Abstract
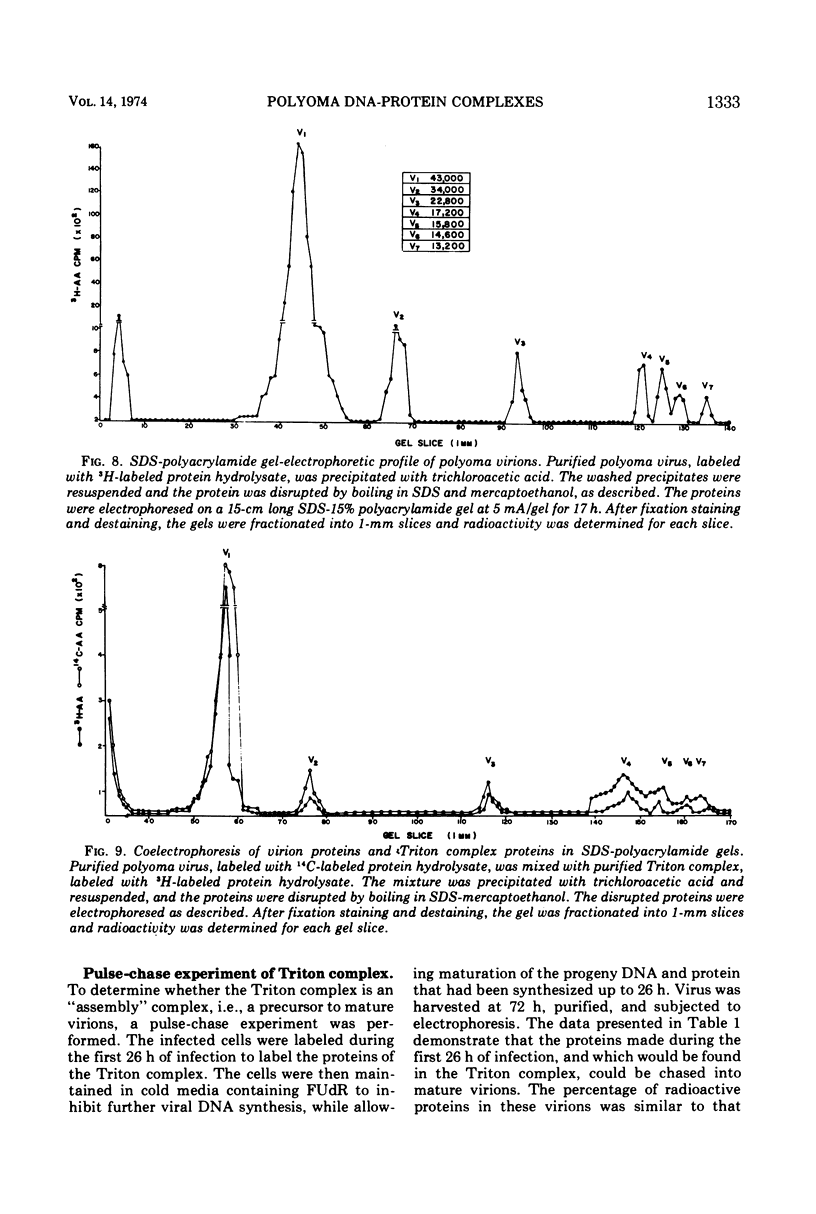
A study was undertaken to examine polyoma DNA-protein complexes. A biophysical characterization of the complexes was made, and the proteins found in such complexes were identified by sodium dodecyl sulfate-polyacrylamide gel electrophoresis. A comparison was made between a 52S nucleoprotein complex isolated from nuclei of 26-h polyoma-infected cells and a 28S virion core complex ejected out of mature virus particles. It was found that both complexes were reduced to a 20S viral DNA component plus free protein after incubation in 1 M NaCl or Sarkosyl. Treatment of the complexes with either Pronase or 0.5 M NaCl resulted in only partial removal of proteins from the viral DNA. After fixation in formaldehyde, the 52S nucleoprotein complex had a buoyant density of 1.45 g/cm3, and the virion core complex had a buoyant density of 1.59 g/cm3. Sodium dodecyl sulfate-polyacrylamide gel profiles of purified polyoma virion proteins, used as a reference marker, demonstrated three capsid proteins, V1 to V3, as well as four histones, V4 to V7, which constituted about 7% of the total virion protein. Electrophoretic analysis of the proteins comprising the 52S nucleoprotein complex revealed that the same seven proteins present in the mature virion were also found in this complex. However, the ratios of the proteins in the complex were quite different from that of the mature virion, with the four histones comprising 48% of the total complex protein. A pulse-chase experiment of the nucleoprotein complex demonstrated that the 26-h complex was chased into mature virions.
Full text
PDF









Selected References
These references are in PubMed. This may not be the complete list of references from this article.
- ALLFREY V. G., LITTAU V. C., MIRSKY A. E. On the role of of histones in regulation ribonucleic acid synthesis in the cell nucleus. Proc Natl Acad Sci U S A. 1963 Mar 15;49:414–421. doi: 10.1073/pnas.49.3.414. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Consigli R. A., Minocha H. C., Abo-Ahmed H. Multiplication of polyoma virus. II. Source of constituents for viral deoxyribonucleic acid and protein synthesis. J Bacteriol. 1966 Sep;92(3):789–791. doi: 10.1128/jb.92.3.789-791.1966. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Consigli R. A., Minocha H. C., Abo-Ahmed H. The effect of 5-fluoro-deoxyuridine on the replication of viral DNA and synthesis of polyoma capsid protein. J Gen Virol. 1968 May;2(3):437–441. doi: 10.1099/0022-1317-2-3-437. [DOI] [PubMed] [Google Scholar]
- Crawford L. V., Gesteland R. F. Synthesis of polyoma proteins in vitro. J Mol Biol. 1973 Mar 15;74(4):627–634. doi: 10.1016/0022-2836(73)90053-3. [DOI] [PubMed] [Google Scholar]
- Frearson P. M., Crawford L. V. Polyoma virus basic proteins. J Gen Virol. 1972 Feb;14(2):141–155. doi: 10.1099/0022-1317-14-2-141. [DOI] [PubMed] [Google Scholar]
- Friedmann T., David D. Structural roles of polyoma virus proteins. J Virol. 1972 Oct;10(4):776–782. doi: 10.1128/jvi.10.4.776-782.1972. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Friedmann T. Genetic economy of polyoma virus: capsid proteins are cleavage products of same viral gene. Proc Natl Acad Sci U S A. 1974 Feb;71(2):257–259. doi: 10.1073/pnas.71.2.257. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Goldstein D. A., Hall M. R., Meinke W. Properties of nucleoprotein complexes containing replicating polyoma DNA. J Virol. 1973 Oct;12(4):887–900. doi: 10.1128/jvi.12.4.887-900.1973. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Green M. H. Biosynthetic properties of a polyoma nucleoprotein complex: evidence for replication sites. J Virol. 1972 Jul;10(1):32–41. doi: 10.1128/jvi.10.1.32-41.1972. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Green M. H., Miller H. I., Hendler S. Isolation of a polyoma-nucleoprotein complex from infected mouse-cell cultures. Proc Natl Acad Sci U S A. 1971 May;68(5):1032–1036. doi: 10.1073/pnas.68.5.1032. [DOI] [PMC free article] [PubMed] [Google Scholar]
- HUANG R. C., BONNER J. Histone, a suppressor of chromosomal RNA synthesis. Proc Natl Acad Sci U S A. 1962 Jul 15;48:1216–1222. doi: 10.1073/pnas.48.7.1216. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Hirt B. Selective extraction of polyoma DNA from infected mouse cell cultures. J Mol Biol. 1967 Jun 14;26(2):365–369. doi: 10.1016/0022-2836(67)90307-5. [DOI] [PubMed] [Google Scholar]
- Huang E. S., Estes M. K., Pagano J. S. Structure and function of the polypeptides in simian virus 40. I. Existence of subviral deoxynucleoprotein complexes. J Virol. 1972 Jun;9(6):923–929. doi: 10.1128/jvi.9.6.923-929.1972. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Huang E. S., Nonoyama M., Pagano J. S. Structure and function of the polypeptides in simian virus 40. II. Transcription of subviral deoxynucleoprotein complexes in vitro. J Virol. 1972 Jun;9(6):930–937. doi: 10.1128/jvi.9.6.930-937.1972. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Khare G. P., Consigli R. A. Cytologic studies in polyoma virus-infected mouse embryo cells. Am J Vet Res. 1967 Sep;28(126):1527–1536. [PubMed] [Google Scholar]
- Lake R. S., Barban S., Salzman N. P. Resolutions and identification of the core deoxynucleoproteins of the simian virus 40. Biochem Biophys Res Commun. 1973 Sep 18;54(2):640–647. doi: 10.1016/0006-291x(73)91471-x. [DOI] [PubMed] [Google Scholar]
- Murakami W. T., Fine R., Harrington M. R., Sassan Z. B. Properties and amino acid composition of polyoma virus purified by zonal ultracentrifugation. J Mol Biol. 1968 Aug 28;36(1):153–166. doi: 10.1016/0022-2836(68)90226-x. [DOI] [PubMed] [Google Scholar]
- Nancock R. Separation by equilibrium centrifugation in CsC1 gradients of density--labelled and normal deoxyribonucleoprotein from chromatin. J Mol Biol. 1970 Mar 14;48(2):357–360. doi: 10.1016/0022-2836(70)90167-1. [DOI] [PubMed] [Google Scholar]
- Roblin R., Härle E., Dulbecco R. Polyoma virus proteins. 1. Multiple virion components. Virology. 1971 Sep;45(3):555–566. doi: 10.1016/0042-6822(71)90171-1. [DOI] [PubMed] [Google Scholar]
- Seebeck T., Weil R. Polyoma viral DNA replicated as a nucleoprotein complex in close association with the host cell chromatin. J Virol. 1974 Mar;13(3):567–576. doi: 10.1128/jvi.13.3.567-576.1974. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Shmookler R. J., Buss J., Green M. H. Properties of the polyoma virus transcription complex obtained from mouse nuclei. Virology. 1974 Jan;57(1):122–127. doi: 10.1016/0042-6822(74)90113-5. [DOI] [PubMed] [Google Scholar]
- Smith G. L., Consigli R. A. Transient inhibition of polyoma virus synthesis by Sendai virus (parainfluenza I). I. Demonstration and nature of the inhibition by inactivated virus. J Virol. 1972 Dec;10(6):1091–1097. doi: 10.1128/jvi.10.6.1091-1097.1972. [DOI] [PMC free article] [PubMed] [Google Scholar]
- WEIL R. The denaturation and the renaturation of the DNA of polyoma virus. Proc Natl Acad Sci U S A. 1963 Apr;49:480–487. doi: 10.1073/pnas.49.4.480. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Weber K., Osborn M. The reliability of molecular weight determinations by dodecyl sulfate-polyacrylamide gel electrophoresis. J Biol Chem. 1969 Aug 25;244(16):4406–4412. [PubMed] [Google Scholar]
- White M., Eason R. Nucleoprotein complexes in simian virus 40-infected cells. J Virol. 1971 Oct;8(4):363–371. doi: 10.1128/jvi.8.4.363-371.1971. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Winocour E., Robbins E. Histone synthesis in polyoma- and SV40-infected cells. Virology. 1970 Feb;40(2):307–315. doi: 10.1016/0042-6822(70)90406-x. [DOI] [PubMed] [Google Scholar]



