Abstract
Vector-borne diseases are particularly responsive to changing environmental conditions. Diurnal temperature variation has been identified as a particularly important factor for the development of malaria parasites within vectors. Here, we conducted a survey across France, screening populations of the house sparrow (Passer domesticus) for malaria (Plasmodium relictum). We investigated whether variation in remotely-sensed environmental variables accounted for the spatial variation observed in prevalence and parasitemia. While prevalence was highly correlated to diurnal temperature range and other measures of temperature variation, environmental conditions could not predict spatial variation in parasitemia. Based on our empirical data, we mapped malaria distribution under climate change scenarios and predicted that Plasmodium occurrence will spread to regions in northern France, and that prevalence levels are likely to increase in locations where transmission already occurs. Our findings, based on remote sensing tools coupled with empirical data suggest that climatic change will significantly alter transmission of malaria parasites.
Climate change and its resulting habitat alteration are expected to have major impacts on the dynamics of infectious diseases1,2,3,4,5. Global warming effects on human vector-borne diseases, such as dengue or malaria, have been extensively investigated6,7; however, it can be extremely challenging to address the impacts of future climate change on human infectious diseases, as the influence of socioeconomic and environmental factors are often intertwined8,9. Although it is now well-documented that the malaria transmission cycle is sensitive to climate, there is a mismatch between the predicted expansion of areas with endemic malaria based on climatic models and the observed reduction in endemicity over the last century10. Therefore, the assumed link between rising temperature and the spread of human malaria is heavily debated8. A way to avoid the problem of the antagonistic effects of socioeconomic and environmental factors is to focus on wildlife parasites, such as the avian malaria.
Recently, temperature constraints on the sporogonic development of the avian Plasmodium parasite have been investigated within the mosquito vector11, demonstrating that any increase of temperature could facilitate the spread of the parasite to areas that present suitable environmental conditions for its development. In addition, a literature survey from more than 3,000 avian species found that the prevalence of Plasmodium has increased in parallel with climate changes12. While knowledge of parasite response to climate change is useful, identifying the environmental drivers of local malaria prevalence and parasitemia is paramount to linking the disease to expected environmental changes.
From a physiological perspective, in addition to mean temperatures, diurnal fluctuations in temperature have been shown to affect the rate of parasite development, and the essential elements of mosquito biology3. In addition, recent work has stressed that vector competence could be reduced at higher temperature, demonstrating that vectorial capacity of malaria mosquitoes needs an optimum temperature13. With the availability of fine-scale remotely sensed variables for both current and future climate conditions14, an investigation of how such variables are related to detailed empirical field data on host-parasite dynamics can add much to our understanding of the potential direction of evolutionary changes induced by environmental modifications15.
Here, we extensively sampled a ubiquitous host, the house sparrow (Passer domesticus) and its parasite Plasmodium relictum across a large region. We predict that prevalence (i.e. the proportion of infected individuals in a population) of Plasmodium relictum should be linked to climatic conditions, temperature and rainfall variables, which affect vector and parasite development16; parasitemia (i.e. the density of parasites within the host) which reflects the ability of the host to control the infection should exhibit a weaker relationship with environmental characteristics17,18. In addition, based on recent work on avian malaria in northern latitude19 and on an elevation gradient20, we predict that under climate change scenarios, the occurrence of Plasmodium should experience significant geographical range changes.
Results
We determined the prevalence and parasitemia of Plasmodium relictum of 1750 individuals sampled in 24 different house sparrow populations in France (Table 1). We found 24% of infected sparrows with the parasite Plasmodium relictum (lineages Plasmodium SGS1, GenBank accession number JN164729 and Plasmodium GRW11 GenBank accession number JN164731). Prevalence varied from 0% to 78% and parasitemia (relative quantification, see methods) from 0 to 3.7−04 between populations (Table 1). In addition to SGS1 and GRW11, we also found seven other parasite lineages, referring to other Plasmodium morphospecies or to Haemoproteus sp. The very low prevalence of these lineages (ranging from 1% to 8%) prevented us to reliably test the association with climatic variables, and we therefore decided to focus on Plasmodium relictum.
Table 1. GPS coordinates, sample size, prevalence (%) and parasitemia (relative quantification log + 1 ± SE) are given for each of the 24 populations sampled from South to North. No SE is given for Quimper and Kerinou since only one individual was infected in each site.
| Site | N | Prevalence | Parasitemia | Latitude | Longitude |
|---|---|---|---|---|---|
| Saintes Maries de la Mer | 112 | 72.32 | 1.01−05 (±1.81−06) | 43°27'10" N | 4°25'43" E |
| Arles | 49 | 78.57 | 2.10−04 (±1.80−04) | 43°40'35.90" N | 4°37'40.12" E |
| Chizé | 54 | 56.52 | 3.83−06 (±1.48−06) | 46°08'49.35" N | 0°25'31.98" W |
| Dijon | 20 | 55 | 9.29−05 (±4.57−05) | 47°19'18" N | 5°02'29" E |
| Hoedic | 685 | 11.09 | 5.26−05 (±3.83−05) | 47°20'23.46" N | 2°52'40.56" W |
| Cosnes Cours sur Loire | 61 | 50 | 3.57−06 (±6.67−05) | 47°23'15.05" N | 2°54'27.88" E |
| Groix | 45 | 0 | 0 | 47°38'21.95" N | 3°27'13.04" W |
| Vannes | 42 | 23.81 | 3.21−05 (±9.40−06) | 47°39'21" N | 2°45'37" W |
| Ploemeur | 15 | 13.33 | 1.35−05 (±3.51−06) | 47°44'08.50" N | 3°25'38.18" W |
| Languidic | 56 | 16.07 | 3.33−05 (±1.53−05) | 47°50'03" N | 3°09'24" W |
| Quimper | 32 | 3.13 | 3.69−04 | 47°59'51.58" N | 4°05'52.76" W |
| Sein | 32 | 0 | 0 | 48°02'13.94" N | 4°51'06.23" W |
| Kerinou | 26 | 3.85 | 1.58−04 | 48°20'32.92" N | 4°45'21.69" W |
| Crennes | 55 | 30.77 | 4.57−05 (±3.22−05) | 48°22'42.54" N | 0°16'44.60" W |
| Molène | 30 | 0 | 0 | 48°23'46.51" N | 4°57'30.36" W |
| Anglus | 58 | 35.71 | 2.81−05 (±1.97−05) | 48°23'54.54" N | 4°44'23.47" E |
| Saint Elven | 15 | 0 | 0 | 48°27'41.15" N | 4°22'22.07" W |
| Ouessant | 64 | 0 | 0 | 48°27'48.15" N | 5°05'16.57" W |
| Wissous | 39 | 48.72 | 8.79−06 (±3.05−06) | 48°43'51.48" N | 2°19'38.69" E |
| Cachan | 20 | 30 | 1.50−05 (±1.40−05) | 48°47'41.08" N | 2°20'06.64" E |
| Rully | 58 | 35.19 | 1.08−04 (±4.76−05) | 48°49'30.91" N | 0°42'52.15" W |
| Paris | 52 | 30.77 | 1.97−06 (±1.53−06) | 48°50'39.47" N | 2°21'43.62" E |
| Crégy les Meaux | 85 | 41.1 | 2.26−06 (±1.90−06) | 48°58'40.54" N | 2°52'36.15" E |
| Thieux | 45 | 33.33 | 4.57−05 (±3.64−05) | 49°32'36.74" N | 2°19'01.13" E |
The characteristics of each site were determined using a set of environmental variables that included nineteen bioclimatic metrics at 1 km from the WorldClim dataset14 (http://www.worldclim.org) and elevation measure. We constructed classification trees using a Random Forest model21 under the R framework22, with climate variables (extracted for each site) as predictors, and both parasitemia and prevalence as response variables.
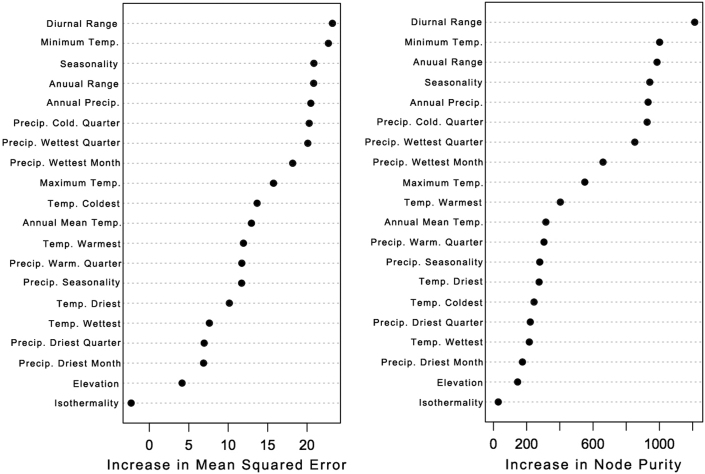
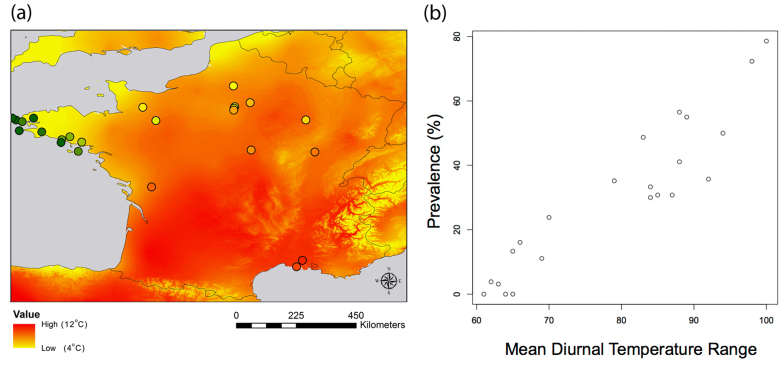
Bioclimatic variables and elevation did little to explain parasitemia, and optimized models never explained more than 2% of variation in parasitemia. In contrast, models attempting to explain variation in malaria prevalence performed well, with up to 83% of all variation explained by only bioclimatic and elevation variables. Of these variables, under both criteria used to evaluate performance (percentage of increase of mean squared error and increase of purity; Fig. 1), temperature variables were the most important in describing where difference in prevalence occurred (Fig. 1). Interestingly, sampled locations with higher diurnal temperature ranges were areas that had higher malaria prevalence in sparrow hosts (Fig. 2). In our predictive map, when extrapolating the correlation between diurnal temperature range and prevalence to the all country, we found that mountains areas, Brittany and the north coast is presenting low prevalence while the south of France is showing high prevalence. A higher temperature seasonality and higher minimum temperature of the coldest month were also bioclimatic variables explaining a higher prevalence in populations.
Figure 1. Importance scores for each environmental variable as related to Plasmodium prevalence, that were used as predictors in random forest algorithm models.
Increase in mean square error (left panel) is calculated as the average increase in squared residuals of the test set when the explanatory variable is randomly permuted. When a given variable has little predictive power, its permutation will not cause substantial difference in model residuals, therefore a higher increase in mean square error is indicative of a more important variable. A larger increase in node purity (right panel) represents more homogeneous calls within partitions of the data. Each measure of variable importance identified diurnal temperature range as the top predictor, along with minimum annual temperature, temperature seasonality, and annual temperature range as being top predictors of Plasmodium prevalence.
Figure 2.
(a) Map showing i) prevalence of Plasmodium at sampled locations (dots in warmer colors equal higher prevalence) overlayed on ii) a spatially continuous map of diurnal temperature range (the most important variable in our models, warmer colors indicate higher diurnal temperature ranges); (b) Correlation between the prevalence (percentage of infected individuals per site) and the mean diurnal temperature range (Celsius degree x10). Map created in ArcMap 10 (Environmental Systems Resource Institute, ArcMap 10.0 ESRI, Redlands, California).
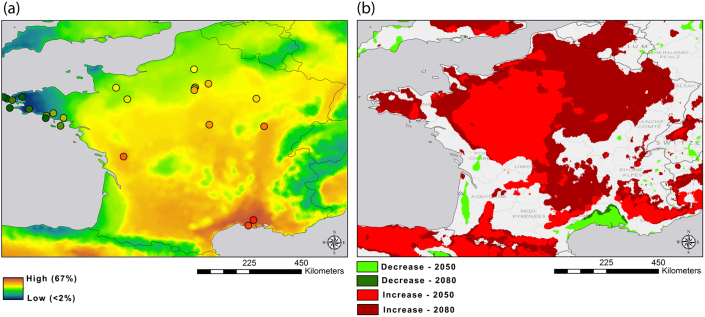
When looking at the map with current environmental conditions, malaria prevalence is low in the north, on the northwest and southwest coast as well as in mountain areas. Prevalence seems especially high in the south of France (Fig. 3a). Under future climate scenarios (IPCC 4th Assessment A1 scenario for the year 2050 and 2080), we found a shift in regions suitable for Plasmodium occurrence. Sparrow populations from most of the geographical area covered by this study are predicted to suffer from a substantial increase of malaria prevalence (Fig. 3b). Interestingly the south of France, where prevalence is high for now, will exhibit a decrease in prevalence in the future. However, the entire north and the northwest coast will show a drastic increase in prevalence, possibly exposing house sparrows and other passerine species to more severe parasite-mediated selection pressures.
Figure 3. Predictive map of malaria prevalence under (a) current environmental conditions and (b) future conditions in years 2050 and 2080.
Green areas indicate those that will experience a predicted decrease in prevalence by at least 1% as compared to current predictions, and red areas are those predicted to have an increase in prevalence by at least 10% over current predictions. Maps created in ArcMap 10 (Environmental Systems Resource Institute, ArcMap Release10.0, ESRI, Redlands, California).
Discussion
The study presented here suggests that avian malaria prevalence is greatly affected by bioclimatic conditions, and that Plasmodium transmission will increase as warming trends continue. As a large portion of the region studied here is predicted to have both mean diurnal and temperature seasonality range increases in the future, and these variables will associated with higher prevalence of avian malaria, we expect avian populations in this region to be under increasing threat from this disease.
Recent laboratory studies suggest differential development and transmission of malaria under varying temperature conditions3. Using a lineage of rodent malaria, Plasmodium chabaudi and the vector Anopheles stephensi, it has been shown that fluctuations around cooler temperatures (18°C) enhanced parasite transmission, whereas fluctuations around a warmer temperature (24°C) impaired it3. Given the fact that our study region is thermally more analogous to the cooler temperature regime in this laboratory study (mean value of 16.6°C), the effects of diurnal temperature range on prevalence in our models is consistent with laboratory findings. Our results suggest that temperature fluctuations may substantially influence malaria transmission even in a natural setting where parasites, vectors, and hosts are exposed to a full range of variable conditions.
Previous work has reported that both prevalence and parasitemia are shaped by a combination of environmental and host traits17,23. While links between prevalence and environmental conditions were made in this study, there was little support for associations between the climate and parasitemia in host populations. The strong response of malaria prevalence to temperature conditions may be due to the combined effect of higher abundance of vectors and accelerated developmental time within the vector in warmer areas. Conversely, parasitemia (the number of parasites in infected sparrows) is likely determined by the balance between the capacity of the parasite to multiply and the capacity of the host to control the infection, factors that may be under less direct influence from environmental variability.
The house sparrow is a ubiquitous passerine host in Europe, and is used in this study as an indicator species to forecast the response of avian malaria within a larger avian community facing undergoing climate changes. We provide here clear-cut results regarding the future distribution of a generalist avian malaria parasite, Plasmodium relictum. Plasmodium relictum is particularly interesting with respect to the potential consequences of climate change because it is maintained by multiple host species and might easily switch to new hosts as long as it spreads into new areas24. The example of the introduction of Plasmodium relictum (the lineage GRW4) and its competent vector, Culex quinquefasciatus, in Hawaii is well known to have decimated immunologically naïve endemic avian species25,26. Of course, each host species is likely to experience differential levels of exposure and response, and as such, this study should only serve as a first prediction of the spatial extent of avian malaria under future conditions. Our study also provides evidence that with impending global warming, malaria will spread in the north of France and that populations already experiencing malaria infections will exhibit higher prevalence. The predictive maps also show that the southwest coast remains relatively unaffected by malaria infection, as well as the center. These findings can be used as a testable spatial template for verification by future ground-truthing efforts.
The impact of climate change on parasite distribution is likely to be extremely complex, and influenced by numerous factors, including but not limited to local environmental conditions. We do not discount here other variables that may affect transmission, such as vector development and its life cycle; a survey of vector abundance and diversity will be performed in parallel with host populations. In this time of rapid global change, the goal of our study is to reveal and emphasize the necessity of studying the effects of ecological change on vector-borne parasites in wildlife, with obvious conservation implications, but also in consideration of larger management and policy decisions, as our methods may be useful when applied to other host-parasite systems.
Methods
Samples were collected across France at 24 sites between 2004 and 2008. In total, 1750 individuals were captured using mist-nets and nest boxes (Table 1). We banded them with a numbered metal ring and blood samples (20 μl) were collected from the brachial vein and stored in lysis buffer (10 mM Tris-HCL pH 8.0, 100 mM EDTA, 2% SDS).
DNA was extracted from whole blood using the Qiaquick 96 Purification Kit (QIAGEN) according to the manufacturer's instructions. For Plasmodium detection, we used a nested PCR to amplify a 600 bp fragment of cytochrome b with the primers HAEMF/HAEMR2 - HAEMNF/HAEMNR227. The PCR products were run out on a 2% agarose gel using 1×TBE, and visualized by an ethidium bromide stain under ultraviolet light to check for positive infections. We identified lineages by sequencing the fragments on an ABI3730XL, Applied Biosystems. For each positive PCR product, we also performed a quantitative PCR to obtain parasitemia28. Briefly, for each individual, we conducted two qPCR in the same run: one targeting the nuclear 18s rDNA gene of Plasmodium and the other targeting the 18s rDNA gene of bird (see ref. 28 for primers and probe sequences). Parasite intensities were calculated as relative quantification values (RQ) as 2e – (Ct18s Plasmodium - Ct18s bird) using the software SDS 2.2 (Applied Biosystem). Ct represents the number of PCR cycles at which fluorescence is first detected as statistically significant above the baseline, which is inversely correlated with the initial amount of DNA in a sample. RQ can be interpreted as the fold-amount of target gene (Plasmodium 18s rDNA) with respect to the amount of the reference gene (host 18s rDNA). All qPCR were run on an ABI 7900HT real-time PCR system (Applied Biosystem).
Modeling under random forest
We used a set of moderately high-resolution climate and satellite remote sensing variables to characterize the environmental differences among our sampling areas. Variables were re-aggregated from their native resolutions to 1 km resolution. We used 19 bioclimatic variables (representing both temperature and precipitation) from the WorldClim database14 which are 50-year averages (1950–2000) of annual means, seasonal extremes and degrees of seasonality in temperature and precipitation, and represent biologically meaningful variables for characterizing species range15,19,29: (http://www.worldclim.org/), and we used elevation measure, downloaded from the Earth Remote Sensing Data Analysis Center (ERSDAC, http://www.jspacesystems.or.jp/ersdac/eng/index.E.html) at ~30 m resolution.
We modelled the ability of bioclimatic variables to predict variation in two response variables in House sparrow hosts, parasitemia and prevalence of avian malaria. We ran 5000 regression trees in a random forest model (RandomForest21) under the R framework (R development Core Team 2004) to measure the percent variation explained in each response. We extracted two measures of variable importance for each model, the percent increase in mean square error when individual variables are randomly permutated, and the total decrease in node impurities from splitting each variable, as measured by the residual sum of squares. Due to the inability of our suite of variables to predict variation in parasitemia (see Results and Discussion), we focused further analyses only on malaria prevalence.
Most statistical procedures and traditional data modeling technique (such as linear regression or ANOVA) measure variable importance indirectly by selecting variables using criteria such as statistical significance and Akaike's Information Criterion. The Random Forest has a different approach; it is a non-parametric algorithm method. We used this method because Random Forest procedures (i) do not require the use of any particular model (which might be difficult to assign given a complex response such as disease prevalence), (ii) do not require normalized data, and (iii) have consistently outperformed traditional regression procedures on a number of datasets15,30,31,32. The advantage of random forest models is their ability to predict a continuous (in this case, prevalence) rather than categorical (presence/absence) variable across a landscape, and their ability to model complex interactions among predictor variables. Also, autocorrelation between predictors is not an obstacle for these algorithms because if climatic variables are highly correlated, then removal of one variable does not affect the model, because an autocorrelated variable can just take its place in the model. In our study, for the case of prevalence, each of the first couple of variables explained unique variation in prevalence; this is not the case for the parasitemia data.
In details, for each tree in the forest, there is a misclassification rate for the out-of-bag observations. To assess the importance of a specific predictor variable, the values of the variable are randomly permuted for the out-of-bag observations, and then the modified out-of-bag data are passed down the tree to get new predictions. The difference between the misclassification rate for the modified and original out-of-bag data, divided by the standard error, is a measure of the importance of the variable. The mean square error is an estimate of the full model's error rate, whereas the purity is a measure of how often an out-of-bag record from the set would be incorrectly called in the “child” leaves of the tree as compared to the “parent”. It is a measure of homogeneity versus heterogeneity in each node of the branch. The purity index represented therefore the sum of how each variable contributes to the homogeneity of out-of-bag calls, and that a larger increase in purity represents more homogeneous calls within partitions of the data.
In addition, we created spatial predictions by applying the relationships determined by the models to 30,000 randomly-selected points within the host range, to predict prevalence in unsampled areas under current climatic conditions. These predictions were then used to create interpolations between points using an Ordinary Kriging33, in order to generate a continuous spatial prediction map of current malaria prevalence (Fig. 3a).
Finally, using the current relationship between climate and malaria prevalence, we projected the spatial variation of prevalence under future climate conditions using environmental data downloaded from the 4th Assessment of the IPCC under an A1 scenario, http://www.worldclim.org). These projections were made for two decadal time periods, 2050 and 2080, in an attempt to understand how patterns of malaria prevalence are likely to change under future climate conditions (Fig. 3b).
Author Contributions
C.L. and G.S. designed the research and wrote the first draft. R.J.H. generated and compiled the environmental data layers used in the analyses. C.B., R.J., S.G., A.Z.L. and O.C. collected field data. C.L. and C.B. performed the molecular analyses. All authors discussed the results and contributed to writing the paper.
Acknowledgments
We thank all the volunteers from the National Museum of Natural History of Paris and from the University of Bourgogne for their help on the field and for providing samples: F. Baroteau, J. Besnault J. Birard, M. Brenier, L. Brucy, P. Bulens, E. Cellier-Holzem, P. Cannesson, G. Chaussi, B. Faivre, P. Fiquet, C. Girard, R. Guerreiro, D. Lavogiez, J. Marion, F. Martayan, G. Massez, P. Ollivier, V. Ternois, and M. Zucca. Financial support was provided by the Région Ile de France to C.L., R.J. and G.S., the Région Bourgogne to G.S., the CNRS Program MIE to G.S.
References
- Harvell C. D. et al. Ecology - Climate warming and disease risks for terrestrial and marine biota. Science 296, 2158–2162 (2002). [DOI] [PubMed] [Google Scholar]
- Massad E., Coutinho F. A. B., Lopez L. F. & da Silva D. R. Modeling the impact of global warming on vector-borne infections. Physics Life Rev. 8, 169–199 (2011). [DOI] [PubMed] [Google Scholar]
- Paaijmans K. P. et al. Influence of climate on malaria transmission depends on daily temperature variation. Proc. Natl. Acad. Sci. USA 107, 15135–15139 (2010). [DOI] [PMC free article] [PubMed] [Google Scholar]
- Rohr J. R. et al. Frontiers in climate change-disease research. Trends Ecol. Evol. 26, 270–277 (2011). [DOI] [PMC free article] [PubMed] [Google Scholar]
- Fuller T. et al. The ecology of emerging infectious diseases in migratory birds: an assessment of the role of climate change and priorities for future research. EcoHealth 9, 80–88 (2012). [DOI] [PubMed] [Google Scholar]
- Patz J. A. & Reisen W. K. Immunology, climate change and vector-borne diseases. Trends Immunol. 22, 171–172 (2011) [DOI] [PubMed] [Google Scholar]
- Patz J. A. & Olson S. H. Malaria risk and temperature: Influences from global climate change and local land use practices. Proc. Natl. Acad. Sci. USA 103, 5635–5636 (2006). [DOI] [PMC free article] [PubMed] [Google Scholar]
- Gething P. W. et al. Climate change and the global malaria recession. Nature 465, 342–345 (2010). [DOI] [PMC free article] [PubMed] [Google Scholar]
- Colwella D. D., Dantas-Torresb F. & Otrantob D. Vector-borne parasitic zoonoses: Emerging scenarios and new perspectives. Vet. Parasitol. 182, 14–21 (2011). [DOI] [PubMed] [Google Scholar]
- Bouma M. J., Baeza A., terVeen A. & Pascual M. Global malaria maps and climate change: a focus on East African highlands. Trends Parasitol. 27, 421–422 (2011). [DOI] [PubMed] [Google Scholar]
- LaPointe D. A., Goff M. L. & Atkinson C. T. Thermal constraints to the sporogonic development and altitudinal distribution of avian malaria Plasmodium relictum in Hawaii. J. Parasitol. 96, 318–324 (2010). [DOI] [PubMed] [Google Scholar]
- Garamszegi L. Z. Climate change increases the risk of malaria in birds. Glob. Change Biol. 17, 1751–1759 (2011). [Google Scholar]
- Paaijmans K. P., Blanford S., Chan B. H. K. & Thomas M. B. Warmer temperatures reduce the vectorial capacity of malaria mosquitoes. Biol. Lett. 8, 465–468 (2012). [DOI] [PMC free article] [PubMed] [Google Scholar]
- Hijmans R. J., Cameron S. E., Parra J. L., Jones P. G. & Jarvis A. Very high resolution interpolated climate surfaces for global land areas. Int. J. Climatol. 25, 1965–1978 (2005). [Google Scholar]
- Sehgal R. N. M. et al. Spatially explicit predictions of blood parasites in a widely distributed African rainforest bird. Proc. R. Soc. Lond. B. 278, 1025–1033 (2010). [DOI] [PMC free article] [PubMed] [Google Scholar]
- Rogers D. J. & Randolph S. E. Climate change and vector- borne diseases. In: Advances in Parasitology, Vol 62, pp 345–381, Elsevier Academic Inc, San Diego (2006). [DOI] [PubMed] [Google Scholar]
- Knowles S. C. L. et al. Molecular epidemiology of malaria prevalence and parasitaemia in a wild bird population. Mol. Ecol. 20, 1062–1076 (2011). [DOI] [PubMed] [Google Scholar]
- Kaslow R. A., McNicholl J. & Hill A. V. S Genetic Susceptibility to Infectious Diseases. Oxford University Press, Oxford. (2008).
- Loiseau C. et al. First evidence and predictions of Plasmodium transmission in Alaskan bird populations. PloS ONE 7, e44729 (2012). [DOI] [PMC free article] [PubMed] [Google Scholar]
- Zamora-Vilchis I., Williams S. E. & Johnson C. N. Environmental temperature affects prevalence of blood parasites of birds on an elevation gradient: implications for disease in a warming climate. PLoS ONE 7, e39208 (2012). [DOI] [PMC free article] [PubMed] [Google Scholar]
- Liaw A. & Wiener M. Classification and regression by randomForest. R News 2, 18–22 (2002). [Google Scholar]
- R Development Core Team: R 2004. A language and environment for statistical computing. Vienna, Austria: R Foundation for Statistical Computing. (2004).
- Lachish S., Knowles S. C. L., Alves R., Wood M. J. & Sheldon B. C. Infection dynamics of endemic malaria in a wild bird population: parasite species-dependent drivers of spatial and temporal variation in transmission rates. J. Anim. Ecol. 80, 1207–1216 (2011). [DOI] [PubMed] [Google Scholar]
- Hellgren O., Perez-Tris J. & Bensch S. A jack-of-all-trades and still a master of some: prevalence and host range in avian malaria and related blood parasites. Ecology 90, 2840–2849 (2009). [DOI] [PubMed] [Google Scholar]
- VanRiper C., VanRiper S. G., Goff M. L. & Laird M. The epizootiology and ecological significance of malaria in Hawaiian land birds. Ecol. Monog. 56, 327–344 (1986). [Google Scholar]
- Samuel M. D. et al. The dynamics, transmission, and population impacts of avian malaria in native Hawaiian birds: a modeling approach. Ecol. Appl. 21, 2960–2973 (2011). [Google Scholar]
- Waldenström J., Bensch S., Hasselquist D. & Östman Ö. A new nested polymerase chain reaction method very efficient in detecting Plasmodium and Haemoproteus infections from avian blood. J. Parasitol. 90, 191–194 (2004). [DOI] [PubMed] [Google Scholar]
- Cellier-Holzem E., Esparza-Salas R., Garnier S. & Sorci G. Effect of repeated exposure to Plasmodium relictum (lineage SGS1) on infection dynamics in domestic canaries. Int. J. Parasitol. 40, 1447–1453 (2010). [DOI] [PubMed] [Google Scholar]
- Nix H. A biogeographic analysis of Australian elapid snakes. Atlas of Elapid Snakes of Australia. Australian Government Publishing Service. (1986).
- Breiman L. Random forests. Mach. Learn. 45, 5–32 (2001). [Google Scholar]
- Cutler D. R. et al. Random forests for classification in ecology. Ecology 88, 2783–2792 (2007). [DOI] [PubMed] [Google Scholar]
- Prasad A. M., Iverson L. R. & Liaw A. Newer classification and regression tree techniques: bagging and random forests for ecological prediction. Ecosystems 9, 181–199 (2006). [Google Scholar]
- Oliver M. A. Kriging: a method of interpolation for Geographical Information Systems. Int. J. GIS 4, 313–332 (1990). [Google Scholar]