Abstract
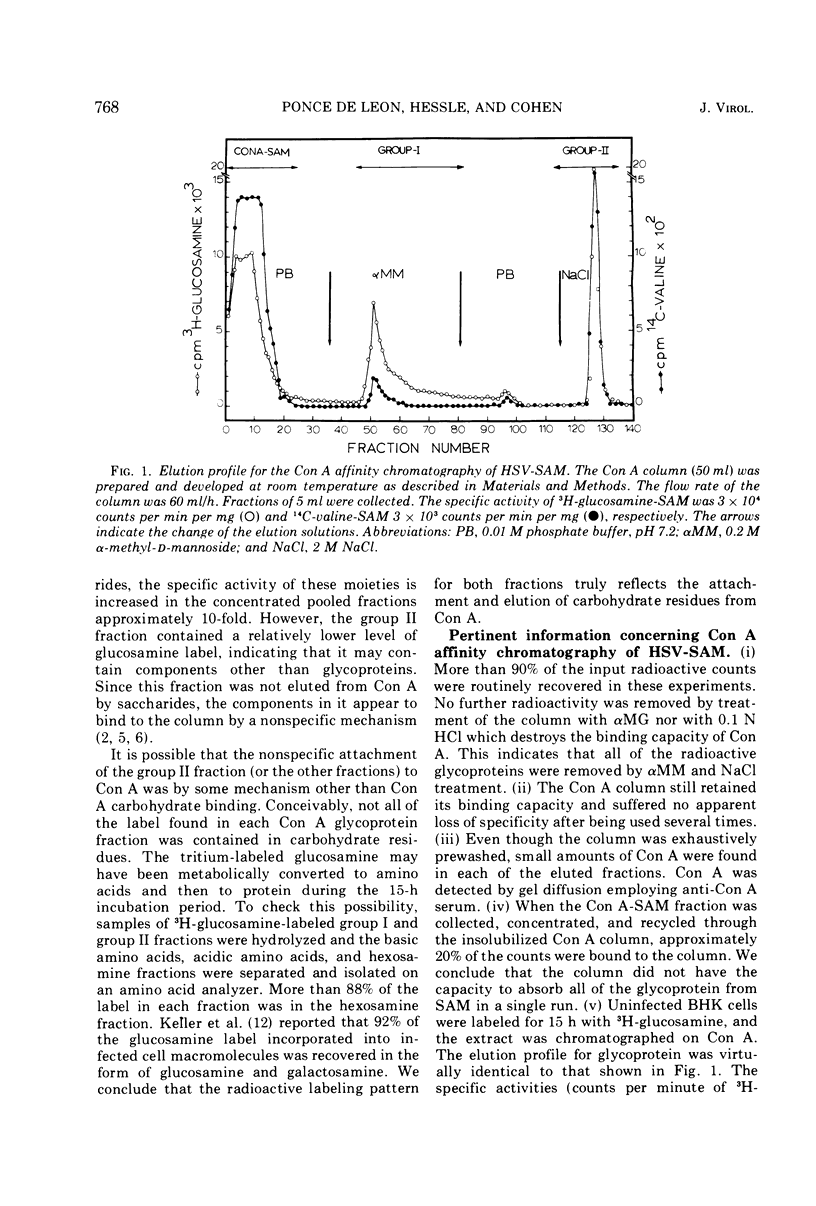
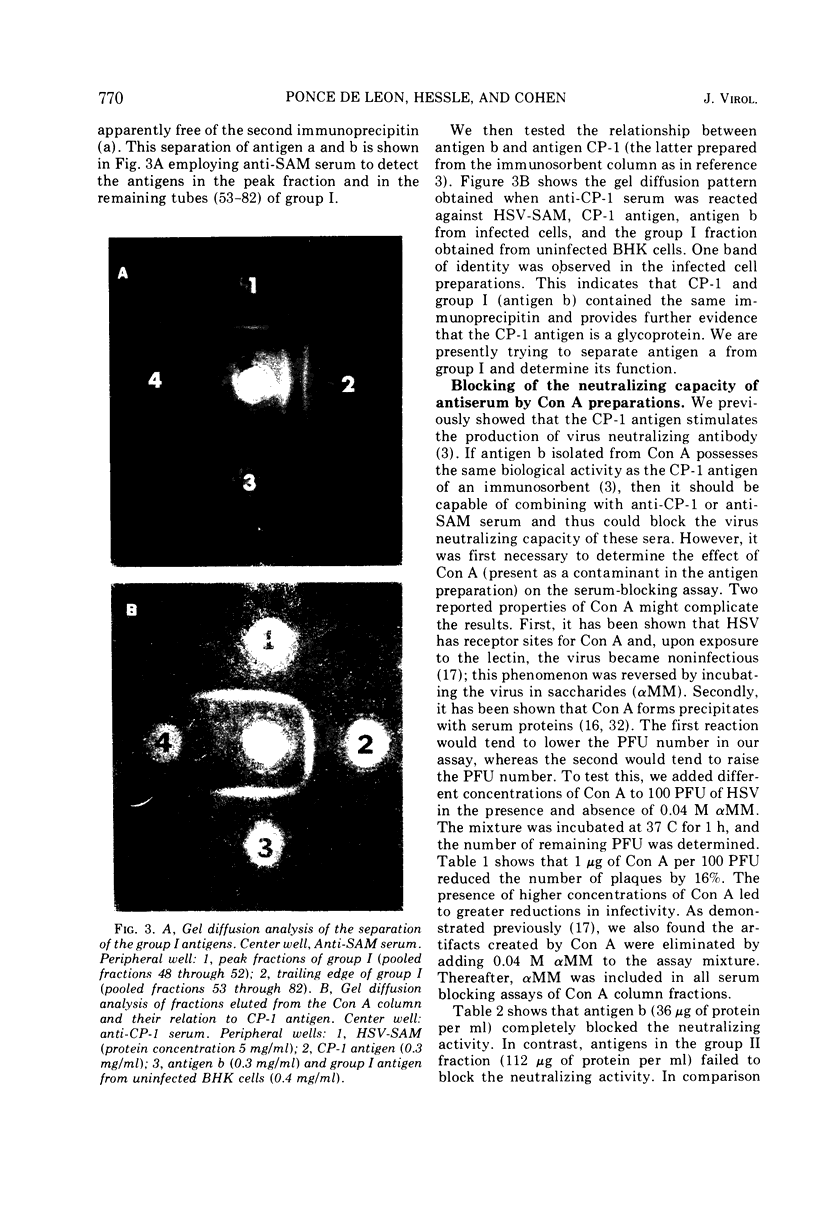
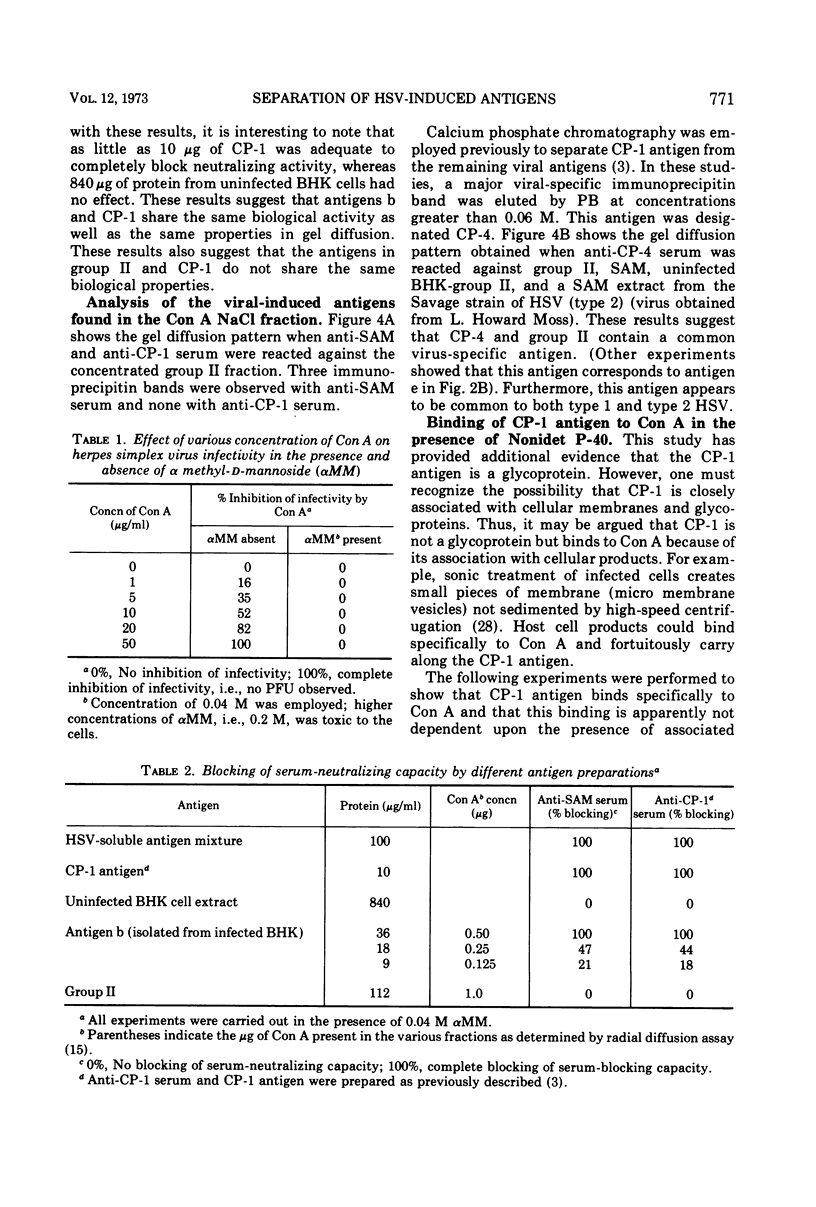
Biologically active herpes simplex virus (HSV)-induced antigens were selectively removed from extracts of infected BHK cells by affinity chromatography by utilizing an insoluble form of concanavalin A (Con A). Soluble extracts of 3H-glucosamine-labeled, HSV-infected cells were absorbed to a Con A column. Bound material was eluted with α-methyl-d-mannoside (αMM) and NaCl. The specific activity of the eluted glycoproteins increased by 10-fold. Two broad groups of viral-induced antigens were isolated from Con A. Group I includes two antigens which bind to Con A by a specific mechanism because the antigens are dissociated by αMM. Group II contains three antigens which bind to Con A but apparently by a nonspecific or electrolytic mechanism. One antigen in group I was identified as the glycoprotein antigen, CP-1, described previously.
Full text
PDF




Images in this article
Selected References
These references are in PubMed. This may not be the complete list of references from this article.
- APPLEYARD G., ZWARTOUW H. T., WESTWOOD J. C. A PROTECTIVE ANTIGEN FROM THE POX-VIRUSES. I. REACTION WITH NEUTRALIZING ANTIBODY. Br J Exp Pathol. 1964 Apr;45:150–161. [PMC free article] [PubMed] [Google Scholar]
- Cohen G. H., Ponce de Leon M., Nichols C. Isolation of a herpes simplex virus-specific antigenic fraction which stimulates the production of neutralizing antibody. J Virol. 1972 Nov;10(5):1021–1030. doi: 10.1128/jvi.10.5.1021-1030.1972. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Doyle R. J., Kan T. -J. Interaction between concanavalin A and heparin. FEBS Lett. 1972 Jan 15;20(1):22–24. doi: 10.1016/0014-5793(72)80007-3. [DOI] [PubMed] [Google Scholar]
- Doyle R. J., Woodside E. E., Fishel C. W. Protein-polyelectrolyte interactions. The concanavalin A precipitin reaction with polyelectrolytes and polysaccharide derivatives. Biochem J. 1968 Jan;106(1):35–40. doi: 10.1042/bj1060035. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Dufau M. L., Tsuruhara T., Catt K. J. Interaction of glycoprotein hormones with agarose-concanavalin A. Biochim Biophys Acta. 1972 Sep 29;278(2):281–292. doi: 10.1016/0005-2795(72)90233-4. [DOI] [PubMed] [Google Scholar]
- GOLDSTEIN I. J., HOLLERMAN C. E., SMITH E. E. PROTEIN-CARBOHYDRATE INTERACTION. II. INHIBITION STUDIES ON THE INTERACTION OF CONCANAVALIN A WITH POLYSACCHARIDES. Biochemistry. 1965 May;4:876–883. doi: 10.1021/bi00881a013. [DOI] [PubMed] [Google Scholar]
- Goldstein I. J., So L. L. Protein-carbonhydrate interaction. 3. Agar gel-diffusion studies on the interaction of Concanavalin A, a lectin isolated from jack bean, with polysaccharides. Arch Biochem Biophys. 1965 Aug;111(2):407–414. doi: 10.1016/0003-9861(65)90203-1. [DOI] [PubMed] [Google Scholar]
- Hayman M. J., Skehel J. J., Crumpton M. J. Purification of virus glycoproteins by affinity chromatography using Lens culinaris phytohaemagglutinin. FEBS Lett. 1973 Jan 15;29(2):185–188. doi: 10.1016/0014-5793(73)80557-5. [DOI] [PubMed] [Google Scholar]
- Helenius A., Simons K. Removal of lipids from human plasma low-density lipoprotein by detergents. Biochemistry. 1971 Jun 22;10(13):2542–2547. doi: 10.1021/bi00789a019. [DOI] [PubMed] [Google Scholar]
- Keller J. M., Spear P. G., Roizman B. Proteins specified by herpes simplex virus. 3. Viruses differing in their effects on the social behavior of infected cells specify different membrane glycoproteins. Proc Natl Acad Sci U S A. 1970 Apr;65(4):865–871. doi: 10.1073/pnas.65.4.865. [DOI] [PMC free article] [PubMed] [Google Scholar]
- LOWRY O. H., ROSEBROUGH N. J., FARR A. L., RANDALL R. J. Protein measurement with the Folin phenol reagent. J Biol Chem. 1951 Nov;193(1):265–275. [PubMed] [Google Scholar]
- Lloyd K. O. The preparation of two insoluble forms of the phytohemagglutinin, concanavalin A, and their interactions with polysaccharides and glycoproteins. Arch Biochem Biophys. 1970 Apr;137(2):460–468. doi: 10.1016/0003-9861(70)90463-7. [DOI] [PubMed] [Google Scholar]
- Mancini G., Carbonara A. O., Heremans J. F. Immunochemical quantitation of antigens by single radial immunodiffusion. Immunochemistry. 1965 Sep;2(3):235–254. doi: 10.1016/0019-2791(65)90004-2. [DOI] [PubMed] [Google Scholar]
- NAKAMURA S., TANAKA K., MURAKAWA S. Specific protein of legumes which reacts with animal proteins. Nature. 1960 Oct 8;188:144–145. doi: 10.1038/188144b0. [DOI] [PubMed] [Google Scholar]
- Okada Y., Kim J. Interaction of concanavalin A with enveloped viruses and host cells. Virology. 1972 Nov;50(2):507–515. doi: 10.1016/0042-6822(72)90401-1. [DOI] [PubMed] [Google Scholar]
- Olshevsky U., Becker Y. Herpes simplex virus structural proteins. Virology. 1970 Apr;40(4):948–960. doi: 10.1016/0042-6822(70)90141-8. [DOI] [PubMed] [Google Scholar]
- Olshevsky U., Becker Y. Surface glycopeptides in the envelope of herpes simplex virions. Virology. 1972 Oct;50(1):277–279. doi: 10.1016/0042-6822(72)90371-6. [DOI] [PubMed] [Google Scholar]
- Poretz R. D., Goldstein I. J. An examination of the topography of the saccharide binding sites of concanavalin A and of the forces involved in complexation. Biochemistry. 1970 Jul 7;9(14):2890–2896. doi: 10.1021/bi00816a021. [DOI] [PubMed] [Google Scholar]
- Sharon N., Lis H. Lectins: cell-agglutinating and sugar-specific proteins. Science. 1972 Sep 15;177(4053):949–959. doi: 10.1126/science.177.4053.949. [DOI] [PubMed] [Google Scholar]
- So L. L., Goldstein I. J. Protein-carbohydrate interaction. 13. The interaction of concanavalin A with alpha-mannans from a variety of microorganisms. J Biol Chem. 1968 Apr 25;243(8):2003–2007. [PubMed] [Google Scholar]
- So L. L., Goldstein I. J. Protein-carbohydrate interaction. IV. Application of the quantitative precipitin method to polysaccharide-concanavalin A interaction. J Biol Chem. 1967 Apr 10;242(7):1617–1622. [PubMed] [Google Scholar]
- Spear P. G., Roizman B. Proteins specified by herpes simplex virus. V. Purification and structural proteins of the herpesvirion. J Virol. 1972 Jan;9(1):143–159. doi: 10.1128/jvi.9.1.143-159.1972. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Sumner J. B., Howell S. F. Identification of Hemagglutinin of Jack Bean with Concanavalin A. J Bacteriol. 1936 Aug;32(2):227–237. doi: 10.1128/jb.32.2.227-237.1936. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Tevethia S. S., Lowry S., Rawls W. E., Melnick J. L., McMillan V. Detection of early cell surface changes in herpes simplex virus infected cells by agglutination with concanavalin A. J Gen Virol. 1972 Apr;15(1):93–97. doi: 10.1099/0022-1317-15-1-93. [DOI] [PubMed] [Google Scholar]
- Watson D. H., Wildy P. The preparation of 'monoprecipitin' antisera to herpes virus specific antigens. J Gen Virol. 1969 Mar;4(2):163–168. doi: 10.1099/0022-1317-4-2-163. [DOI] [PubMed] [Google Scholar]
- Weinstein Y., Givol D., Strausbauch P. H. The fractionation of immunoglobulins with insolubilized concanavalin A. J Immunol. 1972 Dec;109(6):1402–1404. [PubMed] [Google Scholar]