Abstract
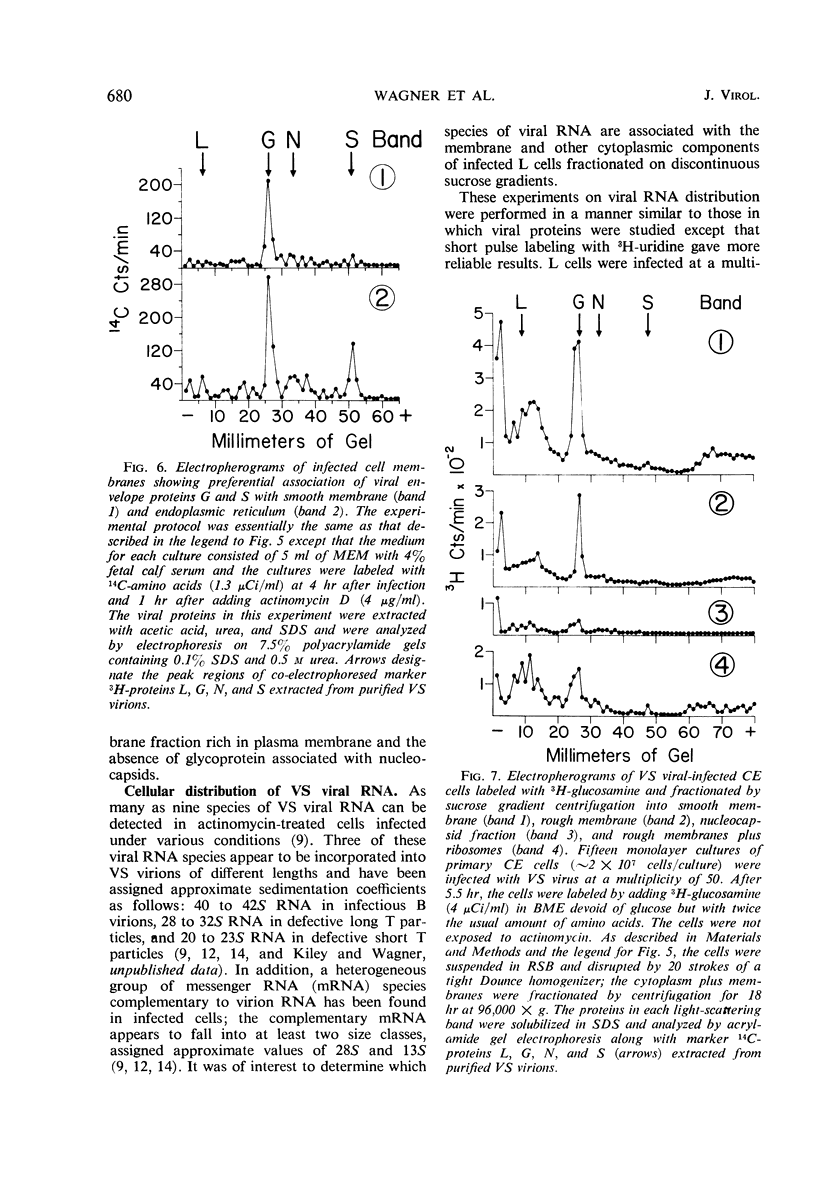
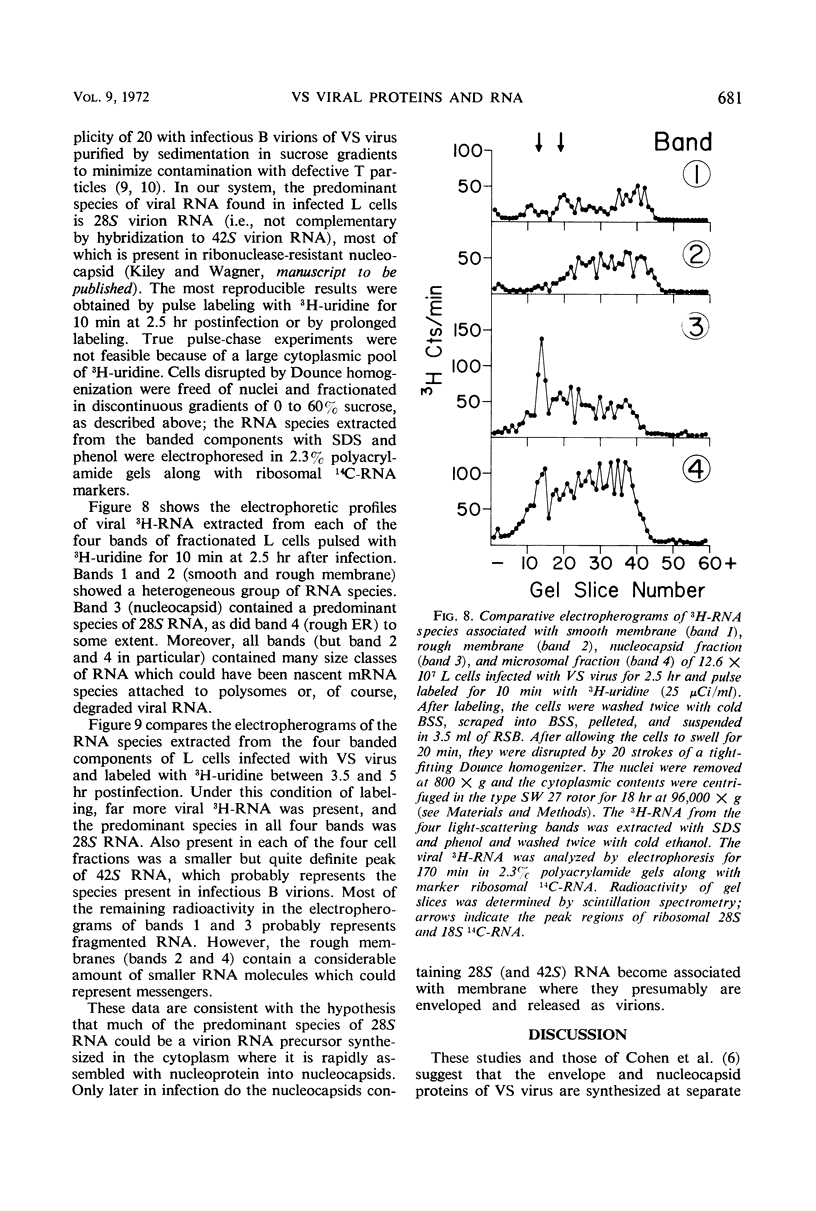
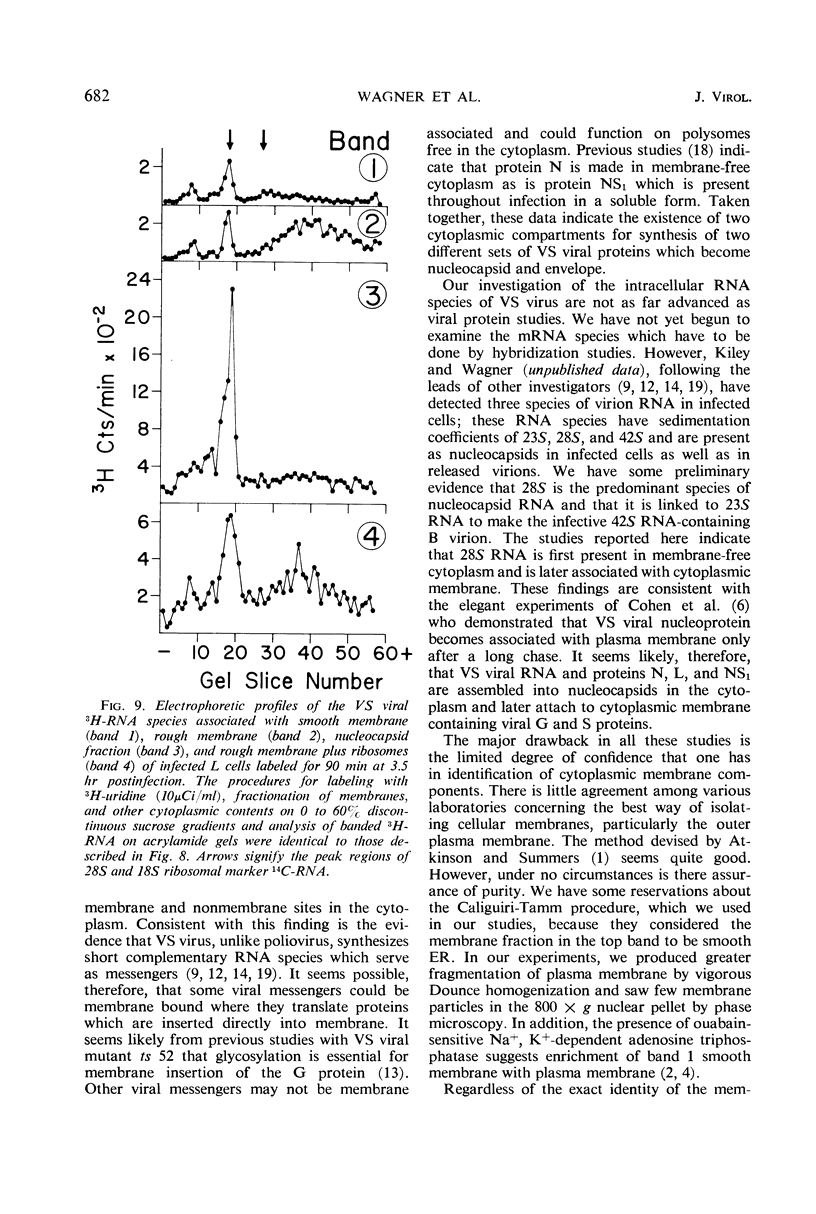
The cytoplasmic sites of synthesis in L cells of the protein and ribonucleic acid species of vesicular stomatitis virus were studied by polyacrylamide gel electrophoresis after fractionation of membrane and other cytoplasmic components by the Caliguiri-Tamm technique. The viral spike protein (glycoprotein G) was found primarily associated with a smooth membrane fraction which is rich in plasma membrane; the G protein was also present in fractions containing rough endoplasmic reticulum. The nonglycosylated envelope protein S (also called M) was found in the smooth membrane fractions but was more abundant in endoplasmic reticulum-enriched fractions. Longer labeling resulted in detection of nucleoprotein N, as well as other minor nucleocapsid proteins L and NS1, in the cellular membrane fractions. The N protein appeared to be made in membrane-free cytoplasm along with progeny ribonucleic acid and later became associated with membrane containing G and S viral proteins.
Full text
PDF


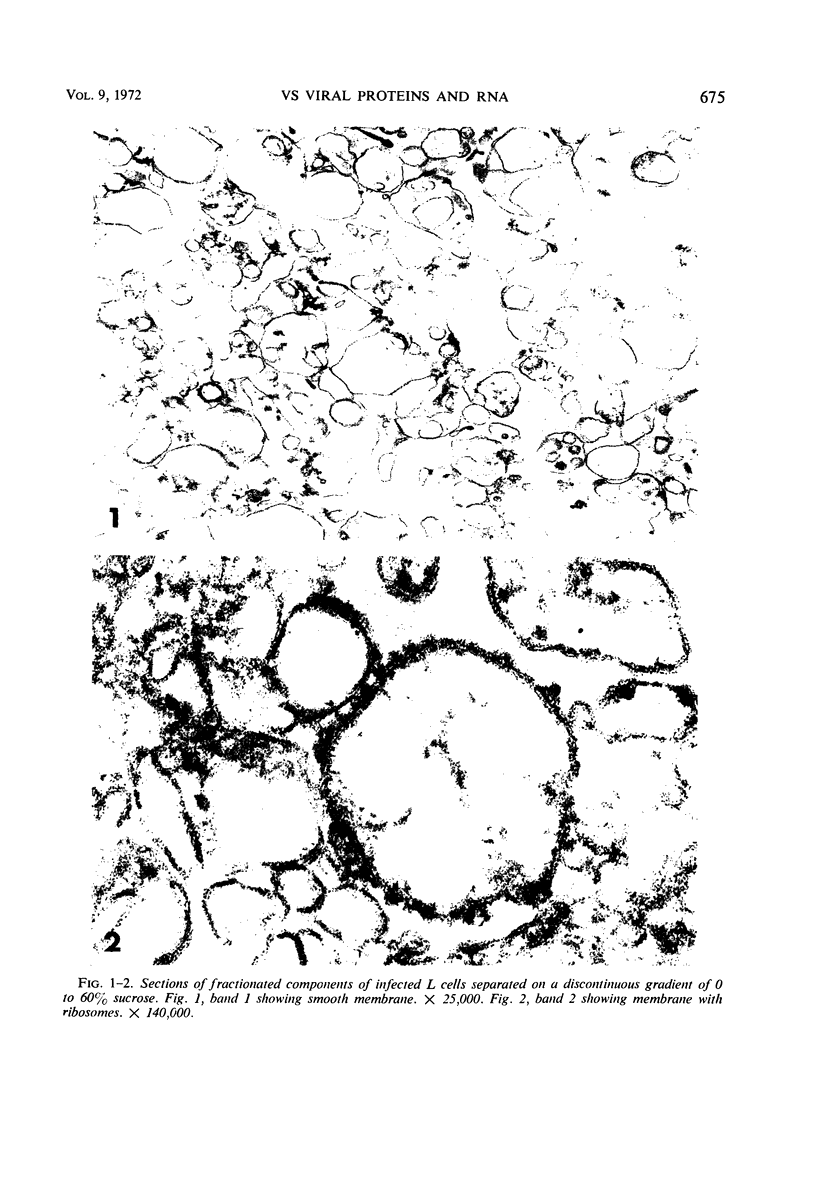

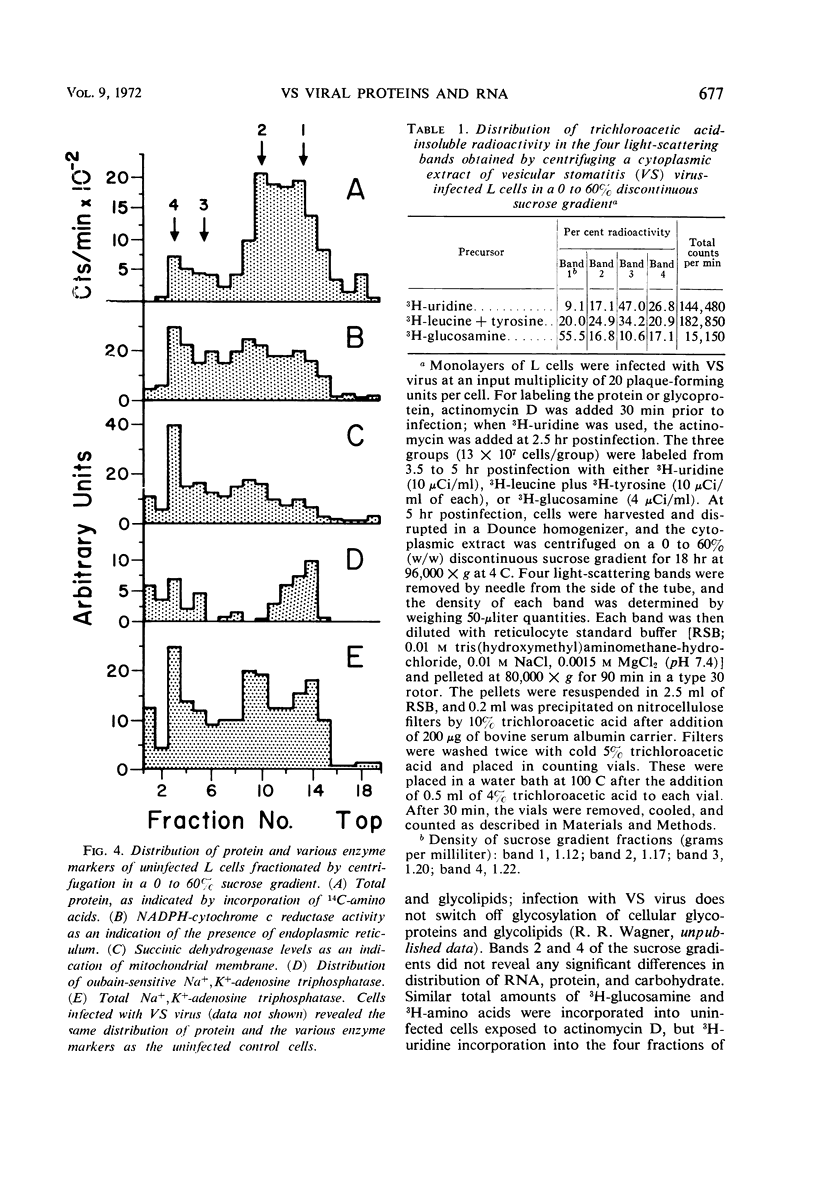

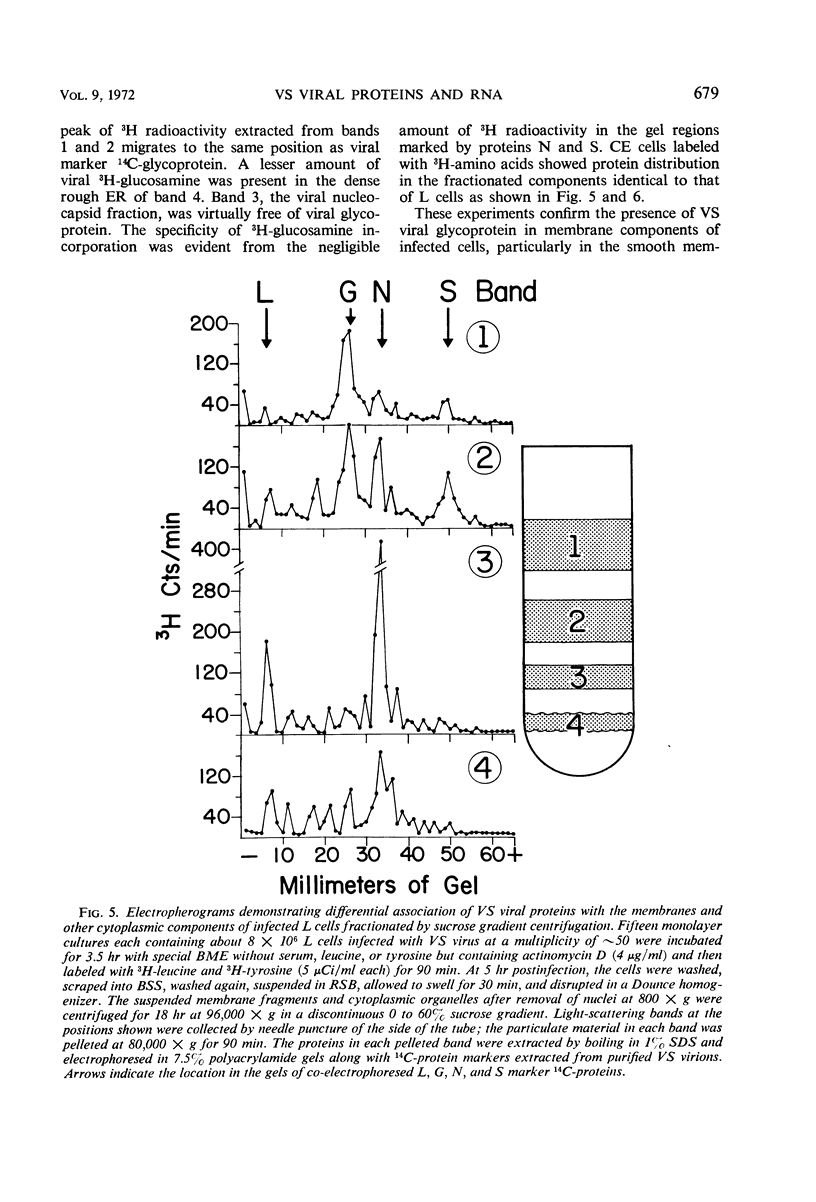

Images in this article
Selected References
These references are in PubMed. This may not be the complete list of references from this article.
- Atkinson P. H., Summers D. F. Purification and properties of HeLa cell plasma membranes. J Biol Chem. 1971 Aug 25;246(16):5162–5175. [PubMed] [Google Scholar]
- Avruch J., Wallach D. F. Preparation and properties of plasma membrane and endoplasmic reticulum fragments from isolated rat fat cells. Biochim Biophys Acta. 1971 Apr 13;233(2):334–347. doi: 10.1016/0005-2736(71)90331-2. [DOI] [PubMed] [Google Scholar]
- Baltimore D., Huang A. S., Stampfer M. Ribonucleic acid synthesis of vesicular stomatitis virus, II. An RNA polymerase in the virion. Proc Natl Acad Sci U S A. 1970 Jun;66(2):572–576. doi: 10.1073/pnas.66.2.572. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Caliguiri L. A., Tamm I. The role of cytoplasmic membranes in poliovirus biosynthesis. Virology. 1970 Sep;42(1):100–111. doi: 10.1016/0042-6822(70)90242-4. [DOI] [PubMed] [Google Scholar]
- Cohen G. H., Atkinson P. H., Summers D. F. Interactions of vesicular stomatitis virus structural proteins with HeLa plasma membranes. Nat New Biol. 1971 May 26;231(21):121–123. doi: 10.1038/newbio231121a0. [DOI] [PubMed] [Google Scholar]
- Duesberg P. H., Blair P. B. Isolation of the nucleic acid of mouse mammary tumor virus (MTV). Proc Natl Acad Sci U S A. 1966 Jun;55(6):1490–1497. doi: 10.1073/pnas.55.6.1490. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Heine J. W., Schnaitman C. A. Entry of vesicular stomatitis virus into L cells. J Virol. 1971 Nov;8(5):786–795. doi: 10.1128/jvi.8.5.786-795.1971. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Huang A. S., Baltimore D., Stampfer M. Ribonucleic acid synthesis of vesicular stomatitis virus. 3. Multiple complementary messenger RNA molecules. Virology. 1970 Dec;42(4):946–957. doi: 10.1016/0042-6822(70)90343-0. [DOI] [PubMed] [Google Scholar]
- Huang A. S., Greenawalt J. W., Wagner R. R. Defective T particles of vesicular stomatitis virus. I. Preparation, morphology, and some biologic properties. Virology. 1966 Oct;30(2):161–172. doi: 10.1016/0042-6822(66)90092-4. [DOI] [PubMed] [Google Scholar]
- MacGregor C. H., Schnaitman C. A. Alterations in the cytoplasmic membrane proteins of various chlorate-resistant mutants of Escherichia coli. J Bacteriol. 1971 Oct;108(1):564–570. doi: 10.1128/jb.108.1.564-570.1971. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Mudd J. A., Summers D. F. Polysomal ribonucleic acid of vesicular stomatitis virus-infected HeLa cells. Virology. 1970 Dec;42(4):958–968. doi: 10.1016/0042-6822(70)90344-2. [DOI] [PubMed] [Google Scholar]
- Printz P., Wagner R. R. Temperature-sensitive mutants of vesicular stomatitis virus: synthesis of virus-specific proteins. J Virol. 1971 May;7(5):651–662. doi: 10.1128/jvi.7.5.651-662.1971. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Schincariol A. L., Howatson A. F. Replication of vesicular stomatitis virus. I. Viral specific RNA and nucleoprotein in infected L cells. Virology. 1970 Nov;42(3):732–743. doi: 10.1016/0042-6822(70)90319-3. [DOI] [PubMed] [Google Scholar]
- Sottocasa G. L., Kuylenstierna B., Ernster L., Bergstrand A. An electron-transport system associated with the outer membrane of liver mitochondria. A biochemical and morphological study. J Cell Biol. 1967 Feb;32(2):415–438. doi: 10.1083/jcb.32.2.415. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Wagner R. R., Heine J. W., Goldstein G., Schnaitman C. A. Use of antiviral-antiferritin hybrid antibody for localization of viral antigen in plasma membrane. J Virol. 1971 Feb;7(2):274–277. doi: 10.1128/jvi.7.2.274-277.1971. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Wagner R. R., Schnaitman T. C., Snyder R. M., Schnaitman C. A. Protein composition of the structural components of vesicular stomatitis virus. J Virol. 1969 Jun;3(6):611–618. doi: 10.1128/jvi.3.6.611-618.1969. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Wagner R. R., Snyder R. M., Yamazaki S. Proteins of vesicular stomatitis virus: kinetics and cellular sites of synthesis. J Virol. 1970 May;5(5):548–558. doi: 10.1128/jvi.5.5.548-558.1970. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Wild T. F. Replication of vesicular stomatitis virus: characterization of the virus-induced RNA. J Gen Virol. 1971 Nov;13(2):295–310. doi: 10.1099/0022-1317-13-2-295. [DOI] [PubMed] [Google Scholar]
- Yamazaki S., Wagner R. R. Action of interferon: kinetics and differential effects on viral functions. J Virol. 1970 Oct;6(4):421–429. doi: 10.1128/jvi.6.4.421-429.1970. [DOI] [PMC free article] [PubMed] [Google Scholar]