Abstract
Effective chemotherapy against esophageal carcinoma is considered achievable with a combination of 5-fluorouracil (5-FU) and cisplatin (CDDP). However, chemo-therapy remains ineffective in certain patients. The aim of this study was to clarify the factors which affect sensitivity to 5-FU and CDDP. The effects of factors known to influence sensitivity to 5-FU and CDDP, namely transporters, DNA repair enzymes and metabolic enzymes, were examined. mRNA levels of four transporters, SLC22A2, SLC23A2, ABCB1 and ABCC2, two DNA repair-related enzymes, Rad51 and MSH2, and one metabolic enzyme, dihydropyrimidine dehydrogenase (DPYD), showed a strong correlation (|r|>0.7) with IC50 values for 5-FU. In addition, the mRNA levels of ABCC2, MSH2 and DPYD showed a strong correlation (|r|>0.7) with the IC50 values for CDDP. Gimeracil, a DPYD inhibitor, enhanced the sensitivity of some cells to 5-FU but decreased the sensitivity of all the cells to CDDP. The inhibitory effects of ABCC2 with MK571 did not correspond to those observed in the correlation analysis. In conclusion, mRNA levels of SLC22A2, SLC23A2, ABCB1, ABCC2, Rad51, MSH2 and DPYD were confirmed to be strongly correlated with IC50 values for 5-FU, and mRNA levels of ABCC2, MSH2 and DPYD were confirmed to be strongly correlated with IC50 values for CDDP. In addition, the inhibition of DPYD appeared to affect the cytotoxicity of CDDP.
Keywords: esophageal carcinoma, cisplatin, 5-fluorouracil, drug sensitivity
Introduction
In Japan, one-third of all mortalities are cancer-related (1). The incidence of lung, colorectal and breast cancer is increasing in Japan as well as worldwide (1). Esophageal carcinoma has a lower incidence than other types of cancer, but 5-fluorouracil (5-FU) and cisplatin (CDDP)-based chemoradiotherapy results in moderately high response and survival rates relative to other types of cancer. In fact, the complete response and 5-year survival rates following 5-FU and CDDP-based chemoradiotherapy have been reported to be 58 and 29%, respectively, among Japanese esophageal carcinoma patients (2). However, chemotherapy remains ineffective in certain patients. Therefore, identifying the factors that affect sensitivity to 5-FU and CDDP is necessary for enhancing the clinical outcome of chemotherapy for esophageal carcinoma.
Certain factors affecting sensitivity to 5-FU or CDDP have previously been revealed, including the molecular mechanisms involved in the cellular kinetics and dynamics of 5-FU and CDDP. For example, overexpression of the ABC transporter superfamily C5 (ABCC5/MRP5) decreases cellular accumulation of 5-FU, resulting in resistance to 5-FU (3). In addition, dihydropyrimidine dehydrogenase (DPYD), a 5-FU metabolizing enzyme, has been correlated with clinical response to 5-FU-based chemotherapy among colon cancer patients (4,5). The cytotoxic effects of CDDP are also attenuated by ERCC1, a DNA repair-related enzyme associated with restoration of DNA damage induced by chemotherapeutic agents or UV rays (6–8). However, there is little information concerning whether the levels of these molecules are predictive of sensitivity to 5-FU or CDDP in esophageal carcinoma.
In the present study, sensitivity to 5-FU and CDDP and mRNA levels of 35 genes, including drug transporters, DNA repair enzymes and metabolic enzymes, were evaluated in 5 human esophageal carcinoma cell lines. Based on these findings, factors affecting the sensitivity of esophageal carcinoma cells to 5-FU and CDDP were examined.
Materials and methods
Chemicals
5-FU was obtained from Sigma-Aldrich Chemical Co. (St. Louis, MO, USA). CDDP was purchased from Wako Pure Chemical Industries, Ltd. (Osaka, Japan). Gimeracil and MK571 were purchased from Toronto Research Chemicals, Inc. (Toronto, ON, Canada) and Cayman Chemical Company (Ann Arbor, MI, USA), respectively. 2-(4-Iodophenyl)-5-(2,4-disulfophenyl)-2H-tetrazolium, monosodium salt (WST-1) and 1-methoxy-5-methylphenazinium methylsulfate were purchased from Dojindo Laboratories (Kumamoto, Japan).
Cell culture
The human esophageal adenocarcinoma cell line OE33 was purchased from DS Pharma Biomedical Co., Ltd. (Osaka, Japan) and the squamous carcinoma cell lines KYSE30, KYSE70, KYSE140 and KYSE150 (9) were obtained from Health Science Research Resources Bank (Osaka, Japan). OE33 and the other cell lines were maintained in RPMI-1640 medium (Invitrogen Corp., Carlsbad, CA, USA) and Dulbecco’s modified Eagle’s medium (Invitrogen), respectively, supplemented with 10% heat-inactivated fetal bovine serum (lot no. 1335770 and 348777, Invitrogen). Cells were cultured in an atmosphere of 95% air and 5% CO2 at 37°C and subcultured every 3 or 4 days at a density of 1×106 cells/25 cm2 culture flask. The number of passages for OE33, KYSE30, KYSE70, KYSE140 and KYSE150 cells was 15–25, 15–28, 15–26, 21–31 and 19–31, respectively.
Growth rate of esophageal carcinoma cell lines
The growth rate of esophageal carcinoma cells was evaluated with a WST-1 assay utilizing succinate dehydrogenase activity. Cells were seeded onto a 96-well plate (Corning Inc., Corning, NY, USA) at a density of 5×103 cells/well/100 μl and cultured in an atmosphere of 95% air and 5% CO2 at 37°C. After 0, 6, 12, 18, 24, 36, 48, 72 and 96 h, the culture medium was exchanged for 110 μl of medium containing WST-1 reagent solution (10 μl WST-1 solution and 100 μl culture medium), and 3 h later the absorbance was determined using a micro-plate reader at 450 nm with a reference wavelength of 620 nm (SpectraFluor™, Tecan Group Ltd., Männedorf, Switzerland). The doubling time for cell growth was calculated from the logarithmic phase of a growth curve (10) as follows: Doubling time = (t1 - t0) × log102/(log10N1 - log10N0). N0 and N1 are the number of cells (% of day 0) at t1 and t0, respectively.
Growth inhibitory activity assay
Cells were seeded onto 96-well plates (Corning Inc.) at a density of 5×103 cells/well/100 μl on day 0. After incubation for 24 h, the culture medium was exchanged for one containing 5-FU or CDDP at various concentrations (day 1). On day 4, a WST-1 assay was performed as described above.
The effects of gimeracil and MK571 on the growth inhibitory effects of 5-FU and CDDP were also evaluated by WST-1 assay. Cells were incubated for 24 h as described above and the culture medium was exchanged for one containing 5-FU or CDDP at various concentrations with or without gimeracil (100 μM) or MK571 (50 μM). Following incubation for 72 h at 37°C, the culture medium was replaced with a medium containing WST-1 and the absorbance was measured.
The 50% growth inhibitory concentrations (IC50) were calculated according to the sigmoid inhibitory effect model: E = Emax × [1 - Cγ/(Cγ + IC50γ)], using the nonlinear least-squares fitting method (Solver, Microsoft® Excel). E and Emax represent the surviving fraction (% of control) and its maximum, respectively. C and γ are the drug concentration in the medium and the sigmoidicity factor, respectively. Relative sensitivity was calculated as follows: Relative sensitivity = IC50 (without gimeracil or MK571)/IC50 (with gimeracil or MK571).
Real-time reverse transcription (RT)-PCR
The mRNA expression levels were measured by real-time RT-PCR. Cells were seeded at a density of 2×106 cells/60 mm culture dish and 48 h later, total RNA was extracted from the cells with a GenEluteTM Mammalian Total RNA Miniprep kit (Sigma-Aldrich). Total RNA (1 μg) was used for RT with a PrimeScriptTM RT reagent kit (Takara Bio, Inc., Shiga, Japan) and a thermal cycler (i-Cycler, Bio-Rad Laboratories, Inc., Hercules, CA, USA). The RT reaction was conducted in 40 μl reaction buffer at 37°C for 15 min and terminated by heating at 85°C for 5 sec followed by cooling at 4°C.
Real-time PCR was performed with a 7500 Real-time PCR system (Applied Biosystems, Carlsbad, CA, USA) and SYBR Premix Ex Taq™ (Takara Bio, Inc.). The primer sequences are shown in Table I. PCR was performed at 95°C for 10 sec, followed by 40 cycles of 95°C for 5 sec and 60°C for 34 sec. Dissociation was initiated at 95°C for 15 sec followed by 60°C for 1 min and 95°C for 15 sec. To compare the relative expression of target mRNA levels between the cell lines, the comparative Ct method was used, as previously described (10); β-actin (ACTB) was used as an internal standard. Samples were prepared in duplicate and three independent sample sets were analyzed.
Table I.
Sequences of oligonucleotide primers designed for real-time PCR.
| Function and gene | Forward (5′–3′) | Reverse (5′–3′) | Reference |
|---|---|---|---|
| ACTB | TCATGAAGTGTGACGTGGACATC | TGCATCCTGTCGGCAATG | 10 |
| Transport | |||
| SLC22A1 | TCTTCCATCGTCACTGAGTTCAAC | AGAAGCCCGCATTCAAACAG | 10 |
| SLC22A2 | TCTACTCTGCCCTGGTTGAATTC | ATGCAGCCCAAGGGTAACG | 10 |
| SLC22A3 | TAGCCCCATTTCTGCTCTTTC | AGATGGATGCCAGGATACCAA | 10 |
| SLC23A2 | TCTTTGTGCTTGGATTTTCGAT | ACGTTCAACACTTGATCGATTC | 23 |
| SLC31A1 | ACAAGTCAGCATTCGCTACAATTC | TTGCAGGAGGTGAGGAAAGC | 9 |
| ABCB1 | TTCCTTCACCCAGGCAATG | ATGAGTTTATGTGCCACCAAGTAG | a |
| ABCC1 | CAGTGACCTCTGGTCCTTAAACAA | TTGGCGCATTCCTTCTTCC | 24 |
| ABCC2 | ACTTGTGACATCGGTAGCATGGA | AAGAGGCAGTTTGTGAGGGATGA | a |
| ABCC3 | GTCCGCAGAATGGACTTGAT | TCACCACTTGGGGATCATTT | 25 |
| ABCC4 | GCTCAGGTTGCCTATGTGCT | CGGTTACATTTCCTCCTCCA | 25 |
| ABCC5 | CGAAGGGTTGTGTGGATCTT | GTTTCACCATGAAGGCTGGT | a |
| ABCC6 | TGTCGCTCTTTGGAAAATCC | AGGAACACTGCGAAGCTCAT | 25 |
| ABCG2 | TGACGGTGAGAGAAAACTTAC | TGCCACTTTATCCAGACCT | 26 |
| ATP7A | AGATACTGGGACACTGGAGAAA | AGGTCATCCCTTCCACTTTCA | 10 |
| ATP7B | TGATTTATAACCTGGTTGGGATACC | ATGAGAGCACCACAGACACAGA | 10 |
| DNA repair | |||
| ERCC1 | TACAAGGCCTATGAGCAGAAACCA | TCTCTTGATGCGGCGATGAG | a |
| ERCC2 | CTGGAGGTGACCAAACTCATCTA | CCTGCTTCTCATAGAAGTTGAGC | 27 |
| ERCC3 | TATCCCAGGACACACAGGAAAT | TCACCTTGAAGCTATAACCTTGA | a |
| XPA | TGCGGCGAGCAGTAAGAAG | TCATGGCCACACATAGTACAAGTC | a |
| Rad51 | TGGGAACTGCAACTCATCTGG | GCGCTCCTCTCTCCAGCAG | 28 |
| BRCA1 | ACAGCTGTGTGGTGCTTCTGTG | CATTGTCCTCTGTCCAGGCATC | 29 |
| BRCA2 | TGAAGAGCAGTTAAGAGCCTTGAA | ACGGTTGTGACATCCCTTGATAAA | a |
| HMGB1 | CAAGCGAACAGCAGGGTTAG | CAGATTGAGTCATTTGCTCCTCTTA | a |
| HMGB2 | TGAACATCGCCCAAAGATCA | TCAGACCACATTTCACCCAATT | a |
| MLH1 | GATTACCCCTTCTGATTGACA | ACTGAGGCTTTCAAAACA | 30 |
| MSH2 | CAGTATATTGGAGAATCGCA | AGGGCATTTGTTTCACC | 30 |
| PMS2 | AGTCAGCGTGCAGCAGTTATT | GACCATTTTGGCATACTCCTTCT | a |
| RPP25 | AGAATGGTGGACAGTGGGATT | TACTTCAGGTGCTCTTCGTGAATG | a |
| Metabolism | |||
| GSTP1 | CTGCGCATGCTGCTGGCAGATC | TTGGACTGGTACAGGGTGAGGTC | 31 |
| GCLC | GGCAAGATACCTTTATGACCAGTT | TGCAGCACTCAAAGCCATAA | 32 |
| GCLM | TGACTGCATTTGCTAAACAATTTGA | CGTGCGCTTGAATGTCAGG | 33 |
| TYSM | GCCTCGGTGTGCCTTTCA | CCCGTGATGTGCGCAAT | 34 |
| DPYD | AATGATTCGAAGAGCTTTTGAAGC | GTTCCCCGGATGATTCTGG | 35 |
| UMPS | TAGTGTTTTGGAAACTGTTGAGGTT | CTTGCCTCCCTGCTCTCTGT | 36 |
| MTHFR | CGGGTTAATTACCACCTTGTCAA | GCATTCGGCTGCAGTTCA | 36 |
Primer sequences were designed using Primer Express® software. ACTB, β-actin.
Statistical analyses
Data are shown as the mean ± standard deviation (SD). Comparisons between 2 and among 3 or more groups were performed with Student’s unpaired t-test and repeated one-way analysis of variance (ANOVA) followed by Scheffe’s F test, respectively. P<0.05 (two-tailed) was considered to indicate a statistically significant result. The correlation analysis was performed using Pearson’s correlation coefficient (r).
Results
Growth rates of esophageal carcinoma cell lines
Table II shows the cell growth doubling times for the 5 esophageal carcinoma cell lines. Doubling times for the cells varied from 20 to 25 h, revealing a significant difference between lines. KYSE30 cells (20.1±1.41 h) had the shortest doubling time and OE33 cells (25.0±0.90 h) the longest.
Table II.
Doubling times of esophageal carcinoma cell lines.
| Cell line | Doubling time, mean ± SD (h) |
|---|---|
| OE33 | 25.0±0.90 |
| KYSE30 | 20.1±1.41 |
| KYSE70 | 21.8±0.51 |
| KYSE140 | 23.3±1.07 |
| KYSE150 | 20.6±0.53 |
n=6.
Sensitivity of esophageal carcinoma cell lines to 5-FU and CDDP
The IC50 values for 5-FU were markedly different among the cell lines (0.524–30.2 μM); the OE33 cells showed the highest sensitivity to 5-FU and the KYSE30 cells the lowest sensitivity (Table III). In the case of CDDP, the IC50 values were also substantially different among the cell lines (2.17–19.5 μM). The rank order of sensitivity to CDDP was comparable to that for 5-FU.
Table III.
IC50 values for 5-FU and CDDP in esophageal carcinoma cell lines.
| IC50 value, mean ± SD (μM)
|
||
|---|---|---|
| Cell line | 5-FU | CDDP |
| OE33 | 0.524±0.08 | 2.17±0.33 |
| KYSE30 | 30.2±8.29 | 19.5±3.67 |
| KYSE70 | 13.1±13.3 | 5.27±0.36 |
| KYSE140 | 1.88±0.38 | 3.09±0.67 |
| KYSE150 | 4.75±1.46 | 14.0±1.02 |
n=4. 5-FU, 5-fluorouracil; CDDP, cisplatin.
Correlation analysis of factors affecting drug sensitivity
The level of mRNA expression differed among the esophageal carcinoma cell lines (Table IV). The correlations between the IC50 values and the mRNA levels of the 35 different genes were analyzed (Table V). SLC22A3 mRNA was not detected in any cells, with the exception of the OE33 cell line. ABCC6 mRNA expression was not observed in KYSE30 and KYSE70 cells.
Table IV.
Expression levels of mRNA in esophageal carcinoma cell lines.
| Expression ratio, mean ± SD (2−ΔCt×10−4)
|
|||||
|---|---|---|---|---|---|
| Function and gene | OE33 | KYSE30 | KYSE70 | KYSE140 | KYSE150 |
| Transport | |||||
| SLC22A1 | 0.11±0.03 | 0.07±0.02 | 0.01±0.003 | 0.03±0.02 | 0.12±0.07 |
| SLC22A2 | 0.49 ±0.15 | 0.16±0.03 | 0.23±0.03 | 0.87±0.45 | 0.47±0.36 |
| SLC22A3 | 36.3±9.24 | ND | ND | ND | ND |
| SLC23A2 | 76.1±13.8 | 36.6±6.39 | 59.8±4.66 | 61.1±43.8 | 92.9±64.0 |
| SLC31A1 | 125±26.6 | 131±10.2 | 179±23.4 | 252±135 | 244±147 |
| ABCB1 | 0.53±0.14 | 0.16±0.03 | 0.25±0.06 | 0.54±0.20 | 0.79±0.59 |
| ABCC1 | 74.7±11.3 | 37.0±3.83 | 246±32.9 | 123±75.6 | 67.8±36.6 |
| ABCC2 | 0.05±0.01 | 2.57±0.89 | 1.36±0.07 | 0.38±0.16 | 0.38±0.23 |
| ABCC3 | 80.5±15.1 | 10.2±2.64 | 60.7±8.62 | 40.6±23.3 | 123±86.7 |
| ABCC4 | 26.1±2.17 | 28.9±1.95 | 52.1±5.53 | 189±105 | 92.5±51.3 |
| ABCC5 | 9.06±1.30 | 62.14±17.0 | 65.38±8.60 | 22.92±10.6 | 26.76±19.2 |
| ABCC6 | 1.79±0.12 | ND | ND | 0.05±0.05 | 0.04±0.04 |
| ABCG2 | 6.25±1.29 | 4.64±0.21 | 1.66±0.34 | 2.47±0.69 | 33.1±18.3 |
| ATP7A | 8.66±1.27 | 7.99±0.70 | 4.68±1.20 | 6.09±3.61 | 15.4±7.98 |
| ATP7B | 1.91±0.25 | 1.89±0.73 | 2.21±0.47 | 5.72±4.77 | 3.37±2.69 |
| DNA repair | |||||
| ERCC1 | 219±66.1 | 143±35.8 | 96.3±13.2 | 241±131 | 296±175 |
| ERCC2 | 43.9±4.57 | 35.1±8.01 | 21.0±2.52 | 47.9±22.4 | 52.2±22.6 |
| ERCC3 | 82.3±11.5 | 79.3±19.4 | 57.4±6.31 | 134±97.9 | 185±104 |
| XPA | 91.4±16.0 | 107±16.7 | 193±14.8 | 461±290 | 283±164 |
| Rad51 | 3.84±1.03 | 2.64±0.37 | 3.67±1.09 | 6.09±3.07 | 4.72±1.75 |
| BRCA1 | 90.5±15.6 | 61.1±2.46 | 65.3±4.56 | 188±116 | 151±78.8 |
| BRCA2 | 110±20.4 | 111±3.99 | 43.5±3.74 | 261±165 | 204±108 |
| HMGB1 | 35.2±7.29 | 35.9±1.84 | 51.4±3.98 | 79.9±47.8 | 37.6±19.1 |
| HMGB2 | 509±87.3 | 1340±150 | 1343±129 | 1980±947 | 1367±679 |
| MLH1 | 27.5±4.55 | 21.3±1.61 | 23.9±1.83 | 28.2±18.4 | 70.9±36.8 |
| MSH2 | 185±39.8 | 540±38.6 | 331±23.5 | 338±154 | 272±114 |
| PMS2 | 25.3±3.22 | 34.2±4.24 | 77.1±12.8 | 123±81.6 | 67.0±42.8 |
| RPP25 | 27.5±4.35 | 6.32±0.99 | 0.13±0.03 | 74.9±59.9 | 0.74±0.47 |
| Metabolism | |||||
| GSTP1 | 2444 ±425 | 2926±644 | 3421±380 | 5784±3549 | 7249±3978 |
| GCLC | 5.21±0.51 | 3.87±1.15 | 45.0±4.30 | 10.8±4.41 | 9.05±5.39 |
| GCLM | 8.38±2.61 | 31.34±4.30 | 75.1±10.8 | 33.0±15.4 | 32.1±22.2 |
| TYMS | 54.0±11.6 | 163±3.10 | 2511±136 | 81.8±32.9 | 215.6±102 |
| DPYD | 5.81±2.03 | 62.4±6.50 | 0.82±0.29 | 1.23±0.87 | 12.4±9.82 |
| UMPS | 85.6±17.4 | 69.0±3.85 | 162±20.6 | 183±108 | 165±73.2 |
| MTHFR | 5.78±1.85 | 10.0±2.39 | 18.6±2.72 | 25.6±23.2 | 23.7±15.3 |
ΔCt = Ct (target gene) - Ct (β-actin). ND, not detected; n=3.
Table V.
Pearson’s correlation coefficient between IC50 values for 5-FU or CDDP and mRNA expression level.
| Pearson’s correlation coefficient (r)
|
||
|---|---|---|
| Function and gene | 5-FU | CDDP |
| Transport | ||
| SLC22A1 | −0.189 | 0.333 |
| SLC22A2 | −0.764 | −0.574 |
| SLC22A3 | ND | ND |
| SLC23A2 | −0.790 | −0.302 |
| SLC31A1 | −0.477 | −0.132 |
| ABCB1 | −0.788 | −0.215 |
| ABCC1 | −0.150 | −0.530 |
| ABCC2 | 0.992b | 0.706 |
| ABCC3 | −0.659 | −0.179 |
| ABCC4 | −0.470 | −0.315 |
| ABCC5 | 0.573 | 0.234 |
| ABCC6 | ND | ND |
| ABCG2 | −0.244 | 0.398 |
| ATP7A | −0.199 | 0.451 |
| ATP7B | −0.485 | −0.314 |
| DNA repair | ||
| ERCC1 | −0.638 | −0.041 |
| ERCC2 | −0.533 | 0.019 |
| ERCC3 | −0.439 | 0.187 |
| XPA | −0.463 | −0.284 |
| Rad51 | −0.756 | −0.523 |
| BRCA1 | −0.653 | −0.274 |
| BRCA2 | −0.455 | −0.049 |
| HMGB1 | −0.341 | −0.507 |
| HMGB2 | 0.010 | 0.125 |
| MLH1 | −0.369 | 0.269 |
| MSH2 | 0.913a | 0.719 |
| PMS2 | −0.363 | −0.365 |
| RPP25 | −0.486 | −0.561 |
| Metabolism | ||
| GSTP1 | −0.401 | 0.121 |
| GCLC | 0.032 | −0.321 |
| GCLM | 0.287 | −0.011 |
| TYMS | 0.163 | −0.211 |
| DPYD | 0.881 | 0.863 |
| UMPS | −0.522 | −0.379 |
| MTHFR | −0.319 | −0.074 |
ND, not detected.
P<0.05 and
P<0.01 significant correlations between IC50 values and mRNA expression levels. 5-FU, 5-fluorouracil; CDDP, cisplatin.
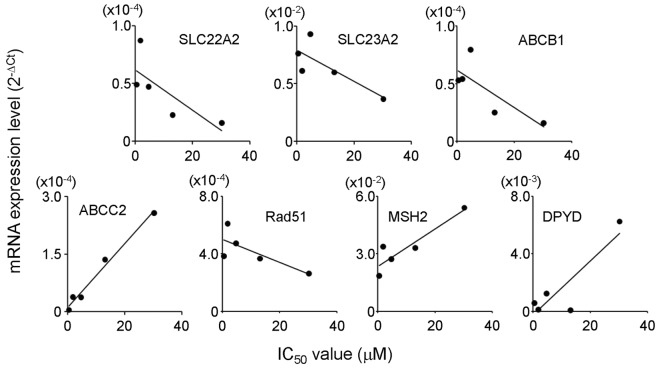
The mRNA levels of SLC22A2, SLC23A2, ABCB1 and Rad51 showed a strong negative correlation (r<−0.7) with the IC50 values for 5-FU. ABCC2, MSH2 and DPYD were positively correlated with the IC50 values for 5-FU (r>0.7; Table V and Fig. 1). In the case of CDDP, a high positive correlation coefficient (r>0.7) was found between the IC50 values and ABCC2, MSH2 and DPYD mRNA expression (Table V and Fig. 2).
Figure 1.
Correlation between IC50 values for 5-FU and mRNA expression levels in the esophageal carcinoma cell lines. The IC50 values for 5-FU were obtained from growth inhibition studies (Table III). The mRNA expression levels (2−ΔCt) in the cells were evaluated by real-time RT-PCR assay using SYBR®-Green. The threshold cycle (Ct) values were used to quantify the PCR product, and the relative expression level of the target gene was expressed as 2−ΔCt. The ΔCt was calculated by subtracting Ct (β-actin; as an internal standard) from Ct (target gene). 5-FU, 5-fluorouracil; RT, reverse transcription.
Figure 2.
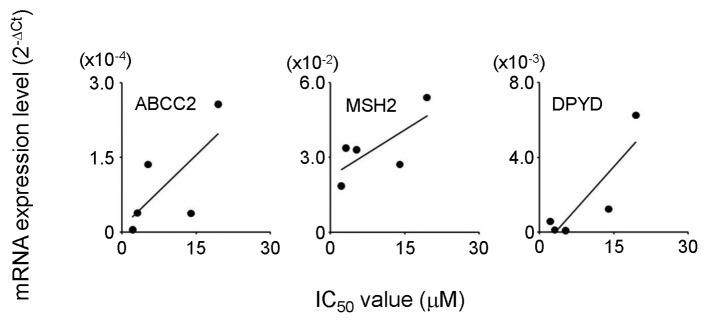
Correlation between the IC50 values for CDDP and mRNA expression levels in the esophageal carcinoma cell lines. The IC50 values for CDDP were obtained from growth inhibition studies (Table III). The mRNA expression levels (2−ΔCt) in the cells were evaluated by real-time RT-PCR assay using SYBR®-Green. The threshold cycle (Ct) values were used to quantify the PCR product, and the relative expression level of the target gene was expressed as 2−ΔCt. ΔCt was calculated by subtracting Ct (β-actin; as an internal standard) from Ct (target gene). CDDP, cisplatin; RT, reverse transcription.
Effects of gimeracil and MK571 on sensitivity of esophageal carcinoma cell lines to 5-FU and CDDP
The sensitivity of KYSE30 cells to 5-FU was enhanced by gimeracil, but in the other cell lines gimeracil had no observable effect (Table VI). In addition, gimeracil showed a tendency to decrease the sensitivity of all the cell lines to CDDP.
Table VI.
Relative sensitivity of the esophageal carcinoma cell lines to 5-FU or CDDP with or without gimeracil.
| Relative sensitivity, mean ± SD (fold)
|
||
|---|---|---|
| Cell line | 5-FU | CDDP |
| OE33 | 1.10±0.37 | 0.579±0.06 |
| KYSE30 | 2.30±0.13 | 0.710±0.03 |
| KYSE70 | 1.16±0.19 | 0.687±0.05 |
| KYSE140 | 0.989±0.15 | 0.691±0.10 |
| KYSE150 | 1.19±0.16 | 0.788±0.25 |
Relative sensitivity, the ratio of IC50 value for 5-FU or CDDP without gimeracil to those with gimeracil (n=4). Gimeracil, 100 μM. 5-FU, 5-fluorouracil; CDDP, cisplatin.
MK571 had no observable effect on the KYSE30, KYSE140 and KYSE150 cells (Table VII). However, the sensitivity of KYSE70 cells to 5-FU was substantially accelerated by the presence of MK571, and the sensitivity of OE33 cells to 5-FU was markedly decreased. However, MK571 showed a tendency to decrease sensitivity to CDDP, with the exception of the KYSE30 and KYSE150 cell lines.
Table VII.
Relative sensitivity of the esophageal carcinoma cell lines to 5-FU or CDDP with or without MK571.
| Relative sensitivity, mean ± SD (fold)
|
||
|---|---|---|
| Cell line | 5-FU | CDDP |
| OE33 | 0.0680±0.01 | 0.852±0.28 |
| KYSE30 | 0.961±0.06 | 0.974±0.09 |
| KYSE70 | 2.36±1.36 | 0.617±0.06 |
| KYSE140 | 0.813±0.16 | 0.803±0.04 |
| KYSE150 | 0.731±0.11 | 1.08±0.26 |
Relative sensitivity, the ratio of IC50 values for 5-FU or CDDP without MK571 to those with MK571 (n=4). MK571, 50 μM. 5-FU, 5-fluorouracil; CDDP, cisplatin.
Discussion
Combination chemotherapy with 5-FU and CDDP is known to be effective against esophageal carcinoma. However, it remains ineffective in certain patients, and the causes for this have not been clarified. The aim of the present study was to examine the factors affecting the sensitivity of esophageal carcinoma cells to 5-FU and CDDP.
The sensitivity of the 5 different esophageal carcinoma cell lines to 5-FU and CDDP differed (Table III). OE33, an adenocarcinoma cell line, showed a high sensitivity to 5-FU and CDDP, whereas the squamous cell carcinoma KYSE30 cells showed low sensitivity to 5-FU and CDDP. In addition, OE33 cells had the longest doubling time (an index of cell growth) of all the cell lines and KYSE30 cells the shortest (Table II), resulting in a trend for lower sensitivity to chemo-therapeutic agents among cells with higher growth activity. These findings suggest that sensitivity to 5-FU and CDDP was influenced by the growth activity of cells, although cytotoxic agents such as 5-FU and CDDP are known to be more toxic in cells with higher growth activity. In order to resolve this discrepancy, further studies concerning the correlation between cell growth and sensitivity to 5-FU or CDDP should be performed.
The correlations between sensitivity to 5-FU and CDDP and the mRNA levels of the 35 genes were then examined. The levels of target mRNA expression differed among the cell lines (Table IV). The mRNA levels of ABCC2, MSH2 and DPYD were positively correlated with the IC50 values of 5-FU (r>0.7; Fig. 1 and Table V). By contrast, a negative correlation between the IC50 values of 5-FU and the mRNA levels of SLC22A2, SLC23A2, ABCB1 and Rad51 was observed. In the light of the biological roles of these genes, the negative correlation between SLC22A2 and SLC23A2 mRNA expression and sensitivity was considered to be noteworthy. SLC22A2 encodes an organic cation transporter which is responsible for cell uptake of various drugs, including CDDP (11,12). A colon carcinoma cell line exhibiting resistance to 5-FU has been reported to show lower expression of SLC23A2 mRNA than its parent cells (13). ABCC2, MSH2 and DPYD are known to act in detoxifying mechanisms; they are an efflux transporter, DNA repair-related protein and metabolic enzyme, respectively. Although ABCB1 is a known efflux transporter that contributes to drug resistance, the cytotoxicity of 5-FU was not influenced by the expression of ABCB1 (14). In addition, the overexpression of DNA-repair related proteins, including Rad51, has been reported to contribute to resistance to DNA damaging agents (15). Although the present findings showing a negative correlation between IC50 values and ABCB1 and Rad51 mRNA expression levels conflict with previous findings, they may indicate that ABCB1 and Rad51 have no significant impact on sensitivity.
In the case of CDDP, a positive correlation (r>0.7) between the IC50 values and the mRNA levels of ABCC2, MSH2 and DPYD was identified. The findings for ABCC2 and MSH2 are supported by their functions; the export of CDDP from cells (16) and repair of DNA damaged by CDDP (17), respectively (Table V and Fig. 2). In addition, proliferating cell nuclear antigen-normalized mRNA expression of DPYD has previously been reported to be associated with sensitivity to CDDP in lung cancer tissues (18). Although the correlation between CDDP and DPYD has not been investigated in detail, these previous results may support the present findings. The mRNA levels of ABCC2, MSH2 and DPYD correlated well with sensitivity to both 5-FU and CDDP, suggesting that these are potent predictive factors for 5-FU and CDDP-based chemotherapy in esophageal carcinoma patients.
Finally, the roles of ABCC2 and DPYD in sensitivity to 5-FU and CDDP were examined, since the knock-down of MSH2 in SW460 and HeLa cells has been reported to have no influence on sensitivity to 5-FU (19). In the present study, 100 μM gimeracil, which showed sufficient inhibition of DPYD (20), enhanced 5-FU sensitivity in the KYSE30 cell line (Table VI), which had the highest level of DPYD mRNA expression of all the cell lines tested (Table IV). The present findings support those of Ando et al(21); that is, DPYD was a predictor of sensitivity to 5-FU. Apart from the correlation analysis, gimeracil decreased sensitivity to CDDP in all cell lines (Table VI), implying that DPYD activity may be required for the cytotoxic effect of CDDP. Further investigations are required to resolve this contradiction. The concomitant administration of 50 μM MK571, a representative ABCC2 inhibitor (22), was found to decrease the sensitivity of OE33 and KYSE150 cells to 5-FU. In addition, the growth inhibitory activity of CDDP was decreased in KYSE30 and KYSE150 cell lines (Table VII). These findings conflict with the function of ABCC2 function as an efflux transporter, and further investigations are required to clarify this situation.
In conclusion, the mRNA levels of SLC22A2, SLC23A2, ABCB1, ABCC2, Rad51, MSH2 and DPYD were confirmed to be strongly correlated with the IC50 values for 5-FU, and those of ABCC2, MSH2 and DPYD were also confirmed to be strongly correlated with the IC50 values for CDDP. These genes have the potential to affect the sensitivity to 5-FU and CDDP. In addition, the inhibition of DPYD was suggested to affect the cytotoxicity of CDDP. These findings provide useful information for improving the clinical outcome of chemotherapy against esophageal carcinoma.
Acknowledgments
This study was supported in part by a grant of Strategic Research Foundation Grant-aided Project for Private Universities from the Ministry of Education, Culture, Sport, Science and Technology, Japan.
References
- 1.National Cancer Center: Cancer statistics in Japan 2011 http://ganjoho.jp/public/statistics/backnumber/2011_jp.html. Accessed June 29, 2012.
- 2.Tahara M, Ohtsu A, Hironaka S, Boku N, Ishikura S, Miyata Y, Ogino T, Yoshida S. Clinical impact of criteria for complete response (CR) of primary site to treatment of esophageal cancer. Jpn J Clin Oncol. 2005;35:316–323. doi: 10.1093/jjco/hyi095. [DOI] [PubMed] [Google Scholar]
- 3.Pratt S, Shepard RL, Kandasamy RA, Johnston PA, Perry W, III, Dantzig AH. The multidrug resistance protein 5 (ABCC5) confers resistance to 5-fluorouracil and transports its monophosphorylated metabolites. Mol Cancer Ther. 2005;4:855–863. doi: 10.1158/1535-7163.MCT-04-0291. [DOI] [PubMed] [Google Scholar]
- 4.Kornmann M, Schwabe W, Sander S, Kron M, Sträter J, Polat S, Kettner E, Weiser HF, Baumann W, Schramm H, Häusler P, Ott K, Behnke D, Staib L, Beger HG, Link KH. Thymidylate synthase and dihydropyrimidine dehydrogenase mRNA expression levels: predictors for survival in colorectal cancer patients receiving adjuvant 5-fluorouracil. Clin Cancer Res. 2003;9:4116–4124. [PubMed] [Google Scholar]
- 5.Ciaparrone M, Quirino M, Schinzari G, Zannoni G, Corsi DC, Vecchio FM, Cassano A, La Torre G, Barone C. Predictive role of thymidylate synthase, dihydropyrimidine dehydrogenase and thymidine phosphorylase expression in colorectal cancer patients receiving adjuvant 5-fluorouracil. Oncology. 2006;70:366–377. doi: 10.1159/000098110. [DOI] [PubMed] [Google Scholar]
- 6.Larminat F, Bohr VA. Role of the human ERCC-1 gene in gene-specific repair of cisplatin-induced DNA damage. Nucleic Acids Res. 1994;22:3005–3010. doi: 10.1093/nar/22.15.3005. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 7.Andersson BS, Sadeghi T, Siciliano MJ, Legerski R, Murray D. Nucleotide excision repair genes as determinants of cellular sensitivity to cyclophosphamide analogs. Cancer Chemother Pharmacol. 1996;38:406–416. doi: 10.1007/s002800050504. [DOI] [PubMed] [Google Scholar]
- 8.Hsu DS, Lan HY, Huang CH, Tai SK, Chang SY, Tsai TL, Chang CC, Tzeng CH, Wu KJ, Kao JY, Yang MH. Regulation of excision repair cross-complementation group 1 by Snail contributes to cisplatin resistance in head and neck cancer. Clin Cancer Res. 2010;16:4561–4571. doi: 10.1158/1078-0432.CCR-10-0593. [DOI] [PubMed] [Google Scholar]
- 9.Shimada Y, Imamura M, Wagata T, Yamaguchi N, Tobe T. Characterization of 21 newly established esophageal cancer cell lines. Cancer. 1992;69:277–284. doi: 10.1002/1097-0142(19920115)69:2<277::aid-cncr2820690202>3.0.co;2-c. [DOI] [PubMed] [Google Scholar]
- 10.Kitada N, Takara K, Minegaki T, Itoh C, Tsujimoto M, Sakaeda T, Yokoyama T. Factors affecting sensitivity to antitumor platinum derivatives of human colorectal tumor cell lines. Cancer Chemother Pharmacol. 2008;62:577–584. doi: 10.1007/s00280-007-0640-3. [DOI] [PubMed] [Google Scholar]
- 11.Kimura N, Masuda S, Tanihara Y, Ueo H, Okuda M, Katsura T, Inui K. Metformin is a superior substrate for renal organic cation transporter OCT2 rather than hepatic OCT1. Drug Metab Pharmacokinet. 2005;20:379–386. doi: 10.2133/dmpk.20.379. [DOI] [PubMed] [Google Scholar]
- 12.Filipski KK, Loos WJ, Verweij J, Sparreboom A. Interaction of Cisplatin with the human organic cation transporter 2. Clin Cancer Res. 2008;14:3875–3880. doi: 10.1158/1078-0432.CCR-07-4793. [DOI] [PubMed] [Google Scholar]
- 13.Karasawa H, Miura K, Fujibuchi W, Ishida K, Kaneko N, Kinouchi M, Okabe M, Ando T, Murata Y, Sasaki H, Takami K, Yamamura A, Shibata C, Sasaki I. Down-regulation of cIAP2 enhances 5-FU sensitivity through the apoptotic pathway in human colon cancer cells. Cancer Sci. 2009;100:903–913. doi: 10.1111/j.1349-7006.2009.01112.x. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 14.Takara K, Obata Y, Yoshikawa E, Kitada N, Sakaeda T, Ohnishi N, Yokoyama T. Molecular changes to HeLa cells on continuous exposure to cisplatin or paclitaxel. Cancer Chemother Pharmacol. 2006;58:785–793. doi: 10.1007/s00280-006-0226-5. [DOI] [PubMed] [Google Scholar]
- 15.Takenaka T, Yoshino I, Kouso H, Ohba T, Yohena T, Osoegawa A, Shoji F, Maehara Y. Combined evaluation of Rad51 and ERCC1 expressions for sensitivity to platinum agents in non-small cell lung cancer. Int J Cancer. 2007;121:895–900. doi: 10.1002/ijc.22738. [DOI] [PubMed] [Google Scholar]
- 16.Noma B, Sasaki T, Fujimoto Y, Serikawa M, Kobayashi K, Inoue M, Itsuki H, Kamigaki M, Minami T, Chayama K. Expression of multidrug resistance-associated protein 2 is involved in chemotherapy resistance in human pancreatic cancer. Int J Oncol. 2008;33:1187–1194. [PubMed] [Google Scholar]
- 17.Lan L, Hayashi T, Rabeya RM, Nakajima S, Kanno S, Takao M, Matsunaga T, Yoshino M, Ichikawa M, Riele H, Tsuchiya S, Tanaka K, Yasui A. Functional and physical interactions between ERCC1 and MSH2 complexes for resistance to cisdiamminedichloroplatinum(II) in mammalian cells. DNA Repair (Amst) 2004;3:135–143. doi: 10.1016/j.dnarep.2003.10.005. [DOI] [PubMed] [Google Scholar]
- 18.Takizawa M, Kawakami K, Obata T, Matsumoto I, Ohta Y, Oda M, Sasaki T, Watanabe G. In vitro sensitivity to platinum-derived drugs is associated with expression of thymidylate synthase and dihydropyrimidine dehydrogenase in human lung cancer. Oncol Rep. 2006;15:1533–1539. [PubMed] [Google Scholar]
- 19.Pettersen HS, Visnes T, Vågbø CB, Svaasand EK, Doseth B, Slupphaug G, Kavli B, Krokan HE. UNG-initiated base excision repair is the major repair route for 5-fluorouracil in DNA, but 5-fluorouracil cytotoxicity depends mainly on RNA incorporation. Nucleic Acids Res. 2011;39:8430–8444. doi: 10.1093/nar/gkr563. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 20.Li Y, Mizutani Y, Shiraishi T, Nakamura T, Mikami K, Takaha N, Okihara K, Kawauchi A, Sakai T, Miki T. The significance of the expression of dihydropyrimidine dehydrogenase in prostate cancer. BJU Int. 2007;99:663–668. doi: 10.1111/j.1464-410X.2006.06606.x. [DOI] [PubMed] [Google Scholar]
- 21.Ando T, Ishiguro H, Kuwabara Y, Kimura M, Mitsui A, Sugito N, Mori R, Ogawa R, Katada T, Fujii Y. Relationship between expression of 5-fluorouracil metabolic enzymes and 5-fluorouracil sensitivity in esophageal carcinoma cell lines. Dis Esophagus. 2008;21:15–20. doi: 10.1111/j.1442-2050.2007.00700.x. [DOI] [PubMed] [Google Scholar]
- 22.Pedersen JM, Matsson P, Bergström CA, Norinder U, Hoogstraate J, Artursson P. Prediction and identification of drug interactions with the human ATP-binding cassette transporter multidrug-resistance associated protein 2 (MRP2; ABCC2) J Med Chem. 2008;51:3275–3287. doi: 10.1021/jm7015683. [DOI] [PubMed] [Google Scholar]
- 23.Reidling JC, Subramanian VS, Dahhan T, Sadat M, Said HM. Mechanisms and regulation of vitamin C uptake: studies of the hSVCT systems in human liver epithelial cells. Am J Physiol Gastrointest Liver Physiol. 2008;295:1217–1227. doi: 10.1152/ajpgi.90399.2008. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 24.Nishimura M, Koeda A, Morikawa H, Satoh T, Narimatsu S, Naito S. Comparison of inducibility of multidrug resistance (MDR)1, multidrug resistance-associated protein (MRP)1, and MRP2 mRNAs by prototypical microsomal enzyme inducers in primary cultures of human and cynomolgus monkey hepatocytes. Biol Pharm Bull. 2008;31:2068–2072. doi: 10.1248/bpb.31.2068. [DOI] [PubMed] [Google Scholar]
- 25.Maubon N, Le Vee M, Fossati L, Audry M, Le Ferrec E, Bolze S, Fardel O. Analysis of drug transporter expression in human intestinal Caco-2 cells by real-time PCR. Fundam Clin Pharmacol. 2007;21:659–663. doi: 10.1111/j.1472-8206.2007.00550.x. [DOI] [PubMed] [Google Scholar]
- 26.Dauchy S, Miller F, Couraud PO, Weaver RJ, Weksler B, Romero IA, Scherrmann JM, De Waziers I, Declèves X. Expression and transcriptional regulation of ABC transporters and cytochromes P450 in hCMEC/D3 human cerebral micro-vascular endothelial cells. Biochem Pharmacol. 2009;77:897–909. doi: 10.1016/j.bcp.2008.11.001. [DOI] [PubMed] [Google Scholar]
- 27.Shimizu J, Horio Y, Osada H, Hida T, Hasegawa Y, Shimokata K, Takahashi T, Sekido Y, Yatabe Y. mRNA expression of RRM1, ERCC1 and ERCC2 is not associated with chemosensitivity to cisplatin, carboplatin and gemcitabine in human lung cancer cell lines. Respirology. 2008;13:510–517. doi: 10.1111/j.1440-1843.2008.01302.x. [DOI] [PubMed] [Google Scholar]
- 28.Sliwinski T, Krupa R, Majsterek I, Rykala J, Kolacinska A, Morawiec Z, Drzewoski J, Zadrozny M, Blasiak J. Polymorphisms of the BRCA2 and RAD51 genes in breast cancer. Breast Cancer Res Treat. 2005;94:105–109. doi: 10.1007/s10549-005-0672-5. [DOI] [PubMed] [Google Scholar]
- 29.Amirrad M, Al-Mulla F, Varadharaj G, John B, Saji T, Anim JT. BRCA1 gene expression in breast cancer in Kuwait: correlation with prognostic parameters. Med Princ Pract. 2005;14:67–72. doi: 10.1159/000083913. [DOI] [PubMed] [Google Scholar]
- 30.Müller A, Zielinski D, Friedrichs N, Oberschmid B, Merkelbach-Bruse S, Schackert HK, Linnebacher M, von Knebel Doeberitz M, Büttner R, Rüschoff J. Reduced mRNA expression in paraffin-embedded tissue identifies MLH1-and MSH2-deficient colorectal tumours and potential mutation carriers. Virchows Arch. 2008;453:9–16. doi: 10.1007/s00428-008-0637-2. [DOI] [PubMed] [Google Scholar]
- 31.Veeriah S, Kautenburger T, Habermann N, Sauer J, Dietrich H, Will F, Pool-Zobel BL. Apple flavonoids inhibit growth of HT29 human colon cancer cells and modulate expression of genes involved in the biotransformation of xenobiotics. Mol Carcinog. 2006;45:164–174. doi: 10.1002/mc.20158. [DOI] [PubMed] [Google Scholar]
- 32.Uthus EO, Reeves PG, Saari JT. Copper deficiency decreases plasma homocysteine in rats. J Nutr. 2007;137:1370–1374. doi: 10.1093/jn/137.6.1370. [DOI] [PubMed] [Google Scholar]
- 33.Hoang YD, Avakian AP, Luderer U. Minimal ovarian upregulation of glutamate cysteine ligase expression in response to suppression of glutathione by buthionine sulfoximine. Reprod Toxicol. 2006;21:186–196. doi: 10.1016/j.reprotox.2005.07.011. [DOI] [PubMed] [Google Scholar]
- 34.Gustavsson B, Kaiser C, Carlsson G, Wettergren Y, Odin E, Lindskog EB, Niyikiza C, Ma D. Molecular determinants of efficacy for 5-FU-based treatments in advanced colorectal cancer: mRNA expression for 18 chemotherapy-related genes. Int J Cancer. 2009;124:1220–1226. doi: 10.1002/ijc.23852. [DOI] [PubMed] [Google Scholar]
- 35.Yoshinare K, Kubota T, Watanabe M, Wada N, Nishibori H, Hasegawa H, Kitajima M, Takechi T, Fukushima M. Gene expression in colorectal cancer and in vitro chemosensitivity to 5-fluorouracil: a study of 88 surgical specimens. Cancer Sci. 2003;94:633–638. doi: 10.1111/j.1349-7006.2003.tb01495.x. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 36.Matsubara J, Nishina T, Yamada Y, Moriwaki T, Shimoda T, Kajiwara T, Nakajima TE, Kato K, Hamaguchi T, Shimada Y, Okayama Y, Oka T, Shirao K. Impacts of excision repair cross-complementing gene 1 (ERCC1), dihydropyrimidine dehydrogenase, and epidermal growth factor receptor on the outcomes of patients with advanced gastric cancer. Br J Cancer. 2008;98:832–839. doi: 10.1038/sj.bjc.6604211. [DOI] [PMC free article] [PubMed] [Google Scholar]