Abstract
Dietary cholesterol regulation of cholesterol 7α-hydroxylase (Cyp7a1), the rate-limiting enzyme in the classical pathway of bile acid synthesis, has been implicated in plasma cholesterol responsiveness. In the current study, the effects of 0.0% and 0.5% cholesterol diets were examined in Cyp7a1 knockout (KO), heterozygous Cyp7a1 KO (Het), and human Cyp7a1 transgenic mice on the mouse Cyp7a1 KO background (Tg+KO). We confirmed previous findings that dietary cholesterol increased mouse Cyp7a1 activity in Het mice but decreased human Cyp7a1 activity in Tg+KO mice. However, in both Het and Tg+KO mice, dietary cholesterol increased bile acid pool size (36% and 72%, respectively) and fecal bile acid excretion (2.2- and 3.6-fold, respectively). The expression of cholesterol 27-hydroxylase (Cyp27), the major enzyme of the alternative pathway of bile acid synthesis, was not significantly different in cholesterol-fed KO, Het, or Tg+KO mice. Furthermore, dietary cholesterol had comparable effects on total plasma cholesterol and non-high-density lipoprotein cholesterol in KO, Het, and Tg+KO mice. Thus, in Tg+KO mice, dietary cholesterol regulates bile acid pool size, fecal bile acid excretion, and plasma cholesterol independently of Cyp7a1 activity. These results challenge the notion that dietary cholesterol regulation of Cyp7a1 is a major determinant of plasma cholesterol responsiveness.
Dietary cholesterol is absorbed in the proximal small intestine and transported to the liver. In the liver, dietary cholesterol regulates the expression of many genes, including some that influence plasma cholesterol levels (1). Differences in regulation of some of these genes could account for the variation in plasma cholesterol response to dietary cholesterol between species or between individual humans. A candidate gene for this role is cholesterol 7α-hydroxylase (Cyp7a1), a liver enzyme that governs the rate-limiting step in the classical pathway of bile acid synthesis (2). In the liver, cholesterol is converted to bile acids, thus removing it from the active cholesterol pool and resulting in up-regulation of hepatic low-density lipoprotein receptors and decreased plasma cholesterol levels. In addition, bile acids are excreted into the small intestine, where they act as detergents to facilitate the absorption of dietary cholesterol, lipids, and fat-soluble vitamins. Most of the bile acids are reabsorbed in the distal ileum and returned to the liver, but in each cycle, ≈5% are excreted into the feces (3). Thus, conversion to bile acids represents a major pathway whereby cholesterol can be removed from the body, which may also influence plasma cholesterol levels.
Mangelsdorf's laboratory (4) has shown that dietary cholesterol can up-regulate mouse and rat Cyp7a1 through activation of the liver X receptor (LXR) transcription factor, which binds to an LXR response element in the Cyp7a1 promoter. The human Cyp7a1 gene contains a mutated LXR response element, and it has been hypothesized that the failure of cholesterol to up-regulate this gene may account for the relatively greater response of plasma cholesterol to dietary cholesterol in humans compared to mice and rats (5–7). One possibility is that this greater response is due to differential dietary cholesterol regulation of Cyp7a1. To examine this question, two recent studies were performed that fed dietary cholesterol to wild-type and human Cyp7a1 transgenic mice on the mouse Cyp7a1 knockout (KO) background. In both studies, wild-type mice responded to dietary cholesterol by up-regulating Cyp7a1 (5, 8). In one study, dietary cholesterol did not up-regulate the human transgene (8); whereas, in the other study, the transgene was down-regulated (5). In both studies the authors suggested that humans are more responsive to dietary cholesterol than mice, because they cannot up-regulate Cyp7a1 activity in response to dietary cholesterol. However, if this were true, then one would expect dietary cholesterol to increase plasma cholesterol levels in human Cyp7a1 transgenic mice on the mouse Cyp7a1 KO background. Yet, in the one study that made this measurement, 2% dietary cholesterol failed to increase plasma cholesterol in these animals or in wild-type mice (8). This brings into question the relevance of dietary cholesterol regulation of Cyp7a1 in plasma cholesterol responsiveness and suggests the existence of other important regulatory processes.
In the current study, the effect of increasing dietary cholesterol from 0.0% and 0.5% was compared between Cyp7a1 KO mice, Cyp7a1 heterozygous (Het) mice, and Cyp7a1 transgenic mice on the KO background (Tg+KO) with regard to Cyp7a1 mRNA and activity levels, bile acid pool size, fecal bile acid excretion, and plasma lipoprotein cholesterol levels. The difference in Cyp7a1 regulation between the human transgene and the mouse gene in response to dietary cholesterol was confirmed. However, in mice expressing either mouse or human Cyp7a1, there were comparable effects of dietary cholesterol on bile acid pool size, fecal bile acid excretion, and plasma lipoprotein cholesterol levels. This similarity suggests that dietary cholesterol regulation of genes other than Cyp7a1 is responsible for these phenomena.
Methods
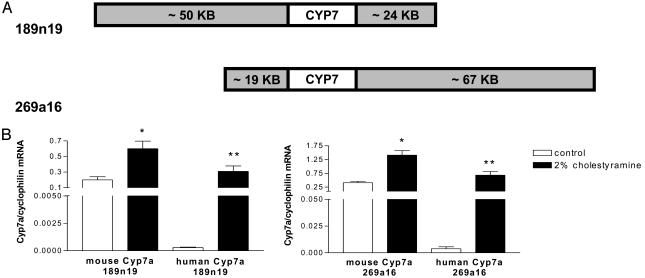
Mice. A human bacterial artificial chromosome (BAC) library (catalog no. BAC-4434; Genome Systems, St. Louis) was screened, and two BACs containing the human Cyp7a1 gene, each with different lengths of upstream and downstream flanking sequence, were identified and mapped (Fig. 1A). BAC-189n19 contained 50 kb of DNA 5′ and 24 kb 3′ to the gene; whereas BAC-269a16 contained 19 kb of DNA 5′ and 67 kb 3′ to the gene. Transgenic mice were produced by microinjecting these BACs into C57BL/6J × CBA/CaJ zygotes. Mice integrating human Cyp7a1 DNA into their genome were identified by PCR from tail DNA by using primers that are specific for the human Cyp7a1 gene (sense, 5′-GTTCTTCGGGTGTGATCATATATC-3′; antisense, 5′TTCCAAATGGCTATTTCCATCCAG-3′). One transgenic line was established for BAC-189n19 and four for BAC-269a16. Each line was backcrossed 10 times to C57BL/6J. In the current study, only heterozygous transgenic mice from the BAC-189n19 line and one of the BAC-269a16 lines were used. Outbred Cyp7a1 KO mice were obtained from D. Russell and backcrossed at least 10 generations to the C57BL/6J background. In contrast to outbred mice described previously (9), C57BL/6J Cyp7a1 KO mice were able to survive without supplementing their or their mothers' diets with vitamins or cholic acid. To generate human Cyp7a1 transgenic mice on the KO background, the Cyp7a1 transgenic line 189n19 was crossed with KO mice, resulting in human Cyp7a1 transgenics on the heterozygous Cyp7a1 KO background. These mice were then crossed with KO mice to obtain mice that were heterozygous for the human Cyp7a1 transgene and homozygous KO for the mouse Cyp7a1 gene. KO and Het littermates were used in experiments as controls. Animals were housed in a humidity- and temperature-controlled room with a 12-h dark/12-h light cycle at the Laboratory Animal Research Center at The Rockefeller University. At the end of all mouse experiments, except for the fecal bile acid excretion study, food was removed from the cage at 10 a.m., and the mice were then fasted for 6 h, sedated with ketamine/xylazine, and killed. All animal protocols were approved by The Rockefeller University Animal Care and Use Committee.
Fig. 1.
Creation of human Cyp7a1 BAC transgenic mice. (A) The two BACs with genomic mouse Cyp7a1. The Cyp7a1 structural gene spans ≈10 kb in the center of each BAC, which have different lengths of 5′ and 3′ flanking sequence. (B) Expression and regulation of the mouse endogenous gene and human Cyp7a1 transgene in different transgenic lines. Female 189n19 and 269a16 BAC transgenic mice on the wild-type background (generation N7; n = 3 per group) were fed the AIN76a diet with or without cholestyramine. Mouse and human Cyp7 mRNA levels were quantified by TaqMan real-time quantitative PCR as described. *, P < 0.05, t test; **, P < 0.01, t test.
Diets. All animals had free access to water and food. After weaning at 4 weeks of age, breeding mice of all genotypes and their pups were fed the pellet form of a cereal-based rodent diet [PicoLab Rodent Diet 20 (Purina) containing 149 ppm cholesterol and 4.5% crude fat]. For dietary studies, 6-week-old female mice were fed the pellet form of a semisynthetic modified AIN76a diet containing 10% kcal as fat and 0.0% cholesterol [Clinton/Cybulsky Rodent Diet D12102N (Research Diets, New Brunswick, NJ) (10)] for 1 week. For the next week, mice received either the same 0.0% cholesterol diet or this diet supplemented with 0.5% (wt/wt) cholesterol (Diet D00083101, Research Diets) or 2% cholestyramine (Diet D1111210, Research Diets). They were then killed at 8 weeks of age. For the fecal bile acid excretion study, 6-week-old mice received the 0.0% cholesterol diet in pellet form in their regular cages. After 1 week, the mice were housed individually in metabolic cages and received a powdered form of either the 0.0% or 0.5% cholesterol diet for 3 days.
Quantification of Human and Mouse Cyp7a1 mRNA and of Cholesterol 27-Hydroxylase (Cyp27) mRNA. Fifty milligrams of liver tissue was placed into 1 ml of RNAlater (Ambion, Austin, TX) and homogenized in TRIzol reagent (Invitrogen), after standing overnight at 4°C. Total RNA was isolated according to the manufacturer's instructions, subjected to RNeasy cleanup (Qiagen) and, afterward, reverse-transcribed with Superscript II (Invitrogen). Real-time quantitative PCR was performed as described (11) with slight modifications by using the Applied Biosystems 7700 Sequence Detection System. Primers were specific for the human or mouse Cyp7a1 genes (human Cyp7a1: forward primer, TGGGCATCGCAAGCAAA; reverse primer, CTTTCATTGCTTCTGGGTTCCTA; and mouse Cyp7a1: forward primer, TGGGCATCTCAAGCAAACAC; reverse primer, TCATTGCTTCAGGGCTCCTG). The probe was the same for both the mouse and human Cyp7a1 (6FAM-TTCCTGCGACCTTCTGGAGTTTATTTCAAATGA). The following probe and primers were used to quantify Cyp27 mRNA levels: forward primer, TGCCTGGGTCGGAGGAT; reverse primer, GAGCCAGGGCAATCTCATACTT; and probe, 6FAM-TGGAGATGCAACTGATGCTGTCAAGGC. Cyclophilin was used as a housekeeping gene in all experiments (forward primer, GGCCGATGACGAGCCC; reverse primer, TGTCTTTGGAACTTTGTCTGCAA; and probe, 6FAM-TGGGCCGCGTCTCCTTCGA). An absolute standard curve was prepared for the human and mouse Cyp7a1 and for cyclophilin by using known concentrations of plasmids that contained specific sequences from these genes for each real-time quantitative PCR experiment. For Cyp27, a 1:20 dilution of mouse liver cDNA was used to generate a relative standard curve by serial dilution.
Cyp7a1 Enzyme Activity Assay. Liver samples were frozen immediately in liquid nitrogen. Microsomes were prepared by differential ultracentrifugation and used for the measurement of Cyp7a1 activity as described (12).
Bile Acid Pool Size and Fecal Bile Acid Excretion. Pool size was determined as the bile acid content of the small intestine, liver, and gallbladder. These tissues were removed and homogenized in double distilled water at room temperature with a Polytron homogenizer (Kinematica, Kùens-Lucerne, Switzerland). The homogenate was then extracted in ethanol as described (13). The total bile acid content of the tissue extracts was measured with an enzymatic assay (Trinity Biotech, Bray, Ireland). For fecal bile acid excretion, stools from individually housed mice were collected on each day of the 3-day feeding period and then dried, weighed, and ground. Bile acids were then extracted in ethanol and determined by an enzymatic assay (Trinity Biotech).
Lipid Analysis. Plasma was separated from whole blood by centrifugation. Plasma density was adjusted to 1.063 g/ml, and non-high-density lipoprotein (HDL) and HDL fractions were isolated after overnight ultracentrifugation at 40,000 rpm (model L8-55M Beckman ultracentrifuge) and 4°C. Total plasma cholesterol and HDL cholesterol (mg/dl) were then measured enzymatically by using the Infinity Cholesterol Reagent (Trinity Biotech). Non-HDL cholesterol was determined by subtraction of HDL cholesterol from total cholesterol.
Statistical Analysis. All data are reported as mean ± SD in a specified number of individual animals. A Student's unpaired t test was used to compare two datasets. For the comparison of three or more datasets, a one-way ANOVA with Tukey's post hoc test was used.
Results
Characterization of Human Cyp7a1 BAC Transgenic Mice on the Mouse Cyp7a1 Wild-Type Background. Two lines of human Cyp7a1 BAC transgenic mice, 189n19 and 269a16, were studied (Tg+). Crossing these Tg+ mice with C57BL/6J wild-type mice produced a Mendelian distribution of wild-type and Tg+ animals. The Tg+ animals were indistinguishable from their wild-type littermates in terms of survival, weight, gross appearance, and behavior. The human Cyp7a1 transgene and the endogenous mouse gene were only expressed in the liver (based on quantitative PCR of tissue RNAs; data not shown).
To compare the expression and regulation of the human Cyp7a1 transgene and the endogenous mouse Cyp7a1 gene, female Tg+ mice of the lines 189n19 and 269a16 were fed the 0.0% cholesterol diet with or without 2% cholestyramine. The results of this study are shown in Fig. 1B. Mouse Cyp7a1 mRNA levels were easily detectable by TaqMan real-time quantitative PCR. In contrast, expression of the human Cyp7a1 transgene was almost at the limits of detection in livers from mice of both transgenic lines fed the AIN76a diet alone. Bile acid depletion by cholestyramine led to an expected ≈3-fold increase in the ratio of the mouse endogenous mRNA to cyclophilin in both lines (P < 0.05). Remarkably, the ratio of the human Cyp7a1 mRNA to cyclophilin went up ≈1,000-fold in Tg+ mice of both lines in response to cholestyramine (P < 0.01) and reached the level of expression of the endogenous mouse gene in mice fed the AIN76a diet without cholestyramine. Although differences in mouse and human Cyp7a1 expression levels were found in Tg+ mice under basal conditions, this was not because of an inability of the human transgene to be expressed at high levels under different conditions.
Characterization of Human Cyp7a1 BAC Transgenic Mice on the Mouse Cyp7a1 KO Background. Mice of the human Cyp7a1 BAC transgenic line 189n19 were bred onto the mouse Cyp7a1 KO background. KO and Tg+KO mice did not differ in weight, gross appearance, or behavior from their mouse Het littermates at the time of study.
To further characterize these animals, KO, Het, and Tg+KO mice were fed the 0.0% cholesterol diet for 2 weeks (weeks 6–8), and their levels of Cyp7a1 mRNA are shown in Fig. 2A. In the KO animals, Cyp7a1 mRNA was completely undetectable. In the Het mice, mouse Cyp7a1 mRNA levels were 18-fold higher than the human Cyp7a1 mRNA levels in the Tg+KO mice (P < 0.001). Although not shown, transgene expression on the Cyp7a1 KO background was 2.6-fold higher than on the wild-type background (0.0107 ± 0.0006 vs. 0.0041 ± 0.0025 mRNA/cyclophilin; P < 0.01). Thus, the human transgene was suppressed somewhat in the presence of the endogenous mouse gene; however, even when the transgene was bred to the KO background, levels of human Cyp7a1 mRNA were still considerably lower than levels of mouse Cyp7a1 mRNA in Het mice.
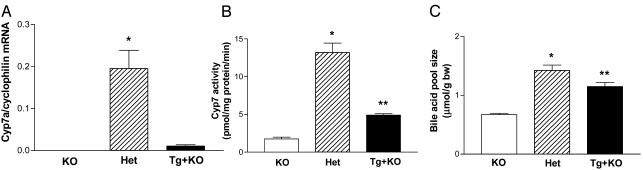
Fig. 2.
Characterization of human Cyp7a1 BAC transgenic mice on the mouse Cyp7a1 KO background. Female Cyp7a1 KO, Het, and Tg+KO mice (n = 4 per group) received the AIN76a 0.0% cholesterol diet for 2 weeks and were killed at 8 weeks of age. (A) Cyp7a1 mRNA levels. *, Het vs. Tg+KO (P < 0.001, t test). (B) Cyp7a1 activity levels. *, Het vs. Tg+KO (P < 0.001, t test); **, Tg+KO vs. KO (P < 0.01, t test). (C) Bile acid pool size. *, Het vs. KO (P < 0.01, t test); **, Tg+KO vs. KO (P < 0.01, t test).
In this study, Cyp7a1 activity was also measured, the results of which are shown in Fig. 2B. The assay showed some background Cyp7a1 activity in the KO livers, as reported by others (14). However, in the Het mice, Cyp7a1 activity was 7.5-fold above background (P < 0.01); whereas, in the Tg+KO mice, Cyp7a1 activity was 2.8-fold above background (P < 0.01). In the Het mice, the Cyp7a1 activity was 2.7-fold that in Tg+KO mice (P < 0.001). Therefore, Cyp7a1 activity was higher in Het mice than in Tg+KO mice, but the difference was not nearly as great as at the mRNA level.
The bile acid pool size was also measured in the same groups of mice on the 0.0% cholesterol diet, and no significant difference was found between Het and Tg+KO mice. However, as shown in Fig. 2C, each had a larger bile acid pool size than the KO mice (2.1- and 1.7-fold, respectively; P < 0.01 and P < 0.01, respectively). Thus, even though Het mice had higher Cyp7a1 mRNA and activity levels than Tg+KO, their bile acid pool sizes did not differ.
Effect of Cholesterol Feeding on Cyp7a1 mRNA and Activity Levels, Bile Acid Pool Size, and Fecal Bile Acid Excretion in Het and Tg+KO Mice. The responses of Cyp7a1 mRNA and activity levels in Het and Tg+KO mice to cholesterol feeding were assessed, and the results are shown in Fig. 3 A and B. Compared with mice fed the 0.0% cholesterol diet, Het mice fed the 0.5% cholesterol diet had 68% higher mouse Cyp7a1 mRNA levels (P < 0.05) and 48% higher Cyp7a1 activity levels (P < 0.01); whereas Tg+KO mice had the same human Cyp7a1 mRNA and 64% lower Cyp7a1 activity levels (P < 0.05). This experiment showed that mouse and human Cyp7a1 are very different in their response to cholesterol feeding.
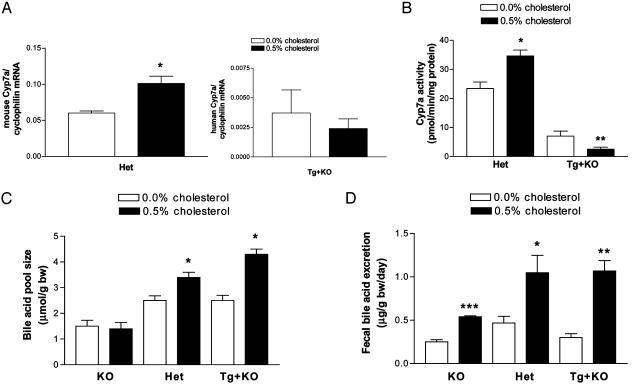
Fig. 3.
Cholesterol feeding: comparison of KO, Het, and Tg+KO mice. The mice received either the AIN76a (0.0% cholesterol) diet or the AIN76a diet supplemented cholesterol (0.5% cholesterol) (n = 4 females per group). (A Left) Mouse Cyp7a1 mRNA in Het mice. *, P < 0.05, t test. (Right) Human Cyp7a1 mRNA levels for Tg+KO mice (P not significant). Note the difference in scale of the y axis between both graphs. (B) Cyp7a1 activity for Het and Tg+KO mice. *, P < 0.01, t test; **, P < 0.05, t test. (C) Bile acid pool size for KO, Het, and Tg+KO mice. *, P < 0.05, t test; **, P < 0.001, t test. (D) Fecal bile acid excretion for KO, Het, and Tg+KO mice. *, P < 0.05, t test; **, P < 0.01, t test; ***, P < 0.001, t test.
The effect of cholesterol feeding on bile acid pool size was also assessed, and the results are shown in Fig. 3C. As a control, measurements were also made in KO mice. Compared with mice fed the 0.0% cholesterol diet, KO mice fed the 0.5% cholesterol diet did not increase their bile acid pool size; whereas Het mice increased their bile acid pool size by 36% (P < 0.05), and Tg+KO mice increased their bile acid pool size by 72% (P < 0.001). There was no significant difference between Het and Tg+KO mice fed the 0.5% cholesterol diet.
The effect of cholesterol feeding on fecal bile acid excretion was next measured, and the results are shown in Fig. 3D. Compared with mice fed the 0.0% cholesterol diet, KO, Het, and Tg+KO mice fed the 0.5% cholesterol diet all significantly increased fecal bile acid excretion. The KO mice began with relatively low excretion, and the absolute level of cholesterol-induced bile acid excretion reached only ≈50% of the level achieved in Het and Tg+K0 mice (P < 0.05 and P < 0.05, respectively, compared with KO mice). Thus, despite the difference between Het and Tg+KO mice in Cyp7a1 mRNA and activity levels in response to cholesterol feeding, bile acid pool size and fecal bile acid excretion showed similar increases.
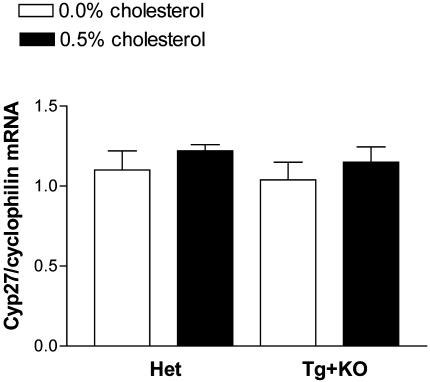
Cyp27 mRNA Levels in KO, Het, and Tg+KO Mice on Low and High Cholesterol Diets. The alternative pathway of bile acid biosynthesis was assessed in Het and Tg+KO mice on 0.0% and 0.5% cholesterol diets by measuring Cyp27 mRNA levels, the results of which are shown in Fig. 4. There were no significant effects of either genotype or diet on Cyp27 mRNA levels, suggesting that the dietary cholesterol effects on bile acid pool size and fecal bile acid excretion were not the result of increased Cyp27 expression. There was also no effect of cholesterol feeding on Cyp27 mRNA levels in KO mice (data not shown).
Fig. 4.
Cholesterol feeding: Cyp27 mRNA levels in Het and Tg+KO mice. Het and Tg+KO mice (n = 4 females per group) received either the 0.0% cholesterol diet or the 0.5% cholesterol diet. There is no significant difference between any of these groups.
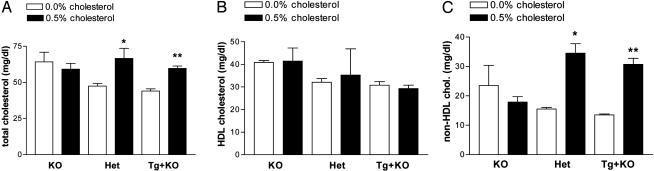
Effect of Cholesterol Feeding on Plasma Total, HDL, and Non-HDL Cholesterol Levels in KO, Het, and Tg+KO Mice. The effect of cholesterol feeding in KO, Het, and Tg+KO mice on plasma total, HDL, and non-HDL cholesterol levels was assessed, and the results are shown in Fig. 5 A, B, and C, respectively. In Het and Tg+KO mice, cholesterol feeding caused a similar magnitude of increase in total cholesterol levels (40% and 36%, respectively; P < 0.05 and P < 0.001, respectively) and non-HDL cholesterol levels (123% and 127%, respectively; P < 0.01 and P < 0.001, respectively). In contrast, KO mice showed no increase in total cholesterol and non-HDL cholesterol levels. In all three genotypes there was no significant effect of cholesterol feeding on HDL cholesterol levels. Thus, the presence of only the mouse Cyp7a1 gene or only the human Cyp7a1 gene resulted in the same response to dietary cholesterol.
Fig. 5.
Plasma cholesterol levels in female KO, Het, and Tg+KO mice (n = 4 per group). The mice received either the 0.0% or 0.5% cholesterol diet. (A) Total cholesterol. *, P < 0.05, t test; **, P < 0.001, t test. (B) HDL cholesterol. (C) non-HDL cholesterol. *, P < 0.01, t test; **, P < 0.001, t test.
Discussion
KO, Het, and Tg+KO mice were used to evaluate the role of Cyp7a1 in dietary cholesterol response. We confirmed that the human transgene and the mouse gene are differentially regulated by dietary cholesterol at the mRNA and activity levels. Nevertheless, this differential regulation was not reflected in the effects of dietary cholesterol on the bile acid pool size, fecal bile acid excretion, or plasma cholesterol levels. Thus, dietary cholesterol regulates these parameters independently of Cyp7a1 mRNA and activity levels.
The current study found that feeding 0.5% cholesterol increased mouse Cyp7a1 mRNA levels by 68% in Het (mouse Cyp7a1 only) but did not significantly change human Cyp7a1 mRNA in Tg+KO (human Cyp7a1 only). This diet also increased Cyp7a1 activity by 48% in Het but decreased Cyp7a1 activity by 64% in Tg+KO mice. In a previous study, Chen et al. (8) found that feeding 2% cholesterol increased mouse Cyp7a1 mRNA levels 2-fold but had no effect on human Cyp7a1 mRNA levels. In another study, Agellon et al. (5) demonstrated that feeding 1% cholesterol increased mouse Cyp7a1 mRNA levels 40% and decreased human Cyp7a1 mRNA levels by 56%. Agellon et al. also showed feeding cholesterol decreased Cyp7a1 activity by 55% in Tg+KO mice. The current results and those previously published are similar. Any minor differences may be due to the purity of the strains on the C57 background and/or the amounts of cholesterol in the different diets.
The Chen and Agellon studies did not measure the effects of dietary cholesterol on bile acid pool size or fecal bile acid excretion, which are major determinants of sterol balance. In the current study, we measured these parameters. Despite the differential regulation of human and mouse Cyp7a1 at the mRNA and activity levels, we found that feeding 0.5% cholesterol caused a similar increase in bile acid pool size and fecal bile acid excretion in both Het and Tg+KO mice. Thus, in Tg+KO mice, dietary cholesterol decreased Cyp7a1 activity; whereas, contrary to expectations, Cyp7a1 activity increased bile acid pool size and fecal bile acid excretion. This dissociation is similar to what has been described in cholesterol-fed rabbits by Xu et al. (15). These authors reported that 1 day of cholesterol feeding in rabbits increased Cyp7a1 activity with no significant effect on bile acid pool size; whereas 10 days of cholesterol feeding decreased Cyp7a1 activity by 62% but almost doubled the bile acid pool size. They suggested that the increase in bile acid pool size induced by several days of dietary cholesterol feeding overrode any direct stimulation of Cyp7a1 by dietary cholesterol and resulted in decreased Cyp7a1 activity. Thus, long-term feeding of dietary cholesterol in rabbits and Tg+KO mice increases bile acid pool size, which in turn suppresses Cyp7a1 activity. The common notion that Cyp7a1 activity regulates bile acid pool size does not apply to rabbits and Tg+KO mice. Instead, the bile acid pool size is the major regulator of Cyp7a1 activity.
As further evidence of the dominant effect of bile acid pool size on human Cyp7a1 mRNA and activity levels, the Tg+KO mice on a 0.0% cholesterol diet had very low Cyp7a1 mRNA and activity levels compared with Het mice with similar bile acid pool size. The increased sensitivity of human Cyp7a1 to bile acids was also shown by feeding cholestyramine to Tg+ mice on the wild-type background. In these mice, depleting the bile acid pool by feeding them 2% cholestyramine increased human Cyp7a1 mRNA ≈1,000-fold in both transgenic lines; whereas 2% cholestyramine increased mouse Cyp7a1 mRNA levels only ≈3-fold. In fact, when the bile acid pool was depleted, the mRNA level for human Cyp7a1 was nearly equal to mouse Cyp7a1 in these mice. Thus, the major effect on human Cyp7a1 expression is exerted by bile acid pool size, as has been suggested for rabbit Cyp7a1 (15).
The question then arises as to how dietary cholesterol increases bile acid pool size. It is clear in comparing KO and Het mice that the presence of mouse Cyp7a1 results in a ≈2-fold increase in bile acid pool size. In addition, feeding dietary cholesterol to KO mice results in no increase in bile acid pool size; whereas, in Het mice, the bile acid pool size increases by 36% (P < 0.05), which is compatible with the dietary cholesterol-induced increase in mouse Cyp7a1 mRNA (68%, P < 0.05) and activity (48%, P < 0.01) levels. Therefore, in mice, Cyp7a1 could be responsible for the dietary cholesterol-induced increase in bile acid pool size. However, in Tg+KO mice with only human Cyp7a1, dietary cholesterol decreased Cyp7a1 activity by 64% (P < 0.05) and increased bile acid pool size by 72% (P < 0.001). Therefore, in these mice, Cyp7a1 could not be responsible for dietary cholesterol-induced increase in bile acid pool size, and other regulatory factors must be at work. In rabbits, 2% cholesterol feeding for 3 days increased the activity of Cyp27, the rate-limiting enzyme in the alternative bile acid biosynthetic pathway, and increased the bile acid pool size (15). The same group showed that 2% cholesterol feeding did not up-regulate Cyp27 activity in rats (16). In the current study, 0.5% cholesterol feeding did not alter Cyp27 mRNA levels in either Het or Tg+KO mice, making it unlikely that dietary cholesterol stimulation of the alternative pathway of bile acid biosynthesis was responsible for the increase observed in the bile acid pool size.
Much of the original interest in Cyp7a1 regulation relates to the phenomenon of plasma lipoprotein level responsiveness to dietary cholesterol. In the study by Xu et al. (16), feeding 2% dietary cholesterol to rabbits raised plasma cholesterol levels 31-fold; whereas the increase in rats was only 2.1-fold. This large difference in responsiveness was attributed to finding that dietary cholesterol down-regulated Cyp7a1 activity and increased bile acid pool size in rabbits, compared to increased Cyp7a1 activity and no change in bile acid pool size in rats. In another study by Poorman et al. (17), feeding 0.1% dietary cholesterol to normal rabbits raised plasma cholesterol levels 10-fold; whereas, in a specially bred strain of low-responder rabbits, plasma cholesterol levels did not increase. In this study, 0.1% dietary cholesterol decreased fecal bile acid excretion ≈67% in normal rabbits and only ≈33% in the low responders. In both types of rabbits, 0.25% dietary cholesterol failed to increase Cyp7a1 activity, although the resistant rabbits had almost double the activity of the normal rabbits. These authors attributed the lack of diet responsiveness to higher Cyp7a1 activity and less dietary cholesterol-induced decrease in fecal bile acid excretion in resistant rabbits. Beynen et al. compared two other inbred strains of rabbits that were hypo- or hyperresponsive to dietary cholesterol (18). They found that feeding a high-cholesterol diet (0.07%) increased fecal bile acid excretion in hyporesponsive, but not in hyperresponsive, rabbits. The authors concluded that the differences in stimulation of fecal bile acid excretion after cholesterol feeding determined the phenomenon of hypo- or hyperresponsiveness in the two strains. In the current study, we found that dietary cholesterol in Het mice increased Cyp7a1 activity, bile acid pool size, and fecal bile acid excretion; whereas, in Tg+KO mice, dietary cholesterol decreased Cyp7a1 activity and increased bile acid pool size and fecal bile acid excretion. Yet in both genotypes, plasma total cholesterol levels rose ≈40%, and non-HDL cholesterol levels rose ≈2.3-fold. Thus, the notion that dietary cholesterol increases plasma cholesterol levels in proportion to decreasing Cyp7a1 activity and increasing bile acid pool size is contradicted by our mouse data. We speculate that the dietary cholesterol-induced increase in fecal bile acid excretion in KO, Het, and Tg+KO mice might be the explanation.
In summary, the current study in genetically altered mice provides fresh insights into diet responsiveness and the role of Cyp7a1 in this process. We confirmed that mouse Cyp7a1 activity is up-regulated by dietary cholesterol and that human Cyp7a1 activity is down-regulated. Yet in mice with mouse Cyp7a1 or human Cyp7a1, dietary cholesterol increased the bile acid pool size and fecal bile acid excretion. These increases could be accounted for by dietary cholesterol regulation of Cyp7a1 in animals with mouse Cyp7a1 but not in animals with human Cyp7a1. We also showed that the relative lack of responsiveness of plasma cholesterol to dietary cholesterol in mice compared to rabbits is not due to differential regulation of Cyp7a1 or the bile acid pool size.
Acknowledgments
We thank David Russell for providing Cyp7a1 KO mice and Edward Rubin and Len Pennacchio for end-sequencing the BACs. This work was supported by National Institutes of Health Grant HL32435 and a scholarship from the Deutsche Forschungsgemeinschaft (DFG Ti 312/1–1) to M.T.
Abbreviations: Cyp7a1, cholesterol 7α-hydroxylase; KO, knockout; Het, Cyp7a1 heterozygous; Tg+KO, Cyp7a1 transgenic on the KO background; BAC, bacterial artificial chromosome; Cyp27, cholesterol 27-hydroxylase; HDL, high-density lipoprotein.
References
- 1.Maxwell, K. N., Soccio, R. E., Duncan, E. M., Sehayek, E. & Breslow, J. L. (2003) J. Lipid Res. 44, 2109–2119. [DOI] [PubMed] [Google Scholar]
- 2.Cohen, J. C., Cali, J. J., Jelinek, D. F., Mehrabian, M., Sparkes, R. S., Lusis, A. J., Russell, D. W. & Hobbs, H. H. (1992) Genomics 14, 153–161. [DOI] [PubMed] [Google Scholar]
- 3.Russell, D. W. & Setchell, K. D. (1992) Biochemistry 31, 4737–4749. [DOI] [PubMed] [Google Scholar]
- 4.Peet, D. J., Turley, S. D., Ma, W., Janowski, B. A., Lobaccaro, J. M., Hammer, R. E. & Mangelsdorf, D. J. (1998) Cell 93, 693–704. [DOI] [PubMed] [Google Scholar]
- 5.Agellon, L. B., Drover, V. A., Cheema, S. K., Gbaguidi, G. F. & Walsh, A. (2002) J. Biol. Chem. 277, 20131–20134. [DOI] [PubMed] [Google Scholar]
- 6.Hegsted, D. M. (1986) Am. J. Clin. Nutr. 44, 299–305. [DOI] [PubMed] [Google Scholar]
- 7.Kuan, S. I. & Dupont, J. (1989) J. Nutr. 119, 349–355. [DOI] [PubMed] [Google Scholar]
- 8.Chen, J. Y., Levy-Wilson, B., Goodart, S. & Cooper, A. D. (2002) J. Biol. Chem. 277, 42588–42595. [DOI] [PubMed] [Google Scholar]
- 9.Ishibashi, S., Schwarz, M., Frykman, P. K., Herz, J. & Russell, D. W. (1996) J. Biol. Chem. 271, 18017–18023. [DOI] [PubMed] [Google Scholar]
- 10.Lichtman, A. H., Clinton, S. K., Iiyama, K., Connelly, P. W., Libby, P. & Cybulsky, M. I. (1999) Arterioscler. Thromb. Vasc. Biol. 19, 1938–1944. [DOI] [PubMed] [Google Scholar]
- 11.Soccio, R. E., Adams, R. M., Romanowski, M. J., Sehayek, E., Burley, S. K. & Breslow, J. L. (2002) Proc. Natl. Acad. Sci. USA 99, 6943–6948. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 12.VanPatten, S., Ranginani, N., Shefer, S., Nguyen, L. B., Rossetti, L. & Cohen, D. E. (2001) Am. J. Physiol. 281, G393–G404. [DOI] [PubMed] [Google Scholar]
- 13.Schwarz, M., Russell, D. W., Dietschy, J. M. & Turley, S. D. (1998) J. Lipid Res. 39, 1833–1843. [PubMed] [Google Scholar]
- 14.Schwarz, M., Russell, D. W., Dietschy, J. M. & Turley, S. D. (2001) J. Lipid Res. 42, 1594–1603. [PubMed] [Google Scholar]
- 15.Xu, G., Salen, G., Shefer, S., Tint, G. S., Nguyen, L. B., Chen, T. S. & Greenblatt, D. (1999) J. Clin. Invest. 103, 89–95. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 16.Xu, G., Shneider, B. L., Shefer, S., Nguyen, L. B., Batta, A. K., Tint, G. S., Arrese, M., Thevananther, S., Ma, L., Stengelin, S., et al. (2000) J. Lipid Res. 41, 298–304. [PubMed] [Google Scholar]
- 17.Poorman, J. A., Buck, R. A., Smith, S. A., Overturf, M. L. & Loose-Mitchell, D. S. (1993) J. Lipid Res. 34, 1675–1685. [PubMed] [Google Scholar]
- 18.Beynen, A. C., Meijer, G. W., Lemmens, A. G., Glatz, J. F., Versluis, A., Katan, M. B. & Van Zutphen, L. F. (1989) Atherosclerosis 77, 151–157. [DOI] [PubMed] [Google Scholar]