Summary
Background
There are only few reports about the use of bone marrow stromal stem cells (BMSCs) for the treatment of traumatic liver injury. This study aimed to study the therapeutic effect of fluorescence-labeled BMSCs administered to rats subject to traumatic liver injury.
Material/Methods
Male SD rats with a 70% resection of the liver were injected with feridex-labeled BMSCs which could be induced to functional hepatocytes in vitro. Liver function was assayed and the liver scanned by 1.5-T MRI at 12 hrs and on days 1, 3, 5, 7, and 14 post-operation. The pathological changes of liver sections were monitored.
Results
The serum levels of alanine aminotransferase, aspartate aminotransferase, alkaline phosphatase, direct bilirubin, and total bilirubin in the transplantation group were significantly lower than the control group. The MRI showed rats of the transplantation group had an oval low signal area at 12 hr after operation; the low signal range gradually expanded and the signal intensity gradually decreased over 14 days after operation. The low signal range in the control group disappeared 12 hr after the operation. After Prussian blue staining, rats of the transplantation group contained blue granules with no significant hypertrophy or edema in hepatocytes, while the control group showed no blue granules with significant hypertrophy and edema.
Conclusions
The BMSCs transplanted into the injured rat liver gradually migrate to the surrounding liver tissue and partially repair the liver surgical injury in rats. BMSCs may represent an effective therapeutic approach for acute liver injury.
Keywords: bone marrow stromal stem cells, traumatic liver injury, cell transplantation, MRI, rat
Background
Liver injury, which occurs in 62.5% of patients suffering abdominal trauma, is the most common cause of death associated with abdominal trauma. In the United States, liver trauma accounts only for 15–20% of blunt abdominal injuries, but is responsible for more than 50% of deaths from abdominal trauma [1]. A recent study reported the mortality rate of liver injury at 9%, partially due to the requirement of liver resection in 30% of liver injuries [2]. Orthotopic liver transplantation is the most effective treatment for severe liver injury, but the widespread clinical application of liver transplantation is restricted by the shortage of available donor livers and multiple postoperative complications. Stem cell therapy has been considered a viable treatment option for liver injury in the future. By eliminated the need for a donor organ, while having the potential to promote liver regeneration with fewer complications than operation, stem cell therapy has raised great hope for the improvement of liver injury treatment.
One of the hallmark features of the liver is its remarkable ability to maintain constant size despite injury. Although the precise molecular mechanisms that modulate liver regrowth are not entirely clear, the ability of the liver to quickly regain cells after injury is well documented [3]. Natural repair of liver is mainly dependent on endogenous cell pools, including hepatocytes, hepatic progenitor cells (HPCs)/oval cells (OCs), and bone marrow stromal stem cells. The role of hepatocyte replication and hepatic progenitor cells in liver regeneration has been researched in great detail. It has been shown that hepatocytes have the ability to replicate and contribute to the regrowth of the liver [4–6]. Hepatic progenitor cells (of the Canal of Hering) are the equivalent of oval cells in a rat model and have also been shown to generate large numbers of hepatocytes in liver regrowth [7–9]. Though evidence has shown differentiation of bone marrow stromal stem cells (BMSCs) into hepatocytes, the extent of the role of BMSCs in liver regeneration and repair is still uncertain. Sources of exogenous stem/progenitor cells that are currently under investigation in the context of repair of liver injury include embryonic stem cells, bone marrow-derived mesenchymal stem cells, fat-derived mesenchymal stem cells, fetal annex stem cells and endothelial progenitor cells (EPCs) [10–15].
BMSCs have been shown to differentiate into hepatocytes in vivo and in vitro under certain conditions [16–19]. MSCs are multipotent adult stem cells that are easily obtained from BM aspirates and expanded into large quantities in vitro. Primary BM-derived stem cells can undergo a process of differentiation; these differentiated cells express various hepatocyte-specific markers and have hepatocyte-specific bioactivities, including urea production, albumin secretion and glycogen storage.
There are a number of studies on the application of BMSCs for the treatment of chronic liver diseases, but only few reports about the use of BMSCs for the treatment of traumatic liver injury. Abdel Aziz et al. [20] reported that injection of BMSCs into rats with CCl4 induced liver injury resulted in protection against the fibrotic process through minimization of collagen deposition. In addition, this study demonstrated the ability of BMSCs to differentiate into hepatocytes. Kuo et al., reported that transplantation of different doses of BMSCs into rat spleen resulted in BMSC differentiation into functional hepatocytes, rescuing rats from liver failure [21].
This study aimed to investigate the effect of BMSCs in liver repair following traumatic injury in rats. We transplanted feridex-labeled BMSCs into rats with 70% excision of the total liver volume. Feridex-labeling of BMSCs does not alter biochemical or hematologic measures of organ function [22]. The indicators of rat liver function were assayed and migration of BMSCs in the rat livers was monitored by 1.5-T superconducting MR imaging equipment and histological observation. Based on the literature available on the efficacy of BMSCs in chronic liver injury, we believe BMSCs will promote liver regeneration and differentiate into functional hepatocytes, thus increasing liver functionality.
Material and Methods
Ethics statement
All animal experiments were conducted by Drs Xiaowu Chen and Dajian Zhu in the Laboratory Animal Center at Southern Medical University, Guangzhou, China. The procedure was approved by the Ethics Committee of Southern Medical University, Guangzhou, China [Permit No.: SCXK (Yue) 2006-0015].
Animals
Male and female Sprague-Dawley (SD) rats aged 5–6 weeks (weighing 100–120 g) were used as BMSC donors (purchased from Sun Yat-sen University Experimental Animal Center, Guangzhou, China). Male SD rats aged eight weeks were used as BMSC recipients.
Isolation and culture of rat BMSCs
BMSCs from SD rats were isolated and cultured as per an established protocol [23]. Five week old rats were anesthetized with 8% chloral hydrate (4.0 ml/kg); the femurs and tibiae of the rats were excised and the soft connective tissue removed. The two ends of the femurs and tibiae were excised and the cells of the bone marrow were harvested by flushing the bone marrow cavity with complete culture medium. The extract was filtered with a 200 mesh filter, centrifuged for 5min at 1,000 × g, resuspended in media, and inoculated into a 25-cm2 flask containing Dulbecco’s modified Eagle’s medium with low glucose (DMEM-LG, GBICO corp., America) supplemented with 10% fetal bovine serum (FBS, BIOIND Inc., Israel) at 37°C in 5% CO2. The medium in the culture flask was replaced with the same volume of fresh 48 hrs later, then replaced every 3–5 days when adherent cells reached 70–80% confluence. Cell collection was completed with 0.25% trypsin-EDTA (GBICO) treatment and subculturing using 25-cm2 flasks.
Feridex-labeling of rat BMSCs
Cultured rat BMSCs were labeled with superparamagnetic nanoparticles as previously described [24,25]. BMSCs (third passage) were collected by 0.25% trypsin-EDTA and complete culture medium containing 10% FBS. The resulting mixture was centrifuged for 5 min at 1,000 × g, washed with phosphate-buffered saline (PBS) twice, and resuspended in 1 ml PBS. Finally, the cells were counted and separated into two aliquots of 1×106 cells: one for flow cytometry analysis with anti-rat CD29, CD44, CD34, or CD45 antibodies (Pharmingen Inc., San Diego, CA) and the other for feridex labeling. The other sample was centrifuged again, resuspended, and inoculated for 12 hr into a 25-cm2 flask containing complete culture medium with a combination of poly-L-lysine at 0.75 μg/ml (from Sigma-Aldrich Co., St Louis, MO) and feridex at 11.2–19.6 μg/ml (from Pharmagenesis Inc., Palo Alto, CA). The feridex-labeled BMSCs were collected using the same method as described above, followed by centrifugation for 5 min at 1,000 × g, and fixation by 2.5% glutaraldehyde at 4°C for 3 days. The intracytoplasmic iron particles within the cells were evaluated with a 60-kV electron microscopy and Prussian blue staining.
Cytotoxicity assay
The cytotoxicity of feridex labeling to primary rat BMSCs was determined using the 3-(4,5-dimethylthiazol-2-yl)-2,5-diphenyltetrazonium bromide (MTT) assay. BMSCs subject to feridex labeling were incubated for 1, 2, 3, 4 or 5 weeks and 20 μl MTT reagent (5.0 mg/ml) was added to each well after removal of medium, and cells incubated for a further 4 h at 37°C. The absorbance of formazan, a metabolite of MTT, in the resulting solution was photometrically measured at a wavelength of 595 nm using a microplate reader (Tecan Instruments Inc., Research Triangle Park, NC).
Induction of differentiation of BMSCs into hepatocyte-like cells
Feridex-labeled BMSCs were incubated with hepatocyte growth factor (HGF) at 20 ng/ml and epidermal growth factor (EGF) at 10 ng/ml for 7 days.
Construction of liver injury rat model with 70% hepatectomy
An acute liver injury rat model with 70% hepatectomy was constructed as described previously [26,27]. 40 SD rats were equally divided into transplantation group and control group randomly. All of the rats had been prohibited from eating and drinking for 12 hr, and were anesthetized with 8% chloral hydrate (4 ml/kg) via injection into peritoneal cavity. The operation was started after tension in rat limb muscles vanished (approximately 3 min). The rat was positioned in the supine position; the fur on the abdomen was scraped off and the abdominal skin was disinfected with alcohol solution of iodine and 75% alcohol. A transverse incision approximately 2 cm was made in the skin just beneath the xiphisternum, and the musculature of the anterior abdominal wall and the peritoneal wall were then incised gradually to expose the liver. Two parts of the median lobe and the left lobe of the liver (which were totally 70% of the liver volume) were divided and a loop of silk suture was maneuvered around the lobes and tied at their narrow pedicle; these portions were then excised.
BMSC transplantation in liver injury rats
Our preliminary study has compared the effect of three transplantation methods in liver injury rats: local liver injection, portal vein injection and femoral vein injection). Our results showed that the local liver injection gave the best therapeutic effect compared to other two approaches (data not shown). In this study, 1.0 ml 1×106/ml feridex-labeled BMSCs suspension was injected into the right lobe slowly in the transplantation group within 15 min; the rats of control group were injected with 1.0 ml saline in the same position. The peritoneal cavity was flushed with metronidazole and saline. In order to close the peritoneal cavity layer by layer with 3-0 silk, the rats were given 4.0 ml of 40,000 U penicillin and 5% glucose by intravenous injection within the first 3 days after operation, twice a day, with free access to water and food for 6 hr.
MRI scanning in liver injury rats with or without BMSC transplantation
We have developed an MRI method to track the BMSCs in the liver in vivo[28]. The rat livers were examined by 1.5-T MR imaging equipment (type: EXCELART/P3, Toshiba) 12 hr and on days 1, 3, 5, 7, 14 after operation. All of the rats that received cells and saline were anesthetized by chloral hydrate (2.0 ml/kg) before examination. The rats were placed in prone position in the knee coil, all the images of rat livers scanned by MRI consisted of T1-weighted images (403/15, repetition time msec/echo time msec) and T2-weighted images (4000/100, repetition time msec/echo time msec).
Assessment of hepatic function in liver injury rats with or without BMSC transplantation
Blood samples were obtained from each rat of the two groups pre-operation, at 12 hr, and on days 1, 3, 5, 7, and 14 after operation. The samples were centrifuged for 10 min at 1,000 × g and 1.0 ml serum was collected, the levels of the liver function indicators, such as alanine transaminase (ALT), aspartate aminotransferase (AST), alkaline phosphatase (ALP), total bilirubin (TB) and direct bilirubin (DBIL), of both the transplantation group and control group were analyzed by an Olympus AU2700 automatic biochemical analyzer (Tokyo, Japan).
Flow cytometry
For each sample, BMSCs were isolated by centrifugation at 1,000 × g for 6 min, and transferred into screw-capped tubes and stored at −80°C. Flow cytometry was performed (i.e. CD29, CD34, CD44 and CD45).
Reverse transcription-polymerase chain reaction (RT-PCR) for the detection of albumin (ALB) and α-fetoprotein (AFP)
The expression and ALB and AFP in BMSCs on days 3, 5, 7, 21 and 28 after induction with growth hormones was determined by RT-PCR amplification using Access RT-PCR System according to the manufacturer’s instructions. The housekeeping gene GADPH was used in the same reaction as a reference for standardization of the procedure. The primer sequences for ALB were as follows: 5′-ATA CAC CCA GAA AGC ACCTC-3′; and 3′-CAC GAA TTG TGC GAATG-5′. The primer sequences for AFP were: 5′-AAC AGC AGA GTG CTG CAAAC-3′, and 3′-AGG TTT CGT CCC TCA GAAAG-3′. The resultant PCR products were separated by 1.0% agarose gel electrophoresis and visualized by means of ethidium bromide staining using a GDS8000 image analysis system (Syngene, Cambridge, the UK). The levels of ALB or AFP mRNA were normalized to that of GADPH present in the same sample.
Histological examination
Rat livers of the two groups were harvested on the 14th day after operation, fixed with 10% formalin, and embedded with paraffin. Histological analysis of liver tissues was conducted by serial tissue section and Prussian blue staining to identify intracytoplasmic iron particles. The liver sections were observed under the light microscopy (×40), and the blue granule cells in each field were calculated for five random fields.
Statistical analysis
Data is expressed as mean ±SD. All analyses were performed with SPSS13.0 software. All variables were determined by a repeated measure analysis of variance (ANOVA) between the transplantation group and the control group, followed by an F-test. Results were considered statistically significant at when P<0.05.
Results
Morphology of cultured rat BMSCs
The adherent cells had a colony-like distribution in 3 days after inoculation into the flask under phase-contrast microscopy. Typically, about 70–90% confluence was reached by days 7–9. Cells of the third passage had a typical fibroblast-like morphology and good refractivity. The profile of the cells could be clearly seen, but the nuclei were not distinctly visible (Figure 1).
Figure 1.
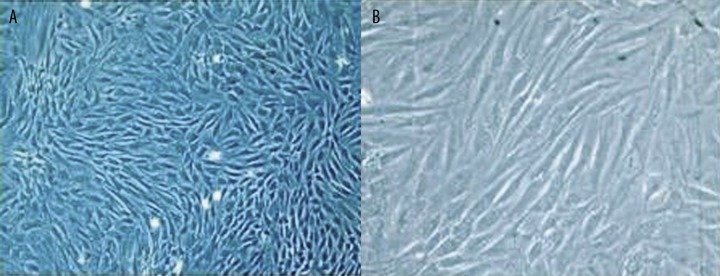
Cultured rat BMSCs of the third passage under phase-contrast microscopy. (A) ×100 magnification; and (B) ×200 magnification.
Surface markers of isolated rat BMSCs and feridex labeling efficiency
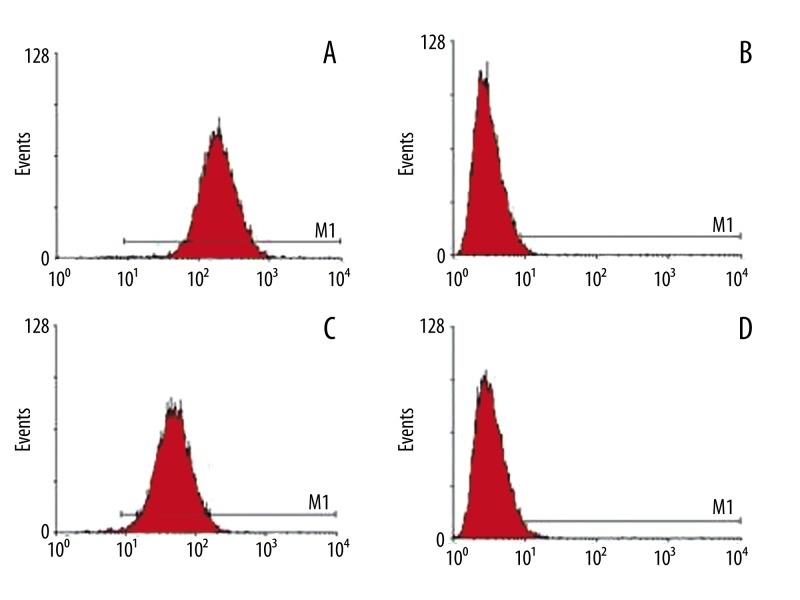
The BMSCs isolated from rat bone marrow were positive for CD29 (99.7%) and CD44 (99.4%) and negative for CD34 (1.3%) and CD45 (2.2%) by flow cytometry (Figure 2), which is in agreement with the results from a previous study [29]. The Prussian blue staining of the feridex-labeled BMSCs showed that iron particles were blue (Figure 3A). When the concentration of feridex was 11.2, 14.0, 16.8 and 19.6 μg/ml, the labeling efficiency for rat BMSCs was 71, 83, 91 and 96%, respectively. Electron micrographs of feridex-labeled BMSCs showed that endosomal vesicles contained iron particles, the area outlined shows high-density granule aspect of endosomes (Figure 3B). Labeling of feridex at concentrations ≤16.8 μg/ml over 5 weeks did not show significant cytotoxicity towards BMSCs (P>0.05), but feridex at 19.6 μg/ml showed significant cytotoxicity to BMSCs (>25%, P<0.05).
Figure 2.
Data from the flow cytometric analysis. (A) CD29; (B) CD34; (C) CD44; and (D) CD45.
Figure 3.
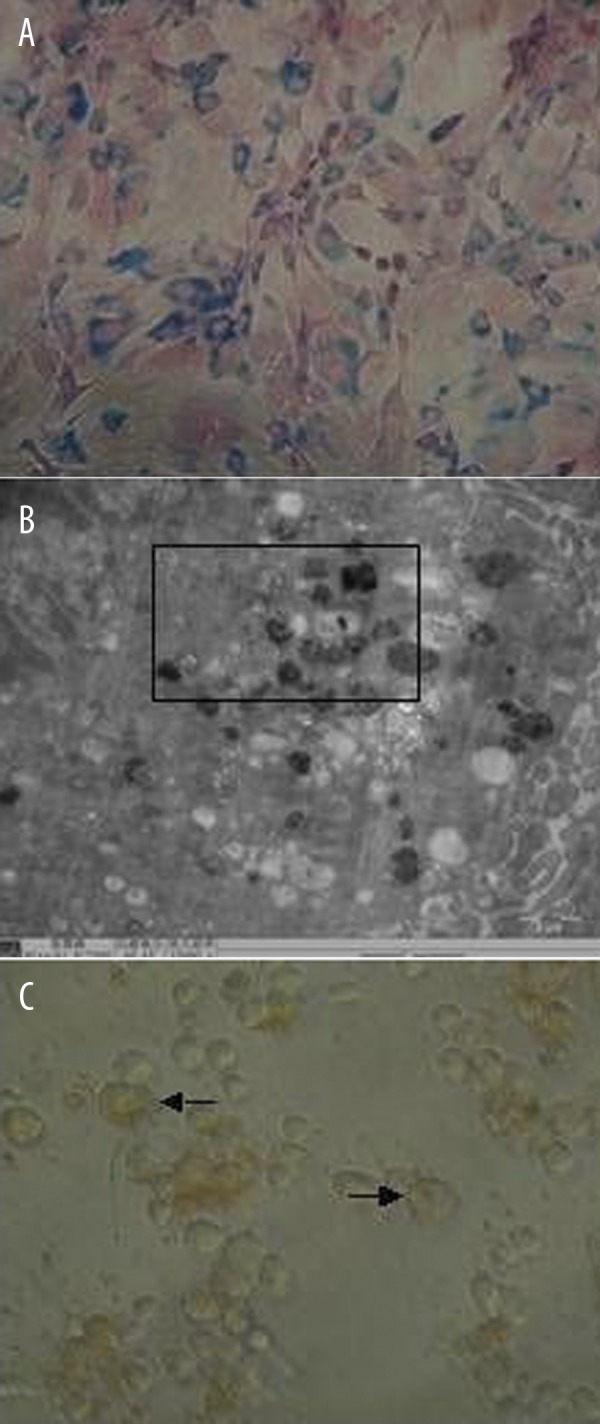
Prussian blue staining of feridex-labeled BMSCs ((A) ×200 magnification), feridex-labeled BMSCs under the electron microscopy ((B) ×15,000 magnification), and BMSCs after incubation with HGF and EGF for 14 days (× 400 magnification).
Differentiation of rat BMSCs into functional hepatocyte-like cells in vitro
After incubation of BMSCs with HGF and EGF for 7 days, the cells became triangle-like, polygon-like, or irregular shape. After 14 days, the cells were oval-like and formed clusters (Figure 3C); after 21 days, there was increased number of oval-shaped cells.
When unlabeled and feridex-labeled BMRCs were incubated with HGF and EGF, there was no detectable AFP mRNA after 3–5 days. AFT mRNA was detectable on day 7 and enhanced on day 14 but declined on day 28. For ALB mRNA, we observed its expression on day 14 and remained until day 28. When BMSCs were labeled with feridex at 19.6 μg/ml, the expression of AFP and ABL was significantly decreased compared to those cells labeled with feridex at concentration ≤16.8 μg/ml.
Effect of BMSC transplantation on liver function in rats with 70% hepatectomy
ALT levels of transplantation group were lower than control group significantly on days 1, 3, 5 post-operation (P<0.05, Table 1). AST and TB levels of transplantation group were lower than control group significantly at 12 hr and on days 1, 3, 5 post-operation (P<0.05). ALP and DBIL levels of transplantation group were lower than the control group significantly 12 hr and on days 1, 3, 5, 7 post-operation (P<0.05). There was no statistically significant difference between indicators of the two groups 14 days after operation (P>0.05) (Table 1).
Table 1.
Effect of BMSC transplantation on serum levels of ALT, AST, ALP, total bilirubin (TB) and direct bilirubin (DBIL) in liver-resected rats (mean ±SD).
| Treatment | Pre-transplantation | Time after operation | |||||
|---|---|---|---|---|---|---|---|
| 12 hr | 1 day | 3 days | 5 days | 7 days | 14 days | ||
| ALT (U/L) | |||||||
| BMSC-treated | 29±5 | 579±90 | 253±49c | 161±43c | 33±6c | 28±7 | 32±5 |
| Sham | 32±3 | 653±118 | 365±38 | 282±40 | 50±6 | 33±6 | 31±3 |
| AST (U/L) | |||||||
| BMSC-treated | 123±15 | 1,155±200a | 542±152c | 119±21c | 114±15b | 113±15 | 110±12 |
| Sham | 117±12 | 1,293±166 | 879±120 | 165±17 | 127±10 | 120±14 | 119±21 |
| ALP (U/L) | |||||||
| BMSCs | 177±37 | 203±27a | 231±33a | 238±34c | 199±45b | 187±40b | 176±43 |
| Sham | 185±40 | 224±29 | 259±32 | 307±44 | 238±39 | 219±32 | 187±18 |
| TB (μmol/L) | |||||||
| BMSC-treated | 0.97±0.38 | 6.54±0.52c | 4.91±0.58c | 3.02±0.73c | 1.12±0.29c | 0.97±0.23 | 0.99±0.21 |
| Sham | 1.19±0.39 | 6.87±0.53 | 5.49±0.70 | 4.70±0.67 | 1.99±0.35 | 0.90±0.13 | 0.94±0.22 |
| DBIL (μmol/L) | |||||||
| BMSC-treated | 0.29±0.11 | 4.79±0.38c | 3.85±0.67c | 2.93±0.40c | 1.85±0.32c | 0.34±0.08c | 0.34±0.13 |
| Sham | 0.28±0.07 | 5.70±0.59 | 4.68±0.63 | 3.63±0.26 | 2.66±0.26 | 1.01±0.12 | 0.32±0.09 |
P<0.05, BMSC-transplanted vs. sham group;
P<0.01, BMSC-transplanted vs. sham group;
P<0.001, BMSC-transplanted vs. sham group.
Tracking of BMSCs in rat liver by MRI imaging technique
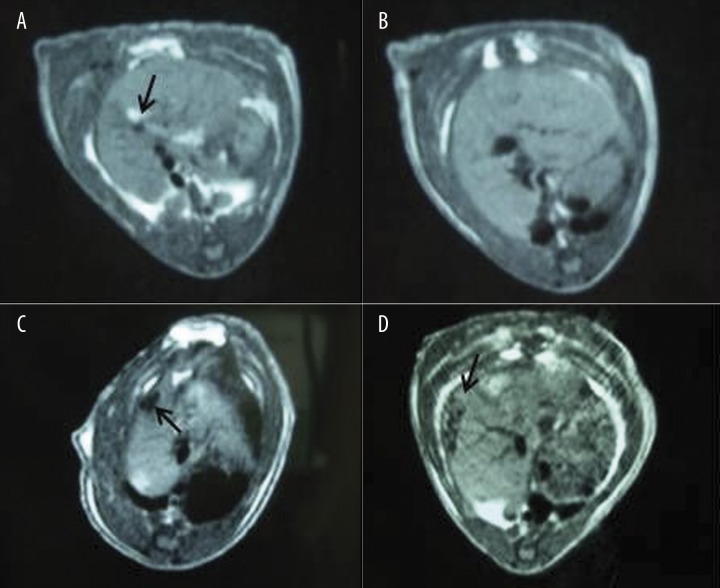
In rats of control group, MRI showed an oval high signal area about 2×1 mm at the site of the liver transplant 12 hr after operation, the oval high signal area was not observed after this viewing (Figure 4A, B). In rats of transplantation group, an oval low signal area of about 3×5 mm was detected by MRI in the site of the liver transplant at 12 hr after operation. The low signal area gradually expanded through the course of the study, as the area expanded to 5×7 mm 14 days post-operation (Figure 4C, D). At the same time, the signal intensity gradually decreased from 12 hr to day 14 after operation.
Figure 4.
MRI images of the rat live 12 hr and 7 days after operation. (A) sham, 12 hr after operation; (B) sham, 7 days after operation; (C) BMSC-transplanted, 12 hr after treatment; and (D) BMSC-transplanted, 7 days after treatment.
Effect of BMSC transplantation on liver histology and BMSC counting in rats with 70%-hepatectomy
In the sham-operated rats, the liver was red-brown with thin lobes and the edge of the liver was sharp (Figure 5A). In the BMSC-transplanted rats, the liver size was larger than before operation with enlarged lobes and stunt edge (Figure 5B). No blue granule cells were observed in the liver of the control group, but significant hypertrophy and edema of the liver cells was apparent (Figure 5C). In rats of the transplantation group, cells with blue granules were distributed in the liver sinusoids, with an average of 40–50 cells in each field. No significant hypertrophy and edema of liver cells was observed (Figure 5D).
Figure 5.
(A) the liver lobes in the sham-operated rats; (B) the liver lobes in BMSC-transplanted rats; (C) prussian blue staining of the rat liver section on 14 days after operation in the sham-operated group; and (D) prussian blue staining of the rat liver section on 14 days after operation in the BMSC-transplanted rats.
Discussion
To track the in vivo paths and disposition of BMSCs from the donors, we labeled these cells with feridex which is often used in clinical settings as a magnetic resonance imaging contrast media. As aqueous colloid of superparamagnetic iron oxide associated with dextran, feridex at 19.6 μg/ml has some cytotoxicity to BMSCs as shown in our in vitro assay. Other studies have found that feridex at concentrations >22.4 or 25 μg/ml showed significant cytotoxicity towards BMSCs [30,31]. High concentrations of feridex also reduce the differentiation of BMSCs into hepatocytes [31]. Thus, we have chosen 16.8 μg/ml as the optimal concentration for feridex labeling of BMSCs.
Regenerative stimuli, such as the partial hepatectomy performed in this experiment, causes most hepatocytes to undergo symmetrical mitosis [32]. However, as suggested by the high mortality rate of liver injury, this mechanism is often not enough to regenerate the liver to functional levels. When this type of regeneration fails, a second mechanism based on stem cells from the canals of Hering can play a role [33,34]. Treatment of liver injury due with exogenous BMSCs is thought to effect liver regeneration through direct differentiation of these BMSCs into hepatocytes, as well as through delivery of growth factors that promote liver regeneration and cell fusion [35,36]. In this study, MRI scanning showed that treatment with exogenous BMSCs does in fact cause gradual BMSC migration to the surrounding liver tissue from the spot of transplantation. In addition, histological observation showed that BMSCs distribute themselves and eventually settle in the liver sinusoids.
The results support our hypothesis that BMSC treatment promotes regeneration resulting in an increase in liver function, though the exact mechanism is not fully elucidated. The levels of the liver functionality enzymes in the BMSC transplantation group were significantly lower than that of the control group at the same period, suggesting damage to liver functionality of the transplantation group was less than damage to the control group. The transplantation group’s lack of pathological changes when compared to the tightly packed, hypertrophic, and hydropic hepatocytes of the control group also indicate a healthier, better functioning rat liver in the transplantation group after treatment.
Our findings have showed that BMSCs recovered rat liver functions and ameliorated the pathological changes in traumatic liver injury. However, the mechanism of how BMSCs migrate and repair injured liver is still unknown. Our in vitro study has shown that BMSCs can be induced to functional hepatocyte-like cells in the presence of HGF and EGF. It can be anticipated that the injected BMSCs might differentiate into functional hepatocytes in vivo and thus improve the liver function. It has been reported that laminin in the liver induces transplanted cells to the hepatic parenchyma, resulting in adherence to these molecules [37]. Generally, the laminins reside in the portal and interlobular veins and would increase in number approximately 3 days after partial hepatectomy. Thus, laminin might play an important role in the biological process of migration and differentiation of BMSCs [38].
The chemokine stromal cell-derived factor-1 (SDF-1), which attracts human and murine progenitors, is expressed by liver bile duct epithelium and mediated by its receptor CXCR4, which is expressed by endothelial and various stromal cell types in the bone marrow. SDF-1 and its receptor CXCR4 participate in the homing of BMSCs [39,40], referred to as the SDF-1/CXCR4 pathway. The serum from rats with injured livers may enable BMSCs expression of α-fetoprotein and albumin mRNA [41], which we believe is closely tied to BMSC liver injury repair. Additionally, participation of hepatic mast cells the regeneration of rat liver may contribute to the overall mechanism of recovery after partial hepatectomy [42].
Conclusions
The usage of BM-derived stem cells for treatment of liver disease might sidestep many obstacles, such as ethical concerns and risks of rejection. In the future, perhaps localization of BMSCs to the canal of Hering (a stem cell “niche”) might provide even more appropriate conditions for stem cell growth and differentiation. This paper has shown the therapeutic effect of BMSC treatment on liver function in traumatic liver injury. As the mechanism behind BMSC treatment is expounded, more powerful stem cell treatments for liver injury using different delivery and targeting techniques can be developed.
Footnotes
Source of support: This study was supported by a grant from the Natural Science Foundation of Guangdong Province (NO. 06008278), China
References
- 1.Murray CJ, Lopez AD. Alternative projections of mortality and disability by cause 1990–2020: Global Burden of Disease Study. Lancet. 1997;349:1498–504. doi: 10.1016/S0140-6736(96)07492-2. [DOI] [PubMed] [Google Scholar]
- 2.Polanco P, Leon S, Pineda J, et al. Hepatic resection in the management of complex injury to the liver. J Trauma. 2008;65:1264–69. doi: 10.1097/TA.0b013e3181904749. discussion 1269–70. [DOI] [PubMed] [Google Scholar]
- 3.Duncan AW, Dorrell C, Grompe M. Stem Cells and Liver Regeneration. Gastroenterology. 2009;137:466–81. doi: 10.1053/j.gastro.2009.05.044. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 4.NLRB, Farmer S. Liver Regeneration Following Partial Hepatectomy: genes and metabolism. In: AS, AMD, editors. Liver Growth and Repair. London: Chapman & Hall; 1998. [Google Scholar]
- 5.Fausto N, Campbell JS, Riehle KJ. Liver regeneration. Hepatology. 2006;43:S45–53. doi: 10.1002/hep.20969. [DOI] [PubMed] [Google Scholar]
- 6.Michalopoulos GK, DeFrances MC. Liver regeneration. Science. 1997;276:60–66. doi: 10.1126/science.276.5309.60. [DOI] [PubMed] [Google Scholar]
- 7.Strain AJ, Crosby HA, Nijjar S, et al. Human liver-derived stem cells. Semin Liver Dis. 2003;23:373–84. doi: 10.1055/s-2004-815563. [DOI] [PubMed] [Google Scholar]
- 8.Sell S. Heterogeneity and plasticity of hepatocyte lineage cells. Hepatology United States. 2001:738–50. doi: 10.1053/jhep.2001.21900. [DOI] [PubMed] [Google Scholar]
- 9.Thorgeirsson SS, Grisham JW. Overview of recent experimental studies on liver stem cells. Semin Liver Dis. 2003;23:303–12. doi: 10.1055/s-2004-815559. [DOI] [PubMed] [Google Scholar]
- 10.Aurich I, Mueller LP, Aurich H, et al. Functional integration of hepatocytes derived from human mesenchymal stem cells into mouse livers. Gut England. 2007:405–15. doi: 10.1136/gut.2005.090050. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 11.Aurich H, Sgodda M, Kaltwasser P, et al. Hepatocyte differentiation of mesenchymal stem cells from human adipose tissue in vitro promotes hepatic integration in vivo. Gut England. 2009:570–81. doi: 10.1136/gut.2008.154880. [DOI] [PubMed] [Google Scholar]
- 12.Asahina K, Teramoto K, Teraoka H. Embryonic stem cells: hepatic differentiation and regenerative medicine for the treatment of liver disease. Curr Stem Cell Res Ther United Arab Emirates. 2006:139–56. doi: 10.2174/157488806776956878. [DOI] [PubMed] [Google Scholar]
- 13.Banas A, Teratani T, Yamamoto Y, et al. Adipose tissue-derived mesenchymal stem cells as a source of human hepatocytes. Hepatology. 2007;46:219–28. doi: 10.1002/hep.21704. [DOI] [PubMed] [Google Scholar]
- 14.Beaudry P, Hida Y, Udagawa T, et al. Endothelial progenitor cells contribute to accelerated liver regeneration. J Pediatr Surg United States. 2007:1190–98. doi: 10.1016/j.jpedsurg.2007.02.034. [DOI] [PubMed] [Google Scholar]
- 15.Habib HS, Halawa TF, Atta HM. Therapeutic applications of mesenchymal stroma cells in pediatric diseases: current aspects and future perspectives. Med Sci Monit. 2011;17(11):RA233–39. doi: 10.12659/MSM.882036. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 16.Sato Y, Araki H, Kato J, et al. Human mesenchymal stem cells xenografted directly to rat liver are differentiated into human hepatocytes without fusion. Blood. 2005;106:756–63. doi: 10.1182/blood-2005-02-0572. [DOI] [PubMed] [Google Scholar]
- 17.Snykers S, De Kock J, Tamara V, Rogiers V. Hepatic differentiation of mesenchymal stem cells: in vitro strategies. Methods Mol Biol. 2011;698:305–14. doi: 10.1007/978-1-60761-999-4_23. [DOI] [PubMed] [Google Scholar]
- 18.Guguen-Guillouzo C, Corlu A, Guillouzo A. Stem cell-derived hepatocytes and their use in toxicology. Toxicology. 2010;270:3–9. doi: 10.1016/j.tox.2009.09.019. [DOI] [PubMed] [Google Scholar]
- 19.Stock P, Staege MS, Muller LP, et al. Hepatocytes derived from adult stem cells. Transplant Proc. 2008;40:620–23. doi: 10.1016/j.transproceed.2008.01.058. [DOI] [PubMed] [Google Scholar]
- 20.Abdel Aziz MT, Atta HM, Mahfouz S, et al. Therapeutic potential of bone marrow-derived mesenchymal stem cells on experimental liver fibrosis. Clin Biochem. 2007;40:893–99. doi: 10.1016/j.clinbiochem.2007.04.017. [DOI] [PubMed] [Google Scholar]
- 21.Kuo TK, Hung SP, Chuang CH, et al. Stem cell therapy for liver disease: parameters governing the success of using bone marrow mesenchymal stem cells. Gastroenterology. 2008;134:2111–21. 21 e1–3. doi: 10.1053/j.gastro.2008.03.015. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 22.Yocum GT, Wilson LB, Ashari P, et al. Effect of human stem cells labeled with ferumoxides-poly-L-lysine on hematologic and biochemical measurements in rats. Radiology. 2005;235:547–52. doi: 10.1148/radiol.2352040383. [DOI] [PubMed] [Google Scholar]
- 23.Zhang GQ, Fang CH, Chi DZ. Hepatocyte growth factor induces differentiation of adult rat mesenchymal stem cells into a hepatocyte lineage in vitro. Zhonghua Wai Ke Za Zhi. 2005;43:716–20. [PubMed] [Google Scholar]
- 24.Jendelova P, Herynek V, DeCroos J, et al. Imaging the fate of implanted bone marrow stromal cells labeled with superparamagnetic nanoparticles. Magn Reson Med. 2003;50:767–76. doi: 10.1002/mrm.10585. [DOI] [PubMed] [Google Scholar]
- 25.Anderson SA, Glod J, Arbab AS, et al. Noninvasive MR imaging of magnetically labeled stem cells to directly identify neovasculature in a glioma model. Blood. 2005;105:420–25. doi: 10.1182/blood-2004-06-2222. [DOI] [PubMed] [Google Scholar]
- 26.Martins PN, Theruvath TP, Neuhaus P. Rodent models of partial hepatectomies. Liver Int. 2008;28:3–11. doi: 10.1111/j.1478-3231.2007.01628.x. [DOI] [PubMed] [Google Scholar]
- 27.Christ B, Bruckner S, Stock P. Hepatic transplantation of mesenchymal stem cells in rodent animal models. Methods Mol Biol. 2011;698:315–30. doi: 10.1007/978-1-60761-999-4_24. [DOI] [PubMed] [Google Scholar]
- 28.Chen XW, Fang CH, Liu SJ, et al. Tracking of allogenically grafted rat bone marrow stem cells labeled with Feridex in rat liver. J Fourth Mil Med Univ. 2006;27:1956–59. [Google Scholar]
- 29.Pittenger MF, Mackay AM, Beck SC, et al. Multilineage potential of adult human mesenchymal stem cells. Science. 1999;284:143–47. doi: 10.1126/science.284.5411.143. [DOI] [PubMed] [Google Scholar]
- 30.Bulte JW, Douglas T, Witwer B, et al. Magnetodendrimers allow endosomal magnetic labeling and in vivo tracking of stem cells. Nat Biotechnol. 2001;19:1141–47. doi: 10.1038/nbt1201-1141. [DOI] [PubMed] [Google Scholar]
- 31.Arbab AS, Bashaw LA, Miller BR, et al. Characterization of biophysical and metabolic properties of cells labeled with superparamagnetic iron oxide nanoparticles and transfection agent for cellular MR imaging. Radiology. 2003;229:838–46. doi: 10.1148/radiol.2293021215. [DOI] [PubMed] [Google Scholar]
- 32.Overturf K, al-Dhalimy M, Ou CN, et al. Serial transplantation reveals the stem-cell-like regenerative potential of adult mouse hepatocytes. Am J Pathol. 1997;151:1273–80. [PMC free article] [PubMed] [Google Scholar]
- 33.Evarts RP, Nagy P, Nakatsukasa H, et al. In vivo differentiation of rat liver oval cells into hepatocytes. Studies on the proliferation and fate of oval cells in the liver of rats treated with 2-acetylaminofluorene and partial hepatectomy. Cancer Res. 1989;49:1541–47. [PubMed] [Google Scholar]
- 34.Tatematsu M, Ho RH, Kaku T, et al. Studies on the proliferation and fate of oval cells in the liver of rats treated with 2-acetylaminofluorene and partial hepatectomy. Am J Pathol. 1984;114:418–30. [PMC free article] [PubMed] [Google Scholar]
- 35.Fausto N. Liver regeneration and repair: hepatocytes, progenitor cells, and stem cells. Hepatology. 2004;39:1477–87. doi: 10.1002/hep.20214. [DOI] [PubMed] [Google Scholar]
- 36.Kallis YN, Alison MR, Forbes SJ. Bone marrow stem cells and liver disease. Gut England. 2007:716–24. doi: 10.1136/gut.2006.098442. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 37.Carvalho S, Cortez E, Stumbo AC, et al. Laminin expression during bone marrow mononuclear cell transplantation in hepatectomized rats. Cell Biol Int. 2008;32:1014–18. doi: 10.1016/j.cellbi.2008.04.003. [DOI] [PubMed] [Google Scholar]
- 38.Sasaki T, Fassler R, Hohenester E. Laminin: the crux of basement membrane assembly. J Cell Biol. 2004;164:959–63. doi: 10.1083/jcb.200401058. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 39.Kollet O, Shivtiel S, Chen YQ, et al. HGF, SDF-1, and MMP-9 are involved in stress-induced human CD34+ stem cell recruitment to the liver. J Clin Invest. 2003;112:160–69. doi: 10.1172/JCI17902. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 40.Dar A, Kollet O, Lapidot T. Mutual, reciprocal SDF-1/CXCR4 interactions between hematopoietic and bone marrow stromal cells regulate human stem cell migration and development in NOD/SCID chimeric mice. Exp Hematol. 2006;34:967–75. doi: 10.1016/j.exphem.2006.04.002. [DOI] [PubMed] [Google Scholar]
- 41.Ma J, Duan FL, Yan FG, et al. Serum from partial hepatectomy rat and hepatocyte growth factor stimulate bone marrow cell expressing albumin and a-fetoprotein. Zhonghua Gan Zang Bing Za Zhi. 2004;12:410–13. [PubMed] [Google Scholar]
- 42.Zweifel M, Breu K, Matozan K, et al. Restoration of hepatic mast cells and expression of a different mast cell protease phenotype in regenerating rat liver after 70%-hepatectomy. Immunol Cell Biol. 2005;83:587–95. doi: 10.1111/j.1440-1711.2005.01368.x. [DOI] [PubMed] [Google Scholar]