Abstract
The Lyme disease spirochete, Borrelia burgdorferi, encounters many environmental signals as it cycles between the arthropod vector and mammalian hosts, including temperature, pH, and other host factors. To test the possibility that dissolved oxygen modulates gene expression in B. burgdorferi, spirochetes were exposed to differential levels of dissolved oxygen, and distinct alterations were observed at both the transcriptional and translational levels. Specifically NapA, a Dps/Dpr homologue involved in the oxidative stress response in other bacteria, was reduced when B. burgdorferi was grown under oxygen-limiting conditions. In contrast, several immunoreactive proteins were altered when tested with infection-derived sera from different hosts. Specifically, OspC, DbpA, and VlsE were synthesized at greater levels when cells were grown in limiting oxygen, whereas VraA was reduced. The levels of oxygen in the medium did not affect OspA production. Real-time reverse transcription-PCR analysis of RNA isolated from infectious isolates of strains B31 and cN40 indicated that the expression of ospC, dbpA, and vlsE increased while napA expression decreased under dissolved-oxygen-limiting conditions, whereas flaB was not affected. The reverse transcription-PCR results corroborated the immunoblot analyses and indicated that the increase in OspC, DbpA, and VlsE was due to regulation at the transcriptional level of the genes encoding these antigens. These results indicate that dissolved oxygen modulates gene expression in B. burgdorferi and imply that the redox environment may be an additional regulatory cue that spirochetes exploit to adapt to the disparate niches that they occupy in nature.
The spirochetal etiologic agent of Lyme disease, Borrelia burgdorferi, is transmitted to humans and other mammals through the bite of infected Ixodes ticks (reviewed in reference 23). Prior to the ingestion of a blood meal by the ticks, B. burgdorferi is primarily restricted to the midgut, and following a blood meal, the spirochetes migrate to the salivary glands and are ultimately transmitted to mammalian hosts. The zoonotic life cycle between tick vector and various mammalian hosts imposes an implicit requirement for adaptation of B. burgdorferi to these different environmental conditions (1, 5, 6, 9, 31, 34, 38).
One of the environmental signals that B. burgdorferi encounters before and after a tick blood meal is the increase from the ambient temperature of the tick to that of a warm-blooded mammal. Several investigators have shown that the surface lipoproteins OspA and OspC are altered by an experimentally imposed temperature differential (31) as well as by host-adapted conditions, whereby dialysis membranes containing spirochetes were implanted into the peritoneal cavity of rats (1). The paradigm that has emerged is that ospA and ospC are diametrically regulated, with ospA synthesized in unfed ticks (ambient temperature) and ospC expressed concurrent with a blood meal or host adaptation (≈37°C or dialysis membrane implants) (1, 5, 9, 31). More recently, genomics-based approaches have confirmed and extended these observations, indicating that many genes are affected by an increase in temperature and host adaptation, most of which encode proteins of unknown function (5, 19, 25, 27).
Another important variable that B. burgdorferi is exposed to during the various stages of its life cycle is pH. Carroll et al. determined that pH differences (6.0 to 8.0) altered the expression of 37 membrane proteins (6). pH and temperature were analyzed simultaneously, and a reciprocal pattern of regulation was observed, such that one set of genes were preferentially expressed at pH 6.8 and 37°C (including ospC) and the other set were expressed at pHs between 7.5 and 8.0 independent of temperature (including ospA) (38). This analysis also revealed that, in addition to ospC and ospA, several other genes also exhibited ospC-like (ospF, mlp-8 and rpoS) or ospA-like (lp6.6 and p22) expression that could be classified into group 1 and group 2 sets of genes, respectively (38).
A balance in the response to the level of dissolved oxygen is likely to be an important cue for B. burgdorferi because levels of dissolved oxygen vary in the different tissues that B. burgdorferi is known to traffic through during infection (32, 37). Specifically, there is a fourfold difference in dissolved oxygen in mammalian tissues between the dermal layers and bloodstream (32, 37). Understanding how B. burgdorferi responds to different oxygen levels may provide important insights into how these pathogens adapt to these disparate niches as they traffic through mammalian tissues and are able to withstand the toxic effect of reactive oxygen species.
We report here the effect of reduced oxygen on gene expression in B. burgdorferi. These data suggest that the level of dissolved oxygen serves as a signal that modulates gene expression and prepares B. burgdorferi for various stages of its infectious cycle.
MATERIALS AND METHODS
Bacterial strains and growth conditions.
Low-passage, infectious isolates of B. burgdorferi B31 derivative MSK5 (16) and sensu stricto isolates cN40 and 297 were used in this study. For the oxygen depletion studies, B. burgdorferi isolates were grown in complete BSK-II medium supplemented with 6% normal rabbit serum to a density of 1 × 105 per ml and then used to inoculate cultures under conventional microaerophilic conditions (referred to as microaerophilic conditions throughout the remainder of this report) or in BSK-II medium depleted of oxygen (referred to as anaerobic conditions throughout the remainder of this report) at 32°C for 96 h. Depletion of oxygen in the medium was achieved by two methods. Complete BSK-II medium was treated with Oxyrase (Oxyrase, Inc., Mansfield, Ohio) for 4 h at 32°C to achieve an approximate 10-fold reduction in the level of dissolved oxygen (from 0.25 to <0.025 mM/ml), or complete BSK-II medium was subjected to displacement of dissolved oxygen with nitrogen gas to reduce the level of dissolved oxygen from 0.25 to < 0.025 mM/ml (i.e., microaerophilic relative to anaerobic conditions, respectively). In both cases the depletion of dissolved oxygen was monitored with a DO-166 dissolved oxygen probe (Lazar Research Labs, Los Angeles, Calif.).
SDS-PAGE and immunoblotting.
Sodium dodecyl sulfate-polyacrylamide gel electrophoresis (SDS-PAGE) and Western blotting with infection-derived and monospecific antisera were conducted as previously described (16, 33).
RNA extraction.
RNA was extracted with a previously published procedure (25). Briefly, B. burgdorferi cultures were inoculated at a density of 106 per ml, and the cells were grown either microaerophilically or anaerobically to a final density of 4 × 107 to 5 × 107 per ml. RNA was obtained independently from untreated (microaerophilically grown) and treated (anaerobically grown) B. burgdorferi cells, and each resulting sample was assayed in triplicate. RNA was extracted from these samples by resuspending the cell pellets in RNA-Bee (Tel-Test, Inc., Friendswood, Tex.) at a ratio of 0.2 ml of RNA-Bee for every 106 cells. The suspension was then extracted with chloroform and precipitated in isopropanol. The pellets were washed with 75% ethanol, air-dried, and resuspended in diethyl pyrocarbonate-treated water. Samples were incubated with DNase I (DNA-free; Ambion Inc., Austin, Tex.) twice at 37°C for 45 min and quantified spectrophotometrically.
The purified RNA was tested for contaminating DNA by real-time PCR with the flaB primer set (see Table 1). Real-time PCRs were set up with SYBR Green PCR master mix with oligonucleotide primers at a final concentration of 900 nM. Real-time quantitative PCR was done with an ABI Prism 7700, and data were collected and analyzed with SDS software v1.7. The RNA samples (devoid of contaminating DNA) were converted to cDNA with TaqMan reverse transcription reagents (Applied Biosystems, Foster City, Calif.) according to the manufacturer's recommendations. Real-time quantitative reverse transcription-PCR was done as described above with cDNA as the template and the various oligonucleotide primer sets indicated in Table 1. The induction of each gene relative to the untreated and oxygen-depleted samples was normalized to the level of flaB as previously described (14) with the ΔΔCt method, in which the quantity of a given transcript is determined by the equation 2 − ΔΔCt, where Ct is the cycle number of the detection threshold.
TABLE 1.
Oligonucleotide primers used for real-time reverse transcription-PCR
| Oligonucleotide | Nucleotide sequence (5′ to 3′) |
|---|---|
| flaBF | CAGCTAATGTTGCAAATCTTTTCTCT |
| flaBR | TTCCTGTTGAACACCCTCTTGA |
| ospAF | GGCGTAAAAGCTGACAAAAGTAAAGTA |
| ospAR | TAGTGTTTTGCCATCTTCTTTGAAAA |
| ospCF | CGGATTCTAATGCGGTTTTACTTG |
| ospCR | CAATAGCTTTAGCAGCAATTTCATCT |
| dbpAF | CAGATGCAGCTGAAGAGAATCCT |
| dbpAR | ACCCTTTGTAATTTTTCTCTCATTTTT |
| vlsEF | AATCAGCGAAGAAAGTAACAACAAGTAA |
| vlsER | GAAAACTCGACCACCTTGATCAC |
| napAF | GAATGCTTGGATATGATTCTGAATTTAG |
| napAR | TCTCATTCCAAAAATATTCTTCAAAATCT |
| vraAF | AAGCAAATAGAATCGGCCTACAA |
| vraAR | CCCAATTCAATCCCCTAAAAGAC |
Statistics.
To determine whether our real-time reverse transcription-PCR data were statistically significant, we determined whether significant differences were seen for the Ct values obtained for anaerobically versus microaerophilically grown B. burgdorferi for both experimental sets tested with a multiple paired t test as per the SPSS statistical package (SPSS Inc., Chicago, Ill.). Statistical significance was accepted when the P values were less than 0.05.
RESULTS
Effect of reduced level of dissolved oxygen on B. burgdorferi growth.
To determine if reduced oxygen influenced the growth characteristics of Borrelia burgdorferi, we used either enzymatic (Oxyrase) or physical (i.e., N2 gas displacement) methods to decrease the level of dissolved oxygen in BSK-II medium. Measurements made with an oxygen electrode indicated that samples treated with either Oxyrase or N2 gas exhibited a 10-fold reduction in dissolved oxygen levels from 0.25 mM/ml in microaerophilic samples to 0.020 to 0.025 mM/ml in samples treated with either the enzymatic or nonenzymatic regimen.
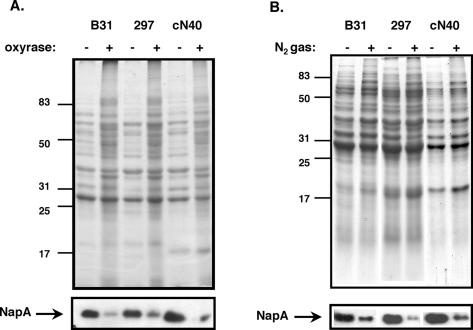
The samples were all grown at 32°C and the pH of the medium was monitored before and after Oxyrase or N2 gas treatment. No change in pH was observed under either condition (data not shown). Low-passage, infectious B. burgdorferi strains B31, 297, and cN40 were all active and motile under these conditions, but exhibited a twofold reduction in growth rate compared to cultivation under conventional microaerophilic conditions (data not shown), and no overt differences were noted in their total protein profiles other than the added Oxyrase reagent, which is a protein-based proprietary reagent that presumably contributes to the additional protein species observed in the Coomassie blue-stained gel shown in Fig. 1A.
FIG. 1.
Level of oxidative stress response protein NapA (BB0690) in B. burgdorferi strains B31, 297, and cN40 grown in O2-depleted medium. The B. burgdorferi isolates were grown under standard microaerophilic conditions in BSK-II medium (lanes −) or in BSK-II depleted of O2 by treatment with Oxyrase (lanes +, panel A) or by displacement with N2 gas (lanes +, panel B). Cultures were harvested after 96 h of treatment, separated on an SDS-PAGE gel stained with Coomassie blue (above), and analyzed by Western immunoblotting with NapA antiserum (shown below).
Nevertheless, we hypothesized that B. burgdorferi oxidative stress proteins would be downregulated when oxygen levels were decreased. To test this, we used antiserum to NapA (designated BB0690 by the Institute for Genomic Research), a 20-kDa protein which is known to be affected by reactive oxygen species (2, 7, 10, 20, 29), to determine the level of this protein in samples grown microaerophilically and anaerobically. For all strains tested, NapA was reduced, particularly when Oxyrase or N2 gas was used to deplete oxygen (Fig. 1A and B, lower panels). These results suggest that oxygen levels have a profound effect on the expression of NapA, and as such, NapA serves as an effective barometer for the oxidative status of B. burgdorferi cells. It should be noted that the levels of the endoflagellar antigen FlaB were unaffected by the redox status of the cells and thus served as a reference control for the growth of B. burgdorferi independent of oxygen levels throughout this study (data not shown).
Effect of reduced dissolved oxygen on B. burgdorferi antigen profiles.
To determine the effect of oxygen limitation on global antigenic differences in B. burgdorferi, we compared the protein profiles of cells grown in the presence and absence of Oxyrase by Western immunoblot analysis (Fig. 2A). When these samples were probed with serum from a patient with chronic Lyme disease or infection-derived mouse serum, profound differences were observed. Specifically, several proteins in the 18- to 20-kDa and 30- to 35-kDa range were synthesized at greater levels in B. burgdorferi grown under limiting O2 conditions (Fig. 2B and 2C) even though equivalent amounts of cells were loaded for these immunoblots, as shown by the total protein profile (Fig. 2A) and the several invariant antigens between the samples.
FIG. 2.
Immunoreactive proteins induced when B. burgdorferi strain B31 is grown under limiting oxygen. B. burgdorferi was grown under standard microaerophilic conditions in BSK-II medium (lanes −) or with added Oxyrase to reduce the level of dissolved oxygen (lanes +). Cells were grown as described in the text, and the samples were resolved by SDS-PAGE and stained with Coomassie blue (panel A) or immunoblotted and probed either with serum from a patient with chronic Lyme borreliosis (panel B) or with infection-derived mouse serum (panel C). Numbers on the left of each panel indicate the molecular mass of protein markers. The arrow denotes the location of the B. burgdorferi OspA lipoprotein.
Effect of reduced dissolved oxygen on the synthesis of lipoprotein antigens.
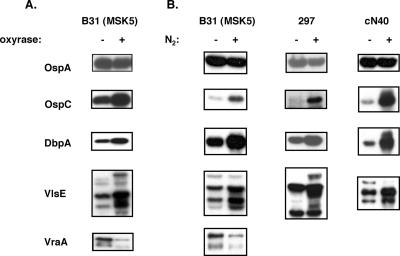
To determine the effect of oxygen limitation on the synthesis of specific borrelial antigens, we used monospecific antisera or monoclonal antibodies to individual B. burgdorferi proteins in Western immunoblot analyses. While outer surface protein A (OspA) remained constant under both microaerophilic and anaerobic conditions, OspC, the decorin binding adhesin DbpA (11, 12), and the antigenic variant VlsE (40) all increased in B. burgdorferi grown under anaerobic conditions when Oxyrase or N2 gas was used (Fig. 3). In contrast, the VraA lipoprotein, a temperature-inducible antigen (15, 33) whose function is unknown, was downregulated when the B. burgdorferi strain B31 derivative MSK5 was grown anaerobically (Fig. 3).
FIG. 3.
Growth with reduced levels of dissolved oxygen modulates the synthesis of several B. burgdorferi antigens from B. burgdorferi sensu stricto strains B31 (MSK5), 297, and cN40. Equivalent numbers of B. burgdorferi cells, grown under standard microaerophilic conditions in BSK-II medium (lanes −, panels A and B) or treated with either Oxyrase (lanes +, panel A) or N2 gas displacement (lanes +, panel B) were resolved by SDS-PAGE, immobilized onto membranes, and probed with antisera to the antigens indicated on the left. The strains tested are indicated above each immunoblot column.
To verify that our observations were not limited to low-passage B. burgdorferi strain B31, we tested infectious B. burgdorferi strains 297 and cN40 because other investigators have noted regulatory differences between B. burgdorferi isolates (38). Although there were some differences between the infectious isolates, overall the same trends were observed for all three strains tested with the notable exception of VraA, which was not detected in either strain 297 or cN40 (Fig. 3).
Effect of O2 levels on transcript synthesis.
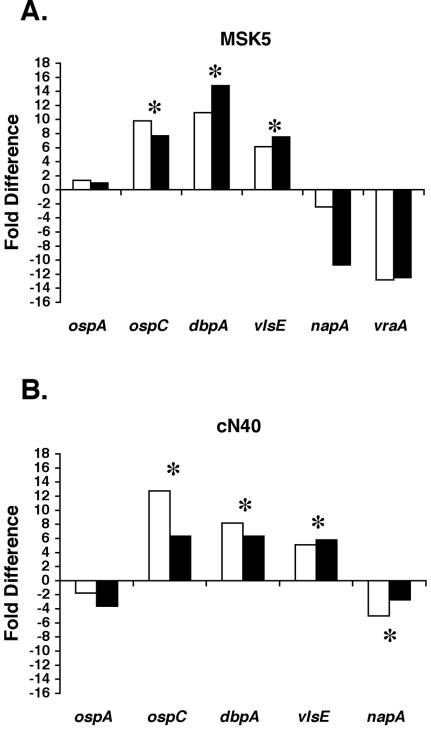
To confirm that changes in protein levels were due to regulation at the transcriptional level, we used quantitative real-time reverse transcription-PCR of total RNA obtained from B. burgdorferi cultivated under microaerophilic and anaerobic conditions. As a normalization control for anaerobic and microaerophilic growth, we used the transcript to flaB, whose transcription level was not significantly affected by growth under either of these conditions (P value of 0.22; data not shown). The results indicated that there was a statistically significant increase in the level of transcripts specific to ospC, dbpA, and vlsE in O2-depleted medium relative to cells grown under microaerophilic conditions (Fig. 4). Specifically, ospC, dbpA, and vlsE transcript levels were 7.7- to 9.8-fold, 10.9- to 14.8-fold, and 6.1- to 7.5-fold greater, respectively, in O2-depleted growth medium than in microaerophilically grown, low-passage B. burgdorferi strain B31 (Fig. 4A). In contrast, under the same conditions, levels of napA and vraA transcripts were decreased 2.4- to 10.7-fold and 12.5- to 12.8-fold, respectively, but were not statistically significant (Fig. 4A). Furthermore, ospA transcripts were unaffected by either condition, consistent with the immunoblot data shown for OspA in Fig. 3. The variations in transcript levels observed were consistent with the increase in protein levels observed for OspC, DbpA, and VlsE and the corresponding decrease in NapA and VraA levels in the earlier immunoblot analyses (Fig. 3) and indicate that the regulation of these antigens occurs at the transcriptional level (Fig. 4).
FIG. 4.
Real-time reverse transcription-PCR indicates that redox regulation of B. burgdorferi genes occurs at the transcriptional level. RNA was isolated from B. burgdorferi strain B31 derivative MSK5 (panel A) and strain cN40 (panel B) following both microaerophilic and anaerobic growth and subjected to real-time reverse transcription-PCR as described in the text. All samples were normalized relative to the flaB transcript and are shown as the ratio of transcripts from cells grown anaerobically relative to cells grown under microaerophilic conditions (represented as fold difference on the y axis). The asterisks (*) indicate samples whose Ct values were statistically significant (i.e., P value less than 0.05) when transcripts from cells grown anaerobically were compared to the same gene transcripts from cells grown microaerophilically. The Ct values from both independent experimental sets were compared with a multiple paired t test. The white and black bars represent data from two independent experiments. The genes analyzed are listed below for each strain tested.
A similar differential in transcript levels was observed in low-passage B. burgdorferi strain cN40 under anaerobic conditions compared to microaerophilically grown spirochetes. Specifically, ospC, dbpA, and vlsE transcripts were increased under anaerobic conditions relative to microaerophilically grown B. burgdorferi by 6.3- to 12.7-fold, 6.3- to 8.2-fold, and 5.1- to 5.8-fold, respectively, whereas napA and ospA transcripts were reduced 2.7- to 5-fold and 1.8- to 3.5-fold, respectively, under the same conditions (Fig. 4). With the exception of ospA, the differences in transcript level observed were statistically significant. The results with cN40 corroborate our earlier antigenic comparisons for OspC, DbpA, VlsE, and NapA under O2-limiting conditions (Fig. 3) and confirm that the redox regulation observed is applicable to an additional infectious borrelial isolate (cN40) (Fig. 4B).
DISCUSSION
Pathogenic bacteria sense and adapt to changes in their environment by coordinately altering gene expression to facilitate survival, transmission and establishment of disease in the host. In this context B. burgdorferi is no different, as it requires rapid adaptation when moving from the arthropod vector to mammalian hosts. Several studies have shown that temperature (25, 28, 31, 34), pH (6), soluble host factors (1, 5, 27, 28), interactions with host cells (14), and a combination of one or more of these factors (38) affect gene expression in B. burgdorferi. Likewise, the effect of dissolved oxygen levels on gene expression represents another potential environmental signal that has not been characterized.
Trafficking through the bloodstream, from the site of deposition or inoculation in the skin after a tick bite to various tissues in the mammalian host, and the host inflammatory response would require a rapid adaptive response to compensate for the four- to fivefold increase in the level of dissolved oxygen between dermal tissue and arterial blood (32, 37). This response is likely to be critical for the survival of B. burgdorferi because high levels of dissolved oxygen are accompanied by greater levels of reactive oxygen species that are toxic or lethal (recently reviewed in references 22 and 35). Since the spirochetal load within a given tissue is too low for direct analysis of B. burgdorferi protein content, the effect of O2 levels on borrelial protein synthesis was determined under in vitro growth conditions where the effect of additional variables such as temperature and pH were kept constant.
Initially we measured the amount of dissolved O2 to be 0.25 mM/ml in conventionally produced BSK-II medium (data not shown). Following either enzymatic (Oxyrase) or physical (N2 gas displacement of dissolved oxygen) depletion of O2 from BSK-II medium, the level of dissolved O2 was reduced to approximately 0.020 to 0.025 mM/ml (data not shown). When three separate, infectious isolates of B. burgdorferi were grown under low oxygen conditions, the amount of NapA (designated BB0690 by the Institute for Genomic Research), a protein shown in other systems to be a nonspecific DNA binding protein that protects cells from oxidative damage (2, 7, 10, 20, 29), was greatly reduced (Fig. 1), consistent with previous reports demonstrating that NapA levels increase in response to greater oxidative stress as a result of regulation by the recently identified BosR regulatory protein (4). Although NapA is purported to bind and protect DNA from oxidative damage in other bacterial systems (2, 7, 10, 20, 29), the exact protective mechanism of B. burgdorferi NapA has not been determined.
We used infection-derived mouse serum and serum from a patient with chronic Lyme borreliosis to determine whether O2 reduction had any effect on the antigenic profile of B. burgdorferi. Although the total protein profiles were primarily unchanged following growth in O2-depleted conditions, we found that several immunoreactive proteins were differentially expressed under these conditions (Fig. 2). With monospecific serum raised against several lipoproteins, we subsequently determined that OspC, DbpA, and VlsE were synthesized at greater levels when the spirochetes were grown in anaerobic conditions, whereas OspA was unchanged (Fig. 3). This profile was common to all three isolates, although subtle differences in expression were observed between low-passage B. burgdorferi strains B31, 297, and cN40 (Fig. 3). Our observations are consistent with recently published reports with genomics-based approaches which demonstrate that dbpA, ospC, and vlsE are upregulated either in mice infected with B. burgdorferi (19) or in dialysis membranes implanted into the peritoneal cavity of rats (1, 27), suggesting that limiting oxygen may be a signal involved in the transcriptional activation of these genes in vivo.
Several recent studies have shown that gene expression within the tick vector following a blood meal can be quite variable (24, 30). For example, Ohnishi et el. demonstrated that ospC and ospA were differentially expressed with all potential combinatorial outcomes possible, indicating that the microenvironment of a given cell within a seemingly identical locale can result in disparate gene expression profiles (24). In addition, Liang et al. demonstrated that B. burgdorferi is able to avoid immune clearance by antibody specific for OspC by downregulating ospC in vivo (17). Although this in vivo observation is likely to be a stochastic phenomenon and dependent on prior exposure to B. burgdorferi and/or persistent infection, it highlights the complexity involved in attempting to explain gene regulatory effects in an equally complex mammalian environment.
Based on these observations and others indicating a differential regulation of lipoprotein-encoding genes over time (3, 9, 18, 19, 26), it is tempting to speculate that the expression level of lipoproteins in B. burgdorferi may also vary in different tissues (i.e., skin tissues relative to blood) where their function could be essential for infection and ultimately favor the persistence of the spirochetes. For example, in the skin or interstitial fluid, increased level of adhesins such as the decorin binding proteins (Dbps), which bind to the proteoglycan decorin that coats host collagen (11, 12), could favor greater attachment of the spirochetes in these tissues. In contrast, within the bloodstream where B. burgdorferi-specific antibodies might be present (depending on the previous exposure to B. burgdorferi), reduced production of Dbp and/or OspC as well as other surface-exposed proteins could favor immune evasion and persistence. Thus, the differential modulation of these and other lipoprotein genes as a function of dissolved O2 may provide another adaptive strategy for B. burgdorferi survival in mammalian tissues.
Several reports have implicated RpoN and RpoS as regulators of both ospC and dbpA in B. burgdorferi (13). As previous investigators have also shown that ospC and dbpA are upregulated in response to temperature, pH, and level of dissolved oxygen, it seems likely that all of these signals are integrated similarly, resulting in RpoN- and RpoS-dependent activation. Recently, Yang et al. demonstrated that the response regulator encoded by BB0763 (designated Rrp2) is required for the induction of ospC and dbpA (39). The activation of Rrp2 is mediated by its cognate membrane-associated histidine kinase encoded by BB0764. This histidine kinase contains a PAS domain that has been shown in other systems to respond to redox stimuli and potentially to as yet unidentified low-molecular-weight species (36). Following sensing by the histidine kinase via the PAS domain, Rrp2 is activated by phosphorylation and coordinates transcription of genes in the context of the RpoN-RNA polymerase complex. In B. burgdorferi, one such gene is rpoS, which has been shown to subsequently activate transcription of dbpA and ospC (13, 39). Our data strongly suggest that oxygen is one of the signals that activates this cascade; however, we cannot exclude the possibility that other molecules may also be involved in the observed induction. The additional signals that may also contribute to this response remain to be determined.
In addition to OspC and DbpA, we determined that VlsE, a lipoprotein involved in antigenic variation (21, 40), was synthesized at greater levels when dissolved O2 was limiting (Fig. 3). A similar derepression of vlsE was observed when B. burgdorferi was cocultivated with intact endothelial cells and also with membranes derived from endothelial cells (14). Although the specific components of the cell membranes that induced vlsE expression have not been determined, it is probable that O2 depletion was mediated by the oxidase activity from the endothelial cell membranes (14). Additionally, Crother et al. recently demonstrated that VlsE levels were higher in skin and joint tissue relative to heart tissue in SCID mice infected with B. burgdorferi (8). Thus, it is tempting to speculate that the differences in VlsE observed in these studies are due to dissolved oxygen levels, inasmuch as temperature and pH would presumably be constant within these tissues.
Real-time reverse transcription-PCR of genes encoding differentially synthesized proteins was conducted under oxygen-limiting conditions to obtain a quantitative measurement of these transcripts (Fig. 4). This analysis corroborated our Western immunoblot data for both strains B31 and cN40, indicating that the synthesis of the lipoprotein antigens OspC, DbpA, and VlsE was due to regulation at the transcriptional level. There were significant differences in the levels of the transcripts that accumulated for strains B31 and cN40, but the patterns of regulation were similar between these two isolates (Fig. 4). Specifically, ospC, dbpA, and vlsE were all transcribed significantly more when oxygen was limiting, whereas ospA transcript levels were unaffected independently of how B. burgdorferi strain B31 derivative MSK5 was cultivated.
In addition to other environmental signals (i.e., pH and temperature), we have determined that B. burgdorferi also alters gene expression in response to the level of dissolved O2. Based on observations, we have developed a model for B. burgdorferi redox regulation. Clearly, redox differences would seem to be operative during the tick life cycle; however, the level of reactive oxygen species within Ixodes spp. is unknown. Once inside the mammal, one could hypothesize that oxygen levels would be variable depending on the tissues to which the spirochete disseminate. For example, within the initial dermal skin deposition site, the level of oxygen would be predicted to be low based on measurements in rats indicating a fourfold reduction of dissolved oxygen in interstitial fluid of the dermis relative to arterial blood (32, 37). Such conditions would result in derepression of dbpBA and thus promote adherence to decorin and subsequent colonization. Daughter cells that could disseminate from the initial site of attachment might then be exposed to different levels of oxygen as they traffic to different tissues within the host.
In the model we propose, the effect of oxygen, which one would predict to be variable within different host tissues, may result in the expression patterns observed. In addition, the localized host inflammatory response may also contribute to the altered gene regulation in order to combat the toxic effect of degranulated neutrophils and/or macrophages. This model does not account for the variability observed for all cells of B. burgdorferi that have been analyzed following tick feeding/host adaptation; however, it serves as a good starting point to understand the role that oxygen plays in this complex regulatory cascade. Along these lines, we infected C3H mice with low-passage, infectious B. burgdorferi grown under oxygen limitation and found that these cells lose their infectious phenotype following this treatment (J. Seshu and J. T. Skare, unpublished data). We are attempting to determine the basis of this observation, as it links the physiological status of the B. burgdorferi cells to their infectivity potential and, as such, may have importance in defining pathogenic mechanisms in Lyme borreliosis.
Host-specific gene expression of B. burgdorferi has been demonstrated (1, 5, 9, 18, 19, 24, 27, 28, 30, 31); however, the mechanisms of host-specific gene expression are not known. In conclusion, it is our contention that, in addition to the previously published signals, dissolved oxygen serves as an environmental factor that also modulates gene expression in B. burgdorferi. Such a response is also important physiologically because exposure to reactive oxygen species derived from either endogenous or exogenous sources is harmful to all living systems. A recent report indicated that a regulatory protein, designated BosR, mediates the response to reactive oxygen species and regulates napA and other genes involved in the response to toxic oxygen intermediates in B. burgdorferi (4). Studies to link this regulatory protein to the redox regulation described herein are under way.
Acknowledgments
We are grateful to Alan Barbour, Robert Gilmore, Magnus Höök, Richard Marconi, Steve Norris, and Allen Steere for providing the various sera used in this study. We also thank John Leong and Michael Norgard for providing B. burgdorferi strains cN40 and 297, respectively, and Rajesh Miranda for assistance with statistical evaluation.
This work was supported by Public Health Service grant AI-42345 from the National Institute of Allergy and Infectious Diseases (to J.T.S.).
Editor: D. L. Burns
REFERENCES
- 1.Akins, D. R., K. W. Bourell, M. J. Caimano, M. V. Norgard, and J. D. Radolf. 1998. A new animal model for studying Lyme disease spirochetes in a mammalian host-adapted state. J. Clin. Investig. 101:2240-2250. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 2.Altuvia, S., M. Almiron, G. Huisman, R. Kolter, and G. Storz. 1994. The dps promoter is activated by OxyR during growth and by IHF and sigma S in stationary phase. Mol. Microbiol. 13:265-272. [DOI] [PubMed] [Google Scholar]
- 3.Anguita, J., M. N. Hedrick, and E. Fikrig. 2003. Adaptation of Borrelia burgdorferi in the tick and the mammalian host. FEMS Microbiol. Rev. 27:493-504. [DOI] [PubMed] [Google Scholar]
- 4.Boylan, J. A., J. E. Posey, and F. C. Gherardini. 2003. Borrelia oxidative stress response regulator, BosR: a distinctive Zn-dependent transcriptional activator. Proc. Natl. Acad. Sci. USA 100:11684-11689. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 5.Brooks, C. S., P. S. Hefty, S. E. Jolliff, and D. R. Akins. 2003. Global analysis of Borrelia burgdorferi genes regulated by mammalian host-specific signals. Infect. Immun. 71:3371-3383. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 6.Carroll, J. A., C. F. Garon, and T. G. Schwan. 1999. Effects of environmental pH on membrane proteins in Borrelia burgdorferi. Infect. Immun. 67:3181-3187. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 7.Cooksley, C., P. J. Jenks, A. Green, A. Cockayne, R. P. Logan, and K. R. Hardie. 2003. NapA protects Helicobacter pylori from oxidative stress damage, and its production is influenced by the ferric uptake regulator. J. Med. Microbiol. 52:461-469. [DOI] [PubMed] [Google Scholar]
- 8.Crother, T. R., C. I. Champion, X. Y. Wu, D. R. Blanco, J. N. Miller, and M. A. Lovett. 2003. Antigenic composition of Borrelia burgdorferi during infection of SCID mice. Infect. Immun. 71:3419-3428. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 9.Das, S., S. W. Barthold, S. S. Giles, R. R. Montgomery, S. R. Telford, and E. Fikrig. 1997. Temporal pattern of Borrelia burgdorferi p21 expression in ticks and the mammalian host. J. Clin. Investig. 99:987-995. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 10.Grant, R. A., D. J. Filman, S. E. Finkel, R. Kolter, and J. M. Hogle. 1998. The crystal structure of Dps, a ferritin homolog that binds and protects DNA. Nat. Struct. Biol. 5:294-303. [DOI] [PubMed] [Google Scholar]
- 11.Guo, B. P., E. L. Brown, D. W. Dorward, L. C. Rosenberg, and M. Hook. 1998. Decorin-binding adhesins from Borrelia burgdorferi. Mol. Microbiol. 30:711-723. [DOI] [PubMed] [Google Scholar]
- 12.Guo, B. P., S. J. Norris, L. C. Rosenberg, and M. Hook. 1995. Adherence of Borrelia burgdorferi to the proteoglycan decorin. Infect. Immun. 63:3467-3472. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 13.Hubner, A., X. Yang, D. M. Nolen, T. G. Popova, F. C. Cabello, and M. V. Norgard. 2001. Expression of Borrelia burgdorferi OspC and DbpA is controlled by a RpoN-RpoS regulatory pathway. Proc. Natl. Acad. Sci. USA 98:12724-12729. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 14.Hudson, C. R., J. G. Frye, F. D. Quinn, and F. C. Gherardini. 2001. Increased expression of Borrelia burgdorferi vlsE in response to human endothelial cell membranes. Mol. Microbiol. 41:229-239. [DOI] [PubMed] [Google Scholar]
- 15.Labandeira-Rey, M., E. A. Baker, and J. T. Skare. 2001. VraA (BBI16) protein of Borrelia burgdorferi is a surface-exposed antigen with a repetitive motif that confers partial protection against experimental Lyme borreliosis. Infect. Immun. 69:1409-1419. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 16.Labandeira-Rey, M., and J. T. Skare. 2001. Decreased infectivity in Borrelia burgdorferi strain B31 is associated with loss of linear plasmid 25 or 28-1. Infect. Immun. 69:446-455. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 17.Liang, F. T., M. B. Jacobs, L. C. Bowers, and M. T. Philipp. 2002. An immune evasion mechanism for spirochetal persistence in Lyme borreliosis. J. Exp. Med. 195:415-422. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 18.Liang, F. T., F. K. Nelson, and E. Fikrig. 2002. DNA microarray assessment of putative Borrelia burgdorferi lipoprotein genes. Infect. Immun. 70:3300-3303. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 19.Liang, F. T., F. K. Nelson, and E. Fikrig. 2002. Molecular adaptation of Borrelia burgdorferi in the murine host. J. Exp. Med. 196:275-280. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 20.Martinez, A., and R. Kolter. 1997. Protection of DNA during oxidative stress by the nonspecific DNA-binding protein Dps. J. Bacteriol. 179:5188-5194. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 21.McDowell, J. V., S. Y. Sung, L. T. Hu, and R. T. Marconi. 2002. Evidence that the variable regions of the central domain of VlsE are antigenic during infection with Lyme disease spirochetes. Infect. Immun. 70:4196-4203. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 22.Mongkolsuk, S., and J. D. Helmann. 2002. Regulation of inducible peroxide stress responses. Mol. Microbiol. 45:9-15. [DOI] [PubMed] [Google Scholar]
- 23.Nadelman, R. B., and G. P. Wormser. 1998. Lyme borreliosis. Lancet 352:557-565. [DOI] [PubMed] [Google Scholar]
- 24.Ohnishi, J., J. Piesman, and A. M. de Silva. 2001. Antigenic and genetic heterogeneity of Borrelia burgdorferi populations transmitted by ticks. Proc. Natl. Acad. Sci. USA 98:670-675. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 25.Ojaimi, C., C. Brooks, S. Casjens, P. Rosa, A. Elias, A. Barbour, A. Jasinskas, J. Benach, L. Katona, J. Radolf, M. Caimano, J. Skare, K. Swingle, D. Akins, and I. Schwartz. 2003. Profiling of temperature-induced changes in Borrelia burgdorferi gene expression by using whole genome arrays. Infect. Immun. 71:1689-1705. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 26.Pal, U., and E. Fikrig. 2003. Adaptation of Borrelia burgdorferi in the vector and vertebrate host. Microbes Infect. 5:659-666. [DOI] [PubMed] [Google Scholar]
- 27.Revel, A. T., A. M. Talaat, and M. V. Norgard. 2002. DNA microarray analysis of differential gene expression in Borrelia burgdorferi, the Lyme disease spirochete. Proc. Natl. Acad. Sci. USA 99:1562-1567. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 28.Roberts, D. M., M. Caimano, J. McDowell, M. Theisen, A. Holm, E. Orff, D. Nelson, S. Wikel, J. Radolf, and R. T. Marconi. 2002. Environmental regulation and differential production of members of the Bdr protein family of Borrelia burgdorferi. Infect. Immun. 70:7033-7041. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 29.Rocha, E. R., G. Owens, Jr., and C. J. Smith. 2000. The redox-sensitive transcriptional activator OxyR regulates the peroxide response regulon in the obligate anaerobe Bacteroides fragilis. J. Bacteriol. 182:5059-5069. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 30.Schwan, T. G., and J. Piesman. 2000. Temporal changes in outer surface proteins A and C of the Lyme disease-associated spirochete, Borrelia burgdorferi, during the chain of infection in ticks and mice. J. Clin. Microbiol. 38:382-388. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 31.Schwan, T. G., J. Piesman, W. T. Golde, M. C. Dolan, and P. A. Rosa. 1995. Induction of an outer surface protein on Borrelia burgdorferi during tick feeding. Proc. Natl. Acad. Sci. USA 92:2909-2913. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 32.Siegemund, M., J. van Bommel, and C. Ince. 1999. Assessment of regional tissue oxygenation. Intensive Care Med. 25:1044-1060. [DOI] [PubMed] [Google Scholar]
- 33.Skare, J. T., D. M. Foley, S. R. Hernandez, D. C. Moore, D. R. Blanco, J. N. Miller, and M. A. Lovett. 1999. Cloning and molecular characterization of plasmid-encoded antigens of Borrelia burgdorferi. Infect. Immun. 67:4407-4417. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 34.Stevenson, B., T. G. Schwan, and P. A. Rosa. 1995. Temperature-related differential expression of antigens in the Lyme disease spirochete, Borrelia burgdorferi. Infect. Immun. 63:4535-4539. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 35.Storz, G., and J. A. Imlay. 1999. Oxidative stress. Curr. Opin. Microbiol. 2:188-194. [DOI] [PubMed] [Google Scholar]
- 36.Taylor, B. L., and I. B. Zhulin. 1999. PAS domains: internal sensors of oxygen, redox potential, and light. Microbiol. Mol. Biol. Rev. 63:479-506. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 37.Venkatesh, B., T. J. Morgan, and J. Lipman. 2000. Subcutaneous oxygen tensions provide similar information to ileal luminal CO2 tensions in an animal model of haemorrhagic shock. Intensive Care Med. 26:592-600. [DOI] [PubMed] [Google Scholar]
- 38.Yang, X., M. S. Goldberg, T. G. Popova, G. B. Schoeler, S. K. Wikel, K. E. Hagman, and M. V. Norgard. 2000. Interdependence of environmental factors influencing reciprocal patterns of gene expression in virulent Borrelia burgdorferi. Mol. Microbiol. 37:1470-1479. [DOI] [PubMed] [Google Scholar]
- 39.Yang, X. F., S. M. Alani, and M. V. Norgard. 2003. The response regulator Rrp2 is essential for the expression of major membrane lipoproteins in Borrelia burgdorferi. Proc. Natl. Acad. Sci. USA 100:11001-11006. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 40.Zhang, J. R., J. M. Hardham, A. G. Barbour, and S. J. Norris. 1997. Antigenic variation in Lyme disease borreliae by promiscuous recombination of VMP-like sequence cassettes. Cell 89:275-285. [DOI] [PubMed] [Google Scholar]