Abstract
The majority of hepatitis C virus (HCV) and human immunodeficiency virus (HIV) coinfection occurs among persons who inject drugs. Rapid improvements in responses to HCV therapy have been observed, but liver-related morbidity rates remain high, given notoriously low uptake of HCV treatment. Advances in HCV therapy will have a limited impact on the burden of HCV-related disease at the population-level unless barriers to HCV education, screening, evaluation, and treatment are addressed and treatment uptake increases. This review will outline barriers to HCV care in HCV/HIV coinfection, with a particular emphasis on persons who inject drugs, proposing strategies to enhance HCV treatment uptake and outcomes.
Keywords: human immunodeficiency virus, hepatitis C virus, drug users, coinfection, treatment, barriers
Overlapping modes of human immunodeficiency virus (HIV) and hepatitis C virus (HCV) acquisition have led to a large burden of HCV/HIV coinfection in many settings [1, 2]. HIV-infected persons who inject drugs (PWID), both active and former injectors comprise the majority of HCV/HIV coinfection in the developed world [2]. Although the incidence of HCV among HIV-infected men who have sex with men has increased (reviewed in [1, 3]), this group represents a minority of those with HCV/HIV coinfection. Among PWID, the higher prevalence and greater risk of HCV transmission through equipment sharing, compared with HIV transmission, means that the vast majority of PWID with HIV will be co-infected with HCV [2]. As reviewed elsewhere [2], HIV alters the natural history of HCV, accelerates liver disease progression, and is associated with considerable morbidity and mortality. For these reasons, the clinical management of HIV in PWID populations must include consideration HCV.
Given the considerable burden of HCV-related disease in co-infection and an aging population, assessment of HCV and liver disease is a crucial part of care for co-infected individuals, particularly among those with factors increasing risk of HCV-related disease progression. Accelerated liver disease progression among persons with HCV/HIV coinfection, coupled with inferior responses to interferon (IFN)–based HCV therapy, highlight the importance of improved direct-acting antiviral (DAA) therapy–based outcomes. Improving HCV therapeutic options will have limited impact, however, if barriers to HCV education, screening, evaluation, and treatment remain and low treatment uptake continues. This review will outline barriers to HCV treatment access in HCV/HIV coinfection, with an emphasis on PWID, and propose strategies to enhance HCV treatment uptake and outcomes.
HCV TREATMENT IS SUCCESSFUL AMONG PWID
The goal of HCV therapy is viral eradication, achieved when HCV RNA is undetectable in the blood 6 months after treatment, known as sustained virological response (SVR) or cure. HCV treatment with pegylated IFN (PEG-IFN) and ribavirin can achieve SVR in 40%–50% of co-infected individuals [2]. Newer DAAs offer further improvements in responses.
Older HCV treatment guidelines excluded substance users from HCV treatment consideration, based on concerns of poor adherence, exacerbation of preexisting substance use and psychiatric comorbid conditions, and reinfection [4]. There are now data from studies including both HCV- and HCV/HIV-infected PWID demonstrating that HCV treatment is successful [5]. Guidelines have been revised to recommend consideration of HCV treatment among PWID on a “case-by-case” basis. Data also suggest that modest increases in HCV treatment uptake among active PWID may achieve substantial reductions in HCV prevalence [6] and are cost-effective [7].
As reviewed elsewhere [4], PEG-IFN/ribavirin is safe and effective among PWID [4, 5]. Studies indicate that a history of injecting drug use (including recent use) and/or hazardous alcohol use does not generally compromise adherence, treatment completion or SVR, although some studies have found lower rates of treatment completion. HCV treatment does not have an impact on drug dependency treatment or increase drug use. Occasional drug use during treatment does not seem to affect outcomes. However, lower rates of adherence and SVR have been observed in persons with frequent drug use (daily or every other day) during treatment. Rates of reinfection are low, 1%–5% per year. Unfortunately, only a minority of participants enrolled in studies to date have been HCV/HIV co-infected. Future data on HCV treatment outcomes among PWID with HCV/HIV coinfection are a priority.
UNACCEPTABLY LOW HCV TREATMENT UPTAKE
Despite the growing burden of HCV-related liver disease among co-infected persons with ongoing substance use and psychiatric conditions, data demonstrating that HCV treatment can be successful in this patient population, and changes to guidelines supporting treatment, HCV treatment uptake remains unacceptably low. Among studies in Canada and the United States, only 1%–7% of those with HCV/HIV coinfection (the majority PWID) have received HCV treatment [8–11]. Data from a large HCV/HIV clinic in Baltimore, Maryland, showed that even among HCV/HIV-infected patients receiving HIV care, treatment uptake was <1% [9]. Compared with those with HCV infection alone, PWID with HCV/HIV coinfection have demonstrated lower rates for acceptance of clinical referral [10], willingness to undergo HCV treatment [11], and HCV treatment uptake [11].
HCV-infected individuals often have complex social, medical, psychiatric, and substance use–associated comorbid conditions, which complicate decisions about care and are further compounded by additional factors related to coinfection [1]. Impaired access to therapy often results from the combination of multiple barriers to antiviral therapy present at the levels of the system, provider, and patient.
SYSTEMS-LEVEL BARRIERS TO HCV CARE
At the systems level, there is lack of consensus about screening and treatment guidelines; thus, HCV testing and evaluation remain low. Furthermore, there is limited infrastructure for providing HCV assessment and treatment, particularly in substance use treatment and primary care services catering to marginalized populations. Limited knowledge about testing and treatment, limited accessibility of testing locations, and long waiting lists for accessing HCV care are cited as barriers to care among patients [4, 12]. In many countries, the high cost of PEG-IFN/ribavirin treatment presents a major barrier to care. In the United States, although HCV treatment is available for the majority of HCV/HIV-infected patients requiring PEG-IFN/ribavirin treatment through the Ryan White Care Act, it is unclear whether DAAs (including protease inhibitors) will be covered.
The prospect of considerably improved DAA-based therapeutic strategies in the next few years, including IFN-free regimens, has increased the importance of liver disease staging and treatment recommendations based on degree of fibrosis. Liver biopsy is problematic, and hepatic elastography (eg, FibroScan) is still not licensed in the United States, thus limiting opportunities to assess disease severity and target therapy to patients with more advanced fibrosis. Finally, reimbursements available to HCV care providers are limited, so there is little monetary incentive for engaging in this otherwise rewarding area of medicine.
PRACTITIONER-LEVEL BARRIERS TO HCV CARE
At the practitioner level, perceptions about poor adherence, ongoing substance use, relapse to substance use, risk of exacerbating comorbid psychiatric disease and potential risk of reinfection have often been used as reasons for withholding therapy [4]. Many physicians are unwilling to treat patients actively using drugs. In a study of Canadian HCV specialists, only 20% would consider providing treatment to active PWID [13]. Among other physicians, HCV treatment is not seen as part of their “core” business. In a national study of addiction medicine physicians in the United States, although 61% reported screening most PWID for HCV antibodies, only 9% reported providing HCV treatment and only 30% were willing to provide HCV treatment, even if given the appropriate training and resources [14]. A lack of confidence in initiating HCV treatment because of low case numbers and inadequate HCV knowledge are noted as factors for persistently low HCV screening, evaluation, and treatment rates [15]. In 3 large HIV clinics in Los Angeles, California, patients beginning HCV treatment more often had providers with higher caseloads of HCV/HIV-infected patients, greater HCV treatment experience, greater confidence in HCV care management, positive expectancies of HCV treatment, and more lenient views about alcohol and depression in their treatment philosophies than those who did not receive treatment [16].
These findings demonstrate that some physicians have suboptimal knowledge about HCV and its treatment, which may be due to limited training and inexperience with HCV treatment in coinfection. In some settings, there is also a low perceived need for treatment due to a lack of realization that fibrosis is advanced (given that liver disease may not have been appropriately assessed) or a belief that other comorbid diseases should take precedence. There are also clear misconceptions about HCV treatment in those with ongoing substance use. Concerns about a high risk-to-benefit ratio and the perception that many patients are poor treatment candidates because of preconceived notions about substance use or psychiatric comorbid conditions that will negatively affect treatment outcomes often lead to delayed or no entry into HCV care. However, data across substance use, primary, and outpatient mental health settings demonstrate that the possibility of initiating antiviral therapy can be a robust motivating factor for decreasing alcohol use in patients with HCV infection [17]. HCV treatment decisions for PWID with coinfection have added complexity because of issues related to the timing of antiretroviral therapy (ART), the severity of HIV disease, and drug-drug interactions [4]. Qualitative research with primary care HIV providers in the United States found that experienced providers agree that stable HIV disease, favorable HCV genotype, and advanced liver disease are all factors supporting a stronger recommendation for HCV treatment [18]. However, many providers are deferring therapy, given that IFN-sparing regimens with increased simplicity (once-daily dosing and shorter therapy duration), improved tolerability and efficacy will probably soon be available.
PATIENT-LEVEL BARRIERS TO HCV CARE
At the patient level, major barriers for accessing HCV care include poor knowledge and inaccurate perceptions about HCV infection and its treatment [4, 11, 19]. Poor knowledge, combined with the absence of noticeable symptoms and perceptions about HCV infection being a benign disease, results in a low perceived need for treatment [4, 12, 20]. Other important barriers to HCV care include unemployment or employment responsibilities, unstable housing, lack of transportation, parental responsibilities, poverty, incarceration, stigma, and inadequate access to healthcare (in uninsured or underinsured patients [4, 21]). Given that HCV prevalence is often higher among ethnic minorities, racial and ethnic disparities in access to healthcare also complicate HCV care. Furthermore, patients may intentionally avoid assessment and treatment because of the “horror stories” about liver biopsies and HCV treatment propagated within peer networks [12]. In addition, persons with HCV/HIV co-infected coinfection may have other medical comorbid conditions (eg, cardiovascular disease) which may require more immediate attention. Sociodemographic and cognitive-affective factors may lead to forgotten appointments and poor adherence to the HCV evaluation and treatment process [12]. Patient-provider relationships often have an important influence on whether patients embark on HCV treatment [12, 20, 21].
STRATEGIES TO ENHANCE HCV CARE IN COINFECTION
At the systems level, improving the proportion of diagnosed individuals is crucial to enhancing HCV assessment. The United States Department of Health and Human Services (DHHS) action plan for viral hepatitis proposes creating standard recommendations to guide HCV testing and referral to care, implement routine HCV testing and linkage to care, and promote health information technology to improve testing and enhance referral to HCV care [22, 23]. Risk-based and birth cohort (1945–1964) screening strategies, now recommended by the Centers for Disease Control and Prevention, have been associated with increases in HCV testing in primary care [24]. Given the risk of HCV among persons infected with HIV, annual testing for HCV antibodies is recommended by some guidelines (eg, those of the European AIDS Clinical Society [25]), but this practice has not yet been adopted in the United States [1]. The licensing and reimbursement for transient elastography (eg, FibroScan) and other noninvasive methods of assessing liver disease in the United States and elsewhere would also enhance HCV evaluation and targeting of therapy to those at greatest need. These noninvasive methods offer simple, effective tools for advanced liver disease screening, enabling the triage of patients for more immediate care.
Enhanced screening will require the appropriate infrastructure for HCV assessment and treatment. This could include implementing HCV care services in HIV, substance use treatment, and primary care settings. Increasing the number of providers offering HCV treatment could also be achieved by improving reimbursement for HCV care. Other strategies for enhancing infrastructure for HCV care for underserved populations focus on the integration of community-based health centers using telehealth technology to provide training and support for primary care providers to deliver HCV care [26]. Peer-support workers may also be important in enhancing HCV assessment, given data suggesting that engagement in HCV care may be facilitated by the influence of peers who completed treatment [4]. There needs to be a paradigm shift toward an HCV “seek, test, treat, and prevent” strategy in coinfection, setting a new standard of universal treatment, with the goal of eradicating HCV in coinfection.
At the practitioner level, enhanced HCV education for providers working in the fields of HIV, addiction, and primary care would enhance HCV assessment and treatment [1, 23]. Consistent with the DHHS action plan for improving provider education in the United States [22, 23], training programs need to educate providers about factors influencing liver disease progression, liver assessment (given that many patients with advanced disease are asymptomatic and/or without physical stigmata or laboratory data suggesting advanced fibrosis), HCV treatment candidacy (based on evidence) and HCV treatment efficacy (addressing myths about the negative impact of psychiatric and substance use on HCV treatment outcomes). Cross-specialty training for untrained providers through collaborations and targeted in-services with trained specialists would also be helpful. The DHHS action plan proposes several of these strategies to help improve provider education in the United States [22, 23].
Given the rapid advancement of DAA-based therapy for HCV, it is realistic that the norm within 4–5 years will be a once-daily, short-duration (12-week), and tolerable regimen (eliminating IFN-mediated side effects) that is curative in the majority of patients. Given the increased simplicity of treatment and need to rapidly expand access to care, HCV infection may move toward becoming a disease managed primarily by infectious disease physicians [27], addiction specialists, and primary care providers. Hepatology specialists will remain crucial, with perhaps a greater focus on the management of patients with more advanced liver disease. This expanded access of treatment and broadened provider base will need a corresponding increase in access to therapy. HCV antiviral therapy access programs will be required, along lines similar to those of successful HIV ART programs, incorporating drug price reform and generic production for resource-limited countries,.
At the patient level, practitioners need to provide greater pretreatment education about HCV transmission, risk factors for fibrosis progression, treatment, reinfection risk, and risk reduction strategies. Pretreatment assessment should include considerations of current psychiatric disorders (mood, anxiety, depression, previous suicide attempts), substance use, sociodemographic factors (lifestyle, daily routine, housing and living environment, transportation), social functioning and support, finances, nutrition, coping skills, history of violence and previous adherence behavior (attending regular medical appointments and adherence to ART for HIV infection).
Psychiatric health and substance use conditions are highly prevalent in co-infected patients and are underdiagnosed [28]. Given that depressive symptoms, anxiety, and substance use can affect both IFN- and non–IFN-containing regimens, it is important to assess these domains in preparation for HCV treatment. Systematic and comprehensive use of brief, validated scales for depression, anxiety, and substance use can prompt providers to address these health issues or refer to behavioral health providers for further detailed assessment and treatment, if required [28]. Furthermore, the systematic use of such scales for all HCV/HIV co-infected individuals may lead to mental health referrals and follow-up by a colocated mental health provider and is associated with high rates of HCV treatment initiation and greater adherence to antiviral therapy [29].
STRATEGIES TO ENHANCE HCV TREATMENT ADHERENCE AND OUTCOMES
A variety of clinical models have been successful in delivering HCV treatment to co-infected PWID including hospital-based liver disease, viral hepatitis and HIV clinics, substance use detoxification clinics, opioid substitution clinics, prisons, and primary care clinics. The basis for effective clinical management within these settings is access to a multidisciplinary team, generally including the treating provider, nurses, access to substance use services, and access to behavioral health providers (psychiatrists, psychologists, social workers) and other social support services (including peer support, if available). Although the level of engagement between disciplines can vary between models, communication between providers across disciplines is essential. Strategies that have proved successful for enhancing HCV evaluation, adherence, or treatment response include hospital-based and primary and specialty care-based integrated care [28], community-based telehealth [26], nurse-led education [30], directly observed therapy [31], and peer-support groups and workers [32–34].
A key component moving forward in the era of DAA-based therapy will be adherence management programs to enhance outcomes for both HCV and HIV treatments among co-infected PWID. Evidence-based strategies to enhance adherence include patient education, cognitive-behavioral interventions to promote adherence, prepackaged medication blister packs, directly observed therapy, prompt management of adverse events, medication managers, individual or group-based peer support and technology-based adherence reminders (text and mobile applications). Although many of these strategies have been assessed for improving ART adherence, there are limited data in the setting of HCV. Further research is needed in this area to demonstrate which evidence-based strategies are effective for enhancing HCV treatment adherence and outcomes. In the future, the goal is to have simple, once-daily, single pill regimens for HCV, as are available for HIV.
FUTURE TREATMENT OF HCV AMONG HCV/HIV
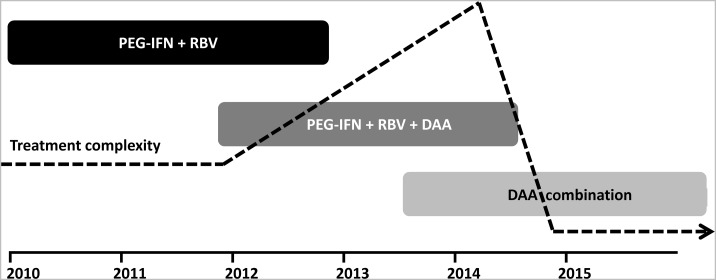
Although time lines for HCV therapeutic strategies are not clear, phase 3 studies of IFN-free therapy commenced in 2012 for HCV-monoinfected patients (Figure 1) and studies in coinfected patients will follow soon [35]. Thus, it is likely that the period of triple therapy with PEG-IFN/ribavirin and a protease inhibitor will be relatively short-lived and replaced by IFN-free therapy for the majority of monoinfected and coinfected patients (Figure 1). Initially, for the next few years, the overall complexity of HCV management will increase with protease inhibitor-based therapy, given the concerns of side effects and drug interactions among those with coinfection. However, a rapid decline in complexity will be observed after the likely switch to IFN-free regimens, given considerably reduced toxicity, shortened treatment durations, and simplified dosing and monitoring schedules.
Figure 1.
Phases of therapeutic development for the treatment of hepatitis C virus infection and the associated complexity of clinical management. Adapted from Dore [35] with permission. Abbreviations: DAA, direct-acting antiviral; PEG-IFN, pegylated interferon; RBV, ribavirin.
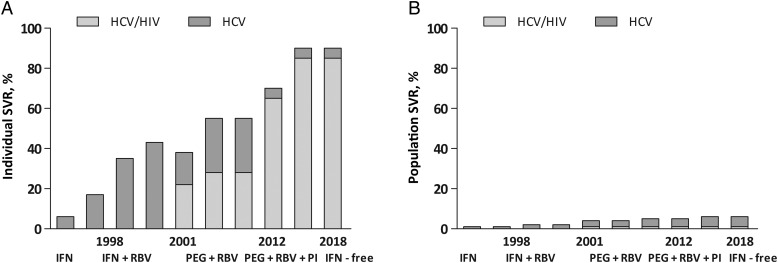
Unless the proportion of individuals screened, assessed, and treated for HCV infection is increased significantly, these impressive anticipated advancements in HCV therapy will have little effect on HCV at the population level (Figure 2). Strategies to increase HCV screening, assessment and treatment are urgently needed. The key will be to ameliorate barriers to care at the system, provider, and patient levels, preparing for the IFN-free era and future eradication of HCV. Finally, the strong association between the level of immunodeficiency and mortality from both AIDS-related illness and liver disease means that effective management of HIV, including optimum ART adherence, should be the foundation on which enhanced HCV management is built.
Figure 2.
Disparity between potential hepatitis C virus (HCV) treatment efficacy and projected HCV treatment effectiveness. A, High HCV treatment efficacy if projected over 5-10 years; the sustained virological response (SVR) will increase from 55% with pegylated-interferon (PEG-IFN) and ribavirin (RBV) to approximately 70% in the era of PEG-IFN (65% in HCV/human immunodeficiency virus [HIV] coinfection), RBV, and a protease inhibitor (genotype 1 only, with HCV genotype 2/3 patients still having an SVR of approximately 80% with PEG-IFN/RBV). It is anticipated that by 2018, IFN-free agents will be available, with SVR equal to 90% in HCV infection (85% in HCV/HIV coinfection). B, The global impact of new treatment is negligible without expanded access; in 2005, estimates in Europe and the United States suggested that approximately 3% of patients had received treatment, with treatment uptake increasing by only 0.5% per year (adapted from Thomas et al [2], with permission). Abbreviations: HCV, hepatitis C virus; HIV, human immunodeficiency virus; IFN, interferon; PEG, pegylated IFN; RBV, ribavirin; SVR, sustained virological response.
Notes
Acknowledgments. The authors would like to thank David L. Thomas (The Johns Hopkins University) for his helpful comments and access to data for the concept previously developed to explore potential HCV treatment efficacy and projected HCV treatment effectiveness.
Financial support. The Kirby Institute is funded by the Australian Government Department of Health and Ageing and is affiliated with the Faculty of Medicine, University of New South Wales. G. J. D. is supported by a National Health and Medical Research Council Practitioner Research Fellowship. J. G. is supported by a National Health and Medical Research Council Career Development Fellowship. This publication resulted in part from research supported by from the National Institute on Drug Abuse (grant K23 DA020383 to L. E. T.) and the Lifespan/Tufts/Brown Center for AIDS Research, supported by the National Institute of Allergy and Infectious Diseases (grant P30AI042853). The content is solely the responsibility of the authors and does not necessarily represent the official views of the National Institute on Drug Abuse, the National Institute of Allergy and Infectious Diseases, or the National Institutes of Health.
Potential conflicts of interest. J. G. is a consultant/advisor for Merck. L. E. T. was a past member of the speakers’ bureau for Genentech. G. J. D. is a consultant/advisor for and has received research grants from Roche, Merck, Janssen, Gilead, and Bristol-Myers Squibb. All other authors report no potential conflicts.
All authors have submitted the ICMJE Form for Disclosure of Potential Conflicts of Interest. Conflicts that the editors consider relevant to the content of the manuscript have been disclosed.
References
- 1.Taylor LE, Swan T, Mayer KH. HIV coinfection with hepatitis C virus: evolving epidemiology and treatment paradigms. Clin Infect Dis. 2012;55(Suppl 1):S33–42. doi: 10.1093/cid/cis367. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 2.Thomas DL, Leoutsakas D, Zabransky T, Kumar MS. Hepatitis C in HIV-infected individuals: cure and control, right now. J Int AIDS Soc. 2011;14:22. doi: 10.1186/1758-2652-14-22. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 3.Kim A. An epidemiologic update on hepatitis C infection in persons living with or at risk of HIV infection. J Infect Dis. 2012 doi: 10.1093/infdis/jis927. doi:10.1093/infdis/jis927. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 4.Grebely J, Tyndall MW. Management of HCV and HIV infections among people who inject drugs. Curr Opin HIV AIDS. 2011;6:501–7. doi: 10.1097/COH.0b013e32834bcb36. [DOI] [PubMed] [Google Scholar]
- 5.Hellard M, Sacks-Davis R, Gold J. Hepatitis C treatment for injection drug users: a review of the available evidence. Clin Infect Dis. 2009;49:561–73. doi: 10.1086/600304. [DOI] [PubMed] [Google Scholar]
- 6.Martin NK, Vickerman P, Foster GR, Hutchinson SJ, Goldberg DJ, Hickman M. Can antiviral therapy for hepatitis C reduce the prevalence of HCV among injecting drug user populations? A modeling analysis of its prevention utility. J Hepatol. 2011;54:1137–44. doi: 10.1016/j.jhep.2010.08.029. [DOI] [PubMed] [Google Scholar]
- 7.Martin NK, Vickerman P, Miners A, et al. Cost-effectiveness of hepatitis C virus antiviral treatment for injection drug user populations. Hepatology. 2012;55:49–57. doi: 10.1002/hep.24656. [DOI] [PubMed] [Google Scholar]
- 8.Grebely J, Raffa JD, Lai C, et al. Low uptake of treatment for hepatitis C virus infection in a large community-based study of inner city residents. J Viral Hepat. 2009;16:352–8. doi: 10.1111/j.1365-2893.2009.01080.x. [DOI] [PubMed] [Google Scholar]
- 9.Mehta SH, Lucas GM, Mirel LB, et al. Limited effectiveness of antiviral treatment for hepatitis C in an urban HIV clinic. AIDS. 2006;20:2361–9. doi: 10.1097/QAD.0b013e32801086da. [DOI] [PubMed] [Google Scholar]
- 10.Fishbein DA, Lo Y, Reinus JF, Gourevitch MN, Klein RS. Factors associated with successful referral for clinical care of drug users with chronic hepatitis C who have or are at risk for HIV infection. J Acquir Immune Defic Syndr. 2004;37:1367–75. doi: 10.1097/01.qai.0000131932.21612.49. [DOI] [PubMed] [Google Scholar]
- 11.Grebely J, Genoway KA, Raffa JD, et al. Barriers associated with the treatment of hepatitis C virus infection among illicit drug users. Drug Alcohol Depend. 2008;93:141–7. doi: 10.1016/j.drugalcdep.2007.09.008. [DOI] [PubMed] [Google Scholar]
- 12.Swan D, Long J, Carr O, et al. Barriers to and facilitators of hepatitis C testing, management, and treatment among current and former injecting drug users: a qualitative exploration. AIDS Patient Care STDS. 2010;24:753–62. doi: 10.1089/apc.2010.0142. [DOI] [PubMed] [Google Scholar]
- 13.Myles A, Mugford GJ, Zhao J, Krahn M, Wang PP. Physicians’ attitudes and practice toward treating injection drug users with hepatitis C: results from a national specialist survey in Canada. Can J Gastroenterol. 2011;25:135–9. doi: 10.1155/2011/810108. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 14.Litwin AH, Kunins HV, Berg KM, et al. Hepatitis C management by addiction medicine physicians: results from a national survey. J Subst Abuse Treat. 2007;33:99–105. doi: 10.1016/j.jsat.2006.12.001. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 15.Lambert SM, Page AN, Wittmann J, et al. General practitioner attitudes to prescribing hepatitis C antiviral therapy in a community setting. Aust J Prim Health. 2011;17:282–7. doi: 10.1071/PY10069. [DOI] [PubMed] [Google Scholar]
- 16.Osilla KC, Wagner G, Garnett J, et al. Patient and provider characteristics associated with the decision of HIV coinfected patients to start hepatitis C treatment. AIDS Patient Care STDS. 2011;25:533–8. doi: 10.1089/apc.2011.0048. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 17.Oser M, Cucciare M, McKellar J, Weingardt K. Correlates of hazardous drinking among veterans with and without hepatitis C. J Behav Med. 2012;35:634–41. doi: 10.1007/s10865-011-9394-9. [DOI] [PubMed] [Google Scholar]
- 18.Wagner G, Ryan G, Osilla KC, Bhatti L, Goetz M, Witt M. Treat early or wait and monitor? A qualitative analysis of provider hepatitis C virus treatment decision-making in the context of HIV coinfection. AIDS Patient Care STDS. 2009;23:715–25. doi: 10.1089/apc.2009.0049. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 19.Treloar C, Hull P, Dore GJ, Grebely J. Knowledge and barriers associated with assessment and treatment for hepatitis C virus infection among people who inject drugs. Drug Alcohol Rev. 2012;31:918–24. doi: 10.1111/j.1465-3362.2012.00468.x. [DOI] [PubMed] [Google Scholar]
- 20.Osilla KC, Ryan G, Bhatti L, Goetz M, Witt M, Wagner G. Factors that influence an HIV coinfected patient's decision to start hepatitis C treatment. AIDS Patient Care STDS. 2009;23:993–9. doi: 10.1089/apc.2009.0153. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 21.Treloar C, Newland J, Rance J, Hopwood M. Uptake and delivery of hepatitis C treatment in opiate substitution treatment: perceptions of clients and health professionals. J Viral Hepat. 2010;17:839–44. doi: 10.1111/j.1365-2893.2009.01250.x. [DOI] [PubMed] [Google Scholar]
- 22.US Department of Health and Human Services. Washington, DC: US Department of Health and Human Services; 2011. Combating the silent epidemic of viral hepatitis: action plan for the prevention, care, and treatment of viral hepatitis; pp. 1–76. [Google Scholar]
- 23.Ward JW, Valdiserri RO, Koh HK. Hepatitis C virus prevention, care, and treatment: from policy to practice. Clin Infect Dis. 2012;55(Suppl 1):S58–63. doi: 10.1093/cid/cis392. [DOI] [PubMed] [Google Scholar]
- 24.Litwin AH, Smith BD, Drainoni ML, et al. Primary care-based interventions are associated with increases in hepatitis C virus testing for patients at risk. Dig Liver Dis. 2012;44:497–503. doi: 10.1016/j.dld.2011.12.014. [DOI] [PubMed] [Google Scholar]
- 25.Rockstroh JK, Bhagani S, Benhamou Y, et al. European AIDS Clinical Society (EACS) guidelines for the clinical management and treatment of chronic hepatitis B and C coinfection in HIV-infected adults. HIV Med. 2008;9:82–8. doi: 10.1111/j.1468-1293.2007.00535.x. [DOI] [PubMed] [Google Scholar]
- 26.Arora S, Thornton K, Murata G, et al. Outcomes of treatment for hepatitis C virus infection by primary care providers. N Engl J Med. 2011;364:2199–207. doi: 10.1056/NEJMoa1009370. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 27.McGovern BH. Editorial commentary: hepatitis C virus and the infectious disease physician: a perfect match. Clin Infect Dis. 2012;55:414–7. doi: 10.1093/cid/cis378. [DOI] [PubMed] [Google Scholar]
- 28.Bonner JE, Barritt ASt, Fried MW, Evon DM. Time to rethink antiviral treatment for hepatitis C in patients with coexisting mental health/substance abuse issues. Dig Dis Sci. 2012;57:1469–74. doi: 10.1007/s10620-012-2141-4. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 29.Knott A, Dieperink E, Willenbring ML, et al. Integrated psychiatric/medical care in a chronic hepatitis C clinic: effect on antiviral treatment evaluation and outcomes. Am J Gastroenterol. 2006;101:2254–62. doi: 10.1111/j.1572-0241.2006.00731.x. [DOI] [PubMed] [Google Scholar]
- 30.Larrey D, Salse A, Ribard D, et al. Education by a nurse increases response of patients with chronic hepatitis C to therapy with peginterferon-alpha2a and ribavirin. Clin Gastroenterol Hepatol. 2011;9:781–5. doi: 10.1016/j.cgh.2011.05.022. [DOI] [PubMed] [Google Scholar]
- 31.Grebely J, Raffa JD, Meagher C, et al. Directly observed therapy for the treatment of hepatitis C virus infection in current and former injection drug users. J Gastroenterol Hepatol. 2007;22:1519–25. doi: 10.1111/j.1440-1746.2007.05032.x. [DOI] [PubMed] [Google Scholar]
- 32.Grebely J, Knight E, Genoway KA, et al. Optimizing assessment and treatment for hepatitis C virus infection in illicit drug users: a novel model incorporating multidisciplinary care and peer support. Eur J Gastroenterol Hepatol. 2010;22:270–7. doi: 10.1097/meg.0b013e32832a8c4c. [DOI] [PubMed] [Google Scholar]
- 33.Sylvestre DL, Zweben JE. Integrating HCV services for drug users: a model to improve engagement and outcomes. Int J Drug Policy. 2007;18:406–10. doi: 10.1016/j.drugpo.2007.01.010. [DOI] [PubMed] [Google Scholar]
- 34.Norman J, Walsh NM, Mugavin J, et al. The acceptability and feasibility of peer worker support role in community based HCV treatment for injecting drug users. Harm Reduct J. 2008;5:8. doi: 10.1186/1477-7517-5-8. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 35.Dore GJ. The changing therapeutic landscape for hepatitis C. Med J Aust. 2012;196:629–32. doi: 10.5694/mja11.11531. [DOI] [PubMed] [Google Scholar]