Abstract
Over the past several decades, invasive cervical cancer (ICC) incidence in the United States has declined dramatically. Much of this decline has been attributed to widespread use of cytology screening followed by treatment of precancerous lesions. Despite available technologies to prevent ICC and screening programs targeting high-risk women, certain populations in the United States experience disproportionately high rates of ICC (e.g., racial/ethnic minorities and rural women). Limited access to and use of screening/follow-up services underlie this disparity. The licensure of the human papillomavirus (HPV) vaccine in 2006 introduced an additional method of ICC prevention. Unfortunately, dissemination of the vaccine to age-eligible females has been lower than expected (32% have received all 3 recommended doses). Decreasing the burden of HPV infection and HPV-related diseases in the United States will require greater dissemination of the HPV vaccine to adolescents and young adults, along with successful implementation of revised ICC screening guidelines that incorporate HPV and cytology cotesting. While a future without ICC is possible, we will need a comprehensive national health care program and innovative approaches to reduce ICC burden and disparities.
Cervical Cancer Burden and Trends in the United States
Invasive cervical cancer (ICC) and its precancerous lesions remain a costly public health problem in the United States. In 2011, there were 12,710 new cases of ICC and 4,290 deaths due to the disease (1), making it the 13th most common cancer among women (2). Incidence and mortality rates have declined by more than 75% since the 1940s, a change attributed to the introduction and widespread use of ICC screening with cervical cytology [Papanicolaou (Pap)] and treatment of precancerous lesions (3). Since 1975, the age-adjusted incidence rate of ICC has decreased from 14.8 per 100,000 women to 6.6 in 2008 (4). This decline has been considered as one of the greatest cancer prevention achievements in the United States to date (3).
Disparities and the Excess Burden of Cervical Cancer
Although ICC incidence and mortality rates declined for the United States as a nation, not all populations experienced similar declines and disparities persist (5, 6). Greater than 60% of ICC cases in the United States occur in underserved populations (3), including racial/ethnic minorities, women residing in rural areas, and women living in poverty. Underserved women are less likely to undergo ICC screening and less likely to receive follow-up care, placing them at higher risk for the development of ICCs (7, 8).
Racial/ethnic disparities
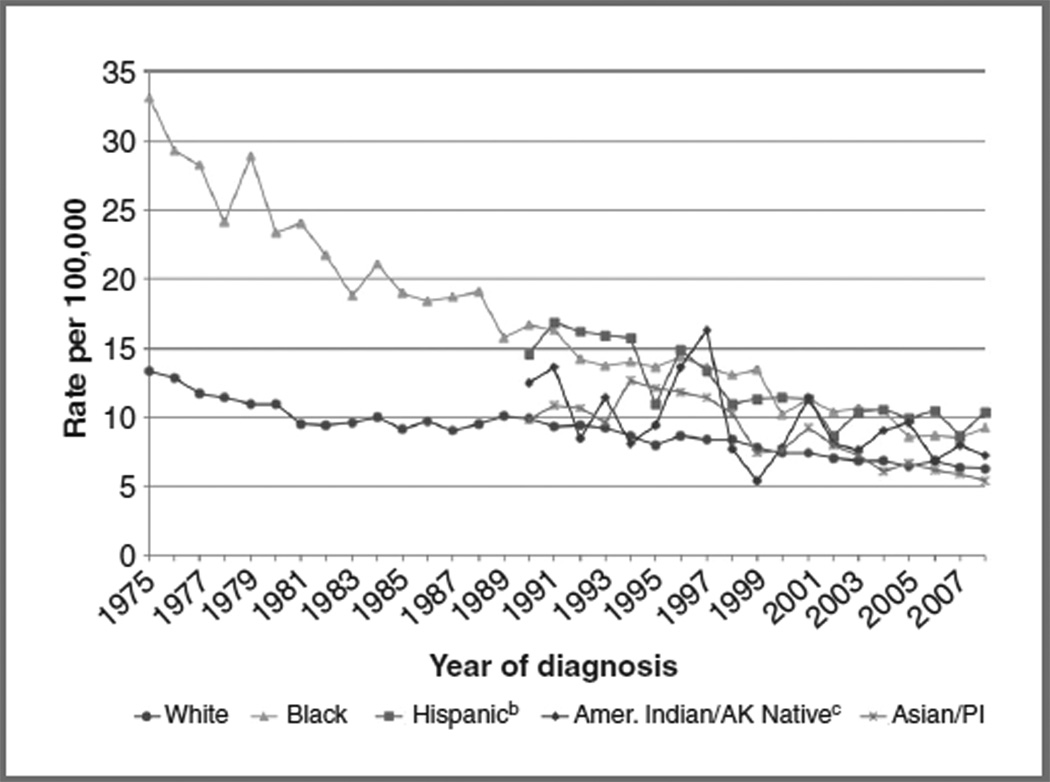
For decades, racial/ethnic minorities have experienced disproportionately higher rates of ICC than whites (Fig. 1; ref. 4). Since 1975, ICC incidence among black women has declined, yet in 2008, it remained 30% higher than that of whites (9.2 vs. 6.3 per 100,000). Although Hispanic women have experienced declining rates since 1990, the incidence of ICC (10.4 per 100,000) remained higher than that of any other racial/ethnic group. American Indian/Alaska Native women have historically suffered from higher rates of ICC than white women; however, time trends are less clear, given the small number of women comprising this population.
Figure 1.
Time trends in age-adjusted incidencea of ICC in the United States, 1975–2008, by race/ethnicity. aRates are age-adjusted to the 2000 United States standard population. Incidence data for whites and blacks are from the Surveillance Epidemiology and End Results (SEER) 9 areas (San Francisco, Connecticut, Detroit, Hawaii, Iowa, New Mexico, Seattle, Utah, and Atlanta). Incidence data for Asian/Pacific Islanders, American Indians/Alaska Natives, and Hispanics are from the SEER 13 Areas (SEER 9 Areas, San Jose-Monterey, Los Angeles, Alaska Native Registry, and Rural Georgia). bHispanic is not mutually exclusive from whites, blacks, Asian/Pacific Islanders, and American Indians/Alaska Natives. Incidence data for Hispanics are based on North American Association of Central Cancer Registries Hispanic Identification Algorithm (NHIA) and exclude cases from the Alaska Native Registry. cRates for American Indian/Alaska Native are based on the CHSDA (Contract Health Service Delivery Area) counties. Notes: SEER 9 and SEER 13 cancer incidence data are collected from population-based cancer registries covering 9.5% and 13.8% of the U.S. population, respectively. Source: SEER (4). AK, Alaskan; Amer, American; PI, Pacific Islanders.
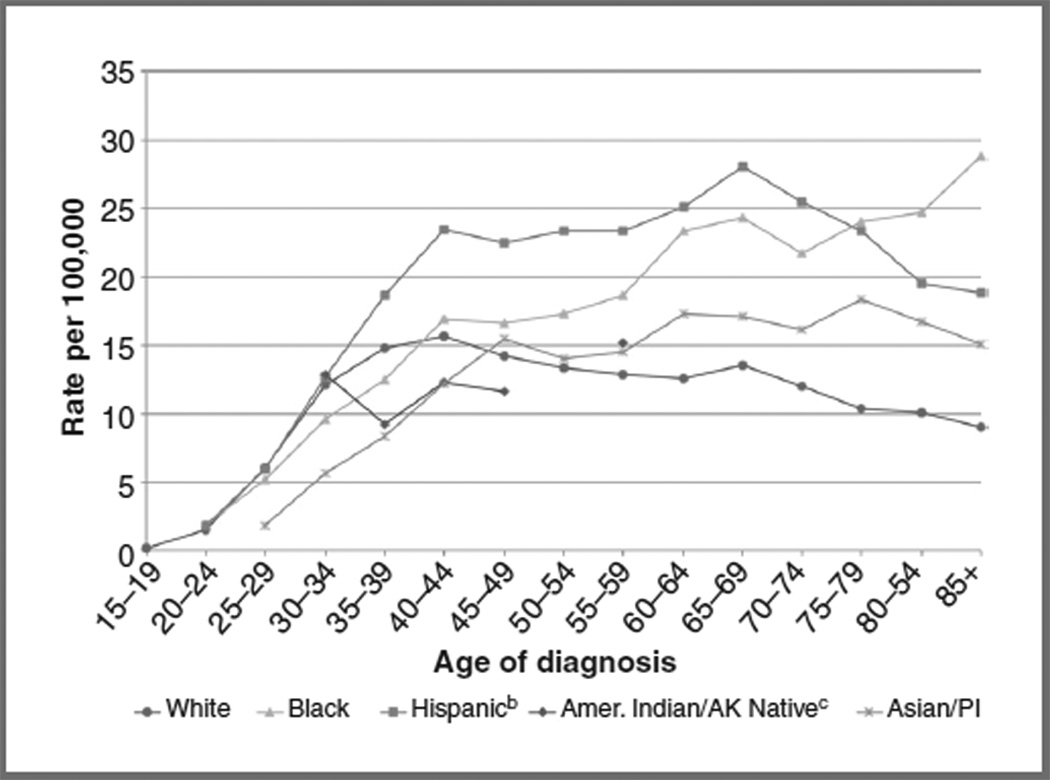
The age at which women are diagnosed with ICC differs by race/ethnicity. There is a sharp increase in incidence among black women with advancing age (Fig. 2; ref. 9). Among black women ≥85 years, the incidence of ICC was 28.9 per 100,000; this rate is 3 times that of white women. In contrast, as women of other racial/ethnic groups age, ICC incidence rates plateau or decline. It is unknown whether this age-related disparity is related to inadequate screening, or differences in socioeconomic factors or comorbid conditions (e.g., diabetes or hypertension) that may influence screening participation and follow-up.
Figure 2.
Age-specific incidencea of ICC in the United States, 2000 to 2008, by race/ethnicity. aIncidence data are from the SEER 17 areas (San Francisco, Connecticut, Detroit, Hawaii, Iowa, New Mexico, Seattle, Utah, Atlanta, San Jose-Monterey, Los Angeles, Alaska Native Registry, Rural Georgia, California excluding San Francisco/San Jose-Monterey/ Los Angeles, Kentucky, Louisiana, and New Jersey). bHispanic is not mutually exclusive from whites, blacks, Asian/Pacific Islanders, and American Indians/Alaska Natives. Incidence data for Hispanics are based on NHIA and exclude cases from the Alaska Native Registry. cRates for American Indian/Alaska Native are based on the CHSDA(Contract Health Service Delivery Area) counties. Notes: SEER 17 cancer incidence data are collected from population-based cancer registries covering 26.2% of the U.S. population. Source: SEER (9). AK, Alaskan; Amer, American; PI, Pacific Islanders.
Geographic disparities
Regional variation in ICC incidence and mortality exists across the United States, with disproportionately high rates concentrated along the United States–Mexico border, in the deep South, and in the Appalachian region (6). Underserved white women in Appalachia experience higher rates of ICC than white women outside Appalachia, illustrating that disparities extend beyond that of race/ethnicity. For example, in Ohio, ICC incidence and mortality are higher among white women in Appalachian counties (9.6 and 3.1 per 100,000, respectively) than among white women in non-Appalachian counties (7.7 and 2.3, respectively; ref. 10).
Socioeconomic disparities
In the United States, cancer registries lack information on socioeconomic status (SES), making it difficult to report ICC incidence by SES. While epidemiologic research on SES and its association with ICC has been complicated by issues of race/ethnicity, studies have shown SES to be a strong predictor of ICC screening, diagnosis, and treatment, even after controlling for race/ethnicity (11). Further studies are needed to examine how SES impacts ICC rates and to elucidate the role of race/ethnicity.
Cervical Cancer Screening
ICC and its precancerous lesions are effectively detected by routine Pap tests. The excess burden of ICC among high-risk populations may be partly explained by an underutilization of screening services. High-risk women may not have adequate access to screening, may not receive physician recommendation for screening (12), or may choose not to participate in these services. Poverty, lack of health care insurance, cultural beliefs and perceptions, fear, embarrassment, lack of knowledge, language differences, and immigration status are among many barriers preventing women from being screened (13, 14).
Cytology screening coverage
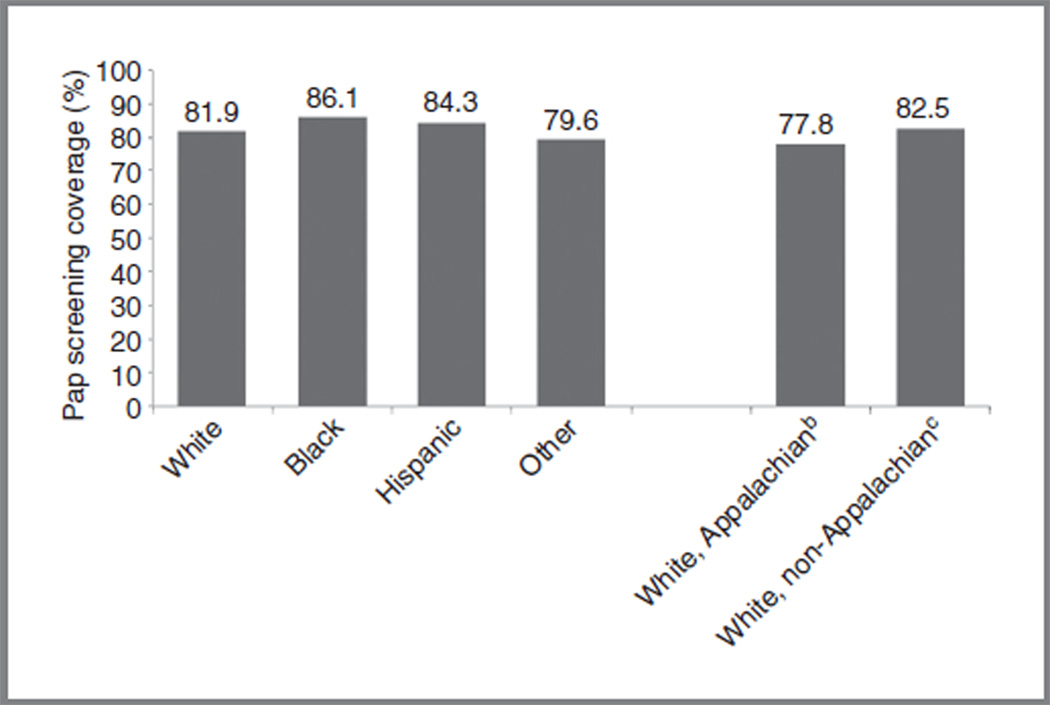
Rates of cytology screening are comparable across racial/ethnic groups, albeit slightly higher among minorities (Fig. 3; ref. 15). Paradoxically, although screening rates among black (86.1%) and Hispanic (84.3%) women are slightly higher than among white women (81.9%), ICC incidence and mortality rates remain higher among black and Hispanic women. For white women, screening is lower among those residing in Appalachian Ohio (77.8%) than among those in non-Appalachian Ohio (82.5%), showing that poor, underserved white women underutilize cervical screening.
Figure 3.
Estimates of recent, self-reported Pap screening among adult womena in the United States, 2010. aWomen 18 years of age and older with an intact cervix who have had a Pap test within the past 3 years. bEstimates are from Ohio counties considered part of the Appalachian region. cEstimates are from Ohio counties not considered part of the Appalachian region. Notes: BRFSS data are collected by each state via landline telephone survey. The median response rate for the 2010 BRFSS (all states combined) was 54.6%. BRFSS prevalence estimates are overestimated when compared with National Health Interview Survey (NHIS) estimates. Sources: BRFSS, United States, 2010 (15).
Availability, utilization, and timeliness of treatment services
Timely diagnostic follow-up of abnormal results and the availability and utilization of treatment services are needed to effectively reduce ICCs (7, 8). Uninsured women and racial/ethnic minorities experience the longest delays in cervical disease evaluation, often due to a lack of health care insurance and site of care (16). Federal and state funding for health-related services among the underserved often exclude high-risk populations such as recent or undocumented immigrants, preventing access to follow- up treatment (3, 17, 18). Among Appalachian women in Kentucky, logistical issues are the most common barriers to ICC follow-up and treatment, including lack of transportation, scheduling appointments, and uncertainty about medical procedures (3). Similar difficulties have been reported by Latina women, including cost, inability to obtain childcare, and fear of the procedure/outcome (3, 19). Physicians have cited reduced funding for followup colposcopy and treatment as key issues (17).
Policy initiatives to improve cervical screening coverage
Federally and locally funded ICC prevention programs have been implemented to promote screening among high-risk women. The Breast and Cervical Cancer Mortality Prevention Act (1990) established the only nationwide screening program, the National Breast and Cervical Cancer Early Detection Program (NBCCEDP), which provides access to screening and diagnostic services for all low-income, uninsured, and underinsured women (20). However, only a small proportion of financially eligible women receive services due to severe funding limitations. Between 2004 and 2006, only 8.7% of eligible women ages 18 to 64 years were screened for ICC (21). This percentage varies by age, state, and race/ethnicity, from 6.5% in blacks to 36.1% in American Indians/Alaska Natives (21). Some women diagnosed with ICC through the NBCCEDP have had access to treatment since 2000 through the Breast and Cervical Cancer Prevention and Treatment Act. Because the NBCCEDP specifically targets underserved women, the program is in a unique position to address issues of access to screening and treatment services. However, decreasing the ICC burden will require increased national funding to support the NBCCEDP, not only to provide Pap tests to all eligible women but also to assure follow-up of abnormal results.
Human Papillomavirus Vaccination
Currently, there are 2 ICC prevention methods: cervical screening followed by treatment of precancerous lesions and human papillomavirus (HPV) vaccination. In 2006, the first HPV vaccine (quadrivalent, Gardasil) was licensed in the United States for use among young females (9–26 years), and in 2009, another HPV vaccine (bivalent, Cervarix) was licensed, also for use among young females (10–25 years).
HPV vaccination coverage among females
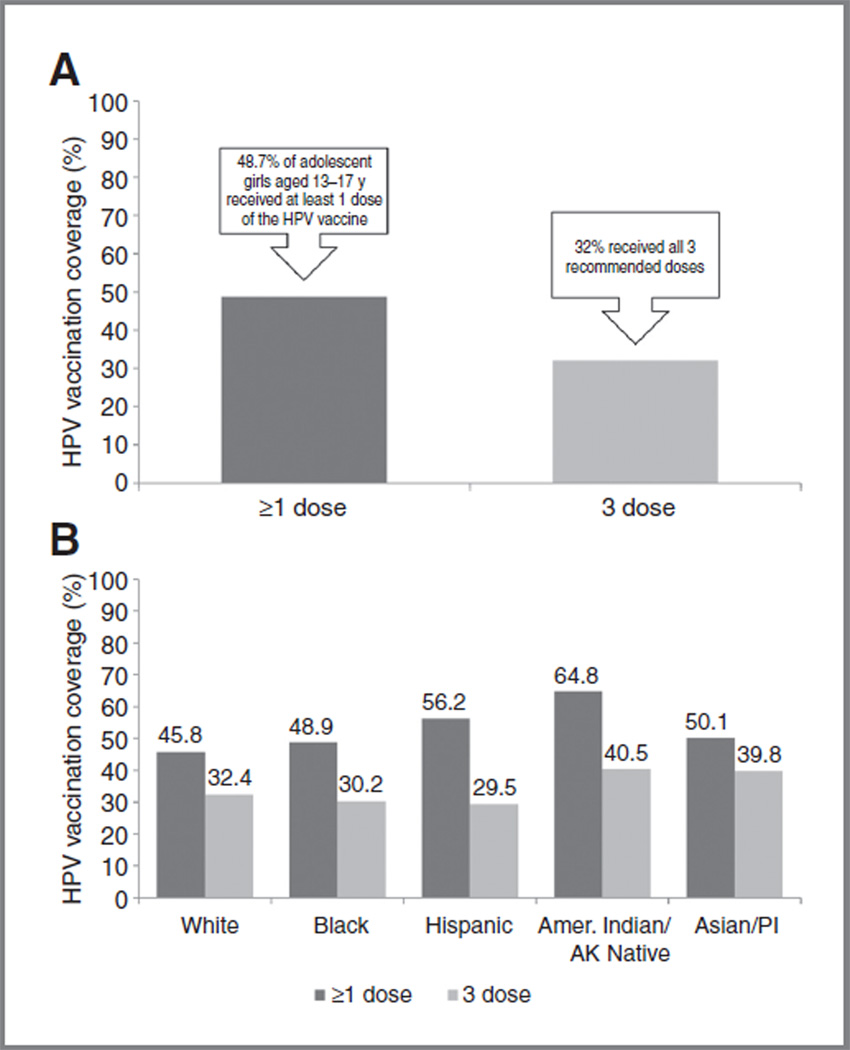
Since 2006, HPV vaccination coverage in the United States has increased among adolescents, nevertheless coverage remains low. Findings from the 2010 National Immunization Survey-Teen (NIS-Teen) indicate that slightly less than half of adolescent girls (13–17 years) received at least 1 dose of the HPV vaccine (quadrivalent or bivalent) and only 32% received all 3 recommended doses (Fig. 4A; ref. 22). Vaccination coverage varied by race/ethnicity (Fig. 4B; ref. 22); initiation (≥1 dose) among whites (45.8%) was significantly lower than among Hispanics (56.2%) or American Indians/Alaska Natives (64.8%).
Figure 4.
A, estimates of HPV vaccinationa coverage among adolescent girls (13–17 years) in the United States, 2010. B, estimates of HPV vaccinationa coverage among adolescent girls (13–17 years) in the United States, 2010, by race/ethnicity. aQuadrivalent or bivalent vaccine. Source: NIS-Teen, United States, 2010 (22). AK, Alaskan; Amer, American; PI, Pacific Islanders.
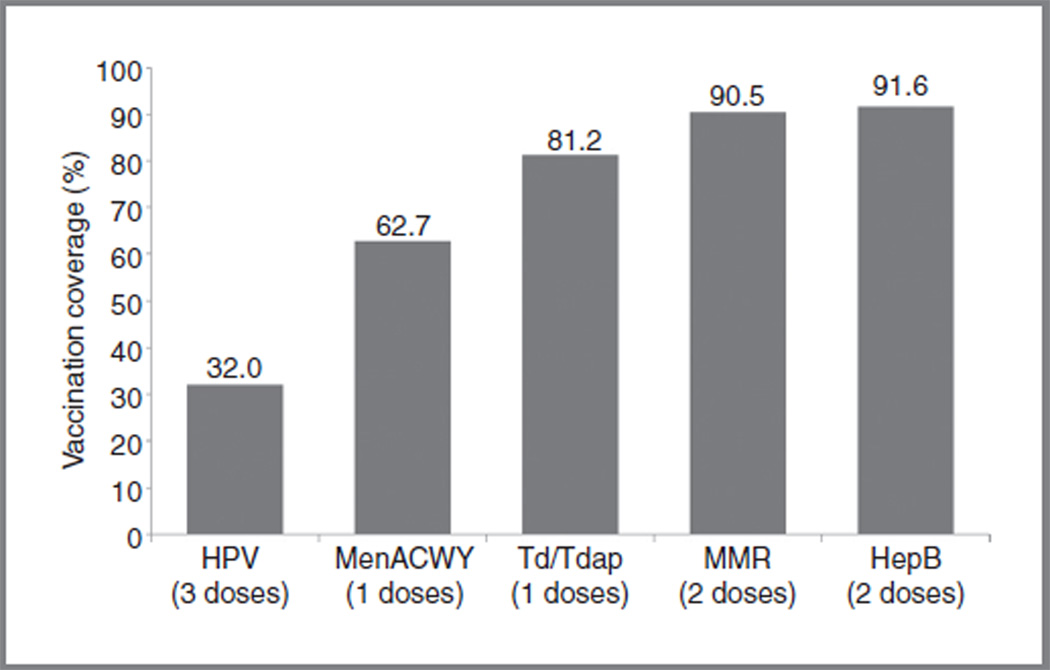
The HPV vaccine lags behind other adolescent vaccines. In 2010, 91.6% of adolescents received 3 recommended doses of Hepatitis B vaccine, whereas only 32% of adolescent females received 3 recommended doses of HPV vaccine (Fig. 5; ref. 22). Efforts are needed to improve HPV vaccine initiation and completion, especially among racial/ethnic minorities at higher risk of ICC.
Figure 5.
Estimates of childhood vaccination coverage among adolescents (13–17 years) in the United States, 2010. HepB, hepatitis B vaccine; MenACWY, meningococcal vaccine; MMR, measles, mumps, rubella vaccine; Td/Tdap, tetanus toxoid-diphtheria vaccine (Td) or tetanus toxoid, diphtheria, pertussis vaccine (Tdap). Notes: HPV vaccine (quadrivalent or bivalent) reported among females only. Source: NIS-Teen, United States, 2010 (22).
Barriers to HPV vaccination
Numerous barriers impeding HPV vaccine uptake have been identified. Among adolescents and young adults, common individual barriers include cost, safety, and the perception that vaccination is unnecessary if not sexually active (23, 24). Parental knowledge, attitudes, and beliefs about vaccine also influence uptake (25, 26). Parents opposing the vaccine reported that their children were too young, the vaccine was not needed, children were not sexually active (27–29), and that receipt of vaccine might increase promiscuity (17, 30, 31). Moreover, the media and public officials have made unsubstantiated claims about vaccine safety, reinforcing negative opinions of the HPV vaccine (32).
Institutional/structural factors that influence vaccine initiation include cultural/linguistic differences, lack of health care access, and lack of physician recommendation. Among minority populations, language difficulties and navigating a complex health care system pose considerable challenges to vaccination. Poor access to health care services also negatively impacts vaccine uptake. A study in Kentucky found a 7-fold decrease in rural women, versus urban women, returning for a follow-up vaccine dose, despite provision of the vaccine at no cost. These findings suggest that distance to the clinic might influence completion rates (33). Insurance status, lack of awareness among those who are eligible for free/discounted vaccines, and lack of a steady primary care source also impact vaccine uptake (23, 34–36).
Findings from the 2010 NIS-Teen survey indicate that physician recommendation of HPV vaccine is a key factor in vaccine initiation (27). Many primary care physicians have not been proactive in promoting the vaccine particularly to young adolescents, the target age for vaccination. In a national study of family physicians, pediatricians, and obstetricians/gynecologists (37), all were least likely to recommend the HPV vaccine to 11- to 12-year-olds (34.6%) compared with 13- to 17-year-olds (52.7%) and 18- to 26-year-olds (50.2%). Other physician-related factors include missed opportunities for catch-up vaccination during routine health visits, vaccine cost and reimbursement, and difficulty discussing sexuality with adolescents (17, 38).
Policy initiatives to promote HPV vaccination
One year after licensure, the Advisory Committee on Immunization Practices (ACIP) recommended routine HPV vaccination for young females (39). With recommendations from ACIP, physicians are more likely to promote the HPV vaccine to patients, and costs are covered by many health care insurance plans. In addition, eligible children can receive the HPV vaccine at no cost through the federally funded Vaccines for Children (VFC) program that provides free ACIP-recommended vaccines to children (≤18 years) unable to pay (e.g., Medicaid eligible, uninsured/underinsured, American Indian/Alaska Native; ref. 40). Therefore, in theory, most young women ages 9 to 18 can receive the HPV vaccine at little or no cost. Still, there are populations who lack health care insurance but are ineligible for VFC and cannot afford the cost of the vaccine or clinic administration fee. Cost may be a more important barrier to vaccination among those already at increased risk of HPV infection and ICCs.
An additional strategy to reduce ICC among females has been HPV vaccination of males. HPV infection in men contributes to HPV infection and subsequent development of ICCs in women (41). Vaccinating males reduces HPV transmission to females and strengthens herd immunity. Model-based studies have shown that a gender-neutral HPV vaccination strategy would result in maximal disease reduction (42, 43). Given the low vaccine coverage among U.S. females, HPV vaccination has recently been recommended for routine use among males. In 2009, the quadrivalent HPV vaccine was approved for use among young males (9–26 years), and in 2011, ACIP officially recommended routine HPV vaccination of both girls (bivalent or quadrivalent) and boys (quadrivalent) ages 11 to 12 years. With ACIP recommendations in place and VFC coverage ensured for both genders, physician recommendation and health care insurance coverage of the HPV vaccine should improve over time, expanding accessibility to all young women and men in the United States.
Reducing the Burden of Cervical Cancer in the United States
A more thorough understanding of the etiologic role of HPV has provided the foundation for rational, targeted, and cost-effective approaches to ICC prevention. HPV vaccination and HPV-based screening hold promise for eliminating disease and disease-related disparities. Universal vaccination against HPV has the potential to reduce the incidence of ICC and its precancerous lesions (grade II/III) by 91% (42). However, continued efforts are needed to advocate for HPV vaccination among adolescents and young adults, especially among racial/ethnic minorities at increased risk of ICCs. New approaches should be considered to broadly disseminate HPV vaccines. History shows that incorporating vaccination into school entry requirements ensures high vaccination coverage in the United States. Campaign strategies should also encourage increased knowledge and awareness of HPV vaccination and improve physician recommendations. With every health care visit is the opportunity to vaccinate.
Increased knowledge of HPV natural history and carcinogenesis has led to improved ICC screening methods, including HPV DNA testing. In March 2012, several U.S. professional organizations released updated, evidence-based cervical screening recommendations that incorporate routine HPV testing (44, 45). For women ages 30 to 65 years, HPV cotesting (cytology + HPV test) should be conducted every 5 years, or cytology alone every 3 years. Additional recommendations include: (i) women ages 21 to 29 years should undergo cytology every 3 years; (ii) certain low-risk groups should no longer be screened (women under age 21, certain women over age 65, and women who have undergone complete hysterectomy); and (iii) women vaccinated against HPV should continue to follow screening guidelines. Through the extension of screening intervals and reduced screening of low-risk women (e.g., adolescents and older women without history of ICC), fewer instances of overscreening and overtreatment should occur (44).
While universal health care does not exist in the United States, health care in general is undergoing rapid change. Under the Affordable Care Act (ACA, 2010), women will be able to receive recommended preventive services, including cervical screening and adolescent HPV vaccination, without cost sharing (co-payment, coinsurance, or deductible; ref. 46), a great stride in improving the affordability and accessibility of ICC prevention for women. Nevertheless, many questions will need to be addressed as we implement the ACA, including: What infrastructure will be required for successful implementation? How do we incorporate revised ICC screening guidelines (e.g., extended screening intervals and routine HPV testing) into current screening programs? Should HPV testing be offered in an alternative sequence (e.g., HPV testing as a primary screen with cytologic triage of HPV+ women; ref. 47–51)? At which age could screening initiation be further delayed? How do we minimize overscreening and over-treating HPV+ women? How should screening guidelines be modified as vaccination rates improve?
In summary, ICCs can be eliminated in the United States. To achieve this goal, the United States will need to adopt a comprehensive national health care program that underscores accessible and equitable health care and delivers compassionate care to all. With the implementation of the ACA, this type of health care is possible. Interest in women’s health has been renewed, and the United States is poised to make even greater progress in cervical cancer prevention and early detection. Through the development of a national ICC control strategy, we can decrease the number of women infected with HPV, increase access to care and improve cancer-related outcomes, and finally, decrease health care disparities. A future without ICC is possible, although we must be innovative and vigilant in our approach to reduce ICC burden and disparities.
Acknowledgments
Grant Support
This work was supported in part by a cancer prevention fellowship for C.M. Pierce Campbell supported by the National Cancer Institute grant R25T CA147832.
Footnotes
This work was presented, in part, at the Opening Ceremony of the 27th International Papillomavirus Conference and Clinical Workshop in Berlin, Germany on September 18, 2011.
Disclosure of Potential Conflicts of Interest
E.D. Paskett received commercial research support from Merck. A.R. Giuliano received a commercial research grant from Merck and commercial research support from GSK. A.R. Giuliano received an honoraria from Speaker’s bureau and is a consultant/advisory board member in Merck. No potential conflicts of interest were disclosed by the other authors.
Authors' Contributions
Conception and design: C.M. Pierce Campbell, L.J. Menezes, A.R. Giuliano
Development of methodology: C.M. Pierce Campbell, L.J. Menezes, A.R. Giuliano
Acquisition of data (provided animals, acquired and managed patients, provided facilities, etc.): E.D. Paskett
Analysis and interpretation of data (e.g., statistical analysis, biostatistics, computational analysis): C.M. Pierce Campbell, L.J. Menezes
Writing, review, and/or revision of the manuscript: C.M. Pierce Campbell, L.J. Menezes, E.D. Paskett, A.R. Giuliano
Administrative, technical, or material support (i.e., reporting or organizing data, constructing databases): L.J. Menezes
References
- 1.American Cancer Society. Cancer facts & figures 2011. Atlanta, GA: American Cancer Society; 2011. [Google Scholar]
- 2.Kohler BA, Ward E, McCarthy BJ, Schymura MJ, Ries LA, Eheman C, et al. Annual report to the nation on the status of cancer, 1975–2007, featuring tumors of the brain and other nervous system. J Natl Cancer Inst. 2011;103:714–736. doi: 10.1093/jnci/djr077. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 3.Scarinci IC, Garcia FA, Kobetz E, Partridge EE, Brandt HM, Bell MC, et al. Cervical cancer prevention: new tools and old barriers. Cancer. 2010;116:2531–2542. doi: 10.1002/cncr.25065. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 4.Howlader N, Noone A, Krapcho M, Neyman N, Aminou R, Waldron W, et al. SEER cancer statistics review, 1975–2008. Bethesda, MD: National Cancer Institute; 2011. [cited 2011 Nov 29]. Available from: http://seer.cancer.gov/csr/1975_2008. [Google Scholar]
- 5.Patel NR, Rollison DE, Barnholtz-Sloan J, Mackinnon J, Green L, Giuliano AR. Racial and ethnic disparities in the incidence of invasive cervical cancer in Florida. Cancer. 2009;115:3991–4000. doi: 10.1002/cncr.24427. [DOI] [PubMed] [Google Scholar]
- 6.Watson M, Saraiya M, Benard V, Coughlin SS, Flowers L, Cokkinides V, et al. Burden of cervical cancer in the United States, 1998–2003. Cancer. 2008;113:2855–2864. doi: 10.1002/cncr.23756. [DOI] [PubMed] [Google Scholar]
- 7.Leyden WA, Manos MM, Geiger AM, Weinmann S, Mouchawar J, Bischoff K, et al. Cervical cancer in women with comprehensive health care access: attributable factors in the screening process. J Natl Cancer Inst. 2005;97:675–683. doi: 10.1093/jnci/dji115. [DOI] [PubMed] [Google Scholar]
- 8.Spence AR, Goggin P, Franco EL. Process of care failures in invasive cervical cancer: systematic review and meta-analysis. Prev Med. 2007;45:93–106. doi: 10.1016/j.ypmed.2007.06.007. [DOI] [PubMed] [Google Scholar]
- 9.Surveillance Research Program, National Cancer Institute. Fast Stats: An interactive tool for access to SEER cancer statistics [homepage on the Internet] Bethesda, MD: National Cancer Institute; [cited 2012 Jan 25]. Available from: http://seer.cancer.gov/faststats/ [Google Scholar]
- 10.Ohio Department of Health. Ohio Cancer Incidence Surveillance System, 2001–2005 [Internet] Columbus, OH: Ohio Department of Health; 2010. [cited 2011 Sep 12]. Available from: http://www.odh.ohio.gov/healthstats/ocisshs/ci_surv1.aspx. [Google Scholar]
- 11.Newmann SJ, Garner EO. Social inequities along the cervical cancer continuum: a structured review. Cancer Causes Control. 2005;16:63–70. doi: 10.1007/s10552-004-1290-y. [DOI] [PubMed] [Google Scholar]
- 12.Nuno T, Castle PE, Harris R, Estrada A, Garcia F. Breast and cervical cancer screening utilization among Hispanic women living near the United States-Mexico border. J Womens Health (Larchmt) 2011;20:685–693. doi: 10.1089/jwh.2010.2205. [DOI] [PubMed] [Google Scholar]
- 13.Akers AY, Newmann SJ, Smith JS. Factors underlying disparities in cervical cancer incidence, screening, and treatment in the United States. Curr Probl Cancer. 2007;31:157–181. doi: 10.1016/j.currproblcancer.2007.01.001. [DOI] [PubMed] [Google Scholar]
- 14.Johnson CE, Mues KE, Mayne SL, Kiblawi AN. Cervical cancer screening among immigrants and ethnic minorities: a systematic review using the health belief model. J Low Genit Tract Dis. 2008;12:232–241. doi: 10.1097/LGT.0b013e31815d8d88. [DOI] [PubMed] [Google Scholar]
- 15.Centers for Disease Control and Prevention. Behavioral risk factor surveillance system survey data [homepage on the Internet] Atlanta, GA: Centers for Disease Control and Prevention; 2010. [cited 2011 Sep 12]. Available from: http://www.cdc.gov/brfss/ [Google Scholar]
- 16.Battaglia TA, Santana MC, Bak S, Gokhale M, Lash TL, Ash AS, et al. Predictors of timely follow-up after abnormal cancer screening among women seeking care at urban community health centers. Cancer. 2010;116:913–921. doi: 10.1002/cncr.24851. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 17.Daley MF, Crane LA, Markowitz LE, Black SR, Beaty BL, Barrow J, et al. Human papillomavirus vaccination practices: a survey of US physicians 18 months after licensure. Pediatrics. 2010;126:425–433. doi: 10.1542/peds.2009-3500. [DOI] [PubMed] [Google Scholar]
- 18.McPhee SJ, Bird JA, Davis T, Ha NT, Jenkins CN, Le B. Barriers to breast and cervical cancer screening among Vietnamese-American women. Am J Prev Med. 1997;13:205–213. [PubMed] [Google Scholar]
- 19.Percac-Lima S, Aldrich LS, Gamba GB, Bearse AM, Atlas SJ. Barriers to follow-up of an abnormal Pap smear in Latina women referred for colposcopy. J Gen Intern Med. 2010;25:1198–1204. doi: 10.1007/s11606-010-1450-6. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 20.Centers for Disease Control and Prevention. National Breast and Cervical Cancer Early Detection Program [homepage on the Internet] Atlanta, GA: Centers for Disease Control and Prevention; 2011. [cited 2011 Nov 29]. Available from: http://www.cdc.gov/cancer/nbccedp/ [Google Scholar]
- 21.Tangka FK, O'Hara B, Gardner JG, Turner J, Royalty J, Shaw K, et al. Meeting the cervical cancer screening needs of underserved women: the National Breast and Cervical Cancer Early Detection Program, 2004–2006. Cancer Causes Control. 2010;21:1081–1090. doi: 10.1007/s10552-010-9536-3. [DOI] [PubMed] [Google Scholar]
- 22.Centers for Disease Control and Prevention. National and state vaccination coverage among adolescents aged 13 through 17 years–United States, 2010. MMWRMorb Mortal Wkly Rep. 2011;60:1117–1123. [PubMed] [Google Scholar]
- 23.Anhang Price R, Koshiol J, Kobrin S, Tiro JA. Knowledge and intention to participate in cervical cancer screening after the human papillomavirus vaccine. Vaccine. 2011;29:4238–4243. doi: 10.1016/j.vaccine.2011.03.076. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 24.Caskey R, Lindau ST, Alexander GC. Knowledge and early adoption of the HPV vaccine among girls and young women: results of a national survey. J Adolesc Health. 2009;45:453–462. doi: 10.1016/j.jadohealth.2009.04.021. [DOI] [PubMed] [Google Scholar]
- 25.Cates JR, Brewer NT, Fazekas KI, Mitchell CE, Smith JS. Racial differences in HPV knowledge, HPV vaccine acceptability, and related beliefs among rural, southern women. J Rural Health. 2009;25:93–97. doi: 10.1111/j.1748-0361.2009.00204.x. [DOI] [PubMed] [Google Scholar]
- 26.Hughes J, Cates JR, Liddon N, Smith JS, Gottlieb SL, Brewer NT. Disparities in how parents are learning about the human papillomavirus vaccine. Cancer Epidemiol Biomarkers Prev. 2009;18:363–372. doi: 10.1158/1055-9965.EPI-08-0418. [DOI] [PubMed] [Google Scholar]
- 27.Dorell CG, Yankey D, Santibanez TA, Markowitz LE. Human papillomavirus vaccination series initiation and completion, 2008–2009. Pediatrics. 2011;128:830–839. doi: 10.1542/peds.2011-0950. [DOI] [PubMed] [Google Scholar]
- 28.Head KJ, Cohen EL. Young women's perspectives on cervical cancer prevention in Appalachian Kentucky. Qual Health Res. 2011;22:476–487. doi: 10.1177/1049732311425053. [DOI] [PubMed] [Google Scholar]
- 29.Stokley S, Cohn A, Dorell C, Hariri S, Yankey D, Messonnier N, et al. Adolescent vaccination-coverage levels in the United States, 2006– 2009. Pediatrics. 2011;128:1078–1086. doi: 10.1542/peds.2011-1048. [DOI] [PubMed] [Google Scholar]
- 30.Allen JD, Othus MK, Shelton RC, Li Y, Norman N, Tom L, et al. Parental decision making about the HPV vaccine. Cancer Epidemiol Biomarkers Prev. 2010;19:2187–2198. doi: 10.1158/1055-9965.EPI-10-0217. [DOI] [PubMed] [Google Scholar]
- 31.Katz ML, Reiter PL, Heaner S, Ruffin MT, Post DM, Paskett ED. Acceptance of the HPV vaccine among women, parents, community leaders, and healthcare providers in Ohio Appalachia. Vaccine. 2009;27:3945–3952. doi: 10.1016/j.vaccine.2009.04.040. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 32.Abdelmutti N, Hoffman-Goetz L. Risk messages about HPV, cervical cancer, and the HPV vaccine Gardasil: a content analysis of Canadian and U.S. national newspaper articles. Women Health. 2009;49:422–440. doi: 10.1080/03630240903238776. [DOI] [PubMed] [Google Scholar]
- 33.Crosby RA, Casey BR, Vanderpool R, Collins T, Moore GR. Uptake of free HPV vaccination among young women: a comparison of rural versus urban rates. J Rural Health. 2011;27:380–384. doi: 10.1111/j.1748-0361.2010.00354.x. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 34.Conroy K, Rosenthal SL, Zimet GD, Jin Y, Bernstein DI, Glynn S, et al. Human papillomavirus vaccine uptake, predictors of vaccination, and self-reported barriers to vaccination. J Womens Health. 2009;18:1679–1686. doi: 10.1089/jwh.2008.1329. [DOI] [PubMed] [Google Scholar]
- 35.Taylor LD, Hariri S, Sternberg M, Dunne EF, Markowitz LE. Human papillomavirus vaccine coverage in the United States, National Health and Nutrition Examination Survey, 2007–2008. Prev Med. 2011;52:398–400. doi: 10.1016/j.ypmed.2010.11.006. [DOI] [PubMed] [Google Scholar]
- 36.Wong CA, Berkowitz Z, Dorell CG, Price RA, Lee J, Saraiya M. Human papillomavirus vaccine uptake among 9- to 17-year-old girls: National Health Interview Survey, 2008. Cancer. 2011;117:5612–5620. doi: 10.1002/cncr.26246. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 37.Vadaparampil ST, Kahn JA, Salmon D, Lee JH, Quinn GP, Roetzheim R, et al. Missed clinical opportunities: provider recommendations for HPV vaccination for 11–12 year old girls are limited. Vaccine. 2011;29:8634–8641. doi: 10.1016/j.vaccine.2011.09.006. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 38.Kahn JA, Rosenthal SL, Tissot AM, Bernstein DI, Wetzel C, Zimet GD. Factors influencing pediatricians' intention to recommend human papillomavirus vaccines. Ambul Pediatr. 2007;7:367–373. doi: 10.1016/j.ambp.2007.05.010. [DOI] [PubMed] [Google Scholar]
- 39.Centers for Disease Control and Prevention. Advisory Committee on Immunization Practices [homepage on the Internet] Atlanta, GA: Centers for Disease Control and Prevention; 2012. [cited 2012 Mar 27]. Available from: http://www.cdc.gov/vaccines/acip/index.html. [Google Scholar]
- 40.Centers for Disease Control and Prevention. Vaccines for Children Program [homepage on the Internet] Atlanta, GA: Centers for Disease Control and Prevention; 2011. [cited 2012 Mar 27]. Available from: http://www.cdc.gov/vaccines/programs/vfc/index.html. [Google Scholar]
- 41.Giuliano AR. Human papillomavirus vaccination in males. Gynecol Oncol. 2007;107:S24–S26. doi: 10.1016/j.ygyno.2007.07.075. [DOI] [PubMed] [Google Scholar]
- 42.Elbasha EH, Dasbach EJ, Insinga RP. Model for assessing human papillomavirus vaccination strategies. Emerg Infect Dis. 2007;13:28–41. doi: 10.3201/eid1301.060438. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 43.Giuliano AR, Salmon D. The case for a gender-neutral (universal) human papillomavirus vaccination policy in the United States: Point. Cancer Epidemiol Biomarkers Prev. 2008;17:805–808. doi: 10.1158/1055-9965.EPI-07-0741. [DOI] [PubMed] [Google Scholar]
- 44.Moyer VA. Screening for cervical cancer: U.S. Preventive Services Task Force recommendation statement. Ann Intern Med. 2012 Mar 14; doi: 10.7326/0003-4819-156-12-201206190-00424. [Epub ahead of print] [DOI] [PubMed] [Google Scholar]
- 45.Saslow D, Solomon D, Lawson HW, Killackey M, Kulasingam SL, Cain J, et al. American Cancer Society, American Society for Colposcopy and Cervical Pathology, and American Society for Clinical Pathology screening guidelines for the prevention and early detection of cervical cancer. CA Cancer J Clin. 2012;137:516–542. doi: 10.1309/AJCPTGD94EVRSJCG. [DOI] [PubMed] [Google Scholar]
- 46.Affordable Care Act rules on expanding access to preventive services for women [Internet] Washington, DC: U.S. Department of Health & Human Services; 2012. [cited 2012 Feb 14]. Available from: http://www.healthcare.gov/news/factsheets/2011/08/womensprevention08012011a.html. [Google Scholar]
- 47.Cuzick J, Arbyn M, Sankaranarayanan R, Tsu V, Ronco G, Mayrand MH, et al. Overview of human papillomavirus-based and other novel options for cervical cancer screening in developed and developing countries. Vaccine. 2008;26(Suppl 10):K29–K41. doi: 10.1016/j.vaccine.2008.06.019. [DOI] [PubMed] [Google Scholar]
- 48.Cuzick J, Clavel C, Petry KU, Meijer CJ, Hoyer H, Ratnam S, et al. Overview of the European and North American studies on HPV testing in primary cervical cancer screening. Int J Cancer. 2006;119:1095–1101. doi: 10.1002/ijc.21955. [DOI] [PubMed] [Google Scholar]
- 49.Kitchener HC, Gilham C, Sargent A, Bailey A, Albrow R, Roberts C, et al. A comparison of HPV DNA testing and liquid based cytology over three rounds of primary cervical screening: extended follow up in the ARTISTIC trial. Eur J Cancer. 2011;47:864–871. doi: 10.1016/j.ejca.2011.01.008. [DOI] [PubMed] [Google Scholar]
- 50.Leinonen M, Nieminen P, Kotaniemi-Talonen L, Malila N, Tarkkanen J, Laurila P, et al. Age-specific evaluation of primary human papillomavirus screening vs conventional cytology in a randomized setting. J Natl Cancer Inst. 2009;101:1612–1623. doi: 10.1093/jnci/djp367. [DOI] [PubMed] [Google Scholar]
- 51.Schiffman M, Wentzensen N, Wacholder S, Kinney W, Gage JC, Castle PE. Human papillomavirus testing in the prevention of cervical cancer. J Natl Cancer Inst. 2011;103:368–383. doi: 10.1093/jnci/djq562. [DOI] [PMC free article] [PubMed] [Google Scholar]