Abstract
The B7 family consists of structurally related, cell-surface proteins that regulate immune responses by delivering costimulatory or coinhibitory signals through their ligands. Eight family members have been identified to date including CD80 (B7-1), CD86 (B7-2), CD274 (programmed cell death-1 ligand [PD-L1]), CD273 (programmed cell death-2 ligand [PD-L2]), CD275 (inducible costimulator ligand [ICOS-L]), CD276 (B7-H3), B7-H4, and B7-H6. B7 ligands are expressed on both lymphoid and nonlymphoid tissues. The importance of the B7 family in regulating immune responses is clear from their demonstrated role in the development of immunodeficiency and autoimmune diseases. Manipulation of the signals delivered by B7 ligands shows great potential in the treatment of cancers including leukemias and lymphomas and in regulating allogeneic T-cell responses after stem cell transplantation.
Introduction
Cancer and the immune system are fundamentally interrelated.1,2 Cancer cells express tumor-specific aberrant antigens3,4 and must therefore evade immune detection to survive,5 either by inducing immunosuppression2 or deriving survival signals from tumor-infiltrating immune cells.6 T cell–mediated antitumor immunity4 requires recognition of cancer-associated antigen by the major histocompatibility complex (MHC), and appropriate costimulatory and repressive secondary signals arising from complex interactions with other immune, stromal, and tumor cells. Dysfunction of costimulation pathways may contribute to failed antitumor immunity.
The B7 system (Table 1) is one of the most important secondary signaling mechanisms and is essential in maintaining the delicate balance between immune potency and suppression of autoimmunity. Potential therapeutic applications include immune-boosting adjuvants to conventional anticancer therapy, hematopoietic stem cell transplantation (HSCT), antitumor vaccines, bioengineered T cells as well as attenuation of graft-versus-host disease (GVHD). The B7 family is only one aspect of a complex signaling network (Figures 1 and 2) that comprises other immunoglobulin superfamily (IGSF) members, the tumor necrosis factor superfamily (TNFRSF), chemokines, cytokines, and adhesion molecules. However, based on a substantial evidence base and a growing therapeutic armory, the B7 family requires particular attention, as summarized in Table 2.7
Table 1.
Nomenclature of B7 family molecules
| CD designation | Molecule | “B7” designation | Eponyms | “Common” name | Major ligands |
|---|---|---|---|---|---|
| CD80 | B7-1 | B7-1 | B7 | CD80 | CD28, CD152 |
| CD86 | B7-2 | B7-2 | — | CD86 | CD28, CD152 |
| CD274 | Programmed cell death ligand 1 | B7-H1 | PD-L1 | PD-L1 | CD279 (PD-1) |
| CD275 | Inducible costimulator ligand | B7-H2 | B7RP-1, B7h, ICOS-L | ICOS-L | CD278 (ICOS) |
| CD276 | B7 homologue 3 | B7-H3 | — | — | ??TLT-2 |
| B7 homologue 4 | B7-H4 | B7S1, B7x | — | ??BTLA | |
| B7 homologue 6 | B7-H6 | — | — | NKp30 | |
| CD273 | Programmed cell death ligand 2 | B7-DC | PD-L2 | PD-L2 | CD279 |
| CD277 | Butyrophilin SF3 | — | BT3.1, BTF5 | — | ? |
The CD designation is used preferentially in this review.
— indicates no established designation in this category; ?, unknown; and ??, evidence is contested or based on limited date.
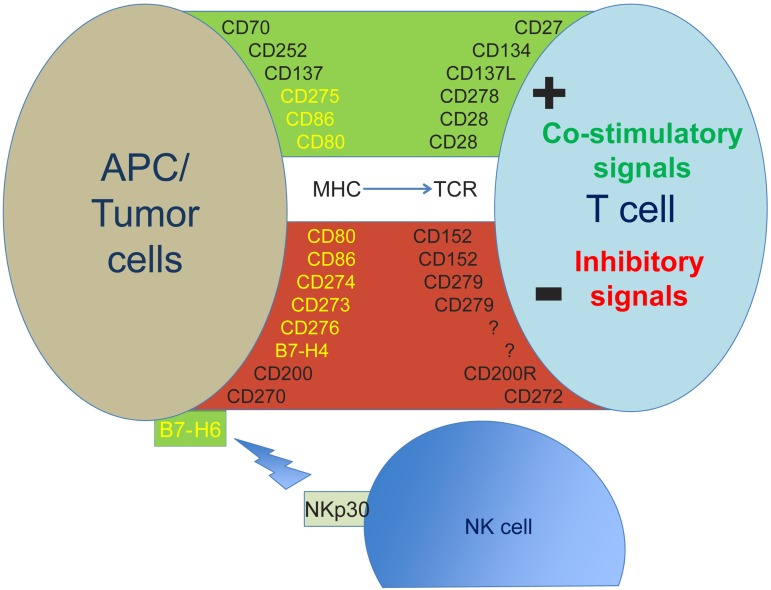
Figure 1.
Expression of selected antigens expressed on the cell surface of PC or tumor cells and their costimulatory or inhibitory ligands on the surface of T cells or NK cells. B7 family members are shown in yellow. APC indicates antigen presenting cell; MHC, major histocompatibility complex; NK, natural killer cell; and TCR, T-cell receptor.
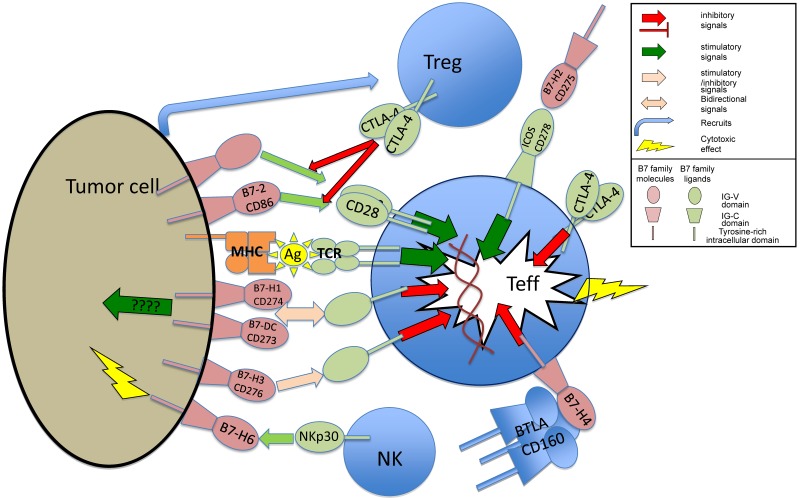
Figure 2.
Expression of B7 family members and their receptors have complex interactions with the tumor immune environment. Treg indicates T regulatory cells; Teff, T effector cells; ????, limited evidence base for mechanism; Ag, antigen presented in MHC complex; MHC, major histocompatibility complex; and TCR, T cell–receptor complex.
Table 2.
Summary of evidence for role and therapeutic potential of the B7 family molecules in hematological malignancy, with citations discussed in this review
| B7 targets | Phase 1/2 trial agent | Preclinical evidence base | Phase 1/2 evidence oncology | Phase 1/2 evidence Hemato-oncology |
|---|---|---|---|---|
| Augmentation of systemic antitumor immunity | ||||
| CTLA4 blockade | Ipilimumab | 18 | 46, 47 | 48 |
| PD-1/PD-L1 blockade | BMS936558, BMS936559, MK-3475 | 60-62 | 92-94 | 95 |
| ICOS pathway modulation | X | 169, 171 | X | X |
| Tumor-directed therapy against B7 targets | ||||
| CD80 | Galiximab | 18 | X | 42–44 |
| PD-L1 | X | 57, 61 | 92-94 | X |
| B7-H6 | X | 191, 192 | X | X |
| Augmentation of GVL | ||||
| CTLA4 blockade | Ipilimumab | 97 | 46, 47 | 96 |
| PD-1/PD-L1 blockade | X | 98 | X | X |
| ICOS pathway modulation | X | 169 | X | X |
| Attenuation of GVHD | ||||
| CTLA4 stimulation | CTLA4.Ig | 127 | X | 128, 129 |
| CD80/86 blockade | CD80/86 mAb | 127 | X | X |
| ICOS pathway modulation | X | 173, 174 | X | X |
| Bioengineered T cells | ||||
| CD28 CAR | Modified T cell | 123 | X | 125, 126 |
| CD80 CAR | Modified T cell | 124 | X | X |
| NKp30 CAR | Modified NK cell | 191 | X | X |
| Antitumor vaccine augmentation | ||||
| CTLA4 blockade | Ipilimumab | 18 | 110 | 48 111 |
| PD-1/PD-L1 blockade | X | 112, 113 | X | X |
| Treg depletion and inactivation | ||||
| CTLA-4 blockade | X | 136, 137 | X | X |
mAb indicates monoclonal antibody; NK, natural killer; CTLA, cytolytic T cell–associated sequence-4; PD, programmed cell death; ICOS, inducible costimulator; CAR, chimeric antigen receptor; and X, there are currently no data or therapeutic agent available.
The standardized approach to nomenclature is the cluster of differentiation (CD), which has not yet been applied to all members of the family. While many researchers are more familiar with their original names, which are still widely used in the literature, for the purposes of this review, the CD designation is used wherever possible.
B7 founder members
CD80, CD28, and CD152 (CTLA-4)
The first B7 family member was a molecule discovered on activated, proliferative splenic B lymphocytes,7 and termed B7 in an early nomenclature convention attributed to B cell–defining markers. Subsequent cloning and sequencing of the B7 gene revealed sequence homology with IGSF members and expression in a variety of lymphoid malignancies.8 B7 was found to bind CD28,9 an IGSF member on T cells, which augmented their activation.10–12 Further molecular profiling identified a CD28 homologue, CD152 (cytolytic T cell–associated sequence-4 [CTLA-4]),13 which mapped to the same chromosomal region as CD2814 and bound B7 more potently.15 The B7 signal blockade using a CTLA-4.Ig construct led to suppression of humoral16 and cell-mediated immune responses,17 while transfection of immunogenic murine melanoma cell lines with B7 enhanced antitumor immunity, which could be abrogated by CTLA-4.Ig.18
Initial experiments suggested that both anti-CD28 and anti–CTLA-4 monoclonal antibodies (mAbs) were T-cell activators,19 but contrary evidence showing a suppressive role for CTLA-4 soon emerged20,21 and was conclusively demonstrated in Ctla-4−/− mice, which rapidly develop fatal lymphoproliferation and multisystemic autoimmunity.22,23 Multiple mechanisms for CTLA-4–mediated immunosuppression are proposed (Figure 3), including competition for CD28 binding and induction of suppressive intracellular signaling pathways (reviewed in Rudd and Schneider24).
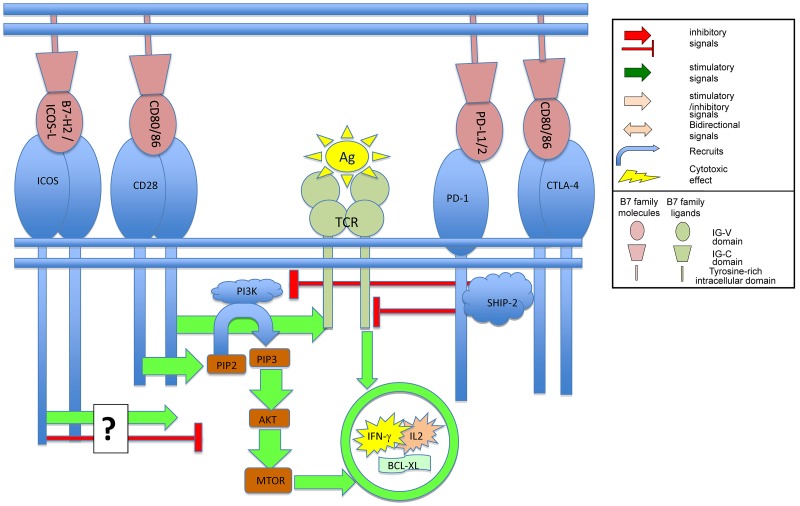
Figure 3.
Binding of B7 family members to their ligands on T cells regulates TCR mediated signaling.
Differential cellular binding of 2 B7-directed mAbs (anti–BB-1 and anti-B7) suggested distinct molecular targets (B7-1/CD80 and B7-2/CD86),25,26 a finding confirmed when the B7-2 gene was cloned from a CTLA-4.Ig-binding activated-B-cell cDNA library.27 Both molecules were demonstrated on antigen-presenting cells (APCs) including dendritic cells (DCs) and activated macrophages.28
CD80/CD86 expression in malignancy
A role for B7 in antitumor immunity was confirmed by the demonstration that cytotoxic T cells better eradicated murine malignancies transfected to express B7-1 and B7-2.18 In contrast to solid tumors, both molecules are expressed innately in many hematologic malignancies.29–31 After in vitro activation, follicular lymphoma (FL) cells up-regulate CD80/CD86 and other costimulatory and adhesion molecules,32 thereby increasing APC activity and augmenting primed T-cell responses. The malignant Hodgkin Reed Sternberg (HRS) cells of classic Hodgkin lymphoma (CHL) express high levels of CD80/CD86.33,34 Expression in multiple myeloma (MM) is variable, and may influence prognosis.35 Expression is low in chronic lymphocytic leukemia (CLL) unless the cells are stimulated,36 and rare in acute leukemia.37,38 However, expression of these costimulatory molecules alone is clearly inadequate for effective antitumor immunity because even malignancies showing high expression levels inevitably progress without therapy. Both molecules are widely expressed in the tumor immune microenvironment, and loss of expression may contribute to the failed antitumor responses.30,39,40
Therapeutic modulation of CD28, CD152, and CD80/86
CD80 has been targeted using the mAb galiximab41 in phase 1 and 2 trials in non-Hodgkin lymphoma (NHL) and CHL, both alone42 and combined with rituximab.43,44 Off-target immune suppression through CD80 blockade may limit the efficacy of such an approach. Anti-CD28 mAb therapy showed preclinical promise as an agent that could specifically expand regulatory T-cell subsets as therapy for autoimmune disorders. However, the subsequent clinical trial led to disastrous toxicity resulting from the use of too high a dose of the antibody and an unanticipated massive cytokine storm,45 urging caution in future therapeutic approaches to B7 pathway modulation. The first B7 pathway-targeting agent, anti–CTLA-4 mAb (ipilimumab) has now been Food and Drug Administration (FDA)–approved for the treatment of metastatic melanoma.46,47 Evidence is accumulating for its efficacy in hematologic malignancies, including in vitro enhancement of antitumor T-cell responses.48 Efficacy as a single agent is limited, although use in combination therapies may improve this.
Expansion of the B7 family and an immunosuppressive axis: CD274 (B7-H1/PD-L1), CD273 (B7-DC /PD-L2), and CD279 (PD-1)
A GenBank database B7 homology search identified a gene (B7-H1/CD274) encoding a protein distinct in amino acid sequence and tissue distribution from the founder B7 members, but with a similar tertiary protein structure.49 Expression in hematologic tissue was restricted to activated T cells and monocytes, although nonhematologic expression was widespread. Its natural ligand was neither CD28 nor CTLA-4 and proved elusive. In contrast to the effects of CD80/CD86.Ig, B7-H1.Ig enhanced proliferation in activated T cells, but induced IL10 and IFN-γ production with negligible IL4 and IL2.
A subtractive cDNA library comparing DCs and macrophages revealed another homologous protein (B7-DC) whose structure and genomic sequence was similar to B7-H1, and which failed to bind CD28 or CTLA-4.Ig.50 B7-DC.Ig stimulated activated T cells more potently than CD80.Ig, inducing IFN-γ and IL2 production and, in contrast to B7-H1, very little IL10. Its ligand, programmed cell death 1 [PD-1], a molecule discovered a decade before, also bound B7-H1. The properties of this new receptor/ligand interaction were not stimulatory like CD28, but profoundly inhibitory.
An experiment designed to reveal mechanisms of programmed cell death, applied subtractive hybridization cDNA library cloning to T-cell lines and identified a cell death–specific gene.51 CD279 (PD-1) was an IGSF homologue and, consistent with a role in T-cell turnover, found predominantly in the thymus. Expression was confirmed in activated circulating human T and B cells.52,53 Pd-1−/− mice developed splenomegaly and a systemic autoimmune syndrome suggesting a role in immunosuppression.54
Structural similarities between CD279 and CD152 (CTLA-4), but without CD80/86 binding,55 suggested an interaction between the B7-stimulatory and PD-1–inhibitory systems. A PD-1–specific ligand, coexpressed with CD80/CD86 on APCs and other nonhematopoietic tissues, was discovered through further B7-homologue searches and cloned from human and murine cDNA libraries.56 Termed CD274 (PD-1-ligand-1 [PD-L1]), ligation with PD-1 induced profound suppression of T-cell proliferation and cytokine secretion, which was overcome by excess anti-CD28 or anti-CD3. PD-L1 and B7-H1 transpired to be the same molecule, while B7-DC,50 independently named PD-L2 (CD273) after an expressed sequence tag (EST) database search for PD-L1 homologues,57 identified the second ligand for CD279.
Although initial in vitro work showed PD-L1/PD-L2.Ig transmitted stimulatory signals to activated lymphocytes,49,50 binding to their ligand PD-1 conveyed potent suppressive signals.56,57 Evidence for bidirectional signaling emerged from murine B cell– and T cell–specific gene deletion and transplantation experiments.58 Knock-out of T cell–specific PD-1 or B cell–specific PD-L2 impaired long-lived plasma cell formation, implying a stimulatory role for reverse signaling. Receptor/ligand pairing is also more promiscuous than originally believed. A search for binding partners for CD80 using Ctla-4−/−Cd28−/− activated CD4+ T cells suggested PD-1 as a candidate, with CD80 signaling through PD-1 independent of CTLA-4 or CD28, to inhibit T-cell proliferation.59 The complexity of the B7 system is becoming clear: context and direction of signaling determines responder cell outcome.
CD279 (PD-1), CD274 (PD-L1), and CD273 (PD-L2) expression in malignancy
The PD-1/PD-L1/PD-L2 axis has been widely demonstrated to contribute to failed antitumor immunity. Pd-1−/− mice and Pd-1wt/wt mice treated with anti–PD-L1 antibody demonstrate superior rejection of PD-L1–expressing tumors60 through PD-1–dependent and –independent pathways.61 Growth of pancreatic cancer cells in immunocompetent B6 mice is impaired by administration of anti–PD-1 or PD-L1 mAbs.62 Tumor-specific T cells fail to lyse melanoma cells in host 2C/Rag2−/− mice despite stimulatory IFN-γ treatment because IFN-γ also induces up-regulation of immunosuppressive PD-L1.63 PD-L1 is expressed in many tumors and cell lines57,61 and appears to be associated with poorer outcome in solid malignancy,62,64–67 while PD-1 expression by tumor-infiltrating lymphocytes (TILs) may also confer poorer outcomes.68 After treatment using a tumor-vaccination strategy of murine acute leukemia, long-lived residual leukemia cells were found to up-regulate PD-L1, suggesting a mechanism of leukemia persistence and relapse.69 These cells resisted CTL-mediated cell lysis, which could be overcome by anti–PD-L1 mAbs. However, blockade using anti–PD-1 mAbs did not reverse CTL resistance, while blocking CD80 had a similar effect to a PD-L1 block. The B7 systemÆs complexity is further illustrated by these findings: CD274 appears to have further immunosuppressive binding partners, while CD80 may be capable of mediating suppressive as well as costimulatory signals.70
There is sparse and conflicting evidence for CD274 (PD-L2) expression in acute leukemia,71,72 although the molecule is apparently up-regulated on malignant cell exposure to IFN-γ or Toll-like receptor (TLR) ligand, and at relapse. Evidence for reduced T cell–mediated specific lysis in vitro correlates with CD274 expression and is reversible by blocking CD274.73 Functional work in CLL has demonstrated a clear role for the PD-1/PD-L1 axis in mediating systemic, as well as antitumor, immune defects.74 Expression of CD279 by circulating T cells and CD274 by leukemia and FL cells is greater than in nonmalignant control cells, while CD274 expression is associated with poorer clinical outcome. T cells derived from patients with various malignancies have impaired in vitro ρ-GTPase signaling with reduced immune synapse formation.74 This defect is reversed by treatment with anti-PD1 and anti–PD-L1 mAbs, along with blockade of further inhibitory molecules CD200, CD270, and CD276 (B7-H3). Lenalidomide exposure also reverses this defect, coincident with down-regulation of CD274 and CD276 expression.74 These findings, along with evidence that CD80 expression is up-regulated on lenalidomide-exposed CLL cells,75 provide some insight into the drugÆs mechanism of action. In patients with adult T-cell leukemia/lymphoma (ATLL), CD279 expression on CD8+ T cells is up-regulated compared with ATLL-free HTLV1+ (human T-cell lymphotrophic virus) controls.76
CD279 is invariably expressed in angioimmunoblastic T-cell lymphoma, frequently in CLL, but absent in most other NHL.77–79 CD279 is expressed in the immune microenvironment of NHL, where it may have a prognostic significance,80 and there are conflicting reports of expression in the CHL microenvironment.81–83 Evidence for expression of CD274 in NHL is sparse and inconsistent. Immunohistochemistry and flow cytometric studies have shown expression by the malignant cell in the majority of primary mediastinal large B-cell lymphoma (PMBL)84 and anaplastic large cell lymphoma (ALCL), a minority of diffuse large B-cell lymphoma (DLBCL), and not at all in most other NHL.85 CD274 is overexpressed in the HRS of CHL82,86 associated with copy number aberrations86,87 and activating translocations involving the 9p24 chromosomal region containing the CD273 and CD274 genes,88 and upstream through the CD274 and CD273 expression-regulating JAK2 pathway.86 Limited in vitro evidence suggests that there is CD274-dependent suppression of T cells in CHL, evidenced by suppressed proliferation and IFN-γ production.82,85
There is variable expression of CD274 in myeloma89,90 with evidence for an important role in myeloma-induced immunosuppression. CTL generated in vitro by exposure to IFN-γ–treated myeloma cell lines show impaired cytolysis of subsequently presented target myeloma cells.89 NK-cell trafficking and cytotoxic potency in vitro is augmented by pretreatment with anti–PD-1 mAb.91 An autologous transplantation and costimulator/vaccine-based treatment strategy for myeloma in a cell line–based mouse model, which had showed evidence for the emergence of dysfunctional circulating CD279-expressing CD8+ T cells, is augmented by the administration of PD-L1 antibodies.90
While somewhat conflicting and lacking functional corroboration, these findings suggest that therapeutic modulation of this axis in hematologic malignancy may have a role.
Therapeutic modulation of CD274, CD279, and CD273
Fully humanized IgG4 mAbs targeting the PD-1/PD-L1 axis have reached clinical trials. Anti PD-L1 therapy (BMS936559) was administered to 160 heavily pretreated patients with a diverse group of solid tumors in a phase 1 trial.92 Objective and often durable responses were observed in up to 19% of patients, along with autoimmune and infusion reactions consistent with the murine PD-L1−/− models. A similar clinical trial of an anti–PD-1 monoclonal antibody therapy (BMS936558) demonstrated comparable response rates in 296 patients.93 There was an association between response and level of tumor PD-L1 expression. Further combination therapy approaches using anti–PD-1 mAbs are under way, including combination with the anti–CTLA-4 mAb ipilimumab in melanoma (NCT01024231), with conventional combination chemotherapy in non-small cell lung cancer (NCT01454102) and with tyrosine kinase inhibitors sunitinib and pazopanib in renal cell carcinoma (NCT01592370). Another PD-1 antibody (MK-3475) has also shown activity in patient with advanced solid tumors.94 A phase 1 safety and pharmacokinetic study of the anti–PD-1 mAb in advanced diverse hematologic malignancies showed a reasonable safety profile and a 33% overall response rate95 with further monotherapy and combination therapy trials proposed. The results are promising, but these drugs remain at an early stage of development and are likely to find their most active role as immune adjuvants to conventional therapy.
B7 pathway modulation to augment immunotherapy
Allogeneic HSCT
Failure of allogeneic HSCT may in part be mediated by suppressive B7 signaling. Anti–CTLA-4 mAb therapy has been applied successfully in phase 2 clinical trials to treat relapsed CHL, myeloma, and leukemias after allogeneic HSCT96 without the anticipated exacerbation of GVHD seen in murine transplantation experiments.97 Use of PD-L1/PD-1 axis blockade to enhance allogeneic T-cell responses has not yet reached clinical trial, although there is promising preclinical data in a murine AML model using TCR-engineered AML antigen-specific allogeneic T cells.98 Late transfer of these cells led to poorer antitumor responses accompanied by PD-1 overexpression. Responses were restored by treatment with anti–PD-L1 mAb. PD-1 blockade, however, accelerated GVHD lethality.99
Tumor vaccination strategies
B7 molecules may play an essential role in cancer vaccination strategies. Mouse model immunization strategies using highly immunogenic cancer cell lines showed early success, particularly with chemokine costimulatory approaches.100 Tumor lysate-pulsed DCs have been incorporated into some vaccination strategies,101 showing modest success in patients with advanced lymphoma.102 Idiotype-specific tumor vaccines have been produced for myeloma103 and NHL,104,105 while immunogenic tumor-specific antigens have been identified in CLL106,107 and acute leukemia.108 However, vaccination has yet to make any real impact on cancer therapy: an informal meta-analysis incorporating almost 1000 patients with a variety of malignancies showed an overall response rate of < 4%.109
Combining vaccination strategies with immunosuppressive B7 pathway blockade may improve these disappointing outcomes. CTLA-4 mAb-enhanced melanoma rejection has been demonstrated in a clinical trial,110 but studies in NHL have been disappointing, with minimal responses achieved in vaccinated patients treated with ipilimumab.48,111 Encouraging in vitro evidence suggests that a similar strategy could be applied to PD-1/PD-L1 axis blockade, although this has yet to be incorporated into any clinical trial. One group showed PD-L1/PD-L2 siRNA silencing improved DC-induced leukemia-specific T-cell responses,112 while another demonstrated enhanced T-cell responses after PD-1 antibody treatment in a myeloma/DC fusion vaccination strategy.113
Bioengineered T cells
In contrast to the active immunization strategies outlined in the previous paragraph, an alternative strategy is to expand and reinfuse autologous bioengineered T cells. Autologous TILs extracted from melanoma biopsies, expanded and reinfused into immunosuppressed recipients, have shown some therapeutic success114–116 but failed to translate to other malignancies. Attempts to augment autologous T-cell specificity for tumor antigens led to TCR bioengineering approaches, first against EBV-driven lymphoproliferative tumors117 and subsequently other tumor-specific antigens.118–120 While clinical responses are reported, reactive clones tend to rapidly lose efficacy or die off altogether.121 Subsequent developments used tumor antigen-inoculated transgenic mice whose T cells express human MHC, expanding tumor-specific T-cell clones and hence deriving ôready rearrangedö TCR genes for transfection into patient-derived T cells.122 Newer techniques incorporate novel receptor constructs into autologous T cells using chimeric antigen receptors (CAR). Here, a rearranged, antigen-specific immunoglobulin variable region gene is combined with intracellular domains of the TCR gene, thus bypassing MHC-restricted antigen recognition. CARs have the advantage of being amenable to further augmentation with costimulatory motifs, of which CD28123 and CD80124 have demonstrated efficacy. CARs incorporating costimulatory modifications have reached phase 1 clinical trials in NHL125 using an anti–CD19-CD28-TCRζ chain construct, and CLL, using an anti–CD19-CD137 construct (a TNFRSF member). Promising results have been observed, even in refractory patients.126 Further optimization is needed and B7 family members will play a central role in this.
GVHD attenuation
Allogeneic organ transplantation tolerance may be induced through CD80/CD86 blockade,17 and this finding has encouraged research into a role for the B7 molecules in GVHD. Fully HLA-mismatched mixed lymphocyte reactions are partially abrogated by CTLA-4.Ig or CD80/CD86 antibody treatment, and fully by combination treatment.127 This observation led to a promising clinical trial using CTLA-4.Ig to anergize donor lymphocytes and attenuate GVHD after mismatched allogeneic HSCT for hematologic malignancy.128,129 Early immune reconstitution with relatively low rates of acute and chronic GVHD and viral infection was observed, without the high relapse rates seen after T-cell depletion. Another potential GVHD-attenuating strategy, through PD-1/PD-L1/2 axis manipulation, is a promising hypothetical concept.99
Regulatory T cells
The role of regulatory T cells (Tregs) in suppression of successful antitumor immunity is becoming more evident. There is an association between adverse outcome in solid tumor and increased FOXP3-expressing cells in the microenvironment130 suggesting that their presence suppresses antitumor immunity.131,132 However, in B-cell malignancies the converse has been found, with increased infiltrate associated with improved outcome.133–135 CTLA-4136,137 and PD1/PD-L1138–140 are key pathways used by Tregs to mediate their suppressive activity, hence therapeutic interference with these pathways may contribute by depleting this profoundly suppressive population. These possibilities are under active investigation.
Another costimulatory B7 pathway: ICOS and B7-H2
A further avenue of B7 family costimulatory molecules opened with the discovery of another CD28-like molecule (CD28-related peptide-1 [CRP-1]).141 The human equivalent was confirmed to be a stimulatory molecule expressed only on activated T cells, hence termed inducible costimulator142 (ICOS/CD278). A second clone from the same library, termed B7-related peptide-1 (B7RP-1), had structural similarities to CD80/CD86. ICOS failed to bind CD80/CD86, B7RP-1 failed to bind to CD28 or CTLA-4, but ICOS and B7RP-1 specifically bound each other. B7RP-1 had already been discovered and functionally characterized by an independent group and named ôB7hö143; its human homolog was identified and named B7-H2.144 This was the same molecule, ICOS-ligand (B7RP-1/B7h/ICOS-L/CD275), and a later study showed that ICOS-L could bind and costimulate via CD28.145
ICOS may play a role in maintaining durable immune reactions146 and is expressed at particularly high levels in germinal center T cells or TFH (follicular helper) cells.147,148 ICOS-L is constitutively expressed by APCs as well as diverse nonhematologic tissues143 and down-regulated with ongoing inflammation,149,150 in contrast to the activation-induced CD28 ligands CD80/CD86.141,151 In CD28−/− mice, germinal center formation is rescued by ICOS overexpression, while inappropriate overexpression in CD28wt/wt mice induces systemic autoimmunity. T cells derived from ICOS−/− mice show normal thymic T-cell development but dysfunctional proliferation, cytokine and immunoglobulin production, and germinal center formation while paradoxically conferring greater susceptibility to Th1/IFN-γ–mediated disease including experimental autoimmune encephalitis (EAE).152 The contradictory functions of ICOS suggest a more complex role in T-cell regulation, perhaps favoring IL4/IL10/IL13 and humoral responses at the expense of IFN-γ–mediated cytotoxic/Th1 responses.153 ICOS also plays a role in maintaining immunosuppressive CD4+ T-cell subsets, with ICOS+ enriched CD4+ cell cultures secreting more IL10 and less TNFα than ICOS-depleted systems154 and ICOS, with FOXP3, essential for proper Treg development in the context of autoimmune diabetes and airway immune tolerance in ICOS−/− mice.155
Mutations in the human ICOS gene lead to an attenuated adult-onset common variable immunodeficiency (CVID),156 a predominantly B-cell defect likely arising from a loss of TFH cell function. The disease manifests with a variety of autoimmune phenomena as well as cancer and infection susceptibility.157 The complexity of ICOS makes any therapeutic applications difficult to predict, but its importance is clear.
ICOS and B7-H2 in cancer
Angioimmunoblastic T-cell lymphoma cells express ICOS, PD-1, and CXCR5, consistent with being the malignant counterpart of TFH lymphocytes.158 ICOS expression has also been described as a potential survival mechanism in NPM-ALK fusion anaplastic large cell lymphoma cells.159 Although induced ICOS-L expression enhances cytotoxic T cell–mediated tumor rejection,160–162 there is sparse and conflicting evidence for its expression in solid tumors,163,164 while expression in myeloma165,166 and acute leukemia is widely heterogeneous.71,166 Despite these sometimes contradictory findings, the ICOS/ICOS-L axis may yet have an indirect role in enhancement of tumor immunity. ICOS is inducible on NK cells, which demonstrate cytotoxicity against ICOS-L–transfected murine leukemia cell lines.167 Cytokine-induced killer cells (CIK) demonstrate MHC-independent cytotoxicity and antitumor activity,168,169 enhanced after ICOS transfection.170 Optimal immunostimulatory therapy using CTLA-4 mAbs may be dependent on an intact ICOS/ICOS-L pathway,171 with CTLA-4 blockade increasing tumor-specific IFN-γ–secreting T cells in cancer patients.172
Therapeutic modulation of ICOS/ICOS-L
ICOS is overexpressed during acute allogeneic solid organ rejection and tolerance is improved with pathway blockade,173 which suggests that this pathway may have importance in GVHD. Murine acute GVHD is reduced if donor Icos−/− mice or ICOS-blocking antibodies are used,174 with less gut and liver GVHD and improved survival175 despite intact Icos−/− donor cell cytotoxic function, cytokine-secretion152,153 and tissue trafficking,175 while Icos−/− recipient mice better tolerate allogeneic HSCT with more rapid and complete donor chimerism.174 However, a further study demonstrated more severe GVHD arising from allogeneic CD8+ Icos−/− cell transplantation compared with wild type,176 a contradiction perhaps explained by the differing mouse genetic backgrounds and variable use of Treg-depleting anti-CD25 mAbs. ICOS appears central to GVHD modulation, although because of these controversies, a therapeutic application has yet to emerge.
Controversial immunosuppressive pathways: B7-H3 and B7-H4
CD276 (B7-H3) was identified soon after ICOS,177 not detectable in resting blood cells but present in various normal tissues, in several tumor cell lines and induced on activated DCs and monocytes. B7-H3.Ig stimulates and increases IFN-γ secretion by activated T cells. Its ligand was eventually isolated from a CD28 family database homology search of a murine splenocyte cDNA library. Only TLT-2 showed specific binding,178 a member of the innate immune response-amplifying TREM (triggering receptor expressed on myeloid cells) molecular family TLT-2 is constitutively expressed on activated CD8+ T cells, inducible on CD4+ T cells and ligation augments T-cell activation. This murine molecule remains controversial in humans, with evidence for179 and against180 its role as CD276 ligand. As such, the function of CD276 also remains contentious, with stimulatory177,181,182 and inhibitory180,183 functions described, and the context of expression clearly central to its role. CD276 expression on solid tumors confers an adverse outcome.184,185 Our group demonstrated expression in CLL,74 down-regulated by lenalidomide, and where antibody-mediated blockade improved in vitro T-cell function.
A further B7/CD28-like molecule expressed on activated T cells, B cells, monocytes, and DCs has been described (B7-H4) which fails to bind CTLA-4.Ig, ICOS.Ig, or PD-1.Ig,186 possesses a binding partner expressed on activated T cells and whose Ig molecular construct, in contrast to that of all other B7-family molecules, inhibits, not stimulates, T-cell function. ‘B7S1’ was discovered by a second group, mAb blockade of which exacerbated murine EAE,187 while a third group identified ‘B7x,’ suggested that it was the same molecule188 and proposed a ligand, a molecule that had been identified during previous gene expression profiling of Th1 polarized cells: BTLA (B and T lymphocyte attenuator). BTLA-binding properties suggested immunosuppressive functions analogous to those mediated by CTLA-4 and PD-1, which was supported by the Btla−/− mouse, which demonstrates enhanced Th1 activity.189 However, subsequent investigations failed to confirm that BTLA is the ligand of B7-H4. A TNFRSF molecule, HVEM, seems to be the major BTLA-interacting immunosuppressive molecule.190
Although B7-H3 and B7-H4 are structurally related B7 family members, there is little consensus on function or binding partners. Crosstalk between other pathways, stimulatory as well as repressive functions, inducible as well as constitutive, tissue-dependent expression and 2-way signaling without clear receptor/ligand polarity are likely complexities existing across all molecules of this class and not only these more recently described members.
B7-H6: a tumor-specific family member
Investigation into natural killer cell (NK) effector mechanisms revealed the most recently identified member of the B7 family, B7-H6, a PD-L1/B7-H3 homologue specifically binding the CTLA-4-homologous NK-effector molecule NKp30.191 Membrane-bound B7-H6 activates NK cells, leading to degranulation and IFN-γ secretion. Unlike other B7 family members, B7-H6 is not expressed in any normal tissue even after activation, but is expressed in a variety of primary tumors and cell lines. The potential for therapeutic manipulation is clear, already inspiring incorporation of NKp30 into CAR constructs.192
An extended B7 definition? Butyrophilin and Skint
Other B7 homologues have been described with potential importance in cancer immune surveillance193,194 and immune modulation, including the diverse Butyrophilin195 family and the epidermal T cell–associated Skint (Selection and upKeep of INTraepithelial T cells) family.196 Butyrophilin subfamily 3 (BTN3) has a structural similarity to B7-H4197 and is expressed on immune cells198 as well as human ovarian cancer,197 while BTN3 agonists may induce immunosuppression.199 However, functional data are conflicting200; no ligand has been determined and no in vivo data generated. These molecules are intriguing but their role in cancer immunology remains speculative.
Conclusion
Our understanding of the B7 family has expanded enormously because the costimulatory pathways were first described. Contemporary models of immune regulatory networks reflect this complexity. The promise of immune-mediated enhancement of autologous and allogeneic responses through pharmacologic manipulation of this pathway shows substantial promise. Ongoing studies investigating the role of the most recently described members of the family: B7-H3, B7-H4, B7-H6, Butyrophilin, and Skint will likely reveal further potentially modifiable pathways as therapeutic targets for intervention. Whereas manipulation of the B7 family members and their ligands in suppressing GVHD and augmenting GVL after allogeneic HSCT has shown benefit in murine studies, and promising results in limited phase 2 clinical studies, this remains an area for clinical trial development. The contribution of ICOS/ICOS-L to GVHD appears to be fundamental; however, based on our current understanding of this pathway, the complexities of the interactions make predictions of clinical responses difficult to predict. After decades of study, strategies incorporating B7 family members, such as CTLA-4 and PD-1 pathway-inhibiting molecules and CAR-T cells, have finally demonstrated clear clinical benefit, fulfilling the early promise of the importance of this family in hematologic malignancies.
Acknowledgments
This work was supported in part by funding from P01 CA81538 from the National Cancer Institute to the CLL Research Consortium (J.G.G.) and by the Baker Foundation (P.G.).
Authorship
Contribution: P.G. and J.G.G. reviewed the literature and wrote the review.
Conflict-of-interest disclosure: J.G.G. has received honoraria from Celgene, Roche, Napp, Merck, and Pharmacyclics. J.G.G. receives grant funding from National Institutes of Health and Cancer Research UK and receives an honorarium from the American Society of Hematology as an associate editor of Blood. The remaining author declares no competing financial interests.
Correspondence: John G. Gribben, Barts Cancer Institute, Charterhouse Square, London EC1M 6BQ, United Kingdom; e-mail: j.gribben@qmul.ac.uk.
References
- 1.Hanahan D, Weinberg RA. Hallmarks of cancer: the next generation. Cell. 2011;144(5):646–674. doi: 10.1016/j.cell.2011.02.013. [DOI] [PubMed] [Google Scholar]
- 2.Schreiber RD, Old LJ, Smyth MJ. Cancer immunoediting: integrating immunity's roles in cancer suppression and promotion. Science. 2011;331(6024):1565–1570. doi: 10.1126/science.1203486. [DOI] [PubMed] [Google Scholar]
- 3.Van Pel A, Vessiere F, Boon T. Protection against two spontaneous mouse leukemias conferred by immunogenic variants obtained by mutagenesis. J Exp Med. 1983;157(6):1992–2001. doi: 10.1084/jem.157.6.1992. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 4.van der Bruggen P, Traversari C, Chomez P, et al. A gene encoding an antigen recognized by cytolytic T lymphocytes on a human melanoma. Science. 1991;254(5038):1643–1647. doi: 10.1126/science.1840703. [DOI] [PubMed] [Google Scholar]
- 5.Prehn RT, Main JM. Immunity to methylcholanthrene-induced sarcomas. J Natl Cancer Inst. 1957;18(6):769–778. [PubMed] [Google Scholar]
- 6.Condeelis J, Pollard JW. Macrophages: obligate partners for tumor cell migration, invasion, and metastasis. Cell. 2006;124(2):263–266. doi: 10.1016/j.cell.2006.01.007. [DOI] [PubMed] [Google Scholar]
- 7.Freedman AS, Freeman G, Horowitz JC, Daley J, Nadler LM. B7, a B-cell-restricted antigen that identifies preactivated B cells. J Immunol. 1987;139(10):3260–3267. [PubMed] [Google Scholar]
- 8.Freeman GJ, Freedman AS, Segil JM, Lee G, Whitman JF, Nadler LM. B7, a new member of the Ig superfamily with unique expression on activated and neoplastic B cells. J Immunol. 1989;143(8):2714–2722. [PubMed] [Google Scholar]
- 9.Linsley PS, Clark EA, Ledbetter JA. T-cell antigen CD28 mediates adhesion with B cells by interacting with activation antigen B7/BB-1. Proc Natl Acad Sci U S A. 1990;87(13):5031–5035. doi: 10.1073/pnas.87.13.5031. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 10.Linsley PS, Brady W, Grosmaire L, Aruffo A, Damle NK, Ledbetter JA. Binding of the B-cell activation antigen B7 to CD28 costimulates T-cell proliferation and interleukin 2 mRNA accumulation. J Exp Med. 1991;173(3):721–730. doi: 10.1084/jem.173.3.721. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 11.Koulova L, Clark EA, Shu G, Dupont B. The CD28 ligand B7/BB1 provides costimulatory signal for alloactivation of CD4+ T cells. J Exp Med. 1991;173(3):759–762. doi: 10.1084/jem.173.3.759. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 12.Gimmi CD, Freeman GJ, Gribben JG, et al. B-cell surface antigen B7 provides a costimulatory signal that induces T cells to proliferate and secrete interleukin 2. Proc Natl Acad Sci U S A. 1991;88(15):6575–6579. doi: 10.1073/pnas.88.15.6575. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 13.Brunet JF, Denizot F, Luciani MF, et al. A new member of the immunoglobulin superfamily CTLA-4. Nature. 1987;328(6127):267–270. doi: 10.1038/328267a0. [DOI] [PubMed] [Google Scholar]
- 14.Dariavach P, Mattei MG, Golstein P, Lefranc MP. Human Ig superfamily CTLA-4 gene: chromosomal localization and identity of protein sequence between murine and human CTLA-4 cytoplasmic domains. Eur J Immunol. 1988;18(12):1901–1905. doi: 10.1002/eji.1830181206. [DOI] [PubMed] [Google Scholar]
- 15.Linsley PS, Brady W, Urnes M, Grosmaire LS, Damle NK, Ledbetter JA. CTLA-4 is a second receptor for the B-cell activation antigen B7. J Exp Med. 1991;174(3):561–569. doi: 10.1084/jem.174.3.561. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 16.Linsley PS, Wallace PM, Johnson J, et al. Immunosuppression in vivo by a soluble form of the CTLA-4 T-cell activation molecule. Science. 1992;257(5071):792–795. doi: 10.1126/science.1496399. [DOI] [PubMed] [Google Scholar]
- 17.Lenschow DJ, Zeng Y, Thistlethwaite JR, et al. Long-term survival of xenogeneic pancreatic islet grafts induced by CTLA4lg. Science. 1992;257(5071):789–792. doi: 10.1126/science.1323143. [DOI] [PubMed] [Google Scholar]
- 18.Chen L, Ashe S, Brady WA, et al. Costimulation of antitumor immunity by the B7 counterreceptor for the T lymphocyte molecules CD28 and CTLA-4. Cell. 1992;71(7):1093–1102. doi: 10.1016/s0092-8674(05)80059-5. [DOI] [PubMed] [Google Scholar]
- 19.Linsley PS, Greene JL, Tan P, et al. Coexpression and functional cooperation of CTLA-4 and CD28 on activated T lymphocytes. J Exp Med. 1992;176(6):1595–1604. doi: 10.1084/jem.176.6.1595. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 20.Walunas TL, Lenschow DJ, Bakker CY, et al. CTLA-4 can function as a negative regulator of T-cell activation. Immunity. 1994;1(5):405–413. doi: 10.1016/1074-7613(94)90071-x. [DOI] [PubMed] [Google Scholar]
- 21.Gribben JG, Freeman GJ, Boussiotis VA, et al. CTLA4 mediates antigen-specific apoptosis of human T cells. Proc Natl Acad Sci U S A. 1995;92(3):811–815. doi: 10.1073/pnas.92.3.811. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 22.Tivol EA, Borriello F, Schweitzer AN, Lynch WP, Bluestone JA, Sharpe AH. Loss of CTLA-4 leads to massive lymphoproliferation and fatal multiorgan tissue destruction, revealing a critical negative regulatory role of CTLA-4. Immunity. 1995;3(5):541–547. doi: 10.1016/1074-7613(95)90125-6. [DOI] [PubMed] [Google Scholar]
- 23.Waterhouse P, Penninger JM, Timms E, et al. Lymphoproliferative disorders with early lethality in mice deficient in Ctla-4. Science. 1995;270(5238):985–988. doi: 10.1126/science.270.5238.985. [DOI] [PubMed] [Google Scholar]
- 24.Rudd CE, Schneider H. Unifying concepts in CD28, ICOS and CTLA4 co-receptor signalling. Nat Rev Immunol. 2003;3(7):544–556. doi: 10.1038/nri1131. [DOI] [PubMed] [Google Scholar]
- 25.Nickoloff BJ, Mitra RS, Lee K, et al. Discordant expression of CD28 ligands, BB-1, and B7 on keratinocytes in vitro and psoriatic cells in vivo. Am J Pathol. 1993;142(4):1029–1040. [PMC free article] [PubMed] [Google Scholar]
- 26.Azuma M, Ito D, Yagita H, et al. B70 antigen is a second ligand for CTLA-4 and CD28. Nature. 1993;366(6450):76–79. doi: 10.1038/366076a0. [DOI] [PubMed] [Google Scholar]
- 27.Freeman GJ, Gribben JG, Boussiotis VA, et al. Cloning of B7-2: a CTLA-4 counter-receptor that costimulates human T-cell proliferation. Science. 1993;262(5135):909–911. doi: 10.1126/science.7694363. [DOI] [PubMed] [Google Scholar]
- 28.Engel P, Gribben JG, Freeman GJ, et al. The B7-2 (B70) costimulatory molecule expressed by monocytes and activated B lymphocytes is the CD86 differentiation antigen. Blood. 1994;84(5):1402–1407. [PubMed] [Google Scholar]
- 29.Dorfman DM, Schultze JL, Shahsafaei A, et al. In vivo expression of B7-1 and B7-2 by follicular lymphoma cells can prevent induction of T-cell anergy but is insufficient to induce significant T-cell proliferation. Blood. 1997;90(11):4297–4306. [PubMed] [Google Scholar]
- 30.Vyth-Dreese FA, Boot H, Dellemijn TA, et al. Localization in situ of costimulatory molecules and cytokines in B-cell non-Hodgkin's lymphoma. Immunology. 1998;94(4):580–586. doi: 10.1046/j.1365-2567.1998.00550.x. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 31.Chaperot L, Plumas J, Jacob MC, et al. Functional expression of CD80 and CD86 allows immunogenicity of malignant B cells from non-Hodgkin's lymphomas. Exp Hematol. 1999;27(3):479–488. doi: 10.1016/s0301-472x(98)00059-9. [DOI] [PubMed] [Google Scholar]
- 32.Schultze JL, Cardoso AA, Freeman GJ, et al. Follicular lymphomas can be induced to present alloantigen efficiently: a conceptual model to improve their tumor immunogenicity. Proc Natl Acad Sci U S A. 1995;92(18):8200–8204. doi: 10.1073/pnas.92.18.8200. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 33.Delabie J, Ceuppens JL, Vandenberghe P, de Boer M, Coorevits L, De Wolf-Peeters C. The B7/BB1 antigen is expressed by Reed-Sternberg cells of Hodgkin's disease and contributes to the stimulating capacity of Hodgkin's disease-derived cell lines. Blood. 1993;82(9):2845–2852. [PubMed] [Google Scholar]
- 34.Murray PG, Oates J, Reynolds GM, Crocker J, Young LS. Expression of B7 (CD80) and CD40 antigens and the CD40 ligand in Hodgkin's disease is independent of latent Epstein-Barr virus infection. Clin Mol Pathol. 1995;48(2):M105–M108. doi: 10.1136/mp.48.2.m105. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 35.Pope B, Brown RD, Gibson J, Yuen E, Joshua D. B7-2-positive myeloma: incidence, clinical characteristics, prognostic significance, and implications for tumor immunotherapy. Blood. 2000;96(4):1274–1279. [PubMed] [Google Scholar]
- 36.Ranheim EA, Kipps TJ. Activated T cells induce expression of B7/BB1 on normal or leukemic B cells through a CD40-dependent signal. J Exp Med. 1993;177(4):925–935. doi: 10.1084/jem.177.4.925. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 37.Hirano N, Takahashi T, Takahashi T, et al. Expression of costimulatory molecules in human leukemias. Leukemia. 1996;10(7):1168–1176. [PubMed] [Google Scholar]
- 38.Costello RT, Mallet F, Sainty D, Maraninchi D, Gastaut JA, Olive D. Regulation of CD80/B7-1 and CD86/B7-2 molecule expression in human primary acute myeloid leukemia and their role in allogenic immune recognition. Eur J Immunol. 1998;28(1):90–103. doi: 10.1002/(SICI)1521-4141(199801)28:01<90::AID-IMMU90>3.0.CO;2-5. [DOI] [PubMed] [Google Scholar]
- 39.Munro JM, Freedman AS, Aster JC, et al. In vivo expression of the B7 costimulatory molecule by subsets of antigen-presenting cells and the malignant cells of Hodgkin's disease. Blood. 1994;83(3):793–798. [PubMed] [Google Scholar]
- 40.Dakappagari N, Ho SN, Gascoyne RD, Ranuio J, Weng AP, Tangri S. CD80 (B7.1) is expressed on both malignant B cells and nonmalignant stromal cells in non-Hodgkin lymphoma. Cytometry B Clin Cytom. 2012;82(2):112–119. doi: 10.1002/cyto.b.20631. [DOI] [PubMed] [Google Scholar]
- 41.Younes A, Hariharan K, Allen RS, Leigh BR. Initial trials of anti-CD80 monoclonal antibody (Galiximab) therapy for patients with relapsed or refractory follicular lymphoma. Clin Lymphoma. 2003;3(4):257–259. doi: 10.3816/clm.2003.n.008. [DOI] [PubMed] [Google Scholar]
- 42.Czuczman MS, Thall A, Witzig TE, et al. Phase I/II study of galiximab, an anti-CD80 antibody, for relapsed or refractory follicular lymphoma. J Clin Oncol. 2005;23(19):4390–4398. doi: 10.1200/JCO.2005.09.018. [DOI] [PubMed] [Google Scholar]
- 43.Leonard JP, Friedberg JW, Younes A, et al. A phase I/II study of galiximab (an anti-CD80 monoclonal antibody) in combination with rituximab for relapsed or refractory, follicular lymphoma. Ann Oncol. 2007;18(7):1216–1223. doi: 10.1093/annonc/mdm114. [DOI] [PubMed] [Google Scholar]
- 44.Smith SM, Bartlett N, Johnson JL, et al. Galiximab, an anti-CD80 primatized monoclonal antibody, in relapsed Hodgkin lymphoma: final results of CALGB 50602. J Clin Oncol. 2010;28(15s):8039. [Google Scholar]
- 45.Suntharalingam G, Perry MR, Ward S, et al. Cytokine storm in a phase 1 trial of the anti-CD28 monoclonal antibody TGN1412. N Engl J Med. 2006;355(10):1018–1028. doi: 10.1056/NEJMoa063842. [DOI] [PubMed] [Google Scholar]
- 46.Phan GQ, Yang JC, Sherry RM, et al. Cancer regression and autoimmunity induced by cytotoxic T lymphocyte-associated antigen 4 blockade in patients with metastatic melanoma. Proc Natl Acad Sci U S A. 2003;100(14):8372–8377. doi: 10.1073/pnas.1533209100. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 47.Hodi FS, O'Day SJ, McDermott DF, et al. Improved survival with ipilimumab in patients with metastatic melanoma. N Engl J Med. 2010;363(8):711–723. doi: 10.1056/NEJMoa1003466. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 48.Ansell SM, Hurvitz SA, Koenig PA, et al. Phase I study of ipilimumab, an anti-CTLA-4 monoclonal antibody, in patients with relapsed and refractory B-cell non-Hodgkin lymphoma. Clin Cancer Res. 2009;15(20):6446–6453. doi: 10.1158/1078-0432.CCR-09-1339. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 49.Dong H, Zhu G, Tamada K, Chen L. B7-H1, a third member of the B7 family, co-stimulates T-cell proliferation and interleukin-10 secretion. Nat Med. 1999;5(12):1365–1369. doi: 10.1038/70932. [DOI] [PubMed] [Google Scholar]
- 50.Tseng SY, Otsuji M, Gorski K, et al. B7-DC, a new dendritic cell molecule with potent costimulatory properties for T cells. J Exp Med. 2001;193(7):839–846. doi: 10.1084/jem.193.7.839. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 51.Ishida Y, Agata Y, Shibahara K, Honjo T. Induced expression of PD-1, a novel member of the immunoglobulin gene superfamily, upon programmed cell death. EMBO J. 1992;11(11):3887–3895. doi: 10.1002/j.1460-2075.1992.tb05481.x. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 52.Shinohara T, Taniwaki M, Ishida Y, Kawaichi M, Honjo T. Structure and chromosomal localization of the human PD-1 gene (PDCD1). Genomics. 1994;23(3):704–706. doi: 10.1006/geno.1994.1562. [DOI] [PubMed] [Google Scholar]
- 53.Nishimura H, Agata Y, Kawasaki A, et al. Developmentally regulated expression of the PD-1 protein on the surface of double-negative (CD4-CD8-) thymocytes. Int Immunol. 1996;8(5):773–780. doi: 10.1093/intimm/8.5.773. [DOI] [PubMed] [Google Scholar]
- 54.Nishimura H, Nose M, Hiai H, Minato N, Honjo T. Development of lupus-like autoimmune diseases by disruption of the PD-1 gene encoding an ITIM motif-carrying immunoreceptor. Immunity. 1999;11(2):141–151. doi: 10.1016/s1074-7613(00)80089-8. [DOI] [PubMed] [Google Scholar]
- 55.Harper K, Balzano C, Rouvier E, Mattei MG, Luciani MF, Golstein P. CTLA-4 and CD28 activated lymphocyte molecules are closely related in both mouse and human as to sequence, message expression, gene structure, and chromosomal location. J Immunol. 1991;147(3):1037–1044. [PubMed] [Google Scholar]
- 56.Freeman GJ, Long AJ, Iwai Y, et al. Engagement of the PD-1 immunoinhibitory receptor by a novel B7 family member leads to negative regulation of lymphocyte activation. J Exp Med. 2000;192(7):1027–1034. doi: 10.1084/jem.192.7.1027. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 57.Latchman Y, Wood CR, Chernova T, et al. PD-L2 is a second ligand for PD-1 and inhibits T-cell activation. Nat Immunol. 2001;2(3):261–268. doi: 10.1038/85330. [DOI] [PubMed] [Google Scholar]
- 58.Good-Jacobson KL, Szumilas CG, Chen L, Sharpe AH, Tomayko MM, Shlomchik MJ. PD-1 regulates germinal center B-cell survival and the formation and affinity of long-lived plasma cells. Nat Immunol. 2010;11(6):535–542. doi: 10.1038/ni.1877. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 59.Butte MJ, Keir ME, Phamduy TB, Sharpe AH, Freeman GJ. Programmed death-1 ligand 1 interacts specifically with the B7-1 costimulatory molecule to inhibit T-cell responses. Immunity. 2007;27(1):111–122. doi: 10.1016/j.immuni.2007.05.016. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 60.Iwai Y, Ishida M, Tanaka Y, Okazaki T, Honjo T, Minato N. Involvement of PD-L1 on tumor cells in the escape from host immune system and tumor immunotherapy by PD-L1 blockade. Proc Natl Acad Sci U S A. 2002;99(19):12293–12297. doi: 10.1073/pnas.192461099. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 61.Dong H, Strome SE, Salomao DR, et al. Tumor-associated B7-H1 promotes T-cell apoptosis: a potential mechanism of immune evasion. Nat Med. 2002;8(8):793–800. doi: 10.1038/nm730. [DOI] [PubMed] [Google Scholar]
- 62.Nomi T, Sho M, Akahori T, et al. Clinical significance and therapeutic potential of the programmed death-1 ligand/programmed death-1 pathway in human pancreatic cancer. Clin Cancer Res. 2007;13(7):2151–2157. doi: 10.1158/1078-0432.CCR-06-2746. [DOI] [PubMed] [Google Scholar]
- 63.Blank C, Brown I, Peterson AC, et al. PD-L1/B7H-1 inhibits the effector phase of tumor rejection by T-cell receptor (TCR) transgenic CD8+ T cells. Cancer Res. 2004;64(3):1140–1145. doi: 10.1158/0008-5472.can-03-3259. [DOI] [PubMed] [Google Scholar]
- 64.Ohigashi Y, Sho M, Yamada Y, et al. Clinical significance of programmed death-1 ligand-1 and programmed death-1 ligand-2 expression in human esophageal cancer. Clin Cancer Res. 2005;11(8):2947–2953. doi: 10.1158/1078-0432.CCR-04-1469. [DOI] [PubMed] [Google Scholar]
- 65.Geng L, Huang D, Liu J, et al. B7-H1 up-regulated expression in human pancreatic carcinoma tissue associates with tumor progression. J Cancer Res Clin Oncol. 2008;134(9):1021–1027. doi: 10.1007/s00432-008-0364-8. [DOI] [PubMed] [Google Scholar]
- 66.Hino R, Kabashima K, Kato Y, et al. Tumor cell expression of programmed cell death-1 ligand 1 is a prognostic factor for malignant melanoma. Cancer. 2010;116(7):1757–1766. doi: 10.1002/cncr.24899. [DOI] [PubMed] [Google Scholar]
- 67.Hamanishi J, Mandai M, Iwasaki M, et al. Programmed cell death 1 ligand 1 and tumor-infiltrating CD8+ T lymphocytes are prognostic factors of human ovarian cancer. Proc Natl Acad Sci U S A. 2007;104(9):3360–3365. doi: 10.1073/pnas.0611533104. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 68.Thompson RH, Dong H, Lohse CM, et al. PD-1 is expressed by tumor-infiltrating immune cells and is associated with poor outcome for patients with renal cell carcinoma. Clin Cancer Res. 2007;13(6):1757–1761. doi: 10.1158/1078-0432.CCR-06-2599. [DOI] [PubMed] [Google Scholar]
- 69.Saudemont A, Buffenoir G, Denys A, et al. Gene transfer of CD154 and IL12 cDNA induces an anti-leukemic immunity in a murine model of acute leukemia. Leukemia. 2002;16(9):1637–1644. doi: 10.1038/sj.leu.2402590. [DOI] [PubMed] [Google Scholar]
- 70.Saudemont A, Quesnel B. In a model of tumor dormancy, long-term persistent leukemic cells have increased B7-H1 and B7.1 expression and resist CTL-mediated lysis. Blood. 2004;104(7):2124–2133. doi: 10.1182/blood-2004-01-0064. [DOI] [PubMed] [Google Scholar]
- 71.Tamura H, Dan K, Tamada K, et al. Expression of functional B7-H2 and B7.2 costimulatory molecules and their prognostic implications in de novo acute myeloid leukemia. Clin Cancer Res. 2005;11(16):5708–5717. doi: 10.1158/1078-0432.CCR-04-2672. [DOI] [PubMed] [Google Scholar]
- 72.Salih HR, Wintterle S, Krusch M, et al. The role of leukemia-derived B7-H1 (PD-L1) in tumor-T-cell interactions in humans. Exp Hematol. 2006;34(7):888–894. doi: 10.1016/j.exphem.2006.03.006. [DOI] [PubMed] [Google Scholar]
- 73.Berthon C, Driss V, Liu J, et al. In acute myeloid leukemia, B7-H1 (PD-L1) protection of blasts from cytotoxic T cells is induced by TLR ligands and interferon-gamma and can be reversed using MEK inhibitors. Cancer Immunol Immunother. 2010;59(12):1839–1849. doi: 10.1007/s00262-010-0909-y. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 74.Ramsay AG, Clear AJ, Fatah R, Gribben JG. Multiple inhibitory ligands induce impaired T-cell immunologic synapse function in chronic lymphocytic leukemia that can be blocked with lenalidomide: establishing a reversible immune evasion mechanism in human cancer. Blood. 2012;120(7):1412–1421. doi: 10.1182/blood-2012-02-411678. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 75.Aue G, Njuguna N, Tian X, et al. Lenalidomide-induced upregulation of CD80 on tumor cells correlates with T-cell activation, the rapid onset of a cytokine release syndrome and leukemic cell clearance in chronic lymphocytic leukemia. Haematologica. 2009;94(9):1266–1273. doi: 10.3324/haematol.2009.005835. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 76.Kozako T, Yoshimitsu M, Fujiwara H, et al. PD-1/PD-L1 expression in human T-cell leukemia virus type 1 carriers and adult T-cell leukemia/lymphoma patients. Leukemia. 2009;23(2):375–382. doi: 10.1038/leu.2008.272. [DOI] [PubMed] [Google Scholar]
- 77.Dorfman DM, Brown JA, Shahsafaei A, Freeman GJ. Programmed death-1 (PD-1) is a marker of germinal center-associated T cells and angioimmunoblastic T-cell lymphoma. Am J Surg Pathol. 2006;30(7):802–810. doi: 10.1097/01.pas.0000209855.28282.ce. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 78.Xerri L, Chetaille B, Serriari N, et al. Programmed death 1 is a marker of angioimmunoblastic T-cell lymphoma and B-cell small lymphocytic lymphoma/chronic lymphocytic leukemia. Hum Pathol. 2008;39(7):1050–1058. doi: 10.1016/j.humpath.2007.11.012. [DOI] [PubMed] [Google Scholar]
- 79.Grzywnowicz M, Zaleska J, Mertens D, et al. Programmed death-1 and its ligand are novel immunotolerant molecules expressed on leukemic B cells in chronic lymphocytic leukemia. PLoS One. 2012;7(4):e35178. doi: 10.1371/journal.pone.0035178. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 80.Carreras J, Lopez-Guillermo A, Roncador G, et al. High numbers of tumor-infiltrating programmed cell death 1-positive regulatory lymphocytes are associated with improved overall survival in follicular lymphoma. J Clin Oncol. 2009;27(9):1470–1476. doi: 10.1200/JCO.2008.18.0513. [DOI] [PubMed] [Google Scholar]
- 81.Greaves P, Clear AJ, Owen A, et al. The PD1/PD-L1 axis in the classical hodgkin lymphoma microenvironment: PD-1 is rarely expressed but identifies patients with high-risk disease [abstract]. Blood (ASH Annual Meeting Abstracts) 2011;118(21) Abstract 1560. [Google Scholar]
- 82.Yamamoto R, Nishikori M, Kitawaki T, et al. PD-1-PD-1 ligand interaction contributes to immunosuppressive microenvironment of Hodgkin lymphoma. Blood. 2008;111(6):3220–3224. doi: 10.1182/blood-2007-05-085159. [DOI] [PubMed] [Google Scholar]
- 83.Muenst S, Hoeller S, Dirnhofer S, Tzankov A. Increased programmed death-1+ tumor-infiltrating lymphocytes in classical Hodgkin lymphoma substantiate reduced overall survival. Hum Pathol. 2009;40(12):1715–1722. doi: 10.1016/j.humpath.2009.03.025. [DOI] [PubMed] [Google Scholar]
- 84.Rosenwald A, Wright G, Leroy K, et al. Molecular diagnosis of primary mediastinal B-cell lymphoma identifies a clinically favorable subgroup of diffuse large B-cell lymphoma related to Hodgkin lymphoma. J Exp Med. 2003;198(6):851–862. doi: 10.1084/jem.20031074. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 85.Andorsky DJ, Yamada RE, Said J, Pinkus GS, Betting DJ, Timmerman JM. Programmed death ligand 1 is expressed by non-Hodgkin lymphomas and inhibits the activity of tumor-associated T cells. Clin Cancer Res. 2011;17(13):4232–4244. doi: 10.1158/1078-0432.CCR-10-2660. [DOI] [PubMed] [Google Scholar]
- 86.Green MR, Monti S, Rodig SJ, et al. Integrative analysis reveals selective 9p24.1 amplification, increased PD-1 ligand expression, and further induction via JAK2 in nodular sclerosing Hodgkin lymphoma and primary mediastinal large B-cell lymphoma. Blood. 2010;116(17):3268–3277. doi: 10.1182/blood-2010-05-282780. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 87.Steidl C, Telenius A, Shah SP, et al. Genome-wide copy number analysis of Hodgkin Reed-Sternberg cells identifies recurrent imbalances with correlations to treatment outcome. Blood. 2010;116(3):418–427. doi: 10.1182/blood-2009-12-257345. [DOI] [PubMed] [Google Scholar]
- 88.Steidl C, Shah SP, Woolcock BW, et al. MHC class II transactivator CIITA is a recurrent gene fusion partner in lymphoid cancers. Nature. 2011;471(7338):377–381. doi: 10.1038/nature09754. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 89.Liu J, Hamrouni A, Wolowiec D, et al. Plasma cells from multiple myeloma patients express B7-H1 (PD-L1) and increase expression after stimulation with IFN-{gamma} and TLR ligands via a MyD88-, TRAF6-, and MEK-dependent pathway. Blood. 2007;110(1):296–304. doi: 10.1182/blood-2006-10-051482. [DOI] [PubMed] [Google Scholar]
- 90.Hallett WHD, Jing W, Drobyski WR, Johnson BD. Immunosuppressive effects of multiple myeloma are overcome by PD-L1 blockade. Biol Blood Marrow Transplant. 2011;17(8):1133–1145. doi: 10.1016/j.bbmt.2011.03.011. [DOI] [PubMed] [Google Scholar]
- 91.Benson DM, Jr, Bakan CE, Mishra A, et al. The PD-1/PD-L1 axis modulates the natural killer cell versus multiple myeloma effect: a therapeutic target for CT-011, a novel monoclonal anti-PD-1 antibody. Blood. 2010;116(13):2286–2294. doi: 10.1182/blood-2010-02-271874. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 92.Brahmer JR, Tykodi SS, Chow LQ, et al. Safety and activity of anti-PD-L1 antibody in patients with advanced cancer. N Engl J Med. 2012;366(26):2455–2465. doi: 10.1056/NEJMoa1200694. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 93.Topalian SL, Hodi FS, Brahmer JR, et al. Safety, activity, and immune correlates of anti-PD-1 antibody in cancer. N Engl J Med. 2012;366(26):2443–2454. doi: 10.1056/NEJMoa1200690. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 94.Patnaik A, Kang SP, Tolcher AW, et al. Phase I study of MK-3475 (anti-PD-1 monoclonal antibody) in patients with advanced solid tumors. J Clin Oncol. 2012;30(Suppl) doi: 10.1158/1078-0432.CCR-14-2607. Abstract 2512. [DOI] [PubMed] [Google Scholar]
- 95.Berger R, Rotem-Yehudar R, Slama G, et al. Phase I safety and pharmacokinetic study of CT-011, a humanized antibody interacting with PD-1, in patients with advanced hematologic malignancies. Clin Cancer Res. 2008;14(10):3044–3051. doi: 10.1158/1078-0432.CCR-07-4079. [DOI] [PubMed] [Google Scholar]
- 96.Bashey A, Medina B, Corringham S, et al. CTLA4 blockade with ipilimumab to treat relapse of malignancy after allogeneic hematopoietic cell transplantation. Blood. 2009;113(7):1581–1588. doi: 10.1182/blood-2008-07-168468. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 97.Blazar BR, Taylor PA, Panoskaltsis-Mortari A, Sharpe AH, Vallera DA. Opposing roles of CD28:B7 and CTLA-4:B7 pathways in regulating in vivo alloresponses in murine recipients of MHC disparate T cells. J Immunol. 1999;162(11):6368–6377. [PubMed] [Google Scholar]
- 98.Koestner W, Hapke M, Herbst J, et al. PD-L1 blockade effectively restores strong graft-versus-leukemia effects without graft-versus-host disease after delayed adoptive transfer of T-cell receptor gene-engineered allogeneic CD8+ T cells. Blood. 2011;117(3):1030–1041. doi: 10.1182/blood-2010-04-283119. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 99.Blazar BR, Carreno BM, Panoskaltsis-Mortari A, et al. Blockade of programmed death-1 engagement accelerates graft-versus-host disease lethality by an IFN-gamma-dependent mechanism. J Immunol. 2003;171(3):1272–1277. doi: 10.4049/jimmunol.171.3.1272. [DOI] [PubMed] [Google Scholar]
- 100.Dranoff G, Jaffee E, Lazenby A, et al. Vaccination with irradiated tumor cells engineered to secrete murine granulocyte-macrophage colony-stimulating factor stimulates potent, specific, and long-lasting anti-tumor immunity. Proc Natl Acad Sci U S A. 1993;90(8):3539–3543. doi: 10.1073/pnas.90.8.3539. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 101.Inaba K, Metlay JP, Crowley MT, Steinman RM. Dendritic cells pulsed with protein antigens in vitro can prime antigen-specific, MHC-restricted T cells in situ. J Exp Med. 1990;172(2):631–640. doi: 10.1084/jem.172.2.631. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 102.Hsu FJ, Benike C, Fagnoni F, et al. Vaccination of patients with B-cell lymphoma using autologous antigen-pulsed dendritic cells. Nat Med. 1996;2(1):52–58. doi: 10.1038/nm0196-52. [DOI] [PubMed] [Google Scholar]
- 103.Osterborg A, Yi Q, Henriksson L, et al. Idiotype immunization combined with granulocyte-macrophage colony-stimulating factor in myeloma patients induced type I, major histocompatibility complex-restricted, CD8- and CD4-specific T-cell responses. Blood. 1998;91(7):2459–2466. [PubMed] [Google Scholar]
- 104.Bertinetti C, Simon F, Zirlik K, et al. Cloning of idiotype immunoglobulin genes in B-cell lymphomas by anchored PCR and production of individual recombinant idiotype vaccines in Escherichia coli. Eur J Haematol. 2006;77(5):395–402. doi: 10.1111/j.1600-0609.2006.00740.x. [DOI] [PubMed] [Google Scholar]
- 105.Navarrete MA, Heining-Mikesch K, Schuler F, et al. Upfront immunization with autologous recombinant idiotype Fab fragment without prior cytoreduction in indolent B-cell lymphoma. Blood. 2011;117(5):1483–1491. doi: 10.1182/blood-2010-06-292342. [DOI] [PubMed] [Google Scholar]
- 106.Krackhardt AM, Witzens M, Harig S, et al. Identification of tumor-associated antigens in chronic lymphocytic leukemia by SEREX. Blood. 2002;100(6):2123–2131. doi: 10.1182/blood-2002-02-0513. [DOI] [PubMed] [Google Scholar]
- 107.Fukuda T, Chen L, Endo T, et al. Antisera induced by infusions of autologous Ad-CD154-leukemia B cells identify ROR1 as an oncofetal antigen and receptor for Wnt5a. Proc Natl Acad Sci U S A. 2008;105(8):3047–3052. doi: 10.1073/pnas.0712148105. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 108.Oka Y, Tsuboi A, Taguchi T, et al. Induction of WT1 (WilmsÆ tumor gene)-specific cytotoxic T lymphocytes by WT1 peptide vaccine and the resultant cancer regression. Proc Natl Acad Sci U S A. 2004;101(38):13885–13890. doi: 10.1073/pnas.0405884101. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 109.Klebanoff CA, Acquavella N, Yu Z, Restifo NP. Therapeutic cancer vaccines: are we there yet? Immunol Rev. 2011;239(1):27–44. doi: 10.1111/j.1600-065X.2010.00979.x. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 110.Ribas A, Comin-Anduix B, Chmielowski B, et al. Dendritic cell vaccination combined with CTLA4 blockade in patients with metastatic melanoma. Clin Cancer Res. 2009;15(19):6267–6276. doi: 10.1158/1078-0432.CCR-09-1254. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 111.O'Mahony D, Morris JC, Quinn C, et al. A pilot study of CTLA-4 blockade after cancer vaccine failure in patients with advanced malignancy. Clin Cancer Res. 2007;13(3):958–964. doi: 10.1158/1078-0432.CCR-06-1974. [DOI] [PubMed] [Google Scholar]
- 112.Hobo W, Maas F, Adisty N, et al. siRNA silencing of PD-L1 and PD-L2 on dendritic cells augments expansion and function of minor histocompatibility antigen-specific CD8+ T cells. Blood. 2010;116(22):4501–4511. doi: 10.1182/blood-2010-04-278739. [DOI] [PubMed] [Google Scholar]
- 113.Rosenblatt J, Glotzbecker B, Mills H, et al. PD-1 blockade by CT-011, anti-PD-1 antibody, enhances ex vivo T-cell responses to autologous dendritic cell/myeloma fusion vaccine. J Immunother. 2011;34(5):409–418. doi: 10.1097/CJI.0b013e31821ca6ce. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 114.Rosenberg SA, Yannelli JR, Yang JC, et al. Treatment of patients with metastatic melanoma with autologous tumor-infiltrating lymphocytes and interleukin 2. J Natl Cancer Inst. 1994;86(15):1159–1166. doi: 10.1093/jnci/86.15.1159. [DOI] [PubMed] [Google Scholar]
- 115.Dudley ME, Wunderlich JR, Robbins PF, et al. Cancer regression and autoimmunity in patients after clonal repopulation with antitumor lymphocytes. Science. 2002;298(5594):850–854. doi: 10.1126/science.1076514. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 116.Rosenberg SA, Yang JC, Sherry RM, et al. Durable complete responses in heavily pretreated patients with metastatic melanoma using T-cell transfer immunotherapy. Clin Cancer Res. 2011;17(13):4550–4557. doi: 10.1158/1078-0432.CCR-11-0116. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 117.Rooney CM, Smith CA, Ng CY, et al. Use of gene-modified virus-specific T lymphocytes to control Epstein-Barr-virus-related lymphoproliferation. Lancet. 1995;345(8941):9–13. doi: 10.1016/s0140-6736(95)91150-2. [DOI] [PubMed] [Google Scholar]
- 118.Kawakami Y, Eliyahu S, Delgado CH, et al. Cloning of the gene coding for a shared human melanoma antigen recognized by autologous T cells infiltrating into tumor. Proc Natl Acad Sci U S A. 1994;91(9):3515–3519. doi: 10.1073/pnas.91.9.3515. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 119.Clay TM, Custer MC, Sachs J, Hwu P, Rosenberg SA, Nishimura MI. Efficient transfer of a tumor antigen-reactive TCR to human peripheral blood lymphocytes confers anti-tumor reactivity. J Immunol. 1999;163(1):507–513. [PubMed] [Google Scholar]
- 120.Stanislawski T, Voss RH, Lotz C, et al. Circumventing tolerance to a human MDM2-derived tumor antigen by TCR gene transfer. Nat Immunol. 2001;2(10):962–970. doi: 10.1038/ni1001-962. [DOI] [PubMed] [Google Scholar]
- 121.Morgan RA, Dudley ME, Wunderlich JR, et al. Cancer regression in patients after transfer of genetically engineered lymphocytes. Science. 2006;314(5796):126–129. doi: 10.1126/science.1129003. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 122.Johnson LA, Morgan RA, Dudley ME, et al. Gene therapy with human and mouse T-cell receptors mediates cancer regression and targets normal tissues expressing cognate antigen. Blood. 2009;114(3):535–546. doi: 10.1182/blood-2009-03-211714. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 123.Maher J, Brentjens RJ, Gunset G, Riviere I, Sadelain M. Human T-lymphocyte cytotoxicity and proliferation directed by a single chimeric TCRzeta /CD28 receptor. Nat Biotech. 2002;20(1):70–75. doi: 10.1038/nbt0102-70. [DOI] [PubMed] [Google Scholar]
- 124.Stephan MT, Ponomarev V, Brentjens RJ, et al. T-cell-encoded CD80 and 4-1BBL induce auto- and transcostimulation, resulting in potent tumor rejection. Nat Med. 2007;13(12):1440–1449. doi: 10.1038/nm1676. [DOI] [PubMed] [Google Scholar]
- 125.Kochenderfer JN, Dudley ME, Feldman SA, et al. B-cell depletion and remissions of malignancy along with cytokine-associated toxicity in a clinical trial of anti-CD19 chimeric-antigen-receptor-transduced T cells. Blood. 2012;119(12):2709–2720. doi: 10.1182/blood-2011-10-384388. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 126.Porter DL, Levine BL, Kalos M, Bagg A, June CH. Chimeric antigen receptor-modified T cells in chronic lymphoid leukemia. N Engl J Med. 2011;365(8):725–733. doi: 10.1056/NEJMoa1103849. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 127.Gribben JG, Guinan EC, Boussiotis VA, et al. Complete blockade of B7 family-mediated costimulation is necessary to induce human alloantigen-specific anergy: a method to ameliorate graft-versus-host disease and extend the donor pool. Blood. 1996;87(11):4887–4893. [PubMed] [Google Scholar]
- 128.Guinan EC, Boussiotis VA, Neuberg D, et al. Transplantation of anergic histoincompatible bone marrow allografts. N Engl J Med. 1999;340(22):1704–1714. doi: 10.1056/NEJM199906033402202. [DOI] [PubMed] [Google Scholar]
- 129.Davies JK, Gribben JG, Brennan LL, Yuk D, Nadler LM, Guinan EC. Outcome of alloanergized haploidentical bone marrow transplantation after ex vivo costimulatory blockade: results of 2 phase 1 studies. Blood. 2008;112(6):2232–2241. doi: 10.1182/blood-2008-03-143636. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 130.Curiel TJ, Coukos G, Zou L, et al. Specific recruitment of regulatory T cells in ovarian carcinoma fosters immune privilege and predicts reduced survival. Nat Med. 2004;10(9):942–949. doi: 10.1038/nm1093. [DOI] [PubMed] [Google Scholar]
- 131.Woo EY, Chu CS, Goletz TJ, et al. Regulatory CD4(+)CD25(+) T cells in tumors from patients with early-stage non-small cell lung cancer and late-stage ovarian cancer. Cancer Res. 2001;61(12):4766–4772. [PubMed] [Google Scholar]
- 132.Woo EY, Yeh H, Chu CS, et al. Cutting edge: Regulatory T cells from lung cancer patients directly inhibit autologous T-cell proliferation. J Immunol. 2002;168(9):4272–4276. doi: 10.4049/jimmunol.168.9.4272. [DOI] [PubMed] [Google Scholar]
- 133.Lee AM, Clear AJ, Calaminici M, et al. Number of CD4+ cells and location of forkhead box protein P3-positive cells in diagnostic follicular lymphoma tissue microarrays correlates with outcome. J Clin Oncol. 2006;24(31):5052–5059. doi: 10.1200/JCO.2006.06.4642. [DOI] [PubMed] [Google Scholar]
- 134.Tzankov A, Meier C, Hirschmann P, Went P, Pileri SA, Dirnhofer S. Correlation of high numbers of intratumoral FOXP3+ regulatory T cells with improved survival in germinal center-like diffuse large B-cell lymphoma, follicular lymphoma and classical Hodgkin's lymphoma. Haematologica. 2008;93(2):193–200. doi: 10.3324/haematol.11702. [DOI] [PubMed] [Google Scholar]
- 135.Greaves P, Clear A, Coutinho R, et al. Expression of FOXP3, CD68, and CD20 at diagnosis in the microenvironment of classical Hodgkin lymphoma is predictive of outcome. J Clin Oncol. 2013;31(2):256–262. doi: 10.1200/JCO.2011.39.9881. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 136.Read S, Malmstrom V, Powrie F. Cytotoxic T lymphocyte-associated antigen 4 plays an essential role in the function of CD25(+)CD4(+) regulatory cells that control intestinal inflammation. J Exp Med. 2000;192(2):295–302. doi: 10.1084/jem.192.2.295. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 137.Takahashi T, Tagami T, Yamazaki S, et al. Immunologic self-tolerance maintained by CD25(+)CD4(+) regulatory T cells constitutively expressing cytotoxic T lymphocyte-associated antigen 4. J Exp Med. 2000;192(2):303–310. doi: 10.1084/jem.192.2.303. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 138.Kitazawa Y, Fujino M, Wang Q, et al. Involvement of the programmed death-1/programmed death-1 ligand pathway in CD4+CD25+ regulatory T-cell activity to suppress alloimmune responses. Transplantation. 2007;83(6):774–782. doi: 10.1097/01.tp.0000256293.90270.e8. [DOI] [PubMed] [Google Scholar]
- 139.Francisco LM, Salinas VH, Brown KE, et al. PD-L1 regulates the development, maintenance, and function of induced regulatory T cells. J Exp Med. 2009;206(13):3015–3029. doi: 10.1084/jem.20090847. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 140.Zhou Q, Munger ME, Highfill SL, et al. Program death-1 signaling and regulatory T cells collaborate to resist the function of adoptively transferred cytotoxic T lymphocytes in advanced acute myeloid leukemia. Blood. 2010;116(14):2484–2493. doi: 10.1182/blood-2010-03-275446. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 141.Yoshinaga SK, Whoriskey JS, Khare SD, et al. T-cell co-stimulation through B7RP-1 and ICOS. Nature. 1999;402(6763):827–832. doi: 10.1038/45582. [DOI] [PubMed] [Google Scholar]
- 142.Hutloff A, Dittrich AM, Beier KC, et al. ICOS is an inducible T-cell co-stimulator structurally and functionally related to CD28. Nature. 1999;397(6716):263–266. doi: 10.1038/16717. [DOI] [PubMed] [Google Scholar]
- 143.Swallow MM, Wallin JJ, Sha WC. B7h, a novel costimulatory homolog of B7.1 and B7.2, is induced by TNFalpha. Immunity. 1999;11(4):423–432. doi: 10.1016/s1074-7613(00)80117-x. [DOI] [PubMed] [Google Scholar]
- 144.Wang S, Zhu G, Chapoval AI, et al. Costimulation of T cells by B7-H2, a B7-like molecule that binds ICOS. Blood. 2000;96(8):2808–2813. [PubMed] [Google Scholar]
- 145.Yao S, Zhu Y, Zhu G, et al. B7-h2 is a costimulatory ligand for CD28 in human. Immunity. 2011;34(5):729–740. doi: 10.1016/j.immuni.2011.03.014. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 146.Linterman MA, Rigby RJ, Wong R, et al. Roquin differentiates the specialized functions of duplicated T-cell costimulatory receptor genes CD28 and ICOS. Immunity. 2009;30(2):228–241. doi: 10.1016/j.immuni.2008.12.015. [DOI] [PubMed] [Google Scholar]
- 147.Breitfeld D, Ohl L, Kremmer E, et al. Follicular B helper T cells express CXC chemokine receptor 5, localize to B-cell follicles, and support immunoglobulin production. J Exp Med. 2000;192(11):1545–1552. doi: 10.1084/jem.192.11.1545. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 148.Schaerli P, Willimann K, Lang AB, Lipp M, Loetscher P, Moser B. CXC chemokine receptor 5 expression defines follicular homing T cells with B-cell helper function. J Exp Med. 2000;192(11):1553–1562. doi: 10.1084/jem.192.11.1553. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 149.Vinuesa CG, Cook MC, Angelucci C, et al. A RING-type ubiquitin ligase family member required to repress follicular helper T cells and autoimmunity. Nature. 2005;435(7041):452–458. doi: 10.1038/nature03555. [DOI] [PubMed] [Google Scholar]
- 150.Yu D, Tan AH, Hu X, et al. Roquin represses autoimmunity by limiting inducible T-cell co-stimulator messenger RNA. Nature. 2007;450(7167):299–303. doi: 10.1038/nature06253. [DOI] [PubMed] [Google Scholar]
- 151.Watanabe M, Takagi Y, Kotani M, et al. Down-regulation of ICOS ligand by interaction with ICOS functions as a regulatory mechanism for immune responses. J Immunol. 2008;180(8):5222–5234. doi: 10.4049/jimmunol.180.8.5222. [DOI] [PubMed] [Google Scholar]
- 152.Rottman JB, Smith T, Tonra JR, et al. The costimulatory molecule ICOS plays an important role in the immunopathogenesis of EAE. Nat Immunol. 2001;2(7):605–611. doi: 10.1038/89750. [DOI] [PubMed] [Google Scholar]
- 153.Dong C, Juedes AE, Temann UA, et al. ICOS co-stimulatory receptor is essential for T-cell activation and function. Nature. 2001;409(6816):97–101. doi: 10.1038/35051100. [DOI] [PubMed] [Google Scholar]
- 154.Lohning M, Hutloff A, Kallinich T, et al. Expression of ICOS in vivo defines CD4+ effector T cells with high inflammatory potential and a strong bias for secretion of interleukin 10. J Exp Med. 2003;197(2):181–193. doi: 10.1084/jem.20020632. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 155.Kornete M, Sgouroudis E, Piccirillo CA. ICOS-dependent homeostasis and function of Foxp3+ regulatory T cells in islets of nonobese diabetic mice. J Immunol. 2012;188(3):1064–1074. doi: 10.4049/jimmunol.1101303. [DOI] [PubMed] [Google Scholar]
- 156.Grimbacher B, Hutloff A, Schlesier M, et al. Homozygous loss of ICOS is associated with adult-onset common variable immunodeficiency. Nat Immunol. 2003;4(3):261–268. doi: 10.1038/ni902. [DOI] [PubMed] [Google Scholar]
- 157.Warnatz K, Bossaller L, Salzer U, et al. Human ICOS deficiency abrogates the germinal center reaction and provides a monogenic model for common variable immunodeficiency. Blood. 2006;107(8):3045–3052. doi: 10.1182/blood-2005-07-2955. [DOI] [PubMed] [Google Scholar]
- 158.Marafioti T, Paterson JC, Ballabio E, et al. The inducible T-cell co-stimulator molecule is expressed on subsets of T cells and is a new marker of lymphomas of T follicular helper cell-derivation. Haematologica. 2010;95(3):432–439. doi: 10.3324/haematol.2009.010991. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 159.Zhang Q, Wang H, Kantekure K, et al. Oncogenic tyrosine kinase NPM-ALK induces expression of the growth-promoting receptor ICOS. Blood. 2011;118(11):3062–3071. doi: 10.1182/blood-2011-01-332916. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 160.Wallin JJ, Liang L, Bakardjiev A, Sha WC. Enhancement of CD8+ T-cell responses by ICOS/B7h costimulation. J Immunol. 2001;167(1):132–139. doi: 10.4049/jimmunol.167.1.132. [DOI] [PubMed] [Google Scholar]
- 161.Liu X, Bai XF, Wen J, et al. B7H costimulates clonal expansion of, and cognate destruction of tumor cells by, CD8(+) T lymphocytes in vivo. J Exp Med. 2001;194(9):1339–1348. doi: 10.1084/jem.194.9.1339. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 162.Zuberek K, Ling V, Wu P, et al. Comparable in vivo efficacy of CD28/B7, ICOS/GL50, and ICOS/GL50B costimulatory pathways in murine tumor models: IFNgamma-dependent enhancement of CTL priming, effector functions, and tumor specific memory CTL. Cell Immunol. 2003;225(1):53–63. doi: 10.1016/j.cellimm.2003.09.002. [DOI] [PubMed] [Google Scholar]
- 163.Martin-Orozco N, Li Y, Wang Y, et al. Melanoma cells express ICOS ligand to promote the activation and expansion of T-regulatory cells. Cancer Res. 2010;70(23):9581–9590. doi: 10.1158/0008-5472.CAN-10-1379. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 164.Schreiner B, Wischhusen J, Mitsdoerffer M, et al. Expression of the B7-related molecule ICOSL by human glioma cells in vitro and in vivo. Glia. 2003;44(3):296–301. doi: 10.1002/glia.10291. [DOI] [PubMed] [Google Scholar]
- 165.Yamashita T, Tamura H, Satoh C, et al. Functional B7.2 and B7-H2 molecules on myeloma cells are associated with a growth advantage. Clin Cancer Res. 2009;15(3):770–777. doi: 10.1158/1078-0432.CCR-08-0501. [DOI] [PubMed] [Google Scholar]
- 166.Wang F, Zhu W, Liu T, et al. The expression analysis of ICOS-L on activated T cells and immature dendritic cells as well as malignant B cells and GraveÆs-disease-derived thyroid tissues by two novel mAbs against human ICOS-L. Tissue Antigens. 2007;69(1):62–72. doi: 10.1111/j.1399-0039.2006.00706.x. [DOI] [PubMed] [Google Scholar]
- 167.Ogasawara K, Yoshinaga SK, Lanier LL. Inducible costimulator costimulates cytotoxic activity and IFN-gamma production in activated murine NK cells. J Immunol. 2002;169(7):3676–3685. doi: 10.4049/jimmunol.169.7.3676. [DOI] [PubMed] [Google Scholar]
- 168.Marten A, Renoth S, von Lilienfeld-Toal M, et al. Enhanced lytic activity of cytokine-induced killer cells against multiple myeloma cells after co-culture with idiotype-pulsed dendritic cells. Haematologica. 2001;86(10):1029–1037. [PubMed] [Google Scholar]
- 169.Leemhuis T, Wells S, Scheffold C, Edinger M, Negrin RS. A phase I trial of autologous cytokine-induced killer cells for the treatment of relapsed Hodgkin disease and non-Hodgkin lymphoma. Biol Blood Marrow Transplant. 2005;11(3):181–187. doi: 10.1016/j.bbmt.2004.11.019. [DOI] [PubMed] [Google Scholar]
- 170.He M, Wang Y, Shi WJ, et al. Immunomodulation of inducible co-stimulator (ICOS) in human cytokine-induced killer cells against cholangiocarcinoma through ICOS/ICOS ligand interaction. J Dig Dis. 2011;12(5):393–400. doi: 10.1111/j.1751-2980.2011.00527.x. [DOI] [PubMed] [Google Scholar]
- 171.Fu T, He Q, Sharma P. The ICOS/ICOSL pathway is required for optimal antitumor responses mediated by anti-CTLA-4 therapy. Cancer Res. 2011;71(16):5445–5454. doi: 10.1158/0008-5472.CAN-11-1138. [DOI] [PubMed] [Google Scholar]
- 172.Liakou CI, Kamat A, Tang DN, et al. CTLA-4 blockade increases IFNgamma-producing CD4+ICOShi cells to shift the ratio of effector to regulatory T cells in cancer patients. Proc Natl Acad Sci U S A. 2008;105(39):14987–14992. doi: 10.1073/pnas.0806075105. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 173.Ozkaynak E, Gao W, Shemmeri N, et al. Importance of ICOS-B7RP-1 costimulation in acute and chronic allograft rejection. Nat Immunol. 2001;2(7):591–596. doi: 10.1038/89731. [DOI] [PubMed] [Google Scholar]
- 174.Taylor PA, Panoskaltsis-Mortari A, Freeman GJ, et al. Targeting of inducible costimulator (ICOS) expressed on alloreactive T cells down-regulates graft-versus-host disease (GVHD) and facilitates engraftment of allogeneic bone marrow (BM). Blood. 2005;105(8):3372–3380. doi: 10.1182/blood-2004-10-3869. [DOI] [PubMed] [Google Scholar]
- 175.Hubbard VM, Eng JM, Ramirez-Montagut T, et al. Absence of inducible costimulator on alloreactive T cells reduces graft versus host disease and induces Th2 deviation. Blood. 2005;106(9):3285–3292. doi: 10.1182/blood-2005-01-0410. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 176.Yu XZ, Liang Y, Nurieva RI, Guo F, Anasetti C, Dong C. Opposing effects of ICOS on graft-versus-host disease mediated by CD4 and CD8 T cells. J Immunol. 2006;176(12):7394–7401. doi: 10.4049/jimmunol.176.12.7394. [DOI] [PubMed] [Google Scholar]
- 177.Chapoval AI, Ni J, Lau JS, et al. B7-H3: a costimulatory molecule for T-cell activation and IFN-gamma production. Nat Immunol. 2001;2(3):269–274. doi: 10.1038/85339. [DOI] [PubMed] [Google Scholar]
- 178.Hashiguchi M, Kobori H, Ritprajak P, Kamimura Y, Kozono H, Azuma M. Triggering receptor expressed on myeloid cell-like transcript 2 (TLT-2) is a counter-receptor for B7-H3 and enhances T-cell responses. Proc Natl Acad Sci U S A. 2008;105(30):10495–10500. doi: 10.1073/pnas.0802423105. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 179.Hashiguchi M, Inamochi Y, Nagai S, et al. Human B7-H3 binds to triggering receptor expressed on myeloid cells-like transcript 2 (TLT-2) and enhances T-cell responses. Open. J Immunol. 2012;2(1):9–16. [Google Scholar]
- 180.Leitner J, Klauser C, Pickl WF, et al. B7-H3 is a potent inhibitor of human T-cell activation: No evidence for B7-H3 and TREML2 interaction. Eur. J Immunol. 2009;39(7):1754–1764. doi: 10.1002/eji.200839028. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 181.Luo L, Chapoval AI, Flies DB, et al. B7-H3 enhances tumor immunity in vivo by costimulating rapid clonal expansion of antigen-specific CD8+ cytolytic T cells. J Immunol. 2004;173(9):5445–5450. doi: 10.4049/jimmunol.173.9.5445. [DOI] [PubMed] [Google Scholar]
- 182.Sun X, Vale M, Leung E, Kanwar JR, Gupta R, Krissansen GW. Mouse B7-H3 induces antitumor immunity. Gene Ther. 2003;10(20):1728–1734. doi: 10.1038/sj.gt.3302070. [DOI] [PubMed] [Google Scholar]
- 183.Suh WK, Gajewska BU, Okada H, et al. The B7 family member B7-H3 preferentially down-regulates T helper type 1-mediated immune responses. Nat Immunol. 2003;4(9):899–906. doi: 10.1038/ni967. [DOI] [PubMed] [Google Scholar]
- 184.Sun Y, Wang Y, Zhao J, et al. B7-H3 and B7-H4 expression in non-small-cell lung cancer. Lung Cancer. 2006;53(2):143–151. doi: 10.1016/j.lungcan.2006.05.012. [DOI] [PubMed] [Google Scholar]
- 185.Zang X, Thompson RH, Al-Ahmadie HA, et al. B7-H3 and B7x are highly expressed in human prostate cancer and associated with disease spread and poor outcome. Proc Natl Acad Sci U S A. 2007;104(49):19458–19463. doi: 10.1073/pnas.0709802104. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 186.Sica GL, Choi IH, Zhu G, et al. B7-H4, a molecule of the B7 family, negatively regulates T-cell immunity. Immunity. 2003;18(6):849–861. doi: 10.1016/s1074-7613(03)00152-3. [DOI] [PubMed] [Google Scholar]
- 187.Prasad DV, Richards S, Mai XM, Dong C. B7S1, a novel B7 family member that negatively regulates T-cell activation. Immunity. 2003;18(6):863–873. doi: 10.1016/s1074-7613(03)00147-x. [DOI] [PubMed] [Google Scholar]
- 188.Zang X, Loke P, Kim J, Murphy K, Waitz R, Allison JP. B7x: a widely expressed B7 family member that inhibits T-cell activation. Proc Natl Acad Sci U S A. 2003;100(18):10388–10392. doi: 10.1073/pnas.1434299100. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 189.Watanabe N, Gavrieli M, Sedy JR, et al. BTLA is a lymphocyte inhibitory receptor with similarities to CTLA-4 and PD-1. Nat Immunol. 2003;4(7):670–679. doi: 10.1038/ni944. [DOI] [PubMed] [Google Scholar]
- 190.Sedy JR, Gavrieli M, Potter KG, et al. B and T lymphocyte attenuator regulates T-cell activation through interaction with herpesvirus entry mediator. Nat Immunol. 2005;6(1):90–98. doi: 10.1038/ni1144. [DOI] [PubMed] [Google Scholar]
- 191.Brandt CS, Baratin M, Yi EC, et al. The B7 family member B7-H6 is a tumor cell ligand for the activating natural killer cell receptor NKp30 in humans. J Exp Med. 2009;206(7):1495–1503. doi: 10.1084/jem.20090681. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 192.Zhang T, Wu MR, Sentman CL. An NKp30-based chimeric antigen receptor promotes T cell effector functions and antitumor efficacy in vivo. J Immunol. 2012;189(5):2290–2299. doi: 10.4049/jimmunol.1103495. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 193.Barbee SD, Woodward MJ, Turchinovich G, et al. Skint-1 is a highly specific, unique selecting component for epidermal T cells. Proc Natl Acad Sci U S A. 2011;108(8):3330–3335. doi: 10.1073/pnas.1010890108. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 194.Harly C, Guillaume Y, Nedellec S, et al. Key implication of CD277/Butyrophilin-3 (BTN3A) in cellular stress sensing by a major human gammadelta T-cell subset. Blood. 2012;120(11):2269–2279. doi: 10.1182/blood-2012-05-430470. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 195.Ogg SL, Komaragiri MV, Mather IH. Structural organization and mammary-specific expression of the butyrophilin gene. Mamm Genome. 1996;7(12):900–905. doi: 10.1007/s003359900265. [DOI] [PubMed] [Google Scholar]
- 196.Boyden LM, Lewis JM, Barbee SD, et al. Skint1, the prototype of a newly identified immunoglobulin superfamily gene cluster, positively selects epidermal gammadelta T cells. Nat Genet. 2008;40(5):656–662. doi: 10.1038/ng.108. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 197.Cubillos-Ruiz JR, Martinez D, Scarlett UK, et al. CD277 is a negative co-stimulatory molecule universally expressed by ovarian cancer microenvironmental cells. Oncotarget. 2010;1(5):329–338. doi: 10.18632/oncotarget.165. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 198.Compte E, Pontarotti P, Collette Y, Lopez M, Olive D. Frontline: characterization of BT3 molecules belonging to the B7 family expressed on immune cells. Eur J Immunol. 2004;34(8):2089–2099. doi: 10.1002/eji.200425227. [DOI] [PubMed] [Google Scholar]
- 199.Yamashiro H, Yoshizaki S, Tadaki T, Egawa K, Seo N. Stimulation of human butyrophilin 3 molecules results in negative regulation of cellular immunity. J Leukocyte Biol. 2010;88(4):757–767. doi: 10.1189/jlb.0309156. [DOI] [PubMed] [Google Scholar]
- 200.Messal N, Mamessier E, Sylvain A, et al. Differential role for CD277 as a co-regulator of the immune signal in T and NK cells. Eur J Immunol. 2011;41(12):3443–3454. doi: 10.1002/eji.201141404. [DOI] [PubMed] [Google Scholar]