Summary
Background and objectives
This study aimed to determine if recovery of kidney function after AKI modifies the association between AKI during hospitalization and adverse outcomes after discharge.
Design, setting, participants, & measurements
The effect of renal recovery after AKI was evaluated in a population-based cohort study (n=190,714) with participants identified from a provincial claims registry in Alberta, Canada, between November 1, 2002 and December 31, 2007. AKI was identified by a two-fold increase between prehospital and peak in-hospital serum creatinine (SCr). Recovery was assessed using SCr drawn closest to 90 days after the AKI event. All-cause mortality and a combined renal outcome of sustained doubling of SCr or progression to kidney failure were evaluated.
Results
Overall, 3.7% of the participants (n=7014) had AKI, 62.7% of whom (n=4400) survived 90 days. In the 3231 patients in whom recovery could be assessed over a median follow-up of 34 months, 30.8% (n=1268) of AKI survivors died and 2.1% (n=85) progressed to kidney failure. Participants who did not recover kidney function had a higher risk for mortality and adverse renal outcomes when AKI participants who recovered to within 25% of baseline SCr were used as the reference group (adjusted mortality hazard ratio (HR), 1.26; 95% confidence interval, 1.10, 1.43) (adjusted renal outcomes HR, 4.13; 95% confidence interval, 3.38, 5.04). Mortality HR was notably higher when participants failed to recover within 55% of baseline.
Conclusions
Renal recovery after AKI is associated with a lower risk of death or adverse renal outcomes after hospital discharge.
Introduction
CKD is a strong predictor of future hospitalization, cardiovascular events, and death as well as considerable health care costs (1–4). AKI is also recognized as a potent predictor of long-term morbidity and mortality, although the mechanism underlying this association remains poorly understood. There are emerging data that lack of recovery of renal function after AKI is an important contributor to the prevalence and progression of CKD (5,6). It remains uncertain to what extent the association between AKI and adverse outcomes is mediated by the AKI event itself, or via the development of de novo or progressive CKD as a result of nonrecovery from AKI.
Recovery of renal function after AKI has been primarily described in patients who required renal replacement therapy, and largely defined as subsequent independence from dialysis. The decision to initiate and discontinue renal replacement therapy for AKI is highly variable, and currently there is lack of consensus for defining renal recovery. In addition, it is established that milder forms of AKI not requiring dialysis are associated with both short-term and long-term mortality; however, little is known about renal recovery and its influence on outcomes in these patients.
Using a population-based cohort of adults with a hospitalization episode, we sought to characterize renal recovery in participants with AKI using the percentage change from the baseline prehospitalization serum creatinine (SCr). Furthermore, we aimed to determine whether recovery to prehospitalization level of kidney function mediates the risk of long-term outcomes associated with AKI, specifically survival, doubling of SCr, and ESRD.
Materials and Methods
Study Population
The study cohort included all adults (aged ≥18 years) residing in Alberta who were admitted to the hospital between November 1, 2002 and December 31, 2007, with at least one outpatient SCr measurement within 180 days before hospitalization, ≥1 measurement during the hospitalization, and ≥1 measurement in the follow-up period. Inclusion also required that the participants survived at least 90 days after hospitalization (non-AKI participants) or the AKI event. If participants had >1 hospitalization episode during this period, only the first hospitalization was considered. Participants with ESRD (estimated GFR [eGFR] <15 ml/min per 1.73 m2, chronic dialysis, prior kidney transplant) at baseline were excluded. All participants were followed from the date of their first available SCr measurement (index date) until March 31, 2009. Data were drawn from hospital discharge abstracts of Alberta Health and Wellness, the Northern and Southern Alberta Renal Programs, and the provincial laboratories of Alberta (7).
Assessment of Baseline Kidney Function
SCr measurements within the province were standardized across provincial laboratories to an isotope dilution mass spectrometry reference standard, and a laboratory-specific correction factor was applied when necessary (7). The Chronic Kidney Disease Epidemiology Collaboration prediction equation was used to determine eGFR (8). Baseline kidney function was defined as the mean of all outpatient SCr in the 6 months before hospitalization. In sensitivity analysis, we alternatively defined baseline kidney function as the last outpatient SCr measurement taken at least 7 days before hospitalization.
Identification of AKI
AKI was identified by changes between baseline (prehospital) and peak in-hospital SCr. AKI was defined as an increase in SCr by ≥100% (>2-fold increase) and/or requirement for acute dialysis during the index hospitalization, which was determined using a validated approach, based on diagnosis and procedural administrative codes (9). This definition is consistent with consensus criteria for AKI staging from the recently published Kidney Disease Improving Global Outcomes (KDIGO) AKI guidelines (10) (AKI stage 2).
Assessment of Comorbid Conditions
Relevant demographic characteristics and preexisting comorbid conditions as well as other details of hospitalization episode, including reasons for hospitalization, any intensive care unit stay, type of hospital, and admission to surgical or nonsurgical ward, were obtained using hospitalization data and claims files, using validated algorithms (Supplemental Table 1) (11). Charlson comorbidity indexes were calculated and reported. We obtained primary International Classification of Diseases (10th revision) codes and used these to classify primary admission diagnoses using a previously published approach (12). Resource intensity weights, similar to diagnostic related group weights used elsewhere, were used to categorize acuity and severity of illness (13,14).
Assessment of Renal Recovery after AKI
Post-AKI renal function was assessed using the first SCr drawn closest to 90 days after the AKI episode (range, 30–150 days) to allow sufficient time for recovery. A 90-day time frame for recovery was used based on the Kidney Disease Outcomes Quality Initiative guidelines (15), which define CKD as a persistent decline in kidney function lasting >90 days. Participants who received their first chronic dialysis treatment between hospital discharge and the end of the recovery assessment period (150 days after AKI) were excluded because creatinine-based measurement of renal recovery was considered not clinically relevant in patients on chronic dialysis, and to avoid misclassification of CKD progression to ESRD as AKI. Recovery was defined as a post-AKI SCr within 25% of the baseline (prehospitalization value) and independence from renal replacement therapy. This definition was based on recommendations by the Acute Dialysis Quality Initiative (16), and is consistent with contemporary definitions of AKI (16,17). In sensitivity analysis, we examined alternative definitions of renal recovery.
Assessment of Mortality, ESRD, and Sustained Doubling of SCr
Study outcomes were assessed from 90 days after the AKI event (or hospital admission for the non-AKI group) to March 31, 2009. The occurrence of ESRD treated with renal replacement therapy was identified using the registries of the Northern and Southern Alberta Renal Programs as previously described (7,18). Sustained doubling of SCr was defined as a doubling of outpatient SCr from the baseline value, which was sustained until the end of the study follow-up period. All-cause mortality was identified using administrative data sources (provincial vital statistics).
Statistical Analyses
Four categories of AKI exposure were defined: no AKI (participants who did not have an AKI episode during hospitalization), AKI with recovery, AKI without recovery, and AKI with unknown recovery (participants with no SCr measurements between 30 and 150 days after the AKI episode). Participants who had an AKI episode during hospitalization and recovered formed the reference group. Continuous variables were described using mean and SD or median with interquartile range as appropriate. Categorical variables were described as proportions of the cohort, with between-group comparisons performed using chi-squared tests and ANOVA or Kruskal–Wallis tests as appropriate.
Separate Cox proportional hazards models were fit for the outcomes of all-cause mortality, and the composite outcome of sustained doubling of SCr or ESRD requiring renal replacement therapy (dialysis or kidney transplantation). All patients were followed-up from 90 days after the AKI event/hospitalization until the end of the study, with further censoring for death in the model fit for the composite renal outcome and for ESRD in the model fit for mortality to address the issue of competing risks. In the base case model, only participants who had an AKI event were included. In sensitivity analysis, the non-AKI group was included to compare outcomes among the four groups. The fully adjusted models included terms for age, sex, primary admission diagnosis grouping, acuity of illness (resource intensity weights), requirement for acute dialysis, and comorbid conditions.
In the sensitivity analyses evaluating various definitions of renal recovery, the referent group was those participants who recovered to within 5% of baseline. Each level of recovery, defined in 10% increments (recovery within 5%–15%, 15%–25%, 25%–35%, 35%–45%, 45%–55%), was modeled individually, adjusting for the previously mentioned covariates. We anticipated that baseline kidney function would confound the association between renal recovery and renal survival and mortality; therefore, we also included baseline eGFR as an independent variable in models using 5 ml/min per 1.73 m2 increments.
The proportional hazards assumption was tested and satisfied. Statistical analyses were performed using STATA MP 11 software (StataCorp, College Station, TX).
Results
Patient Characteristics
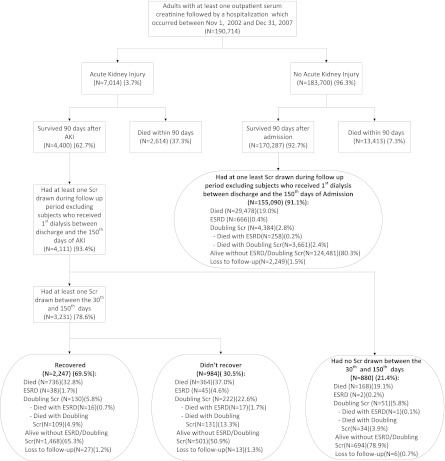
Between November 1, 2002 and December 31, 2007, 190,714 Albertan participants were hospitalized who had at least one outpatient SCr value within 6 months before hospitalization. Of these participants, 7014 (3.7%) experienced an episode of AKI during hospitalization. The 90-day mortality of participants with and without AKI was 37.3% and 7.3%, respectively. Of the AKI survivors, 4111 (93.4%) had at least one SCr drawn during the follow-up period, 78.6% of which had SCr drawn during the recovery period (30–150 days); 85 (2.1%) participants developed ESRD within this recovery assessment period (Figure 1). The characteristics of participants with and without AKI during the index hospitalization are presented in Table 1. Participants with AKI were more likely to be older, were more likely to be male, had greater vascular comorbidity, and were more likely to have a history of complicated diabetes, myocardial infarction, vascular disease, congestive heart failure, and cancer. They also had a higher acuity of illness. There were differences in reasons for hospitalization, with AKI participants more likely to have been admitted for respiratory, cancer, infectious, genitourinary, and cardiovascular conditions.
Figure 1.
Study flowchart. In this figure, ESRD and doubling SCr are mutually exclusive groups. If a participant had both ESRD and sustained doubling SCr, we classified him or her to the ESRD group. SCr, serum creatinine.
Table 1.
Patient characteristics (recovery defined as creatinine within 25% of baseline)
| All Patients | No AKI | AKI | P of AKI versus No AKI | AKI Recovered | AKI Not Recovered | AKI No SCr Drawn | P of AKI Recovered versus Not Recovered | |
|---|---|---|---|---|---|---|---|---|
| Demographics | ||||||||
| Number of participants (%) | 159,201 (100) | 155,090 (97.4) | 4,111 (2.6) | 2247 (54.7) | 984 (23.9) | 880 (21.4) | ||
| Age, mean (SD) | 62.7 (18.1) | 62.7 (18.1) | 65.1 (16.6) | <0.01 | 66 (16.1) | 64.5 (16.9) | 63.3 (17.5) | 0.02 |
| Male sex (%) | 46.8 | 46.6 | 51.5 | <0.01 | 53.0 | 49.3 | 50.3 | 0.05 |
| Comorbid disease (%) | ||||||||
| Myocardial infarction | 22.2 | 22.0 | 28.9 | <0.01 | 31.1 | 31.1 | 21.1 | 0.98 |
| Peripheral vascular disease | 15.2 | 15.1 | 22.5 | <0.01 | 24.0 | 24.7 | 16.3 | 0.69 |
| Cerebrovascular disease | 20.8 | 20.8 | 22.9 | <0.01 | 24.1 | 24.0 | 18.5 | 0.93 |
| Congestive heart failure | 26.4 | 26.0 | 43.1 | <0.01 | 44.6 | 51.4 | 29.9 | <0.01 |
| Uncomplicated diabetes | 19.4 | 19.4 | 19.5 | 0.85 | 19.5 | 17.9 | 21.4 | 0.27 |
| Complicated diabetes | 12.3 | 12.0 | 25.9 | <0.01 | 27.3 | 31.2 | 16.6 | 0.02 |
| Nondermatologic malignancy | 30.0 | 29.9 | 34.6 | <0.01 | 35.1 | 38.2 | 29.3 | 0.09 |
| Mean CCI score (median, interquartile range) | 3.4 (3, 1–5) | 3.4 (3, 1–5) | 4.6 (4, 2–6) | <0.01 | 4.9 (4, 2–7) | 5.2 (5, 3–7) | 3.5 (3, 1–5) | <0.01 |
| CIHI resource intensity weight | 1.9 (3.2) | 1.8 (2.9) | 5.1 (8.7) | <0.01 | 5.7 (9.9) | 5.8 (8.7) | 2.7 (3.6) | 0.71 |
| Primary diagnostic code for hospitalization (%) | ||||||||
| Cardiovascular | 18.6 | 18.6 | 16.8 | <0.01 | 16.6 | 21.2 | 12.3 | <0.01 |
| Respiratory | 6.7 | 6.7 | 8.2 | <0.01 | 8.9 | 5.9 | 8.9 | <0.01 |
| Gastrointestinal | 12.8 | 12.8 | 13.3 | 0.35 | 12.7 | 11.7 | 16.5 | 0.43 |
| Infectious disease | 1.3 | 1.2 | 5.5 | <0.01 | 5.3 | 5.9 | 5.6 | 0.53 |
| Cancer | 10.9 | 11.0 | 9.0 | <0.01 | 8.7 | 10.3 | 8.1 | 0.16 |
| Orthopedics | 9.3 | 9.4 | 4.6 | <0.01 | 4.0 | 4.6 | 6.5 | 0.42 |
| Hematologic | 1.1 | 1.1 | 1.0 | 0.66 | 0.9 | 1.4 | 0.8 | 0.22 |
| Genitourinary | 6.3 | 6.0 | 17.0 | <0.01 | 18.2 | 16.1 | 15.0 | 0.13 |
| Injury/poisoning | 7.9 | 8.0 | 6.2 | <0.01 | 7.1 | 4.9 | 5.5 | 0.02 |
| Other disease | 25.1 | 25.2 | 18.4 | <0.01 | 17.4 | 18.1 | 21.0 | 0.66 |
| Patients required acute dialysis during hospitalization (%) | 0.2 | 0.0 | 9.6 | <0.01 | 7.6 | 19.9 | 3.4 | <0.01 |
| Baseline kidney function | ||||||||
| Serum creatinine (μmol/L) (mean, SD) | 90.4 (32.7) | 90 (31.6) | 105.4 (60.4) | <0.01 | 111.2 (62.8) | 105.1 (63.1) | 91 (47.4) | 0.01 |
| eGFR (ml/min per 1.73 m2) (mean, SD) | 74.3 (24.9) | 74.5 (24.8) | 68.5 (28.3) | <0.01 | 64.9 (27.0) | 70.3 (31.2) | 75.6 (26.7) | <0.01 |
| Categories | ||||||||
| >60 | 71.1 | 71.5 | 59.1 | <0.01 | 53.6 | 61.4 | 70.5 | <0.01 |
| 45–60 | 17.4 | 17.3 | 20.1 | <0.01 | 22.2 | 16.7 | 18.8 | <0.01 |
| 30–44 | 8.7 | 8.5 | 13.1 | <0.01 | 15.1 | 12.9 | 8.2 | 0.10 |
| 20–29 | 2.4 | 2.3 | 5.7 | <0.01 | 6.9 | 6.1 | 2.4 | 0.43 |
| 15–19 | 0.4 | 0.4 | 2.0 | <0.01 | 2.2 | 2.9 | 0.2 | 0.22 |
CCI, Charlson Comorbidity Index; CIHI, Canadian Institute for Health Information.
Baseline kidney function was lower in participants who experienced AKI (mean eGFR 68.5 ml/min per 1.73 m2). Among those participants who developed AKI, 9.6% required acute dialysis. The median duration of follow-up for all study participants was 34 months (range, 0.13–74 months; 25th percentile, 20 months; 75th percentile, 50 months). Overall, 1.4% of study participants were lost to follow-up due to out migration from the province. The average number of SCr measurements drawn per patient in the follow-up period (90 days after AKI/hospitalization) was 25 in the participants who had an episode of AKI and 14 in participants who did not. Serum creatinine measurements used to determine recovery, or postdischarge renal status in the case of the non-AKI participants, were drawn a median of 90 days (25th percentile, 76 days; 75th percentile, 98 days) after the AKI event, or in the case of non-AKI participants, after hospital admission (Supplemental Figure 1).
Recovery of Renal Function
Of the 3231 survivors with AKI in whom recovery could be assessed, 2247 (69.5%) recovered to within 25% of their baseline SCr. Participants with congestive heart failure and complicated diabetes were less likely to recover renal function (Table 1). The mean prehospitalization eGFR was lower in AKI survivors who recovered than in those who did not recover (64.9 versus 70.3 ml/min per 1.73 m2; P<0.01); however, the requirement for renal replacement therapy during hospitalization was greater in those who did not recover renal function.
Renal Recovery and Survival
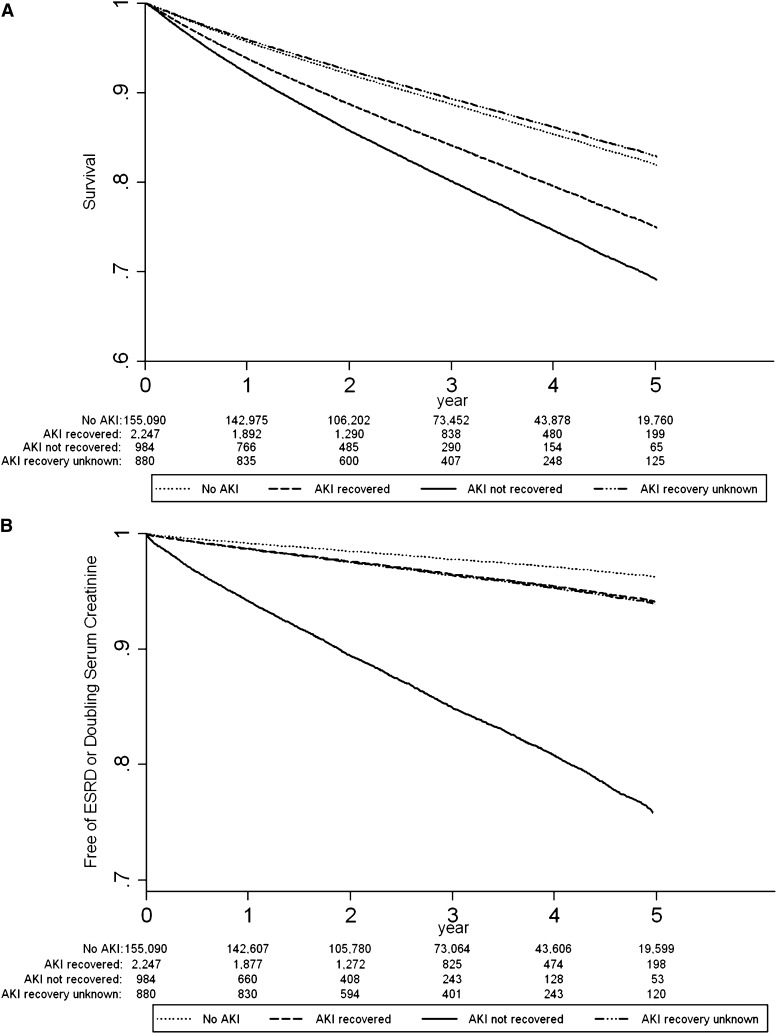
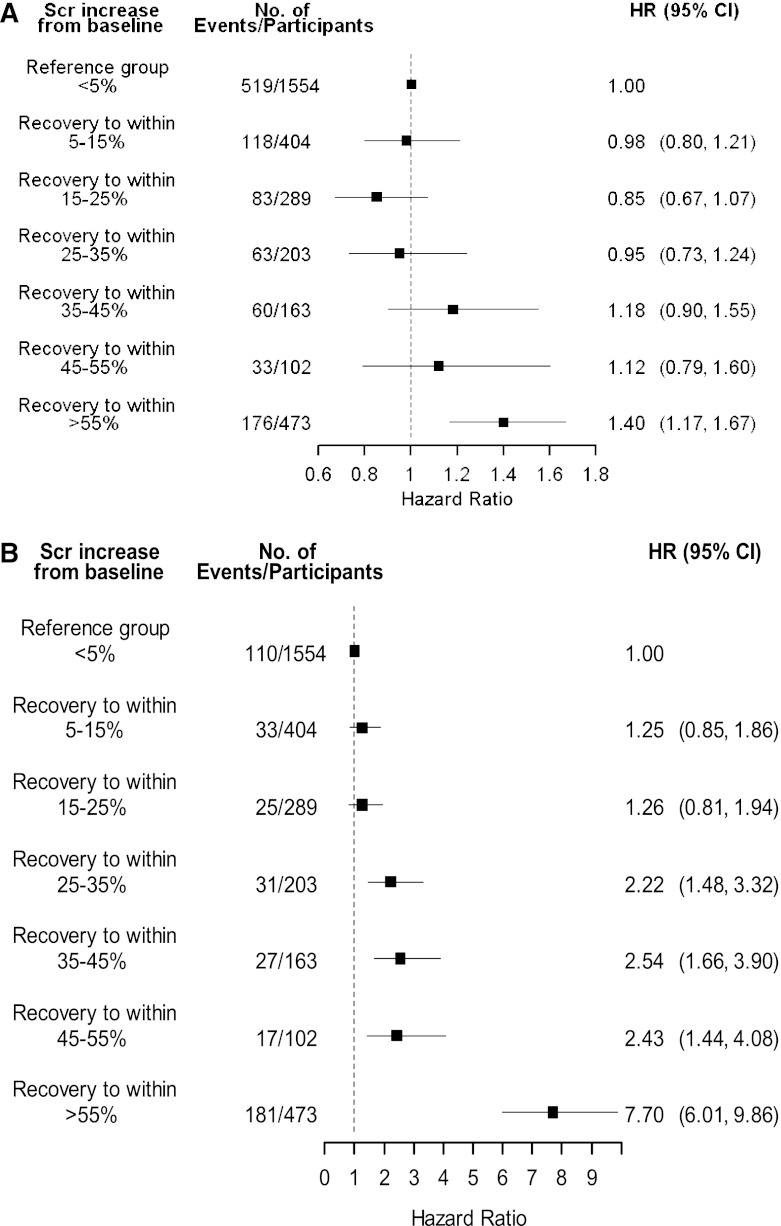
The adjusted survival curves for AKI recovery status and survival are presented in Figure 2A. In AKI survivors who recovered renal function, 67.2% of participants were alive at the end of follow-up compared with 63.0% of participants who did not recover. Those participants with AKI who did not have a SCr drawn within the defined recovery period (AKI recovery unknown) had similar survival to participants without AKI (Figure 1 and Supplemental Table 2). Further analysis of this group showed that 91% (n=801) recovered kidney function within 30 days of the AKI event. The adjusted hazard of mortality for those with AKI who did not recover renal function was 1.26 (95% confidence interval [95% CI], 1.10, 1.43) when participants who recovered were used as the reference group (Table 2). In sensitivity analysis we identified a recovery threshold by comparing mortality outcomes of participants at various levels of recovery to those who recovered to within 5% of baseline (Figure 3A). Mortality differences became significantly different between groups when recovery was defined as within 55% of baseline (adjusted hazard ratio [HR], 1.40; 95% CI, 1.17, 1.67). Sensitivity analysis using the last outpatient SCR >7 days before admission to define baseline kidney function did not change the results (results not shown).
Figure 2.
Renal recovery after AKI is associated with improved patient and renal survival. (A) Adjusted curves for survival in AKI survivors by recovery status (recovery defined as within 25% of baseline). (B) Adjusted curves for renal survival in AKI survivors by recovery status (recovery defined as within 25% of baseline). Adjusted for age, sex, myocardial infarction, peripheral vascular disease, cerebrovascular disease, congestive heart failure, uncomplicated diabetes, complicated diabetes, nondermatologic malignancy, baseline estimated GFR (by 5 ml/min per 1.73 m2 increments), acute dialysis during hospitalization, primary diagnostic code for hospitalization, and Canadian Institute for Health Information resource intensity weight.
Table 2.
Hazard ratios for death and renal outcomes among hospitalized participants by recovery group (recovery defined as within 25% of baseline)
| AKI Status | Participants (n) | Events, n (%) | HR (95% CI) | ||
|---|---|---|---|---|---|
| Doubling SCr/ESRD | Death | Doubling SCr/ESRD | Death | ||
| AKI recovered | |||||
| Crude HR | 2,247 | 168 (7.5) | 720 (32.0) | 1 | 1 |
| Fully adjusted HR | 1 | 1 | |||
| AKI not recovered | |||||
| Crude HR | 984 | 267 (27.1) | 347 (35.3) | 4.46 (3.68, 5.41) | 1.23 (1.08, 1.40) |
| Fully adjusted HR | 4.13 (3.38, 5.04) | 1.26 (1.10, 1.43) | |||
| AKI recovery unknown | |||||
| Crude HR | 880 | 53 (6.0) | 167 (19.0) | 0.71 (0.52, 0.97) | 0.51 (0.43, 0.61) |
| Fully adjusted HR | 0.90 (0.66, 1.24) | 0.59 (0.49, 0.70) | |||
Adjusted factors: age, sex, myocardial infarction, peripheral vascular disease, cerebrovascular disease, congestive heart failure, diabetes uncomplicated, diabetes complicated, nondermatologic malignancy, baseline estimated GFR (by 5 ml/min per 1.73 m2 increments), requirement for acute dialysis, primary diagnostic code for hospitalization, CIHI resource intensity weight. HR, hazard ratio; CI, confidence interval; SCr, serum creatinine.
Figure 3.
Identification of thresholds for significant renal recovery in relation to patient and renal survival. (A) Adjusted HRs for mortality of each mutually exclusive group during the follow-up period (recovery to within 5% is reference group) (n=3188). (B) Adjusted hazard ratios for ESRD/sustained doubling SCr of each mutually exclusive group during follow-up period (recovery to within 5% is reference group) (n=3188). Both A and B were adjusted for age, sex, myocardial infarction, peripheral vascular disease, cerebrovascular disease, congestive heart failure, uncomplicated diabetes, complicated diabetes, nondermatologic malignancy, baseline estimated GFR (by 5 ml/min per 1.73 m2 increments), acute dialysis during hospitalization, primary diagnostic code for hospitalization, and Canadian Institute for Health Information resource intensity weight. Analysis excludes 43 participants who started dialysis during the recovery assessment period. SCr, serum creatinine; HR, hazard ratio; 95% CI, 95% confidence interval.
Renal Recovery and Renal Survival
The adjusted survival curves for renal recovery status and the renal endpoints are presented in Figure 2B. Renal recovery was also associated with improved long-term renal prognosis, when assessed by the composite outcome of sustained doubling of SCr or ESRD requiring renal replacement therapy. Overall, 85 (2.1%) AKI survivors progressed to ESRD and 403 (9.8%) had a sustained doubling of SCr over the follow-up period, compared with 0.4% and 2.8% of participants in the non-AKI group. Compared with AKI survivors who recovered, the AKI nonrecovery group had a higher risk for the composite renal outcome (adjusted HR, 4.13; 95% CI, 3.38, 5.04) (Table 2). Participants in the AKI recovery unknown group were similar to those participants who recovered from AKI (Table 2). In sensitivity analysis, we explored whether varying the definition of renal recovery modified this result. We found that relative to the participants who recovered to within 5% of the baseline SCr, the cut point at which a significant difference in renal survival was observed was when recovery was defined as within 25%–35% of baseline (Figure 3B).
Discussion
We analyzed renal outcomes among survivors of hospitalization over a 6-year period using a large population-based database and found that the majority of participants with moderate to severe AKI (KDIGO stage 2 or greater) do recover renal function. Furthermore, we found that renal recovery status after AKI was associated with both long-term patient survival and progression to kidney failure over a median follow-up period of 2.8 years. Importantly, participants without recovery of renal function after AKI also had a higher long-term risk of doubling of SCr or ESRD. Our findings are consistent with those of others who have reported an association between AKI requiring renal replacement therapy and the development of ESRD, and further extend these findings to include participants with less severe forms of AKI.
Survivors of severe AKI requiring renal replacement therapy experience long-term adverse renal outcomes after hospital discharge; however, it is unclear as to whether these adverse outcomes are attributable to AKI or occur as an expected consequence of CKD resulting from nonrecovery of renal function. A large population-based study by Wald et al. (19) showed that dialysis requirement for AKI during hospitalization was associated with a three-fold increased risk of ESRD (HR, 3.23; 95% CI, 2.70–3.86) without an increase in risk of death. All participants were independent of dialysis at hospital discharge. Other smaller case series of critically ill patients with severe AKI have described varying degrees of renal recovery after hospital discharge—generally defined as independence from dialysis (5,20,21).
Several observational studies suggest that early renal recovery may result in improved short-term mortality; however, long-term renal outcomes have generally not been considered. A systematic review of renal recovery after AKI in elderly individuals reported that patients aged >65 years were less likely to recover kidney function (22). Swaminanthan et al. (23) found that the percentage decrease in the SCr value 24 hours after the peak SCr in AKI after cardiac surgery was associated with a significant decrease in the long-term risk of death (HR per 10% decrease in SCr, 0.82). Other studies that have evaluated the prognostic value of transient changes in kidney function during hospitalization show conflicting results (24–26). Such early assessment of recovery of kidney function likely relates to prerenal versus other causes of AKI, and although this has been linked to in-hospital prognosis, it is less helpful for guiding outpatient follow-up of survivors of AKI at hospital discharge and determining long-term prognosis.
This study has several important clinical and research implications. We were able to demonstrate that recovery of kidney function in AKI survivors is associated with better outcomes after AKI in a general population of hospitalized patients. Our study findings support the view that short-term outcomes after AKI are driven by acuity of illness and adverse long-term outcomes after AKI are, in large part, a consequence of resulting CKD. We were also able to confirm significant renal recovery to be within 25% of the baseline SCr, which may have implications with respect to risk classification and follow-up after AKI.
Several differences were observed between participants who recovered kidney function versus those who did not. In general, patients who recovered were older, male, less likely to have congestive heart failure or complicated diabetes, less likely to be treated with acute dialysis, and more likely to have a lower baseline eGFR. One possible explanation for the seemingly paradoxical association of recovery with baseline eGFR as observed is that SCr-based AKI definitions classify moderate AKI in CKD patients to be the same as severe AKI in patients with normal premorbid kidney function. This association between premorbid eGFR and outcomes after AKI has also been recently shown in cardiac surgery patients (27).
There are limitations to our study. We are limited to available data; however, >90% of participants had at least one SCr drawn after discharge. We were able to show that the participants who did not have SCr drawn during the specified recovery period (21% of the AKI cohort) had outcomes that were comparable with those without AKI (Figure 1); in fact, 91% of these participants recovered from AKI within 30 days. This suggests that our results are unlikely to be biased by missing measurements.
We also excluded participants who started chronic dialysis within 150 days of the AKI episode from further analysis because of concern regarding assessment of recovery as well as the possibility of misclassification of progression to ESRD as AKI. By doing so that we may have excluded patients with severe AKI who did not recover; however, this would bias our result toward the null, meaning our estimates for the HR associated with nonrecovery are likely conservative.
We did not include participants with the mildest form of AKI (KDIGO stage 1) in this study, because of concerns about interlaboratory variability in SCr measurement resulting in misclassification of AKI as well as uncertainty as to how to define recovery in this population. However, further analysis of this group showed that 67% recovered to within 25% of baseline within 30 days of the AKI event (Supplemental Table 3). It remains possible that our definitions of renal recovery are insensitive to clinically important outcomes such as de novo CKD. A recently published analysis of long-term AKI outcomes (26) demonstrated that transient AKI stage 1 is associated with de novo CKD (eGFR <60 ml/min per 1.73 m2); after adjustment for this, the association between AKI and mortality was not significant.
Participants who had SCr drawn before admission may represent a sicker population of participants (ascertainment bias); however, we previously showed that the magnitude of association between AKI and in-hospital mortality is highest in participants without significant CKD (28); therefore, exclusion of participants without prior creatinines who likely have normal or near normal kidney function may underestimate the reported effect estimates in this group. Finally, the use of administrative data does not allow assessment of AKI etiology, which may influence the likelihood of renal recovery and long-term outcomes. Previous studies of AKI in hospitalized patients would suggest that acute tubular necrosis and prerenal causes account for the vast majority of cases (29,30) and the findings of this study likely apply best to these diagnoses.
Recovery of kidney function after an AKI episode is associated with better long-term patient and renal survival than nonrecovery over a range of recovery definitions. Our study findings suggest that assessment of kidney function using SCr drawn 90 days after an AKI event may allow risk stratification of these participants for adverse long-term outcomes and assist in decision making with regard to ongoing follow-up. These findings should be confirmed in prospective studies of AKI.
Disclosures
None.
Acknowledgments
Phoebe Ye provided additional statistical support. N.P. had full access to all the data in the study and takes responsibility for the integrity of the data and the accuracy of the data analysis. A complete list of the membership of the Alberta Kidney Disease Network can be found at http://www.AKDN.info.
B.H. and S.K. were supported by career salary awards from the Alberta Heritage Foundation for Medical Research (AHFMR). B.H. was also supported by a New Investigator Award from the Canadian Institutes of Health Research. M.J. was supported by a Shire Biochem - KRESCENT Joint Fellowship and an AHFMR research award. This work was supported by a research grant from the Kidney Foundation of Canada.
Footnotes
Published online ahead of print. Publication date available at www.cjasn.org.
This article contains supplemental material online at http://cjasn.asnjournals.org/lookup/suppl/doi:10.2215/CJN.06480612/-/DCSupplemental.
See related editorial, “Is it AKI or Nonrecovery of Renal Function That Is Important for Long-Term Outcomes?,” on pages 173–176.
References
- 1.Levey AS, Atkins R, Coresh J, Cohen EP, Collins AJ, Eckardt KU, Nahas ME, Jaber BL, Jadoul M, Levin A, Powe NR, Rossert J, Wheeler DC, Lameire N, Eknoyan G: Chronic kidney disease as a global public health problem: Approaches and initiatives - a position statement from Kidney Disease Improving Global Outcomes. Kidney Int 72: 247–259, 2007 [DOI] [PubMed] [Google Scholar]
- 2.Tonelli M, Wiebe N, Culleton B, House A, Rabbat C, Fok M, McAlister F, Garg AX: Chronic kidney disease and mortality risk: A systematic review. J Am Soc Nephrol 17: 2034–2047, 2006 [DOI] [PubMed] [Google Scholar]
- 3.Trivedi HS, Pang MM, Campbell A, Saab P: Slowing the progression of chronic renal failure: Economic benefits and patients’ perspectives. Am J Kidney Dis 39: 721–729, 2002 [DOI] [PubMed] [Google Scholar]
- 4.Eknoyan G, Lameire N, Barsoum R, Eckardt KU, Levin A, Levin N, Locatelli F, MacLeod A, Vanholder R, Walker R, Wang H: The burden of kidney disease: Improving global outcomes. Kidney Int 66: 1310–1314, 2004 [DOI] [PubMed] [Google Scholar]
- 5.Bagshaw SM, Laupland KB, Doig CJ, Mortis G, Fick GH, Mucenski M, Godinez-Luna T, Svenson LW, Rosenal T: Prognosis for long-term survival and renal recovery in critically ill patients with severe acute renal failure: A population-based study. Crit Care 9: R700–R709, 2005 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 6.Hsu CY, Chertow GM, McCulloch CE, Fan D, Ordoñez JD, Go AS: Nonrecovery of kidney function and death after acute on chronic renal failure. Clin J Am Soc Nephrol 4: 891–898, 2009 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 7.Hemmelgarn BR, Clement F, Manns BJ, Klarenbach S, James MT, Ravani P, Pannu N, Ahmed SB, MacRae J, Scott-Douglas N, Jindal K, Quinn R, Culleton BF, Wiebe N, Krause R, Thorlacius L, Tonelli M: Overview of the Alberta Kidney Disease Network. BMC Nephrol 10: 30, 2009 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 8.Levey AS, Stevens LA, Schmid CH, Zhang YL, Castro AF, 3rd, Feldman HI, Kusek JW, Eggers P, Van Lente F, Greene T, Coresh J, CKD-EPI (Chronic Kidney Disease Epidemiology Collaboration) : A new equation to estimate glomerular filtration rate. Ann Intern Med 150: 604–612, 2009 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 9.Waikar SS, Wald R, Chertow GM, Curhan GC, Winkelmayer WC, Liangos O, Sosa MA, Jaber BL: Validity of International Classification of Diseases, Ninth Revision, Clinical Modification Codes for Acute Renal Failure. J Am Soc Nephrol 17: 1688–1694, 2006 [DOI] [PubMed] [Google Scholar]
- 10.KDIGO : Clinical practice guideline for acute kidney injury. Kidney Int Suppl 2: 1–138, 2012 [Google Scholar]
- 11.Quan H, Sundararajan V, Halfon P, Fong A, Burnand B, Luthi JC, Saunders LD, Beck CA, Feasby TE, Ghali WA: Coding algorithms for defining comorbidities in ICD-9-CM and ICD-10 administrative data. Med Care 43: 1130–1139, 2005 [DOI] [PubMed] [Google Scholar]
- 12.Chertow GM, Burdick E, Honour M, Bonventre JV, Bates DW: Acute kidney injury, mortality, length of stay, and costs in hospitalized patients. J Am Soc Nephrol 16: 3365–3370, 2005 [DOI] [PubMed] [Google Scholar]
- 13.Sarnecki L, Gordon L: Analysis of acuity trends using Resource Intensity Weights via the CIHI Portal. Stud Health Technol Inform 143: 42–46, 2009 [PubMed] [Google Scholar]
- 14.Jacobs P, Yim R: Using Canadian Administrative Databases to Derive Economic Data for Health Technology Assessments, Ottawa, Ontario, Canada, Canadian Agency for Drugs and Technologies in Health, 2009 [Google Scholar]
- 15.National Kidney Foundation : K/DOQI clinical practice guidelines for chronic kidney disease: Evaluation, classification, and stratification. Am J Kidney Dis 39[Suppl 1]: S1–S266, 2002 [PubMed] [Google Scholar]
- 16.Bellomo R, Ronco C, Kellum JA, Mehta RL, Palevsky P, Acute Dialysis Quality Initiative workgroup : Acute renal failure - definition, outcome measures, animal models, fluid therapy and information technology needs: The Second International Consensus Conference of the Acute Dialysis Quality Initiative (ADQI) Group. Crit Care 8: R204–R212, 2004 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 17.Mehta RL, Kellum JA, Shah SV, Molitoris BA, Ronco C, Warnock DG, Levin A, Acute Kidney Injury Network : Acute Kidney Injury Network: Report of an initiative to improve outcomes in acute kidney injury. Crit Care 11: R31, 2007 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 18.Manns BJ, Mortis GP, Taub KJ, McLaughlin K, Donaldson C, Ghali WA: The Southern Alberta Renal Program database: A prototype for patient management and research initiatives. Clin Invest Med 24: 164–170, 2001 [PubMed] [Google Scholar]
- 19.Wald R, Quinn RR, Luo J, Li P, Scales DC, Mamdani MM, Ray JG, University of Toronto Acute Kidney Injury Research Group : Chronic dialysis and death among survivors of acute kidney injury requiring dialysis. JAMA 302: 1179–1185, 2009 [DOI] [PubMed] [Google Scholar]
- 20.Schiffl H: Renal recovery from acute tubular necrosis requiring renal replacement therapy: A prospective study in critically ill patients. Nephrol Dial Transplant 21: 1248–1252, 2006 [DOI] [PubMed] [Google Scholar]
- 21.Lins RL, Elseviers MM, Daelemans R: Severity scoring and mortality 1 year after acute renal failure. Nephrol Dial Transplant 21: 1066–1068, 2006 [DOI] [PubMed] [Google Scholar]
- 22.Schmitt R, Coca S, Kanbay M, Tinetti ME, Cantley LG, Parikh CR: Recovery of kidney function after acute kidney injury in the elderly: A systematic review and meta-analysis. Am J Kidney Dis 52: 262–271, 2008 [DOI] [PubMed] [Google Scholar]
- 23.Swaminathan M, Hudson CC, Phillips-Bute BG, Patel UD, Mathew JP, Newman MF, Milano CA, Shaw AD, Stafford-Smith M: Impact of early renal recovery on survival after cardiac surgery-associated acute kidney injury. Ann Thorac Surg 89: 1098–1104, 2010 [DOI] [PubMed] [Google Scholar]
- 24.Tian J, Barrantes F, Amoateng-Adjepong Y, Manthous CA: Rapid reversal of acute kidney injury and hospital outcomes: A retrospective cohort study. Am J Kidney Dis 53: 974–981, 2009 [DOI] [PubMed] [Google Scholar]
- 25.Goldberg A, Kogan E, Hammerman H, Markiewicz W, Aronson D: The impact of transient and persistent acute kidney injury on long-term outcomes after acute myocardial infarction. Kidney Int 76: 900–906, 2009 [DOI] [PubMed] [Google Scholar]
- 26.Bucaloiu ID, Kirchner HL, Norfolk ER, Hartle JE, 2nd, Perkins RM: Increased risk of death and de novo chronic kidney disease following reversible acute kidney injury. Kidney Int 81: 477–485, 2012 [DOI] [PubMed] [Google Scholar]
- 27.Chawla LS, Zhao Y, Lough FC, Schroeder E, Seneff MG, Brennan JM: Off-pump versus on-pump coronary artery bypass grafting outcomes stratified by preoperative renal function. J Am Soc Nephrol 23: 1389–1397, 2012 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 28.Pannu N, James M, Hemmelgarn BR, Dong J, Tonelli M, Klarenbach S, Alberta Kidney Disease Network : Modification of outcomes after acute kidney injury by the presence of CKD. Am J Kidney Dis 58: 206–213, 2011 [DOI] [PubMed] [Google Scholar]
- 29.Liaño F, Pascual J, Madrid Acute Renal Failure Study Group : Epidemiology of acute renal failure: A prospective, multicenter, community-based study. Kidney Int 50: 811–818, 1996 [DOI] [PubMed] [Google Scholar]
- 30.Cerdá J, Bagga A, Kher V, Chakravarthi RM: The contrasting characteristics of acute kidney injury in developed and developing countries. Nat Clin Pract Nephrol 4: 138–153, 2008 [DOI] [PubMed] [Google Scholar]