Abstract
Dysphagia lusoria occurs secondary to an aberrant right subclavian artery coursing posterior to the oesophagus. Open ligation and transposition to the right carotid artery via a right supraclavicular approach has been described as a minimally invasive method. However, approaching the origin of the aberrant right subclavian artery through this incision can be extremely challenging. A persistent aberrant right subclavian artery stump may account for postoperative residual dysphagia. This article describes a safe, effective and reproducible surgical approach to dysphagia lusoria due to a non-aneurysmal aberrant right subclavian artery.
Keywords: Dysphagia lusoria, Aberrant right subclavian artery, Mediastinoscopy
INTRODUCTION
Dysphagia lusoria occurs secondary to an aberrant right sub-clavian artery (ARSA) coursing posterior to the oesophagus. The anomaly most commonly involves the right subclavian artery (0.5–2.0% of the population), or rarely the left subclavian artery when there is a right-sided aortic arch (0.05–0.1%) [1]. The majority of patients with ARSA remain asymptomatic. Therefore, dysphagia lusoria is exceedingly rare, and a clinician may not encounter even one case during his or her entire career.
A variety of surgical approaches and techniques have been described in the literature; however, because of its rarity, there is no consensus on the best surgical approach to this entity. Satisfactory exposure and ligation of the origin of the ARSA are the most crucial steps for successful surgical treatment. Historically, this process has been a challenge for surgeons and has required invasive or two-staged approaches. In addition, it has often been associated with serious morbidities and even mortalities. We herein describe a safer, more simplified and less invasive one-stage open technique utilizing a mediastinoscope, which allows better exposure and ligation at its origin through a right supraclavicular incision.
CASE REPORT
A previously healthy 48-year old female developed progressively worsening dysphagia for 3 months. She underwent an extensive work-up that included oesophagogastroduodenoscopy, a barium swallow and computed tomography scan of the chest, revealing an ARSA with a retro-oesophageal course compressing the oesophagus (Fig. 1A and B). This finding was substantiated by a 15-kg weight loss over 3 months. The ARSA was patent and had no aneurysmal changes. Her symptoms did not subside despite aggressive medical management, including antacids and dietary modifications. A decision was made to proceed with surgical intervention.
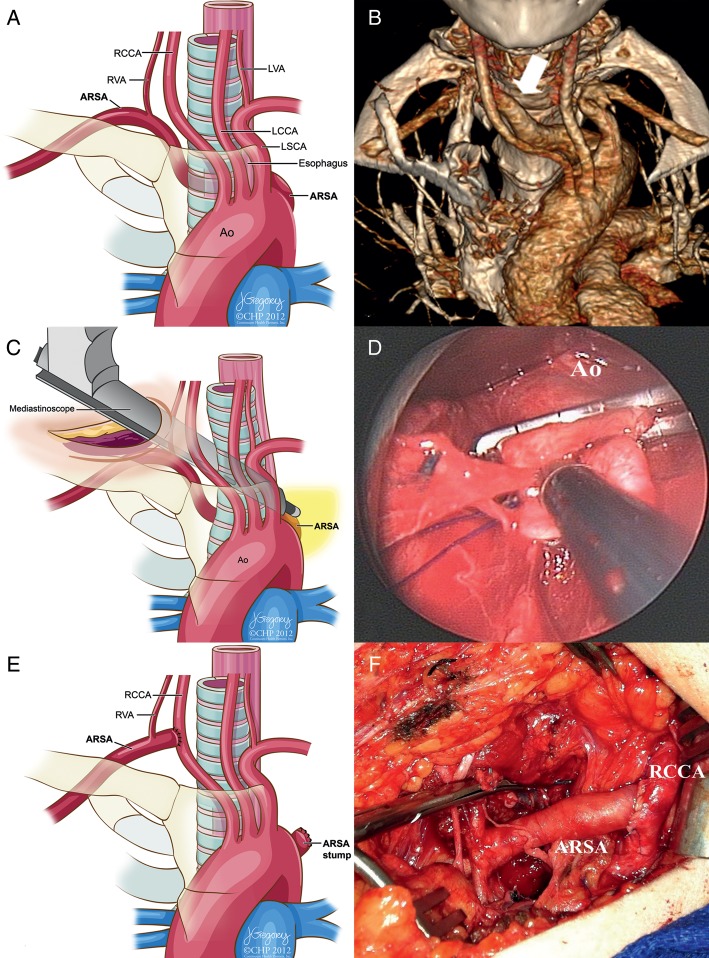
Figure 1:
(A) Illustration of the ARSA. The ARSA is coursing posteriorly behind the oesophagus. ARSA: aberrant right subclavian artery; RCCA: right common carotid artery; LCCA: left common carotid artery; LSCA: left subclavian artery, RVA: right vertebral artery; LVA: left vertebral artery; Ao: Aorta. (B) Preoperative 3D computed tomography reconstruction (anteroposterior view with cranial angulation) showing the origin of the ARSA from the posterior wall of the aortic arch (arrow). (C) Operative illustration showing the mediastinoscope-assisted dissection using the suction cautery device. (D) Operative image from the mediastinoscope demonstrating the aortic wall and the ARSA stump. This plane was created by the use of mediastinoscopic blunt dissection. (E and F) The ARSA was ligated at its origin and reimplanted to the RCCA.
The patient was placed in the supine position under general anaesthesia with a shoulder pillow at the midline for neck extension. A 56-French bougie was inserted to guide oesophageal identification during mobilization of the ARSA. A transverse right supraclavicular incision was made. The clavicular head of the sternocleidomastoid muscle was transected. The phrenic nerve was dissected free from the anterior scalene, which was also divided for further exposure of the ARSA. The common carotid artery and vagal nerve were also identified. The oesophagus was gently retracted anteriorly. A mediastinoscope was inserted to the level of the ARSA (Fig. 1C). Once the origin of the ARSA and aortic arch were dissected using suction-cautery-assisted blunt dissection under mediastinoscopy, systemic heparin was administered. The exposed origin of the ARSA was clamped 1 cm distal to the aortic arch using a small Cooley–Satinsky clamp under mediastinoscopy. Adequate retraction of the oesophagus and trachea was performed, which provided sufficient working space for achieving vascular control under direct vision. The stump of the vessel was oversewn with 3-0 prolene (Fig. 1D), and arterial continuity was restored by transposition of the ARSA into the ipsilateral common carotid artery (Fig. 1E and F). The patient tolerated the procedure without any complications and was neurologically intact, moving her extremities with good bilateral radial and ulnar pulses. Her postoperative course was uneventful and she tolerated regular diet without any difficulties.
DISCUSSION
ARSA is hypothesized to be an abnormal involution or absence of the fourth vascular arch with the right dorsal aorta. The seventh intersegmental artery remains attached to the descending aorta, and this persistent intersegmental artery becomes the right subclavian artery. This leads to an aberrant artery, which often courses posterior to the oesophagus [2]. There are a few suggested mechanisms that cause symptoms: (1) increased oesophageal or vessel wall rigidity with aging; (2) aneurysm formation, especially in the presence of Kommerell's diverticulum; (3) aortic elongation with aging and (4) the presence of a bicarotid trunk [3]. The combination of (1) and (3) may have been the contributing factors in the present case despite the normal ARSA calibre. Although a variety of surgical approaches have been proposed (including thoracotomy, sternotomy, cervical and supraclavicular approach) with many different vascular reconstruction methods [4], each approach has its specific limitations and potential pitfalls. The endovascular approach is the emerging treatment of choice with promising short-term outcomes [5]. On the contrary, long-term outcomes are still unknown. The occlusion device utilized in the endovascular approach may itself cause vascular compression of the oesophagus and could be a source of persistent symptoms; in addition, there is a risk of device migration. Furthermore, this technique may necessitate the use of a prosthetic graft, which carries a risk of future graft infection or thrombosis.
Open ligation and transposition to the right carotid artery via a right supraclavicular approach has been described as having the lowest associated morbidity, while it entails the potential complication of residual dysphagia in cases in which a long stump is left. The key to the success of this procedure is to gain access beyond the left margin of the oesophagus for complete resection of the retro-oesophageal portion of the ARSA. Otherwise, the patient may be at risk of recurrent symptoms in the future. However, oversewing of the subclavian artery stump to the left of the oesophagus through the supraclavicular incision can be extremely challenging because of the limited exposure and the depth of the target vessel. The use of small malleable vascular stapling devices for this type of procedure would be an option although currently available commercial devices were not applicable to the present case.
In our case, optimal exposure of the origin of the aberrant artery was achieved by utilizing a mediastinoscope and its suction cautery device, which allowed not only optimal visualization and secured ligation at its origin, but also safer and bloodless dissection to the target. The vertebrobasilar system was also safely preserved. Reimplantation to the carotid artery was the only required vascular anastomosis in this approach. Theoretically, the same approach can be applied for patients with right-sided aortic arch with an aberrant left subclavian artery if the anatomy permits. In contrast, this approach is not recommended in patients with Kommerell's diverticulum because of suboptimal vascular control of the aortic arch.
Conflict of interest: none declared.
REFERENCES
- 1.Ota T, Okada K, Takanashi S, Yamamoto S, Okita Y. Surgical treatment for Kommerell's diverticulum. J Thorac Cardiovasc Surg. 2006;131:574–8. doi: 10.1016/j.jtcvs.2005.10.012. doi:10.1016/j.jtcvs.2005.10.012. [DOI] [PubMed] [Google Scholar]
- 2.Edwards JE. Congenital malformations of the heart and great vessels. Section H. Malformations of the thoracic aorta. In: Gould SE, editor. Pathology of the Heart. 2nd edn. Springfield, IL: Charles C. Thomas; 1960. pp. 391–462. [Google Scholar]
- 3.Janssen M, Baggen MG, Veen HF, Smout AJ, Bekkers JA, Jonkman JG, et al. Dysphagia lusoria: clinical aspects, manometric findings, diagnosis, and therapy. Am J Gastroenterol. 2000;95:1411–6. doi: 10.1111/j.1572-0241.2000.02071.x. doi:10.1111/j.1572-0241.2000.02071.x. [DOI] [PubMed] [Google Scholar]
- 4.Rogers AD, Nel M, Eloff EP, Naidoo NG. Dysphagia lusoria: a case of an aberrant right subclavian artery and a bicarotidtrunk. ISRN Surg. 2011;2011:819295. doi: 10.5402/2011/819295. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 5.Stone WM, Ricotta JJ, 2nd, Fowl RJ, Garg N, Bower TC, Money SR. Contemporary management of aberrant right subclavian arteries. Ann Vasc Surg. 2011;25:508. doi: 10.1016/j.avsg.2011.02.012. doi:10.1016/j.avsg.2011.02.012. [DOI] [PubMed] [Google Scholar]