Background: FAK has both kinase and scaffolding functions.
Results: Disruption of the function of FAK scaffolding to mediate endophilin A2 phosphorylation inhibits mammary tumor growth and metastasis in vivo by decreasing tumor cell markers for EMT and their MaCSCs activities.
Conclusion: The function of FAK scaffolding is important for promoting mammary tumor progression.
Significance: Targeting the scaffolding function of FAK may be important in breast cancer therapy.
Keywords: Breast Cancer, Epithelial Mesenchymal Transition, Mouse Genetics, Signal Transduction, Tyrosine Kinase
Abstract
Tyrosine kinases have been shown to play critical roles in cancer development and progression, and their inhibitors hold the potential as effective targeted therapies for breast and other cancers. However, some of these kinases like focal adhesion kinase (FAK) also possess scaffolding functions in intracellular signaling, but such kinase-independent functions of FAK or other kinases have not been examined in cancer directly in vivo. Here, we report that disruption of the function of FAK scaffolding through its Pro-878/881 motif suppressed mammary tumor growth and metastasis in a well characterized murine model of human breast cancer. P878A/P881A mutation in the endogenous FAK gene decreased the expression of markers for epithelial-mesenchymal transition (EMT) and mammary cancer stem cell (MaCSC) activities in tumors derived from mutant mice. This mutation disrupted the function of FAK scaffolding to mediate endophilin A2 phosphorylation at Tyr-315 by Src, leading to the decreased surface expression of MT1-MMP, as observed previously in transformed fibroblasts in vitro. Inhibition of the downstream components of this FAK scaffolding function by Y315F endophilin A2 mutant or MT1-MMP knockdown reduced markers for EMT and MaCSC activities. Conversely, bypass of the scaffolding function using the phosphorylation mimic mutant Y315E endophilin A2 or endophilin A2 knockdown rescued the decreased markers for EMT and MaCSCs as well as surface expression of MT1-MMP in tumor cells harboring the P878A/P881A mutation. Together, these results identify a novel role of FAK scaffolding function in breast cancer, which could serve as a new target in combination with kinase inhibition for more effective treatment strategies.
Introduction
Breast cancer exhibits enormous cellular, genomic, and biological complexity wherein multiple gene aberrations act to drive tumorigenesis and cancer progression (1). Although not defined as classical oncogenes or tumor suppressor genes, there is mounting evidence suggesting that many other genes and their associated signaling pathways can regulate tumor initiation and progression (2, 3). It remains as a significant challenge to elucidate the roles and mechanisms of these molecules and their highly interactive signaling pathways in breast cancer and other malignancies. FAK3 is a nonreceptor tyrosine kinase that mediates signal transduction by integrins and other cell surface receptors to regulate cell adhesion, migration, survival, proliferation, and differentiation in a variety of cells (4–8). A large number of previous studies have strongly implicated FAK in the development and progression of breast and other cancers (9, 10). Recently, several groups, including us, have shown that conditional deletion of FAK suppresses tumor formation and progression in mouse models of breast as well as skin cancers (11–15).
Despite the success in establishing a role of FAK in breast cancer, the cellular and molecular mechanisms by which FAK promotes mammary tumorigenesis in vivo are still not well understood. One important issue is the unique role of FAK as both a kinase and a scaffold in mediating various intracellular signaling events. FAK has been shown to directly phosphorylate Grb7 and N-WASP (16, 17). However, it also serves as a scaffold in mediating Src phosphorylation of important targets like p130Cas, paxillin, and endophilin A2 (18–22). The interaction of FAK with endophilin A2, which was recently identified in our laboratory, facilitates the phosphorylation of endophilin A2 by Src, which inhibits endocytosis of MT1-MMP and thereby increases cell invasion in transformed fibroblasts (22). Nevertheless, the potential roles and mechanisms by which this specific FAK scaffolding function contributes to mammary tumorigenesis and progression in vivo remain largely unknown.
In addition to the well established role of FAK in cell survival, proliferation, and migration, recent studies have also revealed potentially novel functions of FAK in the regulation of epithelial-mesenchymal transition (EMT), an important developmental program exploited by cancer cells in their acquisition of invasive and metastatic capacity (10, 23, 24). For example, the expression of FAK mutants resistant to Src phosphorylation significantly decreases Src-mediated disruption of E-cadherin-based cell contacts in colon cancer cells (25). TGF-β-induced EMT has been shown to be mediated by Src or integrin-dependent FAK activation, which results in E-cadherin down-regulation in mouse epithelial cells and hepatocytes (26–30). Increased expression of FAK has also been documented to correlate with the loss of E-cadherin in nodal metastases of laryngeal tumors (31). Interestingly, a number of recent studies have linked many characteristics of MaCSCs to epithelial cells that have undergone EMT (32–35). Our recent study also suggested that FAK may promote mammary tumorigenesis and progression through its effects on MaCSCs (14). Therefore, it is possible that the role of FAK in promoting EMT may closely link to its function in maintaining MaCSCs in breast cancer.
In this paper, we created and analyzed FAK knock-in mice with a P878A/P881A mutation in the MMTV-PyMT mouse model of human breast cancer to investigate the potential role and mechanisms of FAK scaffolding function through Pro-878/881 in breast cancer development and progression in vivo. Our studies showed that disruption of the function of FAK scaffolding to mediate endophilin A2 phosphorylation at Tyr-315 significantly reduced mammary tumor growth and metastasis by decreasing the markers for EMT and impairing tumor cell MaCSC activities. These results provide significant insights into the molecular and cellular mechanisms of FAK signaling through its scaffolding functions in breast cancer development and progression, which may contribute to the future development of novel therapies for breast cancer, especially in combination with inhibitors for kinase activity of FAK to target both kinase-dependent and -independent functions of FAK.
EXPERIMENTAL PROCEDURES
Mice and Genotyping
Based on available mouse genome sequences in the Ensembl database, an isogenic 129SvJ mouse BAC genomic clone containing FAK exon 28 (where the second C-terminal proline-rich motif is encoded) and flanking sequences were obtained from BACPAC Resources Center. The presence of exon 28 in the BAC clone was verified by PCR using two pairs of primers surrounding exon 28 as well as Southern blotting. A targeting vector containing a mutated exon 28 (P878A/P881A) and a neocassette was then constructed for homologous recombination (supplemental Fig. S1A). Gene targeting in 129P2/OlaHsd-derived E14Tg2a mouse embryonic stem cells (36) was performed as described previously (37) with the use of ESGRO (Chemicon, Temecula, CA). Chimeric mice were identified by coat color and then bred to C57BL/6J mice. Transmission of the germ line was identified by PCR and confirmed by Southern blotting (supplemental Fig. S1B). Heterozygous targeted mice bearing PA[neo] allele (+/PA[neo] mice) were obtained and then crossed with EIIa-Cre mice (The Jackson Laboratory), which express Cre in the very early stage of embryogenesis (38), to delete the neomycin cassette to avoid its possible interference with FAK gene expression. The progenies with neomycin cassette removed (+/PA;EIIa-Cre mice) were crossed with C57BL/6J mice to segregate the FAK PA allele from the heterozygote EIIa-Cre transgene. The resulting heterozygous and homozygous FAK P878A/P881A mutation knock-in mice (+/PA and PA/PA mice) were identified by PCR analysis and confirmed by sequencing of tail DNA (supplemental Fig. S1, C and D).
MMTV-PyMT transgenic mice (39) were obtained from the mouse repository of Mouse Models of Human Cancers Consortium at NCI (National Institutes of Health) and have been described previously (14). Mice genotyping for PyMT and wild type FAK alleles were performed as described previously (14, 39). Primers used to genotype FAK P878A/P881A mutation knock-in alleles were P1, 5′-GTCAGAAAATTAGGTATGGT-3′, and P2, 5′-TTTTGTATATCTGCATGACT-3′, and PCR was performed for 30 cycles of 94 °C for 3 min, 55 °C for 2 min, and 72 °C for 4 min. For detection of mammary tumors, mice were palpated every 7 days after weaning, and the size of the tumors was measured with a caliper and recorded. Mice were housed and handled according to local, state, and federal regulations, and all experimental procedures were carried out according to the guidelines of the Institutional Animal Care and Use Committee at the University of Michigan.
Histology, Immunohistochemistry, and Immunofluorescent Labeling
Mammary tumors or lungs were harvested from mice and subjected to analysis by H&E staining or immunohistochemistry using antibodies for E-cadherin (1:250, Cell Signaling), and the micrometastatic nodules per section were quantified, as described previously (14, 40). Cultured primary tumor cells (see below) were subjected to analysis by immunofluorescent labeling using antibodies for E-cadherin (1:250, Cell Signaling) or vimentin (1:150, Santa Cruz Biotechnology), as described previously (41). Texas Red or FITC-labeled secondary antibodies (The Jackson Laboratory) were used at a dilution of 1:250 for immunofluorescence, and nuclei were counterstained with 4′,6-diamidino-2-phenylindole/antifade (Invitrogen).
Preparation of Mammary Tumor Cells and Analysis of Content and Activity of MaCSCs
Mammary tumor cells from +/PA-MT and PA/PA-MT mice were prepared as described previously (14). To determine the content of MaCSCs, the cells were subjected to flow cytometry analysis using Lin−ALDH+ or Lin−CD24+CD29hiCD61hi as markers, as described previously (14). They were also measured for their MaCSC activities using tumor sphere formation assays in vitro and limiting dilution cell transplantation assays in vivo, as described previously (14, 40). In some experiments, MMP inhibitor Marimastat (Tocris Bioscience) was added to the medium at the beginning of tumor sphere formation assays.
Analysis of Tumor Cell Proliferation and Migration in Vitro
Primary mammary tumor cells were analyzed for their proliferation and migration using BrdU incorporation and modified Boyden chamber (Neuro Probe) assays, respectively, as described previously (14, 42).
Experimental Metastasis Assay Using Tail Vein Injection
Tail vein injection experiments to assess metastatic activity of mammary tumor cells were performed as described previously (40). In some experiments, MMP inhibitor Marimastat was administered orally at a dose of 10 mg/kg daily to the recipient mice starting at the day of injections.
Immunoprecipitation, Cell Surface Biotinylation, and Western Blotting
Preparation of lysates from mammary tumor cells and analysis of protein samples in cell lysates and tissue extracts by SDS-PAGE and Western blotting were as described previously (14). Immunoprecipitation and Western blotting were performed using the following antibodies: FAK (1:500; Santa Cruz Biotechnology); endophilin A2 (1:500; Santa Cruz Biotechnology); phosphotyrosine (1:1000; 4G10 from Millipore); E-cadherin (1:1000; Cell Signaling); N-cadherin (1:1000; BD Biosciences); fibronectin (1:500; Sigma); vimentin (1:500; Santa Cruz Biotechnology); vinculin (1:1000; Sigma), and actin (1:1000; Sigma). To determine the cell surface level of MT1-MMP, biotin-labeled cells were lysed, precipitated with streptavidin-conjugated agarose beads, and subjected to Western blotting with anti-MT1-MMP antibody (1:500; Epitomics), as described previously (22).
Recombinant Viruses and Infection of Tumor Cells
The psPAX2, pMD2G, and pGIPZ lentiviral vectors encoding shRNA targeting MT1-MMP or endophilin A2 (MT1-MMP shRNA-1, catalogue no. RMM4431-99010132; MT1-MMP shRNA-2, catalogue no. RMM4431-99951199; endophilin A2 shRNA-1, catalogue no. RMM4431-98857904, and endophilin A2 shRNA-2, catalogue no. RMM4431-98723516) were purchased through the University of Michigan shRNA core facility (Open Biosystems, Huntsville, AL). HEK293 cells were transfected with these three vectors, and the media were collected 12 h after transfection. After centrifugation and filtration, the supernatant was used to infect mammary tumor cells or sorted MaCSCs. The recombinant adenoviruses encoding endophilin A2 mutants Y315F or Y315E or GFP control were generated using the AdEasy-1 system (Stratagene) as described previously (22). Mammary tumor cells or sorted MaCSCs were infected by these adenoviruses at a multiplicity of infection of 100 at suspension condition for 2 h.
Statistical Analysis
Statistical significance was evaluated by a paired t test, using p < 0.05 as indicative of statistical significance. Kaplan-Meier tumor-free survival data were compared using the log rank test. Tumor growth curves were compared using the two-way ANOVA. Frequency of tumorigenic cells (95% confidence interval) was analyzed by Extreme Limiting Dilution Analysis as described previously (43).
RESULTS
Generation of FAK Knock-in Mice with P878A/P881A Mutation
Previous studies in our laboratory have identified a scaffold function of FAK through its C-terminal Pro-rich motif (Pro-878 and Pro-881) to mediate endophilin A2 phosphorylation by Src, which enhances the surface level of MT1-MMP and promotes invasion of Src-transformed fibroblasts in vitro (22). To study the potential role of this FAK scaffolding function in vivo, we generated a mutant FAK allele with residues Pro-878 and Pro-881 mutated to Ala (designated as the PA allele) in the endogenous FAK gene utilizing a gene knock-in approach via homologous recombination. A targeting vector containing the P878A/P881A mutation in exon 28 of FAK genomic DNA and a neocassette (PA[neo] allele) was prepared and used to generate mutant mice containing the knock-in mutant allele (supplemental Fig. S1), as described under “Experimental Procedures.” All FAK+/PA mice (designated as +/PA mice) are viable, fertile, and indistinguishable from wild type mice, indicating that the PA mutant allele did not exhibit any dominant negative effects over the wild type FAK expressed from the wild type allele to cause any apparent phenotypes. Moreover, mating between heterozygous FAK+/PA mice yielded homozygous FAKPA/PA mice (designated as PA/PA mice) at the expected Mendelian ratio. Furthermore, these mice are fertile and indistinguishable from PA/+ or wild type mice, including their ability to nurse pups. Histological examination of female PA/PA mice showed apparently normal mammary gland development in all stages, including branching morphogenesis, lobular-alveolar development, and involution (data not shown). Together, these results suggested that, despite previous findings in transformed cells in vitro (22), FAK C-terminal Pro-rich motif (Pro-878/881) and its scaffolding functions are not required for embryonic development or in adult mice, including mammary gland development and function.
FAK P878A/P881A Mutation Suppresses Mammary Tumor Growth and Metastasis
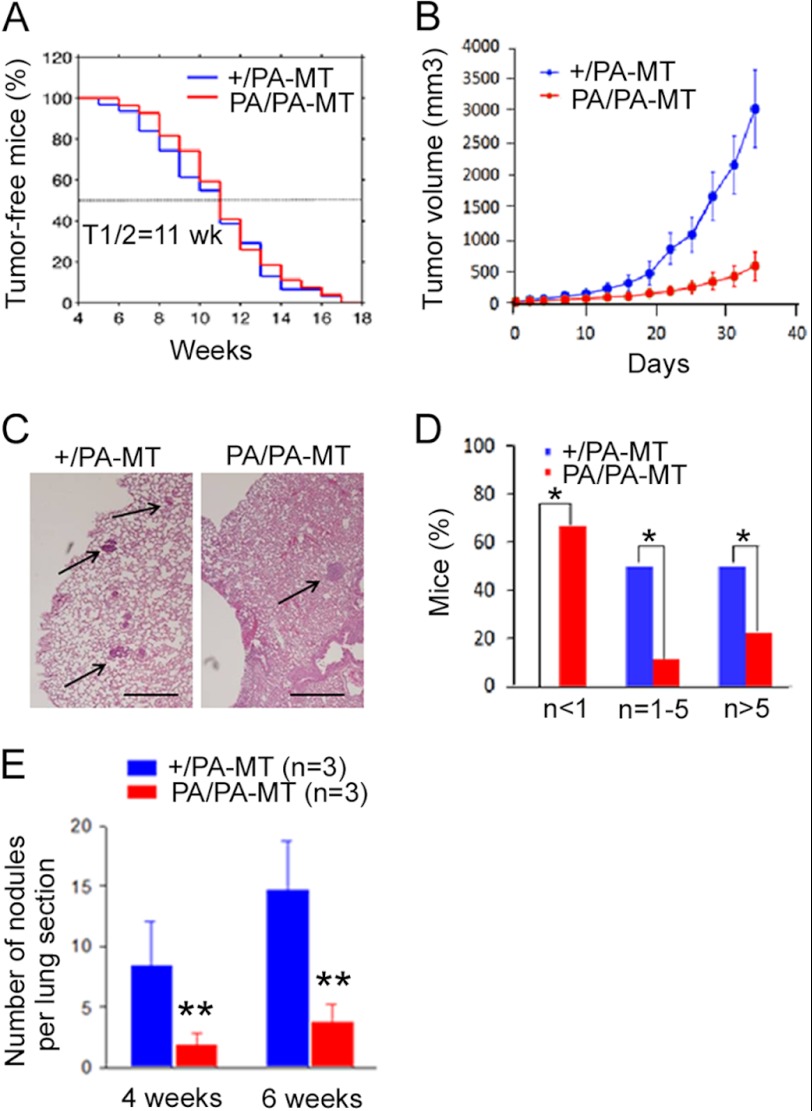
To investigate whether FAK P878A/P881A mutation could affect mammary tumorigenesis and metastasis, we crossed PA/PA mice with the MMTV-PyMT transgenic mouse model of human breast cancer (39) to generate FAKPA/PA;MMTV-PyMT (designated PA/PA-MT) and FAK+/PA;MMTV-PyMT (designated +/PA-MT as a control) mice. Mammary tumor development and metastasis were then evaluated in female mice as described previously (14). Fig. 1A shows that both PA/PA-MT and +/PA-MT mice developed mammary tumors rapidly, with a mean tumor onset age (t50) of 11 weeks, which is comparable with MMTV-PyMT mice (with wild type FAK background) as observed previously (14). Surprisingly, however, measurements at regular intervals of the average volumes of the mammary tumors showed that tumors in PA/PA-MT mice grew at a significantly slower rate from about 2 weeks after the initial appearance of palpable tumors compared with those in +/PA-MT mice (Fig. 1B). Moreover, histological analysis of lung sections at 8 weeks after tumor appearance showed a significant decrease in the number of metastatic nodules in PA/PA-MT mice compared with that in +/PA-MT mice, indicating reduced metastasis in these mice (Fig. 1, C and D). To exclude the possible influence of slower primary tumor growth on the observed reduction in metastasis, we examined experimental metastasis of the same amount of tumor cells isolated from PA/PA-MT or +/PA-MT mice by tail vein injection using recipient nude mice. As shown in Fig. 1E, a significantly reduced number of metastatic nodules was found in the lungs of recipients at both 4 and 6 weeks after injection of PA/PA-MT tumor cells compared with those injected with +/PA-MT tumor cells. Taken together, these results suggested that disruption of FAK scaffold function for endophilin A2 by P878A/P881A mutation in mammary epithelial cells suppressed breast cancer growth and metastasis in vivo.
FIGURE 1.
PA/PA-MT mice had comparable tumor appearance time but suppressed mammary tumor growth and lung metastasis. A, Kaplan-Meier analysis of mammary tumor development in the +/PA-MT (n = 30) and PA/PA-MT (n = 28) mice. B, mean cumulative mammary tumor volume per mouse at different times after primary tumor appearance for +/PA-MT (n = 30) and PA/PA-MT (n = 28). PA/PA-MT versus +/PA-MT, p < 0.05 by the two-way ANOVA. C and D, lung sections (four to six sections per mouse) were prepared 8 weeks after the primary mammary tumor onset and stained with H&E (C), and the micrometastatic nodules (arrows) were quantitated under a microscope for percentages of mice of the indicated genotype with <1, 1–5, or >5 metastases per lung section (D). *, p < 0.05, when the percentage of each group in PA/PA-MT mice is compared with that in +/PA-MT mice. Scale bars, 1 mm. E, 1 × 105 freshly isolated primary tumor cells from +/PA-MT or PA/PA-MT mice were injected into the tail vein of 8-week-old nude mice (n = 3 for each group). The micrometastatic nodules in their lungs were quantitated 4 or 6 weeks after injection. **, p < 0.01, when the number of nodules after injection of tumor cells from PA/PA-MT mice is compared with that from +/PA-MT mice.
FAK P878A/P881A Mutation Leads to Defective EMT of Mammary Tumor Cells from PA/PA-MT Mice
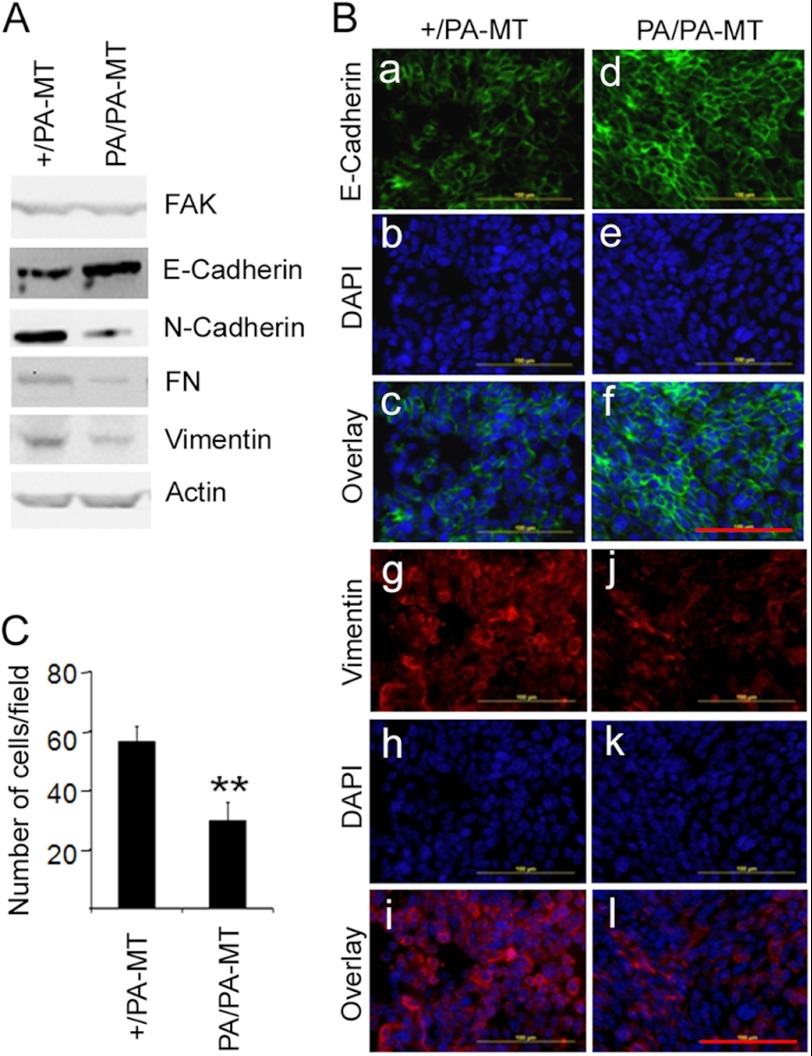
To evaluate whether the reduced tumor growth and metastasis in PA/PA-MT mice are due to intrinsic defects of tumor cells, mammary tumor cells were isolated from PA/PA-MT and +/PA-MT mice and subjected to a variety of analyses in vitro. In contrast to the observation in vivo, tumor cells from PA/PA-MT mice did not show any differences in proliferation (supplemental Fig. S2) or apoptosis (data not shown) when compared with those from +/PA-MT mice. We noted, however, that PA/PA-MT tumor cells tended to form aggregates more readily than the control +/PA-MT tumor cells during passage of these cells (data not shown). Western blotting analyses of tumor cell lysates showed an increased level of epithelial marker E-cadherin and lower levels of mesenchymal markers N-cadherin, fibronectin, and vimentin in PA/PA-MT tumor cells compared with +/PA-MT tumor cells (Fig. 2A). Consistent with these results, immunofluorescent staining of the cultured cells also indicated increased E-cadherin and reduced vimentin in PA/PA-MT tumor cells (Fig. 2B). Elevated E-cadherin expression was also detected in the sections of primary tumors in PA/PA-MT mice by immunohistochemical analysis (supplemental Fig. S3). Moreover, analyses of mRNAs from the tumor cells by quantitative RT-PCR showed decreased expression of Snail1 and Twist1, transcriptional factors critical for EMT (33, 44, 45), in PA/PA-MT tumor cells (supplemental Fig. S4). Finally, PA/PA-MT tumor cells also showed a significant decrease in migration toward fibronectin compared with +/PA-MT tumor cells (Fig. 2C). Taken together, these results suggested that disruption of the function of the FAK scaffold for endophilin A2 reduced markers for EMT in mammary tumor cells, which may contribute to the decreased mammary tumor growth and metastasis in PA/PA-MT mice.
FIGURE 2.
Tumor cell growth, markers for EMT, and migration were suppressed in PA/PA-MT mice. A, lysates of four tumors from two different +/PA-MT or PA/PA-MT mice were prepared and analyzed by immunoblotting using antibodies against various proteins, as indicated. B, cultured primary tumor cells from +/PA-MT or PA/PA-MT mice were analyzed by immunofluorescent labeling using antibody against E-cadherin (panels a–f) or vimentin (panels g–l). Scale bars, 100 μm. C, cell mobility was measured by Boyden chamber assay for the freshly isolated primary tumor cells from +/PA-MT or PA/PA-MT mice attracted by fibronectin. Mean ± S.E. of the number of migrated cells per field from three independent experiments are shown. **, p < 0.01, when the migrated cell number from PA/PA-MT mice are compared with that from +/PA-MT mice.
FAK P878A/P881A Mutation Decreases the Pool of MaCSCs in PA/PA-MT Mice and Compromises Their Tumorigenicity
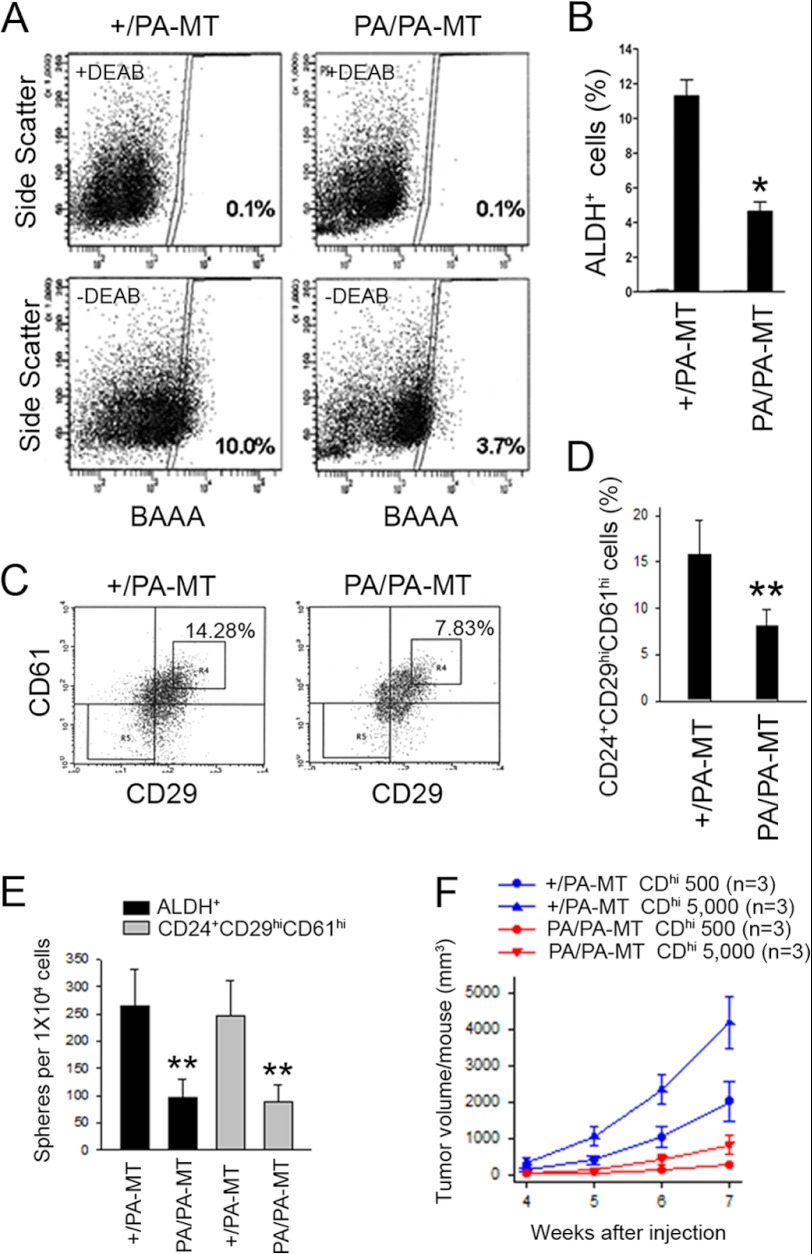
Our previous studies suggested that FAK promoted mammary tumorigenesis and metastasis through regulation of MaCSCs (14). Furthermore, tumor cells exhibiting increased markers for EMT have been shown to share gene expression signatures and other cellular characteristics with that of MaCSCs (32–35). We therefore examined the possibility that the FAK P878A/P881A mutation may also affect MaCSCs in PA/PA-MT mice. The content of MaCSCs in tumor cells from PA/PA-MT and +/PA-MT mice were analyzed using Aldefluor assay as described previously (14). We found that the Aldefluor-positive (ALDH+) subpopulation in mammary tumors from PA/PA-MT mice was significantly decreased compared with that from +/PA-MT mice (Fig. 3, A and B). We also examined the content of MaCSCs using the recently identified Lin−CD24+CD29hiCD61hi signature for MaCSCs in the MMTV-PyMT mouse model (46, 47). Similar to data using Aldefluor assays, a significant decrease of the Lin−CD24+CD29hiCD61hi subpopulation (gated on viable Lin− CD24+ tumor cell population) was observed in PA/PA-MT mice compared with that from +/PA-MT mice (Fig. 3, C and D).
FIGURE 3.
Decreased pool of MaCSCs and their tumorigenicity in PA/PA-MT mice. A and B, Aldefluor assay of freshly isolated tumor cells from +/PA-MT or PA/PA-MT mice. The percentage of ALDH+ cells was determined under the same gating criteria. Results are representative of two separate experiments. *, p < 0.05, when the percentage of ALDH+ cells from PA/PA-MT mice are compared with that from +/PA-MT mice. C and D, freshly isolated tumor cells from +/PA-MT or PA/PA-MT mice were depleted of CD45-positive and CD31-positive cells and labeled with CD24, CD29, and CD61 antibodies. The MaCSC population in each strain of mice was gated as Lin−CD24+CD29hiCD61hi (indicated as R4 quad) under the same gating criteria. Results are representative of two separate experiments. **, p < 0.01, when the percentage of Lin−CD24+CD29hiCD61hi cells from PA/PA-MT mice are compared with that from +/PA-MT mice. E, sorted ALDH+ or Lin−CD24+CD29hiCD61hi tumor cells from +/PA-MT or PA/PA-MT mice were analyzed for primary formation of tumor spheres. Results are generated from eight separated incubations for each sample and are representative of two independent experiments. **, p < 0.01, when the number of spheres by the ALDH+ or Lin−CD24+CD29hiCD61hi cells from PA/PA-MT mice are compared with that from +/PA-MT mice. F, mean cumulative mammary tumor volume per mouse at different times after different numbers of Lin−CD24+CD29hiCD61hi cells from +/PA-MT or PA/PA-MT mice injected into mammary fat pad of 8-week-old nude mice (n = 3 for each group).
To further investigate if interruption of FAK scaffold function for endophilin A2 may affect MaCSC activity and their tumorigenicity, we first examined the effect of FAK P878A/P881A mutation on their self-renewal using tumor sphere formation assay in vitro. As shown in Fig. 3E, the ALDH+ tumor cells isolated from PA/PA-MT mice exhibited a significantly lower capacity to generate tumor spheres compared with those isolated from +/PA-MT mice. Similar results were obtained when the Lin−CD24+CD29hiCD61hi subpopulations from these two mice were measured for tumor sphere formation. Next, ALDH+ tumor cells from PA/PA-MT and +/PA-MT mice were transplanted into mammary fat pads of recipient nude mice at limiting dilutions to assess for their self-renewal and tumorigenicity in vivo. At 3 weeks after transplantation, ALDH+ tumor cells from +/PA-MT mice ranging from 500 to 50,000 cells per injection generated tumors in the majority of recipient mice (Table 1). However, for ALDH+ tumor cells from PA/PA-MT mice, only 50,000 cells per injection, but not 5,000 or 500 cells per injection, generated tumors in the recipient mice. Similarly, Lin−CD24+CD29hiCD61hi tumor cells from PA/PA-MT mice showed significantly reduced tumorigenicity compared with those from +/PA-MT mice (Table 1 and Fig. 3F). Together, these results indicate that FAK P878A/P881A mutation results in impaired self-renewal and a reduced amount of MaCSCs, which could be responsible for the decreased mammary tumor growth and metastasis in PA/PA-MT mice.
TABLE 1.
Tumorigenicity of MaCSCs from PA/PA-MT mice
Mammary tumor cells from +/PA-MT and PA/PA-MT mice were sorted for ALDH+ or CD24+CD29hiCD61hi fractions to enrich for MaCSCs. They were then injected into the inguinal mammary fat pads of 8-week-old nude mice at serial dilutions. The number of sites with tumors grown after 4 weeks post-injection was counted for each condition.
| Cell numbers injected | Tumors/injections |
Frequency of tumorigenic cells (95% confidence interval) | ||
|---|---|---|---|---|
| 500 | 5000 | 50,000 | ||
| +/PA-MT ALDH+ | 5/6 | 6/6 | 6/6 | 1:279 (1:103–1:757) |
| PA/PA-MT ALDH+ | 0/6 | 0/6 | 2/6 | 1:140,015 (1:35,195–1:557,022) |
| +/PA-MT CD24+CD29hiCD61hi | 4/6 | 6/6 | 6/6 | 1:455 (1:163–1:1272) |
| PA/PA-MT CD24+CD29hiCD61hi | 0/6 | 1/6 | 3/6 | 1:61347 (1:22,094–1:170,343) |
Reduced Surface Expression of MT1-MMP Is Responsible for the Decreased Markers for EMT and MaCSCs Activity in PA/PA-MT Mice
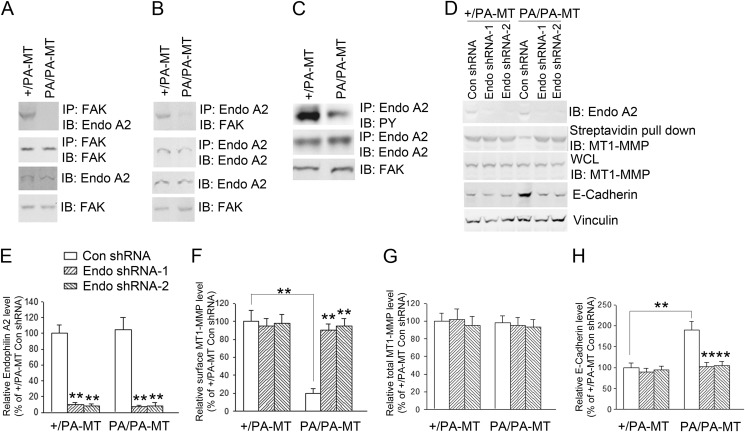
To investigate the mechanisms of reduced markers for EMT and defective MaCSCs activities upon FAK P878A/P881A mutation, we examined FAK interaction with endophilin A2 in mammary tumor cells from PA/PA-MT and +/PA-MT mice, as our previous studies identified the Pro-878/881 motif as a binding site for endophilin A2, which is important in the promotion of cancer cell invasion (22). As expected, wild type FAK in +/PA-MT tumor cells, but not P878A/P881A mutant in PA/PA-MT tumor cells, associated with endogenous endophilin A2 as detected by co-IP using either anti-FAK or anti-endophilin A2 antibodies (Fig. 4, A and B). Moreover, tyrosine phosphorylation of endophilin A2 was also reduced in PA/PA-MT tumor cells (Fig. 4C), which is consistent with data showing a role of this scaffolding interaction in mediating endophilin A2 phosphorylation by Src observed in transformed fibroblasts (22). Interestingly, we also observed that the surface expression of MT1-MMP was significantly decreased in PA/PA-MT tumor cells compared with those in +/PA-MT tumor cells (Fig. 4, D, lanes 1 and 4, and F), whereas the total expression level of MT1-MMP was not altered (Fig. 4G). We next treated PA/PA-MT tumor cells with endophilin A2 shRNA to test whether the reduced cell surface expression of MT1-MMP was dependent on endophilin A2 in these cells. We found that knockdown of endogenous endophilin A2 restored surface expression of MT1-MMP in PA/PA-MT tumor cells, but it did not affect that in +/PA-MT tumor cells, which already have a high level of MT1-MMP on the surface (Fig. 4, D, lanes 2, 3, 5, and 6, and E and F). Moreover, endophilin A2 knockdown also decreased E-cadherin expression in PA/PA-MT tumor cells but not that in +/PA-MT tumor cells (Fig. 4, D and H). These results suggest that FAK P878A/P881A mutation disrupted its interaction with endophilin A2, which may be responsible for the reduced surface expression of MT1-MMP and consequent reduction of markers for EMT in mammary tumor cells of PA/PA-MT mice.
FIGURE 4.
FAK interaction with endophilin A2 controls the surface expression of MT1-MMP in mammary tumor cells. A–C, primary tumor cell lysates from +/PA-MT or PA/PA-MT mice were immunoprecipitated (IP) with anti-FAK (A) or anti-endophilin (Endo) A2 (B and C) followed by Western blotting with antibodies as indicated. Aliquots of lysates were also analyzed by Western blotting directly as indicated. IB, immunoblot. D, freshly isolated primary tumor cells from +/PA-MT or PA/PA-MT mice were infected with recombinant lentiviruses encoding endophilin A2 shRNA or a control shRNA. Three days after infection, surface expression of MT1-MMP as labeled by biotinylation followed by streptavidin precipitation was determined. Aliquots of the lysates were also analyzed directly by Western blotting with antibodies as indicated. E–H, intensity of the endophilin A2 (E), surface MT1-MMP (F), total MT1-MMP (G), and E-cadherin (H) bands was quantified from two independent experiments by densitometry. The mean ± S.D. of relative intensity (normalized to cells from +/PA-MT mice treated with control (Con) shRNA) are shown. **, p < 0.01 compared with cells treated with control shRNA. **, p < 0.01 (linked by lines in F and H) when cells from +/PA-MT and PA/PA-MT mice were compared (both treated with control shRNA).
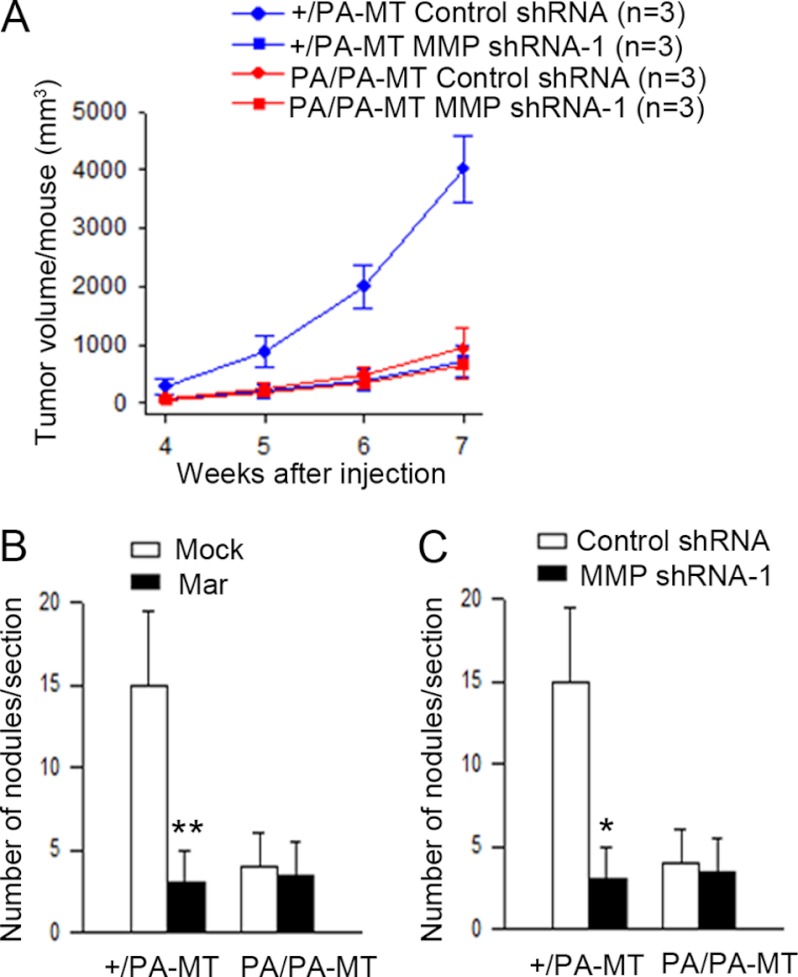
MT1-MMP has been well characterized to promote tumor cell invasion and metastasis in previous studies (22). To determine whether the decreased surface levels of MT1-MMP may mediate the reduced markers for EMT and MaCSC activity in the PA/PA-MT mouse model, we first employed Marimastat, a broad spectrum inhibitor for several MMPs, including MT1-MMP, to test for its effect on +/PA-MT and PA/PA-MT tumor cells. As shown in Fig. 5A, Marimastat treatment increased E-cadherin expression in +/PA-MT tumor cells in a dose-dependent manner, but it did not affect that in PA/PA-MT tumor cells that already have a high E-cadherin expression due to possibly reduced EMT. To exclude the potential effect of Marimastat targeting other MMPs, we also examined the effect of specific MT1-MMP knockdown by shRNA. Two different shRNAs targeting MT1-MMP, but not a control scrambled sequence, reduced the total expression of MT1-MMP in +/PA-MT and PA/PA-MT tumor cells as well as the surface expression of +/PA-MT tumor cells (the surface level was already low in PA/PA tumor cells) (Fig. 5, B–D). Consistent with data from Marimastat treatment, specific knockdown of MT1-MMP also increased E-cadherin expression in +/PA-MT tumor cells (Fig. 5, B and E). We then measured the effect of MT1-MMP inhibition or knockdown on MaCSC activity using tumor sphere formation assays. We found that both Marimastat and MT1-MMP shRNA significantly decreased tumor sphere formation by +/PA-MT, but not PA/PA-MT tumor cells (Fig. 5, F and G). To complement the above in vitro assays, we performed transplantation experiments to assess the role of reduced surface expression of MT1-MMP in the decreased self-renewal and tumorigenicity of PA/PA-MT MaCSCs in vivo. We found that knockdown of MT1-MMP by shRNA significantly reduced tumorigenicity of MaCSCs from +/PA-MT mice in transplantation assays (Table 2 and Fig. 6A). In contrast, the same treatment with MT1-MMP shRNA did not affect tumorigenicity of MaCSCs from PA/PA-MT mice, which already have low levels of MT1-MMP on the surface. MaCSCs from +/PA-MT and PA/PA-MT mice were also injected into the tail vein of recipient nude mice and monitored for metastasis to the lungs with or without treatments of the mice with Marimastat. As shown in Fig. 6B, Marimastat significantly decreased the number of metastatic nodules generated by +/PA-MT MaCSCs to a lower level as PA/PA-MT MaCSCs, which was not further affected by the inhibitor. Similarly, MT1-MMP knockdown reduced metastatic activity of +/PA-MT MaCSCs but not PA/PA-MT MaCSCs (Fig. 6C). Taken together, these results provide strong support that the reduced surface expression of MT1-MMP is responsible for the decreased markers for EMT and MaCSCs activity in PA/PA-MT mice.
FIGURE 5.
Regulation of EMT and tumor sphere formation by MT1-MMP in mammary tumor cells. A, freshly isolated primary tumor cells from +/PA-MT or PA/PA-MT mice were treated with different concentrations of Marimastat as indicated for 8 h. After treatment, total cell lysates were analyzed directly by Western blotting with antibody anti-E-cadherin or anti-actin as a loading control. B, freshly isolated primary tumor cells from +/PA-MT or PA/PA-MT mice were infected with recombinant lentiviruses encoding MT1-MMP shRNA or a control shRNA. Three days after infection, surface expression of MT1-MMP as labeled by biotinylation followed by streptavidin precipitation was determined. Aliquots of the lysates were also analyzed directly by Western blotting with antibodies as indicated. IB, immunoblot. C–E, intensity of the surface MT1-MMP (C), total MT1-MMP (D), and E-cadherin (E) bands was quantified from two independent experiments by densitometry. The mean ± S.D. of relative intensity (normalized to cells from +/PA-MT mice treated with control (Con) shRNA) are shown. **, p < 0.01 compared with cells treated with control shRNA. **, p < 0.01 (linked by lines in C and E) when cells from +/PA-MT and PA/PA-MT mice are compared (both treated with control shRNA). F, sorted ALDH+ tumor cells from +/PA-MT or PA/PA-MT mice were analyzed for primary formation of tumor spheres in the medium contained 100 nm Marimastat. Results are generated from six separated incubations for each sample and are representative of two independent experiments. **, p < 0.01, when the number of spheres by the ALDH+ cells from +/PA-MT mice treated with 100 nm Marimastat are compared with that without treatment. G, sorted ALDH+ tumor cells from +/PA-MT or PA/PA-MT mice were infected with recombinant lentiviruses encoding MT1-MMP shRNA or a control shRNA and then were analyzed for primary formation of tumor spheres. *, p < 0.05, when the number of spheres by the ALDH+ cells from +/PA-MT mice infected with MT1-MMP shRNA virus are compared with that infected with control shRNA virus.
TABLE 2.
Effect of MT1-MMP knockdown on the tumorigenicity of MaCSCs
ALDH+ cells were sorted from mammary tumor cells of +/PA-MT and PA/PA-MT mice and then infected with lentiviruses encoding control shRNA or MT1-MMP shRNA. The infected cells were then injected into the inguinal mammary fat pads of 8-week-old nude mice. The number of sites with tumors grown after 4 weeks post-injection was counted for each condition.
| Cell numbers injected | Tumors/injections |
Frequency of tumorigenic cells (95% confidence interval) | |
|---|---|---|---|
| 5000 | 50,000 | ||
| +/PA-MT control shRNA | 5/6 | 6/6 | 1:2791 (1:1028–1:7575) |
| +/PA-MT MT1-MMP shRNA-1 | 0/6 | 1/6 | 1:304,316 (1:43,069–1:2,150,205) |
| PA/PA-MT control shRNA | 0/6 | 2/6 | 1:138,499 (1:34,774–1:551,621) |
| PA/PA-MT MT1-MMP shRNA-1 | 0/6 | 1/6 | 1:304,316 (1:43,069–1:2,150,205) |
FIGURE 6.
Regulation of MaCSC and metastatic activities by MT1-MMP. A, mean cumulative mammary tumor volume per mouse at different times after 5000 ALDH+ cells from +/PA-MT or PA/PA-MT mice infected with recombinant lentiviruses encoding MT1-MMP shRNA or a control shRNA were injected into mammary fat pads of 8-week-old nude mice (n = 3 for each group). B, 5000 ALDH+ tumor cells from +/PA-MT or PA/PA-MT mice were injected into the tail vein of 8-week-old nude mice (n = 3 for each group). Marimastat were administered orally at a dose of 10 mg/kg daily to the recipient mice right after injection. The micrometastatic nodules in their lungs were quantitated 4 weeks after injection. **, p < 0.01, when the number of nodules after injection of tumor cells from +/PA-MT mice into Marimastat-treated recipient mice compared with that into control recipient mice. C, 5000 ALDH+ tumor cells from +/PA-MT or PA/PA-MT mice were infected with recombinant lentiviruses encoding MT1-MMP shRNA or a control shRNA. Then these cells were injected into the tail vein of 8-week-old nude mice (n = 3 for each group). The micrometastatic nodules in their lungs were quantitated 4 weeks after injection. *, p < 0.05, when the number of nodules after injection of tumor cells from +/PA-MT mice infected with MT1-MMP shRNA virus are compared with that infected with control shRNA virus.
FAK-mediated Phosphorylation of Endophilin A2 Promotes Markers for EMT and MaCSCs Activity of Mammary Tumor Cells
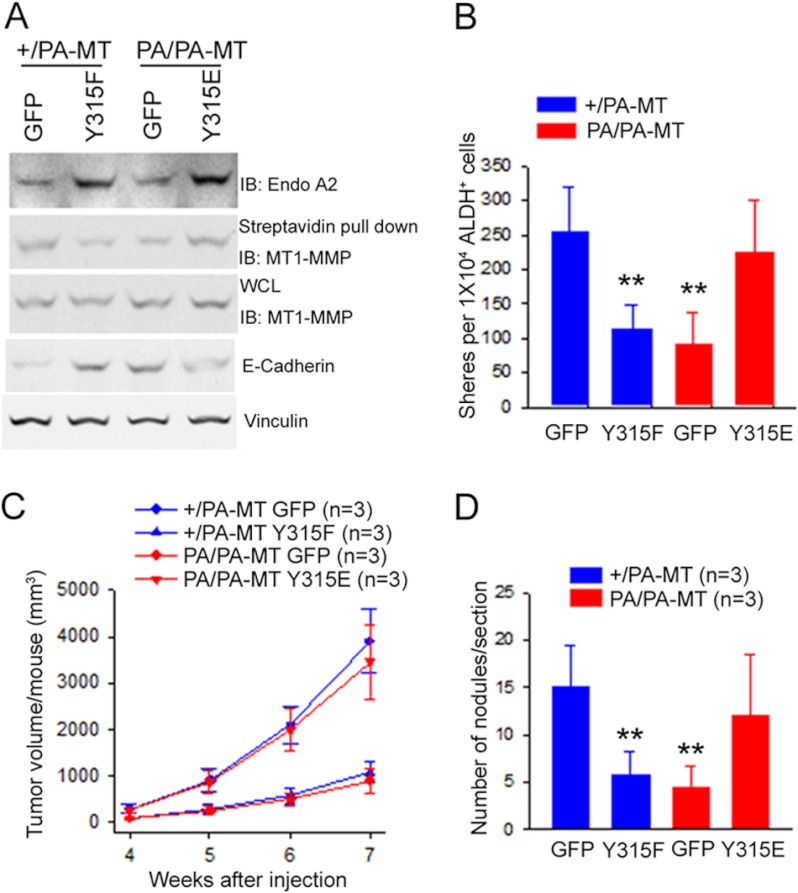
Our previous studies showed that FAK regulates the surface expression of MT1-MMT through mediating the phosphorylation of Tyr-315 of endophilin A2 by Src (22). Thus, the above results suggest a role of FAK-dependent phosphorylation of endophilin A2 at Tyr-315 by Src in promotion of markers for EMT and MaCSC activity. To assess such a possibility directly, we examined the effect of ectopic expression of two endophilin A2 mutants, Y315F (phosphorylation resistant) and Y315E (phosphorylation mimics as a potential gain-of-function mutant), in tumor cells from +/PA-MT and PA/PA-MT mice, respectively. As shown in Fig. 7A, expression of Y315F mutant reduced surface expression of MT1-MMP in +/PA-MT tumor cells. Conversely, expression of Y315E mutant rescued the decreased surface expression of MT1-MMP in PA/PA-MT tumor cells. Furthermore, the Y315F mutant also increased E-cadherin expression in +/PA-MT cells, whereas Y315E decreased it in PA/PA-MT cells (Fig. 7A and supplemental Fig. S5). These results suggest that FAK P878A/P881A mutation reduced surface expression of MT1-MMP and markers for EMT by disrupting its interaction with endophilin A2 (which facilities its phosphorylation at Tyr-315 by Src) (22) rather than potential defective interactions with other proteins.
FIGURE 7.
Regulation of EMT and MaCSC activities by endophilin A2 phosphorylation at Tyr-315 in mammary tumor cells. A, freshly isolated primary tumor cells from +/PA-MT or PA/PA-MT mice were infected with recombinant adenoviruses encoding endophilin A2 mutant Y315F or Y315E or GFP as a control. Three days after infection, surface expression of MT1-MMP was determined. Aliquots of the lysates were also analyzed directly by Western blotting with antibodies as indicated. B–D, freshly isolated primary tumor cells from +/PA-MT or PA/PA-MT mice were sorted for ALDH+ cells and then infected with recombinant adenoviruses encoding endophilin A2 mutants Y315F or Y315E or GFP as a control. B, infected ALDH+ cells were subjected to analysis for tumor sphere formation. **, p < 0.01, when compared with the cells from +/PA-MT mice infected with GFP virus. C, mean cumulative mammary tumor volume per mouse at different times after 5000 infected ALDH+ cells were injected into mammary fat pad of nude mice. +/PA-MT ALDH+ cells expressing Y315F versus +/PA-MT ALDH+ cells expressing GFP, p < 0.01 was done by the two-way ANOVA; PA/PA-MT ALDH+ cells expressing Y315E versus PA/PA-MT ALDH+ cells expressing GFP, p < 0.01 was done by the two-way ANOVA. D, number of nodules per lung section 4 weeks after injection of 5000 ALDH+ tumor cells from +/PA-MT or PA/PA-MT mice infected with different adenoviruses as indicated into tail vein of 8-week-old nude mice, **, p < 0.01, when compared with +/PA-MT ALDH+ cells expressing GFP.
We next evaluated the effects of these mutants on MaCSCs and metastatic activities of tumor cells. We found that expression of the Y315F mutant decreased tumor sphere formation of MaCSCs from +/PA-MT mice to a level comparable with that of MaCSCs from PA/PA-MT mice (Fig. 7B). Conversely, expression of the Y315E mutant rescued the defective self-renewal of MaCSCs from PA/PA-MT mice. Consistent with these data using tumor sphere formation assay in vitro, limiting dilution transplantation experiments showed that the Y315F mutant significantly reduced tumorigenicity of +/PA-MT MaCSCs, whereas the Y315E mutant rescued the defective tumorigenicity of PA/PA-MT MaCSCs in vivo (Table 3 and Fig. 7C). Finally, analysis of these cells by tail vein injection experiments showed a significant inhibition of metastatic activity of +/PA-MT MaCSCs by the Y315F mutant and restoration of the defective metastatic activity of PA/PA-MT MaCSCs by the Y315E mutant (Fig. 7D). Taken together, these results demonstrate that the function of FAK scaffolding to bind endophilin A2 at Pro-878/881, which facilitates its phosphorylation at Tyr-315 by Src, is essential for maintaining MaCSC and tumor metastatic activities by promotion of MT1-MMP surface expression and markers for EMT.
TABLE 3.
Role of endophilin A2 phosphorylation at Y315 in the tumorigenicity of MaCSCs
ALDH+ cells were sorted from mammary tumor cells of +/PA-MT and PA/PA-MT mice and then infected with lentiviruses encoding endophilin A2 mutants Y315F or Y315E or GFP as a control. The infected cells were then injected into the inguinal mammary fat pads of 8-week-old nude mice. The number of sites with tumors grown after 4 weeks post-injection was counted for each condition.
| Cell numbers injected | Tumors/injections |
Frequency of tumorigenic cells (95% confidence interval) | |
|---|---|---|---|
| 5000 | 50,000 | ||
| +/PA-MT GFP | 6/6 | 6/6 | 1 (1–1:5353) |
| +/PA-MT Y315F | 0/6 | 2/6 | 1:138,499 (1:34,774–1:551,621) |
| PA/PA-MT GFP | 0/6 | 1/6 | 1:304,316 (1:43,069–1:2,150,205) |
| PA/PA-MT Y315E | 4/6 | 5/6 | 1:15,032 (1:5748–1:39,313) |
DISCUSSION
Since its initial discovery 2 decades ago, numerous studies have implicated FAK in the development and progression of breast and other cancers (9, 10, 48). Accordingly, several kinase inhibitors for FAK are being developed for targeted cancer therapies (49). Besides functioning as a tyrosine kinase, FAK has been shown to interact with many other proteins, and it acts as a scaffold to regulate diverse cellular activities, including cell survival, proliferation, and migration (4–8). However, the potential role and mechanisms of FAK scaffolding functions have not been examined in any biological or disease process directly in vivo. By generating and analyzing a FAK P878A/P881A mutant knock-in mouse model, here we present data suggesting that disruption of FAK interaction through its Pro-878/881 motif with endophilin A2 attenuated tumor growth and metastasis in a murine model of breast cancer in vivo. Our results provide the first direct evidence for the important role of a scaffolding activity of FAK in promoting breast cancer progression in vivo, which are consistent with and further highlight the importance of kinase-independent functions of FAK in various cells as suggested by several recent studies (50, 51).
Although FAK has been shown to regulate a variety of cellular functions in different cells that could contribute to the observed phenotype in PA/PA-MT mice, our present data suggested that disruption of FAK scaffolding function through the P878A/P881A mutation suppressed mammary tumor growth and metastasis by compromising tumor cell markers for EMT and their MaCSC activities. These results are consistent with a number of recent studies implicating a role for FAK in the regulation of EMT (23, 26–28) as well as MaCSCs (14). Moreover, our data suggested that FAK scaffolding function could contribute to the regulation of markers for EMT and MaCSC activity in breast cancers. Using FAK-null MaCSCs, we recently found that FAK autophosphorylation and subsequent activation of the PI3K/Akt pathway is important for self-renewal and tumorigenicity of MaCSCs, although its potential role has not been assessed directly in vivo (40). It is therefore likely that both kinase-dependent and -independent functions of FAK are required for full activity of MaCSCs, and their relative contributions will be an interesting topic of future investigations. Our findings of decreased markers for EMT and defective MaCSCs self-renewal in PA/PA-MT tumor cells are also consistent with the notion that EMT is a feature of MaCSCs as evidenced by their many shared characteristics, including the expression of signature genes (32–35). However, several recent studies showed that EMT characteristics are not required for the tumor-initiating activity of MaCSCs (52) and that EMT could suppress some attributes of MaCSCs (53). Although it is attractive to hypothesize that FAK signaling regulates MaCSCs through its effects on markers for EMT or vice versa, it is also possible that markers for EMT and MaCSCs are affected by the P878A/P881A mutation through parallel but independent mechanisms. Future studies using PA/PA-MT mice as well as analysis of other FAK mutations will be necessary to determine these possibilities.
Our previous studies in transformed fibroblasts indicate that FAK scaffolding function through Pro-878/881 mediates endophilin A2 phosphorylation by Src to promote surface expression of MT1-MMP via inhibition of its endocytosis (22). Several lines of evidence in this study strongly support that such a molecular mechanism is responsible for the FAK regulation of markers for EMT and MaCSCs of mammary tumor cells in vivo. P878A/P881A mutation in FAK disrupted its interaction with endophilin A2 and reduced endophilin A2 phosphorylation and surface expression of MT1-MMP in mammary tumor cells. In +/PA-MT tumor cells, blocking endophilin A2 phosphorylation by ectopic expression of Y315F mutant also reduced MT1-MMP surface expression. Conversely, either knockdown of endophilin A2 or expression of Y315E mutant mimicking its phosphorylation rescued the reduced MT1-MMP surface expression in PA/PA-MT tumor cells. In all of these cases, increased tumor cell E-cadherin expression and reduced MaCSC self-renewal were observed under the conditions of the reduced MT1-MMP surface expression, and vice versa. Finally, both a broad spectral MMP inhibitor and, more importantly, a specific knockdown of MT1-MMP significantly reduced MaCSCs activity in +/PA-MT tumor cells but not in PA/PA-MT tumor cells where surface MT1-MMP was already low. Therefore, although we cannot completely exclude the potential role of other cellular proteins such as Graf, ASAP1, and amphiphysin that also interact FAK through Pro-878/881 (54–56) in contributing to the defective phenotypes of PA/PA-MT mice, our results presented here strongly suggest a critical role for this FAK scaffolding function to promote mammary tumor cell markers for EMT and MaCSCs activity via controlling MT1-MMP endocytosis and surface expression. In this regard, it is also interesting to note that the first Pro-rich motif in the FAK C-terminal domain (Pro-712/715) is not a consensus site to mediate FAK binding to endophilin A2 (22). Although the potential role of Pro-712/715 has not been assessed directly by a knock-in approach in vivo, our previous studies using FAK-null MaCSCs showed that it was not required for self-renewal and tumorigenicity of MaCSCs (40).
Whereas our results strongly suggest an intrinsic role of FAK scaffolding function through Pro-878/881 in the regulation of tumor cell markers for EMT and MaCSCs, it should be noted that PA/PA-MT mice contain the FAK P878A/P881A mutation in all cells and tissues. Given the critical importance of a tumor microenvironment for tumor growth and progression (57, 58), it is possible that potential defects in tumor stromal cells (e.g. endothelial cells and tumor-associated fibroblasts) may also contribute to the phenotypes observed in PA/PA-MT mice. Nevertheless, we observed similar phenotypes in mammary epithelial cell-specific FAK P878A/P881A mutation conditional knock-in mice (i.e. FAKf/PA;MMTV-Cre;MMTV-PyMT mice with P878A/P881A mutation only in mammary epithelial cells) as that in PA/PA-MT mice (data not shown). Moreover, no differences in angiogenesis were found in PA/PA mice compared with +/PA control mice using either wound- or tumor-induced angiogenesis models (data not shown). Therefore, P878A/P881A mutation likely acted to suppress markers for EMT and MaCSC activities in a tumor cell-autonomous manner, although future studies will be needed to clarify potential contributions from other cells harboring the same FAK mutation.
In contrast to mammary tumor cells, our data indicated that disruption of the function of FAK scaffolding through the P878A/P881A mutation did not cause any apparent developmental or functional defects of any organ systems in PA/PA mice, including mammary glands. Although very surprising, this finding is reminiscent of our previous results where disruption of this scaffolding function only impacted the transformed fibroblasts without any effect on their normal counterpart (22). In fibroblasts, only transformed cells contain significant amounts of surface MT1-MMP and exhibit invasive activities, which is regulated by the FAK scaffolding function to mediate endophilin A2 phosphorylation (22). Therefore, it is conceivable that the MT1-MMP regulation of markers for EMT and MaCSCs activities is also specific for mammary tumor cells. Interestingly, FAK has been shown to regulate EMT during normal embryonic development (59). Our results would suggest that this will be through FAK signaling that is distinct from its scaffolding functions through Pro-878/881. Recent studies in our laboratory suggested that FAK deletion in mammary epithelial cells depletes the pool of normal mammary stem cells (data not shown), contributing to the suppressed mammary tumorigenesis and progression in MFCKO-MT (i.e. FAKf/f;MMTV-Cre;MMTV-PyMT) mice observed previously (14). Given the apparently normal development and function of mammary glands in PA/PA mice, this study indicates that this FAK scaffolding function is not important for mammary stem cells, which could explain the apparently normal mammary tumorigenesis in PA/PA-MT mice as the mammary stem cells pools (putative targets for the generation of MaCSCs) were not depleted as in MFCKO mice.
In summary, our studies identify a novel role for the FAK scaffolding function through Pro-878/881 binding to endophilin A2 in regulating tumor cell markers for EMT and MaCSCs to promote mammary tumor growth and metastasis in vivo. These studies not only provide further insights into the roles and mechanisms of FAK signaling in breast cancer development and progression but also suggest that disruption of such a FAK scaffolding function could serve as a new strategy for human anticancer therapy. The unique requirement of this FAK signaling pathway for MaCSCs but not normal MaSCs would be especially useful for the development of breast cancer therapy targeting MaCSCs. Such a strategy to disrupt FAK scaffolding functions could lead to more successful cancer treatments either alone or when combined with kinase inhibitors for FAK.
Acknowledgments
We are grateful to Dr. Thom Saunders of the University of Michigan for advice on the generation of knock-in mice and C. Bian and A. Serna for technical assistance. We thank Dr. Stephen Weiss of the University of Michigan and members of the Guan laboratory for discussions and helpful comments. We acknowledge E. Hughes, Y. Y. Qu, K. Childs, and G. Gavrilina for the preparation of gene-targeted mice and the University of Michigan Biomedical Research Core Facility, which was supported in part by National Institutes of Health Grant CA046592.
This work was supported, in whole or in part, by National Institutes of Health Grants CA163493 and CA150926. This work was also supported in part by the John S. and Suzanne C. Munn cancer fund of the University of Michigan Comprehensive Cancer Center (to J.-L. G.).

This article contains supplemental Figs. S1–S5.
- FAK
- focal adhesion kinase
- EMT
- epithelial-mesenchymal transition
- MaCSC
- mammary cancer stem cell
- MMTV
- murine mammary tumor virus
- ANOVA
- analysis of variance.
REFERENCES
- 1. Sjöblom T., Jones S., Wood L. D., Parsons D. W., Lin J., Barber T. D., Mandelker D., Leary R. J., Ptak J., Silliman N., Szabo S., Buckhaults P., Farrell C., Meeh P., Markowitz S. D., Willis J., Dawson D., Willson J. K., Gazdar A. F., Hartigan J., Wu L., Liu C., Parmigiani G., Park B. H., Bachman K. E., Papadopoulos N., Vogelstein B., Kinzler K. W., Velculescu V. E. (2006) The consensus coding sequences of human breast and colorectal cancers. Science 314, 268–274 [DOI] [PubMed] [Google Scholar]
- 2. Vogelstein B., Kinzler K. W. (2004) Cancer genes and the pathways they control. Nat. Med. 10, 789–799 [DOI] [PubMed] [Google Scholar]
- 3. Luo J., Solimini N. L., Elledge S. J. (2009) Principles of cancer therapy: oncogene and non-oncogene addiction. Cell 136, 823–837 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 4. Parsons J. T. (2003) Focal adhesion kinase. The first ten years. J. Cell Sci. 116, 1409–1416 [DOI] [PubMed] [Google Scholar]
- 5. Schlaepfer D. D., Mitra S. K. (2004) Multiple connections link FAK to cell motility and invasion. Curr. Opin. Genet. Dev. 14, 92–101 [DOI] [PubMed] [Google Scholar]
- 6. Siesser P. M., Hanks S. K. (2006) The signaling and biological implications of FAK overexpression in cancer. Clin. Cancer Res. 12, 3233–3237 [DOI] [PubMed] [Google Scholar]
- 7. Schaller M. D. (2001) Biochemical signals and biological responses elicited by the focal adhesion kinase. Biochim. Biophys. Acta 1540, 1–21 [DOI] [PubMed] [Google Scholar]
- 8. Zhao J., Guan J. L. (2009) Signal transduction by focal adhesion kinase in cancer. Cancer Metastasis Rev. 28, 35–49 [DOI] [PubMed] [Google Scholar]
- 9. Golubovskaya V. M., Cance W. G. (2007) Focal adhesion kinase and p53 signaling in cancer cells. Int. Rev. Cytol. 263, 103–153 [DOI] [PubMed] [Google Scholar]
- 10. McLean G. W., Carragher N. O., Avizienyte E., Evans J., Brunton V. G., Frame M. C. (2005) The role of focal-adhesion kinase in cancer–a new therapeutic opportunity. Nat. Rev. Cancer 5, 505–515 [DOI] [PubMed] [Google Scholar]
- 11. McLean G. W., Komiyama N. H., Serrels B., Asano H., Reynolds L., Conti F., Hodivala-Dilke K., Metzger D., Chambon P., Grant S. G., Frame M. C. (2004) Specific deletion of focal adhesion kinase suppresses tumor formation and blocks malignant progression. Genes Dev. 18, 2998–3003 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 12. Lahlou H., Sanguin-Gendreau V., Zuo D., Cardiff R. D., McLean G. W., Frame M. C., Muller W. J. (2007) Mammary epithelial-specific disruption of the focal adhesion kinase blocks mammary tumor progression. Proc. Natl. Acad. Sci. U.S.A. 104, 20302–20307 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 13. Pylayeva Y., Gillen K. M., Gerald W., Beggs H. E., Reichardt L. F., Giancotti F. G. (2009) Ras- and PI3K-dependent breast tumorigenesis in mice and humans requires focal adhesion kinase signaling. J. Clin. Invest. 119, 252–266 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 14. Luo M., Fan H., Nagy T., Wei H., Wang C., Liu S., Wicha M. S., Guan J. L. (2009) Mammary epithelial-specific ablation of the focal adhesion kinase suppresses mammary tumorigenesis by affecting mammary cancer stem/progenitor cells. Cancer Res. 69, 466–474 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 15. Provenzano P. P., Inman D. R., Eliceiri K. W., Beggs H. E., Keely P. J. (2008) Mammary epithelial-specific disruption of focal adhesion kinase retards tumor formation and metastasis in a transgenic mouse model of human breast cancer. Am. J. Pathol. 173, 1551–1565 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 16. Han D. C., Shen T. L., Guan J. L. (2000) Role of Grb7 targeting to focal contacts and its phosphorylation by focal adhesion kinase in regulation of cell migration. J. Biol. Chem. 275, 28911–28917 [DOI] [PubMed] [Google Scholar]
- 17. Wu X., Suetsugu S., Cooper L. A., Takenawa T., Guan J. L. (2004) Focal adhesion kinase regulation of N-WASP subcellular localization and function. J. Biol. Chem. 279, 9565–9576 [DOI] [PubMed] [Google Scholar]
- 18. Vuori K., Hirai H., Aizawa S., Ruoslahti E. (1996) Introduction of p130cas signaling complex formation upon integrin-mediated cell adhesion. A role for Src family kinases. Mol. Cell. Biol. 16, 2606–2613 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 19. Ruest P. J., Shin N. Y., Polte T. R., Zhang X., Hanks S. K. (2001) Mechanisms of CAS substrate domain tyrosine phosphorylation by FAK and Src. Mol. Cell. Biol. 21, 7641–7652 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 20. Schaller M. D., Parsons J. T. (1995) pp125FAK-dependent tyrosine phosphorylation of paxillin creates a high-affinity binding site for Crk. Mol. Cell. Biol. 15, 2635–2645 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 21. Petit V., Boyer B., Lentz D., Turner C. E., Thiery J. P., Vallés A. M. (2000) Phosphorylation of tyrosine residues 31 and 118 on paxillin regulates cell migration through an association with CRK in NBT-II cells. J. Cell Biol. 148, 957–970 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 22. Wu X., Gan B., Yoo Y., Guan J. L. (2005) FAK-mediated src phosphorylation of endophilin A2 inhibits endocytosis of MT1-MMP and promotes ECM degradation. Dev. Cell 9, 185–196 [DOI] [PubMed] [Google Scholar]
- 23. Avizienyte E., Frame M. C. (2005) Src and FAK signalling controls adhesion fate and the epithelial-to-mesenchymal transition. Curr. Opin. Cell Biol. 17, 542–547 [DOI] [PubMed] [Google Scholar]
- 24. Thiery J. P., Sleeman J. P. (2006) Complex networks orchestrate epithelial-mesenchymal transitions. Nat. Rev. Mol. Cell Biol. 7, 131–142 [DOI] [PubMed] [Google Scholar]
- 25. Avizienyte E., Wyke A. W., Jones R. J., McLean G. W., Westhoff M. A., Brunton V. G., Frame M. C. (2002) Src-induced de-regulation of E-cadherin in colon cancer cells requires integrin signalling. Nat. Cell Biol. 4, 632–638 [DOI] [PubMed] [Google Scholar]
- 26. Bailey K. M., Liu J. (2008) Caveolin-1 up-regulation during epithelial to mesenchymal transition is mediated by focal adhesion kinase. J. Biol. Chem. 283, 13714–13724 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 27. Cicchini C., Laudadio I., Citarella F., Corazzari M., Steindler C., Conigliaro A., Fantoni A., Amicone L., Tripodi M. (2008) TGFβ-induced EMT requires focal adhesion kinase (FAK) signaling. Exp. Cell Res. 314, 143–152 [DOI] [PubMed] [Google Scholar]
- 28. Nakamura K., Yano H., Schaefer E., Sabe H. (2001) Different modes and qualities of tyrosine phosphorylation of Fak and Pyk2 during epithelial-mesenchymal transdifferentiation and cell migration. Analysis of specific phosphorylation events using site-directed antibodies. Oncogene 20, 2626–2635 [DOI] [PubMed] [Google Scholar]
- 29. Prunier C., Howe P. H. (2005) Disabled-2 (Dab2) is required for transforming growth factor β-induced epithelial to mesenchymal transition (EMT). J. Biol. Chem. 280, 17540–17548 [DOI] [PubMed] [Google Scholar]
- 30. Strizzi L., Bianco C., Normanno N., Seno M., Wechselberger C., Wallace-Jones B., Khan N. I., Hirota M., Sun Y., Sanicola M., Salomon D. S. (2004) Epithelial mesenchymal transition is a characteristic of hyperplasias and tumors in mammary gland from MMTV-Cripto-1 transgenic mice. J. Cell. Physiol. 201, 266–276 [DOI] [PubMed] [Google Scholar]
- 31. Rodrigo J. P., Dominguez F., Suárez V., Canel M., Secades P., Chiara M. D. (2007) Focal adhesion kinase and E-cadherin as markers for nodal metastasis in laryngeal cancer. Arch. Otolaryngol. Head Neck Surg. 133, 145–150 [DOI] [PubMed] [Google Scholar]
- 32. Mani S. A., Guo W., Liao M. J., Eaton E. N., Ayyanan A., Zhou A. Y., Brooks M., Reinhard F., Zhang C. C., Shipitsin M., Campbell L. L., Polyak K., Brisken C., Yang J., Weinberg R. A. (2008) The epithelial-mesenchymal transition generates cells with properties of stem cells. Cell 133, 704–715 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 33. Polyak K., Weinberg R. A. (2009) Transitions between epithelial and mesenchymal states. Acquisition of malignant and stem cell traits. Nat. Rev. Cancer 9, 265–273 [DOI] [PubMed] [Google Scholar]
- 34. Taube J. H., Herschkowitz J. I., Komurov K., Zhou A. Y., Gupta S., Yang J., Hartwell K., Onder T. T., Gupta P. B., Evans K. W., Hollier B. G., Ram P. T., Lander E. S., Rosen J. M., Weinberg R. A., Mani S. A. (2010) Core epithelial-to-mesenchymal transition interactome gene-expression signature is associated with claudin-low and metaplastic breast cancer subtypes. Proc. Natl. Acad. Sci. U.S.A. 107, 15449–15454 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 35. Iliopoulos D., Hirsch H. A., Wang G., Struhl K. (2011) Inducible formation of breast cancer stem cells and their dynamic equilibrium with non-stem cancer cells via IL6 secretion. Proc. Natl. Acad. Sci. U.S.A. 108, 1397–1402 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 36. Skarnes W. C. (2000) Gene trapping methods for the identification and functional analysis of cell surface proteins in mice. Methods Enzymol. 328, 592–615 [DOI] [PubMed] [Google Scholar]
- 37. Kendall S. K., Samuelson L. C., Saunders T. L., Wood R. I., Camper S. A. (1995) Targeted disruption of the pituitary glycoprotein hormone α-subunit produces hypogonadal and hypothyroid mice. Genes Dev. 9, 2007–2019 [DOI] [PubMed] [Google Scholar]
- 38. Lakso M., Pichel J. G., Gorman J. R., Sauer B., Okamoto Y., Lee E., Alt F. W., Westphal H. (1996) Efficient in vivo manipulation of mouse genomic sequences at the zygote stage. Proc. Natl. Acad. Sci. U.S.A. 93, 5860–5865 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 39. Guy C. T., Cardiff R. D., Muller W. J. (1992) Induction of mammary tumors by expression of polyomavirus middle T oncogene: a transgenic mouse model for metastatic disease. Mol. Cell. Biol. 12, 954–961 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 40. Fan H., Guan J. L. (2011) Compensatory function of Pyk2 protein in the promotion of focal adhesion kinase (FAK)-null mammary cancer stem cell tumorigenicity and metastatic activity. J. Biol. Chem. 286, 18573–18582 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 41. Nagy T., Wei H., Shen T. L., Peng X., Liang C. C., Gan B., Guan J. L. (2007) Mammary epithelial-specific deletion of the focal adhesion kinase gene leads to severe lobulo-alveolar hypoplasia and secretory immaturity of the murine mammary gland. J. Biol. Chem. 282, 31766–31776 [DOI] [PubMed] [Google Scholar]
- 42. Wei H., Wei S., Gan B., Peng X., Zou W., Guan J. L. (2011) Suppression of autophagy by FIP200 deletion inhibits mammary tumorigenesis. Genes Dev. 25, 1510–1527 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 43. Hu Y., Smyth G. K. (2009) ELDA. Extreme limiting dilution analysis for comparing depleted and enriched populations in stem cell and other assays. J. Immunol. Methods 347, 70–78 [DOI] [PubMed] [Google Scholar]
- 44. Thiery J. P., Acloque H., Huang R. Y., Nieto M. A. (2009) Epithelial-mesenchymal transitions in development and disease. Cell 139, 871–890 [DOI] [PubMed] [Google Scholar]
- 45. Peinado H., Olmeda D., Cano A. (2007) Snail, Zeb, and bHLH factors in tumour progression. An alliance against the epithelial phenotype? Nat. Rev. Cancer 7, 415–428 [DOI] [PubMed] [Google Scholar]
- 46. Asselin-Labat M. L., Sutherland K. D., Barker H., Thomas R., Shackleton M., Forrest N. C., Hartley L., Robb L., Grosveld F. G., van der Wees J., Lindeman G. J., Visvader J. E. (2007) Gata-3 is an essential regulator of mammary-gland morphogenesis and luminal cell differentiation. Nat. Cell Biol. 9, 201–209 [DOI] [PubMed] [Google Scholar]
- 47. Kouros-Mehr H., Bechis S. K., Slorach E. M., Littlepage L. E., Egeblad M., Ewald A. J., Pai S. Y., Ho I. C., Werb Z. (2008) GATA-3 links tumor differentiation and dissemination in a luminal breast cancer model. Cancer Cell 13, 141–152 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 48. Luo M., Guan J. L. (2010) Focal adhesion kinase: a prominent determinant in breast cancer initiation, progression, and metastasis. Cancer Lett. 289, 127–139 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 49. Parsons J. T., Slack-Davis J., Tilghman R., Roberts W. G. (2008) Focal adhesion kinase. Targeting adhesion signaling pathways for therapeutic intervention. Clin. Cancer Res. 14, 627–632 [DOI] [PubMed] [Google Scholar]
- 50. Zhao X., Peng X., Sun S., Park A. Y., Guan J. L. (2010) Role of kinase-independent and -dependent functions of FAK in endothelial cell survival and barrier function during embryonic development. J. Cell Biol. 189, 955–965 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 51. Lim S. T., Chen X. L., Lim Y., Hanson D. A., Vo T. T., Howerton K., Larocque N., Fisher S. J., Schlaepfer D. D., Ilic D. (2008) Nuclear FAK promotes cell proliferation and survival through FERM-enhanced p53 degradation. Mol. Cell 29, 9–22 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 52. Kim J., Villadsen R., Sørlie T., Fogh L., Grønlund S. Z., Fridriksdottir A. J., Kuhn I., Rank F., Wielenga V. T., Solvang H., Edwards P. A., Børresen-Dale A. L., Rønnov-Jessen L., Bissell M. J., Petersen O. W. (2012) Tumor initiating but differentiated luminal-like breast cancer cells are highly invasive in the absence of basal-like activity. Proc. Natl. Acad. Sci. U.S.A. 109, 6124–6129 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 53. Celià-Terrassa T., Meca-Cortés O., Mateo F., de Paz A. M., Rubio N., Arnal-Estapé A., Ell B. J., Bermudo R., Díaz A., Guerra-Rebollo M., Lozano J. J., Estarás C., Ulloa C., Álvarez-Simón D., Milà J., Vilella R., Paciucci R., Martínez-Balbás M., de Herreros A. G., Gomis R. R., Kang Y., Blanco J., Fernández P. L., Thomson T. M. (2012) Epithelial-mesenchymal transition can suppress major attributes of human epithelial tumor-initiating cells. J. Clin. Invest. 122, 1849–1868 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 54. Hildebrand J. D., Taylor J. M., Parsons J. T. (1996) An SH3 domain-containing GTPase-activating protein for Rho and Cdc42 associates with focal adhesion kinase. Mol. Cell. Biol. 16, 3169–3178 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 55. Liu Y., Loijens J. C., Martin K. H., Karginov A. V., Parsons J. T. (2002) The association of ASAP1, an ADP ribosylation factor-GTPase activating protein, with focal adhesion kinase contributes to the process of focal adhesion assembly. Mol. Biol. Cell 13, 2147–2156 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 56. Messina S., Onofri F., Bongiorno-Borbone L., Giovedì S., Valtorta F., Girault J. A., Benfenati F. (2003) Specific interactions of neuronal focal adhesion kinase isoforms with Src kinases and amphiphysin. J. Neurochem. 84, 253–265 [DOI] [PubMed] [Google Scholar]
- 57. Littlepage L. E., Egeblad M., Werb Z. (2005) Coevolution of cancer and stromal cellular responses. Cancer Cell 7, 499–500 [DOI] [PubMed] [Google Scholar]
- 58. Mueller M. M., Fusenig N. E. (2004) Friends or foes–bipolar effects of the tumour stroma in cancer. Nat. Rev. Cancer 4, 839–849 [DOI] [PubMed] [Google Scholar]
- 59. Li X. Y., Zhou X., Rowe R. G., Hu Y., Schlaepfer D. D., Ilić D., Dressler G., Park A., Guan J. L., Weiss S. J. (2011) Snail1 controls epithelial-mesenchymal lineage commitment in focal adhesion kinase-null embryonic cells. J. Cell Biol. 195, 729–738 [DOI] [PMC free article] [PubMed] [Google Scholar]