Abstract
EMBO J (2013) 32, 324–339 doi:; DOI: 10.1038/emboj.2012.341; published online December 21 2012
The role of lysosomes in important cellular responses, including phagocytosis, cell surface repair, and autophagy underlies a number of human diseases. Furthermore, the role of the lysosomal surface in TORC1 signalling has revealed unexpected properties of these organelles. In this issue, Sridhar et al (2013) uncover an important role for PI(4)P for lysosome function under normal nutrient conditions and after prolonged nutrient deprivation. Ana Maria Cuervo, the late Dennis Shields, and colleagues (Sridhar et al, 2013) conclude that PI4 kinase IIIβ on the surface of the lysosome controls the fidelity of sorting from the lysosome, and is required for autophagic lysosome reformation (ALR). These novel findings provide important insights into the complexities of the lipid composition of the lysosome, and how these lipids may control lysosome function.
Lysosomes, traditionally thought of as inert, terminal endocytic compartments, are in fact dynamic organelles. In addition to their function in degradation of cargo ranging from small vesicles to whole phagosomes and autophagosomes, they respond dynamically to extracellular ion fluxes, and they can generate potent intracellular calcium (and other ionic) signals (Saftig and Klumperman, 2009; Morgan et al, 2011; Shen et al, 2011). The cytoplasmic surface of the lysosome mediates the delivery of cargo-containing vesicles and organelles via SNAREs and tethers, but is also a platform for TORC1 signalling and transcriptional regulation by TFEB (Settembre et al, 2011; Efeyan et al, 2012). The membrane of the lysosome, protected from the degradative enzymes contained in the lumen by highly glycosylated lysosomal membrane proteins (LAMPs, LIMPs, etc.), contains the machinery for its acidification (V-ATPase), as well as a variety of transporters and channels for ions and amino acids. LAMP2A also serves as the receptor for chaperone-mediated autophagy (CMA) that together with the chaperone protein Hsc70 delivers individual proteins into the lysosome (Orenstein and Cuervo, 2010). A key feature of lysosomes is their ability to reform (Luzio et al, 2007; Yu et al, 2010) and noteworthy here is the enhancement of reformation that occurs during prolonged nutrient deprivation described as ALR (Yu et al, 2010).
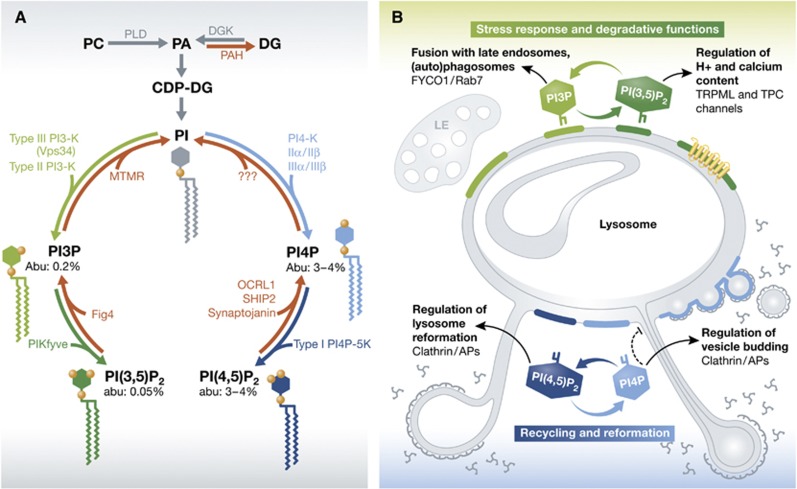
Many of these functions are thought to be dependent on PI(3,5)P2 (phosphatidylinositol (PI)-3,5-bis phosphate), and the cycle of PI-3 phosphate (PI3P) to PI(3,5)P2 (Figure 1A; Michell et al, 2006; Ho et al, 2012). In broad terms, PI3P is required during the fusion of vesicles/(auto)phagosomes with lysosomes, whereas PI(3,5)P2 appears to regulate ion channels that maintain acidification and calcium content optimal for digestive functions (Figure 1B). Indeed, deficiency of PI(3,5)P2 caused by loss of lipid-modifying enzymes (both kinases and phosphatases) is the cause of several human neuropathologies (Ho et al, 2012), and at the cellular level, deficiency in the levels of either PI(3)P or PI(3,5)P2 leads to swelling and loss of lysosomal function (Michell et al, 2006; Ho et al, 2012). This phenotype underscores the fact that defects in lipid-modifying enzymes do not always produce clearly distinct effects because, in addition to lipid inter-conversion (see Figure 1A), deficiencies of one lipid species may feedback to other enzymes, leading to accumulation of unexpected lipids.
Figure 1.
Two phosphoinositide cycles regulate aspects of lysosome function. (A) Pathway of synthesis and consumption of PI3P, PI(3,5)P2, PI(4)P and PI(4,5)P2 from PA via PI. The relative abundance of the various lipid species is shown, alongside the main enzymes responsible for their synthesis and consumption. (B) Stress responses of lysosomes following extracellular or intracellular challenge are regulated in part by PI3P and PI(3,5)P2, with the former involved in fusion with other organelles and the latter in regulation of ion channels (shown in green). A second phosphoinositide cycle dependent on PI(4)P and PI(4,5)P2 was uncovered in the paper by Sridhar et al (2013), which regulates traffic from the lysosomes either in the form of vesicles or tubules (shown in blue, PI(4)P favouring vesicular intermediates). Recent work by Yu and colleagues has also suggested that these lipids regulate lysosome reformation during enhanced flux, also thought to proceed via a tubular intermediate favouring PI(4,5)P2 (shown in blue).
While PI(3,5)P2 is present at very low levels (Figure 1A) it has a well-documented role in lysosome function (Figure 1B). Thus, identification of a role for a PI-4 kinase (PI4KIIIβ), and the more abundant lipids PI(4)P and PI(4,5)P2, in the regulation of lysosome function, in particular vesicle-mediated exit from the lysosome, is surprising and unexpected (Sridhar et al (2013), this issue; Rong et al (2012)). PI(4)P, which is produced in cells by four different enzymes (Figure 1A), plays an important role at the plasma membrane and the trans-Golgi network (TGN), and out of these four enzymes PI4KIIIβ is most important for trafficking out of the TGN (Santiago-Tirado and Bretscher, 2011). Cuervo and colleagues (Sridhar et al, 2013) now show that loss of PI4KIIIβ causes constitutive formation of tubules from lysosomes and loss of retention of lysosomal cargo (Figure 1B). Dynamic LAMP1-positive tubules emanate from lysosomes in the absence of PI4KIIIβ which contain lysosomal content such as cathepsin D, a number of clathrin-coat components, including adaptor protein (AP)-2, kinesin motor Kif13b, and Rab9 (a small GTPase implicated in traffic to late endosomes and lysosome). Sridhar et al (2013) demonstrate the presence of these molecules, some of which are the core machinery for vesicle formation, using elegant fractionation approaches combined with cryoimmunogold labelling and live-cell imaging. Furthermore, preliminary data from two-dimensional electrophoresis suggest that a subpopulation of PI4KIIIβ may undergo a post-translational modification, and it is this subpopulation which is recruited to the lysosome.
Importantly, this work relates to a recent paper from Rong et al (2012), which demonstrates a role for PI(4,5)P2 in ALR, supporting the notion that PI4P-PI(4,5)P2 cycling regulates traffic out of lysosomes and reformation during enhanced flux. Rong et al (2012) used similar approaches including isolation and proteomic analysis of the tubules found during ALR to identify the components required for ALR, which includes clathrin and AP-2, AP-4 and PI(4)P5K1B and 1A. Rong et al (2012) suggested that PI(4)P5K1B was required during ALR for tubule formation, while PI(4)K1A was required for tubule extension and/or fission.
Sridhar et al (2013) recapitulate these findings and show, using LAMP1-RFP and fluorescent pepstatin A (which labels cathespin D) labelling, that loss of PI4KIIIβ results in inefficient sorting in particular into the tubules produced by loss of PI(4)P5K1A. It is proposed that PI(4)P levels control ALR; loss of PI4P causes constitutive formation of tubules and loss of retention of lysosomal cargo, and is required for ALR (Figure 1B). Cuervo and colleagues hypothesize that PI4P is not just a substrate for PI(4)P5Ks (the scenario proposed by Rong et al (2012)) but that it is in fact a critical molecule for determining the balance between tubulation and vesicular transport out of the lysosome during ALR and in normal conditions.
The work of Cuervo and her colleagues following on from Rong et al (2012) shows a masterful control of techniques needed to identify the machinery needed for ALR and efflux from the lysosome. Importantly, both papers looked at the lipid species present using lipid probes, and showed that kinase dead mutants of the kinases do not rescue. However, neither paper actually measured changes in lipid levels as a result of their manipulations. As pointed out above, this may change the interpretation. Some lipid effectors were identified, but no lipid binding mutants of effectors were employed, and no lipid phosphatases were used or identified. These unexplored issues leave open the possibility that further complex regulation is yet to be discovered which may involve effector proteins, and proteins that activate or inhibit the kinases. In summary, Cuervo and her team have provided a clue to how ALR may be mediated by PI(4)P, which combined with data provided by Rong et al (2012), clearly implicates a cycle of PI4P and PI4,5P2 synthesis and hydrolysis in the trafficking out of lysosomes and ALR. Further exciting developments in understanding the dynamic nature of the lysosome have the potential to truly advance our knowledge of cellular homeostasis in both health and disease.
Acknowledgments
NTK is supported by the Biotechnology and Biological Sciences Research Council. SAT is supported by Cancer Research UK.
Footnotes
The authors declare that they have no conflict of interest.
References
- Efeyan A, Zoncu R, Sabatini DM (2012) Amino acids and mTORC1: from lysosomes to disease. Trends Mol Med 18: 524–533 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Ho CY, Alghamdi TA, Botelho RJ (2012) Phosphatidylinositol-3,5-bisphosphate: no longer the poor PIP2. Traffic 13: 1–8 [DOI] [PubMed] [Google Scholar]
- Luzio JP, Pryor PR, Bright NA (2007) Lysosomes: fusion and function. Nat Rev Mol Cell Biol 8: 622–632 [DOI] [PubMed] [Google Scholar]
- Michell RH, Heath VL, Lemmon MA, Dove SK (2006) Phosphatidylinositol 3,5-bisphosphate: metabolism and cellular functions. Trends Biochem Sci 31: 52–63 [DOI] [PubMed] [Google Scholar]
- Morgan AJ, Platt FM, Lloyd-Evans E, Galione A (2011) Molecular mechanisms of endolysosomal Ca2+ signalling in health and disease. Biochem J 439: 349–374 [DOI] [PubMed] [Google Scholar]
- Orenstein SJ, Cuervo AM (2010) Chaperone-mediated autophagy: Molecular mechanisms and physiological relevance. Semin Cell Dev Biol 21: 719–726 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Rong Y, Liu M, Ma L, Du W, Zhang H, Tian Y, Cao Z, Li Y, Ren H, Zhang C, Li L, Chen S, Xi J, Yu L (2012) Clathrin and phosphatidylinositol-4,5-bisphosphate regulate autophagic lysosome reformation. Nat Cell Biol 14: 924–934 [DOI] [PubMed] [Google Scholar]
- Saftig P, Klumperman J (2009) Lysosome biogenesis and lysosomal membrane proteins: trafficking meets function. Nat Rev Mol Cell Biol 10: 623–635 [DOI] [PubMed] [Google Scholar]
- Santiago-Tirado FH, Bretscher A (2011) Membrane-trafficking sorting hubs: cooperation between PI4P and small GTPases at the trans-Golgi network. Trends Cell Biol 21: 515–525 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Settembre C, Di Malta C, Polito VA, Garcia Arencibia M, Vetrini F, Erdin S, Erdin SU, Huynh T, Medina D, Colella P, Sardiello M, Rubinsztein DC, Ballabio A (2011) TFEB links autophagy to lysosomal biogenesis. Science 332: 1429–1433 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Shen D, Wang X, Xu H (2011) Pairing phosphoinositides with calcium ions in endolysosomal dynamics: phosphoinositides control the direction and specificity of membrane trafficking by regulating the activity of calcium channels in the endolysosomes. BioEssays 33: 448–457 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Sridhar S, Patel B, Aphkhazava D, Macian F, Santambrogio L, Shields D, Cuervo AM (2013) The lipid kinase PI4KIIIβ preserves lysosomal identity. EMBO J 32: 324–339 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Yu L, McPhee CK, Zheng L, Mardones GA, Rong Y, Peng J, Mi N, Zhao Y, Liu Z, Wan F, Hailey DW, Oorschot V, Klumperman J, Baehrecke EH, Lenardo MJ (2010) Termination of autophagy and reformation of lysosomes regulated by mTOR. Nature 465: 942–946 [DOI] [PMC free article] [PubMed] [Google Scholar]