Abstract
Itch-specific neurons have been sought for decades. The existence of such neurons is in doubt recently due to the observation that itch-mediating neurons also respond to painful stimuli. Here, we genetically labeled and manipulated MrgprA3+ neurons in dorsal root ganglion (DRG) and found that they exclusively innervate the epidermis of the skin and respond to multiple pruritogens. Ablation of MrgprA3+ neurons led to significant reductions in scratching evoked by multiple pruritogens and occurring spontaneously under chronic itch conditions whereas pain sensitivity remained intact. Importantly, mice with TRPV1 exclusively expressed in MrgprA3+ neurons exhibited only itch- and not pain behavior in response to capsaicin. Although MrgprA3+ neurons are sensitive to noxious heat, activation of TRPV1 in these neurons by noxious heat did not alter pain behavior. These data suggest that MrgprA3 defines a specific subpopulation of DRG neurons mediating itch. Our study opens new avenues for studying itch and developing anti-pruritic therapies.
Pain and itch are two basic modalities initiated and mediated by primary sensory neurons with cell bodies in the dorsal root ganglia (DRG) or trigeminal ganglia. These neurons are highly diverse based on their somal sizes, expression of ion channels and receptors, innervation territories and electrophysiological properties1. Small-diameter DRG neurons with unmyelinated axons (C-fibers) are major neuronal types for mediating pain and itch1–2. The sensations of pain and itch are distinct and can each elicit different behavioral responses such as withdrawal (to avoid tissue injury) and scratching (to remove irritants), respectively. This leads to a fundamental question: are there DRG neurons whose primary function is to elicit itch and not pain? Three important criteria should be met for neurons to be considered as itch- specific. First, these neurons should respond to itchy stimuli (pruritogens). Second, specific loss of these neurons should lead to a reduction in itch but not pain. Third and the most important, specific activation of these neurons should evoke only itch but not pain.
There is ample evidence for different types of primary cutaneous neurons that respond to stimuli that elicit itch sensations in human and itch-associated behavior in animals. These neurons are subsets of nociceptive neurons that also respond to chemical, mechanical or heat stimuli that elicit pain in humans or pain behavior in animals and not itch or itch behavior. For example, in human nerve, there are mechanically insensitive cutaneous C-fibers whose responses to histamine corresponded to the itch produced3 but which also respond to noxious heat and/or one or more painful chemicals such as capsaicin4. There are other C-fibers in humans, monkeys, and mice that respond to heat and mechanical noxious stimuli but whose responses to cowhage spicules correspond to the histamine-independent itch this stimulus produces5–9. Because neurons that are responsive to pruritic chemical agents like histamine are also activated by painful stimuli there is uncertainty as to whether their function is specific to the mediation of itch. The major hurdle to further investigation is the lack of specific molecular markers for labeling these neurons and molecular methods of manipulating their contribution to itch.
We have shown that certain Mrgprs, encoding a large family of G protein-coupled receptors, specifically expressed in subsets of small-diameter DRG neurons, function as receptors for certain pruritogens and mediate itch-associated behavior (i.e. scratching) accordingly. For example, MrgprA3 is a receptor for the anti-malarial drug chloroquine and mediates its direct activation of DRG neurons and chloroquine-induced site-directed scratching10–11. Bovine adrenal medulla peptide 8–22 (BAM8–22) and SLIGRL-NH2, each capable of eliciting a histamine-independent itch, act through MrgprC1111–13. Given that the expression of MrgprA3 and MrgprC11 largely overlaps in DRG neurons14, most chloroquine-sensitive neurons also respond to BAM8–22 and SLIGRL-NH2 11, 13. As histamine can activate all chloroquine-sensitive neurons, MrgprA3+ neurons thus respond to at least 4 different pruritogens11. Therefore, MrgprA3+ neurons are good candidates as neurons specifically dedicated to eliciting itch and itch-associated behavior. To test this hypothesis, we generated a transgenic mouse line in which Cre recombinase is specifically expressed in MrgprA3+ neurons. We used this genetic tool to demonstrate that MrgprA3 defines a specific population of itch-mediating neurons and determine the molecular identity and cellular properties of these neurons.
Results
Generation of MrgprA3GFP-Cre transgenic mouse line
To investigate the function of MrgprA3-expressing neurons, we generated a bacterial artificial chromosome (BAC) transgenic mouse line in which the GFP-Cre fusion protein was expressed under the control of the MrgprA3 promoter (Fig. 1a). The MrgprA3GFP-Cre transgenic line was fertile and showed no obvious phenotypic or behavioral abnormalities. We crossed MrgprA3GFP-Cre with Cre-dependent ROSA26tdTomato reporter mice15 in which Cre active neurons were marked by the expression of tdTomato (Fig. 1a). Because of the strong fluorescence of tdTomato, these neurons can be visualized directly by epifluorescence without immunohistochemical staining (Fig. 1c, e, h). We used antibodies against GFP to stain adult DRG sections from MrgprA3GFP-Cre; ROSA26tdTomato mice. All GFP and tdTomato signals were colocalized in a subset of small-diameter sensory neurons (Fig. 1b–d) (n=3, 312 double-labeled neurons). To characterize Cre activity in the MrgprA3+ neurons, we performed MrgprA3 in situ hybridization on DRG sections from MrgprA3GFP-Cre; ROSA26tdTomato mice. We collected tdTomato fluorescent images and bright field images of each DRG section before the in situ procedure and matched them to the same DRG section afterward (Fig. 1e, f). Of the tdTomato+ neurons examined (389 from 3 mice), 97.2% ± 0.6% exhibited the MrgprA3 in situ signal and 95.5% ± 0.5% of MrgprA3 in situ positive neurons (n=3, 396 MrgprA3+ neurons examined) were also tdTomato+, suggesting that the expression of GFP-Cre is tightly controlled by the endogenous MrgprA3 promoter.
Figure 1. Generation of the MrgprA3GFP-Cre transgenic mouse line.
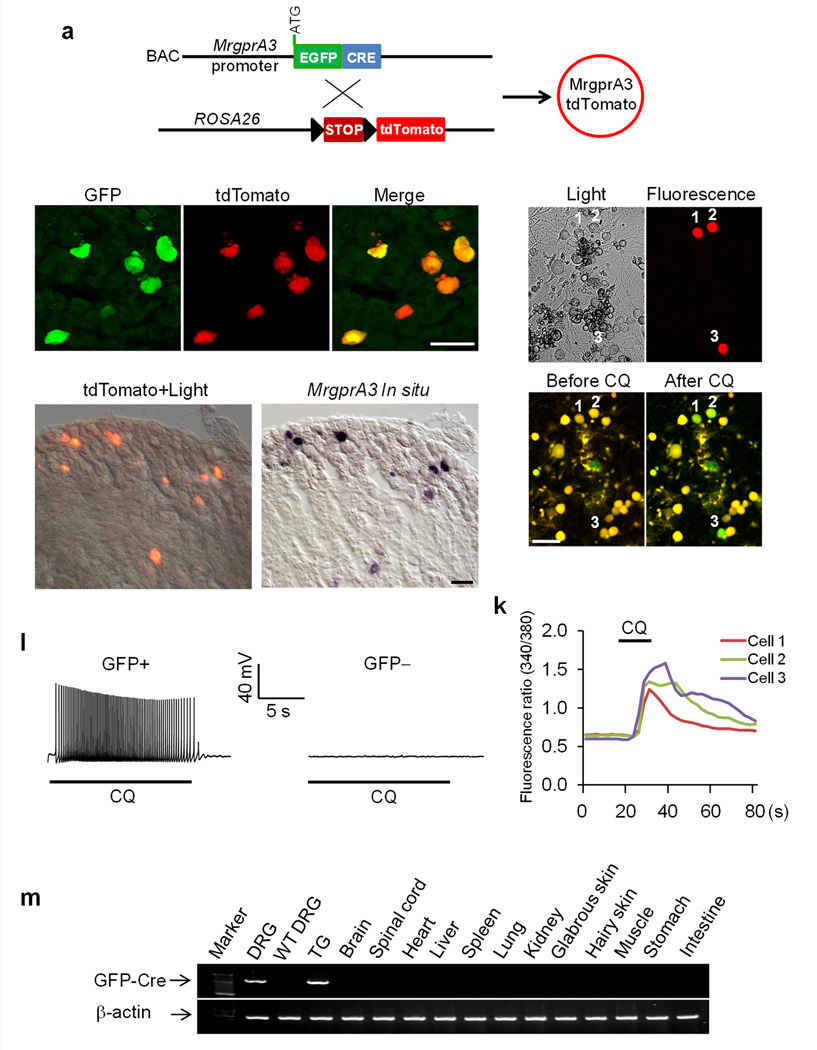
(a) Diagram showing the mating strategy. (b–d) DRG sections from an MrgprA3GFP-Cre; ROSA26tdTomato mouse stained with anti-GFP antibody. TdTomato fluorescence was visualized directly without staining. (e) Merged image of tdTomato fluorescence and bright field view of a DRG section from a MrgprA3GFP-Cre; ROSA26tdTomato mouse. (f) In situ hybridization with an MrgprA3 probe on the same section shown in (e). (g–j) Representative light view (g), fluorescent view (h), and Fura-2 ratiometric images of tdTomato+ DRG neurons (i,j). The color of the neurons switching from yellow to green indicates the increase of the intracellular calcium concentration. (k) Representative traces evoked by chloroquine (CQ; 1 mM) in the calcium imaging assay from the three tdTomato+ neurons labeled in (j). (l) Representative traces of APs induced by chloroquine (1 mM) in neurons from MrgprA3GFP-Cre mice. chloroquine can induce APs in GFP-Cre+ DRG neurons (10 of 11). In contrast, GFP-Cre− DRG neurons (12) do not show any response to chloroquine. (m) RT-PCR analysis of GFP-Cre expression in various tissues from MrgprA3GFP-Cre mice. GFP-Cre was only detected in the DRG and trigeminal ganglia. Scale bar represents 50 μm.
Our previous studies have shown that chloroquine activates MrgprA3 and induces calcium influx and action potentials in dissociated MrgprA3+ neurons11. To further characterize the MrgprA3GFP-Cre transgenic line, we used Ca2+ imaging to examine the responses of dissociated GFP-Cre labeled DRG neurons to chloroquine. Of the 112 tdTomato+ neurons obtained from 3 MrgprA3GFP-Cre; ROSA26tdTomato mice, 90.8% ± 4.6% showed robust increases in intracellular Ca2+ following chloroquine application (Fig. 1g–k). In whole-cell patch clamp recording, 10 of 11 GFP-Cre+ neurons from MrgprA3GFP-Cre transgenic mice displayed a train of action potentials upon chloroquine treatment. In contrast, GFP-Cre-negative neurons failed to generate action potentials with similar treatment (Fig. 1l). To examine the expression pattern of GFP-Cre, we performed RT-PCR with intron-spanning primers on various adult mouse tissues. Among the tissues tested, GFP-Cre was found only in the DRG and trigeminal ganglia, which is consistent with the expression pattern of MrgprA3 11 (Fig. 1m). Taken together, we have successfully generated a transgenic line in which GFP-Cre is specifically expressed in MrgprA3+ neurons and which provides a powerful tool to examine the function of MrgprA3+ neurons.
MrgprA3+ neurons represent a unique IB4+, CGRP+ population
Previously, we have performed in situ hybridization to examine the coexpression of MrgprA3 with different molecular markers and found that MrgprA3+ neurons are a subset of small diameter neurons with nociceptive characteristics16. Using the MrgprA3GFP-Cre transgenic line, we were able to examine the cellular features of MrgprA3 neurons with higher sensitivity. We collected L4-L6 DRG sections from MrgprA3GFP-Cre; ROSA26tdTomato mice and stained them with antibodies against a number of neuronal markers (Fig. 2a–r). Of the 174 tdTomato+ neurons examined, 79.1% were positive for the nonpeptidergic nociceptive marker IB4 (Fig. 2a–c). For 130 other tdTomato+ neurons examined, most 84.7% were labeled with P2X3 and 97.6% with c-RET (Fig. 2d–i). None of the MrgprA3+ neurons were labeled with the peptidergic marker substance P (156 tdTomato+ neurons examined) (Fig. 2m–o) or the myelinated neuronal marker neurofilament 200 (NF200, 200 tdTomato+ neurons examined) (Fig. 2p–r). In addition, of the 184 tdTomato+ neurons examined, 84.2% of MrgprA3+ neurons co-expressed another peptidergic marker, calcitonin gene–related peptide (CGRP). The expression of CGRP appeared lower in MrgprA3+ neurons than in many other neurons that expressed the peptide (Fig. 2j–l). When DRG sections were stained both with anti-CGRP antibody and IB4 labeling, 63.1% of MrgprA3+ neurons were positive for both CGRP and IB4 (106 tdTomato+ neurons examined) (Supplementary Fig. 1a–d). Therefore, MrgprA3+ neurons represent a unique population of DRG neurons that are labeled by both the nonpeptidergic marker IB4 and the peptidergic marker CGRP. In addition, we performed double labeling experiments using anti-TRPV1 antibody to stain TRPV1+ neurons and tdTomato for MrgprA3+ neurons in DRG and found that 19% of TRPV1-positive DRG neurons coexpressed MrgprA3 whereas 88.3% of MrgprA3-positive neurons coexpressed TRPV1 (Supplementary Fig. 1e–g). Furthermore, 93.3% of MrgprA3+ neurons expressed MrgprC11 (Supplementary Fig. 1h–k).
Figure 2. Characterization of MrgprA3+ neurons.
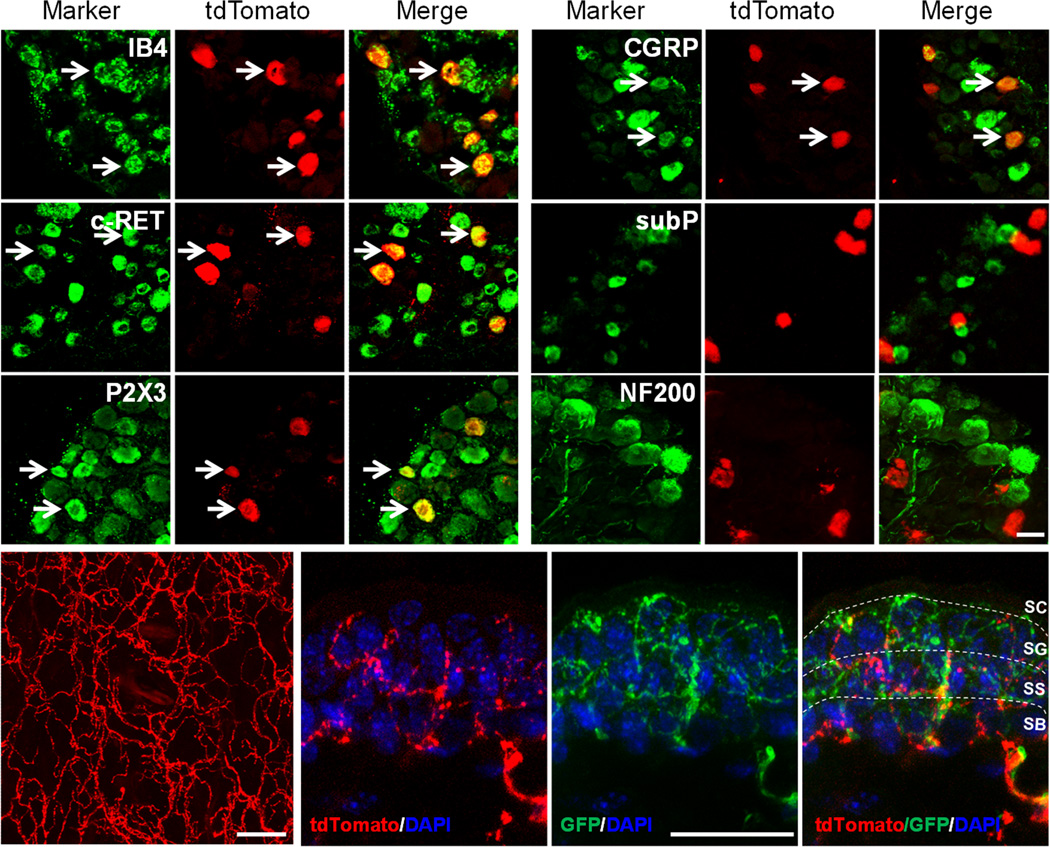
(a–r) L4-L6 DRG sections from MrgprA3GFP-Cre; ROSA26tdTomato mice stained with the indicated markers. Arrows mark representative double-labeled neurons. (s) Whole-mount imaging of the dorsal thoracic skin from MrgprA3GFP-Cre; ROSA26tdTomato mice showing the distribution of MrgprA3+ nerve fibers. (t–v) Section of the dorsal thoracic hairy skin from MrgprA3GFP-Cre; ROSA26tdTomato; MrgprDGFP/+ mice showing MrgprA3+ and MrgprD+ free nerve endings in the superficial epidermis. The section was counterstained with DAPI to label nuclei. Scale bar represents 30 μm. SC, stratum corneum; SG, stratum granulosum; SB, stratum basalis; SS, stratum spinosum. The epidermal subdivisions were identified by keratinocyte nuclear morphology and packing density.
MrgprA3+ neurons exclusively innervate the epidermis
To determine the peripheral targets of MrgprA3+ neurons, we examined an extensive array of tissues from MrgprA3GFP-Cre; ROSA26tdTomato mice by visualizing the tdTomato fluorescence of MrgprA3+ nerve fibers terminals in the skin and other tissues. We found that MrgprA3+ neurons exclusively innervated the epidermis of the skin (Fig. 2s–v, Supplementary Table 1). MrgprA3+ fibers were occasionally seen wrapping around hair follicles. There was also innervation of glabrous skin though to a lesser extent than in hairy skin. In contrast, MrgprA3+ nerve fibers were completely absent from all other tissues examined in the rest of the body, including lung, heart, stomach, muscle and cornea (Supplementary Table 1).
A well-known neuronal population that exclusively innervates the epidermis is the MrgprD-expressing DRG neurons17. To examine if MrgprD+ and MrgprA3+ fibers innervated distinct regions of the epidermis, we crossed MrgprA3GFP-Cre; ROSA26tdTomato mice with MrgprD-GFP knockin mice in which MrgprD-expressing neurons are labeled with GFP17. Sections from the hairy skin of MrgprA3GFP-Cre; ROSA26tdTomato; MrgprDGFP/+mice were collected and stained with an anti-GFP antibody. Since Cre is a nuclear protein, the fusion protein of GFP-Cre is in the nucleus but not in axons. Since tdTomoto is a cytoplasmic protein, it fills the entire axon of the neuron. Therefore, MrgprA3+ fibers in MrgprA3GFP-Cre; Rosa26tdTomoto mice were red not yellow (green+red). MrgprA3+ fibers (red) could be easily distinguished from MrgprD+ fibers which were green. We found that MrgprA3+ and MrgprD+ fibers represent two distinct nerve fiber populations (Fig. 2t–v), which is consistent with previous in situ hybridization results showing that MrgprD and MrgprA3 are expressed in non-overlapping DRG neurons18. Both MrgprA3+ and MrgprD+ free nerve endings penetrate the epidermis and terminate in the same layer of the superficial skin surface, the stratum granulosum (Fig. 2t–v).
The central projections of MrgprA3+ fibers co-terminated with IB4+ fibers in spinal lamina IImiddle (Supplementary Fig. 1l)17. This portion of lamina II is ventral to the CGRP+ lamina and dorsal to the PKCγ lamina (Supplementary Fig. 1m–o). Although we have shown that MrgprA3+ neurons express CGRP in cell bodies within the DRG (Fig. 2j–l), only minimal overlap between the MrgprA3+ central projections and the terminals of other CGRP+ expressing neurons was observed (Supplementary Fig. 1m). This could be explained by the fact that MrgprA3 expression is restricted to cell bodies with low CGRP expression.
MrgprA3+ fibers connect with GRPR+ neurons in spinal cord
There is evidence that spinal dorsal horn neurons expressing gastrin-releasing peptide receptor (GRPR) mediate chemically evoked scratching behavior in mice19–20. The ablation of GRPR+ neurons abolished scratching responses to multiple pruritic stimuli without altering behaviors in standard tests of pain behavior, suggesting GRPR+ neurons are dedicated pruriceptive neurons in the central nervous system20. We asked whether MrgprA3+ central fibers form direct connections with GRPR+ neurons in the spinal cord. We triple stained spinal cord sections for MrgprA3+ fibers (tdTomato), GRPR+ neurons, and the synaptic contacts between them (via the presynaptic marker, synapsin-1). We found that a majority (67%) of GRPR+ neurons formed synapses with MrgprA3 expressing primary afferents in dorsal horn (Fig. 3a–k). We further asked whether chloroquine injection could preferentially activate GRPR+ neurons. Here we used c-Fos induction as the marker for chloroquine-responsive neurons in the spinal cord and examined whether c-Fos was colocalized with GRPR. The FosGFP transgenic mice in which the expression of c-Fos-GFP fusion protein is controlled by the promoter of the c-Fos gene21 were used for this experiment. We injected 10 µl of chloroquine into the dorsal calf of the right hindlimb of the Fos-GFP mice. 90 min after the injection, the L3-L5 frozen spinal cord sections were collected and stained with antibodies against GFP and GRPR. chloroquine injection induced robust c-Fos expression in the superficial dorsal horn. More importantly, 60.9% ± 4.6% of c-Fos+ neurons on the ipsilateral side co-expressed GRPR (Fig. 3o–r) (3 animals, 218 c-Fos+ neurons examined). In contrast, when we applied hot water (50°C) onto the same region of the skin, only 18.7% ± 1.7% of the heat-evoked c-Fos+ neurons coexpressed GRPR (Fig. 3l–n, r) (n=3, 380 c-Fos+ neurons examined). Taken together, our result demonstrated that MrgprA3+ primary afferents formed direct synaptic connections with GRPR+, specific pruriceptive neurons in the dorsal horn. The different patterns of c-Fos induction by chloroquine (itchy) and noxious heat (painful) provide additional support for our conclusion that MrgprA3+ neurons in DRG are itch-mediating neurons.
Figure 3. MrgprA3+ DRG neurons form synaptic connections with GRPR+ neurons in the dorsal spinal cord.
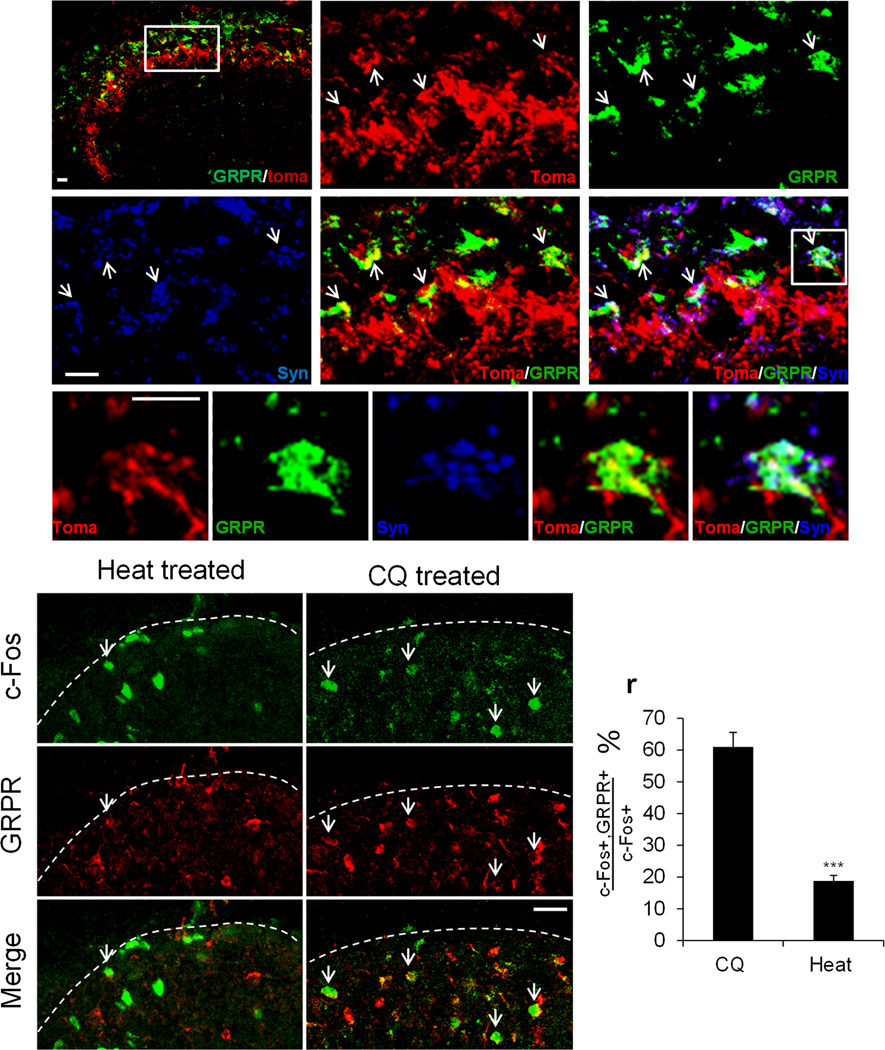
(a–k) spinal cord cross-sections from the thoracic region of adult MrgprA3GFP-Cre; ROSA26tdTomato mice labeled with GRPR (green), tdTomato reporter (red) and presynaptic marker synapsin 1 (blue).The boxed area in a is shown at greater magnification in b–f. The representative neuron in boxed region in f is shown at greater magnification in g–k. Scale bars, 10 µm. Arrows point to representative synaptic connections between tdTomato+ nerve fiber and GRPR+ neurons. (l–q) Double labeling of c-Fos-GFP (green) and GRPR (red) on the L3-L5 spinal cord cross-sections from c-FosGFP mice treated with chloroquine (8 mM) or hot water (50°C). Arrows indicate double labeled cells. Dashed lines define the boundary of spinal cord sections. (r) The percentage of c-Fos+ neurons that coexpress GRPR in the ipsilateral superficial spinal cords. Majority of c-Fos+ neurons induced by chloroquine are GRPR+ dorsal horn neurons whereas majority of heat-induced c-Fos+ neurons do not express GRPR (n=3, p=0.004). Error bars represent SEM.
MrgprA3+ neurons are cutaneous C-polymodal nociceptors
We then studied MrgprA3+ neurons in a more physiologically relevant setting with an in vivo electrophysiological recording method that we recently developed (Fig. 4)22. We first examined MrgprA3+ neurons by visualizing GFP-CRE fluorescence in the nuclei and then applying mechanical, heat, and chemical stimuli to their cutaneous receptive fields (Fig. 4a–c, f). A total of 16 DRG neurons expressing MrgprA3 were identified. All of these MrgprA3+ neurons had cutaneous nociceptors with C-fibers (conduction velocity range: 0.46 – 0.81 m/s) (Fig. 4c) that responded to both mechanical and heat noxious stimuli (Fig. 4d, e) (but not to cold, data not shown). They were therefore classified as C-mechanoheat (CMH) polymodal nociceptors. The following chemicals were injected intradermally into the receptive field in random order: vehicle (saline), histamine, BAM8–22, chloroquine, and capsaicin. At least 5 min elapsed between the termination of action potential discharges evoked by one injection and the application of the next. The neurons were silent (i.e., not spontaneously active) before each chemical application, and the injection of vehicle (n = 9) never evoked any response (i.e., no discharges beyond the insertion of the needle). All neurons tested responded to histamine (n = 9) and to capsaicin (n = 9). Action potentials were evoked by BAM8–22 in 8 of 10 neurons tested, and by chloroquine in 7 of 9 (Fig. 4f). Cowhage spicules (in groups of 3–5) inserted to the RF also evoked responses in all the neurons tested (n = 4). In addition, MrgprA3+ neurons did not respond to β-alanine (50 mM, n=9), an agonist for MrgprD which is expressed in a non-overlaping DRG subpopulation16, 23.
Figure 4. MrgprA3+ neurons have polymodal nociceptors with C-fibers and respond to multiple pruritogens.
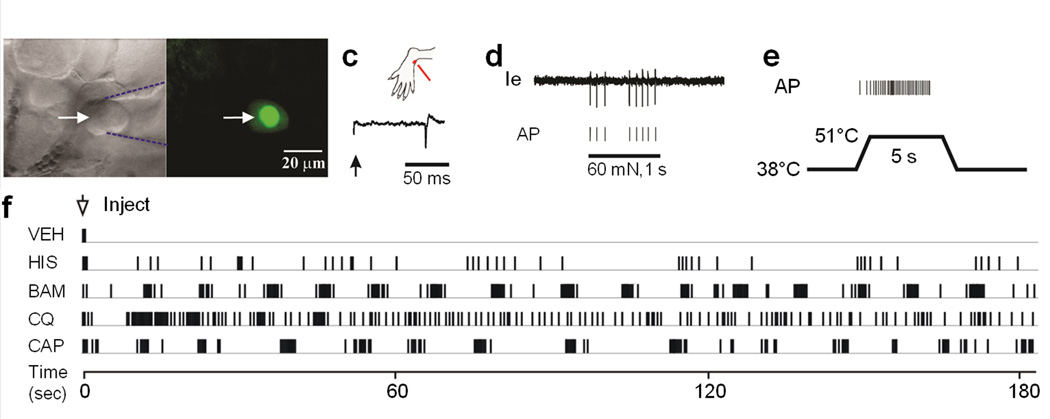
(a–f) In-vivo electrophysiological recording of MrgprA3+ neurons. (a) Bright field image of a neuronal recording (arrow) with an extracellular electrode (outlined with dashed blue lines). (b) Fluorescent microscopy revealed the expression of GFP (nuclear GFP fluorescence in the MrgprA3+ neuron. (c) Location of the cutaneous receptive field (RF, red dot) of this neuron on the hairy skin of the hindpaw and conduction velocity (lower trace, 0.49 m/s for the MrgprA3+ neuron), obtained with electrical stimulation (arrow) of the RF, are shown. (d) Responses of the neuron to a 60 mN force via a 200 μm diameter probe applied to its RF (1 sec) with original extracellular recording trace (Ie) and each action potential (AP) indicated by the vertical tic mark below. (e) Response to heat stimulation (38 to 51°C, 5 sec) indicates that this is a CMH neuron. (f) Responses of the CMH neuron to the intradermal injection for the MrgprA3 neuron of vehicle (VEH), followed by histamine (HIS, 5.4 mM), BAM8–22 (BAM, 0.2 mM), chloroquine (CQ, 1mM), and capsaicin (CAP, 3.3 mM).
Ablation of MrgprA3+ neurons does not alter pain behavior
One efficient and broadly applicable approach to investigate the function of specific cell populations in the context of the whole organism is to ablate the targeted cell population24. Inducible cell ablation in adults is commonly used to avoid compensation during development and can be achieved by expressing human heparin-binding epidermal growth factor-like growth factor precursor (HB-EGF, also called DTR), which is the receptor for the cytotoxic protein diphtheria toxin (DTX)25–26. We selectively ablated MrgprA3+ neurons by crossing the MrgprA3GFP-Cre transgenic line with the Cre-dependent ROSA26DTR line27, followed by DTX injection (Fig. 5a). 100% of MrgprA3+ neurons were lost three weeks after DTX injection (6.8% ± 0.6% vs. 0%) (Fig. 5b, c, and f). In contrast, the percentage of TrkA+ neurons was not changed (23.7% ± 3.6% vs. 24.5% ± 1.8%) (Fig. 5d, e, and f). To confirm the specificity of the ablation, we examined several subpopulations of DRG neurons by staining for various molecular markers. The proportion of CGRP-, IB4-, substance P- and NF200-immunoreactive neurons were similar in DTX-treated MrgprA3GFP-Cre; ROSA26DTR and ROSA26DTR littermates (Supplementary Fig. 2 a–o), suggesting that DTX treatment did not produce any general neurotoxic effects.
Figure 5. The ablation of MrgprA3+ neurons.
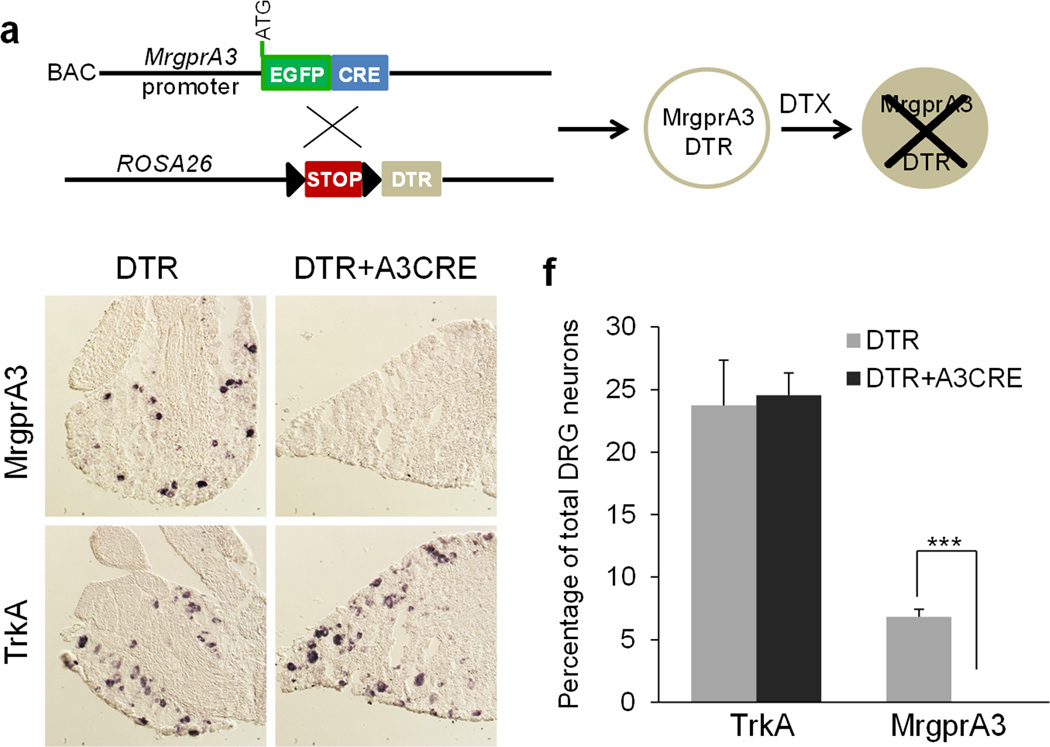
(a) Diagram showing the mating strategy. (b–e) In situ hybridizations of DRG sections from DTX-treated MrgprA3GFP-Cre; ROSA26DTR and ROSA26DTR littermates using MrgprA3 (b, c) and TrkA (d, e) cDNA probes demonstrate that MrgprA3+ neurons were specifically ablated in MrgprA3GFP-Cre; ROSA26DTRmice. (f) Quantitation of the in situ hybridization results (n = 3 mice per genotype, MrgprA3, p = 0.004, TrkA, p = 0.80). ***p < 0.005; two-tailed unpaired Student’s t-test.
We tested the responses of MrgprA3GFP-Cre; ROSA26DTR mice to acute noxious heat, cold, mechanical, and chemical stimulation after DTX administration. DTX-treated MrgprA3GFP-Cre; ROSA26DTR mice exhibited equivalent thermal sensitivities to DTX-treated ROSA26DTR littermates in the Hargreaves, hot plate, cold plate, and tail immersion tests (n≥6 for each group, p>0.5 for each test) (Supplementary Fig. 3a–d). Thus, MrgprA3+ neurons are dispensable for normal pain behavior to heat and cold noxious stimuli. Both groups also showed similar mechanical sensitivity as measured by von Frey filament responses (Supplementary Fig. 3e). Injection of capsaicin or formalin into the right hindpaw of the animals evoked comparable licking and flinching behaviors in the DTX-treated MrgprA3GFP-Cre; ROSA26DTR mice and ROSA26DTR littermates (Supplementary Fig. 3f and 3g). Taken together, these results indicate that MrgprA3+ neurons are not required for acute pain sensation. In addition, inflammatory hyperalgesia, induced by complete Freund’s adjuvant (CFA) injected into the paw, was not affected in DTX-treated MrgprA3GFP-Cre; ROSA26DTR mice compared to DTX-treated ROSA26DTR littermates (Supplementary Fig. 3i and 3j), suggesting that MrgprA3+ neurons are not required for inflammatory pain. Finally, MrgprA3+ neuron ablated mice showed normal motor function as determined by the rotarod test (Supplementary Fig. 2p). Therefore, the genetic manipulations leading to the ablation of MrgprA3+ neurons had no effects on neuronal systems mediating pain or motor function.
Ablation of MrgprA3+ neurons reduces itch behavior
We next evaluated whether itch associated behaviors were affected by the ablation of MrgprA3+ neurons. Indeed, site-directed scratching in response to the subcutaneous injection of chloroquine into the nape of the neck was significantly reduced in DTX-treated MrgprA3GFP-Cre; ROSA26DTR mice compared to DTX-treated ROSA26DTR littermates (Table 1). In addition, the ablation of MrgprA3+ neurons also abolished the site-directed scratching induced by BAM8–22 and SLIGRL-NH2, two pruritogens activating MrgprC11 (Table 1). The total number of scratching bouts exhibited by DTX-treated MrgprA3GFP-Cre; ROSA26DTR mice after BAM8–22 or SLIGRL-NH2 injected into the nape of the neck were comparable to that of WT group injected with saline (Table 1; SLIGRL, p=0.78, BAM8–22, p=0.20). We also tested whether MrgprA3+ neurons are required for histamine-induced scratching. Subcutaneous injection of histamine into the nape of the neck induced strong scratching behavior in DTX-treated ROSA26DTR mice, whereas DTX-treated MrgprA3GFP-Cre; ROSA26DTR mice exhibited a significant decrease in the total number of scratching bouts (Table 1), indicating that MrgprA3+ neurons were important for normal histamine-induced scratching behavior. However, these neurons are not the only neurons responsive to this chemical as evidenced by the residual behavior. We further tested scratching behaviors evoked by two other pruritogens, alpha-methyl-serotonin (α-Me-5HT) and endothelin-1 (ET-1)28. Both α-Me-5HT- and ET-1-induced scratching behaviors were significantly reduced in the DTX-treated MrgprA3GFP-Cre; ROSA26DTR mice compared to the DTX-treated ROSA26DTR littermates (Table 1).
Table 1. The ablation of MrgprA3+ neurons attenuates itch behavior.
Total scratching bouts (mean ± SEM) evoked by injection of different pruritogens or in different chronic itch conditions. “n” in the parenthesis indicate the number of the mice tested. Two-tailed unpaired Student’s t-test.
| pruritogen | DTR (n) | DTR+A3CRE (n) | P value | |
|---|---|---|---|---|
| Neck Injection | Saline | 12 ± 5 (7) | 11 ± 3 (7) | 0.82 |
| CQ | 236 ± 25 (5) | 65 ± 25 (6) | 0.0005 | |
| SLIGRL | 50 ± 8 (8) | 15.6 ± 7.5 (10) | 0.0002 | |
| BAM8–22 | 90 ± 12 (7) | 5.4 ± 2.6 (7) | 0.0002 | |
| Histamine | 60.7 ± 9.1 (7) | 24.2 ± 6.7 (8) | 0.005 | |
| α-Me-5HT | 140.5 ± 18.4 (7) | 81.4 ± 16.7 (7) | 0.024 | |
| ET-1 | 189.8 ± 52.9 (10) | 81.3 ± 24.5 (8) | 0.047 | |
| Cheek Injection | CQ | 66 ± 20.5 (7) | 12.8 ± 3.6 (6) | 0.027 |
| Histamine | 23.6 ± 4.7 (14) | 9.6 ± 3.3 (11) | 0.026 | |
| β-alanine | 29.6 ± 8.7 (5) | 31.5 ± 3.5 (6) | 0.81 | |
| Chronic itch condition | Dry skin | 138.1 ± 18.7 (7) | 36.6 ± 11.4 (7) | 0.0006 |
| Allergic itch | 147.2 ± 16.6 (10) | 79.3 ± 19.4 (9) | 0.01 | |
We next examined the role of MrgprA3+ neurons in chronic itch conditions by using dry skin and allergic itch mouse models. Dry skin pruritus is a common symptom in patients with xerosis and atopic dermatitis, as well as systemic disorders including cholestasis and chronic renal failure29–30. Using a murine model for dry skin pruritus31, we found that the spontaneous scratching response decreased significantly after the ablation of MrgprA3+ neurons (Table 1). Itch is always associated with allergic chronic disease such as, allergic conjunctivitis and allergic contact dermatitis32–34. However, little is known of the sensory neurons that mediate allergic itch. In a mouse model of ovalbumin-induced allergic itch30, we found that the ablation of MrgprA3+ neurons significantly inhibited the scratching behaviors (Table 1).
Previous reports have shown that mice exhibit distinct behaviors directed to cheek injection of pruritogens versus algogens35. Pruritogens elicit scratching with the hindpaw whereas algogens evoke facial wiping with the forelimb. This model provides a reliable means of distinguishing pain and itch behaviors. Injection of chloroquine in the cheek evoked robust hindpaw scratching on the cheek in the DTX-treated ROSA26DTR mice without any facial wiping. However, the scratching responses were completely abolished in the DTX-treated MrgprA3GFP-Cre; ROSA26DTR mice (Table 1) (chloroquine injected ablated mice vs. saline injected WT, 12.8 + 3.6 vs. 6 ± 3.1 bouts, p=0.16) (Table 1). Hindpaw scratching induced by the administration of histamine in the cheek was also significantly reduced though not eliminated after the ablation of MrgprA3+ neurons (Table 1). In contrast, facial wiping by the forepaw induced by capsaicin injection was not altered (Supplementary Fig. 3h), supporting the conclusion that MrgprA3+ neurons are specific for mediating itch- but not required for pain behavior. Recently, we found that intradermal injection of the amino acid, β-alanine, elicited itch in humans and site-directed scratching in mice36. Consistent with the finding that MrgprA3 and MrgprD define two separate subpopulation of DRG neurons18, cheek injection of β-alanine also induced a robust scratching response in DTX-treated MrgprA3GFP-Cre; ROSA26DTR mice (Table 1). The total number of scratching bouts was comparable for the two groups, demonstrating that MrgprA3+ neurons are dispensable for β-alanine-induced itch behavior. In summary, the ablation of MrgprA3+ neurons attenuated itch behaviors not only in acute conditions induced by an array of pruritogens, but also in chronic itch conditions.
MrgprA3+ neurons mediate only itch and not pain behavior
The evidence we have presented for a role in itch does not preclude the possibility that MrgprA3+ neurons may also mediate pain and that their ablation has no significant effect on pain behavior due to compensation from other pain-mediating neurons. One way to rule out this possibility is to selectively activate only MrgprA3+ neurons with a stimulus that normally elicits pain but not itch behavior. If pain as well as itch behavior occur then the activation of these neurons mediates both types of behavior whereas if itch behavior alone occurs, then the activation of these neurons is specifically linked to itch regardless of the type of stimulus that activates the neuron. Our approach was to exclusively express algogen receptor TRPV1 in MrgprA3+ neurons. A previous study has reported that the Cre-dependent ROSA26-TRPV1 knock-in mouse line expresses TRPV1 in a specific neuronal population37. In these transgenic animals, capsaicin can evoke action potential firing in the targeted neurons and induce behavioral responses37–38. We generated the TRPV1−/−; MrgprA3GFP-Cre; ROSA26TRPV1 mice by crossing MrgprA3GFP-Cre mice with ROSA26TRPV1 mice (to re-express TRPV1 in MrgprA3+ neurons) in the TRPV1 knockout background (to eliminate TRPV1 from all cells) (Fig. 6a). We first tested specific functional expression of Trpv1 transgene in MrgprA3+ neurons by calcium imaging using DRG neurons in acute culture. For cultured neurons sampled from WT mice (n = 396 neurons), TRPV1−/− mice (n = 305) and TRPV1−/−; MrgprA3GFP-Cre; ROSA26TRPV1 mice (n = 346), the incidence of responses to chloroquine were virtually the same (5.9% ± 1.2% vs. 5.75% ± 0.4% vs. 8% ±1.3%, n=3 mice per genotype) (Fig.6b, c, e, f, and h). We next analyzed the responsiveness of cultured DRG neurons from the three genotypes of mice to capsaicin. 37.2% of DRG neurons from WT animals responded to capsaicin. Neurons from TRPV1−/− did not respond to capsaicin. However, 6.6% of neurons from TRPV1−/−; MrgprA3GFP-Cre; ROSA26TRPV1 mice responded to capsaicin (Fig. 6d, g, and h). More importantly, every capsaicin-responding neurons from TRPV1−/−; MrgprA3GFP-Cre; ROSA26TRPV1 mice responded to chloroquine (Fig. 6e–g), demonstrating that TRPV1 is exclusively expressed in MrgprA3+ neurons.
Figure 6. Specific activation of MrgprA3+ neurons.
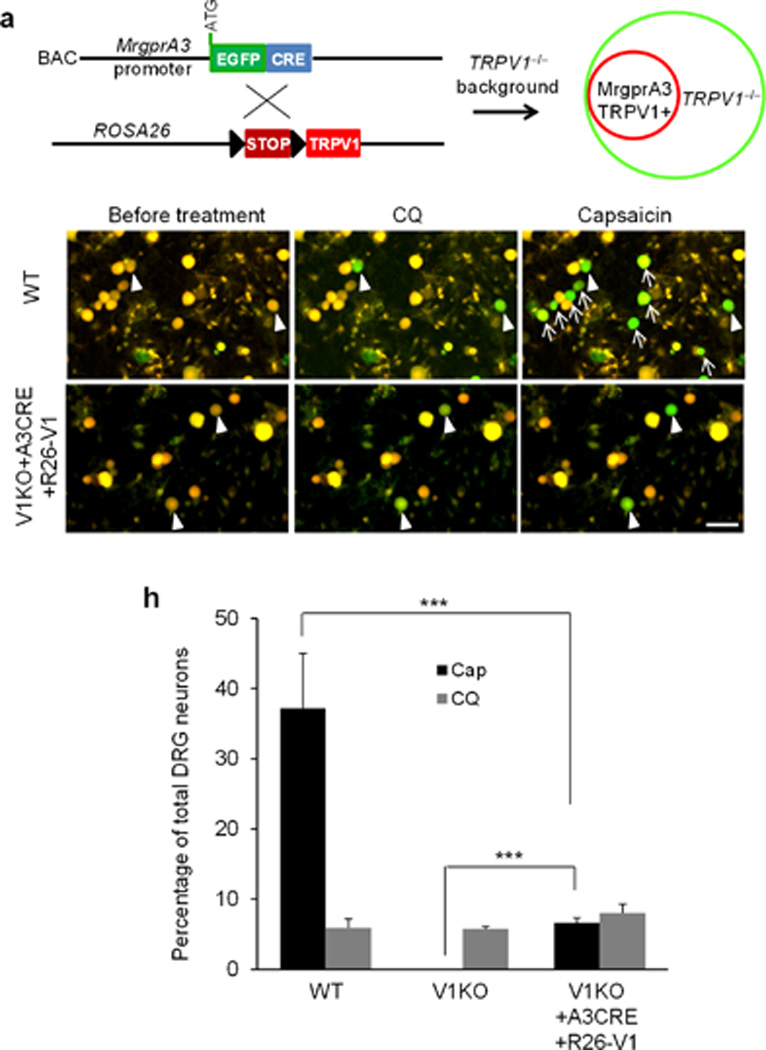
(a) Diagram showing the mating strategy. MrgprA3GFP-Cre; ROSA26TRPV1 mice were generated in TRPV1 knockout background to exclusively express TRPV1 in MrgprA3+ neurons. (b–g) Fura-2 ratiometric images of cultured DRG neurons from WT (b–d) and TRPV1−/−; MrgprA3GFP-Cre; ROSA26TRPV1 mice. The color of the neurons switching from yellow to green indicates the increase of the intracellular calcium concentration (b–g). In WT DRG neurons, capsaicin (1µM) activated a much bigger population than chloroquine (1mM). However, the percentage of TRPV1−/−; MrgprA3GFP-Cre; ROSA26TRPV1 DRG neurons responding to capsaicin and chloroquine are similar and every capsaicin-responding neuron responded to chloroquine. Arrowheads point to the neurons responded to both chloroquine and capsaicin. Arrows point to the neurons only responded to capsaicin. (h) The percentage of total DRG neurons from WT, TRPV1−/− mice, and TRPV1−/−; MrgprA3GFP-Cre; ROSA26TRPV1 mice responded to chloroquine (1mM) and capsaicin (1µM). ***p < 0.005; two-tailed unpaired Student’s t-test.
We then used the cheek injection model35 to examine whether pain- or itch-behaviors or both are triggered by an intradermal injection of capsaicin when TRPV1 is exclusively expressed in MrgprA3+ neurons. In response to capsaicin injected into the cheek, WT mice exhibited robust site-directed wiping (using the forepaw) and little if any scratching (with the hind paw), which is indicative of pain behavior. TRPV1−/− mice exhibited neither wiping nor scratching in response to cheek injection of capsaicin. In contrast, TRPV1−/−; MrgprA3GFP-Cre; ROSA26TRPV1 mice injected with capsaicin each exhibited robust site-directed scratching behaviors with little or no wiping (Fig. 7a, b), demonstrating that the selective activation of MrgprA3+ neurons by capsaicin elicited itch but little or no pain. As a control, cheek injection of mustard oil evoked similar numbers of wiping among all three groups of mice (WT, TRPV1−/−, and TRPV1−/−; MrgprA3GFP-Cre; ROSA26TRPV1) with little or no scratching, suggesting that pain sensation mediated by other TRPs (e.g. TRPA1) are normal in TRPV1−/−; MrgprA3GFP-Cre; ROSA26TRPV1 mice (Fig. 7c, d). In addition, these mice showed similar mechanical sensitivity as measured by von Frey filament responses (Fig. 7g).
Figure 7. Specific activation of MrgprA3+ neurons evokes robust scratching and little or no pain response.
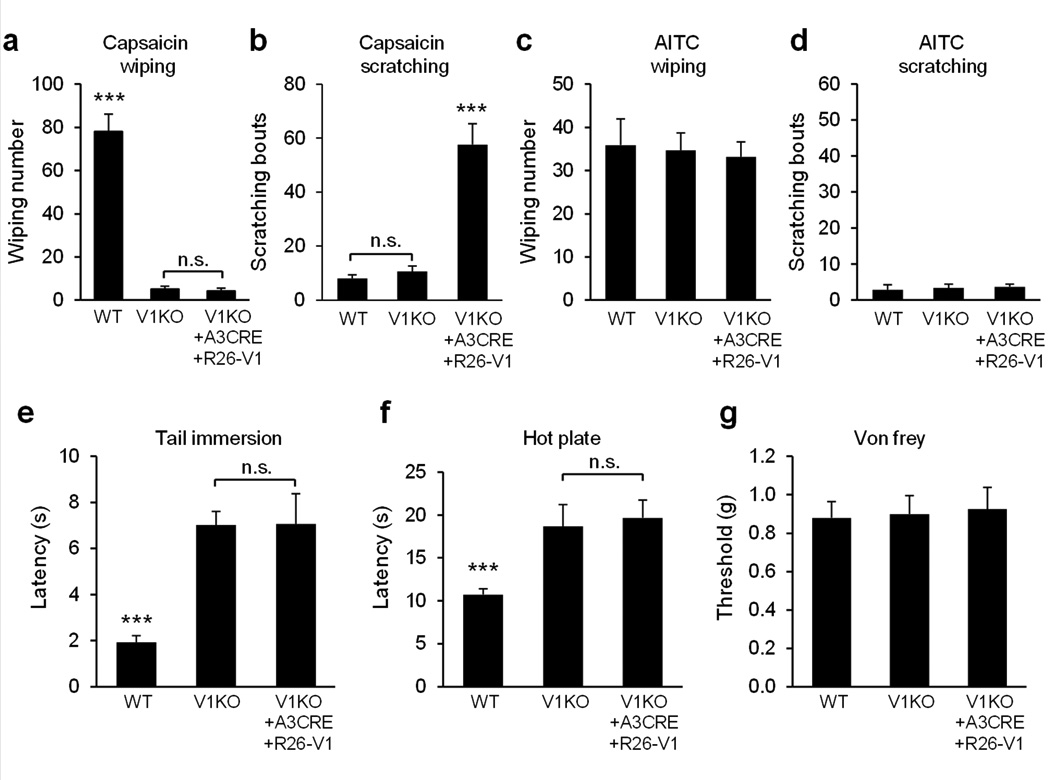
(a) Cheek injection of capsaicin (3.3 mM induced robust, site-directed wiping with the forepaw in WT mice but not in TRPV1−/− mice or TRPV1−/−; MrgprA3GFP-Cre; ROSA26TRPV1 mice (n=13 vs. 13 vs. 10, WT vs. V1KO, p=1.8E-09, V1KO vs. V1KO+A3CRE+R26-V1, p=0.59). (b) Cheek injection of capsaicin (3.3 mM) induced robust site-directed scratching with the hind paw in TRPV1−/−; MrgprA3GFP-Cre; ROSA26TRPV1 mice but not in WT mice or TRPV1−/− mice (WT vs. V1KO vs. V1KO+A3CRE+R26-V1, n=13 vs. 13 vs.10, WT vs. V1KO, p=0.28, V1KO vs. V1KO+A3CRE+R26-V1, p=7.2E-07). (c–d) Cheek injection of allyl isothiocyanate (AITC, 50mM) induced robust wiping (c), not scratching (d) (WT vs. V1KO vs. V1KO+A3CRE+R26-V1, n=8 vs. 8 vs.7). (e) Response latencies of WT, TRPV1−/− and TRPV1−/−; MrgprA3GFP-Cre; ROSA26TRPV1 mice in the tail immersion test (50°C) (WT vs. V1KO vs. V1KO+A3CRE+R26-V1, n=10 vs. 11 vs.8, WT vs. V1KO, p=2.3E-07, V1KO vs. V1KO+A3CRE+R26-V1, p=0.97). (f) Response latencies in the hot plate tests (55°C) (WT vs. V1KO vs. V1KO+A3CRE+R26-V1, n=11 vs. 8 vs. 7, WT vs. V1KO, p=0.0017, V1KO vs. V1KO+A3CRE+R26-V1, p=0.75). (g) Response threshold in the von Frey test (WT vs. V1KO vs. V1KO+A3CRE+R26-V1, n=10 vs. 10 vs. 8, WT vs. V1KO, p=0.87, V1KO vs. V1KO+A3CRE+R26-V1, p=0.86). ***p < 0.005; two-tailed unpaired Student’s t-test. n.s., not significant.
TRPV1 plays an essential role in detecting noxious heat. As previously shown, TRPV1−/− mice exhibited significantly longer tail flick latency in response to 50 °C in tail immersion test as that of WT mice39 (Fig. 7e). A similar impaired response was obtained in the hot plate assay39 (Fig. 7f). Strikingly, TRPV1−/−; MrgprA3GFP-Cre; ROSA26TRPV1 mice have similar latencies in both assays as those in TRPV1−/− mice suggesting that re-introducing TRPV1 in MrgprA3+ neurons in TRPV1−/− mice did not rescue any deficit in sensing noxious heat (Fig. 7e, f).
Discussion
Previously we showed that MrgprA3 functions as a receptor for the pruritogen chloroquine. Because of its specific expression in DRG, the MrgprA3 promoter provided the first means of answering some fundamental questions: Do itch-specific neurons exist? If so, what is the molecular identity of these neurons and what are the cellular and functional properties of these neurons?
A cellular basis for itch being felt only in the skin
An interesting aspect of itch sensation is that itch arises from the skin, but not from deeper tissues such as muscle, bone, and visceral organs from which pain can be felt. Now, we provide a cellular basis of this phenomenon which is that MrgprA3+ axons exclusively innervate the skin. It has also been suggested that itch-mediating nerve fibers terminate in the superficial skin layer. The traditional algogen, capsaicin, generates pain when delivered intradermally but can evoke an initial sensation of itch when applied topically or by means of a cowhage spicule to the skin40–42. One possible explanation is that itch-mediating nerve fibers innervate more superficially and can be chemically activated more selectively by spicule than other fibers that are readily activated by intradermal injection. Indeed, MrgprA3+ axons penetrate the epidermis and terminate in the stratum granulosum layer.
MrgprA3+ neurons mediate multiple types of itch
Our data show that MrgprA3+ neurons respond to multiple chemical pruritogens (Fig. 3). The ablation of MrgprA3+ neurons reduced itch behavior in response to all of the pruritic chemicals that were tested except β-alanine, including those that are known to directly activate Mrgprs (chloroquine, BAM 8–22, and SLIGRL)11, 13 and those that activate other receptors (histamine, ET-1, and α-Me-5HT)1, 43–44. In addition, the responsiveness of MrgprA3+ neurons to algesic stimuli, namely capsaicin, heat and mechanical stimuli provide a mechanism whereby noxious stimuli, such as the punctuate application of such stimuli might under certain conditions elicit itch. Of clinical relevance is that the spontaneous scratching from dry skin or from allergic responses is significantly attenuated in MrgprA3 ablation mice. Together these data implies that MrgprA3+ neurons express a broad array of receptors for pruritic stimuli and play a role in mediating both acute and chronic itch.
The insensitivity of MrgprA3+ neurons to β-alanine and the normal scratching responses to β-alanine in MrgprA3 ablated mice also support the existence of multiple types of itch-mediating neurons. In addition, the residual scratching induced by certain pruritogens especially histamine, ET-1, and α-Me-5HT in MrgprA3 ablation mice indicate that other types of DRG neurons also play a role in mediating itch. For example, at least two types of neurons must mediate histamine evoked itch behavior, namely MrgprA3+ neuron and another, as yet unknown, type that must contribute to the histamine evoked scratching in mice that remains after MrgprA3+ neurons are ablated. Perhaps the later neurons have properties similar to the histamine-responsive, mechanically insensitive, neurons with C-fibers that have been recorded in humans3–4.
TRPV1+ neurons belong to parallel pain and itch pathways
Our data demonstrate that MrgprA3+ neurons are also TRPV1+ and sensitive to capsaicin, and to noxious heat. Specific ablation of this subpopulation of TRPV1+ neurons did not affect the pain behavior induced by noxious thermal, mechanical, and chemical stimuli. Conversely, capsaicin injection in TRPV1−/−; MrgprA3GFP-Cre; ROSA26TRPV1 mice only induced scratching, not wiping, suggesting that the selective activation of the TRPV1+ subpopulation expressing MrgprA3 primarily evokes itch, not pain. In addition, the response latencies to noxious heat of TRPV1−/−; MrgprA3GFP-Cre; ROSA26TRPV1 mice are similar to those of TRPV1−/− mice suggesting that although MrgprA3+ neurons are sensitive to noxious heat, the activation of these neurons by noxious heat did not elicit pain behavior. The specific itch behavior we obtained by selectively activating only the MrgprA3+ neurons provide direct support for the applicability of Muller’s 1826 doctrine of “specific nerve energies” to a submodality of cutaneous sensations, namely the sensation of itch45. That is, the quality of sensation evoked by a stimulus depends on the specific neuronal pathway that is activated regardless of the nature of the stimulus. In this case, if only MrgprA3+ neurons are activated by a stimulus, the sensation should be itch even if the stimulus is noxious mechanical, heat or chemical. However, we cannot rule out the possibility that weaker nociceptive sensations that humans typically report as accompanying itch, may be present in mice but of insufficient magnitude to elicit overt pain behavior.
Our findings suggest that nociceptive, capsaicin-sensitive DRG neurons (that express TRPV1) are of two types: one mediating itch and the other pain. But if this is so, why does the activation of the entire population of TRPV1+ neurons by capsaicin (i.e. in WT mice) only evoke pain and not itch behavior even though MrgprA3+ neurons are also activated? One explanation is that the brain decodes activity in the pruriceptive neurons as “itch” only when there is minimal activity in the nociceptive neurons that do not mediate itch46. Another or additional possibility is that there are central inhibitory mechanisms activated by the pain-mediating neurons that act to block transmission in an itch-mediating pathway47–50. Indeed, in the VGLUT2 conditional knockout mice, i.e. with nociceptors silenced by disrupting glutamatergic transmission, capsaicin evokes itch rather than pain behavior49. It would be important to determine which VGLUT is expressed in MrgprA3+ neurons and whether glutamate is essential for itch signaling transmission in these neurons. To study neurons mediating pain from capsaicin injection, we need to develop new genetic tools to specifically manipulate TRPV1+ in these neurons (vs. itch-mediating, e.g. MrgprA3+ neurons). Currently available DRG-specific Cre lines drive gene expression not only in small diameter neurons with C-fibers but also in most other types of DRG neurons including those with large-diameter Aβ-fibers which would make interpretation of behavioral data difficult.
The search for pruriceptive neurons mediating itch began nearly a century ago. Here, we have defined and characterized a specific subpopulation of itch-mediating neurons which should open new avenues to advancing itch research as well as foster the development of itch therapies that target this population.
ONLINE METHODS
Mouse lines
We purchased a mouse BAC clone (RP23-311C15) containing the entire MrgprA3 gene from the Children's Hospital Oakland Research Institute. We modified the BAC clone using the homologous recombination in bacteria to generate MrgprA3GFP-Cre mouse line (Supplemental Experimental Procedure). We purchased ROSA26Tdtomato, ROSA26DTR, ROSA26TRPV1 and TRPV1−/− mice lines from Jackson Laboratory and crossed with MrgprA3GFP-Cre. We used MrgprA3GFP-Cre transgenic line and both reporter mice lines as hemizygotes or heterozygotes for all the experiments.
Behavioral studies
All behavioral tests were performed with an experimenter blind to genotype. The mice were 2 to 3 month-old males (20–30 g) that had been backcrossed to C57Bl/6 mice for at least six generations. All experiments were performed under the protocol approved by the Animal Care and Use Committee of Johns Hopkins University School of Medicine. 5 weeks old MrgprA3GFP-Cre; ROSA26DTR mice and ROSA26DTR littermates were injected with diphtheria toxin (i.p., 40ug/kg, EMD Biosciences) twice separated by 72 hrs. Behavioral experiments were performed three weeks after the first toxin injection. The day before the behavioral tests, all animals were acclimated for at least 30 min to their testing environment. We housed 4–5 mice in each cage in the vivarium with 12 hr light/dark cycle and all the behavioral tests were performed in the morning.
Back injections and cheek injections of chemicals were performed as previously described 35. Briefly, pruritic compounds were subcutaneously injected into the nape of the neck for back injection and the right cheek of the animal for the cheek injection after acclimatization. Behavioral responses were video recorded for 30 min. The video recording was subsequently played back in slow-motion and the number of bouts of scratching with the hindpaw and also, for the cheek, wiping with the forepaw, each directed toward the injection site, were counted.
In the dry skin model, the rostal back of the mice were treated twice daily with cutaneous application of acetone/ether (1:1) mixture followed by water. After a 6-day treatment, the mice showed robust spontaneous scratching and the treated skin area exhibited decreased stratum corneum hydration and increased trans-epidermal water loss, which mimic the symptoms of dry skin in patients29, 31. In an allergy model of itch, 50 µg ovabumin dissolved in PBS was administered intraperitoneally together with 2 mg of Inject Alum twice separated by 10 days. One week after the second sensitization, 50 µg ovalbumin dissolved in saline was administered in the same manner as other pruritogens and scratching behavior was quantified.
For the hot plate test, a clear plexiglass cylinder was placed on the plate and the mice were placed inside the cylinder. The onset of brisk hindpaw lifts and/or flicking/licking of the hindpaw was assessed at different temperatures.
For the cold plate test, a ceramic plate on a bed of ice was cooled in a −20°C freezer. During the test, the plate was allowed to warm to 0°C as measured by two independent temperature probes. The onset of brisk hindpaw lifts and/or flicking/licking of the hindpaw was assessed.
For the tail immersion test, mice were gently restrained in a 50 ml conical tube into which the mice voluntarily entered. The protruding one third of the tail was then dipped into a water bath of varying temperatures. The latency to respond to the heat stimulus with vigorous flexion of the tail was measured.
For the Hargreaves test, mice were placed under a transparent plastic box (4.5 × 5 × 10 cm) on a glass floor. The infrared source was placed under the glass floor and the infrared light was delivered to the hindpaw. The latency for the animal to withdraw its hindpaw was measured.
For the Von Frey filament test, mice were placed under a transparent plastic box (4.5 × 5 ×10 cm) on a metal mesh. Von Frey filaments, each delivering a different bending force, were applied to the hind paw using the up-down method and the threshold force corresponding to 50% withdrawal determined.
For the chemically-induced pain test, an intraplantar injection was used to administer a total volume of 6 μl of capsaicin (1 mg in 10 ml saline/7% Tween-80), or formalin (2% formalin in saline). The time mice spent licking and flinching was measured for 15 minutes after capsaicin injection and for 1hr after formalin injection.
For the Rotarod test, each mouse was trained for 5 minutes at a constant speed of 4 rpm on the rotarod (Rotamex, Columbus Instruments). The first trial started at least 1 hour after training. Every day, each mouse received 3 trials, separated by 30 minutes, at speeds accelerating from 4 to 40rpm (with a 4 rpm increase every 30 s). Each mouse was tested for 3 consecutive days. The trial was finished when the mouse fell off the rotarod. The latency to falling off the rotarod was recorded and used in subsequent analyses.
Cultures of dissociated DRG neurons
DRG from all spinal levels of 3–4 weeks old mice were collected in cold DH10 medium and treated with enzyme solution at 37°C. After trituration and centrifugation, cells were re-suspended in DH10, plated on glass coverslips coated with poly-D-lysine and laminin, cultured in an incubator at 37°C, and used within 24 hr.
Calcium imaging
Calcium imaging was performed as previously described. Briefly, neurons were loaded with Fura 2-acetomethoxyl ester (Molecular Probes) for 30 min in the dark at room temperature. After washing, cells were imaged at 340 and 380 nm excitation to detect intracellular free calcium. Calcium imaging assays were performed with an experimenter blind to genotype.
Whole-cell current-clamp recordings of cultured DRG neurons
Neurons plated on coverslips were transferred into a chamber with the extracellular solution. Patch pipettes had resistances of 2–4 MΩ. In current-clamp recordings, action potential measurements were performed with an Axon 700B amplifier and the pCLAMP 9.2 software package (Axon Instruments). Neurons were perfused with chemicals for 20 s. All experiments were performed at room temperature (25°C).
Immunofluorescence
Adult mice (8–12 weeks old) were anesthetized with pentobarbital and perfused with 20 ml 0.1M phosphate buffer solution (PBS; pH 7.4; 4°C) followed with 25 ml fixative (4% formaldehyde and 14% [v/v] sat. picric acid in PBS; 4°C). Spinal cord and dorsal root ganglia (DRG) were dissected from the perfused mice. DRG was post-fixed in fixative at 4°C for 30 min, and spinal cord were fixed for 2 hr. Skin was dissected from nonperfused mice and fixed in 2% paraformaldehyde at 4°C for 16–18hr. Tissues were cryoprotected in 20% sucrose for more than 24 hr and were sectioned with a cryostat. The sections on slides were dried at 37°C for 30 min, and fixed with 4% paraformaldehyde at room temperature for 10 min. The slides were pre-incubated in blocking solution (10% normal goat serum, 0.2% Triton X-100 in PBS [pH 7.4]) for 1 or 2 hr at room temperature, then incubated overnight at 4°C in primary antibodies. Secondary antibody incubation was performed at room temperature for 2 hr. The primary antibodies used: rabbit anti-CGRP (T-4239; Peninsula; 1:1000), rabbit anti-NF200 (AB1982;Chemicon; 1:1000), rabbit anti-P2X3 (AB5895; Chemicon; 1:1000), armenian hamster anti-c-RET (Lo and Anderson, 1995) 1:4, rabbit anti-PKCγ (sc-211; Santa Cruz Biotechnology; 1:1000), rabbit anti-GFP (A-11122;Molecular Probes; 1:1000), chicken anti-GFP (GFP-1020; Aves Labs), mouse anti-Neuronal nuclei (MAB377; Chemicon; 1:300). The secondary antibodies used: goat anti-rabbit (A11011, Alexa 568 conjugated; A11008, Alexa 488 conjugated; Molecular Probes); goat anti-chicken (A11039, Alexa 488 conjugated; Molecular Probes) goat anti-Armenian hamster (127-165-160,cy3 conjugated; Jackson Lab); goat anti-mouse IgG1 (A21124, Alexa 568 conjugated; Molecular Probes). All secondary antibodies were 1:500 diluted in blocking solution. To detect IB4 binding, sections were incubated with 1:200 diluted Griffonia simplicifolia isolectin GS-IB4-Alexa 488 (A11001; Molecular Probes).
c-Fos induction in the spinal cord
The dorsal calf of the right hindlimb of c-FosGFP mice were shaved one day before the experiment. The mice were anesthetized with phenobarbital (200mg/kg) before the treatment. chloroquine (10µl of 8mM) was injected into the dorsal calf. For heat stimulation, one drop of 50 °C water was applied to the shaved dorsal calf 10 times, once per minute. The mice were perfused transcardially with 4% paraformaldehyde 90 min after the treatment. Frozen spinal cord sections (20 µm) were obtained from the lumbar level (L3-L5) and immunostained for GFP and GRPR (1:100; LS-A831; MBL).
In situ hybridization
Nonisotopic in situ hybridization on frozen sections from adult MrgprA3GFP-Cre; ROSA26Tdtomato mice was performed as previously described using cRNA probes (Dong et al., 2001). Briefly, MrgprA3 cRNA probes were labeled with digoxigenin-UTP. The probes were detected with an alkaline-phosphatase conjugated anti-digoxigenin antibody (Roche, 1:2000) and the fluorescent signals were developed using NBT/BCIP substrate.
RT-PCR
Total RNA was extracted from various tissues using Trizol reagent (Invitrogen) according to the manufacturer’s instructions. Reverse transcription was done using Superscript first strand (Invitrogen). PCR conditions: 95°C 3 min and 40 cycles of 30 sec at 95°C, 30 sec at 52°C, and 60 sec at 72°C. Intron-spanning primers were used to avoid genomic contamination. The forward primer is located in the first exon of MrgprA3. The reverse primer is located in the Cre sequence which is in the second exon of the GFP-Cre mRNA in transgenic animal. The sequences of the primers are: F: 5' TTCTGTAGTGACTGTATCCTTCCTTC 3'; R: 5' CCGGTTATTCAACTTGCACCAT 3'.
In vivo electrophysiological recording from cutaneous sensory neurons
Adult male mice were used for the in-vivo electrophysiological recording. The use and handling of animals were approved by the Institutional Animal Care and Use Committee of the Yale University School of Medicine and were in accordance with guidelines provided by the National Institutes of Health and the International Association for the Study of Pain. Primary sensory neurons innervating the skin on the hindlimb were extracellularly, electrophysiologicaly recorded using an in-vivo physiological preparation as described8, 22. A peripheral receptive field was identified by exploration of the hindlimb and application of various handheld stimuli. These included the use of a cotton tip (for innocuous mechanical stimuli), gentle pinching or indendations with a glass probe or von Frey filaments with fixed tip diameters (100 or 200 μm) but delivering different bending forces (for noxious mechanical stimuli), a temperature-controlled chip-resister heating probe, or ice (for noxious temperature stimuli). A series of chemicals were injected into the RF, in sequence, each in a volume of 5 μl, and consisting, in random order, of vehicle (0.9% NaCl), histamine (5.4 mM in vehicle), BAM8–22 (0.2 mM), chloroquine (1 mM), and capsaicin (3.3 mM) using a microsyringe with a 31 G needle8.
Statistical analysis
Data are presented as means ± s.e.m. The designation “n” represents the number of animals analyzed. The distribution of the variables ineach experimental group is approximately normal. Statistical comparisons were conducted by two-tailed, unpaired Student’s t-test. Power analysis was used to justify the sample size. Differences were considered statistically significant for P < 0.05. Representative data are from experiments that were replicated biologically at least three times with similar results.
Supplementary Material
ACKNOWLEDGEMENTS
We thank Chip Hawkins and the staff of Transgenic Mouse Core at Johns Hopkins University School of Medicine for assistance with BAC transgenic mouse generation. We thank Drs. David Anderson and Mark Zylka for providing MrgprD-GFP knockin mice and Dr. Ali Guler for providing Rosa26-TRPV1 mice. The work was supported by grants from the NIH to X.D. (NS054791 and GM087369), R.H.L. (NS047399 and NS014624), and Y.G. (NS070814). X.D. is an Early Career Scientist of the Howard Hughes Medical Institute.
Footnotes
AUTHOR CONTRIBUTIONS
L.H. generated the MrgprA3-Cre mice, carried out the genetic manipulation and most of behavioral, immunstaining, Ca2+ imaging experiments, and wrote the manuscript. C.M., H.N., and L.Q. conducted in vivo DRG recordings. Q.L., H.J.W., K.N.P. contributed to behavioral experiments. Y.C. and B.X. made the MrgprA3-Cre BAC construct. Z.T., Y.K. and Z.L. conducted in vitro DRG recordings. B.M., S.H., and Y.G. contributed to immunostaining experiments. R.H. and X.D. supervised the project and wrote the manuscript.
References
- 1.Ikoma A, Steinhoff M, Stander S, Yosipovitch G, Schmelz M. The neurobiology of itch. Nat Rev Neurosci. 2006;7:535–547. doi: 10.1038/nrn1950. [DOI] [PubMed] [Google Scholar]
- 2.Basbaum AI, Bautista DM, Scherrer G, Julius D. Cellular and molecular mechanisms of pain. Cell. 2009;139:267–284. doi: 10.1016/j.cell.2009.09.028. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 3.Schmelz M, Schmidt R, Bickel A, Handwerker HO, Torebjork HE. Specific C-receptors for itch in human skin. J Neurosci. 1997;17:8003–8008. doi: 10.1523/JNEUROSCI.17-20-08003.1997. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 4.Schmelz M, et al. Chemical response pattern of different classes of C-nociceptors to pruritogens and algogens. J Neurophysiol. 2003;89:2441–2448. doi: 10.1152/jn.01139.2002. [DOI] [PubMed] [Google Scholar]
- 5.Johanek LM, et al. A role for polymodal C-fiber afferents in nonhistaminergic itch. J Neurosci. 2008;28:7659–7669. doi: 10.1523/JNEUROSCI.1760-08.2008. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 6.Namer B, et al. Separate peripheral pathways for pruritus in man. J Neurophysiol. 2008;100:2062–2069. doi: 10.1152/jn.90482.2008. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 7.Akiyama T, Carstens MI, Carstens E. Facial injections of pruritogens and algogens excite partly overlapping populations of primary and second-order trigeminal neurons in mice. J Neurophysiol. 2010;104:2442–2450. doi: 10.1152/jn.00563.2010. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 8.Ma C, Nie H, Gu Q, Sikand P, Lamotte RH. In vivo responses of cutaneous C-mechanosensitive neurons in mouse to punctate chemical stimuli that elicit itch and nociceptive sensations in humans. J Neurophysiol. 2012;107:357–363. doi: 10.1152/jn.00801.2011. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 9.LaMotte RH, Shimada SG, Green BG, Zelterman D. Pruritic and nociceptive sensations and dysesthesias from a spicule of cowhage. J Neurophysiol. 2009;101:1430–1443. doi: 10.1152/jn.91268.2008. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 10.Wilson SR, et al. TRPA1 is required for histamine-independent, Mas-related G protein-coupled receptor-mediated itch. Nat Neurosci. 2011;14:595–602. doi: 10.1038/nn.2789. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 11.Liu Q, et al. Sensory neuron-specific GPCR Mrgprs are itch receptors mediating chloroquine-induced pruritus. Cell. 2009;139:1353–1365. doi: 10.1016/j.cell.2009.11.034. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 12.Sikand P, Dong X, LaMotte RH. BAM8-22 peptide produces itch and nociceptive sensations in humans independent of histamine release. J Neurosci. 2011;31:7563–7567. doi: 10.1523/JNEUROSCI.1192-11.2011. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 13.Liu Q, et al. The distinct roles of two GPCRs, MrgprC11 and PAR2, in itch and hyperalgesia. Sci Signal. 2011;4:ra45. doi: 10.1126/scisignal.2001925. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 14.Zylka MJ, Dong X, Southwell AL, Anderson DJ. Atypical expansion in mice of the sensory neuron-specific Mrg G protein-coupled receptor family. Proc Natl Acad Sci U S A. 2003;100:10043–10048. doi: 10.1073/pnas.1732949100. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 15.Madisen L, et al. A robust and high-throughput Cre reporting and characterization system for the whole mouse brain. Nat Neurosci. 2010;13:133–140. doi: 10.1038/nn.2467. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 16.Dong X, Han S, Zylka MJ, Simon MI, Anderson DJ. A diverse family of GPCRs expressed in specific subsets of nociceptive sensory neurons. Cell. 2001;106:619–632. doi: 10.1016/s0092-8674(01)00483-4. [DOI] [PubMed] [Google Scholar]
- 17.Zylka MJ, Rice FL, Anderson DJ. Topographically distinct epidermal nociceptive circuits revealed by axonal tracers targeted to Mrgprd. Neuron. 2005;45:17–25. doi: 10.1016/j.neuron.2004.12.015. [DOI] [PubMed] [Google Scholar]
- 18.Liu Y, et al. Mechanisms of compartmentalized expression of Mrg class G-protein-coupled sensory receptors. J Neurosci. 2008;28:125–132. doi: 10.1523/JNEUROSCI.4472-07.2008. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 19.Sun YG, Chen ZF. A gastrin-releasing peptide receptor mediates the itch sensation in the spinal cord. Nature. 2007;448:700–703. doi: 10.1038/nature06029. [DOI] [PubMed] [Google Scholar]
- 20.Sun YG, et al. Cellular basis of itch sensation. Science. 2009;325:1531–1534. doi: 10.1126/science.1174868. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 21.Barth AL, Gerkin RC, Dean KL. Alteration of neuronal firing properties after in vivo experience in a FosGFP transgenic mouse. J Neurosci. 2004;24:6466–6475. doi: 10.1523/JNEUROSCI.4737-03.2004. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 22.Ma C, Donnelly DF, LaMotte RH. In vivo visualization and functional characterization of primary somatic neurons. J Neurosci Methods. 2010;191:60–65. doi: 10.1016/j.jneumeth.2010.06.010. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 23.Shinohara T, et al. Identification of a G protein-coupled receptor specifically responsive to beta-alanine. J Biol Chem. 2004;279:23559–23564. doi: 10.1074/jbc.M314240200. [DOI] [PubMed] [Google Scholar]
- 24.Abrahamsen B, et al. The cell and molecular basis of mechanical, cold, and inflammatory pain. Science. 2008;321:702–705. doi: 10.1126/science.1156916. [DOI] [PubMed] [Google Scholar]
- 25.Naglich JG, Metherall JE, Russell DW, Eidels L. Expression cloning of a diphtheria toxin receptor: identity with a heparin-binding EGF-like growth factor precursor. Cell. 1992;69:1051–1061. doi: 10.1016/0092-8674(92)90623-k. [DOI] [PubMed] [Google Scholar]
- 26.Saito M, et al. Diphtheria toxin receptor-mediated conditional and targeted cell ablation in transgenic mice. Nat Biotechnol. 2001;19:746–750. doi: 10.1038/90795. [DOI] [PubMed] [Google Scholar]
- 27.Buch T, et al. A Cre-inducible diphtheria toxin receptor mediates cell lineage ablation after toxin administration. Nat Methods. 2005;2:419–426. doi: 10.1038/nmeth762. [DOI] [PubMed] [Google Scholar]
- 28.Imamachi N, et al. TRPV1-expressing primary afferents generate behavioral responses to pruritogens via multiple mechanisms. Proc Natl Acad Sci U S A. 2009;106:11330–11335. doi: 10.1073/pnas.0905605106. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 29.Di Nardo A, Wertz P, Giannetti A, Seidenari S. Ceramide and cholesterol composition of the skin of patients with atopic dermatitis. Acta Derm Venereol. 1998;78:27–30. doi: 10.1080/00015559850135788. [DOI] [PubMed] [Google Scholar]
- 30.Krajnik M, Zylicz Z. Understanding pruritus in systemic disease. J Pain Symptom Manage. 2001;21:151–168. doi: 10.1016/s0885-3924(00)00256-6. [DOI] [PubMed] [Google Scholar]
- 31.Miyamoto T, Nojima H, Shinkado T, Nakahashi T, Kuraishi Y. Itch-associated response induced by experimental dry skin in mice. Jpn J Pharmacol. 2002;88:285–292. doi: 10.1254/jjp.88.285. [DOI] [PubMed] [Google Scholar]
- 32.Saint-Mezard P, et al. Allergic contact dermatitis. Eur J Dermatol. 2004;14:284–295. [PubMed] [Google Scholar]
- 33.Skoner DP. Allergic rhinitis: definition, epidemiology, pathophysiology, detection, and diagnosis. J Allergy Clin Immunol. 2001;108:S2–S8. doi: 10.1067/mai.2001.115569. [DOI] [PubMed] [Google Scholar]
- 34.Ono SJ, Abelson MB. Allergic conjunctivitis: update on pathophysiology and prospects for future treatment. J Allergy Clin Immunol. 2005;115:118–122. doi: 10.1016/j.jaci.2004.10.042. [DOI] [PubMed] [Google Scholar]
- 35.Shimada SG, LaMotte RH. Behavioral differentiation between itch and pain in mouse. Pain. 2008;139:681–687. doi: 10.1016/j.pain.2008.08.002. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 36.Liu Q, et al. Mechanisms of Itch Evoked by beta-Alanine. J Neurosci. 2012;32:14532–14537. doi: 10.1523/JNEUROSCI.3509-12.2012. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 37.Arenkiel BR, Klein ME, Davison IG, Katz LC, Ehlers MD. Genetic control of neuronal activity in mice conditionally expressing TRPV1. Nat Methods. 2008;5:299–302. doi: 10.1038/nmeth.1190. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 38.Guler AD, et al. Transient activation of specific neurons in mice by selective expression of the capsaicin receptor. Nat Commun. 2012;3:746. doi: 10.1038/ncomms1749. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 39.Caterina MJ, et al. Impaired nociception and pain sensation in mice lacking the capsaicin receptor. Science. 2000;288:306–313. doi: 10.1126/science.288.5464.306. [DOI] [PubMed] [Google Scholar]
- 40.Sikand P, Shimada SG, Green BG, LaMotte RH. Sensory responses to injection and punctate application of capsaicin and histamine to the skin. Pain. 2011;152:2485–2494. doi: 10.1016/j.pain.2011.06.001. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 41.Sikand P, Shimada SG, Green BG, LaMotte RH. Similar itch and nociceptive sensations evoked by punctate cutaneous application of capsaicin, histamine and cowhage. Pain. 2009;144:66–75. doi: 10.1016/j.pain.2009.03.001. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 42.Green BG. Spatial summation of chemical irritation and itch produced by topical application of capsaicin. Percept Psychophys. 1990;48:12–18. doi: 10.3758/bf03205007. [DOI] [PubMed] [Google Scholar]
- 43.McQueen DS, Noble MA, Bond SM. Endothelin-1 activates ETA receptors to cause reflex scratching in BALB/c mice. Br J Pharmacol. 2007;151:278–284. doi: 10.1038/sj.bjp.0707216. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 44.Yamaguchi T, Nagasawa T, Satoh M, Kuraishi Y. Itch-associated response induced by intradermal serotonin through 5-HT2 receptors in mice. Neurosci Res. 1999;35:77–83. doi: 10.1016/s0168-0102(99)00070-x. [DOI] [PubMed] [Google Scholar]
- 45.Finger S, Wade NJ. The neuroscience of Helmholtz and the theories of Johannes Muller. Part 2: Sensation and perception. J Hist Neurosci. 2002;11:234–254. doi: 10.1076/jhin.11.3.234.10392. [DOI] [PubMed] [Google Scholar]
- 46.McMahon SB, Koltzenburg M. Itching for an explanation. Trends Neurosci. 1992;15:497–501. doi: 10.1016/0166-2236(92)90102-e. [DOI] [PubMed] [Google Scholar]
- 47.Lagerstrom MC, et al. VGLUT2-dependent sensory neurons in the TRPV1 population regulate pain and itch. Neuron. 2010;68:529–542. doi: 10.1016/j.neuron.2010.09.016. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 48.Ross SE, et al. Loss of inhibitory interneurons in the dorsal spinal cord and elevated itch in Bhlhb5 mutant mice. Neuron. 2010;65:886–898. doi: 10.1016/j.neuron.2010.02.025. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 49.Liu Y, et al. VGLUT2-dependent glutamate release from nociceptors is required to sense pain and suppress itch. Neuron. 2010;68:543–556. doi: 10.1016/j.neuron.2010.09.008. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 50.Davidson S, Giesler GJ. The multiple pathways for itch and their interactions with pain. Trends Neurosci. 2010;33:550–558. doi: 10.1016/j.tins.2010.09.002. [DOI] [PMC free article] [PubMed] [Google Scholar]
Associated Data
This section collects any data citations, data availability statements, or supplementary materials included in this article.


