Abstract
Generation of tumor-antigen specific CD4+ T-helper (TH) lines through in vitro priming is of interest for adoptive cell therapy of cancer, but the development of this approach has been limited by the lack of appropriate tools to identify and isolate low frequency tumor antigen-specific CD4+ T cells. Here, we have used recently developed MHC class II/peptide tetramers incorporating an immunodominant peptide from NY-ESO-1 (ESO), a tumor antigen frequently expressed in different human solid and hematologic cancers, to implement an in vitro priming platform allowing the generation of ESO-specific TH lines. We isolated phenotypically defined CD4+ T-cell subpopulations from circulating lymphocytes of DR52b+ healthy donors by flow cytometry cell sorting and stimulated them in vitro with peptide ESO119-143, autologous APC and IL-2. We assessed the frequency of ESO-specific cells in the cultures by staining with DR52b/ESO119-143 tetramers (ESO-tetramers) and TCR repertoire of ESO-tetramer+ cells by co-staining with TCR variable β chain (BV) specific antibodies. We isolated ESO-tetramer+ cells by flow cytometry cell sorting and expanded them with PHA, APC and IL-2 to generate ESO-specific TH lines. We characterized the lines for antigen recognition, by stimulation with ESO peptide or recombinant protein, cytokine production, by intracellular staining using specific antibodies, and alloreactivity, by stimulation with allo-APC. Using this approach, we could consistently generate ESO-tetramer+ TH lines from conventional CD4+CD25− naïve and central memory populations, but not from effector memory populations or CD4+CD25+ Treg. In vitro primed TH lines recognized ESO with affinities comparable to ESO-tetramer+ cells from patients immunized with an ESO vaccine and used a similar TCR repertoire. In this study, using MHC class II/ESO tetramers, we have implemented an in vitro priming platform allowing the generation of ESO-monospecific polyclonal TH lines from non-immune individuals. This is an approach that is of potential interest for adoptive cell therapy of patients bearing ESO-expressing cancers.
Introduction
Analysis of spontaneous immune responses to tumor antigens in cancer patients has led to the identification of those most relevant for immune-based therapies. One of the most immunogenic of them, called NY-ESO-1 (ESO), belongs to the cancer/testis antigen (CTA) group, including antigens that, in adults, have an expression pattern restricted to testis and tumor cells.1,2 ESO is frequently expressed in various solid (e.g. melanoma, ovarian cancer)3,4 and hematologic (e.g. multiple myeloma (MM) adult T-cell leukemia/lymphoma (ATLL))5-9 tumors, and represents an attractive target for cancer immunotherapy. Different ESO-based immunotherapeutic approaches are under development, including vaccines10,11 and passive adoptive cell transfer (ACT) therapy using adoptively transferred ESO-specific T cells, which is particularly attractive for the treatment of patients with recurrent disease.
ACT using tumor-infiltrating lymphocytes (TIL) amplified in vitro, in association with lymphodepleting chemotherapy and IL-2, has indeed been shown to mediate, in patients with refractory metastatic melanoma, the regression of large established tumors.12 Trials using defined tumor antigen-specific CD8+ T-cell (CTL) populations have shown a more limited efficacy, together with a short in vivo persistence of transferred populations, in the absence of specific CD4+ T-cell help.13,14 A recent study, however, has reported a long-term complete remission in a patient with metastatic melanoma adoptively transferred with an in vitro expanded autologous ESO-specific CD4+ T-cell clone that persisted in vivo and appeared to induce endogenous responses to additional tumor antigens.15 The potential of adoptive transfer of tumor antigen-specific CD4+ T cells for the eradication of established tumors has been further supported by recent studies in murine models.16-18 Together, these results encourage the implementation of further studies assessing the clinical efficacy of ESO-specific CD4+ T cells administered to cancer patients bearing antigen-expressing tumors, alone or in association with ESO-specific CTL.
Whereas the generation of tumor antigen-specific CD4+ TH cell clones for ACT from patients with spontaneous immune responses to the antigen, as currently performed, is labor intensive, not economically advantageous and is not applicable to all patients,19 an alternative approach is to generate tumor-specific TH populations of defined antigen specificity and HLA-restriction through in vitro priming of CD4+ T cells from non-immune individuals, including histocompatible donors. Because an important element for a successful therapy based on the adoptive transfer of tumor-specific T cells is their ability to persist and expand in vivo, populations derived from naïve CD4+ T cells may be, in this respect, superior in their ability to maintain a high replicative potential and resist terminal differentiation.16,20,21 In addition, ex vivo removal of CD25+ regulatory T cells (Treg) from CD4+ T-cell populations may be suitable, to avoid their presence in the transferred TH lines.22 The development of this approach, however, has been limited, to date, by the lack of appropriate tools to specifically identify and separate low frequency tumor antigen-specific CD4+ T cells, particularly from non-immune individuals.
Soluble fluorescent MHC-peptide tetramers that allow the direct identification and separation of antigen-specific T cells, have in recent years become essential tools for T-cell analysis. MHC class I/peptide tetramers have been generated for a large number of murine and human alleles and peptides, and are widely used to assess antigen-specific CD8+ T cells.23,24 The development of MHC class II tetramers, and particularly of those incorporating peptides from tumor and self-antigens, has been more complex and only a few of them have been successfully developed.25-27 Through a strategy that combines the use of Histidine (His)-tagged peptides with isolation of MHC/peptide monomers by affinity purification, we have recently generated MHC class II tetramers incorporating an ESO immunodominant peptide (ESO-tetramers).28,29 We have shown that MHC class II/ESO tetramers specifically and avidly bind to ESO-specific CD4+ T cells, allowing their highly sensitive detection and isolation from polyspecific populations. In this study, we have used ESO-tetramers to implement an in vitro priming platform, allowing the generation of ESO-monospecific polyclonal TH lines from CD4+ T cells of non-immune individuals. This approach can potentially be applied to the treatment of large numbers of patients bearing ESO-expressing cancers.
Design and Methods
Healthy donor samples, cells and ex vivo cell sorting
Peripheral blood samples were collected from healthy donors after obtaining written informed consent and approval from the Institutional Review Board (Etablissement Français du Sang Pays de la Loire, Nantes, France). Peripheral blood mononuclear cells (PBMC) were obtained from peripheral blood by density gradient centrifugation (LSM 1077 lymphocyte separation medium, PAA laboratories GmbH). CD4+ T cells were enriched by positive selection from PBMC by magnetic cell sorting (Miltenyi Biotec). For ex vivo flow cytometry cell sorting, CD4+ T cells were stained with fluorochrome-labeled monoclonal antibodies (mAb) specific for CD45RA (BD Biosciences), CD25 (Beckman Coulter), CD127 (eBioscience) and CCR7 (BD Biosciences) and were separated into conventional naïve (N; CD45RA+CCR7+CD25–), natural naïve Treg (NnTreg; CD45RA+CCR7+CD25+CD127low), conventional memory (M; CD45RA–CCR7+/-CD25–) and memory Treg (MTreg; CD45RA–CCR7+/-CD25+CD127low) populations to high purity (>97%) using a FACSAria (BD Biosciences). In some experiments conventional memory cells were sorted into central memory (CM; CD45RA−CCR7+CD25−) and effector memory (EM; CD45RA− CCR7−CD25−) populations. Monocyte-derived dendritic cells (DC) were generated from enriched CD14+ cells, isolated from PBMC by magnetic sorting (Miltenyi Biotec), by culture in the presence of rhGM-CSF and rhIL-4 (1000 U/ml each; R&D Systems) for five days. The DR52b+ EBV-transformed B-cell line, JBUSH (9035), was obtained from the National Marrow Donor Program/American Society for Histocompatibility and Immunogenetics (NMDP/ASHI) Cell Repository. Healthy donors were functionally typed for DR52b expression based on the capacity of their antigen presenting cells (APC) to present peptide ESO119-143 to a specific DR52b-restricted CD4+ T-cell clone, as previously described.30
Priming of ESO-specific CD4+ T cells, tetramer staining, TCR BV analysis and generation of specific TH lines
Fluorescent HLA-DR52b/ESO119-143 (DR52b/ESO) and HLA-DR52b/Influenza matrix protein73-92 (DR52b/Flu) tetramers were generated using His-tagged antigenic peptides, as previously described.28Ex vivo sorted CD4+ T-cell subpopulations (3-5×106) were stimulated in vitro with peptide ESO119-143 (2 mM) in the presence of irradiated autologous CD4– cells (3-5×106) and were cultured in the presence of rhIL-2 (100 U/mlL, Chiron). Day 12 cultures were incubated with tetramers at a final concentration of 3 g/mL for 1 h at 37°C and then stained with anti-CD4 mAb and analyzed by flow cytometry. TCR variable b chain (BV) usage by ESO-specific cells was determined by flow cytometry analysis following staining of Day 12 cultures with ESO-tetramers and anti-BV2 mAb (Immunotech), as previously described.28 ESO-specific T cells were isolated from peptide-stimulated cultures by tetramer-guided flow cytometry cell sorting, and expanded by stimulation with PHA and irradiated allogeneic PBMC in the presence of IL-2, as previously described.28 The specificity of the obtained polyclonal TH lines was assessed by tetramer staining and flow cytometry analysis.
Antigen recognition, cytokine production and alloreactivity
For the assessment of their functional avidity, ESO-specific polyclonal TH lines (104) were incubated with JBUSH cells (104) in the presence of serial dilutions of peptide ESO119-143 (1 mM) or a control irrelevant peptide (ESO1-20), and IFN-γ was assessed in 24-h culture supernatants by ELISA (Invitrogen). For the assessment of the reactivity of ESO-specific lines to naturally processed full-length ESO, 104 cells were co-cultured with DR52b+ DC (5×104), pre-incubated for 16 h with serial dilutions of recombinant ESO or Melan-A proteins, and IFN-γ was assessed in 24-h culture supernatants by ELISA. The capacity of ESO-specific TH lines to produce cytokines was assessed in a standard 4-h intracellular cytokine staining assay using specific mAb (IL-2, TNF-α, IFN-γ, IL-4 and IL-10, BD Biosciences; IL-17, eBioscience) following stimulation with peptide ESO119-143 (1 mM). To assess the extent of alloreactivity of ESO-specific lines, 104 cells were incubated with DC (5×104) derived from a panel of 8 DR52b+ healthy donors, in the presence or absence of peptide ESO119-143 (1 mM), and IFN-γ was assessed in 24-h culture supernatants by ELISA.
Results
In vitro priming of conventional naïve CD4+CD25− T cells from DR52b+ donors by stimulation with peptide ESO119-143 and detection with DR52b/ESO119-143 tetramers
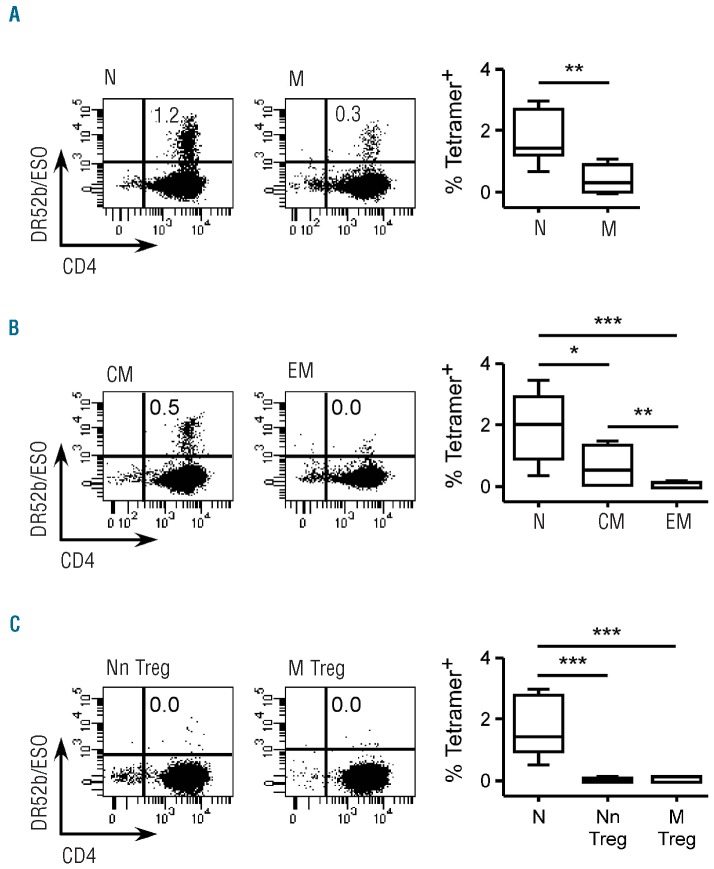
We have recently described the generation and validation of tetrameric complexes of DR52b, DRB3*0202, an alternate MHC class II molecule that is frequently expressed in Caucasians, incorporating the immunodominant peptide ESO119-143.28 In CD4+ T cells of patients immunized with an ESO vaccine, ESO119-143-specific DR52b-restricted cells were detected by DR52b/ESO119-143 tetramers ex vivo at an average frequency of 1/5,000 memory CD4+ T cells. In contrast, in DR52b+ healthy donors, DR52b/ESO119-143 tetramer+ cells were not detectable ex vivo, indicating that they are present in the pre-immune repertoire at frequencies close or below tetramer detection limit (in the range of 1/100,000 cells ex vivo). To assess the feasibility of generating ESO-specific lines from CD4+ T cells of healthy donors through in vitro priming, we enriched CD4+ T cells from DR52b+ donors by magnetic cell sorting and isolated conventional naïve (CD45RA+CCR7+CD25−) populations by flow cytometry cell sorting (Online Supplementary Figure S1). We stimulated isolated conventional naïve CD4+ T cells with peptide ESO119-143 in the presence of autologous APC and IL-2, cultured them for 12 days, and then assessed the cultures by staining with DR52b/ESO119-143 (DR52b/ESO thereafter) tetramers and anti-CD4 mAb. As illustrated in Figure 1A and summarized in Figure 1B, ESO-tetramer+ cells were readily detectable in the cultures from all DR52b+ donors assessed at an average frequency of 1.8 ± 1.1% of CD4+ T cells. No significant staining was obtained using DR52b tetramers incorporating an unrelated peptide from influenza matrix protein (DR52b/Flu). In addition, no significant ESO-tetramer staining was detected by assessing control CD4+ T-cell cultures from DR52b– donors. Together, these results demonstrate that in vitro priming of ESO-tetramer+ cells is consistently and efficiently obtained by antigen stimulation of CD4+ T cells from non-immune individuals.
Figure 1.
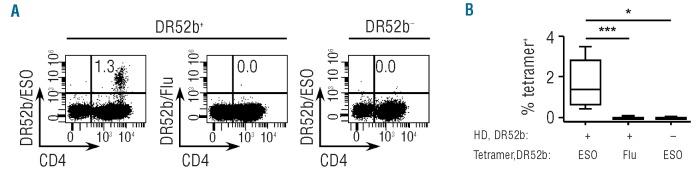
In vitro priming of ESO119-143-specific CD4+ T cells from conventional naïve CD4+CD25− T cells of DR52b+ healthy donors. Conventional naïve (N, CD45RA+CCR7+CD25–) cells isolated ex vivo by flow cytometry cell sorting from CD4+ T cells of DR52b+ (n=11) and DR52b- (n=3) healthy donors (HD), as shown in Online Supplementary Figure S1, were stimulated in vitro with peptide ESO119-143 in the presence of autologous APC and Day 12 cultures were stained with DR52b/ESO or control DR52b/Flu tetramers and anti-CD4 mAb, and analyzed by flow cytometry. (A) Dot plots for one DR52b+ and one DR52b− donor. (B) Percentages of tetramer+ cells in all cultures. Statistical analyses were performed using the Mann-Whitney test. *P<0.05; ***P<0.001.
ESO-tetramer+-TH can be efficiently generated through in vitro priming of conventional central memory but not effector memory or Treg populations
In some experiments, in parallel to conventional naïve CD4+ T-cell populations, we stimulated conventional memory (CD45RA–CCR7+/−CD25−) populations simultaneously isolated by cell sorting (Online Supplementary Figure S1). To our surprise, we obtained efficient in vitro priming of ESO-tetramer+ cells from conventional memory populations, although at lower frequencies (0.4±0.4%) as compared to those obtained with conventional naïve populations (Figure 2A). To further define the subpopulation containing ESO-specific TH precursors in the memory compartment, we separated conventional memory CD4+ T cells into CCR7+ central memory (CM) and CCR7– effector memory (EM) populations and stimulated them under the described conditions. We consistently obtained in vitro priming of ESO-specific TH from CM, at frequencies on average approximately 3-fold lower than those obtained with naïve populations (0.7±0.6%) (Figure 2B). In contrast, we obtained no significant in vitro priming from EM populations (Figure 2B) or from naïve or memory Treg populations (CD25+CD127low) simultaneously isolated and stimulated under the same conditions (Figure 2C)
Figure 2.
ESO119-143-specific CD4+ T cells can be generated from conventional central memory but not effector memory CD4+ T cells or Treg of DR52b+ healthy donors. (A) Conventional naïve (N, CD45RA+CCR7+CD25−) and memory (M, CD45RA−CCR7+/-CD25−) cells isolated ex vivo from CD4+ T cells of DR52b+ healthy donors (n=7), as shown in Online Supplementary Figure S1, were stimulated as in Figure 1 and stained with DR52b/ESO tetramers and anti-CD4 and analyzed by flow cytometry. Dot plots for one donor and data for all donors assessed are shown. (B) Conventional naïve (N, CD45RA+CCR7+CD25−) as well as central memory (CM, CD45RA−CCR7+CD25−) and effector memory (EM, CD45RA−CCR7−CD25−) cells, isolated ex vivo by cell sorting from CD4+ T cells of DR52b+ donors (n=9) as shown in Online Supplementary Figure S1, were assessed as in A. Dot plots for one donor and data for all donors assessed are shown. (C) NnTreg (CD45RA+CCR7+CD25+CD127low) and Mtreg(CD45RA−CCR7+/- CD25+CD127low) populations were isolated ex vivo by cell sorting from CD4+ T cells of DR52b+ donors (n=9), as shown in Online Supplementary Figure S1, and assessed as in A. Dot plots for one donor and data for all donors assessed are shown. Statistical analyses were performed using the Wilcoxon’s matched pairs test. *P<0.05; **P<0.01; ***P< 0.001.
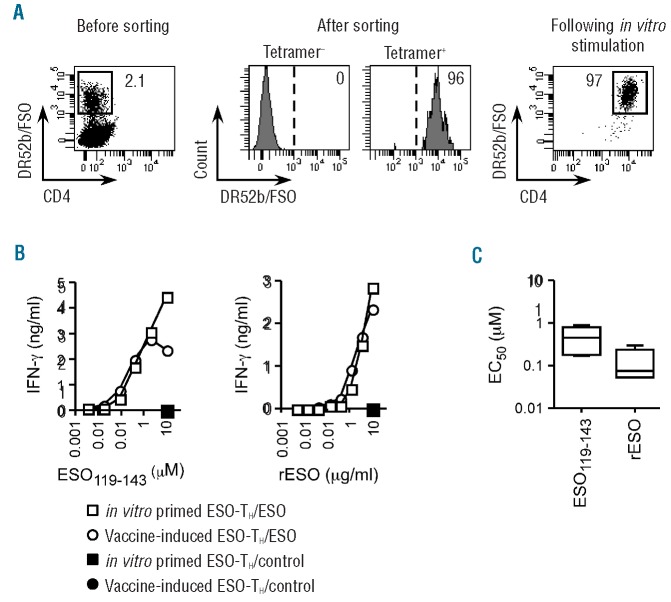
In vitro primed ESO-tetramer+-TH lines display a functional avidity similar to ESO-tetramer+-TH from patients immunized with an ESO vaccine and recognize full-length ESO antigen processed and presented by dendritic cells
To characterize ESO119-143-specific TH cells generated through in vitro priming, we isolated ESO-tetramer+ populations from Day 12 cultures by flow cytometry cell sorting, expanded them by mitogen stimulation in the presence of feeder cells, and assessed the resulting populations. The ESO-tetramer+-TH lines obtained through this procedure typically contained more than 90% of tetramer+ cells (Figure 3A). To assess the functional avidity of antigen recognition of in vitro primed ESO-tetramer+-TH lines, we incubated DR52b+ JBUSH cells with serial dilutions of peptide ESO119-143 and the in vitro primed lines and assessed IFN-γ secretion in the culture supernatant, by ELISA. As illustrated in Figure 3B, in vitro primed ESO-tetramer+-TH lines recognized peptide ESO119-143 with a similar functional avidity as ESO119-143 tetramer+-TH from a cancer patient immunized with a recombinant ESO vaccine. To assess the ability of in vitro primed lines to recognize, in addition to peptide ESO119-143, also the native ESO antigen, we incubated overnight DR52b+ DC, generated from isolated circulating CD14+ monocytes as described,31 with a recombinant full-length ESO protein, and assessed antigen recognition by the lines by measuring IFN-γ secretion.28 Similar to vaccine-induced ESO-TH, in vitro primed ESO-tetramer+-TH lines efficiently recognized the recombinant ESO protein processed and presented by DC, whereas they failed to recognize DC incubated with a control protein (Figure 3C).
Figure 3.
Isolation and functional assessment of ESO-tetramer+-TH lines generated from in vitro primed conventional naïve CD4+ T cells. (A) In vitro stimulated conventional naïve CD4+ T-cell cultures were stained with DR52b/ESO tetramers (left dot plot) and ESO-tetramer+ cells were isolated by flow cytometry cell sorting and expanded in vitro by mitogen stimulation. Aliquots of sorted tetramer+ and control tetramer− populations were analyzed by flow cytometry (middle histograms). Aliquots of TH lines obtained following in vitro expansion of sorted ESO-tetramer TH cells were stained with DR52b/ESO tetramers and anti-CD4 mAb and analyzed by flow cytometry (right dot plot). (B and C) ESO-tetramer+-TH lines obtained following in vitro priming and ESO119-143-specific DR52b-restricted TH obtained from cancer patients vaccinated with rESO were assessed functionally by incubation with either DR52b+ JBUSH cells and serial dilutions of ESO119-143 or control peptide or with DR52b+ DC pre-incubated with serial dilutions of full length recombinant ESO or control protein. IFN-γ was measured in 24-h culture supernatants by ELISA. (B) Examples of peptide and protein recognition. (C) Concentration of peptide and protein resulting in half maximal IFN-γ secretion (EC50) for all in vitro primed ESO-tetramer+- TH lines assessed (n=6).
In vitro primed ESO-tetramer+-TH lines frequently use TCR BV2 similar to ESO-tetramer+-TH from patients immunized with an ESO vaccine
We have previously shown that ESO119-143-specific DR52b-restricted CD4+ T cells induced in vivo by vaccination with a recombinant ESO vaccine exhibit a significant conservation of TCR usage among different individuals, with frequent usage of the TCR BV2.28,30 To assess if this restricted BV usage was a shared feature of DR52b/ESO119-143 tetramer+ populations generated through in vitro priming, we co-stained Day 12 cultures with tetramers and anti-BV2 antibodies. We found a high enrichment of BV2 usage in ESO-tetramer+ cells in in vitro primed cultures (Figure 4A and B). Indeed, whereas BV2+ cells represented, on average, 9.2% (range 6-11.8%) of total CD4+ T cells in the cultures, they represented on average 45% (range 10.9-82%) of in vitro primed ESO-tetramer+ cells. This was comparable to 40.6% of BV2+ among ESO-tetramer+ cells in patients vaccinated with the ESO protein.28 Therefore, ESO119-143-specific DR52b-restricted CD4+ T cells obtained through in vitro priming exhibited a TCR repertoire that frequently used BV2 similar to that found in patients immunized with the recombinant ESO vaccine.
Figure 4.
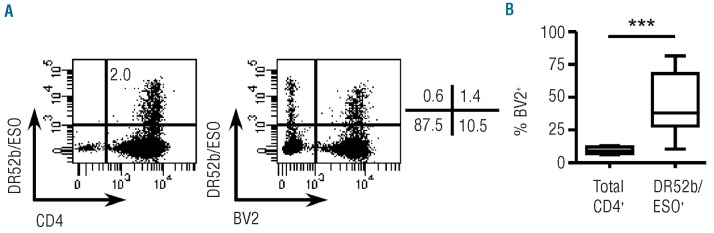
Analysis of TCR BV usage by in vitro primed ESO-tetramer+-TH. Conventional naïve CD4+ T cells from healthy donors were stimulated with peptide ESO119-143 as in Figure 1A and Day 12 cultures were stained with DR52b/ESO tetramers and anti-CD4 and anti-BV2 mAb and analyzed by flow cytometry. (A) Examples of dot plots showing tetramer staining versus anti-CD4 staining in the culture and anti-BV2 staining versus tetramer staining gated on CD4+ cells. (B) The proportions of BV2+ cells in total CD4+ cells in the cultures and in ESO-tetramer+ cells are summarized for all donors assessed (n=11). Statistical analyses were performed using Wilcoxon’s matched pairs test. ***P<0.001.
In virto primed ESO-tetramer+-TH lines secrete IL-2 and effector cytokines and do not recognize allogeneic dendritic cells
To further characterize the ESO-tetramer+-TH lines generated by in vitro priming functionally, we assessed their cytokine secretion profile, by intracellular staining using cytokine-specific mAb, following stimulation with the antigen. Importantly, we found that large proportions of cells in the ESO-tetramer+-TH lines were able to produce IL-2 in response to stimulation with the antigen, suggesting that the lines could support their own growth (Figure 5A). Slightly lower, but significant, proportions of them produced TNF-α and IFN-γ (Figure 5A). IL-4, but not IL-17 or IL-10, was also produced by smaller proportions of cells in the ESO-tetramer+-TH lines (Figure 5A). Because the in vitro primed lines were strictly selected by a single MHC class II allele/peptide complex, their potential alloreactivity was expected to be low. To provide evidence in support of this, we stimulated the lines in the presence of DC from a panel of 8 DR52b-expressing donors that had been pre-incubated or not with peptide ESO119-143, and assessed IFN-γ secretion in the culture supernatants. As shown in Figure 5B, we observed significant recognition of allo-DC only in the presence of the ESO peptide.
Figure 5.
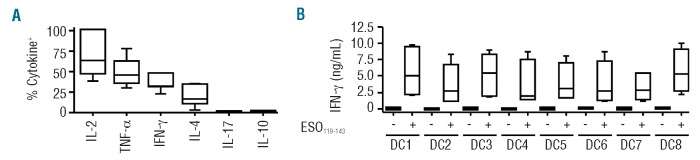
Assessment of cytokine production and alloreactivity of ESO-tetramer+-TH lines generated from in vitro primed conventional naïve CD4+ T cells. (A) ESO-tetramer+-TH lines (n=7) obtained as in Figure 3A from in vitro primed conventional naïve CD4+ T cells were stimulated with peptide ESO119-143 and cytokine production was assessed in a 4-h intracellular cytokine staining assay and analyzed by flow cytometry. The proportions of cells producing the indicated cytokines are summarized for all ESO-tetramer+-TH lines tested. (B) ESO-tetramer+-TH lines (n=6) were incubated with DC from 8 DR52b+ allogeneic healthy donors in the absence or presence of peptide ESO119-143 and IFN-γ was measured in 24-h culture supernatants by ELISA.
Discussion
Recent data encourage us to explore the therapeutic potential of ACT using tumor-specific CD4+ T cells.15-17 There are, however, many problems in the development of this approach, including that of generating tumor-specific CD4+ T clones for all patients. To overcome this limitation, in this study, we have explored the possibility of generating ESO-specific TH lines for ACT through in vitro priming of defined CD4+ T-cell populations from non-immune individuals and isolation with DR52b/ESO119-143 tetramers that we have recently developed.28 We have shown that a single stimulation of circulating conventional naïve CD4+ T cells from DR52b+ donors with peptide ESO119-143, that is highly immunodominant,30 results in the consistent and efficient in vitro priming of ESO-tetramer+ populations, comprising on average more than 1% of CD4+ T cells in Day 12 cultures. Based on this frequency, it can be estimated that ESO119-143-specific DR52b-restricted precursors represent on average approximately 1×10-5 of circulating conventional naïve CD4+ T cells in DR52b-expressing individuals. This frequency, in the range of the limit of detection by the tetramers, explains our inability to detect ESO119-143-specific DR52b-restricted precursors ex vivo. The high specificity of our stimulation and detection methods was confirmed by the lack of detectable responses when stimulating CD4+ T cells from DR52b− donors, along with the lack of significant staining obtained with control tetramers.
After isolation of ESO-tetramer+ populations by flow cytometry cell sorting and expansion by mitogen stimulation, polyclonal monospecific ESO-tetramer+-TH lines were consistently obtained. In support of their physiological relevance, the ESO-tetramer+-TH lines generated through in vitro priming displayed a functional avidity of antigen recognition similar to that of ESO TH from patients immunized with a recombinant ESO vaccine, showed, similar to the latter, a preferential use of TCR BV2, and were able to recognize the full-length ESO antigen processed and presented by DC. In addition, ESO-tetramer+-TH lines produced IL-2, which is in line with their origin from naïve cells, as well as effector cytokine, but did not produce IL-10. Together, these functional characteristics suggest that these populations may be particularly efficient, upon in vivo transfer, at inducing anti-tumor responses.
A surprising finding of our study was the efficient in vitro priming obtained with conventional memory populations from healthy donors, who have no spontaneous immune responses to ESO, as supported by the lack of detectable levels of ESO-specific serum antibodies in this population.32 A comparative assessment of conventional memory subpopulations separated according to their differentiation stage, namely CCR7+ (CM) versus CCR7− (EM), revealed that in vitro priming was consistently obtained with CM cells, a population at an early differentiation stage, but not from EM cells. Interestingly, ESO-tetramer+ lines derived from CM cells exhibited a functional avidity of antigen recognition similar to those derived from naïve populations (data not shown). Human CD4+ CM cells are heterogeneous, including pre-TH1, pre-TH2 and non-polarized populations,33 and it has been shown that DC activated by thymic stromal lymphopoietin stimulate the homeostatic expansion of autologous naïve CD4+ T cells that adopt and maintain a CM phenotype and a polyclonal repertoire.34 Thus, our interpretation of these data is that ESO-specific CD4+ precursors in CM cells could be part of a population that is phenotypically memory but is generated through homeostatic expansion of naïve cells and maintain a similar TCR repertoire composition. This concept, however, still has to be confirmed in further studies, including comparative studies in self- and pathogen-derived antigenic systems. We obtained no significant in vitro priming of ESO-specific CD4+ T cells with CD25+ Treg, both naïve and memory. Treg have been shown by others and us to be enriched in self-reactive cells,35,36 but their antigen specificity is still largely unknown. Thus, although CD4+ T-cell precursors specific for other tumor associated self-antigens may be present at high frequency among Treg, this does not appear to be the case for ESO, which further supports its relevance for use in immunotherapy. However, because Treg have a more limited capacity of expansion in vitro as compared to conventional CD4+ T cells, this result should be considered with caution, and confirmed by assessment through additional approaches.
Because ESO is specifically and frequently expressed in many cancers, both solid and hematologic, ACT with in vitro primed ESO-specific TH lines could be of use in many different clinical settings. For example, their use in the autologous setting, as currently used for melanoma, will extend this application to patients with ESO-expressing tumors but no detectable spontaneous immune response to the antigen. However, we suggest that a major application could be in hematologic tumors such as MM or ATLL, where ESO is frequently expressed, particularly in patients with poor prognosis.6 MM patients who relapse following chemotherapy receive allogeneic stem cell transplantation (alloSCT), and, if they experience further relapse, transfer of additional T cells from the original donor, a therapy known as donor leukocyte infusion. Interestingly, it has been shown that some MM patients who had received alloSCT develop strong antibody as well as CD4+ T-cell responses to CTA, including ESO, suggesting that CTA might represent natural targets for graft-versus-myeloma (GVM) effects.7 Thus, the use of in vitro-primed allogeneic ESO-specific TH lines could represent a very efficient way to guarantee a strong GVM effect in this patient population. Interestingly, with the exception of melanoma, most solid and hematologic malignancies, including MM, do not express MHC class II molecules and cannot, therefore, be directly recognized by tumor antigen-specific CD4+ T cells. The latter, however, have been shown to be able to mediate anti-tumor immune responses through different mechanisms37 including by providing help for the initiation and maintenance of CD8+ T-cell and antibody responses,38 as well as through direct mechanisms in the effector phase of tumor rejection mediated by effector cytokines such as IFN-γ.39,40 This, together with recent studies in cancer patients and in mouse models that have reported tumor regression following ACT therapy with tumor antigen-specific CD4+ T cells, encourage the implementation of studies assessing the efficacy of ACT of tumor antigen-specific TH, alone or in combination with CTL, for the treatment of patients bearing antigen-expressing tumors of different histological types.
Because the specificity of the lines is strictly selected on a single MHC class II allele/peptide complex, their alloreactivity was expected to be limited, although this would need to be confirmed in each case. In support of the limited potential alloreactivity of the in vitro primed ESO-specific lines, we failed to detect significant recognition of DC from DR52b+ donors in the absence of antigen. Interestingly, a similar approach using allogeneic EBV-specific CTL from donors is currently used to treat patients with EBV+ post-transplantation lymphoproliferative disease and has been found to be safe, with very rare occurrence of allo-responses.41 Even in cases in which a degree of alloreactivity is detected, they could still be used through the development of appropriate pre-conditioning regimens, as recently shown in the case of “T-bodies”, re-directed T cells bearing fusion receptor proteins composed of single chain tumor-specific antibodies linked to the signaling part of the TCR.42
Together, the results of this study indicate that an ACT approach based on the use of ESO-specific CD4+ TH generated through in vitro priming is feasible and could be applicable to the treatment of large numbers of patients bearing ESO-expressing cancers. In addition, we expect that the implementation of this versatile in vitro platform, allowing the efficient and consistent priming of tumor-specific CD4+ T cells, will allow further pre-clinical studies to explore the possibility of generating tumor-specific TH lines of different types (e.g. TH1, TH17) and it would be very interesting to compare these in terms of clinical efficacy.43 They could be used in the pre-clinical evaluation of the effects of different vaccine adjuvants (such as TLR ligands)11,44 or immunomodulatory molecules45 that are increasingly used in clinical cancer immunotherapy trials.
Acknowledgments
The authors would like to thank Mrs. P. Pignon and I. Raimbaud for their technical assistance.
Funding: The study was supported by the Ludwig Institute for Cancer Research and the Cancer Research Institute (USA). CP was supported through a fellowship from the Fondation pour la Recherche Médicale (France) and CR from the Ligue Contre le Cancer, France.
Footnotes
The online version of this article has a Supplementary Appendix.
Authorship and Disclosures: Information on authorship, contributions, and financial & other disclosures was provided by the authors and is available with the online version of this article at www.haematologica.org.
References
- 1.Chen YT, Scanlan MJ, Sahin U, Tureci O, Gure AO, Tsang SL, et al. A testicular antigen aberrantly expressed in human cancers detected by autologous antibody screening. Proc Natl Acad Sci USA. 1997;94 (5):1914-8 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 2.Simpson AJ, Caballero OL, Jungbluth A, Chen YT, Old LJ. Cancer/testis antigens, gametogenesis and cancer. Nat Rev Cancer. 2005;5 (8):615-25 [DOI] [PubMed] [Google Scholar]
- 3.Rimoldi D, Rubio-Godoy V, Dutoit V, Lienard D, Salvi S, Guillaume P, et al. Efficient simultaneous presentation of NY-ESO-1/LAGE-1 primary and nonprimary open reading frame-derived CTL epitopes in melanoma. J Immunol. 2000; 165 (12):7253-61 [DOI] [PubMed] [Google Scholar]
- 4.Odunsi K, Jungbluth AA, Stockert E, Qian F, Gnjatic S, Tammela J, et al. NY-ESO-1 and LAGE-1 cancer-testis antigens are potential targets for immunotherapy in epithelial ovarian cancer. Cancer Res. 2003;63 (18):6076-83 [PubMed] [Google Scholar]
- 5.van Baren N, Brasseur F, Godelaine D, Hames G, Ferrant A, Lehmann F, et al. Genes encoding tumor-specific antigens are expressed in human myeloma cells. Blood. 1999;94 (4):1156-64 [PubMed] [Google Scholar]
- 6.van Rhee F, Szmania SM, Zhan F, Gupta SK, Pomtree M, Lin P, et al. NY-ESO-1 is highly expressed in poor-prognosis multiple myeloma and induces spontaneous humoral and cellular immune responses. Blood. 2005;105 (10):3939-44 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 7.Atanackovic D, Arfsten J, Cao Y, Gnjatic S, Schnieders F, Bartels K, et al. Cancer-testis antigens are commonly expressed in multiple myeloma and induce systemic immunity following allogeneic stem cell transplantation. Blood. 2007;109 (3):1103-12 [DOI] [PubMed] [Google Scholar]
- 8.van Duin M, Broyl A, de Knegt Y, Goldschmidt H, Richardson PG, Hop WC, et al. Cancer testis antigens in newly diagnosed and relapse multiple myeloma: prognostic markers and potential targets for immunotherapy. Haematologica. 2011; 96 (11):1662-9 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 9.Nishikawa H, Maeda Y, Ishida T, Gnjatic S, Sato E, Mori F, et al. Cancer/testis antigens are novel targets of immunotherapy for adult T-cell leukemia/lymphoma. Blood. 2012;119 (13): 3097-104 [DOI] [PubMed] [Google Scholar]
- 10.Jager E, Karbach J, Gnjatic S, Neumann A, Bender A, Valmori D, et al. Recombinant vaccinia/fowlpox NY-ESO-1 vaccines induce both humoral and cellular NY-ESO-1-specific immune responses in cancer patients. Proc Natl Acad Sci USA. 2006; 103 (39):14453-8 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 11.Valmori D, Souleimanian NE, Tosello V, Bhardwaj N, Adams S, O'Neill D, et al. Vaccination with NY-ESO-1 protein and CpG in Montanide induces integrated antibody/Th1 responses and CD8 T cells through cross-priming. Proc Natl Acad Sci USA. 2007;104 (21): 8947-52 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 12.Dudley ME, Wunderlich JR, Yang JC, Sherry RM, Topalian SL, Restifo NP, et al. Adoptive cell transfer therapy following non-myeloablative but lymphodepleting chemotherapy for the treatment of patients with refractory metastatic melanoma. J Clin Oncol. 2005;23 (10):2346-57 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 13.Yee C, Thompson JA, Byrd D, Riddell SR, Roche P, Celis E, et al. Adoptive T cell therapy using antigen-specific CD8+ T cell clones for the treatment of patients with metastatic melanoma: in vivo persistence, migration, and antitumor effect of transferred T cells. Proc Natl Acad Sci USA. 2002;99 (25):16168-73 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 14.Mackensen A, Meidenbauer N, Vogl S, Laumer M, Berger J, Andreesen R. Phase I study of adoptive T-cell therapy using antigen-specific CD8+ T cells for the treatment of patients with metastatic melanoma. J Clin Oncol. 2006;24 (31):5060-9 [DOI] [PubMed] [Google Scholar]
- 15.Hunder NN, Wallen H, Cao J, Hendricks DW, Reilly JZ, Rodmyre R, et al. Treatment of metastatic melanoma with autologous CD4+ T cells against NY-ESO-1. N Engl J Med. 2008;358 (25):2698-703 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 16.Xie Y, Akpinarli A, Maris C, Hipkiss EL, Lane M, Kwon EK, et al. Naive tumor-specific CD4(+) T cells differentiated in vivo eradicate established melanoma. J Exp Med. 2010;207 (3): 651-67 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 17.Quezada SA, Simpson TR, Peggs KS, Merghoub T, Vider J, Fan X, et al. Tumor-reactive CD4(+) T cells develop cytotoxic activity and eradicate large established melanoma after transfer into lymphopenic hosts. J Exp Med. 2010;207 (3):637-50 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 18.Muranski P, Restifo NP. Adoptive immunotherapy of cancer using CD4(+) T cells. Curr Opin Immunol. 2009;21 (2):200-8 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 19.Riddell SR, Greenberg PD. The use of anti-CD3 and anti-CD28 monoclonal antibodies to clone and expand human antigen-specific T cells. J Immunol Methods. 1990;128 (2):189-201 [DOI] [PubMed] [Google Scholar]
- 20.Dudley ME, Gross CA, Langhan MM, Garcia MR, Sherry RM, Yang JC, et al. CD8+ enriched “young” tumor infiltrating lymphocytes can mediate regression of metastatic melanoma. Clin Cancer Res. 2010;16 (24):6122-31 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 21.Hinrichs CS, Borman ZA, Gattinoni L, Yu Z, Burns WR, Huang J, et al. Human effector CD8+ T cells derived from naive rather than memory subsets possess superior traits for adoptive immunotherapy. Blood. 2011;117 (3):808-14 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 22.Antony PA, Piccirillo CA, Akpinarli A, Finkelstein SE, Speiss PJ, Surman DR, et al. CD8+ T cell immunity against a tumor/self-antigen is augmented by CD4+ T helper cells and hindered by naturally occurring T regulatory cells. J Immunol. 2005;174 (5):2591-601 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 23.Altman JD, Moss PAH, Goulder PJR, Barouch DH, McHeyzer-Williams MG, Bell JI, et al. Phenotypic analysis of antigen-specific T lymphocytes. Science. 1996;274:94-6 [PubMed] [Google Scholar]
- 24.Guillaume P, Dojcinovic D, Luescher IF. Soluble MHC-peptide complexes: tools for the monitoring of T cell responses in clinical trials and basic research. Cancer Immun. 2009;9:7. [PMC free article] [PubMed] [Google Scholar]
- 25.Nepom GT, Buckner JH, Novak EJ, Reichstetter S, Reijonen H, Gebe J, et al. HLA class II tetramers: tools for direct analysis of antigen-specific CD4+ T cells. Arthritis Rheum. 2002; 46 (1):5-12 [DOI] [PubMed] [Google Scholar]
- 26.Vollers SS, Stern LJ. Class II major histocompatibility complex tetramer staining: progress, problems, and prospects. Immunology. 2008; 123 (3):305-13 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 27.Cecconi V, Moro M, Del Mare S, Dellabona P, Casorati G. Use of MHC class II tetramers to investigate CD4+ T cell responses: problems and solutions. Cytometry A. 2008;73 (11):1010-8 [DOI] [PubMed] [Google Scholar]
- 28.Ayyoub M, Dojcinovic D, Pignon P, Raimbaud I, Schmidt J, Luescher I, et al. Monitoring of NY-ESO-1 specific CD4+ T cells using molecularly defined MHC class II/His-tag-peptide tetramers. Proc Natl Acad Sci USA. 2010;107 (16):7437-42 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 29.Ayyoub M, Pignon P, Dojcinovic D, Raimbaud I, Old LJ, Luescher I, et al. Assessment of vaccine-induced CD4 T cell responses to the 119-143 immunodominant region of the tumor-specific antigen NY-ESO-1 using DRB1*0101 tetramers. Clin Cancer Res. 2010;16 (18):4607-15 [DOI] [PubMed] [Google Scholar]
- 30.Bioley G, Dousset C, Yeh A, Dupont B, Bhardwaj N, Mears G, et al. Vaccination with recombinant NY-ESO-1 protein elicits immunodominant HLA-DR52b-restricted CD4+ T cell responses with a conserved T cell receptor repertoire. Clin Cancer Res. 2009;15 (13):4467-74 [DOI] [PubMed] [Google Scholar]
- 31.Sallusto F, Lanzavecchia A. Efficient presentation of soluble antigen by cultured human dendritic cells is maintained by granulocyte/macrophage colony-stimulating factor plus interleukin 4 and downregulated by tumor necrosis factor α. J Exp Med. 1994;179 (4):1109-18 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 32.Hamai A, Duperrier-Amouriaux K, Pignon P, Raimbaud I, Memeo L, Colarossi C, et al. Antibody responses to NY-ESO-1 in primary breast cancer identify a subtype target for immunotherapy. PLoS One. 2011;6 (6):e21129. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 33.Rivino L, Messi M, Jarrossay D, Lanzavecchia A, Sallusto F, Geginat J. Chemokine receptor expression identifies Pre-T helper (Th)1, Pre-Th2, and nonpolarized cells among human CD4+ central memory T cells. J Exp Med. 2004;200 (6):725-35 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 34.Watanabe N, Hanabuchi S, Soumelis V, Yuan W, Ho S, de Waal Malefyt R, et al. Human thymic stromal lymphopoietin promotes dendritic cell-mediated CD4+ T cell homeostatic expansion. Nat Immunol. 2004;5 (4):426-34 [DOI] [PubMed] [Google Scholar]
- 35.Valmori D, Merlo A, Souleimanian NE, Hesdorffer CS, Ayyoub M. A peripheral circulating compartment of natural naive CD4 Tregs. J Clin Invest. 2005;115 (7):1953-62 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 36.Rivino L, Gruarin P, Haringer B, Steinfelder S, Lozza L, Steckel B, et al. CCR6 is expressed on an IL-10-producing, autoreactive memory T cell population with context-dependent regulatory function. J Exp Med. 2010;207 (3):565-77 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 37.Wang RF. The role of MHC class II-restricted tumor antigens and CD4+ T cells in antitumor immunity. Trends Immunol. 2001;22 (5):269-76 [DOI] [PubMed] [Google Scholar]
- 38.Ossendorp F, Mengede E, Camps M, Filius R, Melief CJ. Specific T helper cell requirement for optimal induction of cytotoxic T lymphocytes against major histocompatibility complex class II negative tumors. J Exp Med. 1998;187 (5):693-702 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 39.Mumberg D, Monach PA, Wanderling S, Philip M, Toledano AY, Schreiber RD, et al. CD4(+) T cells eliminate MHC class II-negative cancer cells in vivo by indirect effects of IFN-γ. Proc Natl Acad Sci USA. 1999; 96 (15):8633-8 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 40.Corthay A, Skovseth DK, Lundin KU, Rosjo E, Omholt H, Hofgaard PO, et al. Primary antitumor immune response mediated by CD4+ T cells. Immunity. 2005; 22 (3):371-83 [DOI] [PubMed] [Google Scholar]
- 41.Haque T, Wilkie GM, Jones MM, Higgins CD, Urquhart G, Wingate P, et al. Allogeneic cytotoxic T-cell therapy for EBV-positive posttrans-plantation lymphoproliferative disease: results of a phase 2 multicenter clinical trial. Blood. 2007;110 (4): 1123-31 [DOI] [PubMed] [Google Scholar]
- 42.Marcus A, Waks T, Eshhar Z. Redirected tumor-specific allogeneic T cells for universal treatment of cancer. Blood. 2011;118 (4): 975-83 [DOI] [PubMed] [Google Scholar]
- 43.Muranski P, Boni A, Antony PA, Cassard L, Irvine KR, Kaiser A, et al. Tumor-specific Th17-polarized cells eradicate large established melanoma. Blood. 2008;112 (2):362-73 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 44.van Duin D, Medzhitov R, Shaw AC. Triggering TLR signaling in vaccination. Trends Immunol. 2006;27 (1):49-55 [DOI] [PubMed] [Google Scholar]
- 45.Peggs KS, Quezada SA, Korman AJ, Allison JP. Principles and use of anti-CTLA4 antibody in human cancer immunotherapy. Curr Opin Immunol. 2006;18 (2):206-13 [DOI] [PubMed] [Google Scholar]