Abstract
Hepatic artery pseudoaneurysm is a rare complication of amebic or pyogenic liver abscesses, and it is generally diagnosed because of hemobilia due to rupture of the aneurysm into the biliary tract. The authors describe a case of vascular complication in a patient affected by amebic liver abscess. Pseudoaneurysm was diagnosed and resolved without hemobilia.
Keywords: Amebiasis, Abscess, Pseudoaneurysm
Sommario
Gli pseudoaneurismi dell'arteria epatica sono una complicanza molto rara degli ascessi epatici amebici e da piogeni e la maggior parte vengono diagnosticati perché si complicano con emobilia.
Gli autori descrivono un caso di complicanza vascolare in un paziente affetto da ascesso amebico del fegato diagnosticato e risolto senza la rottura nelle vie biliari.
Introduction
Amebiasis occurs in 10% of the world's population and is most common in tropical and subtropical regions. Amebic liver abscess is the most frequent extraintestinal manifestation of amebiasis and it may be complicated with rupture into the pleural, pericardial and peritoneal spaces [1,2]. The authors present a case of amebic liver abscess in a patient who developed a rare vascular complication, i.e. a pseudoaneurysm of the hepatic artery. Amoebic abscess and pseudoaneurysm resolved after 3 echo-guided percutaneous aspirations combined with Metronidazole therapy which led to anatomic resolution of the aneurysmal lesion.
Case report
After a stay in the Balearic Isles, a 31-year old man presented with fever, anorexia, cough and left-sided thoracic pain of 7 days' duration. Informed consent was obtained from the patient, who was guaranteed privacy-protection and a proper use of personal data according to the current privacy law.
Clinical examination revealed that the temperature was above normal, but the patient was non-icteric. The liver was soft and smooth, enlarged by 3 cm below the right costal margin and painful but without ascites or splenomegaly. Outcome of central nervous system, cardiovascular and respiratory examinations was normal.
Laboratory examination revealed hemoglobin levels of 11.5 g/dl, leukocyte count 13,000 with 68% neutrophils. Hematic sedimentation velocity, C-reactive protein (RCP) and alkaline phosphatase (AP) values were elevated. Other hepatic and renal function tests were within normal limits.
Ultrasound of the abdomen showed hepatomegaly with two lesions and a slight pericardial effusion (Fig. 1a). One lesion was located in the 8th segment; it measured 75 mm and was hypoechoic with irregular, blurred outlines. The other lesion measured 15 mm in diameter; it appeared anechoic with an echoic rim; color Doppler showed a chaotic arterial flow and a Korean flag-shaped lesion suspicious for hepatic artery aneurysm (Fig. 1b, c).
Fig. 1.
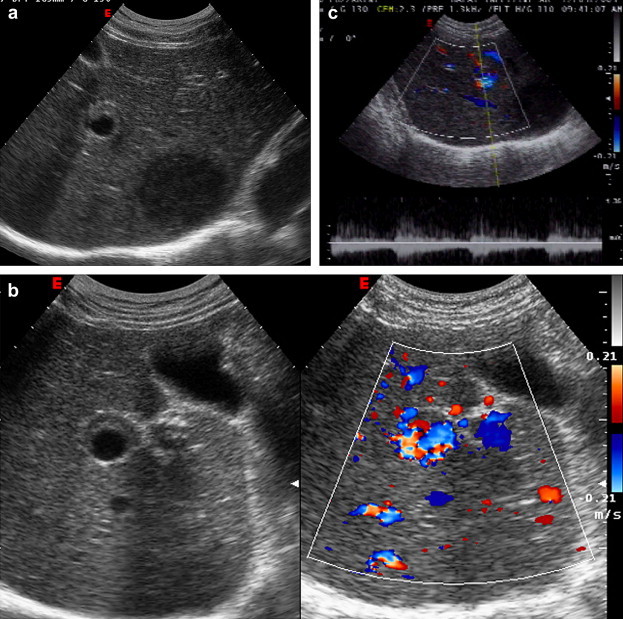
a. Right subcostal oblique scan: in the 8th segment a liver lesion is visible. It is characterized by a hypoechoic aspect with irregular, blurred outlines and alteration of the hepatic artery which appears dilated with echoic contours. b, c. Color Doppler of the hepatic artery shows a Korean flag-shaped lesion with chaotic arterial flow at spectral analysis.
The patient underwent enhanced computed tomography (CT) which evidenced a hepatic artery aneurysm and an abscess located in the 8th hepatic segment (Fig. 2). Subsequently CEUS of the liver confirmed a liver abscess and an avascular lesion around an aneurysmal dilatation of the hepatic artery (Fig. 3).
Fig. 2.

Enhanced CT shows an aneurysm of the hepatic artery.
Fig. 3.

Right subcostal oblique scan: echo-guided aspiration of the liver abscess; the needle has been introduced into the abscessual lesion.
The patient underwent echo-guided aspiration of the liver abscess, and 150 ml of thick, anchovy paste colored pus was drained. The pus resulted negative at direct examination for Entamoeba histolytica (Fig. 4a, b).
Fig. 4.
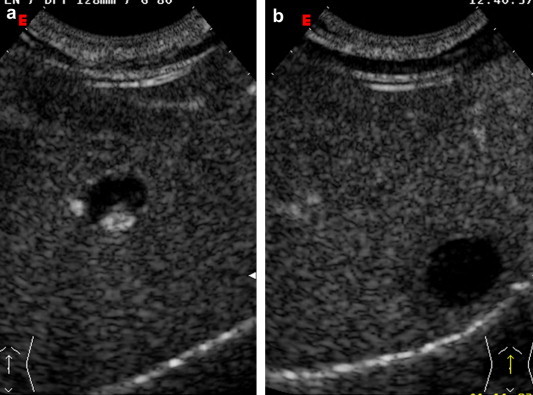
a. Right subcostal oblique scan 30 s after infusion of 2.4 ml Sonovue. The hepatic artery is intensely enhanced, surrounded by an avascular area due to a perivascular necrotic lesion. b. The anechoic lesion is evident, caused by an avascular liver abscess located in the 8th segment.
Blood culture on samples taken during pyrexia was negative, but amebic serology (enzyme-linked immunosorbent) was 1:1024. Sonographic findings in combination with outcome of serology were considered suggestive of hepatic amebic abscess.
The patient was administered intravenous infusions of 500 mg Metronidazole every 8 h and underwent 3 echo-guided percutaneous aspirations of the abscess draining 70 and 30 ml of pus, respectively. The abscess cavity was cleaned with 50 ml of Metronidazole. This therapy made the symptoms disappear in 72 h, and after 15 days the volume of the liver abscess (25 mm in diameter at the first examination) was reduced, leading to anatomic resolution in 60 days.
After 15 days' therapy, color Doppler showed a reduced diameter of the aneurismal lesion with disappearance of the Korean flag-shaped aspect and chaotic arterial flow (Fig. 5). CEUS performed after 15 days' therapy showed a reduced avascular area around the hepatic artery, which disappeared after two months.
Fig. 5.

Color Doppler, right subcostal oblique scan: After 15 days' therapy the hepatic artery appears with normal flow and the diameter of the liver abscess is reduced (arrows).
Discussion
Amebiasis has a high prevalence in tropical and subtropical regions where it affects 50% of the population causing 40,000 deaths each year. In Europe and in North America, the incidence is increasing due to traveling to high risk countries, and sporadic cases and small epidemics in closed communities are reported in the literature [1]. Gastro-intestinal amebiasis develops liver abscess in 2–15% of cases but only 20% of these patients remember a dysenteric episode. Complications of an amebic liver abscess can be widespread and involve any part of the body. Complications can be divided into general complications (cerebral, renal, myocardial lesions, skin and ocular involvement, hepatic coma, hepatorenal failure), local complications (rupture into the peritoneal, pleural or pericardial space, diaphragm or sub-diaphragm abscess, mediastinitis, myocardial abscess and vascular complications), complications of silent lesions, and iatrogenic complications [2].
Early detection of complications is important in order to reduce morbidity and mortality. The most frequent complication of amoebic liver abscess is rupture into the pleural and peritoneal space, but the most dangerous complication is rupture into the pericardial space. Chronic amebic abscess is more frequent in developing countries and may cause skin involvement and/or perforation. Vascular complications reported in the international literature are portal, hepatic and vena cava thrombosis [3,4].
In its passage through the groove of the right lobe, the inferior vena cava may be obstructed and/or thrombosed by an adjacent abscess, particularly if the lesion is very big, and rupture of a liver abscess directly into the inferior vena cava has been reported. Obstruction and thrombosis of the portal or hepatic vein are considered rare vascular complications of hepatic amoebic abscess, and pseudoaneurysm of the hepatic artery is even rarer. This complication occurs due to an abscess forming in the periarterial hepatic tissue and/or to an inflammatory process of the arterial wall.
Most of hepatic artery pseudoaneurysms in patients with amebic or pyogenic liver abscess are diagnosed because of complications involving hemobilia, due to rupture of the aneurysm into the biliary tract [5,6].
The present case of vascular complication occurred in a patient affected by amebic liver abscess, but the pseudoaneurysm was diagnosed and resolved without hemobilia. Color Doppler and enhanced CT showed a hepatic artery aneurysm, but CEUS showed a periarterial abscessual lesion and this permitted diagnosis of hepatic artery pseudoaneurysm.
Color Doppler and CEUS confirmed the complete anatomic resolution of the hepatic vascular lesion.
Conflict of interest statement
The authors have no conflict of interest.
References
- 1.Stanley S.L., Jr. Amoebiasis. Lancet. 2003;361:1025–1034. doi: 10.1016/S0140-6736(03)12830-9. [DOI] [PubMed] [Google Scholar]
- 2.Wells C.D., Arguedas M. Amoebic liver abscess. South Med J. 2004;97:673–682. doi: 10.1097/00007611-200407000-00013. [DOI] [PubMed] [Google Scholar]
- 3.Krishnan K., Badarinath S., Bhusnurmath S.R. Vascular complications of hepatic amoebiasis: a retrospective study. Indian J Pathol Microbiol. 1986;29:293–296. [PubMed] [Google Scholar]
- 4.Sodhi K.S., Ojili V., Sakhuja V., Khandelwal N. Hepatic and inferior vena caval thrombosis: vascular complication of amebic liver abscess. J Emerg Med. 2008;34:155–157. doi: 10.1016/j.jemermed.2007.05.045. [DOI] [PubMed] [Google Scholar]
- 5.Yanagisawa M., Kaneko M., Aizawa T., Michimata T., Takagi H., Mori M. A case of amebic abscess complicated by hemobilia due to rupture of hepatic artery aneurysm. Hepatogastroenterology. 2002;49:375–378. [PubMed] [Google Scholar]
- 6.Gopanpallikar A., Rathi P., Sawant P., Gupta R., Dhadphale S., Deshmukh H.L. Hepatic artery pseudoaneurysm associated with amebic liver abscess presenting as upper GI hemorrage. Am J Gastroenterol. 1997;92(8):1391–1393. [PubMed] [Google Scholar]


