5, E14, E15.5, E17.5 and P0 in PBS then fixed over night at 4°C and transferred to 100% MeOH for long term storage at −20°C.
Temporal bones were rehydrated in ascending grades of PBS and dissected to expose the sensory epithelium of the cochlear duct. Four cochleae at each stage were processed for whole mount in situ hybridization.
Tissue was washed three times in PBST and treated for 20 minutes, three times with detergent mix (50 mM Tris-HCL pH 8, 1 mM EDTA, 150 mM NaCl, 1% IGEPAL, 1% SDS, 0.5% Deoxycholate). Tissue was washed a further three times in PBST, fixed for 20 minutes in 4% PFA and washed in PBST again. Tissue was equilibrated in 50% PBST/50% Hyb mix (50% Formamide, 1.3 X SSC pH 5, 5mM EDTA, 50 μg/ml Yeast RNA (Sigma), 0.2% Tween-20, 0.5% CHAPS, 100 μg/ml Heparin (sigma)) for 10 minutes then soaked in 100% Hyb mix for a further 10 minutes. Tissue was incubated at 65° C in Hyb mix with rocking for 2 hours. Probe was added to pre warmed Hyb mix and applied to the tissue and incubated over night at 65°C with rocking. Probe/hyb mix was retrieved and stored at −20°C for reuse. Tissue was washed in fresh pre-warmed Hyb buffer at 65 °C for 10 minutes followed by two 30-minute washes in 2 X SSC pH 5 at 65°C and three 10 minute washes in MABT at room temperature. Tissue was pre-blocked for one hour in 2% Boehringer blocking reagent (Roche)/MABT and then one hour in 2%BBR/20% Sheep serum/MABT. Tissue was incubated for 4 hours at room temperature in 2% Boehringer blocking reagent 20% sheep serum/MABT/1:2000 anti DIG AP conjugated antibody (Roche). Tissue was washed 6 times in MABT for 30 minutes then washed three times in freshly mixed NTMT buffer (100 mM NaCl, 100 mM Tris pH 9.5, 50 mM MgCl2, 1% Tween- 20). Color was developed on addition of premixed NBT/BCIP (Roche) to fresh NTMT buffer and incubation in the dark until color developed. Tissue was fixed over night in PFA and then mounted on slides for analysis.
In Situ Hybridization on frozen sections
Temporal bones collected and stored in MEOH were rehydrated in PBS the processed for cryosectioning. Sections were cut at 10 μm thickness and dried on slides over night. Sections were stored long term at −80 in a sealed box.
Slides were rested at room temperature for 30 minutes, then fixed in PFA for 10 minutes. Slides were washed three times in PBS then treated with Proteinase K (Sigma) at 1 μg/ml for 30 minutes. Slides were washed in PBS twice before alkylation in triethanolamine with acetic anhydride. Slides were washed three times in PBS and then fixed for 20 minutes with 4% PFA. Slides were washed three times in PBS then incubated in Hyb buffer in a humidified box for 1 hour at 65° C. Fresh Hyb buffer containing 200 ng/ml Dig labeled probe was added and the slides were incubated over night at 65° C. Slides were washed once in 2 X SSC pH 5.4 for 20 minutes at 65° C, cooled to room temperature and then incubated in 200 ng/ml RNaseA for 30 minutes at 37° C. Slides were rinsed twice in 2 X SSC pH 5.4 then incubated twice at 65° C in 0.2 X SSC pH 5.4 for 30 minutes. Slides were equilibrated twice in MAB at room temperature and then blocked for 30 minutes in 1% BBR. Slides were incubated with 1/1000 anti dig AP conjugate antibody (Roche) for one hour and then washed three times in MAB. Slides were soaked in NTMT buffer briefly and NTMT/BCIP/NBT was applied and incubated in darkness until a signal was observed.
Immunofluorescence
Immunofluorescence detection was carried as previously described (Dabdoub et al., 2008). Antibodies used were: Rabbit anti Myosin vi (1:1000, Proteus biosciences), Goat anti Sox2 (1:250, Santa Cruz), Goat anti Prox1 (1:250, R&D Systems), Goat anti jagged1 (1:250, Santa Cruz), Rabbit anti P75 (1:1000, Millipore), mouse anti 2H3 (1:100, DSHB). Phalloidin-488 conjugate (Invitrogen) was used to visualize actin distribution (1:100).
Imaging
Bright field images were captured using an Olympus BX51. Confocal images were obtained using a Zeiss 510 LSM confocal microscope and images were processed using LSM viewer software.
Culturing
Cochlear explants were collected at E13.5, dissected and cultured as previously described (Dabdoub et al., 2008). Three independent experiments were performed for each condition, in each experiment one litter of E13.5 fetuses were used, with at least three cochleae per condition. Recombinant Rspo1 (R &D systems) was added at 5 μg/ml and Rspo2 (R & D systems) at 5 μg/ml or 20 μg/ml in 2% FBS DMEM enriched with N2 supplement and replenished after 24 hours. Explants were cultured for six days then fixed in 4% PFA for 30 minutes.
Cell counts were taken across a 100 μm region at 25%, 50% and 75% points along the length of the duct. Statistics were obtained using a two tailed student’s T test.
Key findings.
Rspo2 is dynamically expressed in the cochlea over a short time period.
Loss of Rspo2 results in supernumerary hair cells in the organ of Corti.
Rspo2 is necessary for normal innervation of outer hair cells.
Acknowledgments
We thank Dr Alan Cheng, Dr Doris Wu and Dr Jeong Kyo Yoon for their valuable comments and insights. Funding for this work was provided by an NIH R01 grant (DC011104) to A.D. Fluorescent images were generated at the UCSD Neuroscience Microscopy Shared Facility supported by grant P30 (NS047101). The hybridoma 2H3 antibody was obtained from the Developmental Studies Hybridoma Bank developed under the auspices of the NICHD and maintained by The University of Iowa, Department of Biology, Iowa City, IA 52242. We also thank Drs. Kazuma Tomizuka (Kyowa Hakko Kirin California, Inc.) and Makoto Kakitani (Kyowa Hakko Kirin Co., Ltd.) for their work managing the Rspo2−/− development and breeding program.
Abbreviations
- IHC
inner hair cell
- OHC
outer hair cell
- SC
support cell
- GER
greater epithelial ridg
- Rspo
R-Spondin
- E
embryonic day
- P
post natal day
- PCP
planar cell polarity
- Myo
Myosin
References
- Aoki M, Kiyonari H, Nakamura H, Okamoto H. R-spondin2 expression in the apical ectodermal ridge is essential for outgrowth and patterning in mouse limb development. Dev Growth Differ. 2008;50:85–95. doi: 10.1111/j.1440-169X.2007.00978.x. [DOI] [PubMed] [Google Scholar]
- Bell SM, Schreiner CM, Wert SE, Mucenski ML, Scott WJ, Whitsett JA. R-spondin 2 is required for normal laryngeal-tracheal, lung and limb morphogenesis. Development. 2008;135:1049–1058. doi: 10.1242/dev.013359. [DOI] [PubMed] [Google Scholar]
- Binnerts ME, Kim KA, Bright JM, Patel SM, Tran K, Zhou M, Leung JM, Liu Y, Lomas WE, 3rd, Dixon M, Hazell SA, Wagle M, Nie WS, Tomasevic N, Williams J, Zhan X, Levy MD, Funk WD, Abo A. R-Spondin1 regulates Wnt signaling by inhibiting internalization of LRP6. Proc Natl Acad Sci. 2007;104:14700–14705. doi: 10.1073/pnas.0702305104. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Carmon KS, Gong X, Lin Q, Thomas A, Liu Q. R-spondins function as ligands of the orphan receptors LGR4 and LGR5 to regulate Wnt/beta-catenin signaling. Proc Natl Acad Sci. 2011;108:11452–11457. doi: 10.1073/pnas.1106083108. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Chai R, Kuo B, Wang T, Liaw EJ, Xia A, Jan TA, Liu Z, Taketo MM, Oghalai JS, Nusse R, Zuo J, Cheng AG. Wnt signaling induces proliferation of sensory precursors in the postnatal mouse cochlea. Proc Natl Acad Sci. 2012;109:8167–8172. doi: 10.1073/pnas.1202774109. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Chassot AA, Ranc F, Gregoire EP, Roepers-Gajadien HL, Taketo MM, Camerino G, de Rooij DG, Schedl A, Chaboissier MC. Activation of beta-catenin signaling by Rspo1 controls differentiation of the mammalian ovary. Hum Mol Genet. 2008;17:1264–1277. doi: 10.1093/hmg/ddn016. [DOI] [PubMed] [Google Scholar]
- Chua AW, Ma D, Gan SU, Fu Z, Han HC, Song C, Sabapathy K, Phan TT. The role of R-spondin2 in keratinocyte proliferation and epidermal thickening in keloid scarring. J Invest Dermatol. 2011;131:644–654. doi: 10.1038/jid.2010.371. [DOI] [PubMed] [Google Scholar]
- Coate TM, Raft S, Zhao X, Ryan AK, Crenshaw EB, 3rd, Kelley MW. Otic mesenchyme cells regulate spiral ganglion axon fasciculation through a Pou3f4/EphA4 signaling pathway. Neuron. 2012;73:49–63. doi: 10.1016/j.neuron.2011.10.029. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Colvin JS, Bohne BA, Harding GW, McEwen DG, Ornitz DM. Skeletal overgrowth and deafness in mice lacking fibroblast growth factor receptor 3. Nature Genetics. 1996;12:390–397. doi: 10.1038/ng0496-390. [DOI] [PubMed] [Google Scholar]
- Dabdoub A, Puligilla C, Jones JM, Fritzsch B, Cheah KS, Pevny LH, Kelley MW. Sox2 signaling in prosensory domain specification and subsequent hair cell differentiation in the developing cochlea. Proc Natl Acad Sci. 2008;105:18396–18401. doi: 10.1073/pnas.0808175105. [DOI] [PMC free article] [PubMed] [Google Scholar]
- de Lau W, Barker N, Low TY, Koo BK, Li VS, Teunissen H, Kujala P, Haegebarth A, Peters PJ, van de Wetering M, Stange DE, van Es JE, Guardavaccaro D, Schasfoort RB, Mohri Y, Nishimori K, Mohammed S, Heck AJ, Clevers H. Lgr5 homologues associate with Wnt receptors and mediate R-spondin signalling. Nature. 2011;476:293–297. doi: 10.1038/nature10337. [DOI] [PubMed] [Google Scholar]
- de Lau WB, Snel B, Clevers HC. The R-spondin protein family. Genome Biol. 2012;13:242. doi: 10.1186/gb-2012-13-3-242. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Driver EC, Pryor SP, Hill P, Turner J, Ruther U, Biesecker LG, Griffith AJ, Kelley MW. Hedgehog signaling regulates sensory cell formation and auditory function in mice and humans. J Neurosci. 2008;28:7350–7358. doi: 10.1523/JNEUROSCI.0312-08.2008. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Friedman MS, Oyserman SM, Hankenson KD. Wnt11 promotes osteoblast maturation and mineralization through R-spondin 2. J Biol Chem. 2009;284:14117–14125. doi: 10.1074/jbc.M808337200. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Fritzsch B, Dillard M, Lavado A, Harvey NL, Jahan I. Canal cristae growth and fiber extension to the outer hair cells of the mouse ear require Prox1 activity. PloS One. 2010;5:e9377. doi: 10.1371/journal.pone.0009377. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Glinka A, Dolde C, Kirsch N, Huang YL, Kazanskaya O, Ingelfinger D, Boutros M, Cruciat CM, Niehrs C. LGR4 and LGR5 are R-spondin receptors mediating Wnt/beta-catenin and Wnt/PCP signalling. EMBO reports. 2011;12:1055–1061. doi: 10.1038/embor.2011.175. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Groves AK, Fekete DM. Shaping sound in space: the regulation of inner ear patterning. Development. 2012;139:245–257. doi: 10.1242/dev.067074. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Han XH, Jin YR, Seto M, Yoon JK. A WNT/beta-catenin signaling activator, R-spondin, plays positive regulatory roles during skeletal myogenesis. J Biol Chem. 2011;286:10649–10659. doi: 10.1074/jbc.M110.169391. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Hao HX, Xie Y, Zhang Y, Charlat O, Oster E, Avello M, Lei H, Mickanin C, Liu D, Ruffner H, Mao X, Ma Q, Zamponi R, Bouwmeester T, Finan PM, Kirschner MW, Porter JA, Serluca FC, Cong F. ZNRF3 promotes Wnt receptor turnover in an R-spondin-sensitive manner. Nature. 2012;485:195–200. doi: 10.1038/nature11019. [DOI] [PubMed] [Google Scholar]
- Hartman BH, Reh TA, Bermingham-McDonogh O. Notch signaling specifies prosensory domains via lateral induction in the developing mammalian inner ear. Proc Natl Acad Sci. 2010;107:15792–15797. doi: 10.1073/pnas.1002827107. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Hayashi T, Cunningham D, Bermingham-McDonogh O. Loss of Fgfr3 leads to excess hair cell development in the mouse organ of Corti. Dev Dyn. 2007;236:525–533. doi: 10.1002/dvdy.21026. [DOI] [PubMed] [Google Scholar]
- Hayashi T, Ray CA, Bermingham-McDonogh O. Fgf20 is required for sensory epithelial specification in the developing cochlea. J Neurosci. 2008;28:5991–5999. doi: 10.1523/JNEUROSCI.1690-08.2008. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Hume CR, Bratt DL, Oesterle EC. Expression of LHX3 and SOX2 during mouse inner ear development. Gene Expr Patterns. 2007;7:798–807. doi: 10.1016/j.modgep.2007.05.002. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Hwang CH, Guo D, Harris MA, Howard O, Mishina Y, Gan L, Harris SE, Wu DK. Role of bone morphogenetic proteins on cochlear hair cell formation: analyses of Noggin and Bmp2 mutant mice. Dev Dyn. 2010;239:505–513. doi: 10.1002/dvdy.22200. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Jacques BE, Puligilla C, Weichert RM, Ferrer-Vaquer A, Hadjantonakis A, Kelley MW, Dabdoub A. A dual function for canonical Wnt/β-catenin signaling in the developing mammalian cochlea. Development. 2012;139:4395–4404. doi: 10.1242/dev.080358. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Jin YR, Turcotte TJ, Crocker AL, Han XH, Yoon JK. The canonical Wnt signaling activator, R-spondin2, regulates craniofacial patterning and morphogenesis within the branchial arch through ectodermal-mesenchymal interaction. Dev Biol. 2011;352:1–13. doi: 10.1016/j.ydbio.2011.01.004. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Kazanskaya O, Glinka A, del Barco Barrantes I, Stannek P, Niehrs C, Wu W. R-Spondin2 is a secreted activator of Wnt/beta-catenin signaling and is required for Xenopus myogenesis. Dev Cell. 2004;7:525–534. doi: 10.1016/j.devcel.2004.07.019. [DOI] [PubMed] [Google Scholar]
- Kiernan AE, Pelling AL, Leung KK, Tang AS, Bell DM, Tease C, Lovell-Badge R, Steel KP, Cheah KS. Sox2 is required for sensory organ development in the mammalian inner ear. Nature. 2005;434:1031–1035. doi: 10.1038/nature03487. [DOI] [PubMed] [Google Scholar]
- Kim KA, Wagle M, Tran K, Zhan X, Dixon MA, Liu S, Gros D, Korver W, Yonkovich S, Tomasevic N, Binnerts M, Abo A. R-Spondin family members regulate the Wnt pathway by a common mechanism. Mol Biol Cell. 2008;19:2588–2596. doi: 10.1091/mbc.E08-02-0187. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Lanford PJ, Lan Y, Jiang R, Lindsell C, Weinmaster G, Gridley T, Kelley MW. Notch signalling pathway mediates hair cell development in mammalian cochlea. Nature Genetics. 1999;21:289–292. doi: 10.1038/6804. [DOI] [PubMed] [Google Scholar]
- Liu S, Li W, Chen Y, Lin Q, Wang Z, Li H. Mouse auditory organ development required bone morphogenetic protein signaling. Neuroreport. 2011;22:396–401. doi: 10.1097/WNR.0b013e328346c10f. [DOI] [PubMed] [Google Scholar]
- Lu W, Kim KA, Liu J, Abo A, Feng X, Cao X, Li Y. R-spondin1 synergizes with Wnt3A in inducing osteoblast differentiation and osteoprotegerin expression. FEBS letters. 2008;582:643–650. doi: 10.1016/j.febslet.2008.01.035. [DOI] [PubMed] [Google Scholar]
- Mathew LK, Sengupta SS, Ladu J, Andreasen EA, Tanguay RL. Crosstalk between AHR and Wnt signaling through R-Spondin1 impairs tissue regeneration in zebrafish. FASEB J. 2008;22:3087–3096. doi: 10.1096/fj.08-109009. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Montcouquiol M, Rachel RA, Lanford PJ, Copeland NG, Jenkins NA, Kelley MW. Identification of Vangl2 and Scrb1 as planar polarity genes in mammals. Nature. 2003;423:173–177. doi: 10.1038/nature01618. [DOI] [PubMed] [Google Scholar]
- Morrison A, Hodgetts C, Gossler A, Hrabe de Angelis M, Lewis J. Expression of Delta1 and Serrate1 (Jagged1) in the mouse inner ear. Mech Dev. 1999;84:169–172. doi: 10.1016/s0925-4773(99)00066-0. [DOI] [PubMed] [Google Scholar]
- Mueller KL, Jacques BE, Kelley MW. Fibroblast growth factor signaling regulates pillar cell development in the organ of corti. J Neurosci. 2002;22:9368–9377. doi: 10.1523/JNEUROSCI.22-21-09368.2002. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Nam JS, Park E, Turcotte TJ, Palencia S, Zhan X, Lee J, Yun K, Funk WD, Yoon JK. Mouse R-spondin2 is required for apical ectodermal ridge maintenance in the hindlimb. Dev Biol. 2007a;311:124–135. doi: 10.1016/j.ydbio.2007.08.023. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Nam JS, Turcotte TJ, Smith PF, Choi S, Yoon JK. Mouse cristin/R-spondin family proteins are novel ligands for the Frizzled 8 and LRP6 receptors and activate beta-catenin-dependent gene expression. J Biol Chem. 2006;281:13247–13257. doi: 10.1074/jbc.M508324200. [DOI] [PubMed] [Google Scholar]
- Nam JS, Turcotte TJ, Yoon JK. Dynamic expression of R-spondin family genes in mouse development. Gene Expr Pattern. 2007b;7:306–312. doi: 10.1016/j.modgep.2006.08.006. [DOI] [PubMed] [Google Scholar]
- Ohkawara B, Glinka A, Niehrs C. Rspo3 binds syndecan 4 and induces Wnt/PCP signaling via clathrin-mediated endocytosis to promote morphogenesis. Dev Cell. 2011;20:303–314. doi: 10.1016/j.devcel.2011.01.006. [DOI] [PubMed] [Google Scholar]
- Ohyama T, Basch ML, Mishina Y, Lyons KM, Segil N, Groves AK. BMP signaling is necessary for patterning the sensory and nonsensory regions of the developing mammalian cochlea. J Neurosci. 2010;30:15044–15051. doi: 10.1523/JNEUROSCI.3547-10.2010. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Ootani A, Li X, Sangiorgi E, Ho QT, Ueno H, Toda S, Sugihara H, Fujimoto K, Weissman IL, Capecchi MR, Kuo CJ. Sustained in vitro intestinal epithelial culture within a Wnt-dependent stem cell niche. Nat Med. 2009;15:701–706. doi: 10.1038/nm.1951. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Pan N, Jahan I, Kersigo J, Kopecky B, Santi P, Johnson S, Schmitz H, Fritzsch B. Conditional deletion of Atoh1 using Pax2-Cre results in viable mice without differentiated cochlear hair cells that have lost most of the organ of Corti. Hear Res. 2011;275:66–80. doi: 10.1016/j.heares.2010.12.002. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Puligilla C, Feng F, Ishikawa K, Bertuzzi S, Dabdoub A, Griffith AJ, Fritzsch B, Kelley MW. Disruption of fibroblast growth factor receptor 3 signaling results in defects in cellular differentiation, neuronal patterning, and hearing impairment. Dev Dyn. 2007;236:1905–1917. doi: 10.1002/dvdy.21192. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Salinas PC. Wnt signaling in the vertebrate central nervous system: from axon guidance to synaptic function. Cold Spring Harb Perspect Biol. 2012:4. doi: 10.1101/cshperspect.a008003. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Whitlon DS, Grover M, Tristano J, Williams T, Coulson MT. Culture conditions determine the prevalence of bipolar and monopolar neurons in cultures of dissociated spiral ganglion. Neuroscience. 2007;146:833–840. doi: 10.1016/j.neuroscience.2007.01.036. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Yamada W, Nagao K, Horikoshi K, Fujikura A, Ikeda E, Inagaki Y, Kakitani M, Tomizuka K, Miyazaki H, Suda T, Takubo K. Craniofacial malformation in R-spondin2 knockout mice. Biochem Biophys Res Co. 2009;381:453–458. doi: 10.1016/j.bbrc.2009.02.066. [DOI] [PubMed] [Google Scholar]
- Yang T, Kersigo J, Jahan I, Pan N, Fritzsch B. The molecular basis of making spiral ganglion neurons and connecting them to hair cells of the organ of Corti. Hear Res. 2011;278:21–33. doi: 10.1016/j.heares.2011.03.002. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Yoon JK, Lee JS. Cellular signaling and biological functions of R-spondins. Cell Signal. 2012;24:369–377. doi: 10.1016/j.cellsig.2011.09.023. [DOI] [PMC free article] [PubMed] [Google Scholar]