Summary
Background
There has been only 1 study on postoperative pain after external dacryocystorhinostomy (DCR) that compared pain between 2 groups of patients; 1 group received local anesthesia and the other received general anesthesia. To further characterize the relationship between these 2 types of anesthesia and postoperative pain, we designed a study in which a single patient received these 2 different anesthesia modalities for a short interval on 2 different sides.
Material/Methods
There were 50 participants in this study. External DCR was performed on the same participant on both sides using local anesthesia on 1 side and general anesthesia on the other. Postoperative pain was measured using the visual analogue scale (VAS), and localization and timing of pain were reported by the participants. Postoperative nausea and vomiting (PONV) were documented if present.
Results
Pain levels were significantly higher with general anesthesia 3 hours post-surgery, and 6 hours post-surgery the pain remains higher following general anesthesia but is borderline insignificant (p=0.051). However, 12 hours post-surgery, there is no significant difference in the pain level (p=0.240). There was no significant difference in the localization of pain with local and general anesthesia. Postoperative nausea is significantly more frequent after general anesthesia, and vomiting only occurs with general anesthesia. Local anesthesia was preferred by 94% of the participants (47 out of 50).
Conclusions
The vast majority of patients in our study who have undergone both GA and LA DCR would choose LA again, providing a compelling case for use of the LA technique.
Keywords: dacryocystorhinostomy, anesthesia, pain, PONV
Background
External dacryocystorhinostomy (DCR) was first described by Toti in 1904 and subsequently modified by Dupuy-Dutemps and Bourguet in 1921 [1]. Since its introduction it has been globally performed, with some modifications. This procedure is still the “gold standard” for lacrimal surgery [2,3] and it can be performed under local or general anesthesia [4,5]. There is has been only 1 study on postoperative pain after external DCR that compared the pain between 2 groups of patients, with 1group undergoing DCR with local anesthesia (LA) and the other receiving general anesthesia (GA). To adequately compare the postoperative pain and adverse effects of these 2 anesthesia modalities, one must perform external DCR in the same patient on both sides, with 1 side receiving LA and the other receiving GA within a short interval. Here, we report the performance of this exact study.
Material and Methods
Our prospective study included 50 participants who underwent external DCR on 1 side with LA and on the other side with GA 2 days afterwards. This study was performed between 2006 and 2008. The same surgeon, anesthesiologist, and scrub nurse performed the surgery on all participants. The same operating room (OR), instruments, and techniques were utilized for all participants.
All participants were older than 30 years and were American Society of Anesthesiology (ASA) status I – III. The inclusion criterion for our study was bilateral dacryocystitis. We selected 73 patients. Twenty-three patients were excluded from the study for 1 of the following reasons: 1) an unwillingness to participate in the research in 14 cases; 2) a history of midface trauma in 1 case; 3) poor candidacy for GA in 6 cases (American Society of Anesthesiology (ASA) status IV); 4) lacrimal sac asymmetry (1 was dilated); and 5) concurrent treatment with warfarin in 1 case. All participants were provided with careful preoperative explanations of the proposed anesthesia and surgery, specifically what to expect during the procedure in terms of sensation, both tactile and auditory. Approval for this study was obtained from the Belgrade University School of Medicine Ethics Committee.
All participants were administered a 0.1% oxymetazoline solution sprayed into their nose 1 hour before surgery. In the OR, 3 to 5 cotton-tipped applicators (depending on the nostril diameter) moisturized with 0.1% oxymetazoline were placed in the nose for 10–15 minutes instead of conventional gauze packing because of the ease of insertion. Topical anesthesia drops (2% tetracaine-hydrochloride) were instilled into both of the conjunctival sacs.
In the LA group, 3 minutes after the installation of cotton-tipped applicators, 5 drops of 2% tetracaine-hydrochloride were instilled into the nose to moisturize the cotton-tipped applicators and anesthetize the nasal mucosa. All participants were injected with a 1: 1 (v/v) mixture of 2% lidocaine – 1: 200 000 adrenalin and 0.75% bupivacaine. The anesthetics were injected through a 16-mm, 25-G needle via the skin in 3 positions (1.5 ml each): 1) above the infraorbital foramen immediately above the lower orbital floor periosteum at the maximum depth allowed by the needle; 2) into the medial extraconal orbit above the medial ligament at the maximum depth allowed by the needle; and 3) subcutaneously into the lacrimal sac region. Following the injections, 0.3 ml of the solution was used to moisturize the cotton-tipped applicators that were previously placed in the nose. Direct injection of anesthetics into the nasal mucosa was not performed in any case. A 22-G cannula was introduced into the cubital vein for intravenous therapy if needed. No intravenous sedation was administered. All participants received supplemental intranasal oxygen at 4 L/min.
The GA group was intravenously administered 2.5 mg/kg of propofol, 1 mcg/kg of fentanyl for induction, and 0.6 mg/kg of rocuronium for muscle relaxation. Anesthesia was maintained with sevoflurane/NO2/O2. All participants were subjected to constant monitoring of their electrocardiogram, blood pressure and pulse oximetry.
Briefly, for the external DCR, a medial canthal incision was made with a blunt dissection of the orbicularis muscle carried down to the level of the medial canthal tendon. The medial canthal tendon and periosteum were incised and reflected with a periosteal elevator to expose the lacrimal fossa. If the angular vessels were bleeding, they were immediately cauterized by bipolar diathermy. Next, we made anterior and posterior flaps in the lacrimal sac. The nasal pack was removed. The thin lacrimal bone was infractured, and a 3-mm-tip Kerrison punch was used to create an osteotomy. The next step was to form anterior and posterior flaps in the nasal mucosa. The 2 posterior flaps were then closed using a 6-0 Vicryl suture. The silicone stents were passed along the canalicular system and into the osteotomy site to exit via the naris. The tubes were pulled, tied, knotted, and spontaneously retracted into the nose. The anterior nasal mucosa flap was anastomosed to the anterior aspect of the lacrimal sac, and the incision was closed in a layered fashion using a 6-0 Vicryl suture. A gauze pack was placed in the nose and removed the next day. The participants were asked to rest for 1–2 hours without applying ice packs and were advised to avoid blowing their noses for 1 week. A fiberoptic coaxial headlight to illuminate the depths of the DCR wound is essential during surgery.
The postoperative nausea and vomiting (PONV), postoperative pain level, and localization were checked at 30 minutes, 90 minutes, 3 hours, 6 hours, 12 hours, and 24 hours after surgery. The visual analogue scale (VAS) was used for clinical pain assessment. Six days following the last procedure, participants were asked about their satisfaction regarding the procedures with the 2 different types of anesthesia. Also, patents stated their subjective opinion about procedure comfort (in GA and LA) on a 3 stage verbal scale designed for the purpose of this study: a) not satisfied, b) satisfied, and c) very satisfied. They were specifically asked about which type of anesthesia (LA or GA) they preferred and if possible, to explain why they preferred it. The participants were observed for 6 months. The Wilcoxon signed-rank test was used to assess the significance of the differences between the groups for VAS pain levels. PONV and satisfaction with anesthesia were compared with chi-squared tests. Data were analyzed with the SPSS 12.0 software (SPSS Inc., 1989–2003) and a P value of less than 0.05 was considered significant.
Results
There were 50 participants aged between 31 and 83 years (64±10) enrolled in this study. There were 40 females, ranging from 31 to 74 years (64±10), and 10 males, ranging from 46 to 83 years (63±11).
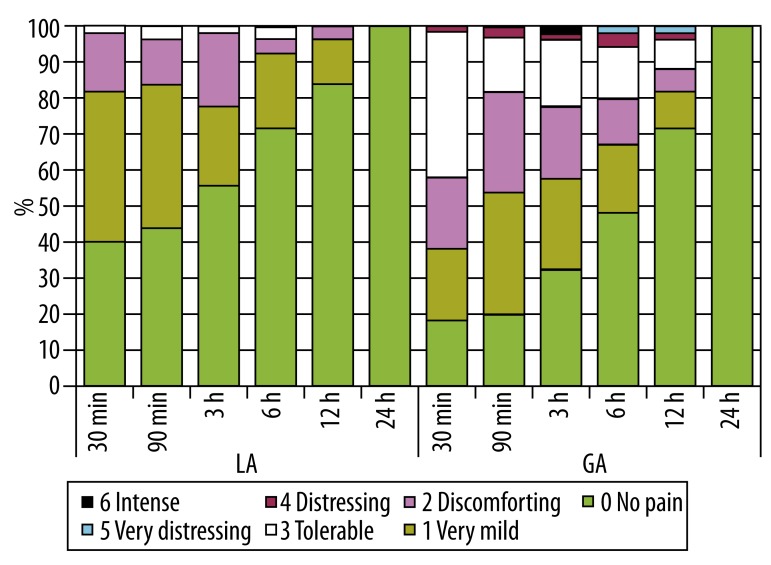
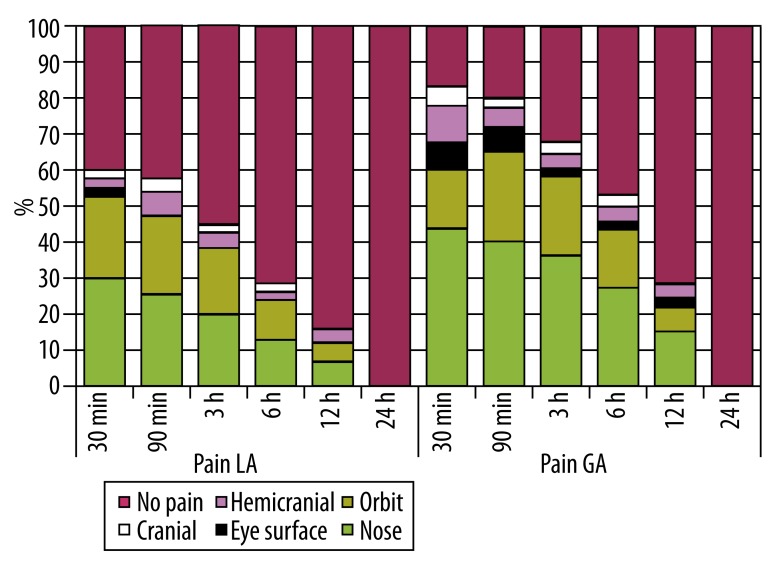
Participants were asked about their level of postoperative pain (Table 1), localization of pain (Table 2), postoperative nausea and vomiting (Table 3) at 30 minutes, 90 minutes, 3 hours, 6 hours, 12 hours, and 24 hours after surgery. The level of pain was lower for LA 3 hours after surgery (p<0.05). At 6 hours, the difference was insignificant but approached significance (p=0.051), and there was no difference after 12 hours (Figure 1). Participants with a pain level of 4 or more received 1 mg/kg of tramadol i.m., and participants with a pain level of 3 or less received methimazole i.m. only when they requested it. After surgery in LA 2 (4%) patients needed pain medication, while in the GA group there were 14 (28%) patients. There was a statistically significant difference in the personal request for pain medication between the LA and GA groups (p<0.05). There was no statistically significant difference in the localization of pain between the LA and GA groups (Figure 2).
Table 1.
Level of pain in our series.
| Time after surgery | Pain with LA | Pain with GA | ||||||
|---|---|---|---|---|---|---|---|---|
| Range | Median | q1 | q3 | Range | Median | q1 | q3 | |
| 30 minutes | 0–3 | 1 | 0 | 1 | 0–4 | 2* | 1 | 3 |
| 90 minutes | 0–3 | 1 | 0 | 1 | 0–4 | 1* | 1 | 2 |
| 3 hours | 0–3 | 0 | 0 | 1 | 0–6 | 1* | 0 | 2 |
| 6 hours | 0–3 | 0 | 0 | 1 | 0–5 | 1 | 0 | 2 |
| 12 hours | 0–2 | 0 | 0 | 0 | 0–5 | 0 | 0 | 1 |
| 24 hours | 0 | 0 | 0 | 0 | 0 | 0 | 0 | 0 |
Statistically significant difference between the LA and GA groups in series; q1 – the first quartile point 1/4 of the data below median (25 percentiles); q3 – the third quartile point 3/4 of the data below median (75 percentiles).
Table 2.
Localization of pain after surgery (numbers represent number of patients).
| Localization of pain | Pain with LA | Pain with GA | ||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|
| 30 min | 90 min | 3 h | 6 h | 12 h | 24 h | 30 min | 90 min | 3 h | 6 h | 12 h | 24 h | |
| Nose | 15 | 13 | 10 | 7 | 4 | 0 | 22 | 20 | 18 | 14 | 8 | 0 |
| Orbit | 12 | 11 | 9 | 5 | 2 | 0 | 8 | 13 | 11 | 8 | 3 | 0 |
| Eye surface | 1 | 0 | 0 | 0 | 0 | 0 | 4 | 3 | 1 | 1 | 1 | 0 |
| Hemicranial | 1 | 3 | 2 | 1 | 2 | 0 | 5 | 2 | 2 | 2 | 2 | 0 |
| Cranial | 1 | 1 | 1 | 1 | 0 | 0 | 2 | 2 | 2 | 1 | 0 | 0 |
| No pain | 20 | 22 | 28 | 36 | 42 | 50 | 9 | 10 | 16 | 24 | 36 | 50 |
Table 3.
PONV in our series, as compared to Ciftci et al. series.
| After surgery | Our series | Ciftci et al. | |||
|---|---|---|---|---|---|
| LA | GA | LA | GA | ||
| Nausea | 5 min | Not measured | 5 (1.6%) | 14 (7.6%)* | |
| 30 min | 1 (2%) | 12 (24%)* | 4 (1.3%) | 9 (4.9%)* | |
| 90 min | 0 | 7 (14%)* | Not measured | ||
| 120 min | Not measured | 1 (0.3%) | 4 (2.1%) | ||
| 180 min | 0 | 0 | Not measured | ||
| Vomiting | 5 min | Not measured | 4 (1.3%) | 10 (5.4%)* | |
| 30 min | 0 | 4 (8%)* | 2 (0.6%) | 6 (3.2%)* | |
| 90 min | 2 (4%) | Not measured | |||
| 120 min | Not measured | 1 (0.3%) | 3 (1.6%) | ||
| 180 min | 0 | 0 | Not measured | ||
Statistically significant difference between the LA and GA groups in series.
Figure 1.
Level of pain after surgery.
Figure 2.
Localization of pain after surgery.
The incidence of PONV with LA was lower than that with GA 30 minutes post-surgery. The incidence of nausea was only lower for LA 90 minutes after the surgery (p<0.05) (Table 3). There was no PONV 3 hours or later following the surgery.
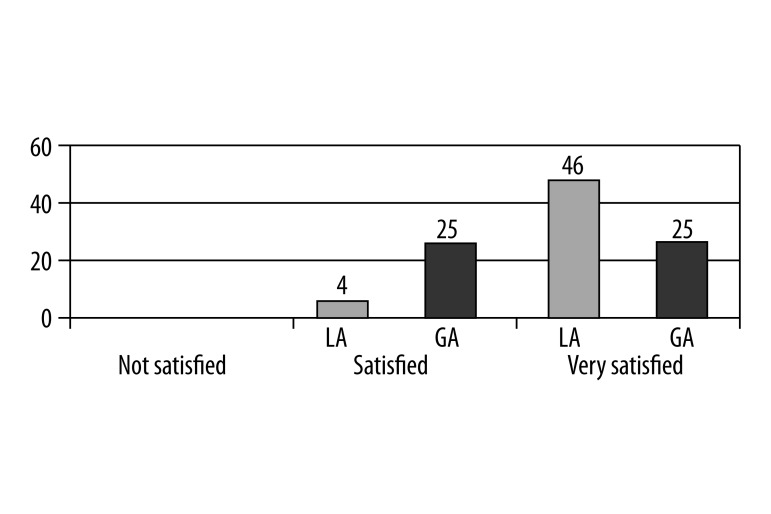
Participants were satisfied with both types of anesthesia; 92% were very satisfied with LA, while 50% were satisfied with GA. The difference is statistically significant (p=0.000, Figure 3). Forty-seven of the 50 (94%) participants preferred that their next procedure be performed under LA. The remaining 3 participants preferred GA. None of the cases required conversion from LA to GA. In addition, none of the participants experienced uncontrolled intranasal bleeding during the procedure. There were no cases of postoperative epistaxis requiring nasal packing. There were no cases of dislodged tubing. The success of the procedure was confirmed by lacrimal passages on irrigation in 49 of the 50 participants after 6 months. One participant had bilateral fibrosis and required further surgery.
Figure 3.
Patients’ satisfaction with procedure under LA and GA.
Discussion
Performing DCR under LA is an accepted procedure and has been performed for many years, especially in elderly patients [6–8]. Obviously, the specific risks of GA are avoided in LA. Previous studies demonstrated that there was a much lower incidence of PONV after DCR with LA than after DCR with GA [10,11]. The anesthetics used for GA also act as vasodilators and enhance bleeding, which brings greater risk of complications [2,4,8–10]. Prior to the current study, it was not known whether LA or GA is more comfortable for the patient, because the same patient was not subjected to the same procedure using different anesthesia.
In our study series, the postoperative pain levels are lower for LA. At 6 hours post-surgery, the difference is insignificant but approaching significance (p=0.051), and 12 hours after surgery there is no significant difference (p=0.240) (Table 1, Figure 1). We propose that this is because we used the long-acting LA bupivacaine in our study. Ciftci et al used only short-acting LA in their study (Table 5). There was no difference in the pain between their patients who received LA and GA 2 hours after surgery [10]. In both studies, the pain decreased with time following the surgery. Also, the level of pain is higher in the Ciftci et al series in comparison to our series. The only common time-point in which the pain levels were measured in these studies was 30 minutes post-surgery. At that time the pain levels, as assessed by VAS, in the Ciftci et al. series were higher than those observed in our series for both LA and GA. We propose that the reason for this difference is because the study population was younger in the Ciftci et al. study (23±2 years), and it has been previously reported that younger patients have lower pain thresholds [12].
Table 5.
Pain in our series, as compared to Ciftci et al. series.
| After surgery | Our series | Ciftci et al. | ||||
|---|---|---|---|---|---|---|
| LA | GA | LA | GA | |||
| Range | Median | Range | Median | Mean ±SD | Mean ±SD | |
| 5 min | Not measured | Not measured | 1.2±1.7 | 4.9±2.5* | ||
| 30 min | 0–3 | 1 | 0–4 | 2* | 1.8±2.0 | 3.6±2.2* |
| 90 min | 0–3 | 1 | 0–4 | 1* | Not measured | |
| 120 min | 2.3±1.4 | 3.1±2.3 | ||||
| 180 min | 0–3 | 0 | 0–6 | 1* | Not measured | |
Statistically significant difference between the LA and GA groups in series.
After surgery in our series, in the LA group 2 (4%) patients needed pain medication, while in the GA group there were 14 (28%) patients. There was a statistically significant difference in the personal request for pain medication between the LA and GA groups (p=0.01). In contrast to Ciftci et al. and our prospective and VAS scale-based methodology for assessing postoperative pain, in a study by Harissi-Dagher et al. the method for assessing pain was retrospective insight into pain with 2 alternative values – either the requirement of postoperative analgesic by the patient, or no requirement. In their series there was no significant difference between LA i GA groups with respect to need of analgesic post-operatively; 50.7% of patinents in the LA group required analgesic and 46% in the GA group. [11] Our mean operating time was 24 minutes for LA (24.24±4.66) and GA (24.32±4.67), rather similar to the 36 minutes found in the Caesar et al. series (longest duration – 65 minutes – was in a patient with persistent ooze from the bony rhinostomy) [2]. Maheshwari’s mean operative time was 15.5 minutes [15]. On the other hand, the significantly longer operating time in the Harissi-Dagher et al. series could have led to more tissue trauma and more frequent post-operative need for analgesics (LA 56.2±15.3 minutes and GA 64.0±18.1minutes) [11].
Most participants complained of pain in their nose or orbit after surgery. The localization of pain is the same for both types of anesthesia following surgery; however, higher levels of pain are experienced after GA (Table 2).
PONV is present in 20% to 30% of patients operated on under GA [13]. In the region of the throat, nose and eye, PONV can be induced in up to 70% of patients after surgery with GA [14]. PONV is more frequently observed when GA is used in both our study, the Ciftci et al. study, and the Harissi-Dagher et al. studies[10,11]. In our series, participants developed PONV more frequently than those in the Ciftci et al. series (Table 3) and less frequently than those in the Harissi-Dagher et al. series (Table 4).
Table 4.
Results of PONV in our series, as compared to Harissi-Dagher et al. series.
| Hours after surgery | Our series | Dagher et al. | |||
|---|---|---|---|---|---|
| LA | GA | LA | GA | ||
| PONV | 0 | Not measured | 61 (86%) | 109 (73%) | |
| 0.5 | 1 (2%) | 12 (24%)* | Not measured | ||
| 1 | Not measured | 10 (14%) | 29 (19%) | ||
| 1.5 | 0 | 7 (14%)* | Not measured | ||
| 2 | Not measured | 0 | 6 (4%) | ||
| 3 | 0 | 0 | 0 | 5 (3%) | |
| 4 | Not measured | 0 | 1 (0.7%) | ||
| 6 | 0 | 0 | Not measured | ||
Statistically significant difference between the LA and GA groups in series.
Our series has a frequency of PONV that is comparable to other previously cited series describing PONV frequency with throat, nose, and eye surgery [13,14]. The Ciftci et al. series has a lower frequency of PONV, while the Harissi–Dagher et al. series has a higher frequency of PONV [10,11].
Patients’ acceptance is very important. However, patients’ acceptance does not necessarily mean that the level of comfort during the procedure was ideal, thus the implemented scale of post-operative comfort in our study.
McNab found the technique acceptable to the patients and none of them stated afterwards that they would have preferred to have had a general anaesthetic [5]. Those patients were not operated on with GA; although they were satisfied with LA, they had not experienced GA at all, so their opinion is not objective.
Other studies examining external DCR and the type of anesthesia found that the procedure is acceptable to the patient based on criteria of how often the patient or surgeon felt the need to convert to GA or how often additional LA had to be used during the procedure because of patient pain [6,7,10,11,15]. Kratky et al. analyzed 25 consecutive DCRs under local anaesthesia in elderly patients and found there was no need for conversion from LA to GA [6]. Hurwitz et al. in 1 case converted anesthesia from LA to GA because of ”non-compliance” [7]. Ciftci et al., in 2 patients operated on with LA, additional LA had to be used during the procedure because of pain [10]. Harissi–Dagher et al. found that patients were comfortable with the technique used and they did not convert LA to GA in any case [11]. Maheshwari found that overall patient satisfaction was good [15]. Except for McNab, none of the authors quoted patients directly on their level of satisfaction with surgical procedure with LA. Indirectly, the number of cases in which no conversion from LA to GA had been noted could be regarded as an indicator of acceptable satisfaction, and this was, seemingly, used by all authors.
Our series is the first to our knowledge in which the same participants experienced both procedures within a short period of time and chose the type of anesthesia for a potential third DCR. The patient choice is LA in 94% of cases (47 of 50 patients). The remaining 6% of patients prefer GA to LA. According to the literature, pain, if it is present, is low during DCR in LA. Knežević et al. made an attempt to quantify the pain of DCR under LA and described it as ”comparable to receiving an i.m. gluteal injection” [16]. None of the patients in our group complained of pain, which is highest during bone removal and flaps formation, in LA [15–17] In 3 cases (6%), the preference for GA over LA must be ascribed to other reasons not measured by parameters included in this study. The only descriptive difference that emerged from our analyses was the patients’ subjective preference for GA; in 2 cases was caused by the fact that these participants did not want to be consciously involved in the procedure (they preferred to sleep and remain unaware). In 1 case, the sound of bone cracking was unpleasant, and the participant did not want to experience it again, but this patient did not complain of pain during the procedure with LA. This complaint is unique in our experience, as no one has complained of this sound in the 300 or more procedures that we have performed using LA.
Conclusions
The vast majority of patients in our study who have undergone both GA and LA DCR would choose LA again, providing a compelling case for use of the LA technique.
GA is a better choice for the rare cases of patients unwilling or unable to cooperate during the procedure (eg, claustrophobia, mental retardation, senile or presenile dementia) and patients who explicitly state that they do not wish to be consciously involved in the procedure.
Acknowledgments
This work was done by the authors without any financial support. All surgical procedures were performed at the University Eye Clinic Clinical Center of Serbia.
There were no research contracts or any kind of financial support (grants) for this study. All authors have no conflicts of interest.
Approval for this study was obtained from the Belgrade University School of Medicine Ethics Committee.
Footnotes
Source of support: Departmental sources
References
- 1.Hughes SM. The History of Lacrimal Surgery. Adv Ophthal Plastic & Reconstruct Surgery. 1986;5:139–68. [PubMed] [Google Scholar]
- 2.Caesar RH, Mc Nab AA. External dacryocystorhinostomy and local anesthesia. Technique to measure minimized blood loss. Opthal Plast Reconst Surg. 2004;20:57–59. doi: 10.1097/01.IOP.0000105567.09310.7C. [DOI] [PubMed] [Google Scholar]
- 3.Moore WMH, Bentley CR, Olver JM. Functional and anatomic results after two types of endoscopic endonasal dacryocystorhinostomy: surgical and holmium laser. Ophthalmology. 2002;109:1575–82. doi: 10.1016/s0161-6420(02)01114-4. [DOI] [PubMed] [Google Scholar]
- 4.Jordan DR. Avoiding blood loss in outpatient DCR. Opthal Plast Reconst Surg. 1991;7:261–66. doi: 10.1097/00002341-199112000-00005. [DOI] [PubMed] [Google Scholar]
- 5.McNab AA, Simmie RJ. Effectiveness of local anesthesia for external dacryocystorhinostomy. Clin Experimental Ophthalmol. 2002;30:270–72. doi: 10.1046/j.1442-9071.2002.00535.x. [DOI] [PubMed] [Google Scholar]
- 6.Kratky V, Hurwitz JJ, Ananthanarayan C, Avram DR. Dacryocystorhinostomy in elderly patients: regional anesthesia without cocaine. Can J Ophthalmol. 1994;29(1):13–16. [PubMed] [Google Scholar]
- 7.Hurwitz JJ, Merkur S, De Angelis D. Outcome of lacrimal surgery in the elderly. Can J Ophthalmol. 2000;35:18–22. doi: 10.1016/s0008-4182(00)80104-0. [DOI] [PubMed] [Google Scholar]
- 8.Woog JJ, Kennedy RH, Custer PL, et al. Endonasal dacryocystorhinostomy: a report by the American Academy of Ophthalmology. Ophthalmology. 2001;108:2369–77. doi: 10.1016/s0161-6420(01)00945-9. [DOI] [PubMed] [Google Scholar]
- 9.Mailer CM, Webster AC. Controlled sedation, sphenopalatine and nasociliary blocks, and bloodless flap suturing in dacryocystorhinostomy. Can J Ophthalmol. 1982;17(5):189–93. [PubMed] [Google Scholar]
- 10.Ciftci F, Pocan S, Karadayi K, Gulecek O. Local versus general anesthesia for external dacryocystorhinostomy in young patients. Ophthal Plast Reconstr Surg. 2005;21(3):201–6. doi: 10.1097/01.iop.0000163317.73873.c9. [DOI] [PubMed] [Google Scholar]
- 11.Harissi-Dagher M, Boulos P, Hardy I, Guay J. Comparison of Anesthetic and Surgical Outcomes of Dacryocystorhinostomy using Loco-regional versus General Anesthesia. Digital Journal of Opthalmology. 2008;14:1. doi: 10.5693/djo.01.2008.001. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 12.Rashiq S, Dick BD. Factors associated with chronic noncancer pain in the Canadian population. Pain Res Manag. 2009;14(6):454–60. doi: 10.1155/2009/919628. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 13.Cohen MM, Duncan PG, DeBoer DP, Tweed WA. The postoperative interview: Assessing risk factros for nausea and vomiting. Anesth Analg. 1994;78:7–16. doi: 10.1213/00000539-199401000-00004. [DOI] [PubMed] [Google Scholar]
- 14.Donlon JV., Jr . Anesthesia for Eye, Nose, and Throat. In: Miller URD, editor. Anesthesia. 5nd ed. New York: Churchill Livingstone; 2000. [Google Scholar]
- 15.Maheshwari R. Single-prick infiltration anesthesia for external dacryocystorhinostomy. Orbit. 2008;27(2):79–82. doi: 10.1080/01676830701377229. [DOI] [PubMed] [Google Scholar]
- 16.Knežević MM, et al. Pain during external dacryocystorhinostomy with local anesthesia. Med Sci Monit. 2011;17(6):CR341–46. doi: 10.12659/MSM.881807. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 17.Smith MG, Kong AS, et al. Lacrimal fossa block: an audit of a minimally invasive regional anaesthetic technique for endoscopic dacryocystorhinostomy. Clin Otolaryngol. 2001;26:407–10. doi: 10.1046/j.1365-2273.2001.00493.x. [DOI] [PubMed] [Google Scholar]