Adenosine – a purinergic signaling molecule
Biochemically, adenosine belongs to a group of molecules referred to as purines. Purines are heterocyclic aromatic molecules that are among the oldest and most influential biochemical compounds in evolutionary history [1]. They are critical building blocks of the genetic code, and therefore the substrate of life, as represented by DNA. These relatively simple molecules are composed from adenine and guanine, and without these molecules, life as we know it would not be possible [2]. In a wide sense, purines are central to the self-sustained and reproducible existence of nucleotide-protein systems, which form cells and tissues that ultimately resulted in an appearance of Homo sapiens [2]. As such, the purine adenosine is well recognized as molecular building block of the genetic code or as part of adenosine triphosphate (ATP) – the universal energy currency of biological reactions [3]. Beyond these function, Alan Drury and Albert Szent-Györgyi from the University of Cambridge introduced in 1929 the idea that purines could also function as extracellular signaling molecules. They injected extracts from cardiac tissues intravenously into a whole animal. They observed a transient slowing of the heart rate [4]. Following several purification steps, they came to the conclusion that the biologic activity in the extract was an “adenine compound”[4]. Today we have genetic evidence that the transient heart-block induced by intravascular adenosine injection is mediated by the activation of an adenosine receptor [5, 6]. Indeed, adenosine signaling can occur through four distinct adenosine receptors – the Adora1, Adora2a, Adora2b and Adora3 - all of them G-protein coupled receptors. Adenosine-induced heart block remains the most famous clinical application for adenosine signaling, as intravenous adenosine injection continues to be a mainstay therapy for the diagnosis and treatment of supraventricular tachycardia [7, 8].
The complex control of extracellular adenosine signaling
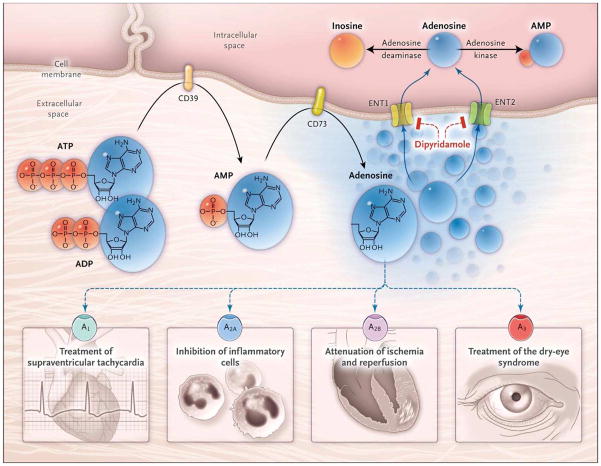
In order to better understand the control mechanism for extracellular adenosine signaling, many studies have addressed the question of where extracellular adenosine comes from. There are examples that extracellular adenosine can be released from intracellular stores [9–11]. However, many studies imply that during disease conditions such as hypoxia, ischemia or inflammation, extracellular adenosine predominantly stems from the breakdown of precursor nucleotides (for example ATP). Stressful conditions are associated with leakage or controlled release of ATP from the intracellular towards the extracellular compartment [12–14]. ATP is subsequently converted via the ectonucleoside triphosphate diphosphohydrolase 1 (CD39) to adenosine monophosphate (AMP) [15–20], and subsequently through the ecto-5′-nucleotidase CD73 to adenosine (Figure 1) [21–23]. Once released into the extracellular compartment, activation of adenosine receptors is responsible for adenosine’s biological activities as signaling molecule. For example, activation of the Adora2b receptor has been implicated in enhancing ischemia tolerance [24–26] and attenuating acute inflammatory responses [21, 25, 27–29].
Figure 1. Extracellular Adenosine Signaling and Its Termination.
In inflammatory conditions, extracellular adenosine is derived predominantly from the enzymatic conversion of the precursor nucleotides ATP and ADP to AMP through the enzymatic activity of the ectonucleoside triphosphate diphosphohydrolase 1 (CD39) and the subsequent conversion of AMP to adenosine through ecto-5′-nucleotidase (CD73). Extracellular adenosine can signal through four distinct adenosine receptors: ADORA1 (A1), ADORA2A (A2A), ADORA2B (A2B), and ADORA3 (A3). An example of the functional role of extracellular adenosine signaling is A1-receptor activation during intravenous administration of adenosine for the treatment of supraventricular tachycardia. In addition, experimental studies implicate activation of A2A that is expressed on inflammatory cells such as neutrophils or lymphocytes in the attenuation of inflammation. Other experimental studies provide evidence of signaling events through A2B in tissue adaptation to hypoxia and attenuation of ischemia and reperfusion. A clinical trial has shown that an oral agonist of the A3 adenosine receptor may be useful in the treatment of the dry-eye syndrome. Adenosine signaling is terminated by adenosine uptake from the extracellular space toward the intracellular space, predominantly through equilibrative nucleoside transporter 1 (ENT1) and equilibrative nucleoside transporter 2 (ENT2), followed by metabolism of adenosine to AMP through the adenosine kinase or to inosine through the adenosine deaminase. Blockade of equilibrative nucleoside transporters by dipyridamole is associated with increased extracellular adenosine concentrations and signaling (e.g., during pharmacologic stress echocardiography or in protection of tissue from ischemia). From Eltzschig HK, Sitkovsky MV, Robson SC. Purinergic Signaling during Inflammation. New England Journal of Medicine 2012;367:2322–33, Massachusetts Medical Society. Reprinted with permission from Massachusetts Medical Society.
There are many control steps regulating extracellular adenosine signaling events – such as ATP release, its conversion to adenosine via CD39 and CD73, or the expression of adenosine receptors (Figure 1). Moreover, there is evidence that alternative molecular pathways exist that can function to enhance extracellular adenosine signaling independent of adenosine, such as enhancement of purinergic signaling events through the neuronal guidance molecule netrin-1 [30–32]. Finally, the termination of extracellular adenosine signaling is a highly complex biological process with many steps that are independently regulated on a transcriptional level [33–35]. As such, adenosine is taken up from the extracellular into the intracellular compartment through adenosine transporters [24], and subsequently converted to inosine via the adenosine deaminase [36, 37], or via the adenosine kinase to AMP (Figure 1) [38]. These processes can function together to fine-tune extracellular adenosine levels and signaling functions [7].
Extracellular adenosine signaling during disease states
Due to the complexity of the system that regulates extracellular adenosine signaling, it has been challenging for many years to characterize biological functions of extracellular adenosine signaling during disease conditions. While first evidence was provided by pharmacological compounds that would specifically modulate signaling through individual adenosine receptors [39, 40], or studies utilizing compounds that would indirectly enhance extracellular adenosine concentrations [41], several breakthrough discoveries in the field were achieved during the past 15 years by generating and studying mice with genetic deletions of specific adenosine receptors or deletions of the enzymatic machinery that controls extracellular adenosine levels. In order to give an overview for the functional role of extracellular adenosine signaling during molecular medicine and its impact on human disease, five different research groups in this field are providing their perspective on recent advances in the field of extracellular adenosine signaling, and how it can be targeted for disease treatment. For example, a land-mark paper from the research group of Dr. Sitkovsky provided the first genetic evidence for an anti-inflammatory role for adenosine signaling as an endogenous feedback loop to limit collateral damage during uncontrolled inflammation via Adora2a receptors [42]. In extension of these findings this team of scientists found that excessive levels of extracellular adenosine may become detrimental during neoplastic disease states, thereby implicating adenosine receptor antagonists in the treatment of cancer [43]. As such, a review by Sitkovsky et al. in this issue is focused on how the hypoxia-adenosinergic signaling pathway can be targeted to improve the adoptive immunotherapy of cancer (49). Studies from the laboratory of Dr. Colgan implicated inflammatory hypoxia in the extracellular production and control of adenosine signaling, and identified hypoxia-induced increases in adenosine signaling as a control mechanism to dampen intestinal inflammation as occurs during inflammatory bowel disease (Figure 2) [29, 44, 45]. Therefore, a review by Colgan et al. is focused on adenosine and gastrointestinal inflammation (50). Studies from the laboratory of Dr. Robson utilized mice with genetic deletions of CD39 and CD73 to describe immunological roles of extracellular adenosine production by enhancing the anti-inflammtory functions of regulatory T-cells [46]. Indeed, a review from Longhi et al. describes biological functions of ecto-enzymes in regulating extracellular adenosine levels in neoplastic and inflammatory disease states [51]. Moreover, studies from the laboratory of Dr. Blackburn examined mice with genetic deletions of the adenosine deaminase [47]. These studies revealed dramatically increased levels of extracellular adenosine, and provide genetic evidence for a potentially detrimental role for prolonged adenosine elevations during chronic inflammatory disease states [36]. In the present issue, a review by Karmouty-Quintana et al. is focused on adenosine signaling during acute versus chronic disease states [52]. Finally, research work from our group has been interested over many years on the transcriptional regulation of adenosine responses, and particularly on the functional role of hypoxia-inducible factors to enhance acute adenosine protection during ischemia or inflammation. Indeed, a review by Poth et al. discusses the transcriptional control of adenosine signaling by hypoxiainducible transcription factors during ischemic or inflammatory disease states [53].
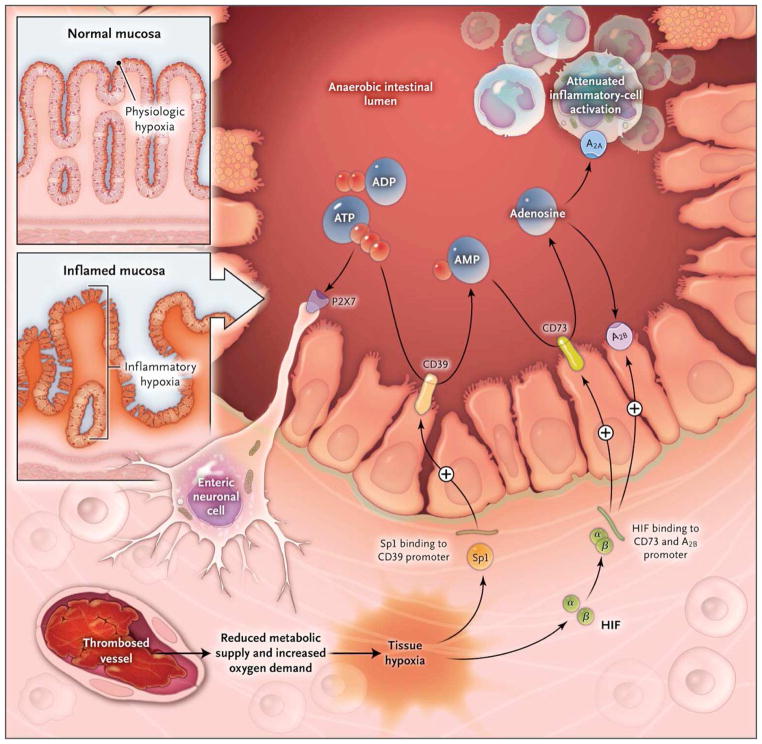
Figure 2. Hypoxia Control of Extracellular Adenosine Generation and Signaling in Intestinal Inflammation.
Histologic staining of intestinal sections for hypoxia shows that hypoxia is present within the apical surface of the intestinal mucosa (orange area in upper insert). This presence is most likely due to the fact that the intestinal lumen is anaerobic, which results in a steep oxygen gradient across the epithelial monolayer. In patients with intestinal inflammation such as that which occurs in the course of inflammatory bowel disease, a decrease in metabolic supply (e.g., due to thrombosed vessels) and profound increases in oxygen demand result in an imbalance in oxygen availability. This imbalance causes severe hypoxia of the inflamed mucosa, as indicated by histologic staining for tissue hypoxia (as shown in the lower insert, the orange staining that extends from the apical aspects of the mucosa into the crypts and submucosal tissues indicates severe tissue hypoxia). Release of ATP or ADP from inflammatory cells, platelets, or epithelial cells results in the activation of P2 receptors such as the P2X7 receptor, expressed on enteric neurons, thereby promoting tissue inflammation and injury. Hypoxia causes the activation of transcriptional programs that result in an Sp1-dependent induction of CD3917 and a hypoxia-inducible factor (HIF)–dependent induction of CD73 and the ADORA2B (A2B) adenosine receptor. These transcriptional changes lead to an increased rate of turnover of the extracellular nucleotides ATP and ADP to AMP (through CD39) and subsequently to adenosine (through CD73). Experimental studies indicate that adenosine-receptor activation — particularly through Adora2a (A2A)89 and A2B90 — dampens intestinal inflammation and promotes epithelial integrity during intestinal inflammation. From Eltzschig HK, Sitkovsky MV, Robson SC. Purinergic Signaling during Inflammation. New England Journal of Medicine 2012;367:2322–33, Massachusetts Medical Society. Reprinted with permission from Massachusetts Medical Society.
As such, there are numerous examples for the importance of extracellular adenosine signaling in molecular medicine. In most instances, extracellular adenosine signaling has anti-inflammatory functions during acute disease states, such as acute lung injury, ischemia and reperfusion or intestinal inflammation [7, 48]. Under such circumstances, pharmacologic approaches to enhance adenosine signaling effects (e.g. via adenosine receptor agonists or adenosine uptake inhibitors) are investigated in preclinical studies [50,51,53]. In contrast, adenosine-elicited inhibition of immune responses during neoplastic disease states contributes to tumor growth and metastasis, thereby implicating adenosine receptor blockers in the treatment of cancer [49]. Similarly, inhibition of adenosine receptors is an evolving therapeutic concept for the treatment chronic disease states, such as pulmonary fibrosis or sickle cell disease. Here, it will be particularly important to identify biomarkers that will help physicians to judge when adenosine protection during an acute disease states turns into promoting its chronicity [52]. Much work will be required to determine the clinical contexts in which the activation or inhibition of specific adenosine receptors can be utilized therapeutically to improve outcomes of acute and chronic inflammatory diseases states, ischemia and reperfusion injury or cancer. The five articles in this special issue of The Journal of Molecular Medicine [49–53] provide a roadmap for further exploration of the field of purinergic signaling for the treatment of human disease states.
Acknowledgments
The present research work was supported by National Heart Institute Grants R01-HL0921, R01-DK083385, R01-HL098294 and a grant by the Crohn’s and Colitis Foundation of America (CCFA) to H. K. E.
Footnotes
Conflict of interest: The author has declared no conflict of interest.
References
- 1.Miller SL, Urey HC. Organic compound synthesis on the primitive earth. Science. 1959;130:245–251. doi: 10.1126/science.130.3370.245. [DOI] [PubMed] [Google Scholar]
- 2.Fredholm B, Verkhratsky A. Purines - 80 years and very much alive. Acta Physiol (Oxf) 2010;199:91–92. doi: 10.1111/j.1748-1716.2010.02113.x. APS2113 [pii] [DOI] [PubMed] [Google Scholar]
- 3.Khakh BS, Burnstock G. The double life of ATP. Sci Am. 2009;301:84–90. 92. doi: 10.1038/scientificamerican1209-84. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 4.Drury AN, Szent-Gyorgyi A. The physiological activity of adenine compounds with especial reference to their action upon the mammalian heart. J Physiol. 1929;68:213–237. doi: 10.1113/jphysiol.1929.sp002608. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 5.Koeppen M, Eckle T, Eltzschig HK. Selective deletion of the A1 adenosine receptor abolishes heart-rate slowing effects of intravascular adenosine in vivo. PLoS One. 2009;4:e6784. doi: 10.1371/journal.pone.0006784. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 6.Eltzschig HK. Adenosine: an old drug newly discovered. Anesthesiology. 2009;111:904–915. doi: 10.1097/ALN.0b013e3181b060f2. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 7.Eltzschig HK, Sitkovsky MV, Robson SC. Purinergic Signaling during Inflammation. New England Journal of Medicine. 2012;367:2322–2333. doi: 10.1056/NEJMra1205750. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 8.Delacretaz E. Clinical practice. Supraventricular tachycardia. N Engl J Med. 2006;354:1039–1051. doi: 10.1056/NEJMcp051145. [DOI] [PubMed] [Google Scholar]
- 9.Lovatt D, Xu Q, Liu W, Takano T, Smith NA, Schnermann J, Tieu K, Nedergaard M. Neuronal adenosine release, and not astrocytic ATP release, mediates feedback inhibition of excitatory activity. Proc Natl Acad Sci U S A. 2012 doi: 10.1073/pnas.1120997109. 1120997109 [pii] [DOI] [PMC free article] [PubMed] [Google Scholar]
- 10.Fredholm BB. Rethinking the purinergic neuron-glia connection. Proc Natl Acad Sci U S A. 2012 doi: 10.1073/pnas.1203764109. 1203764109 [pii] [DOI] [PMC free article] [PubMed] [Google Scholar]
- 11.Decking UKM, Schlieper G, Kroll K, Schrader J. Hypoxia-Induced Inhibition of Adenosine Kinase Potentiates Cardiac Adenosine Release. Circ Res. 1997;81:154–164. doi: 10.1161/01.res.81.2.154. [DOI] [PubMed] [Google Scholar]
- 12.Faigle M, Seessle J, Zug S, El Kasmi KC, Eltzschig HK. ATP release from vascular endothelia occurs across Cx43 hemichannels and is attenuated during hypoxia. PLoS ONE. 2008;3:e2801. doi: 10.1371/journal.pone.0002801. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 13.Eltzschig HK, Weissmuller T, Mager A, Eckle T. Nucleotide metabolism and cell-cell interactions. Methods Mol Biol. 2006;341:73–87. doi: 10.1385/1-59745-113-4:73. [DOI] [PubMed] [Google Scholar]
- 14.Eltzschig HK, Eckle T, Mager A, Kuper N, Karcher C, Weissmuller T, Boengler K, Schulz R, Robson SC, Colgan SP. ATP release from activated neutrophils occurs via connexin 43 and modulates adenosine-dependent endothelial cell function. Circ Res. 2006;99:1100–1108. doi: 10.1161/01.RES.0000250174.31269.70. [DOI] [PubMed] [Google Scholar]
- 15.Hart ML, Gorzolla IC, Schittenhelm J, Robson SC, Eltzschig HK. SP1-dependent induction of CD39 facilitates hepatic ischemic preconditioning. J Immunol. 2010;184:4017–4024. doi: 10.4049/jimmunol.0901851. jimmunol.0901851 [pii] [DOI] [PMC free article] [PubMed] [Google Scholar]
- 16.Eltzschig HK, Kohler D, Eckle T, Kong T, Robson SC, Colgan SP. Central role of Sp1-regulated CD39 in hypoxia/ischemia protection. Blood. 2009;113:224–232. doi: 10.1182/blood-2008-06-165746. blood-2008-06-165746 [pii] [DOI] [PMC free article] [PubMed] [Google Scholar]
- 17.Reutershan J, Vollmer I, Stark S, Wagner R, Ngamsri KC, Eltzschig HK. Adenosine and inflammation: CD39 and CD73 are critical mediators in LPS-induced PMN trafficking into the lungs. FASEB J. 2009;23:473–482. doi: 10.1096/fj.08-119701. fj.08-119701 [pii] [DOI] [PubMed] [Google Scholar]
- 18.Eckle T, Fullbier L, Wehrmann M, Khoury J, Mittelbronn M, Ibla J, Rosenberger P, Eltzschig HK. Identification of ectonucleotidases CD39 and CD73 in innate protection during acute lung injury. J Immunol. 2007;178:8127–8137. doi: 10.4049/jimmunol.178.12.8127. [DOI] [PubMed] [Google Scholar]
- 19.Grenz A, Zhang H, Hermes M, Eckle T, Klingel K, Huang DY, Muller CE, Robson SC, Osswald H, Eltzschig HK. Contribution of E-NTPDase1 (CD39) to renal protection from ischemia-reperfusion injury. FASEB J. 2007;21:2863–2873. doi: 10.1096/fj.06-7947com. fj.06-7947com [pii] [DOI] [PubMed] [Google Scholar]
- 20.Kohler D, Eckle T, Faigle M, Grenz A, Mittelbronn M, Laucher S, Hart ML, Robson SC, Muller CE, Eltzschig HK. CD39/ectonucleoside triphosphate diphosphohydrolase 1 provides myocardial protection during cardiac ischemia/reperfusion injury. Circulation. 2007;116:1784–1794. doi: 10.1161/CIRCULATIONAHA.107.690180. CIRCULATIONAHA.107.690180 [pii] [DOI] [PubMed] [Google Scholar]
- 21.Hart ML, Grenz A, Gorzolla IC, Schittenhelm J, Dalton JH, Eltzschig HK. Hypoxia-inducible factor-1alpha-dependent protection from intestinal ischemia/reperfusion injury involves ecto-5′-nucleotidase (CD73) and the A2B adenosine receptor. J Immunol. 2011;186:4367–4374. doi: 10.4049/jimmunol.0903617. jimmunol.0903617 [pii] [DOI] [PMC free article] [PubMed] [Google Scholar] [Retracted]
- 22.Hart ML, Henn M, Kohler D, Kloor D, Mittelbronn M, Gorzolla IC, Stahl GL, Eltzschig HK. Role of extracellular nucleotide phosphohydrolysis in intestinal ischemia-reperfusion injury. FASEB J. 2008;22:2784–2797. doi: 10.1096/fj.07-103911. fj.07-103911 [pii] [DOI] [PMC free article] [PubMed] [Google Scholar]
- 23.Hart ML, Much C, Gorzolla IC, Schittenhelm J, Kloor D, Stahl GL, Eltzschig HK. Extracellular adenosine production by ecto-5′-nucleotidase protects during murine hepatic ischemic preconditioning. Gastroenterology. 2008;135:1739–1750. e1733. doi: 10.1053/j.gastro.2008.07.064. S0016-5085(08)01404-2 [pii] [DOI] [PubMed] [Google Scholar]
- 24.Grenz A, Bauerle JD, Dalton JH, Ridyard D, Badulak A, Tak E, McNamee EN, Clambey E, Moldovan R, Reyes G, et al. Equilibrative nucleoside transporter 1 (ENT1) regulates postischemic blood flow during acute kidney injury in mice. J Clin Invest. 2012;122:693–710. doi: 10.1172/JCI60214. 60214 [pii] [DOI] [PMC free article] [PubMed] [Google Scholar] [Retracted]
- 25.Ehrentraut H, Westrich JA, Eltzschig HK, Clambey ET. Adora2b Adenosine Receptor Engagement Enhances Regulatory T Cell Abundance during Endotoxin-Induced Pulmonary Inflammation. PLoS One. 2012;7:e32416. doi: 10.1371/journal.pone.0032416. PONE-D-11-16908 [pii] [DOI] [PMC free article] [PubMed] [Google Scholar]
- 26.Eckle T, Hartmann K, Bonney S, Reithel S, Mittelbronn M, Walker LA, Lowes BD, Han J, Borchers CH, Buttrick PM, Kominsky DJ, Colgan SP, Eltzschig HK. Adora2b-elicited Per2 stabilization promotes a HIF-dependent metabolic switch crucial for myocardial adaptation to ischemia. Nat Med. 2012;18:774–782. doi: 10.1038/nm.2728. nm.2728 [pii] [DOI] [PMC free article] [PubMed] [Google Scholar]
- 27.Grenz A, Kim JH, Bauerle JD, Tak E, Eltzschig HK, Clambey ET. Adora2b Adenosine Receptor Signaling Protects during Acute Kidney Injury via Inhibition of Neutrophil-Dependent TNF-alpha Release. J Immunol. 2012;189:4566–4573. doi: 10.4049/jimmunol.1201651. jimmunol.1201651 [pii] [DOI] [PMC free article] [PubMed] [Google Scholar] [Retracted]
- 28.Schingnitz U, Hartmann K, Macmanus CF, Eckle T, Zug S, Colgan SP, Eltzschig HK. Signaling through the A2B adenosine receptor dampens endotoxin-induced acute lung injury. J Immunol. 2010;184:5271–5279. doi: 10.4049/jimmunol.0903035. jimmunol.0903035 [pii] [DOI] [PMC free article] [PubMed] [Google Scholar]
- 29.Frick JS, MacManus CF, Scully M, Glover LE, Eltzschig HK, Colgan SP. Contribution of adenosine A2B receptors to inflammatory parameters of experimental colitis. J Immunol. 2009;182:4957–4964. doi: 10.4049/jimmunol.0801324. 182/8/4957 [pii] [DOI] [PMC free article] [PubMed] [Google Scholar]
- 30.Aherne CM, Collins CB, Masterson JC, Tizzano M, Boyle TA, Westrich JA, Parnes JA, Furuta GT, Rivera-Nieves J, Eltzschig HK. Neuronal guidance molecule netrin-1 attenuates inflammatory cell trafficking during acute experimental colitis. Gut. 2012;61:695–705. doi: 10.1136/gutjnl-2011-300012. gutjnl-2011-300012 [pii] [DOI] [PMC free article] [PubMed] [Google Scholar]
- 31.Grenz A, Dalton JH, Bauerle JD, Badulak A, Ridyard D, Gandjeva A, Aherne CM, Brodsky KS, Kim JH, Tuder RM, Eltzschig HK. Partial netrin-1 deficiency aggravates acute kidney injury. PLoS One. 2011;6:e14812. doi: 10.1371/journal.pone.0014812. 10-PONE-RA-21113R1 [pii] [DOI] [PMC free article] [PubMed] [Google Scholar]
- 32.Rosenberger P, Schwab JM, Mirakaj V, Masekowsky E, Mager A, Morote-Garcia JC, Unertl K, Eltzschig HK. Hypoxia-inducible factor-dependent induction of netrin-1 dampens inflammation caused by hypoxia. Nat Immunol. 2009;10:195–202. doi: 10.1038/ni.1683. [DOI] [PubMed] [Google Scholar]
- 33.Morote-Garcia JC, Rosenberger P, Nivillac NM, Coe IR, Eltzschig HK. Hypoxia-inducible factor-dependent repression of equilibrative nucleoside transporter 2 attenuates mucosal inflammation during intestinal hypoxia. Gastroenterology. 2009;136:607–618. doi: 10.1053/j.gastro.2008.10.037. S0016-5085(08)01874-X [pii] [DOI] [PubMed] [Google Scholar]
- 34.Loffler M, Morote-Garcia JC, Eltzschig SA, Coe IR, Eltzschig HK. Physiological roles of vascular nucleoside transporters. Arterioscler Thromb Vasc Biol. 2007;27:1004–1013. doi: 10.1161/ATVBAHA.106.126714. [DOI] [PubMed] [Google Scholar]
- 35.Eltzschig HK, Abdulla P, Hoffman E, Hamilton KE, Daniels D, Schonfeld C, Loffler M, Reyes G, Duszenko M, Karhausen J, Robinson A, Westerman KA, Coe IR, Colgan SP. HIF-1-dependent repression of equilibrative nucleoside transporter (ENT) in hypoxia. J Exp Med. 2005;202:1493–1505. doi: 10.1084/jem.20050177. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 36.Zhang Y, Dai Y, Wen J, Zhang W, Grenz A, Sun H, Tao L, Lu G, Alexander DC, Milburn MV, et al. Detrimental effects of adenosine signaling in sickle cell disease. Nat Med. 2011;17:79–86. doi: 10.1038/nm.2280. nm.2280 [pii] [DOI] [PMC free article] [PubMed] [Google Scholar]
- 37.Eltzschig HK, Faigle M, Knapp S, Karhausen J, Ibla J, Rosenberger P, Odegard KC, Laussen PC, Thompson LF, Colgan SP. Endothelial catabolism of extracellular adenosine during hypoxia: the role of surface adenosine deaminase and CD26. Blood. 2006;108:1602–1610. doi: 10.1182/blood-2006-02-001016. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 38.Morote-Garcia JC, Rosenberger P, Kuhlicke J, Eltzschig HK. HIF-1-dependent repression of adenosine kinase attenuates hypoxia-induced vascular leak. Blood. 2008;111:5571–5580. doi: 10.1182/blood-2007-11-126763. blood-2007-11-126763 [pii] [DOI] [PubMed] [Google Scholar]
- 39.Jacobson KA, Gao ZG. Adenosine receptors as therapeutic targets. Nat Rev Drug Discov. 2006;5:247–264. doi: 10.1038/nrd1983. nrd1983 [pii] [DOI] [PMC free article] [PubMed] [Google Scholar]
- 40.Cronstein BN, Daguma L, Nichols D, Hutchison AJ, Williams M. The adenosine/neutrophil paradox resolved: human neutrophils possess both A1 and A2 receptors that promote chemotaxis and inhibit O2 generation, respectively. J Clin Invest. 1990;85:1150–1157. doi: 10.1172/JCI114547. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 41.Morabito L, Montesinos MC, Schreibman DM, Balter L, Thompson LF, Resta R, Carlin G, Huie MA, Cronstein BN. Methotrexate and sulfasalazine promote adenosine release by a mechanism that requires ecto-5′-nucleotidase-mediated conversion of adenine nucleotides. J Clin Invest. 1998;101:295–300. doi: 10.1172/JCI1554. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 42.Ohta A, Sitkovsky M. Role of G-protein-coupled adenosine receptors in downregulation of inflammation and protection from tissue damage. Nature. 2001;414:916–920. doi: 10.1038/414916a. [DOI] [PubMed] [Google Scholar]
- 43.Ohta A, Gorelik E, Prasad SJ, Ronchese F, Lukashev D, Wong MK, Huang X, Caldwell S, Liu K, Smith P, et al. A2A adenosine receptor protects tumors from antitumor T cells. Proc Natl Acad Sci U S A. 2006;103:13132–13137. doi: 10.1073/pnas.0605251103. 0605251103 [pii] [DOI] [PMC free article] [PubMed] [Google Scholar]
- 44.Karhausen J, Furuta GT, Tomaszewski JE, Johnson RS, Colgan SP, Haase VH. Epithelial hypoxia-inducible factor-1 is protective in murine experimental colitis. J Clin Invest. 2004;114:1098–1106. doi: 10.1172/JCI21086. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 45.Colgan SP, Eltzschig HK. Adenosine and hypoxia-inducible factor signaling in intestinal injury and recovery. Annu Rev Physiol. 2012;74:153–175. doi: 10.1146/annurev-physiol-020911-153230. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 46.Deaglio S, Dwyer KM, Gao W, Friedman D, Usheva A, Erat A, Chen JF, Enjyoji K, Linden J, Oukka M, Kuchroo VK, Strom TB, Robson SC. Adenosine generation catalyzed by CD39 and CD73 expressed on regulatory T cells mediates immune suppression. J Exp Med. 2007;204:1257–1265. doi: 10.1084/jem.20062512. jem.20062512 [pii] [DOI] [PMC free article] [PubMed] [Google Scholar]
- 47.Blackburn MR, Volmer JB, Thrasher JL, Zhong H, Crosby JR, Lee JJ, Kellems RE. Metabolic Consequences of Adenosine Deaminase Deficiency in Mice Are Associated with Defects in Alveogenesis, Pulmonary Inflammation, and Airway Obstruction. J Exp Med. 2000;192:159–170. doi: 10.1084/jem.192.2.159. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 48.Eltzschig HK, Eckle T. Ischemia and reperfusion--from mechanism to translation. Nat Med. 2011;17:1391–1401. doi: 10.1038/nm.2507. nm.2507 [pii] [DOI] [PMC free article] [PubMed] [Google Scholar]
- 49.Sitkovsky M, Ohta A. Targeting the hypoxia-adenosinergic signaling pathway to improve the adoptive immunotherapy of cancer. J Mol Med. 2013 doi: 10.1007/s00109-013-1001-9. Dennis, please add correct citation. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 50.Colgan SP, Fennimore B, Ehrentraut SF. Adenosine and gastrointestinal inflammation. J Mol Med. 2013 doi: 10.1007/s00109-012-0990-0. add correct citation. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 51.Longhi MS, Robson SC, Bernstein SH, Serra S, Deaglio S. Biological functions of ecto-enzymes in regulating extracellular adenosine levels in neoplastic and inflammatory disease states. J Mol Med. 2013 doi: 10.1007/s00109-012-0991-z. add correct citation. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 52.Karmouty-Quintana H, Yang Xia, Blackburn MR. Adenosine signaling during acute and chronic disease states. J Mol Med. 2013 doi: 10.1007/s00109-013-0997-1. add correct citation. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 53.Poth JM, Brodsky K, Ehrentraut H, Grenz A, Eltzschig HK. Transcriptional control of adenosine signaling by hypoxiainducible transcription factors during ischemic or inflammatory disease. J Mol Med. 2013 doi: 10.1007/s00109-012-0988-7. add correct citation. [DOI] [PMC free article] [PubMed] [Google Scholar]